Abstract
Reintroduction of native freshwater mussels following aquatic habitat restoration is an increasingly used conservation practice to establish self-sustaining populations. Despite the common use of hatchery-grown subadult mussels (< 40 mm) for reintroduction efforts, their physical habitat requirements as well as guidance on identifying suitable release locations for different mussel species and sizes are severely lacking. We addressed this knowledge gap by deploying three-dimensional (3D)-printed subadult mussels equipped with scannable tags in a river as a proxy for hard-to-track live subadult mussels. We tracked mobilization, displacement and resettlement of 3D-printed and live freshwater mussels at five sites in the Mission Reach of the San Antonio River, Texas, USA, over two years. We found mobilization increased at higher discharges, smaller mussels mobilized at lower flows than adult mussels, site-specific geomorphic features reduced displacement distances, and boulder fields, or habitats with large stable roughness elements, resettled mussels at the highest rate. The study methods and results can directly inform freshwater mussel reintroduction efforts and river restoration project designs that include mussel habitats.
Similar content being viewed by others
Introduction
Freshwater mussels (Bivalvia: Unionoida) are a globally diverse species that provide critical ecosystem services for rivers1,2,3. However, their populations have declined precipitously over the past century due to their extreme vulnerability to habitat degradation—both physical and chemical—and freshwater mussels are now among the most imperiled species in North America and worldwide2,4,5. Reintroduction of native mussels following aquatic habitat restoration is an increasingly used conservation practice to establish self-sustaining populations, with multiple federal, state, and university facilities in the U.S. now propagating freshwater mussels to support conservation efforts5,6,7,8. However, the success of these programs, which depends in part on the selection of suitable physical habitat locations and mussel stocking timing, has been variable and poorly monitored8,9,10. While reintroduction success is higher when using larger individuals7, the typical release size of hatchery-propagated mussels is the subadult life-stage between 20 and 40 mm in length (USFW personal comm, 2022) in order to balance the cost of raising mussels with their likelihood of survival2,7,9,11,12. Even so, little is known about the habitat suitability requirements of the subadult life-stage, defined as the period from when a drifting juvenile settles on the river bed to when it becomes a reproducing adult13. This has been attributed in part to the bias of finding larger mussels (> 40 mm) during visual-tactile searches7,14,15,16, leading to inconclusive or overreaching results related to smaller individuals17.
As sessile benthic organisms, mussels are exposed to life-threatening risks during low and high flow events. While high flows play an important role in connecting mussels to downstream habitats18, they can also be a critical limitation to mussels establishing themselves within new habitats7, particularly during smaller life-stages. To date, studies on freshwater mussel physical habitat suitability under high flow conditions have largely focused on adult mussels19,20,21,22. Studies have found that complex hydraulic variables, which consider direct flow measures—such as water velocity and flow depth—as well as sediment and channel characteristics, can be a reasonable proxy for adult mussel habitat suitability at high flows23,24. However, local hydraulic conditions are difficult to monitor in the field at high flows. Field studies have assessed the mussel response to a single high flow event25 and mussel persistence over time26,27, but gaps remain in terms of assessing relative responses across multiple high flow events and have not considered the smaller life-stages. Additionally, studies often focus on assessing physical habitat conditions where mussels are observed without considering when or from where these mussels settled in their current location, both important factors when considering the entire mussel life cycle28.
Principles and techniques from sediment bedload studies can be leveraged to assess physical habitat requirements of these smaller, harder-to-track subadult mussels. Sediment bedload studies track sediment clasts with Passive Integrated Transponders (PIT) drilled in them to understand how the bedload moves through river systems during storm events. These studies have estimated field-based thresholds for particle incipient motion29,30,31,32, highlighted connections between sediment displacement distances and storm flow magnitude30,33,34,35, and found links between tagged sediment deposition and channel morphodynamics36,37. While freshwater mussels could be idealized as pieces of sediment and many of the results of bedload tracking studies are directly applicable to subadult mussels, sediment bedload studies still only use clasts larger than 40 mm due to limitations with PIT tag sizes and recapture32,36, leaving a knowledge gap in how smaller particles and mussels interact with the flow and bed.
To address the critical knowledge gaps related to suitable physical habitat conditions for subadult freshwater mussels, this study employs three dimensional (3D)-printed adult and subadult freshwater mussel replicates of four different species, Pustulosa pustulosa (Pimpleback), Tritogonia verrucosa (Pistolgrip), Amblema plicata (Threeridge), and Lampsilis teres (Yellow Sandshell), equipped with PIT tags to track their mobilization, displacement and settlement through high flow events across two years. We focused on five sites within the Mission Reach of the San Antonio River (Fig. 1), one of the largest urban river restoration projects in the United States, where the first-in-Texas freshwater mussel reintroduction is underway. While the Mission Reach maintains a constant baseflow, removing the threat of desiccation during low flow periods, the system is capable of creating extremely high flow events that cause high bed shear stress (SI Fig. 4). Over the two-year study, we conducted repeat surveys to track the mussels (Fig. 2), recorded sediment composition and bathymetry changes, and analyzed habitat types from aerial imagery and sites visits to assess suitable mussel habitat across species and sizes. We used the PIT tag data to assess what flows mobilized mussels, how far they displaced when mobilized and where they resettled relative to the habitat available downstream of their introduction sites. We compared mobilization and displacement distances between mussel species and sizes, study sites, bed sediment composition and habitat types to understand how suitable conditions change as mussels grow toward reproducing age. Finally, we released live adult mussels in conjunction with 3D-printed adult mussels to compare mobilization flows and displacement distances between replicate and live mussels.
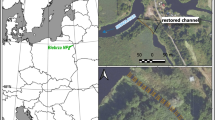
The Mission Reach of the San Antonio River, Texas, USA with the study sites labeled. (a and b) show examples of the 31 concrete riffle structures scattered throughout the Mission Reach that provide aquatic habitat while allowing small watercraft to pass each structure using the canoe chute. Base map sources: Esri, DeLorme, HERE, MapmyIndia (https://www.arcgis.com/home/webmap/viewer.html) with modifications completed using QGIS (version 3.22.3).
Deployment details at a study site in the Mission Reach of the San Antonio River, Texas. (a) The deployment layout at site S3 for Year 1 (green circles) and Year 2 (orange triangles). The inset box in the lower left corner shows an example of the 3D-printed A. plicata mussels in the adult and 20 mm, or subadult, lengths that were deployed throughout the site during both years. (b) Deploying mussels using the layout grid (white square) to ensure each mussel was equidistant apart in Year 1. (c) Scanning the study area for mobilized mussels using a backpack-based antenna with GPS unit.
Results
A total of 1047 mussels were released over two field seasons. 64.8% (n = 678) of those mussels were rescanned at least once and 88.1% (n = 597) of rescanned individuals were included in the analysis (Table 1). 59.3% (n = 354) of the mussels analyzed were 3D-printed 20 mm mussels, 30.0% (n = 179) were 3D-printed adult mussels, and the rest were live adult mussels (10.7%; n = 64). In terms of species, T. verrucosa had the highest rescan rate (70.6%; n = 161) while L. teres had the lowest rescan rate (50.7%; n = 150; Table 1).
Discharge mobilization rates
Overall, the percentage of mussels mobilized increased with peak flow (Fig. 3; SI Table 2). While Year 1 of the field study had lower peak flows compared to Year 2, the mobilization rates observed in Year 1, calculated as the number of mussels rescanned outside their deployment area compared to the total number of mussels not yet scanned as mobilized, were higher than the mobilization rates observed in Year 2 for similar sized peak flows. For example, a 2.0 m3/s event caused 13.4% of mussels to mobilize in Year 1 while a similar-sized storm in Year 2 (3.6 m3/s) caused 7.1% of mussels to mobilize (Fig. 3). The largest storm over the study period (512.5 m3/s, ~ 2-year recurrence event) caused the largest percentage of mussels to mobilize (50%; Fig. 3).
Percent of mussels mobilized by scan visit in the Mission Reach of the San Antonio River, Texas. Percent mobilized is the ratio of mussels that were found displaced during a scan visit compared to the number of mussels that were included in the given study year but not yet mobilized. Scan visits from Year 1 are blue squares and Year 2 are orange circles. The boxed points represent scan visits after similar size flow events for each study year, showing an improvement in percent mobilizations from year to year.
Mussel mobilization rates also varied based on the physical habitat type of the deployment location (Figs. 4a, 5). Mussels were deployed in five habitat types: Bank Habitat (n = 99), Front of Point Bars (n = 34), Back of Point Bars (n = 39), Boulder Fields (n = 135) and Main Channel (n = 290). Boulder Fields and Back of Point Bars both had mobilization rates of 44%, Bank Habitat had a 60% mobilization rate, Main Channel had 61% and Front of Point Bar had a 71% mobilization rate (Fig. 4a).
Mobilization and resettlement differences by habitat type in the Mission Reach of the San Antonio River, Texas (see SI Fig. 1 for example map of habitat types) based on final scan data for each mussel and where those mobilized mussels resettled. (a) shows the percent mobilization categorized by habitat type. These percentages are converted to mussel counts in the center of the diagram, which represents the starting habitat type for all mussels that were mobilized during both study years. The Sankey diagram (b) then shows where those mussels resettled, again categorized by habitat type.
Deployment, mobilization, and resettlement of freshwater mussels across sites S1 (a and b) and S3 (c and d) for Year 1 (a and c) and Year 2 (b and d) of the study. Flow is from top to bottom. Both live and 3D printed mussels of all sizes are included. White triangles and circles represent the initial and secondary deployment locations, respectively, of mussels during a given study year. The size of the deployment symbols indicates the number of mussels that were stationary throughout the study year at a given deployment location. Black circles indicate the resettlement location of mobilized mussels and the color of the path indicates the survey date the displacement was recorded. Mussel displacements are overlaid on habitat type plots derived from aerial imagery and site visits.
Discharge mobilization thresholds
Mobilization thresholds, defined as the maximum flow recorded between site visits during which a mussel mobilized, varied across size classes and species (Table 1). Generally, printed subadult mussels mobilized at a lower median discharge (85.0 m3/s) compared to the printed adult mussels (104.8 m3/s) across all species (Mann–Whitney U; p < 0.05; r = − 0.1962), but they mostly exhibited the same range of mobilizing flow thresholds (Table 1). While the printed 20mm mussels had similar mobilization rates across the four species (Table 1), median mobilization thresholds were more varied with P. pustulosa, T. verrucosa, A. plicata and L. teres having median thresholds of 85.0, 85.0, 41.3, and 41.3 m3/s, respectively, which were significantly different (Kruskal–Wallis H; p < 0.05; ε2 = 0.041). A post-hoc Dunn’s test showed P. pustulosa had significantly different mobilization thresholds from A. plicata (p < 0.05; r = 0.2753). The printed adult mussels had a much larger range in terms of both mobilization rate (42% for P. pustulosa to 70% for L. teres) and median mobilization threshold (46.2 m3/s for A. plicata to 407.8 m3/s for P. pustulosa) but consistently recorded the highest mobilization thresholds within each species. These differences in thresholds can be attributed to the small number of printed adult mussels rescanned, especially after the largest flow events (SI Table 2). The live adult mussels had the same median mobilization thresholds for both species (11.2 m3/s) but these were significantly lower than the thresholds for printed adults of A. plicata (46.2 m3/s; Mann Whitney U; p < 0.05; r = − 0.4609) and L. teres (104.8 m3/s; Mann–Whitney U; p < 0.05; r = − 0.4083).
Mobilization thresholds also varied across study sites, with the lowest median threshold of 41.3 m3/s at S1 and the highest threshold of 407.8 m3/s at S2. Sites S3 and S5 had thresholds of 85.0 m3/s and S4 had a median threshold of 46.2 m3/s. These thresholds were significantly different (Kruskal–Wallis H; p < 0.05; ε2 = 0.024), but the Dunn’s test showed no significance between any individual sites. As for habitat types, both Bank Habitat and Boulder Fields had the highest median mobilization threshold of 104.8 m3/s, while Front of Point Bar had the lowest threshold of 41.3 m3/s. Back of Point Bar had a median threshold of 85.0 m3/s and Main Channel had a threshold of 46.2 m3/s. These thresholds were significantly different (Kruskal Wallis H; p < 0.05; ε2 = 0.037) and the Dunn’s test showed additional significance between Back of Point Bar and Front of Point Bar (p < 0.05; r = 0.4377) along with Back of Point Bar and Main Channel habitat (p < 0.05; r = 0.1847) and Boulder Field and Front of Point Bar (p < 0.05; r = − 0.291). Finally, the bed sediment where mussels were stable versus where mussels were mobilized was very similar. The median D50 was 25.3 mm for mobilized mussels and 24.8 mm for stable mussels, which was not significantly different (Mann–Whitney U; p = 0.26; r = 0.046; SI Fig. 2).
Displacement distance
Across all species and sizes, mussels were displaced a median distance of 55.5 m during the largest observed flow event and a median distance of 4.3 m during the smallest flow event (SI Table 2). Both printed mussel sizes were displaced similar distances (5.7 m for 20 mm printed mussels versus 7.6 m for adult printed mussels; Mann Whitney U; p = 0.07; r = − 0.102), while the live mussels were displaced significantly less median distance (3.1 m; Mann–Whitney U; p < 0.05; r = − 0.40; SI Fig. 5a). By species, P. pustulosa, T. verrucosa, A. plicata and L. teres printed mussels displaced a median distance of 6.6 m, 5.6 m, 6.8 m and 5.5 m, respectively, which was not significantly different (Kruskal Wallis H; p = 0.74; ε2 = − 0.006; SI Fig. 5c). The only exception was that live L. teres mussels displaced significantly shorter distances (3.0 m) than the printed mussels of L. teres (9.0 m; Mann Whitney U; p < 0.05; r = − 0.5072; SI Fig. 5a). When the data was paired down to only the adult mussels released in the second cohort of Year 2, the displacement distances of the live and printed mussels showed no significant difference (Mann–Whitney U test; p = 0.36; r = − 0.127), meaning the travel distance discrepancy was likely a product of the deployment scheme.
Deployment habitat types and study sites both influenced displacement distance. Mussels that were deployed in the Front of Point Bar habitat had the shortest median displacement distance of 4.3 m. Main Channel, Boulder Field, Bank Habitat and Back of Point Bar followed with 5.1 m, 5.3 m, 6.0 m, and 14.4 m, respectively. These distances were statistically significant (Kruskal Wallis H; p < 0.05; ε2 = 0.035) and the post-hoc Dunn’s test found that Back of Point Bar differed from the other groups significantly, but the four other habitat types were similar (SI Table 6). By site, S4 had the shortest median displacement distance of 3.1 m, followed by S3, S1, S5 and S2 with median displacement distances of 4.6 m, 6.4 m, 7.6 m, and 25.0 m, respectively. These displacements were significantly different from one another (Kruskal Wallis H; p < 0.05; ε2 = 0.067) and the post-hoc Dunn’s test showed the most significant difference between S4 and S2 (p < 0.05; r = − 0.58) with other differences of note between S4 and S3 (p < 0.05, r = 0.329), S4 and S2 (p < 0.05; r = 0.328) and S1 and S3 (p < 0.05; r = − 0.220; Fig. 5). Site S4 also had the highest ratio of short-distance displacements (< 10 m), while S1 had the highest ratio of long-distance displacements (> 50 m; Fig. 5; SI Fig. 5b).
Resettlement locations
Resettlement locations were very influenced by habitat type. While at least one mussel resettled in each habitat type (Fig. 4b), Main Channel was the only habitat type with resettlement at all five sites (SI Table 1). Overall, Main Channel habitat had the most mussel resettlement (206 mussels) and the most resettlement from mussels deployed in other habitat types (64 mussels or 31%). Of the 24 mussels that resettled in Back of Point Bars, 66% were deployed in other habitat types, the highest resettlement rate from other habitat types. Front of Point Bars had the lowest resettlement overall with 22 mussels, while Bank Habitats resettled 42 mussels and Boulder Fields resettled 43 mussels. The percent of mussels resettled from other habitats to Boulder Fields was 35%, to Bank Habitats was 48% and to Front of Point Bars was 63%.
When considering resettlement habitat relative to available habitat, Boulder Field habitats captured an average ratio of 3.93 mussels resettled per available habitat area while Bank Habitats captured an average ratio of 0.54 (SI Table 1). The Boulder Field habitat in S3 (Fig. 5) had the highest ratio of mussels resettled per available habitat area (12.59), indicating over twelve times as many mussels settled in Boulder Field habitats at S3 than expected based on the availability of Boulder Field habitat downstream from each mussel’s respective deployment zone. Averages are reported here rather than medians as many locations had no resettlement resulting in median resettlement ratios of 0. The resettlement ratios were significantly different with respect to habitat type (Kruskal Wallis H; p < 0.05; ε2 = 0.17) and the post-hoc Dunn’s test showed significant difference between Main Channel and all but the Boulder Field habitat, as well as Boulder Field being different from both Front and Back of Point Bar Habitats (SI Table 5).
Discussion
This study tracked mobilization, displacement and resettlement of PIT-tagged 3D-printed and live freshwater mussels at five sites in the San Antonio River Mission Reach over two years. Our findings fill a critical knowledge gap in the physical habitat thresholds of subadult mussels13. The study methods and results can directly inform freshwater mussel reintroduction efforts and river restoration project design.
Using 3D-printed mussels in-situ could help researchers and natural resources managers determine where subadult mussels may be most likely to survive to reproducing age, in terms of habitat type, bed composition and flow regime. This novel approach allowed potential reintroduction sites to be conservatively tested for bed stability before introducing live specimens to the river, as monitoring—and therefore deployment recommendations—of translocated or reintroduced populations is rare12. Through our iterative multi-year deployment scheme, the mobilization rates in Year 2 were lower than in Year 1 for similar sized flow events (Fig. 3; SI Table 2), suggesting that informed deployment location selection leads to higher reintroduction success rates for subadult mussels.
Utilizing 3D-printed mussels equipped with internal PIT tags also allowed for tracking of smaller mussels than was previously possible in similar studies using both mussels8 and sediments32,36. The PIT tag rescan rates (63%) were similar to other sediment tracking studies of similar design32,38,39, but higher rescan rates have been seen in other studies that employed different tag types or used larger tag sizes34,40. The rescan rates obtained during this study were similar to those found during an ex-situ scan testing (SI Table 3), indicating we may be at the upper limit for scan efficiency using this combination of tag sizes and experimental set up. The lower rescan rates could be due to tag interference when multiple tags are in close proximity, tag hiding when larger and smaller tags were in close proximity, antenna detection range, tag burial, tags leaving the study area, or tags in pools deeper than was scannable by our equipment38,41. Even with these missed data points, this study showed it is possible to track smaller mussels (20 mm) using PIT tag technology (12 mm tags), but larger numbers of tags are necessary to combat the relatively low rescan rate. This is something that is more easily overcome with the relatively low cost and effort needed to create the 3D-printed replicants as compared to more traditional methods, especially with rare species.
Mussels of all sizes and species exhibited the lowest mobilization rates in Boulder Field and Back of Point Bar habitats. These habitats were defined as having boulder or other large roughness elements that were exposed above the water surface during base flow or on the downstream side of bars and islands, respectively. Both of these habitats exhibit similar flow refugia features. Groupings of boulders are known to cause flow deceleration, interrupt local bed shear stress, entrap incoming sediments and reduce fine sediment erosion42,43,44, all of which could lead to more stable habitat, shorter displacements and frequent resettlement45,46. Hastie et al.25 also found boulder-stabilized substrate to be important for juvenile Margaritifera margaritifera. In addition, bed elevation changes of considerable magnitude (greater than 20mm or the length of the subadult mussels) were seen at some deployment areas (SI Fig. 6), which could be connected to high mobilization rates recorded. While not directly measured, the boulder field habitats could create refuge from these scour events as noted in previous studies43,44.
Mobilization thresholds were dependent on mussel size. Printed 20 mm subadult mussels exhibited lower discharge mobilization thresholds across all species compared to the printed adult mussels of the same species (Table 1), which was a similar result to sediment tracking studies looking at incipient motion thresholds29,30,31,32. Interestingly, the live adult mussels had median mobilization thresholds that were significantly lower than those of the printed adult mussels (Table 1). This was unexpected, as live mussels have behavioral response mechanisms, such as burrowing and horizontal movement47, to help keep them more stable through high flow events2. An unpublished flume study comparing mobilization thresholds between printed and live subadult mussels found these protective behaviors to be important for stability, especially as mussels grew in size. This discrepancy may be attributed to the deployment scheme, as live mussels were not deployed until the second release of Year 2. This meant they were not monitored through a high flow event; the maximum flow felt by this cohort was 46.2 m3/s (~ 0.3-yr recurrence) as compared to the 512.5 m3/s (~ 2-yr recurrence) that was experienced at the beginning of Year 2. The highest flow events caused the highest mobilization rates for printed mussels (Fig. 4; SI Table 2), leaving a gap in the data for live mussels. Multi-phased deployment schemes are common in freshwater mussel studies7,12, but the results can be difficult to interpret if the flow regime felt by each cohort is different, as was the case here. Future studies should consider releasing all size classes of interest in each cohort and minimizing the number of cohorts to simplify size and mobilization relationships.
Channel complexities such as boulder fields and vegetated bars influenced both travel distance and resettlement rates, regardless of mussel size or species. Mussels mobilized at site S1, which had the largest proportion of Main Channel habitat, including pools and runs (SI Table 1), tended to travel further than those at other sites (Fig. 5; SI Fig. 5c). Chapuis et al.37 also found longer displacement distances when tracers were more exposed to flow, as in a run. Conversely, S3 and S4 had the shortest displacement distances, which could be attributed to the greater number of large roughness elements and bars that could slow or catch drifting mussels (Fig. 5). Resettlement numbers were highest in Main Channel habitats, while Boulder Field habitats settled mussels from within the same habitat most frequently, meaning mussels that mobilized within a Boulder Field often also resettled within the same habitat. One limitation in this study was classifying habitat that lies completely under the water surface at regulated baseflow conditions. Future studies should consider further characterizing Main Channel habitat based on the presence of large roughness elements below the surface and other channel forms (i.e., pools, runs) that may create additional suitable habitat conditions.
Identifying areas with suitable habitat for freshwater mussel reintroductions is a challenge for natural resource managers, especially for those species with only remnant populations still present, as is the case in the Mission Reach of the San Antonio River. The methods and results from this study provide novel ways to assess potential subadult mussel habitat without needing to acquire or risking the loss of live specimen. Assessing potential habitat through both qualitative (habitat types) and quantitative measures (3D-printed mussel mobilization trends) as demonstrated here could help focus reintroduction efforts within a river corridor with limited data collection. While fine scale assessments of local hydraulic conditions have been shown to be helpful in determining suitable mussel habitat20,28, the results herein still provide broadly applicable and complementary metrics to assess potential suitable habitats for reintroductions.
Results from this study can also inform river restoration design to provide more suitable habitat conditions for freshwater mussels over their lifecycle, including the subadult life-stage. We recommend building boulder field habitats and other large roughness features that would remain stable at a recurrence interval equal to or beyond that of the maturity age of the target species. Based on our findings that resettlement preferentially occurred within boulder fields, installing boulder-dominated fall out areas downstream of current or planned mussel beds could provide suitable resettlement habitat in the case that mussels get mobilized from their reintroduction habitat.
In conclusion, this study employed 3D-printed mussels to assess the likelihood of subadult freshwater mussel reintroduction success based on easy-to-collect physical parameters. We found subadult mussels to be most stable within boulder fields and where the bed topography was more stable. We also showed boulder fields are the most likely habitat to resettle mobilized mussels during a large flow event in an area-weighted analysis. These findings could inform freshwater mussel reintroduction decisions related to mussel deployment size and locations as well as provide design recommendations for river restoration projects that include mussel habitats.
Methods
Study area
The study area encompasses the Mission Reach, an 8 km stretch of the San Antonio River just downstream of the city of San Antonio, Texas, USA (Fig. 1). The headwaters are controlled by the city of San Antonio for recreation and ecological function and provide a consistent baseflow of approximately 0.4 m3/s throughout the year. The region has hot summers and mild winters, with most precipitation falling in the spring and fall (average annual rainfall 768 mm). The upper watershed is highly urbanized, resulting in very flashy flow events with flood peaks exceeding 300 m3/s, and continued urbanization and climate changes are enhancing these trends48.
To support flood conveyance and provide aquatic habitat, the US Army Corps of Engineers completed a $200 million river restoration project in 201349. This project returned the ecosystem to a more natural channel morphology and included the installation of 31 concrete channel-spanning structures to provide additional aquatic habitat and assist with flood conveyance (Fig. 1b and c). These structures include a canoe chute bisecting the concrete sill to allow personal watercraft to float the river year-round and a semi-permanent boulder field downstream of the sill to create riffle habitat. Since the restoration was completed, fish, birds and insects have recolonized the river50. However, native freshwater mussel populations have not recovered in the expected numbers, despite long term monitoring confirming that the water quality is consistently adequate to support mussels51, minor populations of mussels being present in the unconnected relic side arms of the historic channel (San Antonio River Authority, unpublished data) and recent studies confirming the presence of host fish species50.
The San Antonio River Authority (SARA) is planning to use hatchery-propagated mussels to reintroduce four species of freshwater mussels to the Mission Reach that are currently found in downstream reaches of the San Antonio River: Pustulosa pustulosa (Pimpleback), Tritogonia verrucosa (Pistolgrip), Amblema plicata (Threeridge), and Lampsilis teres (Yellow Sandshell). Five potential reintroduction sites were selected for this study based on general channel morphology, sediment type, and accessibility (Fig. 1). The sites were between 50 and 250 m in length and extended from one of the concrete riffle structures at the upstream end of the site to the next downstream riffle structure or permanent boundary marker. The bed composition of these sites is highly variable, ranging from clay bedrock to fine sand to large cobble and boulders, with gravels and cobbles being the most common.
Experimental design
Mussel 3D-printing
The 3D-printing process for creating subadult mussel replicates included the following key steps. First, fully-intact empty adult mussel shells of the four target species obtained from Texas Parks and Wildlife Division (TPWD) were scanned (NextEngine 3D Scanner, model 2020i) to create 3D renderings (Blender ver. 2.92.0; The Blender Foundation). A shell thickness of 0.1 cm was added to the rendering to provide structural support and ensure no holes would form during printing. Next, mussel mass was determined for the target printing lengths of 20 mm and adult, or the mean size of the live mussels measured, using species-specific power functions created using measurements from known mussel beds in a downstream reach of the San Antonio River (SI Fig. 3). Mussels then were printed in high-visibility colors to aid in tracking efforts and final recovery following the field study. Next, the mussel shells were outfitted with an HDX PIT tag (ORFID, Portland, OR, USA), with the 20 mm and adult sizes having 12 mm and 32 mm tags, respectively. Finally, lead weights and silicone filled the remaining shell space to reach the correct mass and the shell halves were sealed closed.
Data collection
Field data collection related to mussel mobilization, displacement and resettlement took place over the summers of 2022 and 2023 (SI Fig. 4). Data from 2022 were used to inform the deployment experimental design in 2023.
Mussel deployment and tracking
Printed mussels were deployed and tracked over the course of two field seasons, where the second-year deployment scheme was informed by the first-year results. In Year 1, 600 20 mm printed mussels were deployed across three field sites (S1, S3 and S5 in Fig. 1). Each of the four target species were equally represented and the mussels were placed in the corners of a layout grid to ensure the mussels were far enough apart to capture all the tags during a rescanning event (Fig. 2). This grid also kept each deployment group in similar flow and sediment conditions. There were six transects at each site with six deployment locations along each transect plus seven deployment locations along each margin, independent of the transects (Fig. 2). The transect locations were selected in collaboration with SARA to span a wide range of sediment types, geomorphic features and distances from an upstream concrete riffle structure. See Supplemental Information Fig. 1 for all site deployment layouts. Each mussel was partially embedded in the substrate and oriented in the direction of flow at deployment to mimic a live mussel at the surface of the riverbed (personal comm., Clint Robertson, TPWD). Thirty-six printed adult mussels of each species (see SI Table 5 for sizes) were added to this deployment scheme part of the way through Year 1 along two transects within each site. These mussels were embedded around the outside of the 20mm mussel deployment grid to reduce tag interference.
The Year 2 deployment scheme prioritized areas that exhibited high stability within the three Year 1 study sites, as well as added two additional potential reintroduction sites. In Year 2 both sizes (20mm and adult) of each of the four target species were initially deployed in March 2023 (SI Fig. 4), and spread across an area of approximately 1 m x 1 m to reduce tag interference41. There were 2–5 deployment locations per study site (Fig. 2; SI Fig. 1). A secondary deployment occurred at all five sites in July of 2023 that included both 3D-printed adult mussels and live adult mussels (SI Table 5). This release only included L. teres and A. plicata, as these were the only readily available live adult mussels. The live adult mussels were sourced from beds in the Lower San Antonio River Basin. They were transported in buckets to the study area and each mussel had a 23 mm ORFID tag attached to the outside of the shell near the umbo using Loctite glue (Loctite, Connecticut, USA). The pairs of printed and live mussels were embedded approximately 0.5m apart from each other within the selected deployment area.
Rescan visits were triggered by either high flow events or duration elapsed without a significant flow event based on discharge readings from the USGS gage 08178565 for the San Antonio River at Loop 410 (Fig. 1; SI Fig. 4). During each rescan visit, the entire wadable area within a study site was scanned using an Oregon RFID backpack scanner equipped with a GPS to inform PIT tag scan locations. Starting at the downstream end of a site, sweeping motions with the scanning wand were made while moving perpendicular to flow, moving upstream until the entire wadable area of the study site was covered. Additional time was spent scanning deployment areas, as the higher density of tags in these areas made missed or covered tags more likely38. An asynchronous tag scanning test within a replicated deployment area showed closely arranged tags were more difficult to rescan than buried tags, especially when a smaller tag was near a larger tag (SI Table 3). To address this, tags pinged by the backpack scanner were read in real-time on a Bluetooth tablet and checked off from a list to allow more thorough and precise scanning efforts in the higher tag density areas.
Physical habitat measurements
Sediment samples and bathymetry measurements were taken at each mussel deployment location and habitat types were determined from site visits and aerial imagery. Sediment samples were taken in both years of the study at all deployment locations except for the margin sites in Year 1. The margin sites were associated with the closest transect deployment location. Samples were excavated from the center of the deployment site and included sediments up to 5 cm into the bed to account for embedment depth of the various mussel lengths. These samples were air dried to remove standing water and then put in a drying oven for 24 h at 100 °C. A sediment gradation was determined with each sample using sieves sized: 64, 31.5, 16, 8, 4, 2, 1, and 0.5 mm.
Bathymetry was surveyed at all mussel deployment transects in Year 1 and at selected locations in Year 2 based on deployment locations and known areas of channel bed changes (SI Fig. 1). In Year 1, a Trimble TDC150 (cm accuracy model) with RTK network correction (Trimble Inc, Colorado, USA) was used for all data collection. In Year 2, the Trimble unit was used to establish a permanent benchmark and to establish pin locations at the ends of each cross section. The transects were collected using level, stadia rod and tape due to extreme vegetation cover creating high vertical uncertainty in the Trimble points. Elevations were taken every 0.5-1 m along each transect, depending on the transect length.
Habitat types were manually delineated through a combination of site visits, site photos and aerial imagery from Google Earth Pro (Google, California, USA) and Nearmap (Nearmap, Utah, USA). The entire wetted channel at baseflow was classified into five habitat categories following Randklev et al.20 (Fig. 6; SI Fig. 1). To classify a site, first bank lines were delineated, then Bank Habitat was considered to be 1 m into the channel from each bank line. Front of Point Bar and Back of Point Bar included the upstream and downstream ends of instream bars and islands, respectively. Boulder Field included all areas where large roughness elements protruded above the water surface at baseflow and could be seen on aerial imagery. Finally, Main Channel included all other habitat types, such as runs and pools. SI Fig. 1 shows a marked up aerial image for each of the five study sites to show how the habitats were delineated for reproducibility.
Data analysis
Mark-recapture data preprocessing
The protocols for processing mussel mark-recapture data prior to calculating discharge mobilization thresholds and displacement distances are detailed below. Mussels that were never scanned during the entire field season were removed from the dataset. Raw scanner data from each rescan visit was reduced to one point per mussel by averaging all scan coordinates, then the distance between the deployment location and the scanned location was calculated. Mussels with mobilization distances less than 2 m were considered stable, or still in their deployment position, and any displacement beyond 2 m was considered mobilized. This distance was based on the potential offset between the scanning wand and the backpack GPS unit, as well as the accuracy of the backpack GPS unit. Only initial mobilizations, displacement distances and resettlement locations were considered for all analyses due to successive displacements having an undeterminable influence of the unknown mussel orientation within the bed.
Some additional assumptions were made to simplify the dataset based on scan timing and locations. First, if a mussel was not scanned during a rescan visit but was scanned during a future visit in its deployment location, then for all previous scan trips it was assumed that mussel was stable and in its original position52. Second, if a mussel that was marked as displaced but a future scan found it within 2 m of the deployment location, this stable reading overrode the previous displacement and the previous displacement point was dropped from the dataset. Third, due to scanner error (i.e. loss of GPS accuracy) or the position of the backpack relative to the mussel, there were times the mussel’s displacement location was upstream of the deployment location. These upstream displacements were removed from the dataset. Finally, if a mussel was not scanned after a large flow event but was documented being mobilized during a subsequent smaller flow event, the initial mobilization was allocated to the larger previous event, as it is most likely the mussel was displaced during the larger event, but due to burial or tag interference, the mussel was not scanned until a later visit52.
Discharge mobilization rates
To understand how high flow events influenced mobilization, we calculated mobilization rates, or the number of mussels mobilized during a given scan visit compared to those that had not yet been mobilized (SI Table 2). Mobilization rates were determined for each rescan visit and plotted against the peak flow rate between rescan trips (Fig. 3). Mobilization rates were also determined for each mussel species and size (Table 1), which were a cumulative rate of mobilization across all rescan visits for each mussel type.
Discharge mobilization thresholds
Mobilization thresholds were evaluated as the maximum flow rate that occurred between when the mobilization was recorded and the previous field visit for each individual mussel. The discharge mobilization thresholds were grouped by species and size (Table 1), and compared across habitat types, study sites and within species and size groups. A low mobilization threshold meant mussels were mobilized from their initial position at a lower flow rate and infers higher site instability for that mussel species and size than mussels with higher mobilization thresholds. Additionally, we compared the deployment location sediment compositions (D50) and the habitat types between mussels that were stable throughout their respective deployment period and those that were mobilized, considering all mussels sizes and species (SI Fig. 2).
Displacement distance and resettlement
Displacement distances were compared to the maximum flow rate that occurred between the last stable scan and the scan after displacement (SI Table 2). These distances were compared between sizes, species, habitat types and study sites. We also determined the habitat types of each mussel resettlement location following their initial mobilization and calculated resettlement rates within each habitat type. Additionally, the habitat type in which resettlement occurred was compared to the overall habitat type availability downstream of each deployment area following a modified forage ratio approach53,54. While the original intention for this equation was to define an organism’s preference for a certain type of prey, here it was used to calculate the preference of resettlement based on habitat types. For use with drift-prone aquatic species, such as mussels mobilized from the bed during a high flow event, the forage ratio was defined as the percent of mussels resettled into each habitat type relative to the percent of available area of each habitat type perpendicular to and downstream of each deployment zone.
Statistical analysis
The non-parametric Mann–Whitney U and Kruskal–Wallis H tests (with a post-hoc Dunn’s test using a Bonferroni correction) with an alpha of 0.05 were used to assess the significance the results for each study aim. Differences in mobilization flows between the 20 mm printed mussel species (P. pustulosa [n = 69], T. verrucosa [n = 67], A. plicata [n = 39] and L. teres [n = 42]) were compared using a Kruskal–Wallis H test with a post-hoc Dunn’s test. The mobilization flows of live and printed mussels for A. plicata (live [n = 12], printed [n = 29]) and L. teres (live [n = 13], printed [n = 33]) were compared using Mann–Whitney U tests for each species individually. The bed sediment difference between locations where mussels mobilized (n = 337) versus those that were stable (n = 259) was tested using Mann–Whitney U test. Displacement distance differences between the printed mussel sizes (20 mm [n = 216] and adult [n = 96]), as well as between live (n = 25) and printed (n = 96) adult mussels, were compared using Mann–Whitney U tests. Displacement distance differences between study sites (S1 [n = 93], S2 [n = 112], S3 [n = 103], S4 [n = 21], S5 [n = 8]) was compare with a Kruskal–Wallis H test with a post-hoc Dunn’s test. Finally, resettlement ratios across different habitat types for each deployment site used throughout the two-year study (MC [n = 65], RF [n = 45], FBP [n = 50], BPB [n = 58], BH [n = 65]) were also compared using a Kruskal–Wallis H test with a post-hoc Dunn’s test. All statistical analysis was completed using Python version 3.9.13 (Python Software Foundation, Delaware, USA) and the SciPy Stats package (version 1.14.1).
Data availability
The datasets generated during and/or analyzed during the current study are available in the HydroShare repository, http://www.hydroshare.org/resource/202c20c9026e4e278601baeb0e2a9094.
References
Strayer, D. Freshwater Mussel Ecology: A Multifactor Approach to Distribution and Abundance (University of California Press, 2008).
Haag, W. R. North American Freshwater Mussels: Natural History, Ecology, and Conservation (Cambridge University Press, 2012).
Vaughn, C. C. Ecosystem services provided by freshwater mussels. Hydrobiologia 810(1), 15–27. https://doi.org/10.1007/s10750-017-3139-x (2018).
Downing, J. A., Van Meter, P. & Woolnough, D. A. Suspects and evidence: A review of the causes of extirpation and decline in freshwater mussels. Anim. Biodiv. Conserv. 33(2), 151–185. https://doi.org/10.32800/abc.2010.33.0151 (2010).
Haag, W. R. & Williams, J. D. Biodiversity on the brink: An assessment of conservation strategies for North American freshwater mussels. Hydrobiologia 735(1), 45–60. https://doi.org/10.1007/s10750-013-1524-7 (2014).
Hua, D. & Neves, R. J. Captive survival and pearl culture potential of the pink heelsplitter Potamilus alatus. N. Am. J. Aquac. 69(2), 147–158. https://doi.org/10.1577/A05-108.1 (2007).
Carey, C. S., Jones, J. W., Butler, R. S. & Hallerman, E. M. Restoring the endangered oyster mussel ( Epioblasma capsaeformis ) to the upper Clinch River, Virginia: an evaluation of population restoration techniques: Freshwater mussel population restoration techniques. Restor. Ecol. 23(4), 447–454. https://doi.org/10.1111/rec.12195 (2015).
Montesanto, F., Ohlman, L. M. & Pegg, M. A. Survival and growth assessment after reintroduction of the pocketbook mussel, Lampsilis cardium Rafinesque, 1820 among three streams in Nebraska (USA). Aquat. Conserv. 33(6), 535–548. https://doi.org/10.1002/aqc.3942 (2023).
McMurray, S. E. & Roe, K. J. Perspectives on the controlled propagation, augmentation, and reintroduction of freshwater mussels (Mollusca: Bivalvia: Unionoida). Freshwater Mollusk Biol. Conserv. 20(1), 1. https://doi.org/10.31931/fmbc.v20i1.2017.1-12 (2017).
Nakamura, K., Sousa, R. & Mesquita-Joanes, F. Collapse of native freshwater mussel populations: Prospects of a long-term study. Biol. Cons. 279, 109931. https://doi.org/10.1016/j.biocon.2023.109931 (2023).
Thomas, G., Taylor, J. & Garcia de Leaniz, C. Captive breeding of the endangered freshwater pearl mussel Margaritifera margaritifera. Endang. Species. Res. 12(1), 1–9. https://doi.org/10.3354/esr00286 (2010).
Lavictoire, L. & West, C. Population reinforcement of the endangered freshwater pearl mussel (Margaritifera margaritifera): Lessons learned. Diversity 16(3), 187. https://doi.org/10.3390/d16030187 (2024).
Lopez, J. W. & Vaughn, C. C. A review and evaluation of the effects of hydrodynamic variables on freshwater mussel communities. Freshw. Biol. 66(9), 1665–1679. https://doi.org/10.1111/fwb.13784 (2021).
Hornbach, D. J., Allen, D. C., Hove, M. C. & MacGregor, K. R. Long-term decline of native freshwater mussel assemblages in a federally protected river. Freshw. Biol. 63(3), 243–263. https://doi.org/10.1111/fwb.13055 (2018).
Carey, C. S., Jones, J. W., Butler, R. S., Kelly, M. J. & Hallerman, E. M. A comparison of systematic quadrat and capture-mark-recapture sampling designs for assessing freshwater mussel populations. Diversity 11(8), 127. https://doi.org/10.3390/d11080127 (2019).
Pandolfi, G. S., Mays, J. W. & Gangloff, M. M. Riparian land-use and in-stream habitat predict the distribution of a critically endangered freshwater mussel. Hydrobiologia 849(8), 1763–1776. https://doi.org/10.1007/s10750-022-04826-8 (2022).
Lymbery, A. J. et al. Burrowing behavior protects a threatened freshwater mussel in drying rivers. Hydrobiologia 848(12–13), 3141–3152. https://doi.org/10.1007/s10750-020-04268-0 (2021).
Terui, A. et al. Asymmetric dispersal structures a riverine metapopulation of the freshwater pearl mussel Margaritifera laevis. Ecol. Evol. 4, 3004–3014. https://doi.org/10.1002/ece3.1135 (2014).
Maloney, K. O., Lellis, W. A., Bennett, R. M. & Waddle, T. J. Habitat persistence for sedentary organisms in managed rivers: The case for the federally endangered dwarf wedgemussel (Alasmidonta heterodon) in the Delaware River: Quantifying habitat persistence for sedentary taxa. Freshw. Biol. 57(6), 1315–1327. https://doi.org/10.1111/j.1365-2427.2012.02788.x (2012).
Randklev, C. R., Hart, M. A., Khan, J. M., Tsakiris, E. T. & Robertson, C. R. Hydraulic requirements of freshwater mussels (Unionidae) and a conceptual framework for how they respond to high flows. Ecosphere https://doi.org/10.1002/ecs2.2975 (2019).
Hegeman, E. E., Miller, S. W. & Mock, K. E. Modeling freshwater mussel distribution in relation to biotic and abiotic habitat variables at multiple spatial scales. Can. J. Fish. Aquat. Sci. 71(10), 1483–1497. https://doi.org/10.1139/cjfas-2014-0110 (2014).
Flores, N. Y. et al. Assessing habitat suitability for native and alien freshwater mussels in the river Waal (the Netherlands), using hydroacoustics and species sensitivity distributions. Environ. Model Assess 27(1), 187–204. https://doi.org/10.1007/s10666-021-09776-4 (2022).
Zigler, S. J., Newton, T. J., Steuer, J. J., Bartsch, M. R. & Sauer, J. S. Importance of physical and hydraulic characteristics to unionid mussels: A retrospective analysis in a reach of large river. Hydrobiologia 598(1), 343–360. https://doi.org/10.1007/s10750-007-9167-1 (2008).
Roberts, M. O., Jacobson, R. B. & Erwin, S. O. Hydraulics of freshwater mussel habitat in select reaches of the Big River, Missouri. USGS Sci. Investig. Rep. 2022–5002, 49. https://doi.org/10.3133/sir20225002 (2022).
Hastie, L. C., Boon, P. J., Young, M. R. & Way, S. The effects of a major flood on an endangered freshwater mussel population. Biol. Cons. 98, 107–115. https://doi.org/10.1016/S0006-3207(00)00152-X (2001).
Inoue, K., Levine, T. D., Lang, B. K. & Berg, D. J. Long-term mark-and-recapture study of a freshwater mussel reveals patterns of habitat use and an association between survival and river discharge. Freshw. Biol. 59(9), 1872–1883. https://doi.org/10.1111/fwb.12389 (2014).
Hamstead, B. A., Hartfield, P. D., Jones, R. L. & Gangloff, M. M. Changes to freshwater mussel assemblages after 25 years of impoundment and river habitat fragmentation. Aquat. Conserv. 29(12), 2162–2175. https://doi.org/10.1002/aqc.3220 (2019).
Morales, Y., Weber, L. J., Mynett, A. E. & Newton, T. J. Effects of substrate and hydrodynamic conditions on the formation of mussel beds in a large river. J. N. Am. Benthol. Soc. 25(3), 664–676. https://doi.org/10.1899/0887-3593(2006)25[664:EOSAHC]2.0.CO;2 (2006).
Phillips, C. B. & Jerolmack, D. J. Dynamics and mechanics of bed-load tracer particles. Earth Surf. Dynam. 2(2), 513–530. https://doi.org/10.5194/esurf-2-513-2014 (2014).
Houbrechts, G. et al. Evaluation of long-term bedload virtual velocity in gravel-bed rivers (Ardenne, Belgium). Geomorphology 251, 6–19. https://doi.org/10.1016/j.geomorph.2015.05.012 (2015).
Petit, F. et al. Dimensionless critical shear stress in gravel-bed rivers. Geomorphology 250, 308–320. https://doi.org/10.1016/j.geomorph.2015.09.008 (2015).
Ivanov, V., Radice, A., Papini, M. & Longoni, L. Event-scale pebble mobility observed by RFID tracking in a pre-Alpine stream: A field laboratory. Earth Surf. Processes Landf. 45(3), 535–547. https://doi.org/10.1002/esp.4752 (2020).
Hassan, M. A., Church, M. & Schick, A. P. Distance of movement of coarse particles in gravel bed streams. Water Resour. Res. 27(4), 503–511. https://doi.org/10.1029/90WR02762 (1991).
Bradley, N. D. & Tucker, G. E. Measuring gravel transport and dispersion in a mountain river using passive radio tracers. Earth Surf. Processes Landf. 37(10), 1034–1045. https://doi.org/10.1002/esp.3223 (2012).
Phillips, C. B., Martin, R. L. & Jerolmack, D. J. Impulse framework for unsteady flows reveals superdiffusive bed load transport. Geophys. Res. Lett. 40(7), 1328–1333. https://doi.org/10.1002/grl.50323 (2013).
MacVicar, B. J. & Roy, A. G. Sediment mobility in a forced riffle-pool. Geomorphology 125(3), 445–456. https://doi.org/10.1016/j.geomorph.2010.10.031 (2011).
Chapuis, M., Dufour, S., Provansal, M., Couvert, B. & De Linares, M. Coupling channel evolution monitoring and RFID tracking in a large, wandering, gravel-bed river: Insights into sediment routing on geomorphic continuity through a riffle–pool sequence. Geomorphology 231, 258–269. https://doi.org/10.1016/j.geomorph.2014.12.013 (2015).
Stähly, S., Franca, M. J., Robinson, C. T. & Schleiss, A. J. Erosion, transport and deposition of a sediment replenishment under flood conditions. Earth Surf. Processes Landf. 45(13), 3354–3367. https://doi.org/10.1002/esp.4970 (2020).
Liébault, F., Piégay, H., Cassel, M. & Arnaud, F. Bedload tracing with RFID tags in gravel-bed rivers: Review and meta-analysis after 20 years of field and laboratory experiments. Earth Surf. Processes Landf. 49, 147–169. https://doi.org/10.1002/esp.5704 (2023).
Ferguson, R. I., Sharma, B. P., Hodge, R. A., Hardy, R. J. & Warburton, J. Bed load tracer mobility in a mixed bedrock/alluvial channel. JGR Earth Surf. 122(4), 807–822. https://doi.org/10.1002/2016JF003946 (2017).
Chapuis, M., Bright, C. J., Hufnagel, J. & MacVicar, B. Detection ranges and uncertainty of passive Radio Frequency Identification (RFID) transponders for sediment tracking in gravel rivers and coastal environments. Earth Surf. Processes Landf. 39(15), 2109–2120. https://doi.org/10.1002/esp.3620 (2014).
Matthaei, C. D., Arbuckle, C. J. & Townsend, C. R. Stable surface stones as refugia for invertebrates during disturbance in a New Zealand stream. J. N. Am. Benthol. Soc. 19(1), 82–93. https://doi.org/10.2307/1468283 (2000).
Fang, H. W., Liu, Y. & Stoesser, T. Influence of boulder concentration on turbulence and sediment transport in open-channel flow over submerged boulders. JGR Earth Surf. 122(12), 2392–2410. https://doi.org/10.1002/2017JF004221 (2017).
Raus, D., Moulin, F. Y. & Eiff, O. The impact of coarse-grain protrusion on near-bed hydrodynamics. J. Geophys. Res. Earth Surf. 124(7), 1854–1877. https://doi.org/10.1029/2018JF004751 (2019).
Golpira, A., Huang, F. & Baki, A. B. M. The effect of habitat structure boulder spacing on near-bed shear stress and turbulent events in a gravel bed channel. Water 12(5), 1423. https://doi.org/10.3390/w12051423 (2020).
Scully-Engelmeyer, K., Blevins, E., Granek, E. F. & Constable, R. Freshwater mussel populations in Pacific Coast Watersheds (Oregon, USA): Occurrence, condition, habitat, and fish species overlap. Hydrobiologia 850(4), 821–839. https://doi.org/10.1007/s10750-022-05127-w (2023).
Haag, W. R. A hierarchical classification of freshwater mussel diversity in North America: North American freshwater mussel biogeography. J. Biogeogr. 37(1), 12–26. https://doi.org/10.1111/j.1365-2699.2009.02191.x (2009).
Zhao, G., Gao, H. & Cuo, L. Effects of urbanization and climate change on peak flows over the San Antonio River Basin, Texas. J. Hydrometeorol. 17(9), 2371–2389. https://doi.org/10.1175/JHM-D-15-0216.1 (2016).
Lombardi, A. Ecosystem under restoration: A sustainable future for the cultural landscape of San Antonio River, Texas. WIT Trans. Ecol. Environ. 191, 1139–1151. https://doi.org/10.2495/SC140962 (2014).
Turner, R. W. Assessing the Fishery and Economic Value of a Restored Guadalupe Bass Population (University of Texas at San Antonio, 2022).
Opsahl, S. P., Musgrove, M. & Mecum, K. E. Temporal and spatial variability of water quality in the San Antonio segment of the edwards aquifer recharge zone, Texas, with an emphasis on periods of groundwater recharge, September 2017-July 2019. USGS Sci. Investig. Rep. 2020–5033, 37. https://doi.org/10.3133/sir20205033 (2020).
MacVicar, B. J. & Papangelakis, E. Lost and found: Maximizing the information from a series of bedload tracer surveys. Earth Surf. Processes Landf. 47(2), 399–408. https://doi.org/10.1002/esp.5255 (2022).
Kobayashi, D. R. et al. Pelagic habitat characterization of loggerhead sea turtles, Caretta caretta, in the North Pacific Ocean (1997–2006): Insights from satellite tag tracking and remotely sensed data. J. Exp. Mar. Biol. Ecol. 356(1–2), 96–114. https://doi.org/10.1016/j.jembe.2007.12.019 (2008).
Kammel, L. E., Pasternack, G. B., Massa, D. A. & Bratovich, P. M. Near-census ecohydraulics bioverification of Oncorhynchus mykiss spawning microhabitat preferences. J. Ecohydraulics 1(1–2), 62–78. https://doi.org/10.1080/24705357.2016.1237264 (2016).
Acknowledgements
The authors would like to thank the technicians and staff from The Meadows Center for Water and the Environment that were vital to the field campaign: Kannon Byckovski, Shelby Fisher, Aby Panther, Charlotte Piel, Ka’inoa o kekai Lee, Sara Julian, Juliette Barrilleaux, Thomas Heard, Collin Garoutte, and Emily Lorkovic. We would also like to thank the San Antonio River Authority, specifically Austin Davis, Mitch Magruder and Shaun Donovan for their support with site access and local expertise. Finally, we would like to thank Thom Hardy for his guidance and insight in the project conceptualization.
Funding
This work was funded by the US Army Corps of Engineers’ Aquatic Nuisance Species Research Program’s focus on Next Generation Ecological Modeling.
Author information
Authors and Affiliations
Contributions
M.D.: Conceptualization, Data Curation, Methodology, Formal analysis, Writing—original draft, Writing—review and editing. C.R.: Conceptualization, Data Curation, Methodology, Validation, Writing—review and editing. S.S.: Data Curation, Methodology, Validation, Writing—review and editing. C.P.: Methodology, Writing—review and editing. B.L.: Conceptualization, Funding Acquisition, Supervision, Methodology, Writing—original draft, Writing—review and editing.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
DiNicola, M., Riggins, C.L., Schultz, S. et al. Assessing suitable habitat for freshwater mussel reintroductions using 3D-printed subadult replicates. Sci Rep 15, 33119 (2025). https://doi.org/10.1038/s41598-025-18244-y
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-18244-y