Abstract
Cardiovascular health is negatively impacted by arterial stiffening, which increases pulsatile load and elevates left ventricular workload. Aortic dilatation may compensate for the pulsatile overload resulting from arterial stiffening. Previous studies have shown a negative correlation between diabetes and aortic diameter, suggesting that diabetes may impair aortic dilatation and thereby compromise compensatory buffering capacity. This study aims to explore the relationship between diabetes, aortic diameter, and left ventricular hypertrophy. This retrospective study included 271 elderly patients stratified by diabetes status. Aortic and cardiac geometry was measured via echocardiography, and arterial stiffness was assessed using brachial-ankle pulse wave velocity (baPWV). Pulse pressure (PP) was used as an index of pulsatile load. Linear regression and mediation analyses were conducted to examine the associations among diabetes, aortic diameter, arterial stiffness, PP, and left ventricular mass index (LVMI). Diabetes, fasting blood glucose (FPG), and glycated hemoglobin (HbA1c) were independently associated with smaller aortic diameters (AoRi), with regression coefficients of − 0.984, − 0.275, and − 0.479, respectively. BaPWV was positively associated with AoRi (a = 0.942, P = 0.015), whereas AoRi was negatively correlated with PP (b =− 0.990, P < 0.001). BaPWV had a direct positive effect on PP (c′ = 3.336, P = 0.024), which was counteracted by an indirect effect through AoRi (− 0.933, 95% CI − 2.133, − 0.028). Mediation analysis revealed that AoRi attenuated the effect of PP on LVMI (0.439, P < 0.001 vs. 0.308, P = 0.007) and amplified the effect of diabetes on PP (3.903, P = 0.031 vs. 4.732, P = 0.010). In the chain-mediated effect model, diabetes had a significant total effect on LVMI (β = 5.025, P = 0.043). The diabetes → AoRi → PP → LVMI pathway demonstrated a significant indirect effect (0.230, 95% CI 0.013–0.592). Diabetes is associated with impaired aortic dilatation and increased left ventricular hypertrophy, potentially serving as a pathological mechanism driving progression of myocardial hypertrophy.
Similar content being viewed by others
Introduction
Arterial stiffening significantly impacts cardiovascular health by increasing pulsatile load, raising left ventricular workload and impairing tissue perfusion. Such changes contribute to a heightened risk of cardiovascular and age-related diseases. Arterial stiffening is characterized by increased arterial wall stiffness, thickening, and lumen dilatation. These structural alterations reduce arterial compliance and buffering capacity, exacerbate pulsatile load, and enhance the penetration of pulsatile energy into the microvasculature of target organs. Through its elastic reservoir function, the aorta plays a pivotal role in modulating pulsatile load and maintaining hemodynamic stability.
With growing concern about arterial stiffening and pulsatile hemodynamics in cardiovascular disease, recent studies have emphasized the role of aortic dilatation in modulating pulsatile load and left ventricular afterload1,2,3,4. Historically, aortic dilatation has been recognized as a manifestation of target organ damage associated with arterial stiffening and hypertension, both linked to an elevated risk of cardiovascular events5,6. However, more recent evidence suggests that aortic dilatation also serves as an adaptive, compensatory response to increased arterial stiffness and elevated blood pressure. Campos Arias et al. proposed a concept of age-dependent regulatory function of the aorta, where increased aortic stiffness was not always paralleled by elevated aortic impedance, because the hemodynamic impact of aortic stiffening may be overcome by aortic dilatation4. Supporting this, a community-based study found that individuals with smaller aortic diameters and higher pulse wave velocity (PWV) faced greater cardiovascular risk3. These findings align with the water hammer equation, Zc = ρ × PWV/πr2 (where ρ is blood density and r is the aortic radius), indicating that aortic dilatation may compensate for the pulsatile overload from arterial stiffening. Our previous study further demonstrated that aortic dilatation mitigates the impact of pulsatile load on left ventricular remodeling, validating its compensatory role2. Aortic dilatation represents a continuum of vascular remodeling in response to arterial stiffening and hemodynamic stress. Early-stage dilatation serves a compensatory function by buffering increased pulsatile load, while progressive dilatation reflects the cumulative effects of vascular aging and chronic hemodynamic burden. This dynamic spectrum highlights the complex and evolving nature of aortic remodeling in cardiovascular health and disease.
Interestingly, while hypertension and arterial stiffening generally promote aortic dilatation, diabetes mellitus exerts a distinct effect by limiting aortic dilatation. This phenomenon was first observed in patients with aneurysms. A study reported that type 2 diabetes is associated with a reduced incidence of abdominal aortic aneurysm7,8. Further corroborating these findings, the European Society of Cardiology (ESC) noted in a 2019 consensus document that patients with diabetes exhibit a lower prevalence of aortic aneurysm or dissection than age-matched individuals without diabetes9. However, limited studies are exploring this phenomenon in populations without aortic aneurysms. Previous studies offer conflicting findings, with a Western study suggesting a protective effect of diabetes against aortic dilatation while another involving Chinese participants with hypertension found no significant correlation10,11. Our prior research involving 1556 elderly Chinese patients with hypertension identified a significant negative correlation between diabetes and aortic diameter2. These findings suggest that the relationship between diabetes and aortic diameter and its influence on pulsatile hemodynamics warrants further investigation.
Given that diabetes is a known risk factor for arterial stiffening and cardiovascular disease, arterial stiffening is associated with an increase in aortic diameter. This raises an important question: how should we explain the negative correlation between diabetes and aortic diameter10,12,13? We hypothesize that aortic wall remodeling in diabetes restricts aortic dilatation, which compromises its buffering function and elevates cardiovascular risk. This study aims to investigate the relationships between diabetes, aortic diameter, pulsatile load, and their collective impact on left ventricular remodeling in an elderly Chinese population.
Methods
Study population
This single-center, retrospective observational study enrolled consecutive patients aged 60 and above without aortic aneurysm or dissection who underwent brachial-ankle pulse wave velocity (baPWV) measurement at the Department of Geriatrics, First Hospital of China Medical University, between December 2018 and November 2022. Patients were excluded if they had incomplete data, body mass index (BMI) outside the range of 18.5–28 kg/m2, heart failure with reduced ejection fraction or acute heart failure, chronic kidney disease (CKD) stage IV–V, atrial fibrillation, and any acute medical condition. Trained physicians collected demographic and clinical data, which were recorded in the hospital’s electronic information system. Morning fasting blood samples were collected for laboratory analysis. This study was conducted in accordance with the Declaration of Helsinki and approved by the ethical review board of The First Hospital of China Medical University (approval number: [2024] No.130). Informed consent was obtained from all participants. All methods were carried out in accordance with relevant guidelines and regulations.
Evaluation of arterial stiffness
BaPWV was used to assess arterial stiffness, utilizing the OMRON Vascular Profiler (VP-1000; OMRON Healthcare, Kyoto, Japan). Calibrated blood pressure cuffs with integrated sensors were applied bilaterally to the brachial and ankle arteries following a standardized five-minute rest in the supine position. Two automated baPWV measurements were obtained, and the average PWV from the right side was recorded to ensure standardization.
24-Hour ambulatory blood pressure monitoring
Blood pressure (BP) monitoring was performed over 24 h using an ambulatory device (Meditech ABPM-05; Meditech Ltd., Budapest, Hungary) with a cuff applied to the right brachial artery. BP was measured every 30 min from 6:00 AM to 10:00 PM and hourly from 10:00 PM to 6:00 AM. Average 24-h systolic blood pressure (SBP), diastolic blood pressure (DBP), and heart rate (HR) were recorded. Pulse pressure (PP) was calculated as SBP minus DBP and mean arterial pressure (MAP) was calculated using the formula MAP = 1/3 × SBP + 2/3 × DBP. Monitoring data were deemed valid if more than 80% of the scheduled readings were successfully obtained.
Echocardiographic measurement
All transthoracic echocardiography (Philips iE33; Philips Healthcare, Eindhoven, The Netherlands) was performed at rest in the left lateral decubitus position following standard protocols14. Each scan was reviewed by at least two experienced ultrasound experts. Left ventricular (LV) end-diastolic diameter, interventricular septal thickness (IVST), and LV posterior wall thickness (LVPWT) were obtained from parasternal long-axis views using 2D-guided M-mode imaging. LV mass (LVM) was calculated by the Devereux-modified cube formula and indexed to body surface area. End-diastolic LV volume and ejection fraction were derived from biplane Simpson’s method. Aortic diameters were measured at end-diastole from parasternal long-axis 2D images using the leading-edge to leading-edge convention. Measurements were obtained at the sinuses of Valsalva, sinotubular junction, and proximal ascending aorta; the latter was defined as AoR in this study.
Diastolic function was assessed by pulsed-wave Doppler of mitral inflow (E, A waves) at the leaflet tips in the apical four-chamber view. Tissue Doppler imaging (TDI) was performed at the septal mitral annulus to obtain early diastolic velocity (e′), and the E/ e′ ratio was calculated to estimate LV filling pressure.
Definitions and grouping
Diabetes mellitus (DM) was defined as fulfilling any of the following criteria: (1) glycated hemoglobin (HbA1c) ≥ 6.5%; (2) random plasma glucose ≥ 11.1 mmol/L; (3) fasting plasma glucose (FPG) ≥ 7.0 mmol/L; or (4) 2-h plasma glucose ≥ 11.1 mmol/L following an oral glucose tolerance test. Hypertension was defined as either office SBP ≥ 140 mmHg or office DBP ≥ 90 mmHg at rest, or the current use of antihypertensive medication. CKD was defined as an estimated glomerular filtration rate (eGFR) of less than 90 mL/min/1.73 m2, calculated using the CKD-EPI equation. Arterial stiffening was classified as PP ≥ 60 mmHg15. Body surface area (BSA) was calculated with the Mosteller formula. Both LVM and AoR were normalized to BSA. Left ventricular hypertrophy (LVH) was defined as an LVMI ≥ 115 g/m2 for men or ≥ 95 g/m2 for women, or IVST (interventricular septal thickness) or LVPWT (left ventricular posterior wall thickness) > 11 mm for men and > 10.5 mm for women15,16.
Statistical analysis
The normality of continuous variables was assessed using the Shapiro–Wilk test. The Mann–Whitney U test was applied for variables that did not meet normality assumptions. Descriptive statistics are reported as mean ± standard deviation for continuous variables and as frequencies and percentages for categorical variables. Continuous variables were compared between groups using the Student’s t-test or ANOVA, and categorical variables were compared using the chi-square test. Univariate linear regression analysis was first conducted to examine the individual associations between variables. Multivariate linear regression analysis was then used to evaluate the independent effects of each variable while adjusting for potential confounders. Mediation analysis was conducted to explore the mechanisms among the independent variable, dependent variable, and mediator variable. The bootstrap method was used for significance testing with 1,000 resamples, applied through the SPSS PROCESS macro version 4.3. Analysis of covariance (ANCOVA) was employed to compare means across groups, adjusting for relevant covariates.
All statistical tests were two-tailed, with a significance level set at a P-value of 0.05. All analyses were conducted using IBM SPSS version 26 (SPSS Inc, Chicago, IL, USA).
Results
Population characteristics
A total of 375 patients were initially assessed, with 104 excluded based on predefined criteria, resulting in a final sample of 271 patients included in the study (Supplementary Fig. 1).
This study analyzed patients aged 60–100 years (mean age 74.06 ± 9.71 years), predominantly male (83.39%). Hypertension and diabetes prevalences were 77.12 percent and 45.02 percent, respectively. Twenty-four-hour mean SBP averaged 128.27 ± 13.57 mmHg, DBP 69.54 ± 9.09 mmHg, and MAP 89.13 ± 9.05 mmHg. HbA1c was 6.27 ± 1.03 percent, and FBG averaged 5.89 ± 1.44 mmol/L, respectively. Total cholesterol, low-density lipoprotein cholesterol (LDL-C) and triglycerides measured 4.08 ± 0.98, 2.42 ± 0.88 and 1.35 ± 0.82 mmol/L, respectively.
Table 1 summarizes the population characteristics, categorized into two groups based on diabetes status. There was no significant age difference between the two groups (74.47 ± 10.02 vs. 73.54 ± 9.27 years old, P = 0.463), but the group with diabetes had a higher proportion of males (77.18% vs.90.98%, P = 0.002), greater BSA (1.79 ± 0.15 vs. 1.84 ± 0.14 m2, P = 0.002) and marginally higher smoking prevalence (34.23% vs. 36.89%, P > 0.05) . Patients with diabetes had higher FPG (5.15 ± 0.54 vs. 6.80 ± 1.66 mmol/L, P < 0.001), HbA1c (5.73 ± 0.40% vs. 6.94 ± 1.18%, P < 0.001) and triglycerides (1.52 ± 0.95 vs. 1.21 ± 0.68 mmol/L, P < 0.001), but lower high-density lipoprotein cholesterol (HDL-C) (1.34 ± 0.90 vs. 1.13 ± 0.33 mmol/L, P = 0.003) and slightly lower LDL-C (2.52 ± 0.84 vs. 2.31 ± 0.92 mmol/L; P = 0.050).
Absolute aortic diameter did not differ between patients with and without diabetes (36.17 ± 3.83 vs. 35.72 ± 3.56 mm; P > 0.05). After adjustment for BSA, AoRi (20.37 ± 2.67 vs. 19.49 ± 2.16 mm/m2, P = 0.003) was smaller in patients with diabetes. Patients with diabetes also exhibited higher left atrial diameter (LAD), IVST, LVPWT, LVMI and E/e’(P < 0.05). Patients with diabetes exhibited a widened PP (55.85 ± 12.09 vs. 62.37 ± 12.08 mmHg, P < 0.001) despite similar baPWV (1.80 ± 0.42 vs. 1.83 ± 0.42 m/s, P = 0.525), with no significant differences in 24-h mean DBP or 24-h mean MAP between the groups(P > 0.05).
Correlation between aortic diameter and glycemic traits
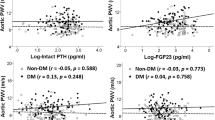
In both univariate and multivariate analyses, diabetes, FBG, and HbA1c were inversely associated with aortic diameter. Diabetes correlated with smaller aortic diameter (univariate: B = − 0.886, P = 0.004; multivariate: B = − 0.984, P = 0.039). FBG (univariate: B = − 0.286, P = 0.006; multivariate: B = − 0.275, P = 0.017) and HbA1c (univariate: B = − 0.327, P = 0.025; multivariate: B = − 0.479, P = 0.007) showed similar associations (Table 2).
Mediation analysis of aortic diameter between baPWV, PP, and LVMI relationships
In mediation models (Fig. 1), arterial stiffness (baPWV) positively associated with AoRi (path a = 0.942, P = 0.015), AoRi negatively associated with PP (path b = − 0.990, P < 0.001). The direct effect of baPWV on PP was positive (c′ = 3.336, P = 0.024), while the total effect was not statistically significant (c = 2.404, P = 0.109). The indirect effect mediated via AoRi was − 0.933 (95%CI − 2.133, − 0.028).
The mediation effect models for AoRi between baPWV, PP, LVMI, and diabetes. (A) The mediation effect model for baPWV, AoRi and PP. (B) The mediation effect model for PP, AoRi and LVMI. (C) The mediation effect model for DM, AoRi and LVMI. Age, gender, smoking status, triglycerides, total cholesterol, creatinine, uric acid, 24-h mean MAP, 24-h mean HR, hypertension, use of antidiabetic medication, insulin, and antihypertensive medication were adjusted in the mediation effect model. AoRi aortic diameter normalized to BSA, BaPWV brachial-ankle pulse wave velocity, PP 24-h mean pulse pressure, LVMI left ventricular mass index, DM diabetes mellitus.
PP demonstrated a total effect on LVMI of 0.308 (P = 0.007) and a direct effect of 0.439 (P < 0.001), with an indirect attenuation via AoRi (− 0.131, 95%CI − 0.230, − 0.058).
Mediation analysis of diabetes, aortic diameter, and PP
When diabetes status was the independent variable, the effect on AoRi was negative (path a = − 0.984, P = 0.039), and AoRi negatively influenced PP (path b = − 0.843, P < 0.001). The direct effect of diabetes on PP was 3.903 (P = 0.031); the total effect was 4.732 (P = 0.010), with the indirect effect via aortic diameter index accounting for the difference.
Subgroup and chain mediation analyses
Forest plots (Fig. 2) show stronger inverse correlations between glycemic traits AoRi in elderly and subgroup with hypertension. In patients with hypertension, chain mediation (Fig. 3) demonstrated that diabetes had a significant total effect on LVMI (P = 0.043) but a non-significant direct effect after adjustment (P = 0.138). The specific indirect pathway Diabetes → AoRi → PP → LVMI was significant (0.230, 95% CI 0.013 to 0.592).
Forest plot of glycemic traits association with AoRi calculated using a linear regression model. (A) Forest plot of diabetes association with AoRi calculated using a linear regression model. (B) Forest plot of FPG association with AoRi calculated using a linear regression model. (C) Forest plot of HbA1c association with AoRi calculated using a linear regression model. Age, gender, smoking status, triglycerides, total cholesterol, creatinine, uric acid, 24-h mean MAP, 24-h mean HR, hypertension, use of antidiabetic medication, insulin, and antihypertensive medication were adjusted in multivariable regression. DM diabetes mellitus, FBG fasting blood glucose, HbA1c glycated hemoglobin, AoRi aortic diameter normalized to BSA, CKD chronic kidney disease, LVH left ventricular hypertrophy.
The chain mediation effect model for DM, AoRi, PP and LVMI. Age, gender, smoking status, 24-h mean MAP, and 24-h mean HR were adjusted in the mediation effect model. DM diabetes mellitus, AoRi aortic diameter normalized to BSA, PP 24-h mean pulse pressure, LVMI left ventricular mass index.
Comparison of aortic diameter stratified by glycemic traits
Among patients with hypertension, those with diabetes had smaller AoRi than those without diabetes (19.45 ± 2.11 vs. 20.43 ± 2.67 mm/m2, P = 0.004). ANCOVA adjustment increased this difference (19.41 ± 0.30 vs. 20.48 ± 0.30 mm/m2; P < 0.001) (Supplementary Fig. 2). AoRi decreased progressively across HbA1c tertiles (P for trend < 0.001).
Discussion
Our findings demonstrate that diabetes, FPG and HbA1c were significantly negatively associated with aortic diameter even in a general, elderly Chinese population. Through both basic and chain mediation models, we confirmed that restricted aortic dilatation in diabetes increases pulsatile load, which in turn exacerbates LVH. Notably, these mechanisms were most pronounced in participants with hypertension, underscoring the synergistic impact of elevated blood pressure and hyperglycemia on aortic compliance and cardiac structure.
Aortic dilatation plays a dual role, representing a continuum from adaptive to maladaptive remodeling rather than distinct stages. Initially, it compensates for increased arterial stiffness by expanding reservoir capacity during systole, thereby buffering pulsatile load. Over time, however, this dilatation reflects chronic hemodynamic overload. Substantial evidence has linked aging and hypertension to a positive association with increased aortic diameter17. While some studies associate larger aortic diameter with elevated LVMI and incident heart failure, others find smaller diameters predict higher cardiovascular risk17,18,19. Studies focusing on pulsatile hemodynamics suggest that aortic dilatation may compensate for impaired aortic elastic reservoir function by increasing the local storage capacity during systole3,20. We reconcile these findings by proposing a time-dependent framework: early compensatory dilatation can buffer pulsatile stress, but persistent hypertension burden eventually deplete vascular reserve, leading to maladaptive remodeling. This model is supported by observations that individuals with elevated PWV but limited aortic expansion exhibit the widest PP and the highest cardiovascular risk3. Nevertheless, it remains inherently difficult to clearly separate compensatory from pathological dilatation, as these processes overlap continuously without a clear boundary between beneficial and detrimental remodeling.
Some early studies have reported a negative correlation between diabetes and aortic diameter, but they largely focused on patients with thoracic or abdominal aortic aneurysms7. Patients with diabetes not only have a lower prevalence of aortic aneurysms, but those with aneurysms also have a lower postoperative re-rupture rate9. These findings suggest that diabetes may significantly slow the progression of aortic dilatation. In recent years, a growing number of studies have explored the relationship between diabetes and aortic diameter in various populations12,21. Several small-scale studies in populations with end-stage renal disease have identified the same negative correlation between diabetes and aortic diameter22. Furthermore, large studies in the general population have further confirmed this inverse association23,24. Additionally, we found that both short-term blood glucose levels (FPG) and long-term blood glucose levels (HbA1c) were significantly negatively correlated with aortic diameter. In a prediction model for ascending aortic diameter based on a training cohort of 30,018 participants, diabetes was included as a key clinical variable. Although its independent effect was small, it had a significant combined impact when considered alongside age and blood pressure23. This aligns with our finding that the correlation coefficient between diabetes and aortic diameter is greater in the context of hypertension.
The impact of diabetes on aortic diameter, whether protective or detrimental to cardiovascular health, remains an issue of ongoing debate25. Diabetes is closely associated with arterial stiffening, correlating with elevated PWV and PP, and increasing the risk of heart failure with preserved ejection fraction (HFpEF) through its adverse impact on pulsatile hemodynamics26,27. Patients with diabetes in our study also exhibited higher LVMI and impaired diastolic function, both recognized features of HFpEF. While we observed associations suggesting that diabetes may contribute to increased pulsatile load potentially by restricting aortic dilatation, as supported by our mediation models, we refrain from making definitive conclusions regarding the development of HFpEF or the predictive value of aortic diameter due to the absence of longitudinal follow-up data. Precision medicine approaches in HFpEF emphasize targeting underlying pathophysiological mechanisms, particularly in diabetic patients who often exhibit a restrictive HFpEF phenotype characterized by severe diastolic dysfunction, elevated left ventricular filling pressures, and concentric hypertrophy28. PP serves as an independent predictor of HFpEF29, and increased pulsatile load associated with reduced aortic dilatation may play a role in HFpEF pathogenesis, especially in diabetes; however, this hypothesis requires further investigation in prospective studies.
Some viewpoints attribute the negative relationship between diabetes and aortic diameter to the protective effects of anti-diabetic medications30. However, our study and others have found a significant negative correlation between blood glucose traits and aortic diameter independent of anti-diabetic treatment, indicating that anti-diabetic medications can only partially explain this relationship. In our analysis, although anti-diabetic treatment was adjusted as a covariate, we did not perform separate analyses for different classes of anti-diabetic drugs (e.g., Metformin, Thiazolidinediones, Dipeptidyl Peptidase-4 inhibitors, Glucagon-Like Peptide-1 receptor agonists, Sodium-Glucose Cotransporter-2 inhibitors) due to limited sample size and statistical power. Therefore, the potential differential effects of these drug classes on aortic remodeling remain unclear. Future studies with larger cohorts should stratify patients according to specific anti-diabetic therapies to elucidate their distinct impacts on aortic structure and compliance. Additionally, increased arterial wall thickness in individuals with diabetes mellitus may partly contribute to their smaller aortic diameter31, which warrants further investigation.
Some studies pay attention to gender differences in aortic remodeling. A study based on UK biobank data showed females had greater hypertension-related reduction in aortic compliance32. In our study, females also showed a stronger correlation between blood glucose traits and aortic diameter, with a statistically significant interaction. However, due to the smaller proportion of females in our study population, this finding requires further validation in future research.
This study has several limitations. First, its cross-sectional design precludes any inference of causality between diabetes, aortic remodeling, and subsequent cardiovascular events; longitudinal data are required to determine temporal relationships and causative effects. Second, the relatively modest sample size limited statistical power, restricting detailed adjustment for individual classes of antihypertensive and antidiabetic therapies, as well as for potential confounders. Although we included medication use as a covariate, we could not stratify analyses by specific drug classes. Third, because our cohort consisted predominantly of older, male, ethnically Chinese participants, the external validity of our findings is constrained; extrapolation to younger individuals, females, or other ethnic groups should be undertaken with caution. Finally, while we measured fasting plasma glucose and glycated hemoglobin as markers of glycemic control, additional biomarkers—such as advanced glycation end products and inflammatory mediators—may provide deeper insight into the molecular mechanisms of aortic extracellular matrix remodeling. Future prospective studies with larger, more diverse populations and serial measurements are needed to evaluate specific therapeutic regimens’ effects on aortic diameter, PWV, and the development of HFpEF, as well as to elucidate underlying pathophysiological pathways under hyperglycemic and oxidative stress conditions.
Conclusions
Our results highlight a previously underrecognized mechanism by which diabetes contributes to cardiovascular remodeling. In an elderly, predominantly male Chinese population, particularly among individuals with hypertension, diabetes and elevated blood glucose levels were significantly associated with smaller ascending aortic diameters. Impaired aortic dilatation increased pulsatile load, accelerating left ventricular hypertrophy and potentially promoting HFpEF. These findings highlight aortic remodeling as a distinct pathological process in diabetes and suggest that preserving aortic compliance may offer a novel therapeutic target. Future research should elucidate the molecular and biomechanical mechanisms underlying these changes and develop interventions to mitigate cardiovascular risk in patients with diabetes.
What is already known on this topic
Arterial stiffening and impaired aortic compliance are key drivers of cardiovascular morbidity. While diabetes reduces the risk of aortic aneurysm, its role in altering aortic structure and disrupting hemodynamic compensation in elderly patients without aneurysms remains unclear.
What this study adds
This study identifies impaired ascending aortic dilatation as a novel mechanism by which diabetes exacerbates pulsatile load and promotes left ventricular hypertrophy, independent of arterial stiffness severity.
How this study might affect research, practice or policy
These findings suggest that targeting aortic compliance could offer a new therapeutic strategy to prevent myocardial hypertrophy and heart failure with preserved ejection fraction (HFpEF) in elderly patients with diabetes and hypertension.
Data availability
The data that support the findings of this study are not publicly available due to institutional regulations and patient confidentiality. The data were obtained from the hospital health information system (HIS) and contain sensitive personal information. They are stored in secure, controlled-access storage at The First Hospital of China Medical University. Requests for access can be directed to the corresponding author and will require appropriate ethical and institutional approvals.
Abbreviations
- AoRi:
-
Aortic diameter
- BP:
-
Blood pressure
- BMI:
-
Body mass index
- baPWV:
-
Brachial-ankle pulse wave velocity
- BSA:
-
Body surface area
- CKD:
-
Chronic kidney disease
- DBP:
-
Diastolic blood pressure
- eGFR:
-
Estimated glomerular filtration rate
- ESC:
-
European society of cardiology
- FPG:
-
Fasting plasma glucose
- HFpEF:
-
Heart failure with preserved ejection fraction
- HbA1c:
-
Glycated hemoglobin
- HR:
-
Heart rate
- LAD:
-
Left atrial diameter
- LDL-C:
-
Low-density lipoprotein cholesterol
- LVM:
-
Left ventricular mass
- LVH:
-
Left ventricular hypertrophy
- LVMI:
-
Left ventricular mass index
- IVST:
-
Interventricular septal thickness
- LVPWT:
-
Left ventricular posterior wall thickness
- PP:
-
Pulse pressure
- SBP:
-
Systolic blood pressure
References
Redheuil, A. et al. Left ventricular and proximal aorta coupling in magnetic resonance imaging: Aging together?. Am. J. Physiol. Heart Circ. Physiol. 317(2), H300-h307 (2019).
Jiang, Y. Q. et al. Ascending aorta dilatation reduces the influence of elevated pulse pressure on left ventricular hypertrophy: findings from a Chinese elderly hypertensive population. J. Geriatr. Cardiol. 20(2), 100–108 (2023).
Vasan, R. S., Song, R. J., Xanthakis, V. & Mitchell, G. F. Aortic root diameter and arterial stiffness: Conjoint relations to the incidence of cardiovascular disease in the framingham heart study. Hypertension 78(5), 1278–1286 (2021).
Campos-Arias, D., De Buyzere, M. L., Chirinos, J. A., Rietzschel, E. R. & Segers, P. Longitudinal changes of input impedance, pulse wave velocity, and wave reflection in a middle-aged population: The asklepios study. Hypertension 77(4), 1154–1165 (2021).
Mancia, G. et al. 2023 ESH guidelines for the management of arterial hypertension the task force for the management of arterial hypertension of the European society of hypertension: Endorsed by the international society of hypertension (ISH) and the European renal association (ERA). J. Hypertens 41(12), 1874–2071 (2023).
Leone, D. et al. Prognostic role of the ascending aorta dilatation in patients with arterial hypertension. J. Hypertens 39(6), 1163–1169 (2021).
Shah, A. D. et al. Type 2 diabetes and incidence of cardiovascular diseases: A cohort study in 1·9 million people. Lan. Diabet. Endocrinol. 3(2), 105–113 (2015).
Dinesh Shah, A. et al. Type 2 diabetes and incidence of a wide range of cardiovascular diseases: a cohort study in 1·9 million people. Lancet 385(Suppl 1), S86 (2015).
Raffort, J. et al. Diabetes and aortic aneurysm: Current state of the art. Cardiovasc. Res. 114(13), 1702–1713 (2018).
Nardi, E., Mulè, G., Nardi, C., Geraci, G. & Averna, M. Inverse association between type 2 diabetes and aortic root dimension in hypertensive patients. Int. J. Cardiol. 228, 233–237 (2017).
Wang, H., Sun, Y., Wang, S. & Zhang, L. Is diabetes correlating with small aortic root dimension in general Chinese hypertensive patients?. Int. J. Cardiol. 264, 189 (2018).
Li, Z. et al. Effects of diabetes mellitus and glycemic traits on cardiovascular morpho-functional phenotypes. Cardiovasc. Diabetol. 22(1), 336 (2023).
Ning, X. et al. Diabetes, its duration, and the long-term risk of abdominal aortic aneurysm: The atherosclerosis risk in Communities (ARIC) Study. Atherosclerosis 313, 137–143 (2020).
Lang, R. M. et al. Recommendations for cardiac chamber quantification by echocardiography in adults: an update from the American society of echocardiography and the European association of cardiovascular imaging. Eur. Heart J. Cardiovasc. Imag. 16(3), 233–270 (2015).
Williams, B. et al. 2018 ESC/ESH Guidelines for the management of arterial hypertension. Eur. Heart J. 39(33), 3021–3104 (2018).
Yao, G. H. et al. Echocardiographic measurements in normal chinese adults focusing on cardiac chambers and great arteries: a prospective, nationwide, and multicenter study. J. Am. Soc. Echocardiogr. 28(5), 570–579 (2015).
Tadic, M., Gherbesi, E., Sala, C., Carugo, S. & Cuspidi, C. Is thoracic aortic diameter an independent predictor of cardiovascular disease and mortality? A Narrative Review. Front Cardiovasc. Med. 9, 867026 (2022).
Cuspidi, C. et al. Incident aortic root dilatation in the general population: Findings from the Pamela study. J. Hypertens 40(3), 544–552 (2022).
Cuspidi, C. et al. New-onset aortic dilatation in the population: a quarter-century follow-up. Clin. Res. Cardiol. 112(11), 1529–1540 (2023).
Torjesen, A. A. et al. Pulse pressure relation to aortic and left ventricular structure in the age, gene/environment susceptibility (AGES)-Reykjavik study. Hypertension 64(4), 756–761 (2014).
Bons, L. R. et al. Sex-specific distributions and determinants of thoracic aortic diameters in the elderly. Heart 106(2), 133–139 (2020).
Ye, M. et al. Diabetes attenuated age-related aortic root dilatation in end-stage renal disease patients receiving peritoneal dialysis. J. Diabet. Invest. 10(6), 1550–1557 (2019).
Pirruccello, J. P. et al. Development of a prediction model for ascending aortic diameter among asymptomatic individuals. JAMA 328(19), 1935–1944 (2022).
Pirruccello, J. P. et al. Deep learning enables genetic analysis of the human thoracic aorta. Nat. Genet. 54(1), 40–51 (2022).
Tian, X. et al. Hypertension, arterial stiffness, and diabetes: A prospective cohort study. Hypertension 79(7), 1487–1496 (2022).
Zheng, M. et al. Arterial stiffness preceding diabetes: A longitudinal study. Circ. Res. 127(12), 1491–1498 (2020).
Chirinos, J. A. et al. Impact of diabetes mellitus on ventricular structure, arterial stiffness, and pulsatile hemodynamics in heart failure with preserved ejection fraction. J. Am. Heart Assoc. 8(4), e011457 (2019).
Cohen, J. B. et al. Clinical phenogroups in heart failure with preserved ejection fraction: Detailed phenotypes, prognosis, and response to spironolactone. JACC Heart Fail 8(3), 172–184 (2020).
Suzuki, K. et al. Pulse pressure, prognosis, and influence of sacubitril/valsartan in heart failure with preserved ejection fraction. Hypertension 77(2), 546–556 (2021).
Arun, D. et al. Exploring the correlation and protective role of diabetes mellitus in aortic aneurysm disease. Front Cardiovasc. Med. 8, 769343 (2021).
Iwamoto, Y. et al. Morphological differences in the abdominal aorta between subjects with and without type 2 diabetes. Cureus 16(9), e68567 (2024).
Elghazaly, H. et al. Characterizing the hypertensive cardiovascular phenotype in the UK Biobank. Eur. Heart J. Cardiovasc. Imag. 24(10), 1352–1360 (2023).
Acknowledgements
None.
Funding
This work was supported by the Liaoning Provincial Applied Basic Research Program (2022JH2/101300061) and Shenyang Science and Technology Project (2024)—Special Project for Public Health Research and Development (24-214-3-10).
Author information
Authors and Affiliations
Contributions
Yuqi Jiang and Wen Tian contributed to the conception and design of the study; Yuqi Jiang and Ziwei Zhao contributed to manuscript drafting; JunFeng Jiang, Yanxia Lin, Pengcheng Jiang, and Yimeng Niu contributed to the statistical analysis and acquisition of data; Huanrui Zhang contributed to the statistical consultation and statistical analysis reviewing; all authors contributed to critical revisions of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval and consent to participate
Ethical approval for this program was granted by the Biomedical Ethics Review Board of The First Hospital of China Medical University (approval number: [2024] No.130).
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Jiang, Y., Zhao, Z., Jiang, J. et al. Diabetes mellitus exacerbates cardiovascular remodeling in elderly Chinese by affecting aortic diameter and pulsatile load. Sci Rep 15, 32275 (2025). https://doi.org/10.1038/s41598-025-17924-z
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-17924-z