Abstract
Obstructive sleep apnea (OSA) is a heterogeneous disorder and the severe OSA could lead to cardiovascular and metabolic comorbidities. However, the OSA diagnosis focuses on the respiratory system according to guidelines. In this study, we aim to explore the clinical phenotypes of severe OSA based on comprehensive assessment of multiple organs function using cluster analysis. Patients with severe OSA were studied with data of age, sex, anthropometric examination, blood pressure, liver and renal function, lipid levels, fasting blood glucose, apnea index (AI), hyponea index (HI), apnea-hyponea index (AHI), lowest Sao2 during sleep-monitor, and disorders of nonalcoholic fatty liver disease (NAFLD) and carotid atherosclerosis (CAS). Cluster analysis was performed using k-medoids algorithm. A total of 503 subjects were clustered into two clusters: Cluster 1 (n = 136), middle-aged women; Cluster 2 (n = 367), middle-aged men. When compared with cluster 1, the cluster 2 showed an increased AI, AHI, BMI, neck and waist circumferences, fasting blood glucose, NAFLD and CAS prevalence, decreased liver and renal function, and decreased HI. The results suggest the phenotypes of severe OSA were identified especially based on multiple organ functions, creating the need to rethink these patients’ comprehensive assessment. The findings from our study suggest that for severe OSA patients, middle-aged men are more likely accompanied worse systematic condition than women, which should be taken into account by physicians.
Similar content being viewed by others
Introduction
Obstructive sleep apnea (OSA) is a sleep disorder characterized by recurrent episodes of collapse of the upper airway during sleep. It’s estimated that 936 million adults aged 30–69 years have mild to severe OSA, among which 425 million have moderate to severe OSA in the world1. The number of OSA patients was highest in China, followed by the USA, Brazil, and India in global1. Patients suffering severe OSA are more likely to develop cardiometabolic comorbidities2. In particular, severe OSAS was found to be a high-risk phenotype closely associated with severe oxygen desaturation, hypertension and cardiovascular diseases3. Thus, this study focused on severe OSA.
OSA is a heterogeneous condition with various predisposing factors, pathophysiological mechanisms, clinical manifestations and consequences of respiratory events4,5,6. Thus, it has not been formally characterized, which poses a challenge in early diagnosis. However, the OSA diagnosis focuses on the respiratory system and is based on apnea-hyponea index (AHI) according to guidelines7. It could not capture OSA patients’ heterogeneity that use AHI as the only measurement to diagnose OSA8,9. OSA phenotypes has been cluster analyzed in previous studies4,5,6. However, those previous studies were performed among patients with mild to severe OSA. Therefore, in this study, we aim to explore the clinical phenotypes of severe OSA based on multiple organs function using cluster analysis.
Results
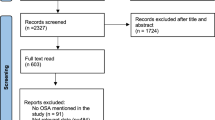
A total of 503 patients with severe OSA were included in the cluster analysis. Subjects for cluster analysis included 372 males (74%), and were overweight (26.8 ± 3.0 kg/m2) with an age of 52.2 ± 7.9 years. The AHI was 53.6 ± 15.6 according to AI (41.8 ± 17.0) and HI (11.8 ± 9.2). The multiple organ functions of patients with severe OSA were worse than those of health controls (Supplement file 1).
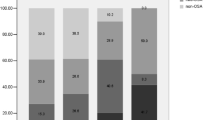
Two distinct clusters were identified. Both clusters included middle-aged subjects. The cluster 1 was composed of women (71.3%) in majority, whereas men compose 90.7% subjects in cluster 2. When compared with cluster 1, the subjects in cluster 2 showed increased BMI, neck circumference, waist circumference, SP, DP (Table 1; Fig. 1). For the AI and AHI, the cluster 2 were higher than cluster 1. However, the HI of subjects in cluster 2 were lower than that in cluster 1 (10.5 ± 7.2 vs. 15.5 ± 12.4 events/h).
Radar plot of severe obstructive sleep apnea patients’ clinical characteristics distribution. BMI body mass index. AI apnea index, HI hyponea index, AHI apnea-hyponea index, LSao2 lowest oxygen saturation in polysomnography, SP systolic blood pressure, DP diastolic blood pressure, UA uric acid, BUN blood urea nitrogen, Cr creatinine, AST aspartate transaminase, ALT alanine transaminase, TG triglycerides, CHOL cholesterol, LDLc low density lipoprotein cholesterol, HDLc high density lipoprotein cholesterol, FPG fasting blood glucose, γ-GT γ-Glutamyl transpeptidase, HCY homocysteine, CAS carotid atherosclerosis, NAFLD nonalcoholic fatty liver disease.
For the systematic assessment, in cluster 2, the levels of UA, BUN, Cr, AST, ALT, TG, CHOL, LDLc, FPG, γ-GT, HCY were all higher than those in cluster 1. In contrast, the level of HDLc in cluster 2 was lower than cluster 1. The prevalence of NAFLD (42.2% vs. 15.4%) and arteriosclerosis (39.8% vs. 16.2%) in cluster 2 were also higher than those in cluster 1 (Table 2; Fig. 1).
Discussions
This study explored possible phenotypes of severe OSA by cluster analysis. Our results showed that the two phenotypes can be identified in the severe OSA patients referred to the sleep laboratory, when they’re comprehensive assessed with demography, physical examination and multiple organ functions.
Our study showed that in severe OSA, one sub-phenotype was characterized as middle-aged women, and the other one was middle-aged men. The middle-aged men sub-phenotype showed worse performance in the comprehensive assessment than the middle-aged women sub-phenotype, including BMI, blood pressure, liver and renal function, blood lipids level, HCY, FPG, prevalence of CAS and NAFLD, AI and AHI. These findings show that subjects in middle-aged men sub-phenotype usually have worse systematic performance than those of middle-aged women sub-phenotype, in consistent with the severity of OSA based on AHI. These findings confirmed that systematic function is consistent with the severity of OSA based on AHI10,11,12. The higher prevalence of CAS and NAFLD in middle-aged men cluster in our study may be due to dyslipidaemia and apnea13,14. That also may be related with an increased neck circumference, which could be a predictor of cardiovascular disease in OSA patients15. Moreover, in our study, the middle-aged male patients with severe OSA were more serious than female patients at the similar age. The sex-stratified analysis shows that the differences in multiple organ functions between males and females was similar with those between Cluster 1 and Cluster 2 (Supplement file 2). That suggests the sub-phenotypes may be related with sex. The differences between men and women may be related with the alterations in the hypothalamus-pituitary axis and the altered secretion of hormones from the peripheral endocrine glands16. It would provide further understanding on it by testing hormone or endocrine in future study.
Furthermore, in our study, AHI and AI in middle-aged men sub-phenotype were higher than middle-aged women sub-phenotype, but the HI were lower. These findings suggest that for severe OSA patients, apnea should have more and worse influence on systematic function than hyponea. It’s consistent with an assumption that apnea, defined by complete cessation of airflow, should be more severe and worse than hypopnea, which had only a partial reduction in airflow. While both apnea and hypopnea involve disruptions in breathing during sleep, there’s different impact on sleep quality and daytime functioning between the two conditions17. Apnea tends to result in more severe sleep disruptions and symptoms such as loud snoring and gasping for air. Hypopnea, while still affecting sleep quality, may have a milder impact depending on the frequency and duration of the events. That may be related with the different impact on the organ functions. In addition, the lowest SaO2 during PSG was not significantly different in two phenotypes from our results. Those results indicate that an increased apnea may have an effect on the lowest SaO2, which was similar with an increased hyponea.
Clinical phenotypes of OSA has been explored in previous studies4,5,6. With different from those previous ones, our study focused on only severe OSA instead of mild to severe OSA. Second, our study focused on systematic assessment with multiple organ functions instead of Epworth Sleepiness Scale or PSG parameters. Thus, the clusters in our study showed different phenotypes from the previous ones. Our results were different from the previous study reported by Ye et al.4, in which the clusters reflected characteristics of symptom experiences and comorbidities. Moreover, our results were similar with that reported by Ferreira-Santos et al.6, in which the models showed middle-aged males cluster, middle-age females cluster and obese elderly males. However, our clusters showed distinct characteristics in multiple organs functions among subjects with severe OSA, whereas the previous one found characteristics in symptom and comorbidities among subjects with mild-to-severe OSA6. Thus, the phenotypes of severe OSA in our study focused on comprehensive assessment for patients.
In this study, the k-medoids algorism was performed in the cluster analysis. K-medoids is an unsupervised method with unlabeled data to be clustered18. It is an improvised version of the k-means algorithm mainly designed to deal with outlier data sensitivity. Compared to other partitioning algorithms, k-medoids algorithm is simple, fast, and easy to implement. It has been used in medical research for disease subtypes, and was identified as one of the most representative algorithm among various cluster algorithm19,20,21.
In conclusion, the phenotypes of severe OSA was identified especially based on multiple organ functions, creating the need to rethink these patients’ comprehensive assessment. The findings from our study suggest that for severe OSA patients, middle-aged men are more likely accompanied worse systematic condition than women, which should be taken into account by physicians.
Methods
Study population
From September 2021 to December 2023, adult patients with severe OSA diagnosed by pulmonologist were recruited from the Affiliated Hospital of Guilin Medical University. The subjects were excluded if he had accepted treatment for OSA, had other sleep apnea, had obstructive pulmonary disease, or had taken sedative or hypnotic medicine. The study protocol was approved by the institutional review board at the Affiliated Hospital of Guilin Medical University and conformed to the Declaration of Helsinki. Written informed consent was obtained from each subject.
Diagnostic criteria
The OSA was diagnosed according to American Academy of Sleep Medicine Clinical Practice Guideline6. OSA was diagnosed based on polysomnography (PSG) in hospital. The severe OSA was identified when apnea-hyponea index (AHI) was more than 30.
Carotid atherosclerosis (CAS) was screened for all subjects. CAS was determined if carotid intima-media thickness (CIMT) ≥ 1 mm with or without atherosclerotic plaque22. The CIMT was automatically measured on the far wall of the left common carotid artery 10 mm proximal to the carotid bifurcation at end-diastole22,23 using color Doppler ultrasound with a 3–12 MHZ probe (PHILIPS HD7, Netherlands) by exporters with at least 5 years’ experience.
Nonalcoholic fatty liver disease (NAFLD) was screened for all subjects as well. NAFLD was diagnosed by exporters with at least 5 years’ experience, when there was evidence of hepatic steatosis by color Doppler ultrasound with a 3–5 MHZ probe (ALOKA F75, Japan) and there was no history of significant alcohol consumption, use of steatogenic medication, viral hepatitis, or hereditary disorders24.
Cluster analysis
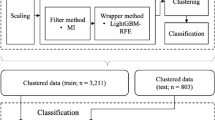
A total of 25 variables were incorporated into the cluster analysis: age, sex, CAS, NAFLD, body mass index (BMI), systolic blood pressure (SP), diastolic blood pressure (DP), uric acid (UA), blood urea nitrogen (BUN), creatinine (Cr), aspartate transaminase (AST), alanine transaminase (ALT), triglycerides (TG), cholesterol (CHOL), low density lipoprotein cholesterol (LDLc), high density lipoprotein cholesterol (HDLc), fasting blood glucose (FPG), γ-Glutamyl transpeptidase (γ-GT), homocysteine (HCY), neckline, waistline, apnea index (AI), hyponea index (HI), apnea-hyponea index (AHI), and lowest oxygen saturation (LSao2) during the PSG. All data were performed t-Distributed stochastic neighbor embedding (TSNE) method, and were standardized using z-score transformation. The k-medoids clustering was performed by using Python3.7.6 programming language (http://www.python.org), scikit-learn22.2 library (https://scikit-learn.org/stable/). The k value was selected by elbow method.
Statistical analysis
The Student’s t test or Mann–Whitney U test was used for comparing continuous data, and the Chi-square test was used for categorical variables. P-values < 0.05 were considered statistically significant. The statistical analyses were performed using SPSS 21.0 (IBM SPSS Inc).
Data availability
The dataset generated and/or analyzed during the current study are not publicly available but are available from the corresponding author and the first author on reasonable request.
References
Benjafield, A. V. et al. Estimation of the global prevalence and burden of obstructive sleep apnoea: A literature-based analysis. Lancet Respir Med. 7 (8), 687–698 (2019).
André, S. et al. Cardiometabolic comorbidities in obstructive sleep apnea patients are related to disease severity, nocturnal hypoxemia, and decreased sleep quality. Respir Res. 21 (1), 35 (2020).
Yeşildağ, M. & Duksal, F. Comorbidities and anthropometric parameters in obstructive sleep apnea syndrome: A phenotype-based study. Clin. Exp. Hypertens. 47 (1), 2512136 (2025).
Ye, L. et al. The different clinical faces of obstructive sleep apnoea: A cluster analysis. Eur. Respir J. 44 (6), 1600–1607 (2014).
Zhang, X. L. et al. Multidimensional assessment and cluster analysis for OSA phenotyping. J. Clin. Sleep. Med. 18 (7), 1779–1788 (2022).
Ferreira-Santos, D. & Rodrigues, P. P. Obstructive sleep apnea: A categorical cluster analysis and visualization. Pulmonology 29 (3), 207–213 (2023).
Kapur, V. K. et al. Clinical practice guideline for diagnostic testing for adult obstructive sleep apnea: An American academy of sleep medicine clinical practice guideline. J. Clin. Sleep. Med. 13 (3), 479–504 (2017).
Zinchuk, A. & Yaggi, H. K. Phenotypic subtypes of OSA: Aa challenge and opportunity for precision medicine. Chest 157 (2), 403–420 (2020).
Borsini, E., Nogueira, F. & Nigro, C. Apnea-hypopnea index in sleep studies and the risk of over-simplification. Sleep. Sci. 11 (1), 45–48 (2018).
Bajpai, J. et al. Prevalence of dyslipidaemia in OSA patients at a tertiary care center. Am. J. Cardiovasc. Dis. 13 (1), 1–9 (2023).
Hwu, D. W., Lin, K. D., Lin, K. C., Lee, Y. J. & Chang, Y. H. The association of obstructive sleep apnea and renal outcomes-a systematic review and meta-analysis. BMC Nephrol. 18 (1), 313 (2017).
Ji, Y., Liang, Y., Mak, J. C. W. & Ip, M. S. M. Obstructive sleep apnea, intermittent hypoxia and non-alcoholic fatty liver disease. Sleep. Med. 95, 16–28 (2022).
Drager, L. F., Polotsky, V. Y. & Lorenzi-Filho, G. Obstructive sleep apnea: an emerging risk factor for atherosclerosis. Chest 140 (2), 534–542 (2011).
Jin, Y. X. et al. Relationship between Obstructive sleep apnea and liver abnormalities in older patients: A cross-sectional study. Int. J. Clin. Pract. 9310588 (2023).
Yeşildağ, M., Şentürk, Z., Bekci, T. T. & Guney, İ. The usefulness of new body indices in determining the risk of cardiovascular disease in cases with obstructive sleep apnea syndrome. Int. J. Gen. Med. 17, 5523–5534 (2024).
Martins, F. O. & Conde, S. V. Gender differences in the context of obstructive sleep apnea and metabolic diseases. Front. Physiol. 12, 792633 (2021).
Mbata, G. & Chukwuka, J. Obstructive sleep apnea hypopnea syndrome. Ann. Med. Health Sci. Res. 2 (1), 74–77 (2012).
Kaufman, L. & Rousseeuw, P. J. -03-08), partitioning around medoids (Program PAM) (in en), Wiley Series in Probability and Statistics (Hoboken, NJ, USA: John Wiley & Sons, Inc.): 68–125, (1990). retrieved 2021-06-13.
Castaldi, P. J. et al. Do COPD subtypes really exist? COPD heterogeneity and clustering in 10 independent cohorts. Thorax 72 (11), 998–1006 (2017).
Michel, L. J. et al. Deep learning to estimate impaired glucose metabolism from magnetic resonance imaging of the liver: An opportunistic population screening approach. PLOS Digit. Health. 3 (1), e0000429 (2024).
Banerjee, A. et al. Identifying subtypes of heart failure from three electronic health record sources with machine learning: an external, prognostic, and genetic validation study. Lancet Digit. Health. 5 (6), e370–e379 (2023).
Chinese ultersound doctors association. Guidelines for vascular ultrasound examination. Chin. J. Ultrasonogr. 11, 993–1012 (2009).
Magnussen, C. G. et al. Use of B-mode ultrasound to examine preclinical markers of atherosclerosis: Image quality May bias associations between adiposity and measures of vascular structure and function. J. Ultrasound Med. 30 (3), 363–369 (2011).
Fan, J. G., Wei, L., Zhuang, H. National Workshop on Fatty Liver and Alcoholic Liver Disease, Chinese Society of Hepatology, Chinese Medical Association; Fatty Liver Disease Expert Committee, Chinese Medical Doctor Association. Guidelines of prevention and treatment of nonalcoholic fatty liver disease (2018, China). J. Dig. Dis. 20 (4), 163–173 (2019).
Acknowledgments
This work was supported by grants from the National Natural Science Foundation of China (No. 81460019), the Guangxi Natural Science Foundation (No. 2022JJA140014) in China.
Author information
Authors and Affiliations
Contributions
Yan Zhou, Jian Yu and Shuyuan Chu designed the study. Yan Zhou, Peng Li and Jimao Rong collected the data. Shuyuan Chu and Yan Zhou developed the models and wrote the manuscript. All authors contributed to the revision and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Zhou, Y., Yu, J., Li, P. et al. Severe obstructive sleep apnea phenotypes by cluster analysis based on multiple organs function. Sci Rep 15, 33709 (2025). https://doi.org/10.1038/s41598-025-19062-y
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-19062-y