Abstract
This article aim is to assess the association between the aggregate index of systemic inflammation (AISI) and long-term outcomes of coronary artery disease (CAD) patients after percutaneous coronary intervention (PCI). 3482 patients are divided into the higher group (AISI ≥ 366) and the lower group (AISI < 366) according to cut-off value of AISI. The mean follow-up time was 37.59 ± 22.24 months. The primary endpoint includes all-cause mortality (ACM) and cardiac mortality (CM) and secondary endpoints include major adverse cardiovascular events (MACEs) and major adverse cardiovascular and cerebrovascular events (MACCEs). The results show that there are statistically significant differences in primary endpoint incidence. The incidence of ACM is 6.6% in higher group and 2.6% in lower group (P < 0.001) while the incidence of CM is 4.1% in higher group and 1.7% in lower group (P < 0.001). Cox regression analysis shows that the risk of ACM was increased to 2.298 times (HR = 2.298, 95% CI: 1.595–3.310, P < 0.001) and the risk of CM was increased to 2.160 times (HR = 2.160, 95% CI: 1.358–3.436, P = 0.001) in the higher group. Therefore, AISI is an independent predictor for CAD patients after PCI.
Similar content being viewed by others
Introduction
Coronary artery disease (CAD) is a heart disease that atherosclerosis causes the narrowing or occlusion of the lumen of the coronary arteries and leads to myocardial ischemia or necrosis, which is one of the leading causes of death in chronic diseases throughout the world. It has been the leading cause of death from chronic diseases worldwide1.With the development of cardiac catheterization technique, percutaneous coronary intervention (PCI) has been an effective treatment for coronary artery disease2,3.
However, cardiovascular adverse events can still occur in some patients undergoing PCI4. Therefore, it is particularly important to analyze the prediction of long-term adverse outcomes in patients with CAD after PCI. Recently, increasing evidence has recently suggested that inflammation plays an important role in the prognosis of cardiovascular disease5.
The aggregate index of systemic inflammation (AISI), calculated as neutrophils (NEU) multiplied by platelets (PLT) and monocytes (MONO) divided by lymphocytes (LYM), serves as a marker for evaluating the overall inflammatory condition using whole blood cells6, which is an easily accessible metric. It has been widely used to predict the outcome of various diseases, such as idiopathic pulmonary fibrosis7 hypertension8, subarachnoid hemorrhage9, age-related macular degeneration10 and COVID-1911. AISI is not the same as acute-phase proteins like CRP or cytokines like IL-6, yet it shows equal or improved predictive performance in various settings by integrating simple CBC-based measures. This is in line with the larger clinical practice trends that prioritize cost-effective and easily accessible indices, such as NLR, PLR, and SIRI, which have shown a strong correlation with traditional inflammatory biomarkers.
Since there are only a few studies indicating a relationship between AISI and cardiovascular disease, we conducted a study with a larger sample size to investigate the link between AISI and clinical outcomes in CAD patients who underwent PCI.
Methods
Clinical data and demographic characteristics collection
This was a large, single-center, retrospective cohort study (CORFCHD-ZZ, identifier: ChiCTR1800019699), 3561 CAD patients (including Chronic Coronary Syndrome and Acute Coronary Syndrome) who performed PCI procedures and hospitalized at the First Affiliated Hospital of Zhengzhou University from 2013 to 2017 were collected. We collected these patients’ clinical and demographic characteristics data, including sex, age, history of smoking and drinking history of hypertension and diabetes, and family history of CAD through the inpatient records at the First Affiliated Hospital of Zhengzhou University.
Blood samples were collected from patients for laboratory tests, and they were directed to fast for a minimum of 12 h beforehand and then blood parameters were measured using a standard method following the central laboratory guidelines of the First Affiliated Hospital of Zhengzhou University. The data included plasma and biochemical parameters, such as the levels of neutrophil count (NEU), platelet count (PLT), monocyte count (MONO), lymphocyte count (LYM), creatinine (Cr), triglycerides (TG), total cholesterol (TC), low-density lipoprotein cholesterol (LDL-C), high-density lipoprotein cholesterol (HDL-C), uric acid (UA).
We also obtained the angiographic results data from patients, including lesion extent, lesion stenosis, lesion location, stent implantation site and number, preoperative TIMI grading, etc. We collected these data from cardiac catheterization operating rooms at the First Affiliated Hospital of Zhengzhou University.
Inclusion and exclusion criteria
Inclusion criteria of this study were as follows: (1) Age: older than 18 years old and younger than 80 years old; (2) Patients met the diagnostic criteria for coronary heart disease and PCI treatment: (a) at least one clinical phenotype of coronary artery disease -- chronic coronary syndrome or acute coronary syndrome; (b)at least one coronary artery with ≥ 50% stenosis confirmed by coronary angiography; (c) an objective test with evidence of myocardial ischemia -- positive plate test; fractional flow reserve(FFR) < 0.8; optical coherence tomography or intravenous ultrasound suggestive of unstable plaque.
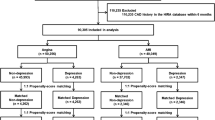
Exclusion criteria of this study were as follows: (1) Patients were excluded because baseline data for NEU, PLT, MONO, or LYM was missing. (2) younger than 18 years of age or older than 80 years of age; (3) combined with severe comorbidities: such as valvular heart disease; pulmonary heart disease; congenital heart disease; hyperthyroidism, anemia or hyperdynamic heart disease; hypertrophic obstructive cardiomyopathy; hepatic insufficiency (defined as alanine aminotransferase or total bilirubin levels more than three times above the normal maximum); renal insufficiency (defined as serum creatinine levels more than 1.5 times above the normal upper limit); disorders - such as various types of anemia, lymphoma, leukemia and other diseases; (4) Patients with known autoimmune diseases (such as systemic lupus erythematosus, rheumatoid arthritis, vasculitis, etc.); (5) Patients with acute or chronic infections within 2 weeks prior to admission (including respiratory, urinary, or systemic infections), or receiving antibiotics or immunosuppressive therapy; (6) Any history of malignancy or ongoing anti-tumor treatment. The flow chart was shown in Fig. 1.
The flowchart of patient’s enrollment.
Follow-up and endpoints
Patients were separated into two groups according to AISI and followed up after PCI by telephone. The mean follow-up was 37.59 ± 22.24 months and the range for follow-up was from 15 to 50 months. Patients’ compliance with drugs and adverse events were carefully assessed by well-trained clinical physicians during follow-up. The primary endpoint of follow-up was long-term mortality, including all-cause mortality (ACM) and cardiac mortality (CM). The secondary endpoints of follow-up were major adverse cardiovascular events (MACEs) and the composite of major adverse cardiovascular and cerebrovascular events (MACCEs). MACEs were defined as readmission, heart failure, cardiac death and bleeding events. MACCEs were defined as MACEs combined with stroke.
Statistical analysis
To justify our sample size, we performed power analyses use PASS (15.0.5). All data were analyzed using SPSS software (version 26.0; SPSS). ROC curves were utilized to determine the cut-off value of the AISI. The AISI was divided into two groups according to AISI cut-off value. Continuous variables are expressed as the mean ± standard deviation (normally distributed) and the median and interquartile range (not normally distributed). Data between two groups were compared using t-tests (for data complying with a normal distribution) or Mann–Whitney U-tests (for data complying with an abnormal distribution). Categorical variables are expressed as frequencies and percentages. They were compared using the Chi-Square test. The Kaplan-Meier method and the log-rank test were used to estimate the cumulative incidence of long-term adverse outcomes. Univariate Cox Regression Analysis models were used to assess the independent predictive value of the AISI for long-term adverse outcomes. We performed multivariable regression analyses, adjusting for clinically relevant covariates such as age, sex, comorbidities (e.g., hypertension, diabetes. Hazard ratios (HRs) and 95% confidence intervals (CIs) were calculated to estimate the association between AISI and outcomes. P < 0.05 was considered statistically significant.
Results
Power and sample size calculation
To justify our samples, we used the Test for Two Survival Curves with Cox’s Proportional Hazards Model, setting the power at 0.99, and found that 317 patients were estimated for this study. We enrolled 3482 patients, which can be regarded as reliable and persuasive.
Patient characteristics
Patient characteristics are summarized, there are 3482 patients were enrolled into this trial. Among these patients, 68.90% were male, the mean age of patients enrolled into this study was 63.27 ± 10.59. Comorbid diagnosis of hypertension was present in 2399(68.90%) patients, while 825(23.96%) were suffering from diabetes mellitus; 1062(30.50%) patients had a history of smoking, 561(16.11%) had a history of drinking. We also collected Cr, UA, TG, TC, HDL-C, LDL-C data.
ROC curve analysis
As shown in Fig. 2, ROC analysis indicated the prediction outcome value of the AISI in CAD after PCI patients with an AUC 0.606 [P < 0.001, 95% CI(0.556–0.656)] The cut-off values of AISI is 366 evaluated by receiver operating characteristic (ROC) curve, so the patients (n = 3482) with CAD after PCI were divided into two groups according to AISI cut-off value. The lower group was AISI < 366 (n = 2483) and the higher group was AISI ≥ 366 (n = 898).
The ROC of AISI.
Demographic and clinical characteristics of lower group and higher group
In the analysis, we found that there were significant differences in age, sex, Cr, UA, LDL-C, smoking and drinking history (all P < 0.05) between these two groups, however, the following variables were not significantly different between these two groups: TC, TG, HDL-C, hypertension and diabetes (all P ≥ 0.05) (Shown in Table 1).
The incidence of endpoint events
As shown in Table 2, for the primary end point, the incidences of ACM (0.03% versus 0.07%, P < 0.001) and CM (0.02% versus 0.04%, P < 0.001) were significantly different among the two groups, but for the secondary end points, we found that there were no significant differences between these two groups in the incidence of MACEs (11.0% versus 12.3%, P = 0.135) and MACCEs (14.2% versus 16.2%, P = 0.082).
Furthermore, as shown in Figs. 3 and 4, the Kaplan-Meier analyses showed that an elevated AISI was significantly related to long-term ACM (log-rank, P < 0.001) and CM (log-rank, P < 0.001).
Cumulative Kaplan–Meier estimates of the time to the first adjudicated occurrence of ACM. The X axis represents the follow-up time, and the Y axis represents the cumulative incidence of ACM. The green line indicates the higher group and the blue line indicates the lower group.
Cumulative Kaplan–Meier estimates of the time to the first adjudicated occurrence of CM. The X axis represents the follow-up time, and the Y axis represents the cumulative incidence of CM. The green line indicates the higher group and the blue line indicates the lower group.
Multivariate Cox proportional hazards regression models were conducted to evaluate the correlation between the AISI and outcomes; the models were adjusted for confounding factors, including age, sex, smoking and drinking history, Cr, UA, LDL-C. The results suggested that, compared with the patients in the lower group, the risk of ACM was increased to 2.298 times (95% CI: 1.595–3.310, P < 0.001) in the higher group, while the risk of CM was increased to 2.160 times (95% CI: 1.358–3.436, P = 0.001) in the higher group. Therefore, long-term ACM and CM will increase with AISI increases. In addition, the confounding variables related to long-term ACM and CM are displayed in Tables 3 and 4. Besides, we also included forest plots of the Cox proportional hazards regression analyses for all-cause mortality (Supplement Fig. 1) and cardiac mortality (Supplement Fig. 2) to make the data visualized.
Discussion
Coronary artery disease (CAD), is a common disease worldwide, and the most effective treatment method is percutaneous coronary intervention (PCI). Therefore, it is very important to explore noninvasive biomarker to predict outcome of CAD after PCI, the application of noninvasive biomarker not only enhances the diagnostic accuracy of coronary heart disease but also provides a basis for personalized treatment, thereby improving long-term patient outcomes. In our study, we found that the AISI was independently associated with the long-term mortality of CAD patients who underwent PCI and found that a higher AISI was independently associated with adverse outcomes in our study. In previous studies, the role of the inflammatory response in adverse cardiovascular events has been increasingly reported in recent years14, neutrophil to ltmphocyte ratio(NLR), platelet to lymphocyte(PLR) and C-reactive protein to albumin ratio(CAR) were considered to be associated with the outcomes of patients with CAD15, which demonstrated the role of inflammatory parameters in CAD patients.
Several mechanistic pathways could connect the AISI to adverse prognosis in patients with CAD after PCI. Below are explanations for the association between the AISI and adverse events from the mechanistic point of view.
First, previous studies have suggested that the neutrophils are closely associated with major adverse cardiac events in patients with CAD16,17. In the early stage of atherosclerosis, neutrophils induce endothelial dysfunction and promote foam cell formation and inflammatory response, accelerating atherosclerosis plaque formation and progression18; in the late stage of atherosclerosis, neutrophils are involved in necrotic nuclei formation and fibrous cap thinning within the plaques, promoting unstable plaque rupture19. Therefore, a high neutrophil count is considered a risk factor for adverse cardiovascular events. In addition, relevant research has stated that platelets play a central role in the process of thrombosis20. Once platelets are activated, they will promote the progression and even deterioration of CAD. It is recognized worldwide that the main cause of mortality and morbidity in coronary heart disease is the excessive activation and aggregation of blood platelets triggered by atherosclerosis plaque rupture21, which leads to thrombosis and vascular occlusion. Therefore, a high platelet count is considered a risk factor for adverse cardiovascular events. Moreover, a large of studies have demonstrated circulating monocytes play an important role in the progression of coronary plaque22. One of the characteristic features of a vulnerable plaque is a higher predominance of lipid-laden macrophages which are derived from circulating monocytes in the cap23. That means monocytes and monocytes-derived macrophages contribute to the initiation development and rupture of plaques in atherosclerosis. Therefore, a high monocyte count is considered a risk factor for adverse cardiovascular events. Last but not least, an increasing number of studies have shown that a decrease in blood lymphocyte count is strongly correlated with the development of premature coronary artery disease24. The redistribution of T cells from the circulation to lymphoid tissue could exacerbate lymphocyte growth; it might induce compensatory proliferation of antigen-experienced T cells, which could increase the risk of cardiovascular disease25.Therefore, a low lymphocyte count is considered a risk factor for adverse cardiovascular events. Based on the calculation formula of AISI is neutrophils (NEU) * platelets (PLT) * monocytes (MONO) / lymphocytes (LYM), it is obviously that AISI is strongly associated with inflammation which could cause adverse events.
The significance of our study was that we provide strong support that the inflammatory response plays an important role in the development of CAD. AISI is derived from routine complete blood count parameters, it is simple, inexpensive and widely available. This makes it a potentially useful tool for risk stratification in patients with cardiovascular disease, particularly for early identification of individuals at higher risk who may benefit from more intensive monitoring or tailored therapeutic strategies. The potential benefits of incorporating AISI into clinical practice include its non-invasive nature, cost-effectiveness, and ability to complement established inflammatory markers such as C-reactive protein (CRP) and interleukin-6 (IL-6). Moreover, in resource-limited settings where advanced biomarker assays may not be feasible, AISI could provide an accessible alternative for assessing systemic inflammation and prognosis. However, there were also some limitations to our research, which are as follows. First, compared to prospective observational study, the findings can only indicate associations rather than establish causal relationships between AISI and adverse outcomes. Second, the study was conducted in a single center research, which may limit the generalizability of the results to other populations and clinical settings. Third, our analysis was based on clinical data alone, without incorporating experimental or mechanistic studies to further elucidate the underlying biological pathways. Further prospective and multi-center studies are needed to validate its prognostic value and to define optimal thresholds for clinical application.
For further studies, we advise scholars to conduct analyses of the prognostic value of AISI in subgroup classification of patients with CAD after PCI as well as comparisons of the predictive efficacy of other inflammation index like monocyte-to-lymphocyte ratio (MLR), neutrophil-to-lymphocyte ratio (NLR), platelet-to-lymphocyte ratio (PLR), systemic immune-inflammation index (SII) and systemic inflammation response index (SIRI) on the prognosis of coronary artery disease in order to systematically and comprehensively explore the role of inflammation in the prognosis of patients with coronary artery disease. On the mechanisms by which systemic inflammation affects long-term outcomes in CAD patients, we need focus on the role of blood components such as neutrophils, monocytes, platelets and lymphocytes in outcomes of patients with coronary artery disease.
Conclusion
The research shows that AISI is an independent predictor of CAD patients following PCI, providing a valuable marker for inflammation on risk assessment and clinical decision-making in cardiovascular diseases. Furthermore, we require additional prospective trials to further validate the prognostic value of AISI and related indices such as NLR, MLR, and PLR in patients with coronary artery disease.
Data availability
The data will not be shared, because the identified participant information is included in the data. Specifically, requests for data related to this study should be directed to the corresponding author, Dr. Ying-Ying Zheng, zhengying527@163.com.
References
GBD 2019 Diseases and Injuries Collaborators. Global burden of 369 diseases and injuries in 204 countries and territories, 1990–2019: a systematic analysis for the global burden of disease study 2019. Lancet 396 (10258), 1204–1222 (2020).
Park, S. J. et al. Preventive percutaneous coronary intervention versus optimal medical therapy alone for the treatment of vulnerable atherosclerotic coronary plaques (PREVENT): a multicentre, open-label, randomised controlled trial. Lancet. 2024 Apr 4:S0140-6736(24)00413-6.
Sibbing, D. Modulation of antiplatelet therapy in PCI-Treated patients: A Rocky road toward more individualized Treatment. J. Am. Coll. Cardiol. 83 (15), 1382–1385 (2024).
Batchelor, W. B. & Damluji, A. A. Rehospitalization After PCI vs CABG for left main disease: refocusing our lens. J. Am. Coll. Cardiol. 83 (11), 1082–1084 (2024).
Farhan, S. et al. Determinants of growth differentiation factor 15 in patients with stable and acute coronary artery disease. Prospective Observational Study Cardiovasc. Diabetol. 15, 60 (2016).
Paliogiannis, P. et al. Inflammatory cell indexes as preoperative predictors of hospital stay in open elective thoracic surgery. ANZ J. Surg. 88, 616–620 (2018).
Zinellu, A. et al. The Aggregate Index of Systemic Inflammation (AISI): A Novel Prognostic Biomarker in Idiopathic Pulmonary Fibrosis. J Clin Med. ;10(18):4134. (2021).
Xiu, J. et al. The aggregate index of systemic inflammation (AISI): a novel predictor for hypertension. Front. Cardiovasc. Med. 10, 1163900 (2023).
Huang, Y. W. et al. Association between a four-parameter inflammatory index and all-cause mortality in critical ill patients with non-traumatic subarachnoid hemorrhage: a retrospective analysis of the MIMIC-IV database (2012–2019). Front. Immunol. 14, 1235266 (2023).
Sannan, N. S. Assessment of aggregate index of systemic inflammation and systemic inflammatory response index in dry age-related macular degeneration: a retrospective study. Front. Med. (Lausanne). 10, 1143045 (2023).
Zinellu, A., Paliogiannis, P. & Mangoni, A. A. Aggregate index of systemic inflammation (AISI), disease Severity, and mortality in COVID-19: A systematic review and Meta-Analysis. J. Clin. Med. 12 (14), 4584 (2023).
Aubert, C. E. et al. Adding a new medication versus maximizing dose to intensify hypertension treatment in older adults: A prospective observational study. Ann. Intern. Med. Oct 05. (2021).
Jongs, N. et al. Effect of Dapagliflozin on urinary albumin excretion in patients with chronic kidney disease with and without type 2 diabetes: a prespecified analysis from the DAPA-CKD trial. Lancet Diabetes Endocrinol. Oct 04. (2021).
Jon, W. et al. Association of lipid fractions with risks for coronary artery disease and diabetes. JAMA Cardiol. 1 (6), 692–699 (2016).
Tanriverdi, Z., Gungoren, F., Tascanov, M. B., Besli, F. & Altiparmak, I. H. Comparing the diagnostic value of the C-Reactive protein to albumin ratio with other inflammatory markers in patients with stable angina pectoris. Angiology 71 (4), 360–365 (2020).
Bao, Q. et al. CNP Ameliorates Macrophage Inflamm. Response Atherosclerosis Circ. Res. ;134(8):e72–e91. (2024).
Tudurachi, B. S. et al. Assessment of inflammatory hematological ratios (NLR, PLR, MLR, LMR and Monocyte/HDL-Cholesterol Ratio) in acute myocardial infarction and particularities in young Patients. Int. J. Mol. Sci. 24 (18), 14378 (2023).
Thazhathveettil, J. et al. Vascular smooth muscle cells in response to cholesterol crystals modulates inflammatory cytokines release and promotes neutrophil extracellular trap formation. Mol. Med. 30 (1), 42 (2024).
Song, L. et al. Significance of neutrophil extracellular traps-related gene in the diagnosis and classification of atherosclerosis. Apoptosis. Feb 17. (2024).
Kristiansen, J. et al. Acute and subacute effects of strenuous exercise on platelet aggregation, coagulation and fibrinolysis in patients with stable coronary artery disease. Thromb. Res 236, 220–227 (2024).
Xu, X. R. et al. Platelets are versatile cells: new discoveries in hemostasis, thrombosis, immune responses, tumor metastasis and beyond. Crit. Rev. Clin. Lab. Sci. 53 (6), 409–430 (2016).
Nozawa, N. et al. Association between Circulating monocytes and coronary plaque progression in patients with acute myocardial infarction. Circ. J. 74 (7), 1384–1391 (2010).
Ghattas, A. et al. Monocytes in coronary artery disease and atherosclerosis: where are we now? J. Am. Coll. Cardiol. 62 (17), 1541–1551 (2013).
Nunez, J. et al. Low lymphocyte count and cardiovascular diseases. Curr. Med. Chem. 18 (21), 3226–3233 (2011).
Zhao, Z. et al. Prognostic value of systemic immune- inflammation index in CAD patients: systematic review and meta-analyses. Eur. J. Clin. Invest. 54 (2), e14100 (2024).
Acknowledgements
Acknowledgements: The authors are grateful to the Department of Cardiology at the First Affiliated Hospital of Zhengzhou University for their help and expertise in conducting this study.
Funding
This research was funded by the National Natural Science Foundation of China (82000238) and the Natural Science Foundation of Xinjiang Uygur Autonomous Region, the Outstanding Youth Fund (2022D01E70).
Author information
Authors and Affiliations
Contributions
Authors’ Contributions(I)Conception and design: TDL and YYZ; (II) Administrative support: JYZ; (III) Provision of study materials or patients: All authors; (IV) Collection and assembly of data: TDL and SS; (V) Data analysis and interpretation: TDL and YYZ; (VI) Manuscript writing: All authors; (VII) Final approval of manuscript: All authors.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics statement
This study protocol was approved by the ethics committee of the First Affiliated Hospital of Zhengzhou University, and was in line with the Declaration of Helsinki. Due to the retrospective design of the study while the data are anonymous, the need to obtain informed consent from eligible patients was waived by the Ethics Review Committee for Scientific Research Projects of the First Affiliated Hospital of Zhengzhou University. The authors were accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.


Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Liu, TD., Sun, S., Zheng, YY. et al. Aggregate index of systemic inflammation as a new prognostic marker in patients with coronary artery disease undergoing PCI. Sci Rep 15, 37623 (2025). https://doi.org/10.1038/s41598-025-21485-6
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-21485-6