Abstract
COVID-19 primarily affects the lungs, but it can also cause neurological, psychological, and cardiovascular complications. Myocardial injury and myocarditis are of significant concerns that are often indicated by elevated troponin levels, a marker of cardiac involvement. This study analyzed risk factors and outcome of elevated troponin-I levels in COVID-19 patients compared to those with normal levels. We included 766 COVID-19 patients of which 50.6% were female, and 74.2% having co-morbidities. Patients were divided into two groups: 383 with elevated troponin levels and 383 with normal levels. We compared their demographics, clinical characteristics and outcome to identify the risk factors for cardiac injury during COVID-19 infection. Data was analyzed through SPSS V.24. Patients with elevated troponin were predominantly male (63.7%) and younger (mean age 58.42 years) than those with normal levels (64.86 years). Despite having fewer co-morbidities and milder COVID-19 cases, participants with raised troponin had lower ICU admission (37.02% vs. 60.2%) and ARDS rates (38.78% vs. 61.2%), yet mortality rates were similar. Ejection fraction remained stable across groups, but Pro-BNP levels were significantly lower in those with raised troponin (8.4% vs. 91.5%). This study highlights significant associations between elevated troponin levels in COVID-19 patients and factors like male gender, comorbid conditions, and ARDS. However, contrary to other studies, higher troponin levels were often found in patients with less severe COVID-19 with negative association with pro-BNP levels. The findings suggest that elevated troponin in COVID-19 does not directly correlate with disease severity or mortality. Further research with longitudinal follow-up and more granular biomarker analysis is recommended to fully elucidate the relationship between troponin levels and cardiac outcomes in COVID-19 patients.
Similar content being viewed by others
Introduction
COVID-19 primarily affects the lungs, but besides that it causes a range of secondary effects, including neurological, psychological, and cardiac complications1. Neurological sequelae include cognitive, memory and peripheral nervous system disorders2. Psychological complications include anxiety and depression which may last for years in some individuals3. Cardiovascular complications present in many ways such as arrhythmias, acute myocardial infarction, heart failure, venous thromboembolic events and myocarditis4. Elevated troponin level is one of the primary markers for detection of cardiac involvement and its severity5. Myocardial injury and myocarditis continue to pose a persistent threat and should not be overlooked in patients with COVID-196.
Association of concomitant co-morbid conditions in the presence of high inflammatory activity may further predispose COVID-19 patients to myocarditis which results in progressive morbidity leading to mortality. Elevated D-dimer and fibrinogen variation have already been reported as indicators of imminent disease severity7. A newly identified predictive value of C-reactive protein/albumin ratio (CAR) for in-hospital mortality in patients with COVID-19 is of importance8. While troponin-I may predict death in patients without pre-existing cardiovascular disease9. Understanding the associated risk factors may help in unraveling the underlying mechanisms involved in the pathophysiological basis of this process. Involvement of ACE2 receptors in the pathogenesis of the COVID 19 disease may lead to the eventual cardiac injury and dysfunction10.
The objective of this study was to determine the risk factors in COVID-19 patients with raised troponin-I levels versus those with normal troponin-I levels by comparing their demographic clinical characteristics. The results of this study may help in estimating the implications for patient outcomes and long-term cardiovascular health. They may also lead to a better understanding of the risks and outcomes of these patients and may facilitate informed clinical decision making.
Methodology
Study design and setting
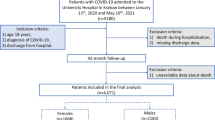
It was a retrospective study conducted from July 2020 till March 2021. The study was conducted at the Sindh Infectious Diseases Hospital and Research Center of Dow University of Health Sciences.
Inclusion criteria
Data of a total of 766 admitted patients with COVID-19 infection was included. Adult patients more than 18 years of age, with proven COVID-19 infection (positive SARS-COV2 polymerase chain reaction) were included. Only those COVID-19 patients were included whose baseline troponin levels were checked at the time of admission. For comparison, two groups of patients were created: one with elevated serum troponin levels at the time of admission or during hospitalization and one with normal troponin levels throughout hospital stay, each containing an equal number of 383 patients. Consecutive sampling technique was used to collect the retrospective data to reach the sample size.
Exclusion criteria
We excluded patients with baseline ejection fraction of less than 45%, unstable ischemic heart disease and patients with recurrent COVID-19 infection. Patients who had COVID-19 symptoms appeared more than 48 h were also excluded from the study.
Data collection
Data for variables used in study like age, sex, co-morbid, disease severity, ICU stay, ARDS, echocardiography finding, serum Pro-BNP and mortality was obtained on a structured proforma from medical records.
WHO guidelines were used to define disease severity and Cytokine Release Syndrome (CRS)11. Acute respiratory distress syndrome (ARDS) was defined as onset of hypoxic respiratory failure (within 1 week), bilateral opacities on chest x-ray and PaO₂/FiO₂ (arterial to inspired oxygen) ratio of ≤ 300 on positive end-expiratory pressure or continuous positive airway pressure ≥ 5 cm H₂O12. COVID-19 disease severity was also defined as per WHO guidelines13.
Measurement of troponin levels
Serum troponin levels were measured using a CLIA (Chemiluminescence immunoassay) method in Dow Diagnostic Research and Reference Laboratory (DDRRL) within 48 h of appearance of COVID-19 symptoms. Raised troponin levels were defined as per institutional laboratory ranges and American Heart Association guidelines (> 53pg/ml in male, > 34pg/ml in female)14. These elevated levels were further categorized into two groups: equivocal troponin levels, defined as values up to twice the upper limit of normal (≤ 106 pg/ml in males and ≤ 68 pg/ml in females), and abnormally elevated troponin levels, defined as values exceeding twice the upper limit (> 106 pg/ml in males and > 68 pg/ml in females).
Statistical analysis
We used SPSS software version 24.0 for data analysis. Categorical variables are given in frequencies and percentages. Continuous variables like age were given in mean and standard deviation. Cross-tabulation/ Pearsons Chi Square test was used to see mortality association with different variables. Fisher’s exact test was applied to see for significant values. Continuous data was compared by Mann witney U test. A p-value of < 0.05 was considered as significant at 95% CI. To avoid confounders, we used multivariate logistic regression model, and results were given as odds ratio: 95% confidence intervals (OR: 95% CI).
Results
A total of 766 COVID-19 patients were studied, of which 388 (50.6%) were females. The mean age was 58.4 ± 13.9 years, and the majority of 569 (74.2%) patients had co-morbidities.
All patients in this study had their baseline troponin levels measured on the day of admission. For those with initially elevated troponin levels or evidence of clinical worsening, repeated measurements were conducted on days 3, 7, 10, and 14.
On comparison of COVID-19 patients having raised troponin with normal serum troponin, it has been found that majority of the patients with raised troponin were males (63.7% vs. 36.24%, p-value < 0.001). In raised troponin group, patients were younger (mean age 58.42 vs. 64.86 years), had less co-morbidities (45.5% vs. 54.4%, p-value = < 0.001), less severe COVID ( 46.75% vs. 53.2%, p-value < 0.001), more mild COVID cases (63.6% vs. 36.3%, p-value < 0.001), less ICU admission (37.02% vs. 60.2%, p-value < 0.001) and less ARDS (38.78% vs. 61.2%, p-value < 0.001) compared to COVID patients with normal troponin. Ejection fraction on Echocardiography was same in both the groups, however, COVID-19 patients with raised troponin had a lower Pro-BNP (8.4% vs. 91.5%, p-value < 0.001) compared to patients with normal troponin. Mortality was the same in both the groups as shown in Table 1.
Severity of rise in troponin levels
Of the 383 patients with raised troponin levels, 338 (88%) had raised troponin on the very first day of admission. The mean serial troponin levels are shown in Fig. 1. In the raised troponin group, 251 (65%) patients had abnormally raised troponin levels of which 123 (49%) patients expired. It was interesting to find that among the raised troponin group, only 34 (10%) patients had ischemic changes on ECG during illness, and 24 (6.2%) patients had abnormal echocardiography findings. To classify further, 72 had levels between 100 and 500 pg/ml, 26 had levels between 501 and 1000 pg/ml, and 68 had levels exceeding 1000 pg/ml. Of those with troponin levels between 100 and 500 pg/ml, 46 (58.97%) required ICU admission and 40 (51.2%) expired. In the 501–1000 pg/ml group, 14 (51.8%) patients required ICU care and 13 (48.1%) expired. Among those with troponin levels greater than 1000 pg/ml, 46 (67.6%) were admitted to the ICU and 30 (44.1%) died, as detailed in Table 2.
Mean of Serial Troponin Levels Measured in Patients with Raised Troponin.
Predictors of raised troponin levels
On multivariate logistic regression analysis, raised troponin in COVID patients is associated with male gender (OR 1.49, 95% CI 1.05–2.1, p-value 0.026), co-morbidities (OR 1.63, 95% CI 0.9–2.6, p-value 0.05) and ARDS (OR 1.69, 95% CI 1.1–2.5, p-value 0.013). However, raised Pro-BNP was inversely related to raised troponin levels (OR 0.73, 95% CI 0.40 − 0.13, p-value < 0.001; Table 3).
Discussion
This retrospective analysis compared patients of COVID-19 having raised cardiac biomarkers Troponin I and pro-BNP versus those having normal cardiac biomarkers and outlined the risk factors associated with increased mortality. The most important findings were that male gender, co-morbid conditions and ARDS were associated with raised Troponin I. Increased Troponin I was also seen more frequently in patients with mild –moderate disease rather than the more severe disease categories and there was no difference in mortality in both the groups.
Cardiovascular involvement in COVID-19 has been well documented in studies from across the world and is considered multifactorial15,16,17.
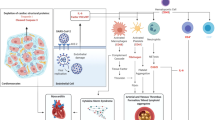
The raised troponin levels may reflect underlying myocarditis in COVID-19 patients18. The multiple mechanisms involved in causing myocardial injury include hypoxia due to severe respiratory compromise, cytokine storm, and direct damage to the vascular endothelium and myocardium, consequential to the increased number of ACE 2 receptors present on it19. The prothrombotic phenomenon seen in SARS-COV 2 infections is also a risk factor for ischemia leading to myocardial infarction, observed in patients with no previous cardiovascular abnormalities as well20,21,22,23. The most common cardiac manifestations reported in COVID-19 include myocarditis, myocardial infarction, arrhythmias and heart failure. These manifestations indicative of myocardial injury are identified by raised cardiac biomarkers like Troponin I or hs-Troponin I, CK –MB, myoglobin and pro-BNP (brain natriuretic peptide) along with ECG and echocardiographic findings22,23,24. Studies have examined the possible association between the prognostic nutritional index (PNI) and in-hospital mortality rates in cases with a high cardiovascular risk burden and hospitalized with COVID-19, which could not be studied in our study due to retrospective nature of the study25.
Previous studies have demonstrated the association between raised cardiac biomarkers, especially Trop I and pro-BNP, and severe COVID-19 irrespective of the presence of an underlying cardiovascular disease22,23,24,26. A meta –analysis by Wen An have shown that Trop I and other cardiac biomarkers like Trop T, CK MB and myoglobin when found to be raised at admission were associated with severity and increased mortality in COVID-1923. Similarly, a large retrospective study from Wuhan, China evaluating 28 days’ mortality in COVID-19 patients also found a statistically significant association between elevated cardiac enzymes including pro-BNP with disease progression and mortality21. In contrast to the above findings, we saw an increased number of patients with mild and moderate COVID-19 with higher levels of Trop I at admission.
The results of our study contrasted with other published studies. Our findings indicated that a higher number of patients with elevated Troponin I levels had mild to moderate disease at admission, presenting with less severe illness, fewer ICU admissions, and fewer comorbidities at a younger age. However, on multivariate logistic regression analysis, our findings corroborated with those of the existing literature. We found that raised Troponin I in COVID − 19 was positively associated with male gender, co-morbidities, ARDS, findings supported by multiple studies both locally and internationally. A prospective study from Pakistan, comparing characteristics of patients with raised Trop I versus normal Trop I showed that males outnumbered females (33.5% vs. 25%) in the group with raised cardiac biomarkers27. Similarly, a meta-analysis by Shafi et al., analyzing sixty-one articles also identified male gender as a risk factor for cardiac injury in multiple studies23.
The most common comorbidities identified in our patients included diabetes, hypertension and cardiovascular diseases (CVD), having a positive association with raised cardiac biomarkers. This supports the findings of previous studies, including the meta-analysis by Shafi et al. which reported hypertension to be the most common comorbidity, identified as a risk factor for acute cardiac injury in patients with COVID 19, followed by CVD and diabetes23. Guo et al. in his study emphasized the importance of Troponin I levels as predictors of cardiovascular complications especially arrhythmias when detected earlier in the disease28. Mumtaz et al. in their retrospective analysis also found a positive association between almost all comorbid conditions and cardiac injury in COVID-19 patients24.
Acute respiratory distress syndrome (ARDS) is an indicator of severe respiratory compromise, a well-known complication of COVID-19, and a hallmark of severe inflammatory reaction including cytokine storm. Studies have shown that ARDS is associated with raised cardiac biomarkers including Trop I and pro-BNP without associated electrocardiographic changes22,24. Similarly, on multivariate analysis we also identified ARDS to be positively associated with raised Trop I, validating the phenomenon of a severe inflammatory reaction leading to cardiac injury.
Pro-BNP has been identified as a quantitative marker of myocardial dysfunction and heart failure and has been found elevated in severe bacterial pneumonias, ARDS, COVID − 19 and other severe viral pneumonias29. In our study we found 166 patients with elevated pro-BNP levels out of which only 8.4% had simultaneously increased Troponin I. This again is in contrast with the existing literature which shows a linear relationship between these two significant markers of cardiac injury, their association with disease severity and even mortality21,22,23. In our study, only a few patients had evidence of myocardial infarction or reduced ejection fraction, suggesting that the aforementioned factors like cytokine storm, hypoxic injury, microvascular thrombosis, and sepsis-induced cardiomyopathy are more likely responsible for troponin elevation than large-vessel arterial thrombosis alone. This is also supported by the inverse relationship between elevated troponin and ProBNP levels.
Limitations of the study
As a retrospective study, it cannot establish causality and may have unmeasured confounding variables.
Absence of Specific Troponin Cutoffs: Abnormally raised troponin levels were not categorized into mild, moderate, and severe elevations, which could provide more detailed insight into the severity of cardiac injury.
Limited Data on ECG Changes: The study does not include ECG data, which could have provided additional insights into cardiac involvement.
No Follow-up for Long-Term Outcomes: Patients were not followed longitudinally, limiting the understanding of the long-term cardiac impact in COVID-19 patients.
Conclusion
This study highlights significant associations between elevated troponin levels in COVID-19 patients and factors like male gender, comorbid conditions, and ARDS. However, contrary to other studies, higher troponin levels were often found in patients with less severe COVID-19. The findings suggest that elevated troponin in COVID-19 does not directly correlate with disease severity or mortality. Further research with longitudinal follow-up and more granular biomarker analysis is recommended to fully elucidate the relationship between troponin levels and cardiac outcomes in COVID-19 patients.
Data availability
All data is provided within the manuscript.
Abbreviations
- ARDS:
-
Acute respiratory distress syndrome
- COVID-19:
-
Corona virus infectious disease 2019
- CRS:
-
Cytokine release syndrome
- CVD:
-
Cardiovascular disease
- DUHS:
-
Dow university of health sciences
- ECG:
-
Electrocardiograph
- IRB:
-
institutional review board
References
Suvvari, T. K., Kutikuppala, L. S., Tsagkaris, C., Corriero, A. C. & Kandi, V. Post-COVID‐19 complications: multisystemic approach. J. Med. Virol. 93 (12), 6451 (2021).
Al-Aly, Z. Long-term neurological sequelae of SARS-CoV-2 infection. Nat. Med. 28 (11), 2269–2270 (2022).
Mazza, M. G. et al. Anxiety and depression in COVID-19 survivors: role of inflammatory and clinical predictors. Brain. Behav. Immun. 89, 594–600 (2020).
Batta, Y. et al. Sequelae and comorbidities of COVID-19 manifestations on the cardiac and the vascular systems. Front. Physiol. 12, 748972 (2022).
Potluri, S., Ventura, H. O., Mulumudi, M. & Mehra, M. R. Cardiac troponin levels in heart failure. Cardiol. Rev. 12 (1), 21–25 (2004).
Long, B., Brady, W. J., Koyfman, A. & Gottlieb, M. Cardiovascular complications in COVID-19. Am. J. Emerg. Med. 38 (7), 1504–1507 (2020).
Hayıroğlu, M. İ., Çınar, T. & Tekkeşin, A. İ. Fibrinogen and D-dimer variances and anticoagulation recommendations in Covid-19: current literature review. Revista Da Associação Médica Brasileira. 66, 842–848 (2020).
Güney, B. Ç. et al. Predictive value of CAR for in-hospital mortality in patients with COVID-19 pneumonia: a retrospective cohort study. Arch. Med. Res. 52 (5), 554–560 (2021).
Çınar, T. et al. Prognostic significance of cardiac troponin level in Covid-19 patients without known cardiovascular risk factors. Am. J. Emerg. Med. 45, 595 (2020).
Babapoor-Farrokhran, S. et al. Myocardial injury and COVID-19: possible mechanisms. Life Sci. 253, 117723 (2020).
Que, Y. et al. Cytokine release syndrome in COVID-19: a major mechanism of morbidity and mortality. Int. Rev. Immunol. 41 (2), 217–230 (2022).
Tasaka, S. et al. ARDS clinical practice guideline 2021. J. Intensive Care. 10 (1), 32 (2022).
Bhimraj, A. et al. Infectious diseases society of America guidelines on the treatment and management of patients with COVID-19 (September 2022). Clin. Infect. Dis. 78 (7), e250–e349 (2024).
Sandoval, Y. et al. High-sensitivity cardiac troponin and the 2021 AHA/ACC/ASE/CHEST/SAEM/SCCT/SCMR guidelines for the evaluation and diagnosis of acute chest pain. Circulation 146 (7), 569–581 (2022).
Aghagoli, G., Gallo Marin, B., Soliman, L. B. & Sellke, F. W. Cardiac involvement in COVID-19 patients: risk factors, predictors, and complications: A review. J. Card. Surg. 35 (6), 1302–1305 (2020).
Bansal, M. Cardiovascular disease and COVID-19. Diabetes & metabolic syndrome. Clin. Res. Rev.. 14 (3), 247–250 (2020).
Inciardi, R. M. et al. Cardiac involvement in a patient with coronavirus disease 2019 (COVID-19). JAMA Cardiol. 5 (7), 819–824 (2020).
Çınar, T., Hayıroğlu, M. İ., Çiçek, V., Uzun, M. & Orhan, A. L. COVID-19 and acute myocarditis: current literature review and diagnostic challenges. Revista Da Associação Médica Brasileira. 66, 48–54 (2020).
Magadum, A. & Kishore, R. Cardiovascular manifestations of COVID-19 infection. Cells 9 (11), 2508 (2020).
Siripanthong, B. et al. The pathogenesis and long-term consequences of COVID-19 cardiac injury. Basic. Translational Sci. 7 (3_Part_1), 294–308 (2022).
Qin, J-J. et al. Redefining cardiac biomarkers in predicting mortality of inpatients with COVID-19. Hypertension 76 (4), 1104–1112 (2020).
An, W., Kang, J-S., Wang, Q. & Kim, T-E. Cardiac biomarkers and COVID-19: A systematic review and meta-analysis. J. Infect. Public Health. 14 (9), 1191–1197 (2021).
Shafi, A. M., Shaikh, S. A., Shirke, M. M., Iddawela, S. & Harky, A. Cardiac manifestations in COVID-19 patients—A systematic review. J. Card. Surg. 35 (8), 1988–2008 (2020).
Mumtaz, A., Rehman, E., Rahaman, M. A. & Rehman, S. Inflammatory biomarkers and cardiac injury in COVID-19 patients. Front. Public. Health. 10, 1024535 (2022).
Çınar, T. et al. Is prognostic nutritional index a predictive marker for estimating all-cause in-hospital mortality in COVID-19 patients with cardiovascular risk factors? Heart Lung. 50 (2), 307–312 (2021).
Liaqat, A., Ali-Khan, R. S., Asad, M. & Rafique, Z. Evaluation of myocardial injury patterns and ST changes among critical and non-critical patients with coronavirus-19 disease. Sci. Rep. 11 (1), 4828 (2021).
Raja, W. et al. Cardiac involvement in patients suffering from covid-19 and its association with diseases severity and prognosis.
Guo, T. et al. Cardiovascular implications of fatal outcomes of patients with coronavirus disease 2019 (COVID-19). JAMA Cardiol. 5 (7), 811–818 (2020).
Mueller, C. et al. Cardiovascular biomarkers in patients with COVID-19. Eur. Heart J. Acute Cardiovasc. Care. 10 (3), 310–319 (2021).
Acknowledgements
We acknowledge the administrative support of staff and faculty of Sindh Infectious Diseases Hospital and Research center.
Author information
Authors and Affiliations
Contributions
Conception/design of the work: IA, UA, SA, FF Data collection: BS, UA, IA, NH, KF, MH, MAS, SA, SKZ, SF, MK, MUK, MM Data analysis and interpretation: BS, UA, IA, NH, KF, MH, MAS, SA, SKZ, SF, MK, MM, MUK Drafting the article: BS, UA, IA, NH, KF, MH, MAS, SA, SKZ, SF Critical revision of the article, and final approval: BS, UA, IA, NH, KF, MH, MAS, SA, SKZ, SF, MUK, MK, MM All authors have read and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approvval
The study was performed according to guidelines of declaration of Helsinki. Study was approved by Institutional Review Board (IRB) of Dow University of Health Sciences (IRB-2111/DUHS/Approval/2021/578). The data was collected retrospectively from the hospital record of admitted patients.
Informed consent statement
All participants were recruited in the study after written informed consent. Informed consent was obtained from all subjects and/or their legal guardians during admission in the hospital as per routine hospital policy.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Ahmed, I., Syed, B., Sayeed, M.A. et al. Comparison of risk factors and outcomes of raised vs normal cardiac troponin levels in COVID-19 patients: a retrospective study. Sci Rep 15, 33385 (2025). https://doi.org/10.1038/s41598-025-18397-w
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-18397-w