Abstract
The Incentive Spirometer (IS) encourages deep breaths to improve lung volume and prevent respiratory complications, but its cost limits some populations. The low-cost "modified Pachon Incentive Spirometer (mPIS)" was developed, and this study aims to compare the distribution of pulmonary ventilation between a low-cost IS named mPIS and another commercial flow-based IS using electrical impedance tomography (EIT). A randomized clinical trial that includes healthy volunteers aged 18–65 years. Responses obtained with the mPIS, and a branded IS were compared. Randomization was used to assign treatments. Spirometry was performed to rule out chronic lung disease, and EIT was used to evaluate the distribution of pulmonary ventilation, measuring variables such as Tidal Variation Minute and End-expiratory lung volume. When comparing the %TVM in the different regions of interest (ROIs) before and after using the mPIS and the branded IS, no changes were found in any of the ROIs for both devices. However, when comparing the global ΔEELIT3-T1 change, the mPIS showed a increase compared to the branded IS (p = 0.049). Additionally, in ROI1 (right anterior), the mPIS showed a increase in ΔEELIT3-T1 (p = 0.000), while in the other ROIs no differences were identified with the use of both incentives. The study demonstrated that the mPIS, is a valid option to improve pulmonary ventilation, as it showed a significant increase in global ΔEELIT3-T1 compared to a commercial device. This suggests that the mPIS can be as effective as more expensive options, representing an accessible alternative in resource-limited settings.
Similar content being viewed by others
Introduction
The Incentive Spirometer (IS) is a visual feedback device designed to visually stimulate patients as they perform slow, deep inspirations1. It is used to increase lung volume and improve gas exchange2.
In the clinical context, it is employed to prevent respiratory complications and to treat postoperative atelectasis in patients undergoing abdominal, thoracic, and neurosurgical procedures3. Its effectiveness has been validated in research supporting improvements in lung capacities and volumes1,2,3,4,5,6,7.
Devices oriented by flow or volume have been designed3, leading to the emergence of various brands in recent years2,4. However, in many cases, their prices can be unaffordable for users with limited resources, especially in rural and remote areas. Additionally, many developing countries face challenges in accessing the medical equipment necessary to provide adequate care, contributing to high mortality rates from conditions that could be prevented with appropriate technology and resources. This public health issue suggests an urgent need to design low-cost medical equipment to increase affordability8–10. The development of medical technologies has historically been concentrated in high-income countries, creating a technological gap that reinforces global health inequities.
In the 1990s, a Colombian anesthesiologist designed an IS that can be manufactured using low-cost materials. After some modifications, this device was named the "Modified Pachón Incentive Spirometer (mPIS)" and was registered with the Superintendence of Industry and Commerce of Colombia at the end of 2019. Laboratory tests characterized it as a flow-based incentive spirometer capable of mobilizing inspiratory flows ranging from 600 cc/second to over 1400 cc/second.
A guide detailing the manufacturing process is available and can be used without the need for licenses; however, this device lacks studies that allow for an objective assessment of how ventilation is distributed compared to a commercial IS. The mPIS operates on fundamental principles of respiratory physiology and fluid mechanics. During inspiration, the reduction in intrapleural pressure generates a pressure gradient that drives airflow from the atmosphere into the buretrol chamber. This airflow displaces a latex condom inside the buretrol, providing visual feedback to the patient. The magnitude of the displacement depends on the balance between the pressure-driven force and the elastic resistance of the condom. To maintain consistent displacement and effective visual feedback, the patient must generate a steady inspiratory flow. Additionally, the device is designed to limit the maximum displacement of the condom, enhancing the perception of peak inspiratory flow and reinforcing patient engagement during the maneuver11.
Despite its promising design and affordability, a key challenge remains: how to objectively evaluate its effectiveness in promoting optimal ventilation. In the clinical setting, the evaluation of lung expansion has relied on global parameters such as oxygen saturation via pulse oximetry, arterial blood gas measurements, lung auscultation, and radiographic imaging12, However, these do not provide the necessary sensitivity to objectively and in real-time assess ventilation distribution when using IS. On the other hand, chest X-rays and computed tomography provide images that healthcare professionals can use to assess changes in regional lung volume, but they have limitations such as delays in detecting dynamic changes, the need to reposition the patient, and radiation exposure13.
In light of this situation and the need for real-time monitoring, Electrical Impedance Tomography (EIT) emerges as a diagnostic tool that utilizes the electrical characteristics of tissue. This technique provides information in a non-invasive, continuous manner, at the bedside, and without radiation14,15. Its ability to monitor pulmonary ventilation and perfusion allows for repeated imaging of tidal volume distribution, as well as distinguishing the characteristic regional filling and emptying of each lung, all in real time and safely.
However, most studies using EIT have focused on critically ill or mechanically ventilated patients. Few have evaluated its application during respiratory therapy maneuvers in healthy individuals, particularly in the context of using low-cost or non-commercial devices like the mPIS. This gap in the literature limits our understanding of how affordable innovations perform in terms of physiological outcomes. This research was motivated by the need to objectively assess whether the mPIS, a low-cost and easily reproducible device, produces comparable regional lung ventilation patterns to a branded commercial IS in healthy individuals. This is particularly relevant in low-resource contexts where access to standard devices may be limited.
The objective of this study was to compare the distribution of pulmonary ventilation between the mPIS and another commercial flow-based IS using EIT in a healthy population in a city in southwestern Colombia.
Related work
Electrical impedance tomography (EIT) has been widely studied for its potential in evaluating regional pulmonary ventilation in both healthy and clinical populations. Frerichs et al. through the TREND consensus, established the technical and clinical foundation for thoracic EIT, offering a structured framework for data analysis, terminology, and clinical applicability. This work remains pivotal for ensuring reproducibility and standardization across EIT research lines16.
In healthy adults, studies have explored the behavior of pulmonary ventilation and muscle activation under different conditions. For example, Sugimoto et al.17 analyzed correlations between muscle mass and strength using bioelectrical impedance analysis, providing indirect evidence on how impedance-based methods can reflect physiological changes in healthy individuals. Though not focused on pulmonary EIT, this study demonstrates how bioimpedance tools contribute to understanding functional responses in health.
Rowley et al. conducted a randomized trial comparing EzPAP and incentive spirometry in postoperative patients, evaluating the impact of both techniques on dorsal ventilation redistribution using EIT. Although no significant differences were found between devices, the study highlighted the ability of EIT to quantify dynamic changes in end-expiratory lung impedance (ΔEELI) and demonstrated its utility as a bedside tool13.
Similarly, Reychler et al. used EIT to compare incentive spirometry and positive expiratory pressure in post-anesthesia recovery. Their findings revealed significant increases in ventilation and recruitment during device use, although these effects were not sustained post-intervention. This underscores the value of EIT in evaluating short-term therapeutic effects in real time18.
In the neonatal population, Ako et al. emphasized the unique respiratory characteristics of neonates and the potential of EIT for continuous, non-invasive monitoring. The authors advocate for broader integration of EIT in neonatal intensive care, noting its relevance in minimizing ventilator-induced lung injury and supporting individualized treatment strategies19.
In contrast to most prior works, which focus either on critically ill patients or general functional characterization, the present study uniquely contributes by evaluating two types of incentive spirometers (one commercial and one low-cost prototype) in a controlled, healthy population, using EIT to detect subtle physiological changes in regional ventilation and end-expiratory lung volume. This not only addresses the need for accessible therapeutic devices but also validates the potential of EIT for comparative device testing.
Methodology
Design
Randomized crossover clinical trial approved by the Human Ethics Committee of the Universidad del Valle, Cali, Colombia (No. 009-022), and registered on ClinicalTrials.gov (NCT05532748, 04/09/2022). The research was conducted in accordance with the ethical principles established in the declaration of Helsinki.
Participants
Healthy volunteers aged 18–65 who signed the informed consent form were included, while individuals with any condition that could alter the tomographic signal (pacemakers, cardioverters, defibrillators, metal implants), pregnant women, individuals with a BMI > 35 kg/m2, individuals with skin lesions preventing the placement of the electrode belt, individuals with a high level of physical activity according to the short version of the International Physical Activity Questionnaire (IPAQ), individuals whose spirometry indicated any alteration, and individuals who did not understand the verbal command of the incentive technique were excluded20.
Volunteers were recruited through communication channels established by the University of Valle, including social media, emails, and word of mouth to create a chain effect. The call provided participants with all necessary information before contacting the researchers. Individuals who decided to enroll in the study were informed about the date, time, and location of the study, details organized according to availability and capacity, as well as safety protocols. Compliance with these measures was essential.
Randomization
This study employed a simple randomization procedure to assign participants to the different treatment sequences, ensuring an unbiased and equitable allocation between groups. The random sequence was generated using the online platform https://www.randomizer.org/, and an allocation concealment mechanism was implemented to maintain blinding of both investigators and participants regarding group assignment. This process was overseen by an independent researcher not involved in data collection, thereby reducing the risk of allocation bias.
Although both the randomization process and data analysis were conducted under blinded conditions, it is important to note that full blinding of investigators and participants was not feasible, as both were aware of the device used in the intervention. This limitation was inherent to the nature of the device, which precluded complete blinding. Nevertheless, additional strategies were adopted to minimize the impact of this factor and reduce the potential for bias related to knowledge of the intervention by study personnel and participants.
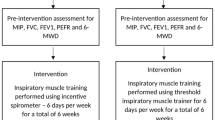
Furthermore, to mitigate other potential sources of bias, such as information bias, random data audits were implemented. An external evaluator, not involved in the study measurements, conducted biweekly random reviews to verify consistency between the original data from the tomography device, the recorded forms, and the study database. This external verification strategy contributed to ensuring the validity and reliability of the data collected throughout the study (Fig. 1).
Study flowchart.
Sample size calculation
This study was classified as a phase 0 clinical trial, primarily aimed at exploring the initial behavior of a new respiratory device in a small population under controlled conditions. As this was a pilot study and in accordance with relevant recommendations for this type of research, a formal sample size calculation was not performed. Phase 0 clinical trials are characterized by small sample sizes and short-duration interventions, with the objective of obtaining preliminary data on the safety and performance of the device, rather than drawing definitive conclusions about its efficacy21,22.
The sample consisted of 30 subjects (15 men and 15 women), all healthy volunteers. This sample size is consistent with the standards for this type of trial. Participant selection was based on non-probabilistic convenience sampling, a common strategy in exploratory studies, particularly in early phases of device development. Although the number of participants was limited, it was appropriate for this initial evaluation phase, as it allows the identification of relevant trends and assessment of protocol feasibility without attempting to generalize the findings to a broader population at this stage of the research process.
Measurements
Spirometry
A baseline spirometry was performed on all participants before the start of the study using the Medgraphics Cardiorespiratory Diagnostics® equipment to obtain an objective measure of pulmonary function and confirm that the volunteers had normal pulmonary function. This ensured that no preexisting differences, which could influence the results, were present. The test was conducted under an established safety protocol, ensuring the safety of participants, witnesses, and researchers. Standards from the American Thoracic Society and the European Respiratory Society were employed23 Participants were required to abstain from physical activity, smoking, vaping, or consuming caffeinated beverages for at least 12 h before the examination. FEV1, FVC, and the FEV1/FVC ratio were recorded.
Electrical impedance tomography
The variables evaluated were Minute Tidal Variation (%TVM), representing the regional relative impedance over the last minute expressed as a percentage, which refers to the distribution of pulmonary ventilation in a specific region of a cross-section of the thorax; and the second variable was ΔEELIT3-T1, the change in average pulmonary impedance from the end of expiration at two different moments, which can be interpreted as changes in lung volume at the end of expiration in the electrode plane. This represents functional residual volume, which can be interpreted as the pulmonary recruitment generated in a specific lung region of a cross-section of the thorax.
The EIT measurement was conducted using the PulmoVista 500 by Dräger. A belt with 16 integrated electrodes was placed around the patient’s thorax, positioned between the fourth and fifth intercostal spaces. When selecting the belt, the participant’s thoracic diameter was considered to ensure a proper fit. The patient was seated in a high-backed chair, ensuring that the belt was always in contact with a surface and that the signal from the equipment was optimal. To minimize measurement errors and interindividual variability, several standardization strategies were implemented, including precise electrode placement, consistent acquisition and image processing protocols, periodic calibration of the EIT device, and control of patient movement during data collection. These measures ensured the reliability and reproducibility of the impedance data obtained throughout the study.
Before initiating the protocol, a researcher not involved in the EIT measurements explained to the participant, via a video, the correct use of the assigned incentive according to randomization. Once the patient was prepared, recording began. Initially, two minutes of quiet breathing were recorded, marking this moment as the first event corresponding to Time 1 (T1). Next, the pulmonary re-expansion protocol was carried out, consisting of three sets of ten breaths through the IS, with a 30-s rest between each set. During the last repetition of the final set, the second event corresponding to Time 2 (T2) was recorded. Finally, the participant performed another two minutes of quiet breathing, marking this moment as the third event corresponding to Time 3 (T3).
The tomograph variables corresponding to events T1 to T3 were obtained from the “Data Analysis” section, which allows reviewing previously recorded data files, and from the “ΔEELI trend” view, which shows changes in end-expiratory lung impedance. These changes can be interpreted as variations in lung volume at the end of expiration in the electrode plane. The analysis focused on the spatial distribution of ventilation. To evaluate regional ventilation, regions of interest (ROIs) were defined. For this purpose, the cross-section of the thorax was divided into four equal-sized quadrants: ROI1 (right anterior), ROI2 (left anterior), ROI3 (right posterior), and ROI4 (left posterior). This quadrant-based division is a commonly used and validated approach in electrical impedance tomography (EIT) studies for characterizing the spatial heterogeneity of lung ventilation. It allows for the evaluation of gravitational and lateral distribution patterns and facilitates comparison between anterior vs. posterior and right vs. left lung regions. Although EIT images capture the entire thoracic cross-section, lung ROIs are typically defined functionally by ventilation-related impedance changes. By segmenting the image into quadrants, this method provides a practical and reproducible framework to detect regional variations in ventilation, as previously described in the EIT consensus statement and related literature (Fig. 2)16.
Region of interest (ROI): ROI1 (right anterior), ROI2 (left anterior), ROI3 (right posterior), and ROI4 (left posterior). The distribution of the ROIs configured in quadrants: ROI1 (right anterior), ROI2 (left anterior), ROI3 (right posterior), and ROI4 (left posterior). The dynamic image (upper left) shows the distribution of ventilation in a transverse thoracic plane over time, with more intense blue indicating areas with greater impedance change (i.e., higher ventilation). The minute image (lower left) represents the sum of impedance changes recorded over one minute, divided into four regions of interest (ROIs): ROI 1 (upper left), ROI 2 (upper right), ROI 3 (lower left), and ROI 4 (lower right). On the right, the impedance variation curves over time are shown for each ROI. Original image.
Vital signs were monitored throughout the protocol, and dyspnea was assessed before and after the use of the incentive spirometer using the Borg scale.
Incentive spirometers
The mPIS was composed of a burette and a condom from the brand Today®, without lubricant. It was crucial to avoid using lubricated condoms, as they could cause discomfort during handling, making the condom adhere to the walls of the burette and hindering its movement. These devices were produced in series by a mechanical engineer using sterile materials. The mPIS mobilizes flows ranging from 600 cc/sec to over 1400 cc/sec (the manufacturing manual is attached).
The branded device used was the Triflo® II from HUDSON RCI®, which mobilizes flows of 600, 900, and 1200 ml/sec.
Statistical analysis
The data were analyzed using IBM SPSS Statistics V23.0. In the descriptive phase, frequencies were obtained for categorical variables, and mean, median, standard deviation, quartiles, and box plots were generated for quantitative variables.
The Kolmogorov–Smirnov test was used to verify the normality of the variables. Since not all variables followed a normal distribution, the non-parametric Wilcoxon test was used to determine whether the differences observed between the initial, intra-, and final measurements were significant. Additionally, the effect size was estimated using Cohen’s d to complement the hypothesis testing.
Results
Demographic and clinical characteristics of the participants are presented in Table 1. The study included 30 individual, 15 men and 15 women aged between 18 and 65 years. Participants were evenly distributed across the predefined age groups established at the start of the study.
All participants showed no comorbidities as measured by the Charlson index, with a score of 0–1 points indicating the absence of comorbidity. The most common level of physical activity was low, representing 66.7% of the sample, as measured by the short version of the IPAQ. Spirometry was performed to rule out any coexisting alterations in pulmonary function.
When comparing the minute tidal variation (VTM) in each of the ROIs before and after the use of each device, it was found that for the mPIS, no ROI showed statistically significant changes in the distribution of pulmonary ventilation before and after its use (p = 0.567; p = 0.812; p = 0.524; p = 0.510) for ROI 1, 2, 3, and 4, respectively. Similarly, with the branded device, there were also no statistically significant changes in the distribution of pulmonary ventilation before and after its use (p = 0.846; p = 0.331; p = 0.524; p = 0.854) for ROI 1, 2, 3, and 4, respectively (Fig. 3).
Comparison of the ventilation distribution percentage of the mPIS and branded IS. A diagram representing the distribution of pulmonary ventilation (%VTM), comparing each ROI at T1 vs. T3 with the use of the Triflo and the mPIS. The upper left quadrant (ROI1), the upper right quadrant (ROI2), the lower left quadrant (ROI3), and the lower right quadrant (ROI4) are displayed. The X-axis represents each of the times (Before: T1; After: T3), while the Y-axis represents the percentage of ventilation distribution for each ROI. ROI: Region of interest, mPIS: modified Pachón Incentive Spirometer.
In Fig. 2, the distribution of ventilation expressed in percentages (%VTM) in each of the ROIs before and after the use of each device is compared. It was found that for the Triflo, no ROI showed statistically significant changes in the distribution of pulmonary ventilation before and after its use (p = 0.932; p = 0.212; p = 0.379; p = 0.837) for ROI 1, 2, 3, and 4, respectively. Likewise, no statistically significant changes in the distribution of pulmonary ventilation measured with arbitrary units were identified in any ROI for the mPIS (ROI1 p = 0.319; ROI2 p = 0.436; ROI3 p = 0.596; ROI4 p = 0.796) (Fig. 4).
Minute tidal volume and ventilation distribution of the mPIS and branded IS. A diagram representing the minute tidal variation (VTM), comparing each ROI at T1 vs. T3 with the use of the Triflo and the mPIS. The upper left quadrant (ROI1), the upper right quadrant (ROI2), the lower left quadrant (ROI3), and the lower right quadrant (ROI4) are displayed. The X-axis represents each of the times (Before: T1; After: T3), while the Y-axis represents the value of the VTM in arbitrary units (AU): Arbitrary units. ROI: Region of interest, mPIS: modified Pachón Incentive Spirometer.
In the comparison of the global ΔEELIT3-T1 between the use of the mPIS and the Triflo, it was found that the mPIS produced a statistically significant increase in end-expiratory lung impedance compared to the Triflo (p = 0.049). The mean ΔEELI was 0.09 (± 0.08) arbitrary units for the mPIS, while for the Triflo it was 0.04 (± 0.05). To better understand the magnitude of this difference, the effect size was calculated using Cohen’s d for paired samples, yielding a value of 0.45. According to conventional benchmarks, this corresponds to a moderate effect size, suggesting that the mPIS has a clinically relevant advantage over the Triflo in promoting end-expiratory lung volume recruitment. Regarding the regions of interest (ROIs), when comparing the ΔEELIT3-T1 for each ROI, a statistically significant increase was observed in ROI1 (right anterior) with the use of the mPIS (p = 0.000). No significant changes were found in the remaining ROIs with either incentive device (Fig. 5).
Comparison of ΔEELI between the mPIS and branded IS. A diagram representing the behavior of the change in average pulmonary impedance at the end of regional expiration (ΔEELI) between T1 vs. T3, comparing the mPIS and the Triflo in each ROI and overall. The X-axis represents the ΔEELI, while the Y-axis represents the devices being compared. ROI: Regions of interest, EELI: End-expiratory lung volume, mPIS: modified Pachón Incentive Spirometer.
Discussion
The aim of this study was to compare the regional distribution of pulmonary ventilation using electrical impedance tomography (EIT) during the use of a modified incentive spirometer (mIPS) and a commercially available incentive spirometer in healthy adults from Cali, Colombia. The findings revealed a similar overall ventilation pattern with both devices; however, the mIPS promoted a greater redistribution of ventilation toward the right anterior lung region.
Access to quality services within the framework of primary health care is increasingly recognized as a key determinant of health equity. Nonetheless, this access remains uneven across countries, with low-income nations facing the most significant barriers to timely diagnosis and effective treatment24.
The development of medical devices has mainly focused on high-income countries, highlighting an imperative need for middle- and low-income countries to access essential health care devices25. In response to this situation, alternatives for low-cost medical devices have emerged that meet the quality standards of commercial devices and offer similar functions with a lower economic impact26.
In this context, the mIPS is proposed as a low-cost and freely accessible device, representing a viable alternative for patients with restrictive pulmonary disorders. A crossover design was used, where participants received interventions with both incentives, with a washout period of 7 days. The duration of this period varies between studies; some have used 5 min18, while others did not report a specific time27. In our research, a longer washout period was established, as there is no standardized time. This choice was based on the idea that a prolonged washout period can improve internal validity, although there is a risk of participant dropout.
This study employed electrical impedance tomography, a non-invasive imaging technique widely validated in the scientific community28. This technique creates images based on the electrical conductivity of biological tissue; it is non-invasive, does not produce ionizing radiation, and offers real-time imaging29.
The evaluation protocol adopted in this study included two minutes of quiet breathing, three cycles of ten breaths with each incentive, and two minutes of quiet breathing after each cycle13. This evidence-based protocol allowed measurements at different moments and captured both deep inspirations and subsequent quiet breaths, thus facilitating a detailed analysis.
The IS has been used to promote lung re-expansion, and although its results are controversial, its use remains common. Research has shown that diaphragmatic mobility is significantly greater when using volume devices compared to flow devices and manual physiotherapy techniques30. Regarding respiratory muscle activity, it has been reported that flow and volume IS generate an increase in mobilized volumes31.
In patients undergoing cardiac surgery, it has been shown that using RIs reduces pulmonary complications32, which also improves adherence to interventions and decreases ICU stay33. In patients with rib trauma, an improvement in pulmonary function has been observed34. However, other studies indicate that no significant changes are evident with the use of IS35 which may be partly due to the type of patient and the protocol used.
In our study, conducted in individuals with normal pulmonary function, changes were recorded with an increase in volume and improvements in ventilation distribution, both with the brand-name device and the mIPS, highlighting the utility of EIT, as previous studies had more global measurements.
The measurement protocol used in this study was based on previous research13, and included phases of quiet breathing and cycles of breathing with incentives. This structure was designed to ensure the quality of the procedures. Three series of ten repetitions were prescribed, supported by other studies, highlighting the importance of reproducibility in experiments36.
The results showed a significant increase in global pulmonary impedance during T2, which is related to the maximum air entry when using the RI. However, the differences between T1 and T3 were not significant. These findings coincide with research in postoperative patients18, which also observed an increase in ventilation measured with EIT following the use of the RI. This increase occurred with both the low-cost RI and the brand-name RI, generating significant changes in regional pulmonary ventilation.
Regarding the distribution of regional pulmonary ventilation, changes were observed in the ROI corresponding to the right anterior region when using the mIPS, in contrast to the brand-name incentive. We consider that the redistribution of air towards the right lung can be explained by the greater lung capacity on that side, attributable to anatomical differences between the right and left lungs, such as the morphology of each lung and the position of the heart. The right lung is generally larger than the left due to its anatomical position, with three lobes compared to the left lung’s two. This anatomical configuration results in a greater total lung capacity on the right side, which may influence ventilation distribution, particularly in body positions that favor the right lung. Moreover, the more vertical orientation of the right main bronchus promotes more efficient airflow, potentially contributing to a higher ventilation-perfusion ratio on this side. In the T3 phase, both devices exhibited no significant differences in regional ventilation distribution compared to quiet breathing, aligning with previous studies that reported stable ventilation patterns during spontaneous respiration18. These findings support the idea that, while anatomical factors may influence ventilation patterns, the devices themselves did not induce major shifts in ventilation beyond what would be expected from regular breathing.
The homogeneity in the results obtained between the mPIS and the brand-name RI was lost when analyzing the variation of global pulmonary impedance at the end of expiration (ΔEELI). In this variable, a significant increase in variation was observed with the use of the low-cost incentive, particularly in the right anterior region (ROI1) and in global ΔEELI. This difference may be attributed to the anatomical variations previously discussed. Although this difference may not have specific clinical implications in healthy individuals, it does suggest that the proposed device in this research meets the necessary standards for its use. Furthermore, the calculated effect size (Cohen’s d = 0.45) for the global ΔEELI comparison indicates a moderate magnitude of effect, reinforcing that the observed differences are not only statistically significant (p = 0.049) but also represent a non-negligible physiological impact. In fact, a moderate effect size in this context suggests that the intervention produced measurable changes in regional lung ventilation and end-expiratory lung volume, which are physiologically relevant parameters in the assessment of respiratory function. These changes reflect a redistribution of ventilation that may enhance alveolar recruitment and improve ventilation-perfusion matching, particularly relevant in populations with compromised lung function. While the clinical relevance of the observed ΔEELI changes may be limited in healthy participants, it is important to highlight that ΔEELI, as measured by electrical impedance tomography (EIT), enables real-time monitoring of pulmonary ventilation distribution. Previous studies by Rowley et al., Segura et al., and Reychler et al. have demonstrated growing interest in ΔEELI as a physiological marker of response to respiratory interventions13,18.
Beyond their physiological implications, these findings also underscore a broader socioeconomic dimension concerning equitable access to medical devices. The mIPS is not proposed as a replacement for commercial devices, but rather as an alternative that benefits vulnerable populations with economic limitations and restricted access to brand-name devices. The cost difference (2 USD versus 40 USD) is a real issue that generates controversy and may be attributed, in part, to rising personnel costs and demographic trends, as well as excessive increases driven by regulatory agencies or the industry37. Therefore, it is essential to promote the search for low-cost devices that meet high-quality standards.
Although measures were taken to mitigate various biases, a sampling bias was identified due to the convenience sampling method. However, since it was conducted in healthy subjects, the authors believe this does not compromise the validity of the results.
We recognize that a key limitation of EIT lies in its reliance on impedance as an indirect indicator of ventilation, complicating direct comparison with standard outcome measures. Additionally, this study included only participants with healthy lungs, which limits the direct applicability of the findings to individuals with pulmonary diseases.
However, the inclusion of healthy individuals is justified by the need to establish baseline measurements, control for confounding variables, and assess the intrinsic performance and safety of the respiratory devices under standardized conditions. This approach is widely accepted in preliminary research, as it allows for an initial evaluation of the intervention’s effects prior to its application in clinical populations. While the results may not be directly extrapolated to patients with respiratory conditions, they provide valuable insights that contribute to clinical reasoning and guide future research. Future research is suggested regarding the cumulative behavior of sessions with the mIPS in individuals with restrictive pulmonary disorders, considering aspects such as frequency and duration.
Overhead and implementation burden of the mPIS
The proposed modified incentive spirometer (mPIS) is characterized by its low operational burden and ease of implementation. Its design, based on accessible materials and simple mechanical components, allows for quick assembly, minimal training requirements, and straightforward use by both health professionals and patients.
Unlike other devices that may require calibration, maintenance, or specialized handling, the mPIS can be deployed in various care settings with no additional logistical demands. Its portability, absence of external power sources, and intuitive operation make it ideal for use in community health programs, rehabilitation services, and even at home.
Furthermore, its use within the standardized evaluation protocol did not prolong the intervention time compared to the commercial spirometer, indicating that it does not add complexity or delays to clinical workflows. This reinforces its potential for integration into resource-sensitive contexts without compromising efficiency or therapeutic outcomes.
Evaluation of the proposed solution under different practical scenarios
Although the concept of execution time typically applies to computational systems, in the context of the proposed mechanical incentive spirometer (mPIS), this can be interpreted as the duration required for setup, instruction, and effective patient use across diverse settings. To evaluate the usability and operational efficiency of the mPIS, three practical scenarios were considered:
The proposed mIPS device demonstrated efficient implementation times across various scenarios: approximately 4 min for setup in home-based programs, under 3 min in hospital wards, and less than 10 min in remote outpatient clinics—showing its practicality and adaptability in diverse clinical contexts.
These evaluations show that the mPIS maintains consistently short execution times across different use scenarios, reinforcing its practical utility and scalability for respiratory care in both clinical and community contexts.
Conclusion
This study demonstrated that a low-cost incentive spirometer (mPIS) produces comparable regional ventilation patterns to a commercially available device when assessed using electrical impedance tomography in healthy adults. While both devices performed similarly in terms of tidal volume mobilization and overall ventilation distribution, the mPIS led to a greater increase in the ΔEELI, particularly in the right anterior region, likely due to anatomical factors.
These findings carry important clinical implications. The observed changes in ΔEELI suggest that the mPIS not only facilitates lung expansion but also enhances regional ventilation, a key target in the management of restrictive pulmonary disorders. Improved end-expiratory lung volume may support better alveolar recruitment and ventilation-perfusion matching, especially in patients with compromised lung mechanics.
Although this study was conducted in healthy individuals, the physiological responses observed support the use of mPIS as a safe and effective tool, and justify future trials in clinical populations. The results lay the groundwork for integrating cost-effective respiratory interventions into broader public health strategies aimed at improving respiratory outcomes in underserved communities.
Data availability
For ethical considerations, the data from this research will be made available upon reasonable request. For inquiries, please contact Vicente Benavides at vicente.benavides@correounivalle.edu.co.
References
Miranda, G. et al. Effects of breathing exercises on breathing pattern and thoracoabdominal motion after gastroplasty*, ** Efeitos de exercícios respiratórios sobre o padrão respiratório e movimento toracoabdominal após gastroplastia. J. Bras. Pneumol. 36(2), 197–204 (2010).
Sanesteban, Y. Rol Del Espirómetro De Incentivo En El Postoperatorio De Cirugía Bariátrica- Una Revisión Sistemática. (Universidade Da Coruña, 2018).
Rafea, A., Wagih, K., Amin, H., El-Sabagh, R. & Yousef, S. Flow-oriented incentive spirometer versus volume-oriented spirometer training on pulmonary ventilation after upper abdominal surgery. Egypt. J. Bronchol. 3(2), 110–118 (2009).
Moreno Rodríguez, L. K. Efectividad de la espirometría incentivada vs entrenamiento de músculos respiratorios, en el postoperatorio de pacientes con cirugía de abdomen y tórax (Universidad Nacional de Colombia, 2016).
Quiles Mateo, A., Ríos Cortés, A. T., De, M. R. & CayuelaGarcía, A. M. Inspiratorio Máximo Obtenido Mediante El Uso De Dos Tipos De Pulmonares. NPunto III(28), 80–86 (2020).
Sánchez Riera, H. et al. Comparison of the efficacy of two chest physiotherapy protocols for patients with cystic fibrosis. Arch. Bronconeumol. 35(6), 275–279 (1999).
Overend, T. J. et al. The effect of incentive spirometry on postoperative pulmonary complications: A systematic review. Chest 120(3), 971–978 (2001).
Mullally, S. A toolkit for medical equipment donations to low-resource settings : Making It work. Trop. Health Educ. Trust (2013).
Neighbour, R. & Eltringham, R. The reality of designing appropriate “low cost” medical products for developing countries and their unintended consequences.
Worrell, G., Pan, X., K, Y., He, B., Institute of Electrical and Electronics Engineers., et al. IEEE Engineering in Medicine and Biology Society. Annual Conference (31st : 2009 : Minneapolis Minn). In EMBC 2009 : Proceedings of the 31st Annual International Conference of the IEEE Engineering in Medicine and Biology Society : “engineering the future of biomedicine” : 2–6 September, 2009, Hilton Minneapolis, Minnesota (IEEE, 2009).
Fernandez, E. & Garcia, Y. G. C. Caracterización y mejoramiento del “incentivo de pachón” utilizado en el hospital universitario del valle (Universidad del Valle, 2004).
Branson, R. D. The scientific basis for postoperative respiratory care. Respir Care. 58(11), 1974–1984 (2013).
Rowley, D. D. et al. A randomized controlled trial comparing two lung expansion therapies after upper abdominal surgery. Respir. Care. 64(10), 1181–1192 (2019).
Riera, J., Riu, P., Casan, P. & Masclans, J. Tomografía de impedancia eléctrica en la lesión pulmonar aguda. Med. Intensiva. 35(8), 509–517 (2011).
De Lema, B., Casan, P. & Riu, P. Electrical impedance tomography: Standardizing the procedure in pneumology. Arch. Bronconeumol. 42(6), 299–301 (2006).
Frerichs, I. et al. Chest electrical impedance tomography examination, data analysis, terminology, clinical use and recommendations: Consensus statement of the TRanslational EIT developmeNt stuDy group. Thorax 72(1), 83 (2016).
Sugimoto, H., Taga, M., Fukushima, K., Akashi, Y. & Momose, K. Relationship between 10 repetition maximum for chest press, leg press, and muscle mass measured using bioelectrical impedance analysis in healthy young adults. J. Phys. Ther. Sci. 37(2), 77–83 (2025).
Reychler, G. et al. Incentive spirometry and positive expiratory pressure improve ventilation and recruitment in postoperative recovery: A randomized crossover study. Physiother. Theory Pract. 35(3), 199–205 (2019).
Ako, A. A., Ismaiel, A. & Rastogi, S. Electrical impedance tomography in neonates: A review. Pediatr. Res. 2025, 1–11 (2025).
Mera, N., Wilches Luna, E. C. & Benavides-Cordoba, V. Comparative analysis of pulmonary ventilation distribution between low-cost and branded incentive spirometers using electrical impedance tomography in healthy adults: Study protocol. PLoS One. 19(5) (2024).
Whitehead, A. L., Julious, S. A., Cooper, C. L. & Campbell, M. J. Estimating the sample size for a pilot randomised trial to minimise the overall trial sample size for the external pilot and main trial for a continuous outcome variable. Stat. Methods Med. Res. 25(3), 1057–1073 (2016).
Browne, R. H. On the use of a pilot sample for sample size determination. Stat. Med. 14(17), 1933–1940 (1995).
Graham, B. L. et al. Standardization of spirometry 2019 update. An official american thoracic society and european respiratory society technical statement. Am. J. Respir. Crit. Care Med. 200(8), E70-88 (2019).
Inagaki, D., Nakahara, S., Chung, U., Shimaoka, M. & Shoji, K. Need for Improvements in medical device management in low- and middle-income countries: Applying learnings from Japan’s experience. JMA J. 6(2), 188 (2023).
Vasan, A. & Friend, J. Medical devices for low- and middle-income countries: A review and directions for development. J. Med. Device. 14(1) (2020).
Liu, Z., Cascioli, V. & McCarthy, P. W. Healthcare monitoring using low-cost sensors to supplement and replace human sensation: Does it have potential to increase independent living and prevent disease?. Sensors 23(4), 2139 (2023).
Morais, C. C. et al. Acute effects of lung expansion maneuvers in comatose subjects with prolonged bed rest. Respir. Care 66(2), 240–247 (2021).
Sigmen, Y. Conception d’un module d’électrodes actives pour un système de tomographie d’impédance électrique bifréquence (2012).
Medina, D. Q., Benavides-Cordoba, V. & Luna, E. C. W. Outpatient use of electrical impedance tomography in patients with chronic obstructive pulmonary disease: A scoping review. Curr. Respir. Med. Rev. 18(3), 208–216 (2022).
dos Yamaguti, W. P. S. et al. Diaphragmatic mobility in healthy subjects during incentive spirometry with a flow-oriented device and with a volume-oriented device. J. Bras. Pneumol. 36(6), 738–745 (2010).
Paisani, D. D. et al. Volume rather than flow incentive spirometry is effective in improving chest wall expansion and abdominal displacement using optoelectronic plethysmography. Respir Care 58(8), 1360–1366 (2013).
Alwekhyan, S. A., Alshraideh, J. A., Yousef, K. M. d. & Hayajneh, F. Nurse-guided incentive spirometry use and postoperative pulmonary complications among cardiac surgery patients: A randomized controlled trial. Int. J. Nurs. Pract. 28(2) (2022).
Eltorai, A. E. M. et al. Effect of an incentive spirometer patient reminder after coronary artery bypass grafting: A randomized clinical trial. JAMA Surg. 154(7), 579–588 (2019).
Sum, S. K., Peng, Y. C., Yin, S. Y., Huang, P. F., Wang, Y. C., Chen, T. P., et al. Using an incentive spirometer reduces pulmonary complications in patients with traumatic rib fractures: A randomized controlled trial. Trials. 20(1) (2019).
Carvalho, C. R. F., Paisani, D. M. & Lunardi, A. C. Incentive spirometry in major surgeries: A systematic review. Rev. Bras. Fisioter. 15(5), 343–350 (2011).
Urell, C. et al. Deep breathing exercises with positive expiratory pressure at a higher rate improve oxygenation in the early period after cardiac surgery–a randomised controlled trial. Eur. J. Cardiothorac. Surg. 40(1), 162–167 (2011).
Reinhardt, R. & Oliver, W. J. The cost problem in health care. Chall. Oppor. Health Care Manag. https://doi.org/10.1007/978-3-319-12178-9_1 (2015).
Acknowledgements
Thank to the participants who contributed to the study.
Funding
None.
Author information
Authors and Affiliations
Contributions
NM, VB-C, and E W-L wrote the main manuscript text. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Mera, N., Benavides-Cordoba, V. & Wilches-Luna, E.C. Pulmonary ventilation distribution with low-cost and commercial incentive spirometers measured by electrical impedance tomography in a randomized clinical trial. Sci Rep 15, 39276 (2025). https://doi.org/10.1038/s41598-025-23034-7
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-23034-7