Abstract
Post spinal-fusion surgery pain levels are high, posing challenges for pain management. We introduced around-the-clock (ATC) administration of intravenous acetaminophen in the clinical pathway for spinal-fusion surgery. This single-center, retrospective observational study investigated effects of its introduction on postoperative pain levels and recovery adjusting for background variables. Adult patients who underwent elective spinal-fusion surgery were categorized into pre- and post-groups based on the timing of ATC acetaminophen introduction. The primary endpoint was the numerical rating scale (NRS) score at rest in the morning of the first postoperative day (POD). Secondary endpoints were NRS scores on POD1-3, rescue analgesic use, postoperative nausea and vomiting within 48 h, food intake, ambulation, and hospitalization length. The pre- and post-groups included 31 and 47 patients, respectively. The estimated means of NRS scores on POD1 were significantly lower in the post-group (1.21) than in the pre-group (3.91) (P < 0.0001). NRS scores until POD3 and number of rescue analgesics used were significantly lower in the post-group. There were no significant differences in the postoperative recovery endpoints, barring a trend toward shorter hospitalization in the post-group. In elective spinal-fusion surgery, the introduction of ATC intravenous acetaminophen into the clinical pathway improved postoperative analgesia.
Similar content being viewed by others
Introduction
In Japan, intravenous acetaminophen (acelio®, Terumo Corporation, Tokyo) has received marketing approval since 2013. Various postoperative analgesia guidelines1,2,3,4 have positioned this agent for multimodal analgesia (MMA) and around-the-clock (ATC) administration. Specific recommendations for ATC include the administration of coxib drugs, non-steroidal anti-inflammatory drugs (NSAIDs), and acetaminophen unless contraindicated. However, ATC administration has not fully permeated the current state of postoperative pain management in Japan.
Pain management is challenging in surgeries with high levels of postoperative pain. Spinal-fusion surgery, in particular, has a high level of pain5, and we found that pain management for this surgery was inadequate at our institution. However, although guidelines for pain management after spinal surgery exist, there are few reports describing specific pain management menus and their effectiveness in spinal-fusion surgery. We have implemented ATC intravenous acetaminophen in the clinical pathway for gynecological and gastrointestinal surgery at our institution and have realized its usefulness6. Therefore, we have included ATC intravenous acetaminophen in the analgesic pathway for spinal surgery.
Japan is a super-aging society; consequently, the number of surgeries, particularly spinal surgeries, among the elderly is increasing. With the increasing emphasis on maintaining mobility, QOL, and activity levels in the elderly population, the numbers of spinal-fusion surgeries, such as posterior lumbar interbody fusion (PLIF) and transforaminal lumbar interbody fusion (TLIF), are expected to increase further7. As mentioned previously, spinal-fusion surgeries are associated with high postoperative pain levels5. According to Radnovich et al.., poor pain control after surgery hinders postoperative rehabilitation, leads to a decline in the patient’s health-related QOL, imposes a significant personal burden, and contributes to increased national healthcare costs8. Furthermore, according to Fletcher et al.., inadequate acute postoperative pain control induces chronic pain, suggesting an association with significant postoperative cognitive impairment9. Therefore, improving acute postoperative pain management is meaningful in the context of spinal surgery.
Herein, we conducted a retrospective study of the effects of the introduction of intravenous acetaminophen in the analgesic pathway for spinal-fusion surgery in November 2018 on postoperative pain and postoperative recovery. Our institution has a patient-support center, and standard procedures and postoperative management have been consistently implemented. The primary endpoint was the postoperative Numerical Rating Scale (NRS) score. Furthermore, we investigated other indicators of postoperative recovery, including food intake, ambulation, and length of hospital stay.
Methods
The present study was approved by the Institutional Review Board of Saiseikai Yokohamashi Tobu Hospital (identifier number: 20220138), was registered with University Hospital Medical Information Network (UMIN) Clinical Trials Registry (identifier number: UMIN000051149) and was conducted after disclosure of information. All methods were performed in accordance with relevant guidelines and regulations. Owing to the retrospective nature of the study, the need to obtain the informed consent was waived by the Institutional Review Board of Saiseikai Yokohamashi Tobu Hospital. The research content was posted in the outpatient department of Saiseikai Yokohamashi Tobu Hospital after approval. Eligible patients were provided with information about the study and an opportunity to decline participation. Data from patients who ultimately did not decline were used for analysis.
This was a retrospective observational study adjusted for background factors, including patients who underwent elective spinal-fusion surgery, specifically PLIF or TLIF, with a fusion segment number of 1 (mono-segment). PLIF and TLIF are spinal-fusion procedures that are performed for degenerative spinal disease such as lumbar spinal stenosis. The inclusion criteria were as follows: ① age ≥ 20 years (no upper limit), ② any sex, ③ American Society of Anesthesiologists physical status (ASA-PS) of I or II, ④ undergoing elective surgery, ⑤ attendance in our patient-support center before undergoing surgery, and ⑥ receiving intravenous acetaminophen four times, including during the surgery, in the group receiving ATC acetaminophen administration (Post-group). The exclusion criteria were as follows: ① clear elevation of liver enzyme levels from preoperative levels (aspartate aminotransferase or alanine aminotransferase ≥ 100 U/L); ② presence of concurrent alcoholic liver disease; ③ pain and PONV assessment not possible; ④ anterior fusion surgery or cases where both anterior and posterior fusions were used in multiple vertebrae; ⑤ development of neurological complications during or after surgery, such as dural tears; and ⑥ presence of metastasis or malignant tumors.
Eligible patients were admitted the day before surgery, did not receive preanesthetic medication, and underwent spinal-fusion surgery under general anesthesia. The surgery was performed with the patient in the prone position using a conventional open approach, with the placement of pedicle screws and bone grafting into the intervertebral space. The surgeons ranged from residents to board-certified spine specialists. General anesthesia was administered via inhalation anesthesia [desflurane (DES) or sevoflurane (SEV)], and fentanyl and remifentanil were administered intraoperatively. The endotracheal tube was removed after patients awakened from anesthesia; patients were managed in the intensive care unit for the first 24 h postoperatively. Postoperative plans for mobilization, meals, pain management, and discharge schedules were implemented according to the clinical pathway. Deviation from the clinical pathway was allowed based on the physician’s judgment in cases of a deteriorating overall condition, poor pain or PONV control, occurrence of postoperative complications, or patient preference. The dosage of intravenous acetaminophen administered during and after surgery was determined as follows. For patients weighing ≥ 50 kg, acetaminophen was intravenously administered at 1,000 mg over 15 min, with an interval of 6 h between administrations and a maximum daily dose of 4,000 mg. For patients weighing < 50 kg, acetaminophen was intravenously administered at 15 mg/kg over 15 min, with an interval of 6 h between administrations and a maximum daily dose of 60 mg/kg.
In this study, to investigate the effectiveness of ATC intravenous acetaminophen administration for elective spinal-fusion surgery, patients were categorized into a pre-introduction group (Pre-group) and a post-introduction group (Post-group), based on the introduction of ATC acetaminophen administration into the clinical pathway, and an observational study was conducted. In our hospital, ATC intravenous acetaminophen administration was introduced into the clinical pathway for elective spinal-fusion surgery, as a postoperative analgesic method, on November 28, 2018. For the comparison before and after this introduction, this study defined a transition period of 2 months and set the study period accordingly. Consequently, the study period for each group was set as follows: Pre-group, for 2 years retroactively from October 28, 2018; Post-group, for 3 years from January 1, 2019. To ensure uniformity in perioperative management and data collection, the Pre-group was retrospectively defined until the patient support center became operational. Information on eligible cases during each period was collected from the medical records (HOPE EGMAIN-GX, Fujitsu Limited, Japan).
Postoperative pain management and intraoperative postoperative nausea and vomiting (PONV) prevention protocols
Postoperative pain management protocol
Protocol common to both groups
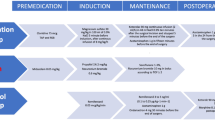
An anesthesiologist intravenously administered acetaminophen 30 min prior to the end of surgery. Local infiltration anesthesia with 1% lidocaine (15 mL) was administered to the surgical field during wound closure. A continuous intravenous infusion of fentanyl at a rate of 20 µg/h was initiated immediately after surgery and discontinued after 50 h postoperatively.
Pre-group and post-group protocol
From the day post-surgery, scheduled prescription of oral acetaminophen was initiated. Rescue medication included the administration of NSAIDs.
Pre-group: On the day of surgery, flurbiprofen axetil injection or intravenous acetaminophen, diclofenac sodium suppositories, and tramadol injection were administered as needed for pain.
Post-group: Postoperatively, intravenous acetaminophen was administered every 6 h, with three administrations (at 16:00, 22:00 on the day of surgery, and 4:00 on the day after surgery). On the day of surgery, flurbiprofen axetil injection, diclofenac sodium suppositories, and tramadol injection were administered as needed for pain. The only change in the protocol between the Pre- and Post-groups was the introduction of the ATC intravenous acetaminophen administration.
Intraoperative PONV prevention protocol
The attending anesthesiologist confirmed the following risk factors for PONV proposed by Apfel et al.10 before surgery: ① nonsmoking, ② female sex, ③ history of motion sickness (sea or car sickness), and ④ postoperative opioid use. If three or more risk factors were present, the following PONV prevention measures were implemented:
-
Dexamethasone (6.6 mg) intravenous administration at the induction of anesthesia.
-
Droperidol (0.02 mL/kg, equivalent to 0.05 mg/kg of droperidol) intravenous administration at the end of surgery.
The primary endpoint of this study was the NRS (rated on an 11-point scale from 0 to 10) score at rest at 6:00 am on the first postoperative day (points). The NRS assessment at rest was conducted by multiple ward nurses who were trained for NRS assessment. The patients received education about the NRS during the preoperative outpatient visit, and they were consequently able to report their NRS values independently. Measurements were obtained at three time points each day: 6:00 am, 12:00 pm, and 6:00 pm. The secondary endpoints were the NRS score at rest from the maximum score on the day of surgery (points) to 6:00 pm on the third postoperative day (points), frequency of rescue analgesic use until the third postoperative day (times), frequency of PONV episodes within 48 h postoperatively (times), amount of food intake at the start of meals (points), score at the start of ambulation (points), and postoperative length of hospital stay (days). Furthermore, demographic information, physical measurements, and laboratory tests were collected for all participants, including sex, age, weight, height, serum hemoglobin level, ASA-PS, anesthesia used during surgery, and operation type.
For each secondary endpoint item other than the NRS scores, the assessment was conducted described in the following sections.
Frequency of rescue analgesic use until postoperative day 3 (times)
Rescue analgesics were administered at the discretion of the ward nurses when additional analgesics were considered necessary. Both before and after the protocol change, rescue medication was administered according to the clinical pathway when the resting NRS score was ≥ 4. The drugs administered were preregistered by the attending physician, following the prescribed instructions for use. The rescue analgesics included acetaminophen (an intravenous formulation used only in the Pre-group), NSAIDs, and weak opioids. The number of rescue analgesics used until postoperative day (POD) 3 was determined based on medical records.
Frequency of PONV episodes within 48 h postoperatively (times)
In cases where PONV remained uncontrolled despite the mentioned PONV preventive measures, additional antiemetic drugs were administered at the discretion of the ward nurses. Intravenous metoclopramide solution or dimenhydrinate tablets were administered as antiemetic drugs. In this study, the frequency of PONV occurrence was defined as the number of times antiemetic drugs were additionally administered within 48 h postoperatively.
Amount of food intake at the start of meals (points)
The amount of food consumed at the start of the meal intake was examined, representing the portion of the meal provided for which the main and side dishes were ingested (in points). The intake of main and side dishes was evaluated by nursing assistants in the ward through visual inspection, with a total score of 20 points (0–10 points each). This evaluation was re-checked by dietitians. Additionally, the time taken from the end of surgery until the intake of the main and side dishes reached 16 points was noted.
Score at the beginning of ambulation (points)
The level of ambulation on the day of getting out of bed was scored using the Intensive Care Unit Mobility Scale, which ranges from 0 points for no activity to 10 points for independent walking on an 11-point scale11.
Discharge criteria
The following were the discharge criteria:
-
Pain was controlled with oral medication.
-
The patient could independently perform oral intake, urination, and defecation.
-
The mobility score was 9 or 10 points, indicating good ambulation.
-
Blood test results had returned to preoperative levels.
-
Imaging did not reveal any abnormalities at the surgical site.
Sample size calculation
The setting of the number of target patients in this study was based on pilot data, as similar studies have not been conducted previously. In the pilot data, the standardized effect difference between the Pre- and Post-groups for the primary endpoint of this study, “NRS score at rest at 6 am on the morning of POD1,” and a similar evaluation indicator, “maximum NRS score on POD1,” was 0.67. In addition, from the perspective of feasibility, the number of evaluable patients in the Pre-group was set at 30. With this sample size in the Pre-group, the Post-group required 46 evaluable patients to detect the effect difference between the Pre- and Post-groups with a power of 0.8. Sample-size calculation and statistical analysis were using SAS version 9.4 (SAS Institute Inc., Cary, NC).
Statistical analyses
In this study, to eliminate the effect of participant demographics on the primary endpoint evaluation, a mixed-effects model was used for analysis. Items influencing the balance of both groups were incorporated as covariates into the model to determine the effect difference between the Pre- and Post-groups without bias in demographic information. Because the statistical analysis requires the data to be normally distributed, we checked the normality of the data distribution. The analysis of the primary endpoint used the NRS score as the response variable, and time points, groups (Pre- and Post-groups), adjusted background factor items, and the interaction of time points and groups were considered fixed effects, whereas cases were treated as variable effects. The linear mixed-effects model was used to estimate the mean NRS scores at each time point for each group, along with 95% CI, and to compare the intergroup differences. The correlation structure between time points was modeled using the CS structure. Differences in characteristics between the Pre- and Post-groups were assessed using t-tests for continuous variables and Fisher’s exact tests for categorical items. Linear models were used to compare the amount of initial food intake, ambulation score, frequency of rescue analgesics used over the first 3 days postoperatively, frequency of PONV occurrence within 48 h, and length of hospital stay. The time taken from the end of surgery until the total eating score reached 16 points for the main and side dishes was compared using the Kaplan–Meier method for survival analysis, and log-rank tests were performed. Additionally, Cox regression was used to calculate the adjusted incidence rates using balanced covariates and to determine the adjusted hazard between the two groups. Linear models were applied with adjustments for balanced covariates for the amount of initial meal intake, ambulation score, frequency of rescue analgesics used over the first 3 days postoperatively, and frequency of occurrence of PONV within 48 h.
We performed a univariate analysis of background items that could affect NRS scores (0–1 day) (univariate analysis, continuous variables). For all items used in the univariate analysis, we selected multivariate items that influenced the NRS scores on POD0 and POD1 by using a forward-backward stepwise selection method. The selected items were surgical procedure (TLIF/PLIF), inhalation anesthetic (SEV/DES), body mass index (BMI) (/5), age (/10), and preoperative hemoglobin level (g/dL). These items were adjusted as covariates (Supplementary Table S1).
Results
The final sample size for analysis included 31 patients in the Pre-group and 47 patients in the Post-group (Fig. 1). Baseline characteristics are shown in Table 1; no significant differences were observed except for the type of inhalation anesthesia used.
Study flowchart. According to the inclusion and exclusion criteria of this study, there were 35 patients in the pre-group and 57 patients in the Post-group. In the pre-group, two patients were excluded due to dural tear, and in the post-group, three patients were excluded due to dural tear and one patient due to unknown fusion segment number. Furthermore, during the creation of the dataset, missing data affecting the evaluation criteria were observed in two patients in the pre-group and six patients in the Post-group and were excluded. As a result, the numbers of participants for analysis were 31 in the pre-group and 47 in the post-group.
Regarding results for the primary endpoint, NRS score at rest on POD1 morning, after balance adjustment, the Post-group [1.21 (0.81–1.60)] showed a significantly lower value than the Pre-group [3.91 (3.41–4.41)], with a difference of − 2.70 (–3.35 to − 2.05, P < 0.0001) (Table 2).
Results for the secondary endpoint, the NRS score at rest at points are presented in Table 2. The temporal changes until POD3 are shown in Fig. 2. NRS scores were consistently lower in the Post-group than in the Pre-group at all time points. Results for the other secondary endpoints are listed in Table 3. Regarding frequencies of rescue analgesics use until POD3, after balancing adjustments, the difference between groups was − 2.33 (–3.28 to − 1.39, P < 0.0001), indicating a lower frequency in the Post-group than in the Pre-group.
NRS scores at rest in both groups. Pre: non-scheduled intravenous acetaminophen. Post: scheduled intravenous acetaminophen. The NRS scores consistently showed lower values in the post-group compared with the pre-group across all time points. Particularly, significant differences between the groups were observed, especially during POD 0 Max, POD1 morning, POD1 afternoon, and POD1 night.
P < 0.05. ***P < 0.0001, **P = 0.0001, *P = 0.0024, ✝P = 0.0036, §P = 0.0066, ¶P = 0.0218.
There were no significant differences between Pre- and Post-groups in terms of the frequencies of PONV onset within 48 h postoperatively, initial amounts of food intake, ambulation scores at the time of getting out of bed, and geometric means of the length of hospital stay, although there was a trend towards a reduction in hospital stay by approximately 2 days in the Post-group.
Regarding the time at which the eating rate reached 80% after surgery, there was no significant difference between the two groups after covariate adjustment (log-rank test, P = 0.1161) (Fig. 3).
Timing to reach 80% eating rate (adjusted) in both groups. Pre: non-scheduled intravenous acetaminophen. Post: scheduled intravenous acetaminophen. *P < 0.05. After surgery, regarding the timing at which the eating rate reached 80%, there was no significant difference between the two groups after adjusting for covariates (log-rank test P = 0.1161).
Discussion
This study investigated the effects of ATC intravenous acetaminophen infusion in patients undergoing elective spinal-fusion surgery (PLIF or TLIF). This observational study grouped patients into a Pre-group (before) and Post-group (after) based on when ATC administration was introduced into the clinical pathway. The primary endpoint, NRS score at rest on POD1 morning, was significantly lower in the Post-group than in the Pre-group. Additionally, the secondary endpoints, such as the NRS score at rest and the frequency of rescue analgesic use until POD3, were significantly lower in the Post-group than in the Pre-group. However, no significant differences were observed in the amount of food intake at the start of meals, ambulation score, and length of hospital stay in the Post-group compared with that in the Pre-group.
The postoperative recovery strategy known as the ERAS protocol, developed in the Nordic countries in 2005, has demonstrated its effectiveness in reducing postoperative complications, improving safety in perioperative management, and shortening hospital stays, particularly in the field of gastrointestinal surgery3. Subsequently, its application was expanded to various surgical procedures; in 2021, an ERAS protocol for spinal surgery was also published12. This protocol aims to enhance postoperative recovery by preventing postoperative pain, nausea, and vomiting and achieving early mobilization and oral meal intake. The pain management strategy involves MMA to enhance analgesic effects and reduce opioid usage. Specifically, intraoperative approaches include infiltration anesthesia with long-acting local anesthetics and intrathecal analgesia. Furthermore, during and after surgery, non-opioid medications, such as acetaminophen, NSAIDs, and intravenous opioids, are concurrently used. However, high-level evidence-based conclusions regarding postoperative pain management in spinal surgery have not yet been established or are lacking as per existing research12. A study conducted in Canada in 2020 highlighted that in 1,300 scheduled spinal surgeries within 24 h postoperatively, inadequate pain control was observed in 57% patients13.
In this study, improvements in the primary endpoint and a secondary endpoint (i.e., NRS score at rest) were observed in the Post-group. Furthermore, the Post-group showed a lower usage of rescue analgesics until POD3 than the Pre-group, suggesting that higher-quality pain management was achieved. When compared with results of our previous study, wherein we introduced ATC intravenous injection as part of an analgesic protocol for postoperative pain management in gynecological surgeries, the current study demonstrated a more significant improvement in pain levels6. The reason behind this could be attributed to the generally higher postoperative pain levels in spinal surgeries targeted in this study, which resulted in more pronounced effect of the innovative pain management strategies5. Furthermore, in this study, correction for the imbalance in background factors was performed by incorporating items that could affect NRS score as covariates into the statistical model, thereby adjusting for differences between the Pre- and Post-groups. By employing such statistical methods, the evaluation of the NRS score at rest in this study achieved high reliability, because it allowed for the precise correction of background factor imbalances. However, because the NRS itself is essentially a subjective assessment, there is a risk of variation between patients. Accordingly, careful consideration is required when assessing pain.
In this study, we examined postoperative recovery ability by assessing the initial food intake level at the start of meals, ambulation scores at the time of getting out of bed, and length of hospital stay as secondary endpoints. These items were considered as secondary endpoints because of a study conducted by Maessen et al.., which investigated effects of various measures to promote postoperative recovery after gastrointestinal surgery on the length of hospital stay. Maessen et al.. demonstrated that achieving early drinking, eating, and ambulation postoperatively contributed to a reduction in hospital stay14. In the present study, there was no significant difference in the ambulation scores at the time of getting out of bed between the two groups. This may reflect the standardized ambulation schedule outlined in the clinical pathway. Furthermore, although we anticipated an increase in food intake at the start of meals due to effective pain control, there was no significant change. This may be explained by the fact that unlike gastrointestinal surgery, spinal surgery allows for the quicker recovery of digestive function, and patients in the Pre-group were able to obtain sufficient food intake immediately after surgery, making the difference less noticeable. However, although there was no statistical difference, the Post-group showed a tendency towards a reduction of about 2 days in the length of hospital stay. This aligns with the results of a meta-analysis conducted by Licina et al.. who investigated the use of MMA for pain management in spinal surgery15. According to Licina et al.., the use of a three-drug MMA regimen resulted in a shorter hospital stay compared with the use of a two-drug regimen15. We suppose that the implementation of MMA with a three-drug regimen, including opioids, acetaminophen, and NSAIDs, in the Post-group improved the quality of pain management, making it easier to meet the discharge criteria at our hospital.
In the postoperative pain management protocol for the Post-group, ATC acetaminophen intravenous infusion was the core of MMA. This protocol was developed based on information obtained from several previous studies and guidelines1,2,4,5,16. Initially, referring to the three-step analgesic ladder for postoperative pain proposed by Crews in 2002, we created a protocol considering PLIF equivalent to Step 3, which represents the most intense postoperative pain17. Additionally, the protocol was aligned with the ERAS protocol for spinal surgery, consistent with our hospital’s postoperative management policy incorporating the principles of MMA, ATC, and opioid-sparing12. The choice of intravenous acetaminophen infusion for MMA and ATC was influenced by the findings of Cakan et al.16. , who combined ATC intravenous acetaminophen infusion every 6 h for 24 h with morphine PCA (without basal infusion) as a postoperative pain management protocol for laminectomy or discectomy. Their results indicated no difference in opioid consumption, but the postoperative 24 h visual analog scale score, vomiting, and patient satisfaction improved compared with morphine alone16. As mentioned earlier, some country-specific guidelines for spinal surgery recommend this approach as an effective analgesic method18,19,20. Based on this information, the postoperative pain management protocol implemented in the Post-group of our study resulted in lower NRS score at rest over 3 postoperative days at all time points compared with the Pre-group. The establishment and implementation of a postoperative pain management protocol in the clinical pathway have been crucial for perioperative pain management in spinal surgery. Similar to other fields, spinal surgery is expected to evolve towards minimally invasive surgery (MIS). In this study, two patients in the Post-group underwent MIS-TLIF. Although the number of patients was limited for a detailed analysis, considering the anticipated increase in MIS cases, it would be advisable to reassess the clinical pathway, including the pain management protocol, in line with the trend toward less invasive surgeries.
This study has three limitations. First, the NRS score used as the primary endpoint in this study is a subjective indicator influenced by the patient’s sensitivity to pain, which may have affected the evaluation level. Ideally, a more specialized and objective pain assessment tool for assessment of pain levels after spine surgery, such as the Calgary Postoperative Pain After Spine Surgery (CAPPS) score, should be utilized The CAPPS score numerically evaluates the quality of pain management based on eight factors: age, sex, opioid use, pain intensity, number of operated vertebral bodies, presence of fixation, and psychological state21. In this study, owing to limitations in extracting psychological state from these factors, the decision was made to use the NRS. We would like to implement pain assessment using CAPPS and conduct evaluations in future studies. Second, it was not possible to establish the effect of ATC intravenous acetaminophen through a prospective study design. At the beginning of the study, ATC intravenous acetaminophen had already been initiated for spinal surgery, making it impractical to plan new interventional trials. Therefore, as shown in Supplementary Table S1, the possibility that various confounding factors influenced the results could not be ruled out. However, in this study, we adjusted for this limitation by using a mixed-effects model as a statistical method. This method allows us to consider correlations between consecutive data points and improve the accuracy of estimations at each time point, even for time-series data such as NRS scores. Third, anesthesia management using motor evoked potential (MEP) monitoring could not be performed. In this study, inhalation anesthesia was primarily used, and remifentanil was used as a supplementary agent and in small amounts. Therefore, owing to its short-acting nature, the impact of remifentanil on the outcomes was considered minimal.
Conclusions
In patients undergoing elective spinal-fusion surgery, the introduction of ATC intravenous acetaminophen into the clinical pathway results in significantly lower NRS scores at rest to POD3, along with reduced use of rescue analgesics. Postoperative recovery items showed no statistical differences between groups, although the Post-group showed a tendency towards the reduction of the length of hospital stay. The implementation of a postoperative pain management protocol within the clinical pathway is crucial for reducing postoperative pain levels and enhancing postoperative recovery capabilities in spinal surgery. Furthermore, given the limitations identified in this study, we believe that the external validity of this protocol should be confirmed through future multicenter prospective studies.
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
American Society of Anesthesiologists Task Force on Acute Pain Management. Practice guidelines for acute pain management in the perioperative setting: an updated report by the American society of anesthesiologists task force on acute pain management. Anesthesiology 116, 248–273 (2012).
Chou, R. et al. Management of postoperative pain: a clinical practice guideline from the American pain society, the American society of regional anesthesia and pain Medicine, and the American society of anesthesiologists’ committee on regional anesthesia, executive committee, and administrative Council. J. Pain. 17, 131–157 (2016).
Fearon, K. C. et al. Enhanced recovery after surgery: a consensus review of clinical care for patients undergoing colonic resection. Clin. Nutr. 24, 466–477 (2005).
European Society of Regional Anaesthesia and Pain Therapy. Prospect https://esraeurope.org/pain-management/ (2024).
Gerbershagen, H. J. et al. Pain intensity on the first day after surgery: a prospective cohort study comparing 179 surgical procedures. Anesthesiology 118, 934–944 (2013).
Taniguchi, H. et al. Effect of scheduled intravenous acetaminophen on postoperative nausea and vomiting in patients undergoing laparoscopic gynecologic surgery. J. Anesth. 34, 502–511 (2020).
Tsujimoto, T. et al. Clinical outcomes of short-segment lumbar fusion in patients older than 85 years with a minimum 2-year follow-up. J. Neurosurg. Spine. 39, 40–46 (2023).
Radnovich, R. et al. Acute pain: effective management requires comprehensive assessment. Postgrad. Med. 126, 59–72 (2014).
Fletcher, D. et al. Chronic postsurgical pain in europe: an observational study. Eur. J. Anaesthesiol. 32, 725–734 (2015).
Apfel, C. C. et al. Evidence-based analysis of risk factors for postoperative nausea and vomiting. Br. J. Anaesth. 109, 742–753 (2012).
Hodgson, C. et al. Feasibility and inter-rater reliability of the ICU mobility scale. Heart Lung. 43, 19–24 (2014).
Debono, B. et al. Consensus statement for perioperative care in lumbar spinal fusion: enhanced recovery after surgery (ERAS®) society recommendations. Spine J. 21, 729–752 (2021).
Yang, M. M. H. et al. Development and validation of a clinical prediction score for poor postoperative pain control following elective spine surgery. J. Neurosurg. Spine. 34, 3–12 (2021).
Maessen, J. et al. A protocol is not enough to implement an enhanced recovery programme for colorectal resection. Br. J. Surg. 94, 224–231 (2007).
Licina, A. & Silvers, A. Perioperative multimodal analgesia for adults undergoing surgery of the spine-a systematic review and meta-analysis of three or more modalities. World Neurosurg. 163, 11–23 (2022).
Cakan, T., Inan, N., Culhaoglu, S., Bakkal, K. & Başar, H. Intravenous Paracetamol improves the quality of postoperative analgesia but does not decrease narcotic requirements. J. Neurosurg. Anesthesiol. 20, 169–173 (2008).
Crews, J. C. Multimodal pain management strategies for office-based and ambulatory procedures. JAMA 288, 629–632 (2002).
Waelkens, P., Alsabbagh, E., Sauter, A., Joshi, G. P. & Beloeil, H. Pain management after complex spine surgery: a systematic review and procedure-specific postoperative pain management recommendations. Eur. J. Anaesthesiol. 38, 985–994 (2021).
Bae, S. et al. Efficacy of perioperative Pharmacological and regional pain interventions in adult spine surgery: a network meta-analysis and systematic review of randomised controlled trials. Br. J. Anaesth. 128, 98–117 (2022).
Brigato, P. et al. Enhanced recovery after surgery for adolescent idiopathic scoliosis: a revised systematic review and meta-analysis. Spine Deform. 13, 647–665 (2025).
Yang, M. M. H., Riva-Cambrin, J., Cunningham, J. & Casha, S. Validation of the Calgary postoperative pain after spine surgery score for poor postoperative pain control after spine surgery. Can. J. Neurol. Sci. 50, 687–693 (2023).
Acknowledgements
The lead author, HT, received research funding from Terumo Corporation, Tokyo. The funder had a role in statistical analysis and publication-related expenses. We would like to thank Editage (http://www.editage.jp) for their assistance with English language editing and the Biostatistical Research Corporation (http://www.biostar.co.jp/) for their assistance with the research protocol and statistical analyses.
Author information
Authors and Affiliations
Contributions
All authors contributed equally to the study design, data collection, data interpretation, figure creation, and manuscript writing. All authors approved the final version to be published and agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Corresponding author
Ethics declarations
Competing interests
The lead author, HT, received research funding from Terumo Corporation, Tokyo, indicating a potential conflict of interest. The other authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Taniguchi, H., Fukuda, K., Sato, T. et al. Effect of the intravenous acetaminophen clinical pathway on postoperative analgesia in spinal fusion surgery. Sci Rep 15, 36644 (2025). https://doi.org/10.1038/s41598-025-20499-4
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-20499-4