Abstract
The current experiment intended to investigate the plant growth-promoting effects of the bacterial endophytes, identified from Moringa oleifera, under salinity stress. These endophytic isolates, Serratia marcescens (KR-27), Klebsiella aerogenes (KL-4), and Lelliottia amnigena (KS-7) were checked for the phosphate solubilization activity, production of indole acetic acid (IAA) and siderophores, employing the quantitative calorimetric techniques. Amongst the tested strains, KS-7 produced substantial amount of IAA while the isolate KR-27 exhibit strong siderophores production and effective phosphate solubilization. Secondly, the effect of endophytic bacterial strains was checked on the rice plants’ morphological parameters in vitro. As a result, the endophytes slightly enhanced the growth indices and fresh/dry biomass of the rice plantlets. Furthermore, the biochemical characterization of the rice plants using standard calorimetric assays revealed that the endophytes inoculation substantially enhanced the level of phytohormones; including IAA, gibberellic acid (GA3), and salicylic acid (SA), while reducing the level of abscisic acid (ABA). The photosynthetic pigments, including chlorophyll a, b, and carotenoids are also elevated, thereby exhibiting the plant growth-promoting effect of the rice plants. Additionally, among the various metabolites, phenolic acid and flavonoids are substantially enhanced, while low level of proline indicates the efficiency of endophytes in the presence of salinity stress. The basic mechanism of endophytes in mitigating salt stress is the regulation of the imbalanced ionic concentrations. Besides, the introduction of the bacterial strains to the growth culture media in the presence of 200 mM NaCl induced the enzymatic antioxidant system of the rice plants. The strains; KS-7 and KR-27 effectively amplified the enzymatic activities of catalase (CAT), ascorbate peroxidase (APX), and glutathione. All these outcomes recommend the applications of newly identified strains of M. oleifera in mitigating salinity stress in sustainable agricultural practices.
Similar content being viewed by others
Introduction
Plants are considered major source of food for human which contain almost all the dietary nutrients required for their normal physiological functions. Due to rapid increase in population and subsequent urbanization, a twofold negative impact has been noted on arable land including the reduction in size and its exposure to numerous biotic and abiotic stress factors1,2. On the flip-side, the increasing world population demands for more food and more crops production. However, the crops productivity is critically reduced by the biotic and abiotic factors such as drought, salinity, high temperature, and contamination of the lands. Therefore, it will be a great challenge in the near future to feed the large swath of population. To address the food insecurities, current agricultural practices which rely mainly on the applications of synthetic fertilizers and pesticides, would need to be replaced by employing sustainable agricultural practices. These include the use of transgenic technology and application of beneficial microorganisms which are both environment- and human-friendly3,4,5,6.
Among the various abiotic factors, salinity affects approximately eight hundred million hectares of the world’s arable land, severely reducing the agronomic attributes of the host in terms of osmotic stress and imbalance the nutrient ratio. Salinity also causes a reduced water uptake and ultimately led to plant cells death and injury7,8,9. Symbiotic microorganisms such as bacteria and fungi which colonize the plant tissue for at least one life cycle, without any negative impact on their host plant, are known as endophytic microorganism10. Plants and their respective endophytes develop several defense mechanisms such as production of EPS, proline accumulation, deaminase activities, siderophores formation, IAA production and phosphate solubilization against salinity stress11,12,13,14,15. During the symbiotic association, the endophytes produce various plant growth promoting phytohormones, defensive metabolites, exopolysaccharides (EPS) and siderophores while the host plants provide shelter and organic compounds to their symbiotic partners. Almost all the plant species, studied so far, contain at least one endophyte16,17.
Rice is consumed as staple food by more than half of the world population and is considered as important cereal crop18. According to the FAO20, the consumption of rice is increasing by 520 million annually21. Susceptibility of the rice plants to various environmental stresses and the increasing world population demand for increased production of rice in the near future22. Annually, a number of environmental factors reduced the rice production by 15–50% all over the world23. Therefore, advanced and innovative agriculture practices must need to be manipulated for enhancing the production of rice in a sustainable way.
Medicinal plants-associated endophytic microorganisms are considered as a rich source of agriculturally and pharmaceutically important secondary metabolites with strong antioxidant, anti-microbial and plant growth promoting potential24. Moringa oleifera Lam. belongs to the Moringaceae family, is a nutritionally-rich and medicinally important plant which is used all over the world for the treatment of various diseases and nutritional deficiencies25,26. Although this plant is nutritionally rich, yet its bacterial endophytes and their applications for agriculture and pharmaceutical industries is still an unexplored area of research. Previously the study conducted by27, isolated antifungal producing endophytic fungi Nigrospora sp for the first time from M. oleifera. Similarly, Dhanalakshmi et al.28 isolated five endophytic fungal strains, including Aspergillus nidulans, Alternaria sp., Exophilia sp, Penicillum sp and Bipolaris sp from the stem and leaves of Moringa. Two bacterial strains, Pantoea agglomerans and Pectobacterium cypripedii, responsible for plant growth promoting activity, were characterized from the roots of Moringa29. However, more research is needed to explore the rich microflora of this miracle tree.
According to recent studies, species related to genus Klebsiella attracted attention due to their plant growth promotion activities which could be used as important growth stimulator in sustainable agriculture practices in future. Although some of the species of this genus such as K. pneumonia, are opportunistic pathogens of Human and animals30,31. In the case of plants, majorities of the species of this genus, such as Klebsiella sp. SBP-832 and Klebsiella sp. D5A33 promote plant growth by enhancing nutrient availability in various biotic and abiotic stress conditions and act as nonpathogenic. The study conducted by34 showed that the plant growth promoting endophytic bacterial strain K. aerogenes HGG15 did not show any side effect and pathogenicity on silkworm. Therefore, we intended to determine the plant growth promoting potential of the Moringa’ underexplored endophytic bacterial strains Serratia marcescens (KR-27), Klebsiella aerogenes (KL-4), and Lelliottia amnigena (KS-7) on rice plant under salt stress, and to the best of our knowledge, this is the first study to explore the potential of these strains under salinity stress in rice plants.
Material and methods
Collection of plant material and microbial strains
The salt-susceptible rice variety, IR-36 was used in the current experiments. Glycerol stock of pre-identified EPS-producing endophytic bacterial strains; Serratia marcescens (KR-27), Klebsiella aerogenes (KL-4) and Lelliottia amnigena (KS-7) with NCBI accession no PP892069, PP854706 and PQ218332, respectively, isolated from M. oleifera, were obtained from the Plant Biotechnology Laboratory Abdul Wali Khan University Mardan, KP, Pakistan35.
Salt tolerance assay of the selected bacterial endophytes
To assess the salinity tolerance of the selected bacterial endophytes, the nutrient broth were supplemented with 50, 100 and 200 mM NaCl and incubated at 28 ± 2 °C for 72 h while the growth of the bacterial isolates were checked after every 6 h using spectrophotometer for growth curve at 600 nm. A salt free-medium was used as a control and each experiment were recorded in triplicate for statistical analysis36.
Assessment of the selected endophytes for plant growth-promoting traits
The selected bacterial endophytes were screened out for the phosphate solubilization, IAA and siderophores production by colorimetric methods. The methods described by Patten and Glick37 were used to quantify IAA produced by selected bacterial endophytes. To determine the quantity of siderophores produced by endophytic bacterial strain, the modified micro-plate method described by Arora and Verma38 was used. While the method described by De et al.39 was employed to determine the ability of the selected strains to solubilize the inorganic phosphate using the Pikovskaya’s (PVK) broth containing tricalcium phosphate as a source of inorganic phosphate (Pi) (see detailed protocols in supplementary file).
Preparation of the bacterial suspension
Rice seedlings were inoculated with the bacterial suspensions at the second week of the culturing. The cultures of each bacterial strain were inoculated in nutrient broth until the OD reached to 0.8. After that, each bacterial culture was centrifuged at 4 ºC for ten min at 8000 rpm, while separating the cell pellet in a 0.03 M MgSO4. The absorbance was adjusted to OD600 0.8 (cell count ~ 1.0 × 108 CFU/mL). Finally, 10 mL of each bacterial suspension was applied to the root zone of rice seedlings. The un-inoculated control plants were treated with only 0.03 M MgSO440.
Sterilization of the rice seeds
Salt-susceptible rice seeds (IR-36) were thoroughly rinsed with tap water to remove any surface dust followed by treating with 70% ethanol for 1 min and 2% sodium hypochlorite for 25 min (Gamborg and Phillips 2013). Final washing (5 times) of the seeds was carried out with sterilized distilled water to remove any traces of the chemical disinfectants.
Assessment of the growth condition in pot experiment
To ascertain the potential of the bacterial strains in promoting plant growth in pots, the uniformly germinated plants in sterilized petri plates were transferred to plastic pots containing 500 g autoclaved soil composed of 1:1:1 peat, sand and perlite after six days of the germination41. The pots were designated as,
-
1.
Control = plants with no salt no endophyte
-
2.
Treatment 1 = 200 mM NaCl solution was applied to rice seedling
-
3.
Treatment 2 = 10 mL of bacterial suspension of isolate KR-27 was applied to the root zone of rice seedlings
-
4.
Treatment 3 = 10 mL of bacterial suspension of isolate KL-4 was applied to the root zone of rice seedlings
-
5.
Treatment 4 = 10 mL of bacterial suspension of isolate KS-7 was applied to the root zone of rice seedlings
-
6.
Treatment 5 = 10 mL of bacterial suspension of isolate KR-27 was applied to the root zone of rice seedlings in the presence of 200 mM NaCl.
-
7.
Treatment 6 = 10 mL of bacterial suspension of isolate KL-4 was applied to the root zone of rice seedlings in the presence of 200 mM salt, and
-
8.
Treatment 7 = 10 mL of bacterial suspension of isolate KS-7 was applied to the root zone of rice seedlings in the presence of 200 mM salt.
All the inoculated plants and control cultures were incubated in growth chamber at 28 ± 2 ºC in the presence of 12 h light with intensity of approximately 250 μmol/m2 s, and 70% humidity. In case of plants exposed to salt stress, a 50 mL of 200 Mm NaCl was added to the plants after 7th day and then left for about two weeks to recuperate. All the plants were consistently irrigated for one month and the data was collected for further analysis.
Morphological characterization of the rice plants
Data for root and shoot length (in cm), number of roots per seedling and plant fresh and dry weight (in grams) was recorded from three replicates of each treatment after a month of transplantation to pots. To determine the dry weight, the plants were washed with water to remove soil particles and then dried in hot air oven at 60 °C until the plants gained a stable weight.
Biochemical, phytohormonal, elemental, and antioxidant analyses of the rice plants
To assess the physiological and biochemical responses of rice plants under salt stress and endophytic bacterial inoculation, various parameters were evaluated. The photosynthetic pigments (chlorophyll and carotenoids) were quantified using the modified method of Maclachlan and Zalik42, while proline, total phenolics, and flavonoids were assessed following the protocols by Ábrahám et al. (2010), Lamuela-Raventós (2018), and Maldonado-Garcia et al.43, respectively. Quantification of the key phytohormones was performed to evaluate plant hormonal responses. The phytohormones including IAA, SA, GA, and ABA were quantified using methods by Manna et al.44, Warrier et al.45, Dumale et al.46, and Materán et al.47. The elemental composition was analyzed using atomic absorption spectrophotometry (Shimadzu AA-670) after acid digestion48. The enzymatic antioxidant activities for Antioxidant enzyme activities, including catalase (CAT), glutathione reductase (GR), and ascorbate peroxidase (APX), were assessed via colorimetric assays described by Nalina et al.49, Carlberg and Mannervik50, and Asada51, while the lipid peroxidation was evaluated by estimating malondialdehyde (MDA) content employing a modified method of Del Buono et al.52 (see supplementary file for detailed procedures and protocols).
Statistical analysis
All the experiments were conducted in three replicates and the recorded data were analyzed using version 6.0 of SPSS software. One way analysis of variance (ANOVA) followed by Duncan’s multiple range test at a 5% significance level (p < 0.05) were used to evaluate variations amongst different treatment groups and the results were represented in graphs and table as mean ± standard deviation (SD) along with letter of significant differences. The data were graphically represented using Graph Pad Prism (version 8.0.1, build 244).
Results
The data in Fig. 1 represent the growth of selected bacterial strains on nutrient agar and broth media. All the isolates were further screened out for plant growth promotion assay and salinity tolerance.
Growth of bacterial strains (KR-27), S. marcescens, (KL-4) K. aerogenes and (KS-7) L. amnigena on nutrient agar and nutrient broth media.
Growth curve of selected bacterial endophytes
All the selected bacterial isolates were found resistant to 200 mM of NaCl stress. A dose dependent increase in the growth was recorded up to 200 mM which was severely impacted beyond 200 mM. In the same way a positive correlation was recorded with time and decline was recorded in the growth after 48 h of exposure to various levels of salt stress (Fig. 2).
OD (600 nm) of bacterial growth treated with 100, 200 and 300 mM NaCl, (A) S. marecescens, (B) K. aerogenes and (C) L. amnigena.
Plant growth-promoting potential of the bacterial endophytes in vitro
The data in Table 1 represents the values of plant growth-promoting metabolites produced by the endophytes isolated from M. oleifera. All the tested bacterial strains were positive for IAA production (Fig. 3). Amongst the three strains, L. amnigena produced 15.21 µg/ml IAA followed by S. marecescens and K. aerogenes which produced 11.43 and 8.21 µg/ml of IAA, respectively. Based on quantitative determination of the siderophores production, the bacterial isolate S. marcescens produced highest amount of siderophores (59%) followed by L. amnigena with percent production of 30.87% and K. aerogenes (15.21%). The bacterial isolates were ascertained for phosphate solubilization index. As compared to other tested isolates, the isolate KR-27 solubilized inorganic phosphate more efficiently at the rate of 22.45 µg/mL of the Pi.
In vitro plant growth promoting traits of the selected endophytic bacterial strains (A) indole acetic acid production, (B) Siderophores production, (C) Phosphate solubilization.
Morphological growth indices of the rice plants
Table 2 consists of the values for growth parameters of the rice plants, recorded after a month of endophytes inoculation. The overall effect of the bacterial endophytes is found satisfactory in inducing the growth of rice plants in normal and saline environments.
Effect of endophytes on length of shoots and roots in rice plants
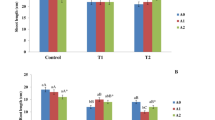
In the present investigation, we found that in comparison to control plants, the rice plants treated with 200 mM NaCl exhibited a considerable reduction in root and shoot length. The salinized plant’s root length was measured as 7.6 cm, but in the control plants root length were 10.83 cm. However, in both normal and saline conditions, the rice plants treated with the bacterial strains S. marecescens and L. amnigena dramatically increased the root length (11.89 and 11.48 cm), respectively. When exposed to salt, the rice plants inoculated with L. amnigena showed the highest root length value (12.73 cm). In case of shoot length, salinity stress reduced it to 29.06 cm from 33.31 cm for control plants, while the plant treated with bacterial strains S. marecescens and L. amnigena significantly enhanced the shoot length to 35.53 and 35.97 cm in the control and 34.73 and 35.41 cm in saline condition, respectively (Table 2).
Effect of endophytes on number of roots and shoots in rice plants
In comparison to the control plants where mean value for numbers of roots were recorded as 16, the salt stress dramatically decreased the number of roots to 11 per plantlet. However, in both normal and saline environment, the administration of endophytic bacteria resulted in an increase in the number of roots per plantlet. Every bacterial endophyte produced more roots and plants. In a similar way, salt stress was found to reduce the number of shoots, whereas the endophytes application enhanced the number of shoots per plant (Table 2).
Effect of endophytic bacteria on plant biomass
Plant fresh and dry biomass is negatively impacted by salinity when compared to non-salinized plants. In case of the control plants, fresh and dry mass were 0.39 and 0.11 g, respectively, whereas in salinized plants, it was 0.29 and 0.07 g. On the other hand, the rice plants inoculated with bacterial strains, S. marecescens and L. amnigena significantly increased the fresh and dry biomass to 0.44 and 0.46 g, while the strain K. aerogenes slightly reduced shoot fresh biomass. In salinity stress, the isolates L. amnigena and S. marecescens increased plant fresh biomass to 0.32 and 0.33 g in comparison to the non-inoculated plants.
Effect of endophytic bacteria on photosynthetic pigments
The data in Table 3 represents the photosynthetic pigments of rice plants inoculated with bacterial isolates in normal and salinized conditions. Normal growth and development of plants depends on photosynthetic pigments and are important growth indictors for plant health. The photosynthetic potential of plants is adversely affected by salinity and thus the productivity of crop plants can be reduced. Likewise, in the current experiment, chlorophyll a and b were significantly decreased by the salt stress; however, the application of endophytic bacterial strains reversed the inhibition of photosynthetic pigments formation in the presence of salt stress. In the control plants, the photosynthetic pigments were found highest in plant inoculated with L. amnigena followed by S. marecescens. In the same condition, the chlorophyll a was found as 2.01 µg/g of fresh weight of rice plant while the plant exposed to salt stress showed 0.22 µg/g chlorophyll a. The plant inoculated with S. marecescens, K. aerogenes and L. amnigena in control environment showed 2.2, 1.92 and 2.32 µg/g of fresh mass, respectively. In salt stress, the application of isolates, S. marecescens, K. aerogenes and L. amnigena enhanced the chlorophyll a contents to 1.23, 1.12 and 1.56 µg/g as compared to the negative control plant which was 0.22 µg/g. In case of chlorophyll b, similar pattern was observed in the plants after the application of endophytic bacterial strain in salt stress as well in non-salinized plants. Carotenoids in the rice plants were considerably elevated by all bacterial isolates. L. amnigena and K. aerogenes were found to have carotenoids at 1.23 and 1.18 µg/g, respectively, whereas the plants inoculated with the bacterial isolate S. marecescens had the highest value, 1.29 µg/g.
Effect of the endophytes on phytohormones level of rice plants
The growth and development of roots and shoots depend on IAA production. In the current experiment, salt stress inhibited the production of IAA and their mean value was found as 2.03 µg/g. The inoculation of endophytic bacterial strains enhanced the concentration of IAA in both saline and in normal conditions. Rice plants inoculated with bacterial strains; S. marecescens, K. aerogenes and L. amnigena without salt stress, produced 4.21 ± 0.7, 3.1 ± 0.8 and 4.1 ± 0.2 µg/g of IAA, respectively. The rice plants under salt stress produced 3.3 ± 0.1, 2.9 ± 0.9 and 4.6 µg/g IAA with the strains S. marecescens, K. aerogenes and L. amnigena, respectively. Under salinity stress, the endophytic bacteria enhanced the plant growth and GA3 production. Comparing the bacterized rice plants to the negative control plants, which were merely exposed to 200 mM salt, the bacterized plants all generated GA3 at levels between 1.9 and 2.6 µg/g. While under normal conditions, the L. amnigena, K. aerogenes, and S. marecescens produced 2.2, 2.3, and 2.5 µg/g GA3, respectively. However, during the salt stress, the KS-7 substantially enhanced the GA3 level to 4.6 µg/g. The level of SA was drastically reduced by salinity stress to 7.6 µg/g as compared to the control plants (27.9 µg/g). While the production of SA in rice plants, after the addition of K. aerogenes was calculated to be 3.4 µg/g in normal conditions, and 4.2 µg/g under saline conditions. The content of SA was markedly elevated by endophytic bacteria L. amnigena among the all strains in the presence of salt stress (Table 3).
When rice plants are exposed to salt, their growth is inhibited as a decrease was recorded in the biomass of their roots and shoots, a reduction in photosynthetic activity, the production of ROS, and an increase in endogenous ABA levels. In the rice plant exposed to 200 mM NaCl, the ABA was found increased from 20.3 (in control) to 31.6 µg/g. However, the rice plants inoculated with bacterial strains in normal condition did not affect the ABA content but in salinity stress the S. marecescens and L. amnigena considerably reduced the level of ABA (Table 3).
Effect of endophytes inoculation on rice plants metabolites
Proline levels in the plants treated with 200 mM salt were increased (to 3.5 µg/g) in comparison to the control plants (1.3 µg/g). The bacterial isolates have no significant effect on proline contents in control condition while in saline condition the isolate S. marecescens and L. amnigena significantly reduced the amount of total proline contents. The rice plants’ total phenolic acid content increased dramatically in response to salinity, reaching 61.44 µg/g compared to 44.94 µg/g of the control. The total phenolic compounds under normal conditions were considerably increased by the bacterial endophytes, S. marecescens and L. amnigena, which were recorded as 67 and 69 µg/g, respectively. Under normal conditions, the total flavonoid concentration was considerably elevated by the isolates, S. marecescens and L. amnigena. The total flavonoid concentrations of the plants treated with salinity were drastically reduced to 0.6 µg/g, whereas the total flavonoid contents in the control plants were 5.11 µg/g. The rice plants inoculated with S. marecescens and L. amnigena resulted in the greatest total flavonoid levels, measuring 7.30 and 5.177 µg/g, respectively (Table 3).
Effect of endophytes on ionic concentrations of the rice plants
As compared to the control, the non-salinized plants and the plants treated with 200 Mm NaCl showed a considerable rise in their Na+ concentration. The plant treated with 200 mM salt has a substantially higher Na+ ion concentration (22.33 mg/g) than the control plant, which has a concentration of 12.6 mg/g of the rice plants dry weight. The concentration of Na+ ions under salinity stress was considerably decreased by the endophytic bacterial isolates, L. amnigena and S. marecescens. When the rice plants treated with the S. marecescens and L. amnigena and specified doses of NaCl, the concentration of Na+ ions decreased to 12.05 and 17.33 mg/g, respectively (Fig. 4A). Salinity dramatically decreased the K+ ion concentration in the plant treated with 200 Mm salt, from 14.6 mg/g in the control to 10.33 mg/g in the treated plant. The isolate KS-7 considerably increased the K+ ion concentrations in plants under both normal and saline stress conditions when compared to other endophytic bacterial treatments. The rice plant treated with salt and the isolate L. amnigena had the highest K+ ion content, measuring 15.0 mg/g (Fig. 4C). When rice plants were exposed to salinity stress, their iron concentration was considerably lower than that of non-salinized plants. In contrast to the control plants, which has 6.0 mg/g of Fe+, the plant subjected to salinity was found with 5 mg/g. Rice plants’ Fe+ accumulation was markedly increased by S. marecescens under both normal and salt stress conditions (Fig. 4B).
Effect of endophytic bacterial strains on morphological parameters of rice plants in control and saline condition. (A) non-salinized control plant, (B) plant inoculated with S. marecescens, (C) plant inoculated with K. aerogenes, (D) plant inoculated with L. amnigena, (E) non inoculated salinized plant, (F) salinized plant inoculated with S. marecescens, (G) salinized plant inoculated with K. aerogenes, (H) salinized plant inoculated with L. amnigena.
Effect of endophytes on MDA contents and enzymatic antioxidant system of the rice plants
Salinity has a major impact on the plant’s antioxidant system. MDA contents was significantly increased in the rice plants exposed to salt stress and was found 68.21 nM/g as compared to the control plants which resulted in 12.32 nM/g (Fig. 5D). However, the application of endophytic bacterial strains significantly reduced the MDA contents in rice plant exposed to salt stress and hence decreased the lipid peroxidation of membrane by changing the host antioxidant system. In comparison to the control plants, a 200 mM NaCl treatment considerably increased the catalase activity up to 19.1 mM. While all of the chosen endophytes increased the catalases in salinity, with the exception of strain K. aerogenes, which decreased the activity (Fig. 5A). Similar pattern was seen in Ascorbate peroxidase, which demonstrated an elevated activity in the rice plants treated with salt and endophytic bacterial strains. Ascorbate peroxidase level in the salinized rice plants was 27.52 µM. After addition of the isolates; S. marecescens and L. amnigena the enzymatic activity was considerably increased to 32.86 and 28.08 µM, respectively (Fig. 5B). The salt concentration substantially lowered glutathione production to 12.45 µg/g compared to the control, which was 17.05 µg/g. In contrast, endophyte treatment greatly increased glutathione production. The strains, L. amnigena and S. marecescens increased the production of glutathione within different endophytes to 22.41 and 19.2 µg/g, respectively (Fig. 5C).
Effect of endophytic bacterial strains on ionic contents of Rice plant in control and salt stress. (A) Na+ concentration, (B) Fe+ Concentration, (C), K+ concentration.
Effect of endophytic bacterial strain on rice enzymatic antioxidant system and MDA contents in control and salt stress. (A) Catalase activities (B) Ascorbate peroxidases activities (C) Glutathione production, (D) MDA Contents.
Discussion
This study demonstrated the ability of the bacterial endophytes to generate plant growth-promoting metabolites such as IAA, siderophores, and phosphate solubilization ability. Previously, several endophytic bacteria are reported to produce auxin-like compounds and function as plant growth-promoting organisms, enhancing plant growth under various abiotic and biotic stresses53,54,55,56. Phytohormones, including auxin can control several processes like cell differentiation, stem extension, and elongation in plants, acting as stimulants for crop enhancement. In some recent findings by Qadir et al.57, it has been found that IAA has a significant role in stress mitigation. When plant sense stress, a significant amount of IAA is produced and in most cases it triggers the cellular division. In such conditions of rapid cellular division, plant uses each cell as a compartment to divide the stress in order to mitigate it. A similar pattern of IAA production was also recorded in the current experiment where it has been found that the bacterial strain L. amnigena resulted in the highest rate of IAA production as compared to other strains. IAA-producing endophytic bacterial strains are previously reported to regulate the plant growth by promoting cell cleavage, differentiation, and elongation under different conditions58. Recently59, demonstrated the isolation of the endophytic strain L. amnigena J29 from the ginger plants with the ability to produce the IAA. Additionally, the two endophytes, S. marcescens and K. aerogenes were also reported to produce significant amounts of IAA60. In the current findings, higher IAA production was attributed to host stress resilience against salinity stress (Fig. 6).
Similarly, siderophores produced by the endophytic bacterial strains have a direct effect on plant growth by enhancing the acquisition of iron. In the current scenario, it has been found that bacterial endophyte S. marcescens produced a significant amount of siderophores in order to get sufficient iron needed by the host instead of Na+ which induces cellular toxicity as plant exposed to higher levels of Na+, hence preventing the host plant from its direct negative impacts. On the other hand, the endophytic strains K. aerogenes and L. amnigenus produced relatively low amounts of siderophores. Previous research conducted by various scientists identified species of Serratia that can produce IAA and siderophores, which supports our study61,62. Furthermore, Barzanti et al.63 demonstrated to isolate a siderophores-producing endophytic bacterial strain from Alyssum bertolonii, while Sessitsch et al.64 identified the similar strain from potato plants. These reports strongly support our findings that bacterial endophytes could be a source of siderophores production for enhanced plant growth. In the current finding, the parallel production of the IAA and siderophore in significant quantities have multi-fold functions i.e., preventing host from excessive Na+ uptake, compartmentalization of the absorbed Na+ to lower the stressful impacts while helps the host plant to grow normally in harsh conditions. Apart from siderophores production and iron uptake, these strains were also capable to solubilize the inorganic phosphate which is far more crucial for normal plant growth and development65,66. Microorganisms found in the endosphere and rhizosphere of plants secrete various types of phosphatases, organic acids, siderophores, and hydroxyl groups, which work together to solubilize inorganic phosphorus into organic soluble phosphate, thus increasing its availability to the host plant67,68,69. It has been found that the S. marcescens and L. amnigena effectively solubilized inorganic phosphate present in the culture media. Siderophore production is one of the mechanisms by which these microorganisms solubilize inorganic phosphate. Our findings indicate that S. marcescens produced 56% siderophores, while L. amnigena produced 32%, indicating their role as phosphate solubilizing.
During the stressful conditions, plant and their associate microbes release a range of metabolite in order to tackle down the stress they are facing while maintaining their normal growth attributes. In such conditions, a higher GA3, SA and ABA were recorded in bacterial endophyte inoculated conditions as compared to the control plant which further suggests their efficacy in mitigating the stress in the host plants. IAA is known for promoting root growth and root surface area, and allows plants’ better access to nutrients in saline soils. Similarly, the role of GA and SA were also determined in mitigating the salt-stress. Gibberellic acid promotes stem growth through cell division, while salicylic acid (SA) acts as a stress regulator. Exposure to salinity encourages microbes to release excess SA, which is converted into SA-based metabolites like siderophores, aiding in iron acquisition and detoxifying heavy metals. SA also affects various organisms, suggesting that increased production by certain microbial strains could help plants cope with stress. It triggers the expression of genes related to stress resistance, including those for antioxidants and secondary metabolites70,71,72. Furthermore, bacterial endophytes can down-regulate abscisic acid (ABA) levels and increase glutathione production, enhancing the host plant’s ability to scavenge free radicals in saline conditions73.
In the current work, an increase in the production of total phenolic acid and flavonoids, in addition to a decrease in the proline content, has been observed. The upsurge in the levels of phenolic acids and flavonoids in the host plant also activate its defense system and mitigate the harsh environmental conditions. The phenolic and flavonoids are the direct ROS quenchers aiding in lowering the secondary oxidative stress due to salinity. These compounds also serve as chemical signals that microbes use to engage in a dialog for establishing symbiotic associations with the host roots. An increase in the proline contents during saline condition aids in lowering the toxicity of Na+ by acting as an osmolyte and direct ROS quencher. In the current instance, declines in the proline contents with the inoculation of bacterial endophyte suggest a potential alleviation of salinity stress in the host plant. Various organic and inorganic amendments are used to eradicate high concentration of Na + salt from soils but these methods have several limitations such as laborious, cost effective and cause environmental pollution74. The use of endophytic and rhizospheric plant growth promoting bacteria residing in the stressful environment minimized the negative effect of stresses by producing various secondary and primary metabolites, these metabolites regulate K + /Na + ratio by maintaining ionic transportation, solubilized nutrients like zinc, phosphorus and potassium and maintain the cellular osmotic potential75,76. Besides these microbes mitigate the salt stress by producing various enzymes such as proteases, oxidases ACC deaminase, siderophores and produced exopolysaccharides which restrict the uptake Na + ions uptake by plant roots77. Some of the plant growth promoting bacterial isolates solubilized potassium and make it available for the uptake of plants in salt stress.
The over production of ROS in plant cell due to salinity stress, damaged the membrane permeability owing to the oxidation of fatty acid present in the membrane oozing out of the essential ions and cause severe symptoms of oxidative stress which ultimately inhibited the growth and development of plants. When polyunsaturated fatty acid (present in the membrane) under goes the oxidation in the presence of ROS, it produces malondialdehyde or thiobarbituric acid (TBARS or MDA), which acts as an indicator for damage of plant tissue in the presence of stresses78,79,80,81. In the current work, it was also noticed that the rice plant exposed to salt stress increased the amount of MDA which indicate that the plants are in severe oxidative stress as compared to the non-salinized plant. Nonetheless, bacterial endophyte inoculated plants showed a significantly lower amount of the MDA production indicating lower stress due to the plant growth promoting and stress alleviating potential of the bacterial endophytes. An increased in K+/Na+ and decline in the MDA contents in the current experiment suggest that the inoculated bacterial endophytic strain, KR-27 and KS-7 counteract the negative effect of salt stress in rice plants. Similarly, the previous research conducted by Tewari and Arora82, Aizaz et al.83 showed that plant growth promoting endophytic bacterial strain enhanced the growth of soybean and sunflower in different level of salt stress.
In order to cope with the resultant oxidative stress due to higher salinity condition, host plant produce tremendous amount of antioxidant enzymes. These enzymes tackle down the excessive ROS. In the current scenario, the KS-7 and KR27 triggered the host plant to produce higher levels of the catalases, Ascorbate peroxidase and glutathione reductases which potentially aid the host plant to cope the excessive ROS produced as a result of salinity. A similar study was also recorded by Hasegawa et al.84 explaining the positive correlation salinity and enhanced activity of antioxidant defense system. Some other studies have shown that salinity increases the CAT activities of salt-resistant rice types while decreasing those of salt-sensitive rice varieties78,85,86. In crops such as soybean, the Ascorbate peroxidase was also found to be directly proportional to the salinity stress (Weisany et al. 2020). Our findings are in line with numerous researchers who have reported that rice and cotton are salt-tolerant due to APX activity in conjunction with CAT and POX activities87,88,89,90.
According to previous study, Rice seed inoculated with bacterial strain S. marcescens S-JS1 enhanced the IAA production, root and shoot length and biomass in host plant91. The study conducted by Parashar et al.92 demonstrated that endophytic bacterial strain L. amnigena isolated from Euphorbia prostrata significantly enhanced seed germination and plant growth physiological response in tomato and wheat plant. El-Akhdar et al.93 isolated salt tolerant rhizospheric bacterial strain Lelliottia amnigena MSR-M49 from wheat and investigate its plant growth promoting potential on wheat plant under salinity stress. Their finding demonstrates that inoculation of wheat plant with L. amnigena MSR-M49 significantly enhanced various morphological, biochemical and physiological parameters of plant under salinity stress.
Conclusion
From the current results and evidences, it was concluded that how endophytic bacterial strains of M. oleifera affected the rice plants in both saline and non-saline conditions in terms of stimulating plant development. It was found that S. marcescens, K. aerogenes and L. amnigena enhanced the growth attributes of rice plants exposed to salt stress by modulating and enhancing various metabolic pathways for the production of phytohormones, photosynthetic pigments, changing the antioxidant system of host plant and enhancing the absorption of macronutrients. Furthermore, our finding suggests that the bacterial isolates S. marcescens and L. amnigena could be used as potential bio fertilizers for the growth of economically important crops like maize, rice, wheat, tomato and potato in phosphate- and iron- deficient soils. Further research is needed to uncover the molecular mechanism, to evaluate the biosafety, and to assess the performance of these strain in large scale agriculture applications in abiotic stresses.
Data availability
All data are fully available and can be found within the manuscript.
References
Hu, G. et al. Mitigating the ripple effects of urbanization on farmland productivity and ecological security through inclusive urbanization strategies. Npj Urban Sustain. 5(1), 12. https://doi.org/10.1038/s42949-025-00199-8 (2025).
Nawaz, M. et al. A review of plants strategies to resist biotic and abiotic environmental stressors. Sci. Total Env. 900, 165832. https://doi.org/10.1016/j.scitotenv.2023.165832 (2023).
Baweja, P., Kumar, S. & Kumar, G. Fertilizers and pesticides: their impact on soil health and environment. In Soil Biology 265–285 (Springer International Publishing, 2020).
Fedoroff, N. V. & Cohen, J. E. Plants and population: is there time?. Proc. Natl. Acad. Sci. U. S. A. 96, 5903–5907 (1999).
Glick, B. R. Plant growth-promoting bacteria: mechanisms and applications. Scientifica (Cairo) 2012, 963401 (2012).
Hussain, S. S. (2019) Microbe-mediated tolerance in plants against biotic and abiotic stresses. In Microbial Interventions in Agriculture and Environment 173–217 (Springer, 2019).
Majeed, A. & Muhammad, Z. Salinity: A major agricultural problem—causes, impacts on crop productivity and management strategies. In Plant Abiotic Stress Tolerance 83–99 (Springer International Publishing, 2019).
Riaz, M. et al A comprehensive review on rice responses and tolerance to salt stress. In Advances in Rice Research for Abiotic Stress Tolerance (eds. Hasanuzzaman, M. et al.) 133–158 (Elsevier, 2019).
Shan, C. et al. Writers, readers, and erasers of N6-Methyladenosine (m6A) methylomes in oilseed rape: identification, molecular evolution, and expression profiling. BMC Plant Biol. 25(1), 147 (2025).
Omomowo, O. I. & Babalola, O. O. Bacterial and fungal endophytes: tiny giants with immense beneficial potential for plant growth and sustainable agricultural productivity. Microorganisms 7, 481 (2019).
Abdalla, M. A. & McGaw, L. J. Bioprospecting of South African plants as a unique resource for bioactive endophytic microbes. Front. Pharmacol. 9, 456 (2018).
Etesami, H. & Maheshwari, D. K. Use of plant growth promoting rhizobacteria (PGPRs) with multiple plant growth promoting traits in stress agriculture: action mechanisms and future prospects. Ecotoxicol. Environ. Saf. 156, 225–246 (2018).
Ha-Tran, D. M. et al. Roles of plant growth-promoting rhizobacteria (PGPR) in stimulating salinity stress defense in plants: a review. Int. J. Mol. Sci. 22, 3154 (2021).
Venugopalan, A. & Srivastava, S. Endophytes as in vitro production platforms of high value plant secondary metabolites. Biotechnol. Adv. 33, 873–887 (2015).
Verma, H. et al. The potential application of endophytes in management of stress from drought and salinity in crop plants. Microorganisms 9, 1729 (2021).
de Borges, W. et al. Endophytic fungi: natural products, enzymes and biotransformation reactions. Curr. Org. Chem. 13(12), 1137–1163 (2009).
Eid, A. M. et al. Harnessing bacterial endophytes for promotion of plant growth and biotechnological applications: an overview. Plants 10, 935 (2021).
Zubair, A., Zaib, S., Ul Ghani, N. N. & Mehboob, S. Zinc-fortified rice: approach to improve dietary zinc consumption and effect on human health. ACS Agric. Sci. Technol. 3, 926–938 (2023).
Weisany, W. et al. Changes in antioxidant enzymes activity and plant performance by salinity stress and zinc application in soybean (Glycine max L.). Plant Omics 5, 60–67 (2012).
Fao, F. A. O. S. T. A. T. Food and agriculture organization of the United Nations. Rome. http://faostat.fao.org. (2021).
Bin Rahman, A. N. M. R. & Zhang, J. Trends in rice research: 2030 and beyond. Food Energy Secur. https://doi.org/10.1002/fes3.390 (2022).
Salgotra, R. K. & Chauhan, B. S. Ecophysiological responses of rice (Oryza sativa L.) to drought and high temperature. Agronomy (Basel) 13, 1877 (2023).
Anami, B. S., Malvade, N. N. & Palaiah, S. Classification of yield affecting biotic and abiotic paddy crop stresses using field images. Inf. Process. Agric. 7, 272–285 (2020).
Tripathi, A., Pandey, P., Tripathi, S. N. & Kalra, A. Perspectives and potential applications of endophytic microorganisms in cultivation of medicinal and aromatic plants. Front. Plant Sci. 13, 985429 (2022).
Kashyap, P. et al. Recent advances in drumstick (Moringa oleifera) leaves bioactive compounds: composition, health benefits, bioaccessibility, and dietary applications. Antioxidants (Basel) 11, 402 (2022).
Saini, R. K., Sivanesan, I. & Keum, Y.-S. Phytochemicals of Moringa oleifera: a review of their nutritional, therapeutic and industrial significance. 3 Biotech 6, 203 (2016).
Zhao, J. H. et al. Bioactive secondary metabolites from Nigrospora sp. LLGLM003, an endophytic fungus of the medicinal plant Moringa oleifera Lam. World J. Microbiol. Biotechnol. 28, 2107–2112 (2012).
Dhanalakshmi, R., Umamaheswari, S., Sugandhi, P. & Arvind Prasanth, D. Biodiversity of the endophytic fungi isolated from Moringa oleifera of yercaud hills. Int. J. Pharmacuit. Sci. Res. 4, 1064–1068 (2013).
Fürnkranz, M., Müller, H. & Berg, G. Characterization of plant growth promoting bacteria from crops in Bolivia. J. Plant Dis. Prot. 116, 149–155 (2009).
Álvarez-Marín, R. et al. Clinical characteristics and outcome of bacteraemia caused by Enterobacter cloacae and Klebsiella aerogenes: more similarities than differences. J. Glob. Antimicrobial Resistance 25, 351–358. https://doi.org/10.1016/j.jgar.2021.04.008 (2021).
Li, L. & Huang, H. Risk factors of mortality in bloodstream infections caused by Klebsiella pneumonia: a single-center retrospective study in China. Medicine 96(35), e7924. https://doi.org/10.1097/MD.0000000000007924 (2017).
Singh, R. P., Jha, P. & Jha, P. N. The plant-growth-promoting bacterium Klebsiella sp. SBP-8 confers induced systemic tolerance in wheat (Triticum aestivum) under salt stress. J. Plant Physiol. 184, 57–67. https://doi.org/10.1016/j.jplph.2015.07.002 (2015).
Liu, W. et al. Whole genome analysis of halotolerant and alkalotolerant plant growth-promoting rhizobacterium Klebsiella sp. D5A. Sci. Rep. 6(1), 26710. https://doi.org/10.1038/srep26710 (2016).
Shang, N. et al. Biodegradation of malachite green by an endophytic bacterium Klebsiella aerogenes S27 involving a novel oxidoreductase. Appl. Microbiol. Biotechnol. 103(5), 2141–2153. https://doi.org/10.1007/s00253-018-09583-0 (2019).
Gul, K. et al. Isolation, biochemical characterization, and antimicrobial activities of EPS-producing bacterial endophytes from Moringa oleifera. Folia Microbiol. https://doi.org/10.1007/s12223-025-01312-1 (2025).
Kaur, M. & Karnwal, A. Screening of endophytic Bacteria from stress-tolerating plants for abiotic stress tolerance and plant growth-promoting properties: identification of potential strains for bioremediation and crop enhancement. J. Agric. Food Res. 14, 100723 (2023).
Patten, C. L. & Glick, B. R. Role of Pseudomonas putida indoleacetic acid in development of the host plant root system. Appl. Environ. Microbiol. 68, 3795–3801 (2002).
Arora, N. K. & Verma, M. Modified microplate method for rapid and efficient estimation of siderophore produced by bacteria. Biotech 7, 381 (2017).
De, P., Choudhury, A., Panda, P. & Hoque, A. Complementation of biochemical and physiological assays with functional PGPR based assays to screen potential Isolates. Int. J. Environ. Clim. Change 12, 640–647 (2022).
Fan, D., Subramanian, S. & Smith, D. L. Plant endophytes promote growth and alleviate salt stress in Arabidopsis thaliana. Sci. Rep. 10, 12740 (2020).
Bennett, G. & Lloyd, J. Seed Inoculation, Coating and Precision Pelleting: Science, Technology and Practical Applications (Springer, 2015). https://doi.org/10.1201/b19298.
Maclachlan, S. & Zalik, S. Plastid structure, chlorophyll concentration, and free amino acid composition of a chlorophyll mutant of barley. Can. J. Bot. 41, 1053–1062 (1963).
Maldonado-Garcia, M. et al. Antioxidant and immunostimulant potentials of Chenopodium ambrosioides L. in Pacific red snapper (Lutjanus peru). Aquaculture 513, 734414 (2019).
Manna, M., Rengasamy, B. & Sinha, A. K. A rapid and robust colorimetric method for measuring relative abundance of auxins in plant tissues. Phytochem. Anal. 35, 1052–1062 (2024).
Warrier, R. R., Paul, M. & Vineetha, M. V. Estimation of salicylic acid in Eucalyptus leaves using spectrophotometric methods. Genet Plant Physiol. 3, 90–97 (2013).
Dumale, J. V., Gamoso, G. R. & Divina, C. C. Plant growth promoting effect, gibberellic acid and auxin like activity of liquid extract of Caulerpa racemosa on rice seed germination. Int. J. Agric. Technol. 12, 2219–2226 (2016).
Materán, M. E. et al. Abscisic acid and 3-indolacetic acid levels during the reinvigoration process of Pinus radiata D. Don adult material. Plant Growth Regul. 59, 171–177 (2009).
Munns, R., Wallace, P. A., Teakle, N. L. & Colmer, T. D. Measuring soluble ion concentrations (Na(+), K(+), Cl(-)) in salt-treated plants. Methods Mol. Biol. 639, 371–382 (2010).
Nalina, M. et al. Water deficit-induced oxidative stress and differential response in antioxidant enzymes of tolerant and susceptible tea cultivars under field condition. Acta Physiol. Plant 43, 10 (2021).
Carlberg, I. & Mannervik, B. Purification and characterization of the flavoenzyme glutathione reductase from rat liver. J. Biol. Chem. 250, 5475–5480 (1975).
Asada, K. Ascorbate peroxidase—a hydrogen peroxide-scavenging enzyme in plants. Physiol. Plant 85, 235–241 (1992).
Del Buono, D., Ioli, G., Nasini, L. & Proietti, P. A comparative study on the interference of two herbicides in wheat and italian ryegrass and on their antioxidant activities and detoxification rates. J. Agric. Food Chem. 59, 12109–12115 (2011).
Akram, J. et al. Role of modern biotechnology in improving crop protection. Bull. Biol. Allied Sci. Res. 2025(1), 100. https://doi.org/10.54112/bbasr.v2025i1.100 (2025).
Bashir, H. et al. Integrating genomics and biotechnological approaches to enhance abiotic stress tolerance in sesame (Sesamum indicum L.). Bull. Biol. Allied Sci. Res. 2023(1), 37. https://doi.org/10.54112/bbasr.v2023i1.37 (2023).
Tanimoto, E. Regulation of root growth by plant hormones roles for auxin and gibberellin. CRC Crit. Rev. Plant Sci. 24, 249–265 (2005).
Zahid, A. et al. Chlorophyll degradation under smog exposure: unveiling the molecular and ecological consequences. Bull. Biol. Allied Sci. Res. 1, 96. https://doi.org/10.54112/bbasr.v2025i1.96 (2025).
Qadir, M. et al. Comparative assessment of chromate bioremediation potential of Pantoea conspicua and Aspergillus niger. J. Hazard Mater. 424, 127314 (2022).
Rana, K. L. et al. Endophytic microbes: biodiversity, plant growth-promoting mechanisms and potential applications for agricultural sustainability. Antonie Van Leeuwenhoek 113, 1075–1107 (2020).
Bódalo, A. et al. In vitro studies of endophytic bacteria isolated from ginger (Zingiber officinale) as potential plant-growth-promoting and biocontrol agents against Botrytis cinerea and Colletotrichum acutatum. Plants 12(23), 4032 (2023).
Selvakumar, G. et al. Cold tolerance and plant growth promotion potential of Serratia marcescens strain SRM (MTCC 8708) isolated from flowers of summer squash (Cucurbita pepo): Cold-tolerant Serratia marcescens. Lett. Appl. Microbiol. 46, 171–175 (2008).
Mahdi, I., Hafidi, M., Allaoui, A. & Biskri, L. Halotolerant endophytic bacterium Serratia rubidaea ED1 enhances phosphate solubilization and promotes seed germination. Agriculture 11, 224 (2021).
Trinh, L. L. & Nguyen, H. H. Role of plant-associated microbes in plant health and development: the case of the Serratia genus. Technol. Agronomy 4, 0024–0025 (2024).
Barzanti, R. et al. Isolation and characterization of endophytic bacteria from the nickel hyperaccumulator plant Alyssum bertolonii. Microb. Ecol. 53, 306–316 (2007).
Sessitsch, A., Reiter, B. & Berg, G. Endophytic bacterial communities of field-grown potato plants and their plant-growth-promoting and antagonistic abilities. Can. J. Microbiol. 50, 239–249 (2004).
An, X. et al. NaCl as an excellent trigger-induced biodiesel production and phenol-containing wastewater treatment in a novel salt-tolerant microalgae Ankistrodesmus sp. ACC. Bioresource Technol. 429, 132515 (2025).
Saharan, B. S. & Nehra, V. Assessment of plant growth promoting attributes of cotton (Gossypium hirsutum) rhizosphere isolates and their potential as bio-inoculants. J. Environ. Res. Develop. 5, 575–583 (2011).
Ahemad, M. & Khan, M. S. Phosphate-solubilizing and plant-growth-promoting Pseudomonas aeruginosa PS1 improves greengram performance in quizalafop-p-ethyl and clodinafop amended soil. Arch. Environ. Contam. Toxicol. 58, 361–372 (2010).
Mohammadi, K. & Sohrabi, Y. Bacterial biofertilizers for sustainable crop production: a review. ARPN J. Agric. Biol. Sci. 7, 307–316 (2012).
Sharma, S. B., Sayyed, R. Z., Trivedi, M. H. & Gobi, T. A. Phosphate solubilizing microbes sustainable approach for managing phosphorus deficiency in agricultural soils. Springer plus 2, 587 (2013).
Abbas, A. et al. Exploring the response mechanisms of rice to salinity stress. Bull. Biol. Allied Sci. Res. 2024(1), 58. https://doi.org/10.54112/bbasr.v2024i1.58 (2024).
Qadir, M. et al. Phytohormones producing rhizobacterium alleviates chromium toxicity in Helianthus annuus L. by reducing chromate uptake and strengthening antioxidant system. Chemosphere 258, 127386 (2020).
Rasheed, M. & Malik, A. Mechanism of drought stress tolerance in wheat. Bull. Biol. Allied Sci. Res. 2022(1), 23. https://doi.org/10.54112/bbasr.v2022i1.23 (2022).
Khan, M. A. et al. Alleviation of salt stress response in soybean plants with the endophytic bacterial isolate Curtobacterium sp. SAK1. Ann. Microbiol. 69, 797–808 (2019).
Nawaz, A. et al. Potential of salt tolerant pgpr in growth and yield augmentation of wheat (Triticum aestivum L) under saline conditions. Front. Microbiol. 2020, 11. https://doi.org/10.3389/fmicb.2020.02019 (2020).
Alaylar, B. et al. Integration of molecular tools in microbial phosphate solubilization research in agriculture perspective. World J. Microbiol. Biotechnol. 36(7), 93. https://doi.org/10.1007/s11274-020-02870-x (2020).
Egamberdieva, D., Wirth, S., Bellingrath-Kimura, S. D., Mishra, J. & Arora, N. K. Salt-tolerant plant growth promoting rhizobacteria for enhancing crop productivityof saline soils. Front. Microbiol. https://doi.org/10.3389/fmicb.2019.02791 (2019).
Bhise, K. K. & Dandge, P. B. Alleviation of salinity stress in rice plant by encapsulated salt tolerant plant growth promoting bacteria Pantoea agglomerans strain KL and its root colonization ability. Arch. Agronomy Soil Sci. 65(14), 1955–1968. https://doi.org/10.1080/03650340.2019.1584395 (2019).
Demiral, T. & Turkan, I. Comparative lipid peroxidation, antioxidant defense systems and proline content in roots of two rice cultivars differing in salt tolerance. Environ. Exp. Bot. 53, 247–257 (2005).
Gómez-Gómez, L., Felix, G. & Boller, T. A single locus determines sensitivity to bacterial flagellin in Arabidopsis thaliana: Sensitivity of A. thaliana to bacterial flagellin. Plant J 18, 277–284 (1999).
Hernández, J. A., Jiménez, A., Mullineaux, P. & Sevilia, F. Tolerance of pea (Pisum sativum L.) to long-term salt stress is associated with induction of antioxidant defences. Plant Cell Environ. 23, 853–862 (2000).
Mandhania, S., Madan, S. & Sawhney, V. Antioxidant defense mechanism under salt stress in wheat seedlings. Biol. Plant 50, 227–231 (2006).
Tewari, S. & Arora, N. K. Multifunctional exopolysaccharides from Pseudomonas aeruginosa PF23 involved in plant growth stimulation, biocontrol and stress amelioration in sunflower under saline conditions. Curr. Microbiol. 69, 484–494 (2014).
Aizaz, M. et al. Exploring the potential of halotolerant bacteria from coastal regions to mitigate salinity stress in wheat: physiological, molecular, and biochemical insights. Front. Plant Sci. 14, 1224731 (2023).
Hasegawa, P. M., Bressan, R. A., Zhu, J.-K. & Bohnert, H. J. Plant cellular and molecular responses to high salinity. Annu. Rev. Plant Physiol. Plant Mol. Biol. 51, 463–499 (2000).
El-Shabrawi, H. et al. Redox homeostasis, antioxidant defense, and methylglyoxal detoxification as markers for salt tolerance in Pokkali rice. Protoplasma 245, 85–96 (2010).
Khan, F. et al. Impact of various concentrations of nacl on morphological attributes of different citrus rootstocks under field conditions. Bull. Biol. Allied Sci. Res. 2023(1), 50. https://doi.org/10.54112/bbasr.v2023i1.50 (2023).
Chattha, M. et al. The impact of seed size on initial drought stress resilience and yield in wheat cultivation. Bul. Biol. Allied Sci. Res. 2024(1), 79. https://doi.org/10.54112/bbasr.v2024i1.79 (2024).
Meloni, D. A., Oliva, M. A., Martinez, C. A. & Cambraia, J. Photosynthesis and activity of superoxide dismutase, peroxidase and glutathione reductase in cotton under salt stress. Environ. Exp. Bot. 49, 69–76 (2003).
Mushtaq, R. et al. Impact of zinc oxide nanoparticles under lead toxicity on chilli. J. Phys. Biomed. Biol. Sci. 2025(1), 38. https://doi.org/10.64013/jpbab.v2025i1.38 (2025).
Tahir, A., Ashraf, M. & Shabir, F. Biochemical, physiological and molecular responses of the horticultural crops to cold responses. J. Phys. Biomed. Biol. Sci. 2025(1), 43. https://doi.org/10.64013/jpbab.v2025i1.43 (2025).
Niu, H. et al. The endophytic bacterial entomopathogen Serratia marcescens promotes plant growth and improves resistance against Nilaparvata lugens in rice. Microbiol. Res. 256, 126956 (2022).
Parashar, M. et al. Two novel plant-growth-promoting lelliottia amnigena isolates from Euphorbia prostrata aiton enhance the overall productivity of wheat and tomato. Plants 12, 17 (2023).
El-Akhdar, I., Elsakhawy, T. & Abo-Koura, H. A. Alleviation of Salt Stress on Wheat (Triticum aestivum L.) by Plant Growth Promoting Bacteria strains Bacillus halotolerans MSR-H4 and Lelliottia amnigena MSR-M49. J. Adv. Microbiol. 2020, 44–58 (2020).
Abrahám, E., Hourton-Cabassa, C., Erdei, L. & Szabados, L. Methods for determination of proline in plants. Methods Mol. Biol. 639, 317–331 (2010).
Berríos, J., Illanes, A. & Aroca, G. Spectrophotometric method for determining gibberellic acid in fermentation broths. Biotechnol. Lett. 26, 67–70 (2004).
Devi, K. A., Pandey, P. & Sharma, G. D. Plant growth-promoting endophyte Serratia marcescens AL2-16 enhances the growth of Achyranthes aspera L., a medicinal plant. Hayati 23, 173–180 (2016).
Gamborg, O. & Phillips, G. C. Plant Cell, Tissue and Organ Culture: Fundamental Methods 1995th edn. (Springer, 1995).
Ghazi, A., Atia, E. & Elsakhawy, T. Evaluation of an endophytic plant growth-promoting bacterium, Klebsiella variicola, in mitigation of salt stress in tuberose (Polianthes tuberosa L.). J. Hortic. Sci. Biotechnol. 96, 770–782 (2021).
Gupta, D. K. et al. (eds) Antioxidants and Antioxidant Enzymes in Higher Plants 1st edn. (Springer International Publishing, 2018).
Gyaneshwar, P. et al. Endophytic colonization of rice by a diazotrophic strain of Serratia marcescens. J. Bacteriol. 183, 2634–2645 (2001).
Hassan, S. & Mathesius, U. The role of flavonoids in root-rhizosphere signalling: opportunities and challenges for improving plant-microbe interactions. J. Exp. Bot. 63, 3429–3444 (2012).
Iniguez, A. L., Dong, Y. & Triplett, E. W. Nitrogen fixation in wheat provided by Klebsiella pneumoniae 342. Mol. Plant Microbe Interact 17, 1078–1085 (2004).
Ismail, M. M., El Zokm, G. M. & El-Sayed, A. A. M. Variation in biochemical constituents and master elements in common seaweeds from Alexandria Coast, Egypt, with special reference to their antioxidant activity and potential food uses: prospective equations. Environ. Monit. Assess. 189, 648 (2017).
Gul, K. Klebsiella aerogenes strain KL-4 16S ribosomal RNA gene, partial sequence – Nucleotide – NCBI. https://www.ncbi.nlm.nih.gov/nuccore/PP854706.1 (2024).
Gul, K. Lelliottia amnigena strain KS7 16S ribosomal RNA gene, partial sequence – Nucleotide - NCBI. https://www.ncbi.nlm.nih.gov/nuccore/PQ218332.1(2024).
Gul, K. Serratia marcescens strain FJ530951.1 16S ribosomal RNA gene, partial sequence - Nucleotide - NCBI. https://www.ncbi.nlm.nih.gov/nuccore/2741075396 (2024).
Khan, N. et al. Insights into the interactions among roots, rhizosphere, and rhizobacteria for improving plant growth and tolerance to abiotic stresses: a review. Cells 10, 1551 (2021).
Lamuela-Raventós, R. M. Folin-Ciocalteu method for the measurement of total phenolic content and antioxidant capacity. In Measurement of Antioxidant Activity & Capacity 107–115 (Wiley, 2017).
Mayak, S., Tirosh, T. & Glick, B. R. Plant growth-promoting bacteria confer resistance in tomato plants to salt stress. Plant Physiol. Biochem. 42, 565–572 (2004).
Morcillo, R. L. & Manzanera, M. The effects of plant-associated bacterial exopolysaccharides on plant abiotic stress tolerance. Metabolites 11, 337 (2021).
Mosleh, M. K., Hassan, Q. K. & Chowdhury, E. H. Application of remote sensors in mapping rice area and forecasting its production: a review. Sensors (Basel) 15, 769–791 (2015).
Munns, R. Comparative physiology of salt and water stress. Plant Cell Environ. 25, 239–250 (2002).
Potshangbam, M., Devi, S. I., Sahoo, D. & Strobel, G. A. Functional characterization of endophytic fungal community associated with Oryza sativa L. and Zea mays L.. Front. Microbiol. 8, 325 (2017).
Rudenko, N. N., Vetoshkina, D. V., Marenkova, T. V. & Borisova-Mubarakshina, M. M. Antioxidants of non-enzymatic nature: Their function in higher plant cells and the ways of boosting their biosynthesis. Antioxidants (Basel) 12, 2014 (2023).
Sarker, U. & Oba, S. Drought stress enhances nutritional and bioactive compounds, phenolic acids and antioxidant capacity of Amaranthus leafy vegetable. BMC Plant Biol. 18, 258 (2018).
Acknowledgements
The authors would like to extend their sincere appreciation to the Ongoing Research Funding Program (ORF 2025-165), King Saud University, Riyadh, Saudi Arabia. The authors would also like to extend their sincere appreciation to Abdul Wali Khan University Mardan, Pakistan for their financial support in this research project.
Author information
Authors and Affiliations
Contributions
Conceptualization by KG., RSK and AI; methodology by RSK. and AH; software by KG; validation by AI., RSK and QA.; resources by AH and RSK; data curation by SH., DA and QA; writing—original draft preparation was prepared by KG., writing—review and editing by QA., AH., and RSK; visualization by SH and DA; supervision, RSK and AH; project administration, RSK, DA and QA.; All authors have read and agreed to the published version of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors have no relevant financial or non-financial interests to disclose.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Gul, K., Khan, R.S., Iqbal, A. et al. Salt stress mitigating potential of bacterial endophytes isolated from Moringa oleifera. Sci Rep 15, 39813 (2025). https://doi.org/10.1038/s41598-025-23411-2
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-23411-2