Abstract
Terrestrial ecosystems exhibit varied land uses as a result of both anthropogenic activities and natural processes. These variations in land use alter plant composition, soil characteristics, topography, and management practices, and hence lead to significant differences in soil microbial communities and their properties. This study evaluates the impact of distinct land use types (riparian, forest, pasture) on soil microbial biomass and microbial stoichiometric indices under uniform climatic and pedological conditions within a micro-basin in the Eastern Mediterranean region. The microbial biomass C (Cmic) in the riparian area was 2.5 and 4 times lower than in the meadow and forest areas, respectively. Additionally, the riparian zone’s microbial quotient (qMic) was 0.5 times higher than the forest and meadow areas. Microbial stoichiometric indices, particularly qMic and metabolic quotient (qCO2), across all land uses, indicated that soils within this micro-basin were healthy and exhibited no signs of stress. The study further corroborated that land use exerts significant effects on soil microbial communities, with microbial biomass and activities largely influenced by soil organic matter. Notably, the Cmic/Nmic ratio remained within the range of 10–12 across all land uses, illustrating a fungal dominance in the microbial biomass. These findings underscore the role of land use patterns in altering soil properties, thereby influencing microbial biomass, microbial respiration, and stoichiometry in soils under similar environmental conditions.
Similar content being viewed by others
Introduction
A comprehensive understanding of soil properties within areas delineated by land use capabilities is essential for sustainable environmental management. In Turkey, as in other regions, land degradation is occurring at an alarming rate due to inappropriate land use practices1. Land use change and degradation lead to intense erosion. Kara, et al.2 reported that 86% of Turkey is vulnerable to varying degrees of erosion. Erosion predominantly affects the topsoil, where soil microbial communities are most active, significantly impacting microbial populations and their functional activities3.
Soils serve as the foundation for primary production, host diverse microbial communities that drive material cycling, and provide vital nutrients and hydrological reservoirs essential for the survival of higher organisms4. Therefore, soils in sensitive areas must be protected from changes such as erosion, inappropriate forestry practices, and other anthropogenic impacts. Over the last 30 years, the increasing demand for agricultural products has triggered the conversion of forests to agricultural land, with land use change increasing at least fivefold worldwide (from ~ 3 to ~ 15 million km2)5.
Topography, climate variations, soil properties, aboveground vegetation diversity, and land use are the main factors controlling the microbial properties and dynamics of soils6. Ecosystem health and continuity depend on carbon and nutrient flow mediated by microbial organisms in the soil–plant-atmosphere cycle6. However, anthropogenic effects and climate variability impact microbial population and diversity7. Microorganisms are sensitive to the aboveground litter composition, root structure, and root enzyme ratios of vegetation, i.e., to changes in land use8. So, the substrate quality of the organic matter entering the soil plays an important role in the diversity and populations of microorganisms as it regulates the microbial decomposition rate9.
Recently, there has been growing interest in assessing the role of soil microbial biomass and basal respiration in ecosystem functioning, particularly their influence on soil quality following land use changes10. Therefore, microbial biomass, basal respiration, and their stoichiometric properties serve as early warning indicators, exhibiting rapid and heterogeneous responses to environmental disturbances, making them invaluable for ecosystem monitoring and management11,12. Soil microorganisms are essential for terrestrial carbon and nutrient cycling, ecosystem stability, and climate regulation, as they drive organic matter decomposition and regulate nutrient dynamics, thereby enhancing soil organic carbon storage13. Notably, microbial biomass carbon contributes approximately 1–3% of the total soil organic carbon pool, underscoring its ecological significance14. Unfortunately, disruptions beyond microbial adaptability can lead to the collapse of essential ecosystem functions, such as nutrient cycling and soil structure maintenance, threatening overall ecological balance and resilience11,15.
Riparian areas differ from other regions in terms of soil and hydrological properties, yet few studies have focused on the microbial properties, soil respiration, and stoichiometric indices of soils in riparian areas16. Positioned between agricultural lands, pastures, meadows, and streams, riparian zones play a crucial role in protecting stream and lake shores from erosion by acting as buffers that filter sediments, nutrients, and pesticides17. To perform these functions effectively, riparian soils must support distinct microbial biomass and activity, reflecting characteristics of both terrestrial land uses and aquatic systems 18. Studies on soil microbial biomass carbon in different land-use systems have been conducted by many researchers worldwide18,19,20. However, most research has focused on forests and their conversion to agricultural land12,21, with limited studies on adjacent forests, pasture, and riparian areas in natural water production basins.
The Mediterranean basin, which is rich in biodiversity but highly sensitive to global warming and fires, is particularly fragile due to frequent land use transformation/change through deforestation, land degradation, and hydrological disruption22. These changes and transformations significantly impact microbial community compositions and activities23, with variations in soil biodiversity, substrate quality, and quantity, microbial carbon productivity, and stoichiometric limitations depending on soil type24,25.
Land use patterns play a crucial role in shaping microbial activity within the Mediterranean region’s micro-basins, a particularly fragile ecosystem vulnerable to the impacts of global warming21. Although numerous studies have examined the physical, chemical, and microbial properties of soils developed under different tree species16,19,21, seasons20,21, and land uses12,13,14,17,18, comprehensive research on soils within adjacent land use patterns at the micro-catchment scale remains limited. Therefore, to ensure the sustainability of ecosystem health, it is crucial to understand the soil physicochemical and microbial properties in various land uses, particularly within drinking-water collection basins26,27. In this context, this study focuses on the interactions between microbial biomass and respiration as influenced by organic matter availability and other soil health indices. Moreover, more in-depth studies to increase understanding of how land use modifications impact soil health, microbial functions, and ecosystem stability, may provide valuable implications for sustainable land and water management practices. Therefore, this research was conducted to evaluate the effects of land use on soil microbial respiration, microbial biomass, and stoichiometric properties within the Meydan Pond micro-basin in the Andırın District of Kahramanmaraş. It aims to deepen our understanding of microbes’ regulatory roles within various ecosystems and the mechanisms underlying soil microbial responses. Additionally, it provides a scientific foundation for enhancing watershed management practices, protecting vegetation, and preserving adjacent zonal ecosystems.
Material and method
Study sites
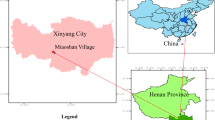
This study was conducted within a 50 km distance from Kahramanmaraş Province, located in the Northeast Mediterranean Sea mountainous region of Turkey. Except for the different land uses in each case, the sampling area has similar topographic, geological, and climatic conditions characterized by the presence of a typical karstic topography (Fig. 1). This mountain range varies from the Upper Cretaceous to Upper Miocene and a Mosezoic limestone succession; it is derived from the Taurus System28. The elevation ranges from 1200 to 1350 m, with steep slopes (50%). The forest canopy in the research area consists primarily of mixed coniferous tree species, aged 80–100 years, including black pine (Pinus nigra Arn.), Taurus fir (Abies cilicica subsp. cilicica), Lebanon cedar (Cedrus libani A. Rich.). The pasture area is typically populated by species from the genera Trifolium sp., Cynodon sp., Astragalus sp., Salvia sp., and others29. In the riparian zone, Phragmites australis is present.
Locations of the study site (a), sampling points (b) and field study (c). The map was created using Google Earth Pro Maps (version 7.3.6.9796) and ArcGIS (version 10.4.1, https://www.esri.com/en-us/arcgis/products/arcgis-pro).
The region has a hot-dry summer and cool-wet winter climate type, Mediterranean climate, (Csa) according to the Köppen-Geiger climate classification (www.koeppen-geiger.vu-wien.ac.at/). Weather data, including long-term (at least 30 years 1990–2020) averages for monthly air temperature and precipitation in Andırın district, were obtained from the Turkish State Meteorological Service (www.mgm.gov.tr). The annual mean temperature is 13.0 °C, with a high of 30 °C in August and a low of − 2.5 °C in February. Annual precipitation is 729 mm, with about 80% occurring during the winter and early spring seasons. The main soil types are generally brown forest soils, classified as Alfisols by the U. S. Soil Taxonomy30. The rugged topography consists of andesitic and limestone bedrock. Table 1 provides a detailed characterization of the study site.
Experimental design, soil sampling, and pretreatment
This study was conducted in the forest, pasture, and riparian areas of the Baskonus Plateau Meydan Pond micro-basin in May 2019. Five different sampling points were chosen randomly from each land use type (forest, pasture, and riparian) at the 0–10 cm depth, a total of fifteen soil samples for this study. A maximum depth of 10 cm was chosen for soil sampling because of the most intense microbial activity and the significant limiting effect of riparian areas beyond 10 cm depth due to the presence of water tables and rocks. These areas are designated as protected zones and are generally restricted from human activity, minimizing anthropogenic influence. Human access is typically limited to recreational use.
For microbial analysis, the litter, root debris, and stones were removed from all soil samples, then sieved through a < 2 mm mesh. Distilled water was added to until 50–60% moisture content, and the samples were stored in a portable coolant below + 4 °C until promptly transported to the laboratory for storage at + 4 °C before analysis for the maintains microbial activity. Physicochemical properties and erodibility indices were determined by Babur, et al.31.
Microbial Biomass C (C mic ) and Microbial Biomass N (N mic )
The chloroform-fumigation-extraction (CFE) method was performed to determine soil microbial biomass carbon (Cmic) and biomass nitrogen (Nmic)32. For CFE, each soil sample was divided into 2 portions of 30 g, and the first half of the soils stored for microbial analyses were fumigated with ethanol-free chloroform at 25 °C for 24 h in a vacuum desiccator in the dark incubator, while the second portion of samples was used as a control without fumigation. Both fumigated and non-fumigated soil samples were mixed in 0.5 M K2SO4 (1:4 w/v), followed by shaking the suspension for 30 min at 200 rpm on a reciprocating oscillator and supernatants were filtered through a Whatman No. 42 filter paper and stored in below + 4 °C before titration. Total organic carbon in both fumigated and non-fumigated extracts was estimated as specified by the Walkley Black method33,34. Briefly, 8 ml aliquot of K2SO4 extracts after oxidation with 0.4N K2Cr2O7 and back-titration with ferrous ammonium sulfate. Cmic was calculated from the difference in extractable organic C between fumigated and non-fumigated soil samples using the formula as follows in Eq. 132,34.
where EC refers to the difference in extractable organic C between the treatments; 2.64 is the proportionality factor for biomass C released by fumigation extraction.
The microbial quotient (Cmic/Corg or qMic) was calculated by expressing microbial C as a percentage of total soil organic C.
The Kjeldahl digestion-distillation-titration method was used to determine the total N in the extracted K2SO4 solution. Briefly, a mixture was prepared by adding 15 ml of the extract solution + 0.4 ml of 0.2 M CuSO4 + to promote organic matter degradation and then 10 ml of high purity (97%) H2SO4 and placed in the digestion unit. This mixture was digested at 380 °C for at least 3 h (until all organic compounds were decomposed). The solution was made up to 250 ml with deionized water. A 50 ml subsample was taken and distilled in the distillation unit with 10 M NaOH (strong base). The distillate was collected in an indicator solution mixed with boric acid and the solution was titrated. Nmic was calculated from the difference in extractable N between fumigated and non-fumigated soil samples by using the following Eq. 233,35,36:
where FN is the difference between N extracted from fumigated and non-fumigated samples and 0.54 is the fraction of microbial biomass N released by the fumigation extraction process.
Microbial respiration (MR)
Microbial respiration was estimated by the sodium hydroxide (NaOH) trap method (absorption method) to measure carbon dioxide (CO2)37. Briefly, 30 g of oven-dried equivalent weight moist soils were weighed and transferred to separate 500 mL capacity airtight beakers. Small beakers filled with 0.1 M NaOH were placed in the center of the bottom of the jar without soil underneath to capture the CO2 released as a result of microbial respiration in the beakers containing the soil sample. The jars were hermetically sealed and incubated at 25 °C for 7 days. After 1 week of incubation, the small beaker containing the CO2 absorbed in the NaOH solution was removed and titrated with 0.05 M HCl after adding BaCl2. An empty jar without soil was used as the control. Then, MR was calculated by dividing the amount of CO2 released during the incubation period by the dry mass of the soil. Metabolic quotient qCO2, an important parameter that can be used to predict soil quality and stress response, was calculated as the ratio between MR ratios (mg CO2-C g Cmic−1 h−1) and Cmic35,38.
Statistical analyses
The data were assessed for normality and homogeneity of variance using skewness, kurtosis, and the Shapiro–Wilk test. Parameters that deviated from a normal distribution were log-transformed (log10) before conducting variance analysis (ANOVA). Once the data met the assumptions for parametric testing, a one-way ANOVA was performed to evaluate the effects of land use type on all measured parameters, followed by Duncan’s multiple range tests (α = 0.05) for post-hoc comparisons. Pearson’s linear correlation analysis was conducted to investigate the relationships between microbial soil properties and stoichiometric parameters across different land uses. Principal component analysis (PCA) was performed using RStudio software, based on the correlation matrix of soil properties with sufficient replicate data across different land use types. This approach allowed for the visualization of overall similarities and differences among the soil properties of various land uses. PCA was used to calculate the differences between data groups, employing one or more discriminant functions to optimally distinguish among the identified groups. Statistical analyses were conducted using the SPSS Inc. (Statistical Package for Social Sciences, Chicago, IL, USA), RStudio, and figures were generated with the Rstudio and XLSTAT software package for Windows.
Results and discussion
Changes in microbial biomass C (C mic ), N (N mic ), and microbial respiration (MR) under different land-use
This study revealed significant differences in microbial properties and stoichiometric parameters across land use types (Fig. 2 and Table 2). Soil microbial biomass decreased from forest to pasture and riparian ecosystems, reflecting a transition from well-developed to sparser vegetation. Forest ecosystems supported more active microbial communities compared to pasture and riparian zones.
Changes in microbial biomass C (a), microbial biomass N (b), and microbial respiration (MR) (c) with afforestation in three land-use types. Different letters above the bars indicate significant differences at p < 0.05 among the land use types (Error bars show ± standard errors).
The Cmic and Nmic varied significantly across land-use types (p < 0.001) with forest soil being significantly higher than both pasture and riparian soils (Fig. 2; Table 2). Briefly, the soil Cmic in the topsoil (0–10 cm), the forest soils had 1487 µg g⁻1 Cmic, 63% and 280% higher than those under the pasture, 913 µg g⁻1, and the riparian soils, 391 µg g⁻1 (Table 2 and Fig. 2a). Determination of soil Cmic is one of the main factors in most soil monitoring studies and programs39. Previous studies conducted in different forest soils have reported microbial biomass C ranging from 470 to 1600 μg g⁻1 in beech forests, 586 to 790 μg g⁻1 in high-altitude (1750 m) cedar forests, and 251 to 838 μg g⁻1 in pine forests12,13,40,41. In addition, Cmic concentrations of soils in the rainy season were found to be 1330.00 μg g−1 in mixed forest ecosystems, 1222.98 μg g−1 in pastures, 380.33 μg g−1 in afforestation areas, and 284.30 μg g−1 in agricultural areas12,21. In our study, the mean Cmic values were found within these previously reported ranges. These results suggest that the high nutrient availability in mixed coniferous forests, driven by substantial litter input and composition, creates more favorable conditions for microbial decomposers. Chen, et al.42 stated in their study that the amount and type of organic matter caused significant differences in the Cmic content of soils. In addition, Vidyanagar43 found that there were significant differences in the Cmic content of topsoil (0–10 cm) developed under different land use types.
This study revealed that the average Nmic levels in soil samples varied significantly across different land use types. Nmic represents the total nitrogen content of soil microorganisms and plays a crucial role in evaluating the contribution of soil microorganisms to the nitrogen cycle, as well as in monitoring soil fertility, health, and quality11. In this study, the differences soil Nmic were statistically significant (p < 0.001) with forest soil containing significantly more Nmic than both pasture and riparian soils (Table 2). For example, the forest vegetation had 122.6 µg g⁻1 Nmic which was 74% and 183% higher than those under pasture, 70.4 µg g⁻1, and riparian soils, 43.3 µg g⁻1 (Fig. 2b). Forest soils exhibited higher Nmic contents compared to other land uses, which can be attributed to the high levels of organic carbon and total nitrogen contributed by the rapid decomposition of cedar needles in the areas12. Díaz-Raviña, et al.44 reported that Nmic content in topsoil under different tree species ranges between 42 and 191 μg g⁻1, Arunachalam and Arunachalam45 found higher Nmic content ranging between 172 and 331 μg g⁻1 in soils from humid forests in Northeast India. Other studies have found Nmic values ranging from 42 to 142 μg g⁻1 across various forest ecosystems21,46. Vidyanagar43 observed Nmic values of 123.30 μg g⁻1 in mixed forest ecosystems, 107.85 μg g⁻1 in pastures, 47.85 μg g⁻1 in afforested areas, and 42.83 μg g⁻1 in agricultural lands. These findings indicate that Nmic content is highest in forested areas, while it is lowest in agricultural and other land-use types. The lignin and nitrogen content in organic material serves as indices of litter resistance to decomposition and substrate quality47. The differences in Cmic and Nmic of soils developed under different land uses may be due to the variability in soil type. Although the soils are derived from similar parent material, they have different soil properties due to different plant biodiversity and land use7,19. For example, forest soils have higher organic matter inputs than pastures and riparian areas43.
Soil microorganisms metabolize organic carbon sources such as plant residues and dead organisms to obtain energy, resulting in the release of carbon dioxide (CO₂)35,48. This represents microbial respiration. MR plays a critical role in maintaining the carbon cycle within the soil ecosystem, facilitating the decomposition of organic matter, and contributing to soil fertility12,49. Soils of this study further showed that soil microbial respiration (MR) rates were significantly altered by land use impacts (Fig. 2c). MR in riparian soils was lower than those under both forest and pasture (p < 0.001; Table 2) where forest soils exhibited the highest MR of 1.5 µg CO₂-C g⁻1 h⁻1, which was followed by pasture soils with 1.21 µg CO₂-C g⁻1 h⁻1 MR, and riparian soils with 0.6 µg CO₂-C g⁻1 h⁻1, respectively. In this study, as a result of distinct land use among microbial respiration trends, forest > pasture > riparian areas are ranked. Some researchers have noted that increased organic matter or optimal climatic conditions enhance microbial activity, which is positively correlated with MR50,51. Similarly, in our study, it is estimated that the high OC and Cmic amounts in the soils in forest areas are due to the high tendency of microorganisms in the soil to produce energy by breaking down organic matter. VM and Marcos ML46 determined in their study on oak forest soils that the amount of MR in different seasons varied between 0.30 and 1.20 μg CO2–C g−1 h−1. Chen, et al.52 reported that MS values of soils in mixed pine forests and pastures varied between 0.17–0.65 and 0.11–0.84 μg CO2–C g−1 h−1, respectively. However, Bolat53 found the highest MR value in fir forest soils with 1.79 μg CO2–C g−1 h−1. In cases where soils carry stress conditions, the MR value will be higher as microbial organisms will produce more CO2 to provide the energy they need.
The pattern of microbial stoichiometry in the three land-use
The findings from this study also showed a significantly altered pattern of microbial stoichiometry impacted by different land uses (Tables 2 and 3). In this study, the pattern of microbial stoichiometry includes all soil qMic, qCO2, Cmic/Nmic, and Nmic/TN indicies.
The results of this study indicate significant differences in qMic in the topsoil (0–10 cm) across the three land uses: forest, pasture, and riparian (p < 0.05; Table 2). Notably, the forest soils had significantly lower qMic (2.31) where the riparian soils had the highest qMic (2.90). A similar difference was also observed between pasture soils and riparian soils where pasture soils had significantly lower qMic (2.06) than riparian soils. However, although differences between forest and pasture soils were not significant, forest soils had slightly higher qMic compared to those of pasture soils. The qMic (Cmic/Corg) ratio is a crucial parameter for assessing soil microbial activity, fertility, and overall health, and it can also serve as a tool for monitoring the effectiveness of soil management practices8. In addition, the qMic ratio is widely recognized as an effective microbial characteristic for monitoring spatial and temporal changes in soils11. Those like Jenkinson and Ladd54 suggested that a qMic ratio of 2.0 is a balance threshold for soils. The mean qMic percentage values in our study were above this threshold, reflecting the efficient use of organic substrates by microbial biomass across all land uses. According to Anderson48, a low qMic ratio results from excessive carbon being utilized for respiration, with minimal microbial biomass production, which is indicative of environmental stress conditions. Low qMic ratios suggest that only a small fraction of organic carbon is accessible to soil microorganisms, indicating poor substrate quality55, whereas a high ratio is often considered a sign of carbon immobilization43. The values obtained align with previous studies, where the qMic ratio was arranged from a minimum of 0.27 to a maximum of 9.943,44,48,56,57.
The mean qCO₂ values and standard deviations for soils across different land uses are presented in Table 3. In contrast to qMic, qCO2 did not show a significant variability (p > 0.05) across different land-use types (Table 2). The qCO2 of riparian soils (1.54 mg CO2-C g Cmic−1 h−1) was higher than those of both pasture (1.32 mg CO2-C g Cmic−1 h−1) and forest (1.00 mg CO2-C g Cmic−1 h−1) soils by 14.3% and 35.1% However, differences were not always statistically significant indicating that great variability in data may lead higher standard deviations (~ 0.59) and in turn kept p-values higher than 0.05 significant level. The metabolic quotient (qCO₂) represents one of the key indicators of the functioning and health of soil ecosystems. To assess the stress conditions within an ecosystem, it is essential to determine qCO₂ values alongside other indicators, such as the microbial quotient (qMic = Cmic/Corg)12,49. According to the ANOVA test, the qCO₂ values for soils from different land uses were not significantly different (p = 0.09; Table 2). However, post hoc analysis revealed that the land uses formed distinct groups based on their qCO₂ values, with pasture soils showing similarity to both forest and riparian soils in terms of their qCO₂ averages (Table 3). This may be attributed to the relatively close and small differences in qCO₂ average values across the land uses. Babur, et al.12 found that the average qCO2 amount of soils varied between 1.55 and 2.63 mg CO2-C g−1 Cmic h−1 in different forest ecosystems. Again, in the same study, it was stated that the average qCO2 amount increased to 4.99 mg CO2-C g−1 Cmic h−1 in the winter season when the microbial population was stressed with the decrease in air temperature and had to respire more to convert frozen organic carbon into nutrient energy. Some researchers have determined that this value varies between 0.74 and 162 mg CO2-C g−1 Cmic h−146,49,53.
When comparing the average Cmic/Nmic ratios of soils across different land uses during the sampling time, the highest average ratio was observed in pasture areas (13.14), while the lowest was found in riparian areas (9.18). Statistical analysis also showed that the Cmic/Nmic ratio in forest and pasture was significantly higher than in riparian soils (p ≤ 0.01, Tables 2 and 3), indicating more fungal dominance in forest and pasture than riparian soils which eventually may have more bacteria domination. The Cmic/Nmic ratio is a valuable indicator for determining the dominant type, structure, and status of the microorganisms that constitute the microbial biomass in the study areas58. According to Jenkinson and Ladd54, a Cmic/Nmic ratio of approximately 10–12 suggests that the microbial biomass is predominantly composed of fungi, whereas a ratio between 3 and 5 indicates bacterial dominance. Based on these threshold values, the Cmic/Nmic ratios obtained from the soils in our study indicate that the microbial community in our research fields is primarily composed of fungal species. Bauhus and Khanna56 emphasized that fungi play a more critical role than bacteria in the initial decomposition of litter, and thus, a higher Cmic/Nmic ratio is expected. Other studies have reported that the Cmic/Nmic ratio can range from 3.0 to 17.3, depending on land use and tree species12,13,53,59.
The average Nmic/TN ratios of soils across different land uses in the study areas were highest in riparian areas (4.95%) and lowest in pasture areas (2.10%). In forests, this value was found to be 4.02 (Table 3). The Nmic/TN values differed significantly among soils from different land uses (p ≤ 0.001, Table 2). The mean and standard deviation values of the Nmic/TN ratios for soils from different land uses are presented in Table 3. However, differences between forest and riparian were not statistically significant (p > 0.05). Soil Nmic/TN ratio, like other stoichiometric indices, is an important biological indicator used to evaluate soil microbial activity, nitrogen cycle, soil fertility, and general health57. It has been shown that a high Nmic/TN ratio in soils does not significantly impact the availability of nitrogen (N). In environments with elevated Nmic/TN ratios, organic compounds can be more readily utilized, leading to regular decomposition of organic matter and a subsequent decrease in its content60. According to Bauhus, et al.55, both low and very high Nmic/TN ratios may indicate limitations in substrate availability. This is because the retention of released nitrogen by microbial organisms can reduce the amount of nitrogen available for plant uptake. The low Nmic/TN ratio observed in pasture areas may be attributed to a low microbial biomass N content, increased nitrogen uptake by plants or nitrogen loss from the environment and subsequent accumulation in the riparian zone due to leaching. The riparian areas are adjacent to pasture lands and contain substantial pond water, which could contribute to this effect. Additionally, the low Nmic/TN ratio could be explained by the fixation of free nitrogen in the soil by microbial organisms. The Nmic/TN values observed in this study (0.5–12.40%) are consistent with findings from previous research12,43,49,53,56.
Relationship between microbial properties and stoichiometric indices
Pearson’s correlation analysis showed that soil MR had a significant positive correlation with Cmic, Nmic, and Cmic/Nmic (p < 0.05). About 92% of MR was explained by Cmic whereas Nmic explained 82% of MR (Table 4). A similar relationship was observed between Cmic and Nmic (p < 0.05) where these properties explained about 96% variability. Another important positive correlation was observed between qMic and Nmic/TN (p < 0.05) where both qMic explained about 76% variability in Nmic/TN. Also, the negative correlation between Cmic (58%) and Nmic (61%) with qCO2 was statistically significant (p < 0.01). Finally, A significant negative correlation was found between Cmic/Nmic and Nmic/TN (p < 0.05 with r = 74%) (Table 4).
The chemical and biochemical properties of soils interact in ways that can either enhance or hinder each other8. Since microbial stoichiometric indices are influenced by these properties, strong correlations between them are expected. For example, Sharma et al.61 reported a significant relationship between microbial biomass C and microbial biomass N (r = 0.869, p < 0.0005), a finding supported by Babur et al.21 (r = 0.580, p < 0.05). Our study corroborates these observations. Microbial biomass and activity are generally highest in topsoil, where organic material is abundant58. This is largely due to favorable climatic conditions and the higher nutrient availability in these layers21. However, as microbial populations increase, so does competition for nutrients, leading to elevated microbial activity and respiration12,28. This increased metabolic demand can cause higher qCO₂ levels, reflecting a stressed environment. Moreover, microorganisms must compete with plant roots for limited resources, which further exacerbates energy expenditure and soil stress28.
The principal component analysis (PCA; Fig. 3) exhibits the relationships between microbial communities and their stoichiometric indices across three land uses where the first two principal components explain 57.5% and 22.8% of the total variability, respectively. The first Dim (Dim1) explains about 57.5% of the variability in the data, likely indicating a gradient of microbial activity and carbon utilization efficiency which perhaps led by a positive correlation with qCO2, qMic, and Nmic/TN, and negative correlations with Cmic/Nmic, MR, Cmic, and Nmic. The second Dim (Dim 2) explains about 22.8% variability in data, perhaps reflecting a gradient of nitrogen availability and a positive correlation with Nmic and Nmic/TN and countered by Cmic/Nmic and qCO2. Perhaps due to environmental stressors, riparian soils appear to have higher qCO2, qMic, and Nmic/TN and clustered on the positive side of the Dim 1. However, pasture soils clustered on the negative side of Dim 1 and Dim 2, having high Cmic/Nmic. PCA also shows a higher response of Cmic and Nmic to the forest. These patterns suggest that Dim 1 (PC1) captures ecological shifts in microbial efficiency and activity, while Dim 2 (PC2) reflects nitrogen dynamics. The principal component analysis (PCA) results (Fig. 3) also shows 80.3% of the total variation along with the two principal components. Changes in land use had a significant impact on the ability to characterize soil microbial properties and stoichiometric indices62. These indices driven by riparian areas appear to be qCO2, qMic, and Nmic/TN where qMic is the highest affected property. In addition, Cmic/Nmic was the main driver of pastureland. Those of Cmic, Nmic, C/N, OC, and MR are drivers of the forest. The negative correlation of qCO2 with Cmic and Nmic played a critical role in differentiating the microbial properties of forests in comparison to riparian areas. On the other hand, a distinct association was observed between Cmic/Nmic and Nmic/TN, which differentiates soils from pasture areas and riparian zones.
Principle component analysis of microbial properties and stoichiometric indices.
Conclusions
The results of this study demonstrate that land use significantly influences soil microbial properties and stoichiometric indices. In tree-based systems, the increased organic matter input enhances soil organic carbon, promoting better soil health and productivity, which in turn supports greater microbial diversity. Forest soils exhibited the highest microbial biomass carbon and the lowest microbial coefficient, whereas riparian areas had the lowest values. This can be attributed to the fact that organic carbon in riparian zones is often washed into rivers and lakes, leading to nutrient-poor conditions. The low SOC and MBC in riparian areas, coupled with the lack of organic matter inputs and frequent flooding, result in poor aeration, which likely reduces soil fertility and microbial activity. When humidity, temperature, and nutrient levels are adequate, microbial biomass and activity are expected to increase. However, in riparian zones, excessive soil water and nutrient leaching contribute to diminished microbial communities and activity. These study results emphasize the need for strategic land management practices to increase soil organic matter, mitigate nutrient leaching, and enhance soil health and microbial activity in riparian zones in the Mediterranean basin. In addition, the results obtained indicate the need to improve management practices, especially in watersheds, preserve forest vegetation, and protect and enhance adjacent regional ecosystems.
Data availability
All data generated or analyzed during this study are included in this published article (and its Supplementary Information files).
References
Camci Çetin, S., Karaca, A., Haktanır, K. & Yildiz, H. Global attention to Turkey due to desertification. Environ. Monit. Assess. 128, 489–493 (2007).
Kara, Ö., Şensoy, H. & Bolat, İ. Slope length effects on microbial biomass and activity of eroded sediments. J. Soils Sediments 10, 434–439. https://doi.org/10.1007/s11368-010-0192-8 (2010).
Barto, E. K., Alt, F., Oelmann, Y., Wilcke, W. & Rillig, M. C. Contributions of biotic and abiotic factors to soil aggregation across a land use gradient. Soil Biol. Biochem. 42, 2316–2324. https://doi.org/10.1016/j.soilbio.2010.09.008 (2010).
Chen, Q., Song, Y., An, Y., Lu, Y. & Zhong, G. Soil microorganisms: Their role in enhancing crop nutrition and health. Diversity 16, 734 (2024).
Ramankutty, N. et al. Trends in global agricultural land use: Implications for environmental health and food security. Annu. Rev. Plant Biol. 69, 789–815. https://doi.org/10.1146/annurev-arplant-042817-040256 (2018).
Singh, J. S. & Gupta, V. K. Soil microbial biomass: A key soil driver in management of ecosystem functioning. Sci. Total Environ. 634, 497–500. https://doi.org/10.1016/j.scitotenv.2018.03.373 (2018).
Babur, E. Significant differences in microbial soil properties, stoichiometry and tree growth occurred within 15 years after afforestation on different parent material. Life 14, 1139 (2024).
Babur, E. et al. in Microbial Syntrophy-Mediated Eco-enterprising (eds Raghvendra Pratap Singh, Geetanjali Manchanda, Kaushik Bhattacharjee, & Hovik Panosyan) 225–245 (Academic Press, 2022).
Jagadamma, S., Mayes, M. A., Steinweg, J. M. & Schaeffer, S. M. Substrate quality alters the microbial mineralization of added substrate and soil organic carbon. Biogeosciences 11, 4665–4678. https://doi.org/10.5194/bg-11-4665-2014 (2014).
Bini, D. et al. Effects of land use on soil organic carbon and microbial processes associated with soil health in southern Brazil. Eur. J. Soil Biol. 55, 117–123. https://doi.org/10.1016/j.ejsobi.2012.12.010 (2013).
Babur, E. et al. Relationship between organic matter and microbial biomass in different vegetation types. Microbial syntrophy-mediated eco-enterprising 225–245 (Elsevier, 2022).
Babur, E., Dindaroğlu, T., Solaiman, Z. M. & Battaglia, M. L. Microbial respiration, microbial biomass and activity are highly sensitive to forest tree species and seasonal patterns in the Eastern Mediterranean Karst Ecosystems. Sci. Total Environ. 775, 145868. https://doi.org/10.1016/j.scitotenv.2021.145868 (2021).
Kara, Ö. & Bolat, İ. Soil microbial biomass C and N changes in relation to forest conversion in the Northwestern Turkey. Land Degrad. Dev. 19, 421–428. https://doi.org/10.1002/ldr.850 (2008).
Dilly, O., Blume, H.-P., Sehy, U., Jimenez, M. & Munch, J. C. Variation of stabilised, microbial and biologically active carbon and nitrogen in soil under contrasting land use and agricultural management practices. Chemosphere 52, 557–569. https://doi.org/10.1016/S0045-6535(03)00237-6 (2003).
Brady, N. C., Weil, R. R. & Weil, R. R. The nature and properties of soils. Vol. 13 (Prentice Hall Upper Saddle River, NJ, 2008).
Griffiths, R. P., Entry, J. A., Ingham, E. R. & Emmingham, W. H. Chemistry and microbial activity of forest and pasture riparian-zone soils along three Pacific Northwest streams. Plant Soil 190, 169–178. https://doi.org/10.1023/A:1004211121095 (1997).
Burchell, M. & Rivers, E. Agricultural riparian buffers. NC Cooperative Extension, NC (2017).
Bargali, K., Manral, V., Padalia, K., Bargali, S. S. & Upadhyay, V. P. Effect of vegetation type and season on microbial biomass carbon in Central Himalayan forest soils, India. CATENA 171, 125–135. https://doi.org/10.1016/j.catena.2018.07.001 (2018).
Soleimani, A., Hosseini, S. M., Massah Bavani, A. R., Jafari, M. & Francaviglia, R. Influence of land use and land cover change on soil organic carbon and microbial activity in the forests of northern Iran. CATENA 177, 227–237. https://doi.org/10.1016/j.catena.2019.02.018 (2019).
Devi, N. B., Singh, L. I., Yadava, P. S. & Khan, M. R. Shift in microbial biomass, soil and microbial stoichiometry in different land uses of Northeast India. Microbe 3, 100085. https://doi.org/10.1016/j.microb.2024.100085 (2024).
Devi, N. B. & Yadava, P. S. Seasonal dynamics in soil microbial biomass C, N and P in a mixed-oak forest ecosystem of Manipur, North-east India. Appl. Soil Ecol. 31, 220–227. https://doi.org/10.1016/j.apsoil.2005.05.005 (2006).
Dindaroglu, T., Boran, B., Babur, E. & Menshov, O. Long-term temporal variation of land use transition on soil carbon stocks in mediterranean karst ecosystems. Forestist 74, 94–101 (2024).
Rosenzweig, S. T., Carson, M. A., Baer, S. G. & Blair, J. M. Changes in soil properties, microbial biomass, and fluxes of C and N in soil following post-agricultural grassland restoration. Appl. Soil. Ecol. 100, 186–194. https://doi.org/10.1016/j.apsoil.2016.01.001 (2016).
Lee, Z. M. & Schmidt, T. M. Bacterial growth efficiency varies in soils under different land management practices. Soil Biol. Biochem. 69, 282–290. https://doi.org/10.1016/j.soilbio.2013.11.012 (2014).
Sinsabaugh, R. L. et al. Stoichiometry of microbial carbon use efficiency in soils. Ecol. Monogr. 86, 172–189. https://doi.org/10.1890/15-2110.1 (2016).
Kara, O., Babur, E., Altun, L. & Seyis, M. Effects of afforestation on microbial biomass C and respiration in eroded soils of Turkey. J. Sustain. For. 35, 385–396 (2016).
Babur, E. & Dindaroglu, T. in Environmental Factors Affecting Human Health (ed Uher Ivan) Ch. 7 (IntechOpen, 2020).
Yılmaz, Y. & Gürer, Ö. The geology and evaluation of Misis-Andırın Belt, Aroun Andırın (Kahramanmaraş). Turkish J. Earth Sci. 5(3), 39–55 (1996).
Anonymous. Kahramanmaraş Onikişubat Orman İşletme Müdürlügü, Başkonuş Orman Amenajman Planı. Kahramanmraş Orman Bölge Müdürlüğü. Orman Genel Müdürlüğü (2014–2033). (2012).
Service, N. R. C. & Department, A. Keys to soil taxonomy: 2010. (Government Printing Office, 2010).
Babur, E. et al. Studying soil erosion by evaluating changes in physico-chemical properties of soils under different land-use types. J. Saudi Soc. Agric. Sci. 20, 190–197. https://doi.org/10.1016/j.jssas.2021.01.005 (2021).
Horwath, W. R. & Paul, E. A. Microbial biomass. Methods Soil Anal. 1(5), 753–773 (1994).
Brookes, P. C., Landman, A., Pruden, G. & Jenkinson, D. S. Chloroform fumigation and the release of soil nitrogen: A rapid direct extraction method to measure microbial biomass nitrogen in soil. Soil Biol. Biochem. 17, 837–842. https://doi.org/10.1016/0038-0717(85)90144-0 (1985).
Vance, E. D., Brookes, P. C. & Jenkinson, D. S. An extraction method for measuring soil microbial biomass C. Soil Biol. Biochem. 19, 703–707. https://doi.org/10.1016/0038-0717(87)90052-6 (1987).
Anderson, T.-H. & Domsch, K. H. The metabolic quotient for CO2 (qCO2) as a specific activity parameter to assess the effects of environmental conditions, such as ph, on the microbial biomass of forest soils. Soil Biol. Biochem. 25, 393–395. https://doi.org/10.1016/0038-0717(93)90140-7 (1993).
Anderson, J. M. & Ingram, J. S. Tropical soil biology and fertility: A handbook of methods. Soil Sci. 157, 265 (1994).
Alef, K. in Methods in applied soil microbiology and biochemistry Vol. 214 222 (Academic Press London, 1995).
Anderson, T.-H. & Domsch, K. Application of eco-physiological quotients (qCO2 and qD) on microbial biomasses from soils of different cropping histories. Soil Biol. Biochem. 22, 251–255 (1990).
Winding, A., Hund-Rinke, K. & Rutgers, M. The use of microorganisms in ecological soil classification and assessment concepts. Ecotoxicol. Environ. Saf. 62, 230–248. https://doi.org/10.1016/j.ecoenv.2005.03.026 (2005).
Dumontet, S., Dinel, H., Scopa, A., Mazzatura, A. & Saracino, A. Post-fire soil microbial biomass and nutrient content of a pine forest soil from a dunal Mediterranean environment. Soil Biol. Biochem. 28, 1467–1475. https://doi.org/10.1016/S0038-0717(96)00160-5 (1996).
Joergensen, R. G. & Scheu, S. Response of soil microorganisms to the addition of carbon, nitrogen and phosphorus in a forest Rendzina. Soil Biol. Biochem. 31, 859–866 (1999).
Chen, C. R., Condron, L. M., Davis, M. R. & Sherlock, R. R. Effects of afforestation on phosphorus dynamics and biological properties in a New Zealand grassland soil. Plant Soil 220, 151–163. https://doi.org/10.1023/A:1004712401721 (2000).
Vidyanagar, V. Seasonal and temporal variation in soil microbial biomass C, N and P in different types land uses of dry deciduous forest ecosystem of Udaipur, Rajasthan, Western India. Appl. Ecol. Environ. Res. 8, 377–390 (2010).
Díaz-Raviña, M., Acea, M. J. & Carballas, T. Seasonal changes in microbial biomass and nutrient flush in forest soils. Biol. Fertil. Soils 19, 220–226. https://doi.org/10.1007/BF00336163 (1995).
Arunachalam, A. & Arunachalam, K. Influence of gap size and soil properties on microbial biomass in a subtropical humid forest of north-east India. Plant Soil 223, 187–195 (2000).
Vm, T. & Marcos, M. L. F. Microbial biomass and activity in a forest soil under different tree species. Electron J. Environ. Agric. Food Chem. 8(9), 878–887 (2009).
White, D. L., Haines, B. L. & Boring, L. R. Litter decomposition in southern Appalachian black locust and pine–hardwood stands: Litter quality and nitrogen dynamics. Can. J. For. Res. 18, 54–63. https://doi.org/10.1139/x88-009 (1988).
Anderson, T.-H. Microbial eco-physiological indicators to asses soil quality. Agr. Ecosyst. Environ. 98, 285–293 (2003).
Tian, Y. et al. Microbial biomass and activity along a natural pH gradient in forest soils in a karst region of the upper Yangtze River. China. J. For. Res. 13, 205–214. https://doi.org/10.1007/s10310-008-0073-9 (2008).
Jenkinson, D. Determination of microbial biomass carbon and nitrogen in soil. Advances in nitrogen cycling in agricultural systems. Ed. JR Wilson. Wallingford: CABI, 368–386 (1988).
Tang, X.-L. et al. Dependence of soil respiration on soil temperature and soil moisture in successional forests in southern China. J. Integr. Plant Biol. 48, 654–663. https://doi.org/10.1111/j.1744-7909.2006.00263.x (2006).
Chen, C. R., Condron, L. M., Davis, M. R. & Sherlock, R. R. Seasonal changes in soil phosphorus and associated microbial properties under adjacent grassland and forest in New Zealand. For. Ecol. Manage. 177, 539–557. https://doi.org/10.1016/S0378-1127(02)00450-4 (2003).
Bolat, I. Seasonal changes in microbial biomass carbon (cmic), nitrogen (nmic), phosphorus (pmic), and microbial respiration of forest floor and top soil under beech, fir and mixed fir-beech forest stands [thesis]. Graduate School of Natural and Applied Sciences, Department of Forestry Engineering. Turkey: Bartın University (in Turkish) (2011).
Jenkinson, D. S. & Ladd, J. Microbial biomass in soil: measurement and turnover. Soil Biochem. 5, 415–471 (1981).
Bauhus, J. & Paré, D. Effects of tree species, stand age and soil type on soil microbial biomass and its activity in a southern boreal forest. Soil Biol. Biochem. 30, 1077–1089. https://doi.org/10.1016/S0038-0717(97)00213-7 (1998).
Bauhus, J. & Khanna, P. The significance of microbial biomass in forest soils. Going underground-ecological studies in forest soils. Trivandrum, India: Research Signpost, 77–110 (1999).
Babur, E. et al. Wattle fencing improved soil aggregate stability, organic carbon stocks and biochemical quality by restoring highly eroded mountain region soil. J. Environ. Manage. 288, 112489. https://doi.org/10.1016/j.jenvman.2021.112489 (2021).
Joergensen, R. G., Anderson, T. H. & Wolters, V. Carbon and nitrogen relationships in the microbial biomass of soils in beech (Fagus sylvatica L.) forests. Biol. Fertility Soils 19, 141–147. https://doi.org/10.1007/BF00336150 (1995).
Chen, T.-H., Chiu, C.-Y. & Tian, G. Seasonal dynamics of soil microbial biomass in coastal sand dune forest. Pedobiologia 49, 645–653. https://doi.org/10.1016/j.pedobi.2005.06.005 (2005).
Khan, K. S. & Joergensen, R. G. Microbial C, N, and P relationships in moisture-stressed soils of Potohar, Pakistan. J. Plant Nutr. Soil Sci. 169, 494–500. https://doi.org/10.1002/jpln.200521904 (2006).
Sharma, P., Rai, S. C., Sharma, R. & Sharma, E. Effects of land-use change on soil microbial C, N and P in a Himalayan watershed. Pedobiologia 48, 83–92 (2004).
Lepcha, N. T. & Devi, N. B. Effect of land use, season, and soil depth on soil microbial biomass carbon of Eastern Himalayas. Ecol. Process. 9, 1–14 (2020).
Author information
Authors and Affiliations
Contributions
E.B.: Conceptualization, Investigation, Methodology, Data curation, Software, Visualization, Writing—Original, Draft, Writing—review and editing. E.O.: Investigation, Normal analysis, Methodology, Data collection, Software, Writing—Original Draft, Writing—review and editing. O.S.U.: Software, Validation, Methodology, Visualization, Software, Writing—Original, Draft, Writing—review and editing.
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflicts of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Babur, E., Ozlu, E. & Uslu, O.S. Soil respiration, microbial biomass, and stoichiometry within riparian buffers and adjacent land use. Sci Rep 15, 40445 (2025). https://doi.org/10.1038/s41598-025-23152-2
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-23152-2