Abstract
The myodural bridge (MDB) is an anatomical structure located in the suboccipital region, consisting of a dense fibrous connective tissue bridge connecting the suboccipital muscles to the dura mater (DM). These fibres interweave with their originating muscles and the dura, forming a functional unit known as the “myodural bridge complex” (MDBC). Previous studies, employing plastination sections, gross dissection, histological staining, and electron microscopy, have confirmed the widespread and highly conserved presence of the MDB in mammals, birds, and reptiles. To further investigate the presence of the MDB across vertebrates, this study selected the African clawed frog (Xenopus laevis), a model amphibian species, as the subject of investigation. Morphological methods, including gross dissection, paraffin section staining, and scanning electron microscopy (SEM), were used to investigate the existence and characteristics of the MDB in this species. A total of 10 sexually Mature African clawed frogs were used for the study: 3 for gross dissection, 5 for histological staining, and 2 for SEM observation. The study confirmed the presence of the MDBC in adult African clawed frogs, located in the atlanto-occipital space, where it connects the perimysium of the longissimus dorsi muscle (LGD) and the interarcualis muscle (IAR) to the dorsal atlanto-occipital membrane (DAOM) and DM. The characteristics of the MDB in adult African clawed frogs were as follows: Its fibrous components consisted predominantly of collagen fibres, with type I and type III collagen being the most abundant and elastic fibres being almost absent. The fibres between the perimysium, DAOM, and DM are interwoven. Considering the unique physiological structure and evolutionary significance of amphibians, our findings extend the known distribution of the MDB across vertebrates, enhance our understanding of adaptive changes in the MDBC among different species, and support the hypothesis that the MDBC plays a regulatory role in cerebrospinal fluid (CSF) dynamics.
Similar content being viewed by others
Introduction
In 1995, Hack et al.1 first described the myodural bridge (MDB) in the human suboccipital region, which they characterized as a dense fibrous connection between the rectus capitis posterior minor (RCPmi), the posterior atlantooccipital membrane (PAOM), and the cervical dura mater (DM). Subsequently, the MDB has been investigated more extensively from various perspectives using diverse experimental methods. Structurally, in addition to the RCPmi, the rectus capitis posterior major (RCPma), obliquus capitis inferior (OCI), and ligamentum flavum are also involved in forming the MDB. The dense fibres emanating from these structures connect either directly or indirectly to the DM, spanning the atlanto-occipital and atlanto-axial spaces. These fibres connect with each other and thus, along with the involved suboccipital musculature, form a functional unit termed the myodural bridge complex (MDBC)2,3. Considering that collagen fibres primarily provide tensile strength, the MDB, primarily composed of these fibres, may similarly possess corresponding functions4.
Comparative anatomical studies have shown that the MDB is a highly conserved anatomical structure across evolution. Zheng et al.5 found that the MDB is present in macaques, dogs, cats, rabbits, mice, guinea pigs, and finless porpoises, based on morphological studies of seven mammalian species from four different orders, suggesting its broad existence across various mammalian species. In addition, the MDB has been found in birds, such as rock pigeons6domestic chickens7penguins8and reptiles, such as turtles9crocodiles10,11and snake12suggesting its widespread presence and potential for important physiological functions.
Functionally, the MDB connects the dura mater to the muscle. However, the precise functions of the MDB remain unclear. Many scholars suggest that the MDB may play a role in preventing dural infolding and maintaining patency of the cerebellar medullary cistern and subarachnoid space13,14,15,16. MRI studies have shown that flexion, extension, and rotation movements of the head can influence cerebrospinal fluid (CSF) flow at the craniocervical junction17,18. Experiments involving suboccipital muscle stimulation in dogs have shown that suboccipital muscle stimulation increased CSF pressure, an effect that subsequently disappeared following MDB dissociation19. Studies using a rat model of suboccipital muscle hyperplasia showed that suboccipital muscle hyperplasia accompanied by increased muscle contractility led to elevated CSF pressure20. Based on these functional studies, our research group hypothesized that the MDB may be a potential driving force for CSF circulation. This hypothesis suggests that during suboccipital muscle contraction, the generated force is transmitted to the DM via the MDB. This transmission is proposed to pull the DM, altering local subarachnoid volume and generating a local negative pressure, thereby influencing local and potentially overall CSF circulation. CSF is an important extracellular fluid that plays a significant role not only in providing buoyancy, cushioning and shock absorption, and metabolic waste removal from the central nervous system (CNS), but also in maintaining CNS homeostasis. Furthermore, changes in CSF pressure are often associated with headache21,22. By comparing the RCPmi in patients with chronic headache and healthy volunteers, Yuan et al.23 suggested that hypertrophy of the RCPmi may be associated with chronic headache. Case reports have also indicated that chronic headache patients responded to MDB dissociation24. Therefore, it has been hypothesized that the MDB is not only a commonly found structure across multiple species but may also possess important physiological or pathological significance.
Understanding the developmental process of a tissue is often crucial for comprehending the mechanisms of its damage repair and the restoration of homeostasis. To further investigate the characteristics and functions of the MDB, and to explore its development and injury repair mechanisms, developmental anatomical studies are essential. Previous studies on MDB developmental anatomy in Sprague-Dawley (SD) rats have shown that the MDB begins to form around embryonic day 18 (E18) and strengthens postnatally, concurrent with increased head movements, with fibre properties gradually changing from a mixture of type I and type III to almost exclusively type I collagen fibres25,26.
The structural basis for the MDB involves the occiput, upper cervical vertebrae, the musculature of the suboccipital region, and the DM. Available studies indicate that the MDB is present in terrestrial tetrapods and even marine mammals. From an evolutionary perspective, amphibians are crucial in the vertebrate transition to land, providing a link between fish and terrestrial tetrapods. Skeletally, amphibians show the earliest differentiation of cervical vertebrae; extant anuran amphibians possess a single cervical vertebra, the atlas, concurrent with the differentiation of the DM from the brain/spinal membranes27,28. The presence of these prerequisite structures suggests that the MDB may exist in amphibians.
The African clawed frog (Xenopus laevis), as a model anuran amphibian, was selected as the subject for this study. Gross dissection, histological section staining, and scanning electron microscopy were utilized to investigate the morphology and characteristics of the MDB in anuran amphibians. Based on the findings of this study and previous comparative anatomical studies, the evolutionary significance of the MDB was discussed.
Materials and methods
Ethics statement
All procedures in this study were performed in strict accordance with the recommendations in the Guide for the Care and Use of Laboratory Animals published by the National Institutes of Health. The protocol was approved by the Committee on the Ethics of Animal Experiments of Dalian Medical University. All procedures involving animals were performed under MS-222 euthanasia, and efforts were made to minimize animal suffering. This study was carried out in compliance with the ARRIVE guidelines.
Animals
Sexually mature male African clawed frogs were purchased from the Clawed Frog Resource Centre and reared according to the standardized Clawed Frog breeding protocols at Cold Spring Harbor29,30. A total of 10 African clawed frogs were used for this study, allocated as follows: 3 for gross dissection, 5 for histological staining, and 2 for scanning electron microscopy.
Gross anatomy
Solutions of MS-222 for immersion at 5 g/L were prepared by mixing MS-222 powder with fresh water from the housing tank on the day of euthanasia. The solution was buffered with pharmaceutical-grade sodium bicarbonate to a pH of 7 to 7.5, and temperature was Maintained between 17 and 22 °C. All frogs were individually immersed in 2 L of solution for 1.5 h, across different days of experimental procedure. Following euthanasia, the frog was placed in a supine position. The chest was opened, the pericardium removed, and a needle inserted into the aortic root via the apex of the heart. The right ventricle was then incised, and perfusion with room temperature Ringer’s solution was performed to clear the vasculature of blood.
Once the perfusate exiting the right ventricle became clear, the thoracic and abdominal organs were removed, and the frog was positioned prone for subsequent fixation. Following skin removal, the abdominal wall and superficial back musculature were sharply dissected. The limb muscles were then dissected, the brachial and lumbar plexuses severed, and the limbs removed, leaving the head and trunk for subsequent dissection under a dissecting microscope.
The longissimus dorsi muscle (LGD) was severed from its attachment to the occipital bone, lifted dorsally to expose the atlanto-occipital joint, and observed for the connection between the muscle and the dura Mater. All images were captured using a Canon EOS 7D Mark II digital SLR camera (Canon Inc., Tokyo, Japan).
Histology and microscopy
Conventional perfusion and fixation were performed on adult African clawed frogs. Tissue blocks were subsequently decalcified using ethylenediaminetetraacetic acid disodium salt (EDTA-2Na). Thereafter, conventional paraffin embedding was performed, and 8-µm thick sections were obtained using a sliding microtome (Leica Micro HM450; Leica Microsystems GmbH, Wetzlar, Germany). For the detection of the MDB and the analysis of its characteristics, sections were stained with Masson, Picrosirius red, and Verhoeff stains using standard protocols. Masson and Verhoeff stained sections were imaged using a Nikon NIS imaging system (Nikon Eclipse 80i, Nikon, Tokyo, Japan), and Picrosirius red stained sections were observed under a polarizing microscope (Olympus BH-2; Olympus Corp., Tokyo, Japan).
Scanning electron microscopy
Samples for scanning electron microscopy (SEM) analysis were prepared by fixation in 2.5% glutaraldehyde solution, dehydration through a graded series of tert-butyl alcohol solutions, drying under vacuum, sputter coating with platinum, and visualized using a scanning electron microscope (Fei Quanta 200, FEI Company, Hillsboro, OR, USA).
Results
The presence of MDB in adult African clawed frog
Three of the 10 adult specimens were used for anatomical dissection to investigate the potential presence of the MDB structure in the suboccipital and intervertebral regions. Gross anatomical examination revealed that adult Xenopus possess only one cervical vertebra, the atlas (the first vertebra). The deep dorsal trunk muscles of Xenopus did not exhibit the distinct differentiation seen in higher vertebrates, such as spinous and semispinalis muscles, but largely comprised segmented LGD. The LGD originated from the transverse and spinous processes of the vertebrae and the exoccipital bone, and terminated at the anterior portion of the urostyle.
Exposure of the LGD revealed smaller muscles between adjacent vertebrae and between the exoccipital bone and the atlas, identified as the interarcualis muscle (IAR). Starting from the cranial attachment of the LGD, the muscle was carefully lifted dorsally to expose the exoccipital bone and then lifted caudally to expose the dorsal atlanto-occipital membrane (DAOM). Subsequently, the muscle was gently retracted to separate the DAOM from its bony attachment and the underlying DM.
During this separation, a cord-like structure was observed connecting the DAOM and DM, which was identified as the MDB in adult African clawed frogs (Fig. 1A-F). Continuing to lift the LGD caudally, the atlas and the remaining vertebrae were sequentially exposed. Following removal of the IAR from the deep aspect of the LGD, dense fibrous connections were observed between the adjacent vertebrae; however, no distinct membranous structures or cord-like connections similar to the MDB were identified.
Layer-by-layer gross anatomy of an adult Xenopus laevis and a schematic representation of the MDB. A-E. Results of the layer-by-layer gross anatomy of Xenopus laevis, and each scale is shown. F A schematic diagram of the gross anatomical results of the site of the muscle dural bridge in Xenopus laevis. TPL: temporalis muscle; LTD: latissimus dorsi muscle; ExOb: external oblique muscle; RBA: rhomboideus anterior muscle; DSC: dorsalis scapulae muscle; LGD: longissimus dorsi muscle; CGI: coccygeoiliacus muscle; EO: exoccipital bone; DM: dura mater. MDB: myodural bridge. The area marked by the red rectangle is the area of the MDB. The green dot line is where the DAOM (dorsal atlantooccipital membrane) is located.
Form of existence of MDB in adult African clawed frog
Histological staining was performed to further characterize the MDB in adult African clawed frogs. Masson trichrome staining was performed to observe the features of collagen fibers.
In sagittal sections, observations showed that the perimysium between muscles in the atlanto-occipital interspace extended ventrally at various angles, attaching to the DAOM and DM, thus forming the MDB in African clawed frogs (Fig. 2A1,A2). In transverse sections, the perimysium between the LGD and IAR thickened, forming a mesh-like structure, attaching ventrally to the dome-shaped DAOM and DM (Fig. 2B1,B2). Furthermore, these fascial bundles converged dorsally towards the midline, presenting a slightly ‘flame-like’ appearance. In coronal sections, due to the close apposition of the dome-shaped DAOM and DM, a complete view of the structure was challenging to obtain in a single section; however, these sections provided supplementary information regarding the three-dimensional features of the MDB meshwork in African clawed frogs (Fig. 2C1,C2).
The DAOM adhered to the DM, which showed continuity with the posterior choroid plexus (PCP). In sagittal sections, a large blood vessel was observed dorsal to the spinal cord and beneath the DM (Fig. 4A1). This vessel was also discernible during gross dissection, along with a major vessel located ventrally. This vessel connected to the PCP (Fig. 4A2). Furthermore, in some sections of the atlanto-occipital interspace, branches of this vessel were observed piercing the DAOM, suggesting potential vascular communication between intracranial and extracranial spaces (Fig. 4A1,A2).
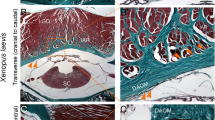
Results of Masson staining in each section of adult Xenopus laevis. A1 Whole slide image (WSI) of the sagittal section with Masson staining. A2 A local magnification of A1. B1 WSI of the transverse section result. B2 A local magnification of B1. C1 WSI of the coronal section result. C2 A local magnification of C1. Each scale is shown. LGD: longissimus dorsi muscle; IAR: interarcualis muscle; EO: exoccipital bone; V1: the first vertebra, the atlas; PCP: posterior choroid plexus; SC: spinal cord; Blue circle: DAOM (dorsal atlantooccipital membrane); Green circle: DM (dura mater); White pentagram: MDB (myodural bridge).
Collagen properties of MDB in adult African clawed frog
To further investigate the fibrous composition of the MDB, Picrosirius red staining was performed. Bright-field and polarized light microscopy views of the Picrosirius red stained sections were observed.
Results indicated that under bright-field microscopy, muscle fibers appeared yellow, while collagen fibers stained red-yellow (Fig. 3A1,A3,B1,B3,C1,C3). Under polarized light microscopy, the collagen fibers of the DM exhibited strong red-yellow birefringence, whereas the perimysium and the DAOM also displayed strong red-yellow birefringence but with a significant admixture of green birefringence (Fig. 3A2,A4,B2,B4,C2,C4). This indicates that in African clawed frogs, the DM is primarily composed of Type I collagen fibers, while the perimysium and the DAOM primarily composed of Type I and Type III collagen fibers. These interwoven collagen fibers, together with the IAR, the LGD, and the bony structures at the atlanto-occipital interspace, constitute the MDBC in African clawed frogs.
Results of Sirius red staining in adult Xenopus laevis. A0, B0, C0 The Masson staining results in the sagittal, transverse, and coronal section of Xenopus laevis, respectively, which are used for localization and comparison. A1, B1, C1. The bright field plots of Sirius red staining results corresponding to A0, B0, and A0, B0, respectively. A2, B2, C2. The dark field plots of Sirius red staining results corresponding to A0, B0 and C0, respectively. A3, B3, C3. Local enlargements corresponding to A1, B1 and C1, respectively. A4, B4, C4 Local enlargements corresponding to A2, B2, and C2, respectively. Each scale is shown. LGD: longissimus dorsi muscle; IAR: interarcualis muscle; EO: exoccipital bone; V1: the first vertebra, the atlas; PCP: posterior choroid plexus; SC: spinal cord; Blue circle: DAOM (dorsal atlantooccipital membrane); Green circle: DM (dura mater); Black pentacle and white pentacle: MDB (myodural bridge).
Rare elastic fibers were observed in MDB in adult African clawed frog
Given that amphibians represent the evolutionary stage where cervical vertebrae first appear, possessing only a single cervical vertebra, it is unknown whether differences exist in the composition of intervertebral connections between the atlanto-occipital space and other intervertebral spaces. Seeing that in mammals, the atlanto-occipital and atlanto-axial spaces are spanned by predominantly collagenous fibrous membranes, whereas other intervertebral spaces are spanned by the ligamentum flavum. On account of elastic fibers is hard to observe on Masson staining slides (Fig. 4A2,A3), Verhoeff staining, a specific stain for elastic fibers, was utilized to investigate whether a similar compositional difference exists in Xenopus laevis.
Verhoeff staining was performed on sagittal sections from adult African clawed frogs to compare the atlanto-occipital interspace with other vertebral interspaces, specifically comparing it to the 6th vertebral interspace, presented as a representative example in the figure. The staining results indicated that within the atlanto-occipital interspace, the MDB, DAOM, and DM were predominantly composed of collagen fibers, which stained red (Fig. 4B1,B3); whereas the fibrous connections at other intervertebral spaces contained a significant number of elastic fibers, appearing as black, wavy structures, in addition to collagen fibers, suggesting these represent the ligamentum flavum or a ligamentum flavum-like structure rich in elastic fibers (Fig. 4 B2,B4). Collectively, these findings suggest that African clawed frogs appear to have a differentiated ligamentum flavum structure, whereas its DAOM does not share the characteristics of the ligamentum flavum.
Masson and Verhoeff staining results of adult Xenopus laevis. A1 The results of local staining in the sagittal section of Xenopus laevis. A2 A local magnification of A1 at the red rectangle. A3 The results of Masson staining of the fifth intervertebral space of Xenopus. B1 The results of Verhoeff staining in the sagittal section of the atlas-occipital space of Xenopus laevis. B2 The results of Verhoeff staining in the sagittal section of the sixth intervertebral space of Xenopus laevis. B3, B4 Local enlargements of B1 and B2, respectively. Each scale is shown. LGD: longissimus dorsi muscle; IAR: interarcualis muscle; EO: exoccipital bone; V1: the first vertebra, the atlas; PCP: posterior choroid plexus; SC: spinal cord; DM: dura mater; LF: ligamentum flavum; V5: fifth vertebra; V6: sixth vertebra; V7: seventh vertebra. Green arrow: large blood vessels running in the dorsal, subdural space of the spinal cord. Red arrow: communicating branch vessels passing through the DAOM and the DM.
SEM results were consistent with histology results
Building upon the gross anatomical and histological findings, the MDB of African clawed frogs was further examined using SEM in the transverse and sagittal planes. Transverse sections clearly demonstrated that the perimysium was ventrally integrated into the DAOM (Fig. 5A1-A4). Sagittal sections revealed that strands of perimysium were ventrally connected to the DAOM, which exhibited a multilayered fiber structure (Fig. 5B1,B2). Dense fibers were also observed interwoven between the exposed DM and the DAOM. These results were consistent with the histological findings.
Scanning Electron microscopic findings of the atlantooccipital space in adult Xenopus laevis. A1-A4. SEM images of the cross section of the atlas occipital space in Xenopus laevis. B1, B2. The SEM images of the sagittal plane of the atlas occipital space in Xenopus laevis. Each scale is shown. LGD: longissimus dorsi muscle; IAR: interarcualis muscle; DAOM/white circle: DAOM (dorsal atlantooccipital membrane); SDM: spinal dura mater; PMS/white arrow: perimysium; Yellow arrow: fiber connections.
Discussion
The suboccipital region, particularly the craniocervical junction, is recognized as one of the most anatomically complex areas. Its anatomy is complex, characterized by intricate vascular and osseous structures, containing multiple pairs of short muscles and a high density of muscle spindles, which contribute significantly to proprioception14,31. Recent studies have established the MDB as a consistent anatomical structure within the suboccipital region. Its presence has been demonstrated not only in humans but also across a wide range of other vertebrates, including numerous mammals (such as marine mammals), reptiles (such as snakes, turtles, and crocodiles), and birds (such as rock pigeons, chickens, and penguins). This widespread occurrence suggests that the MDB may be a physiological structure prevalent among tetrapods6,7,8,9,10,11,12. However, amphibians, an important transition group for vertebrates from water to land, have not been included in these comparative anatomical studies. Therefore, this study selected the amphibian model organism Xenopus laevis as the subject of investigation to investigate the existence and characteristics of the MDB in amphibians.
MDB exists in atlanto-occipital space in xenopus
MDB in Xenopus exist in the form of junctions between the perimysium and the DAOM and DM
The deep anatomy of the atlanto-occipital space in Xenopus laevis was investigated using gross anatomy, histological staining, and SEM. The results confirmed the presence of the MDB in African clawed frogs, located in the atlanto-occipital space, where it forms a connection between the thickened perimysium of the LGD and IAR and the DAOM and DM.
A significant difference between fish and tetrapods lies in the organization of their intervertebral connections27. Studies in mammals have shown that the atlanto-occipital and atlanto-axial spaces are spanned by distinct membranous structures, namely the posterior atlanto-occipital membrane and the posterior atlanto-axial membrane, respectively, while the intervertebral spaces below the axis are predominantly composed of the ligamentum flavum32. Because the vertebral morphology and intervertebral joint movement in fish are relatively uniform, their intervertebral joints are relatively simple and uniform membranous connections. Given the musculoskeletal changes during metamorphosis from aquatic larva to terrestrial adult, we hypothesized that the composition of the atlanto-occipital connections in the adult frog might differ from that of other intervertebral spaces. Verhoeff staining is an elastic fiber-specific stain that is used to distinguish between predominantly collagenous structures and those rich in elastic fibers, such as the ligamentum flavum33. The staining results indicated that the atlanto-occipital space was spanned by a membranous structure predominantly composed of collagen fibers, while other intervertebral spaces were spanned by ligamentum flavum-like structures rich in elastic fibers (Fig. 4A2-B4). This observation aligned with our gross anatomical findings. Dissection readily revealed that the MDB connections in the atlanto-occipital interspace formed a broad, sheet-like membranous structure—significantly larger than those in other intervertebral spaces. In contrast, intervertebral connections outside this region presented as dense, ligamentous bands linearly arranged along the vertebral laminae, creating a stark morphological distinction from the MDB (supplementary figure). Besides, coronal and sagittal sections consistently demonstrated that while the perimysium in the atlanto-occipital interspace is markedly thickened and closely connected to the DAOM (a membranous structure), such adaptations were absent in other intervertebral spaces (Fig. 2A1,C1). Collectively, these differences—in surface area, muscular attachment robustness, and collagen/elastic fiber composition—established the atlanto-occipital interspace as the region of greatest mobility within the Xenopus’ spine. Moreover, the ligamentum flavum-like structures interconnecting the trunk vertebrae provided essential stabilization and mechanical support to the spinal column. This suggested that amphibians may have evolved an analogous connective tissue structure to the ligamentum flavum. Therefore, we conclude that the MDB structure specifically exists in the atlanto-occipital space of African clawed frogs, representing a clear departure from the uniform intervertebral junctions found in fish, resulting in functional and structural differentiation.
In other tetrapods studied to date, the MDB primarily exists as fibrous connective tissue bridges emanating from muscles and connecting directly or indirectly to the DM, a morphology significantly different from the features observed in Xenopus5,7,10. However, considering the overall structure of the suboccipital region, this difference can be attributed to the degree of differentiation of the epaxial musculature. Although the limb girdle bones and musculature exhibit clear differentiation associated with the emergence of limbs, the primary deep back muscle in Xenopus, the LGD, retains a highly segmented form characteristic of a low degree of differentiation, notably lacking the specialized suboccipital muscles seen in other tetrapods34. This indicates that the MDB in Xenopus, while representing a specialized structure, is clearly distinguishable from the ligamentum flavum, and its degree of differentiation aligns with evolutionary progression, suggesting the MDB is likely a highly conserved homologous structure across tetrapods.
The MDB of Xenopus is mainly composed of type I and Type III collagen, which may have a certain self-healing function
Masson staining of the MDB in African clawed frogs revealed that the MDB was predominantly composed of collagen fibers. Further analysis of collagen fiber properties using Picrosirius red staining indicated that the DM was primarily composed of Type I collagen fibers, while the perimysium and DAOM were primarily composed of Type I and Type III collagen fibers. These components were observed to be interwoven. These interwoven collagen fibers, together with the LGD, IAR, and the bony structures of the atlanto-occipital space, constitute the MDBC in African clawed frogs.
Recent studies have shown that the MDB is predominantly composed of Type I collagen fibers in adult Sprague-Dawley (SD) rats, but primarily composed of Type I and Type III collagen fibers in embryonic SD rats. Type III collagen is known to play an important role in regulating the formation of Type I collagen fibrils35which is consistent with observations regarding MDB development in embryonic mice25,26. In the adult individuals of most species we have examined, the myodural bridges (MDBs) are predominantly composed of type I collagen fibers4,6,7,25,36. We have yet to determine the mechanisms underlying: (a) interspecies divergence in MDB collagen composition, and (b) developmental variations between embryonic and adult stages within same species. Nevertheless, these coould be recognized as promising directions for our future research.
The MDB of xenopus May be involved in regulating cerebrospinal fluid circulation in multiple ways
Analysis of serial sections through the head and trunk of African clawed frogs revealed that the MDB, DAOM, DM, and PCP were sequentially connected. The PCP was connected to a large longitudinal blood vessel, identified as the dorsal spinal vein (DSV), located dorsal to the spinal cord and extending throughout the spinal canal. This vein bifurcates at the caudal tip of the PCP, forming oblique occipital veins (OOC), while the PCP itself was drained by several choroidal veins into the OOC.
Based on extensive observations of the MDB in mammals, our research group has previously hypothesized that the MDB may act as a driving force for CSF circulation17,18. This hypothesis suggests that during suboccipital muscle contraction, the force generated is transmitted via the MDB to the DM, thereby pulling the DM and altering the local subarachnoid space volume, resulting in local negative pressure that influences local and potentially overall CSF circulation19,20. In African clawed frogs, the mesh-like MDB anchored to the DAOM and DM could potentially influence CSF circulation by, on one hand, altering local pressure through tension on the DM. On the other hand, in conjunction with the suggested regulatory role of intracranial blood flow by previous studiesthe observed vascular communication branches traversing the DAOM and the presence of large vessels in the subdural space in African clawed frogs suggest that changes in MDBC tension resulting from muscle contraction may also affect CSF dynamics by altering blood flow within the cranial cavity and spinal canal. These vascular connections may enable the MDBC in African clawed frogs to regulate CSF in a more direct and responsive manner. Although observations of this communicating branch were limited by the nature of the sectioning technique and sample size, this finding supports the speculation that such a regulatory mechanism exists.
As an important transitional stage in vertebrate evolution towards terrestrial life, amphibians exhibit unique morphological adaptations. Extant anuran amphibians uniformly possess only a single cervical vertebra, which results in relatively limited neck mobility compared to other quadrupeds. Despite possessing four limbs, their locomotion patterns are relatively simple, with minimal spinal mobility. The process of metamorphosis necessitates rapid adaptation to the terrestrial environment. These unique anatomical and functional features suggest that their CSF dynamics may be regulated in a manner significantly different from that of other tetrapods, and the control exerted by the MDBC on CSF may similarly manifest in varied forms. To investigate the CSF dynamic patterns in Xenopus laevis and their interrelationship with the MDBC, we have conducted further experimental studies, the results of which will be published in the near future.
Evolutionary significance of research on the development of MDB in xenopus
From an evolutionary perspective, amphibians occupy a unique and crucial position, representing the vital transition from aquatic to terrestrial vertebrates. During this transition, overcoming the effects of gravity and the loss of aquatic buoyancy required adaptations, leading to the evolution of a more rigid axial skeleton and the development of limbs capable of lifting the trunk off the ground. Consequently, the separation of the head from the trunk, allowing independent head movement from the forelimbs, became necessary, leading to the appearance of cervical vertebrae37.
Following the transition to a terrestrial environment, gravity also significantly impacts the central nervous system. Cerebrospinal fluid (CSF) plays a crucial role in providing buoyancy to reduce the effective weight of the brain and spinal cord, thus protecting them from compression and impact. Research indicates that in fish, the meninges are relatively simple, consisting primarily of the meninx primitiva. In contrast, amphibians, reptiles, and birds possess a differentiated dura mater (DM) and leptomeninx27. Mammals exhibit further differentiation, possessing the DM, arachnoid mater, and pia mater. This meningeal differentiation facilitates a more organized containment and distribution of CSF, thereby enhancing the buoyancy-based support of the central nervous system.
As transitional organisms from aquatic to terrestrial environments, amphibians possess only a single cervical vertebra and exhibit a relatively low degree of epaxial muscle differentiation38. Their head movement is consequently relatively simple, with limited range of motion. The robust, mesh-like structure of the MDB in amphibians likely contributes to head stabilization. Furthermore, tension changes within the MDBC are hypothesized to regulate CSF dynamics by locally displacing the DM and influencing blood flow within the cranial and vertebral cavities.
Previous studies have documented the existence of the MDB across mammals, birds, and reptiles, revealing variations in its morphology. For example, the basic MDB morphology in birds, reptiles, and mammals is similar, characterized by dense fibrous connective tissue emanating from suboccipital muscles and connecting to the DM via the atlanto-occipital and atlanto-axial interspaces. However, further observations have revealed variations. In marine mammals, the MDB is notably robust, likely an adaptation related to their large body size and deep diving behavior. In some species, such as the narrow-ridged finless porpoise, an “Occipital-Dural Muscle” has been described, directly terminating at the DM; the termination site of this muscle is considered analogous to the MDB structure39. This morphology may be related to the demands of maintaining CSF circulation during deep dives. Similarly, in diving seabirds like penguins, the MDB exhibits differences compared to non-diving birds such as rock pigeons and chickens. Considering the interspecies structural variations at the craniocervical junction, it is evident that the MDB is widely present in tetrapods, with its structure and function adapting accordingly, demonstrating various adaptive changes across species.
As previously mentioned, compared to mammals, African clawed frogs are relatively primitive, exhibiting restricted differentiation of their deep dorsal muscles and limited head and neck range of motion. Therefore, maintaining CSF dynamics in this species may necessitate a more direct regulatory mechanism to facilitate rapid adjustments. In contrast, mammals, with their highly flexible necks and greater mobility of the cervical spine, experience significant modulation of CSF dynamics through spinal movement. Consequently, while vascular communication exists in mammals, the direct regulatory role via tension on these vessels may be relatively less pronounced. With evolutionary progression, the fundamental regulatory role of the MDBC has persisted; however, it has evolved to modulate CSF dynamics in a more indirect manner, coordinated with head and neck movements, reflecting the increasing differentiation of suboccipital muscles and the greater number of cervical vertebrae. These observations suggest that the MDBC’s regulatory function on CSF dynamics is relatively independent of overall spinal mobility. Rather than being lost during evolution, this function has evolved in parallel with the MDBC’s evolutionary and adaptive changes.
Summary and prospect
This study provides the first confirmation of the existence of the MDB in amphibians, using the model organism, the African clawed frog, as the subject of investigation. It described its unique morphology and offered insights into its potential function. These findings fill the gap in the understanding of the MDB’s presence in amphibians, completing the documentation of its presence across the major tetrapod lineages. By comparing findings in amphibians with those from other tetrapods and integrating structural observations with functional hypotheses, this study also discusses the diversity of the MDB’s morphology and potential function across vertebrates. The MDB is a structurally consistent component that, while not classified as a distinct organ, exhibits varied characteristics depending on the specific anatomy of the craniocervical junction in tetrapods.
The presence of the MDB across diverse tetrapods, including amphibians, highlights its evolutionary significance as a conserved structure. The transition of vertebrates from aquatic to terrestrial life involved numerous anatomical and physiological adaptations to overcome novel environmental challenges, such as gravity and changes in musculoskeletal demands. Amphibians, particularly those undergoing metamorphosis like the African clawed frog, serve as a crucial model for studying this transition, as their development from an aquatic tadpole to a terrestrial adult recapitulates key evolutionary changes. These adaptations were likely driven by accumulated genetic changes, some of which may have initially been neutral but acquired significance under new selective pressures.
Therefore, our research group will further investigate the developmental process in the African clawed frog, analyzing the morphological and molecular characteristics of the MDB during this metamorphosis. Additionally, the transparent nature of the African clawed frog tadpole facilitates in vivo visualization of CSF, enabling tracking analysis using fluorescent particles or other indicators under microscopy. This approach will provide more direct evidence of the MDB’s impact on CSF dynamics, moving beyond speculation. Future in-depth studies of the MDB in the African clawed frog aim to enhance our understanding of its molecular regulatory mechanisms and functions, and revealing its unique characteristics and evolutionary significance.
Data availability
The datasets used and/or analysed during the current study would be available from the corresponding author on reasonable request.
References
Hack, G. D., Koritzer, R. T., Robinson, W. L., Hallgren, R. C. & Greenman, P. E. Anatomic relation between the rectus capitis posterior minor muscle and the dura mater. Spine 20, 2484–2486. https://doi.org/10.1097/00007632-199512000-00003 (1995).
Sui, H. J. et al. Anatomical study on the connections between the suboccipital structures and the spinal dura mater. Chin. J. Clin. Anat. 31, 489–490. https://doi.org/10.13418/j.issn.1001-165x.2013.04.011 (2013).
Zheng, N. et al. The myodural Bridge complex defined as a new functional structure. Surg. Radiologic Anatomy: SRA. 42, 143–153. https://doi.org/10.1007/s00276-019-02340-6 (2020).
Zheng, N. et al. Orientation and property of fibers of the myodural Bridge in humans. Spine J.: Offic. J. North. Am. Spine Soc. 18, 1081–1087. https://doi.org/10.1016/j.spinee.2018.02.006 (2018).
Zheng, N. et al. The universal existence of myodural Bridge in mammals: an indication of a necessary function. Sci. Rep. 7, 8248. https://doi.org/10.1038/s41598-017-06863-z (2017).
Okoye, C. S., Zheng, N., Yu, S. B. & Sui, H. J. The myodural Bridge in the common rock pigeon (Columbia livia): morphology and possible physiological implications. J. Morphol. 279, 1524–1531. https://doi.org/10.1002/jmor.20890 (2018).
Dou, Y. R. et al. Existence and features of the myodural Bridge in Gallus domesticus: indication of its important physiological function. Anat. Sci. Int. 94, 184–191. https://doi.org/10.1007/s12565-018-00470-2 (2019).
Chen, C. et al. Existence and features of the myodural Bridge in Gentoo penguins: A morphological study. PLoS One. 16, e0244774. https://doi.org/10.1371/journal.pone.0244774 (2021).
Huangfu, Z. et al. Existence of myodural Bridge in the trachemys scripta elegans: indication of its important physiological function. Int. J. Morphology. 37 https://doi.org/10.4067/s0717-95022019000401353 (2019).
Zhang, J. H. et al. Connection of the posterior occipital muscle and dura mater of the Siamese crocodile. Anat. Rec. 299, 1402–14081932. https://doi.org/10.1002/ar.23445 (2016).
Young, B. A. et al. Variations in the cerebrospinal fluid dynamics of the American alligator (Alligator mississippiensis). Fluids Barriers CNS. 18, 1–15. https://doi.org/10.1186/s12987-021-00248-1 (2021).
Li, C. et al. Identification of the myodural Bridge in a venomous snake, the gloydius shedaoensis: what is the functional significance?? Int. J. Morphology. 40 https://doi.org/10.4067/s0717-95022022000200304 (2022).
Peck, D., Buxton, D. F. & Nitz, A. A comparison of spindle concentrations in large and small muscles acting in parallel combinations. J. Morphol. 180, 243–252. https://doi.org/10.1002/jmor.1051800307 (1984).
Kulkarni, V., Chandy, M. J. & Babu, K. S. Quantitative study of muscle spindles in suboccipital muscles of human foetuses. Neurol. India. 49, 355–359 (2001).
Scali, F., Pontell, M. E., Enix, D. E. & Marshall, E. Histological analysis of the rectus capitis posterior major’s myodural Bridge. Spine J.: Offic. J. North. Am. Spine Soc. 13, 558–563. https://doi.org/10.1016/j.spinee.2013.01.015 (2013).
Enix, D. E., Scali, F. & Pontell, M. E. The cervical myodural bridge, a review of literature and clinical implications. J. Can. Chiropr. Assoc. 58, 184–192 (2014).
Xu, Q. et al. Head movement, an important contributor to human cerebrospinal fluid circulation. Sci. Rep. 6, 31787. https://doi.org/10.1038/srep31787 (2016).
Xu, Q. et al. Head-nodding: a driving force for the circulation of cerebrospinal fluid. Sci. Rep. 11, 14233. https://doi.org/10.1038/s41598-021-93767-8 (2021).
Ma, Y. et al. The morphology, biomechanics, and physiological function of the suboccipital myodural connections. Sci. Rep. 11, 8064. https://doi.org/10.1038/s41598-021-86934-4 (2021).
Li, C. et al. The relationship between myodural bridges, hyperplasia of the suboccipital musculature, and intracranial pressure. PLoS One. 17, e0273193. https://doi.org/10.1371/journal.pone.0273193 (2022).
Sakka, L., Coll, G. & Chazal, J. Anatomy and physiology of cerebrospinal fluid. Eur. Ann. Otorhinolaryngol. Head Neck Dis. 128, 309–316. https://doi.org/10.1016/j.anorl.2011.03.002 (2011).
Bothwell, S. W., Janigro, D. & Patabendige, A. Cerebrospinal fluid dynamics and intracranial pressure elevation in neurological diseases. Fluids Barriers CNS. 16 https://doi.org/10.1186/s12987-019-0129-6 (2019).
Yuan, X. Y. et al. Correlation between chronic headaches and the rectus capitis posterior minor muscle: A comparative analysis of cross-sectional trail. Cephalalgia 37, 1051–1056. https://doi.org/10.1177/0333102416664775 (2017).
Hack, G. D. & Hallgren, R. C. Chronic headache relief after section of suboccipital muscle dural connections: a case report. Headache 44, 84–89. https://doi.org/10.1111/j.1526-4610.2004.04015.x (2004).
Lai, H. X. et al. Development of myodural Bridge located within the atlanto-occipital interspace of rats. Anat. Rec (Hoboken). 304, 1541–1550. https://doi.org/10.1002/ar.24568 (2021).
Lai, H. X. et al. Development, maturation, and growth of the myodural Bridge within the posterior atlanto-axial interspace in the rat. J. Morphol. 283, 993–1002. https://doi.org/10.1002/jmor.21467 (2022).
Yang, A., Cheng, H. & Yao, J. Comparative Vertebrate Anatomy 2nd edn (Peking University, 2008).
Fei, L. et al. Fauna Sinica, Amphibian (Science, 2009).
McNamara, S., Wlizla, M., Horb, M. E. & Husbandry General care, and transportation of xenopus laevis and xenopus tropicalis. Methods Mol. Biol. 1865, 1–17. https://doi.org/10.1007/978-1-4939-8784-9_1 (2018).
Ishibashi, S. & Amaya, E. How to grow xenopus laevis tadpole stages to adult. Cold Spring Harb Protoc. 2021 https://doi.org/10.1101/pdb.prot106245 (2021).
Yu, X. The Craniovertebral Junction Abnormalities—Fundamentals and Surgical Treatment (People’s Military Medical, 2015).
Susan, S. Gray’s Anatomy: the Anatomical Basis of Clinical Practice (Elsevier, 2015).
Piccinin, M. A. & Schwartz, J. in StatPearlsStatPearls Publishing, (2025).
Ryke, P. A. J. The ontogenetic development of the somatic musculature of the trunk of the aglossal Anuran xenopus laevis (Daudin). Acta Zool. 34, 1–700001. https://doi.org/10.1111/j.1463-6395.1953.tb00367.x (1953).
Niederreither, K. et al. Coordinate patterns of expression of type I and III collagens during mouse development. Matrix Biol. 14, 705–713. https://doi.org/10.1016/s0945-053x(05)80013-7 (1995).
Liu, P. et al. The myodural Bridge existing in the nephocaena phocaenoides. PLoS One. 12, e0173630. https://doi.org/10.1371/journal.pone.0173630 (2017).
Camp, A. L. J. I. & Biology, C. What fish can teach Us about the feeding functions of postcranial muscles and joints. 59, 383–393 (2019).
McGowan, G. The development and function of the atlanto-axial joint in albanerpetontid amphibians. J. J. J. O H. 32, 116–122 (1998).
Zhang, Z. X. et al. A specialized myodural Bridge named occipital-dural muscle in the narrow-ridged finless porpoise (Neophocaena asiaeorientalis). Sci. Rep. 11, 15485. https://doi.org/10.1038/s41598-021-95070-y (2021).
Acknowledgements
The authors would like to thank Miss Bei-Bei Sun for her help with the schematic diagram of the anatomical pattern.
Funding
This work was supported by Natural Science Foundation of China (NSFC32471192); Innovation Support Project for Science and Technology Talent of Dalian (2023RG003); Interdisciplinary Project of Dalian University (DLUXK-2024-QN-015). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Author information
Authors and Affiliations
Contributions
Heng Yang: Methodology, Formal analysis, Investigation, Writing-original draft, Writing-review & editing, Visualization.Xue Song: Methodology, Formal analysis, Investigation.Chen Yue: Data curation, Formal analysis, Funding acquisition.Jin Gong: Methodology.Wei Ma: Methodology, Investigation.Ruo-Tong Zhang: Methodology, Investigation.Jing-Xian Sun: Methodology.Tai-Lai Li: Visualization.Campbell Gilmore: Writing-review & editing.Xu-Hui Zhang: Investigation, Methodology.Jing Zhang: Investigation, Methodology.Yang Song: Investigation.Sheng-Bo Yu: Supervision.Cong-Yi Li: Investigation.Wei Tang: Methodology.Chan Li: Methodology, Formal analysis, Investigation, Resources, Data curation, Writing-review & editing, Funding acquisition.Hong-Jin Sui: Conceptualization, Validation, Resources, Writing-review & editing, Supervision, Project administration, Funding acquisition.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Yang, H., Song, X., Yue, C. et al. Identification, structural features, and potential functional significance of the myodural Bridge in African clawed frog, the Xenopus laevis. Sci Rep 15, 35144 (2025). https://doi.org/10.1038/s41598-025-19100-9
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-19100-9