Abstract
The PromarkerD test accurately predicts diabetic kidney disease. We aimed to determine whether PromarkerD and/or its constituent biomarkers would also identify an increased risk of distal sensory polyneuropathy (DSPN) complicating type 2 diabetes (T2D). We selected 160 community-based adults without baseline DSPN (mean age 69 years, 56% males), 80 of whom were free of DSPN after four years and 80 who developed DSN based on the vibration perception threshold. Baseline plasma apolipoprotein A-IV (ApoA4), CD5 antigen-like protein (CD5L) and insulin-like growth factor binding protein 3 (IBP3) concentrations were measured and PromarkerD risk scores were generated. Logistic regression modelling assessed associations between PromarkerD risk and its component proteins and incident DSPN. At Year 4, 77 of 151 with valid data (51%) had developed DSPN. The PromarkerD area under the receiver operating characteristic curve was 0.53 (95% CI 0.43–0.62). The odds ratio for the PromarkerD score for identifying incident DSPN was 0.97 (95% CI 0.82–1.16, P = 0.76), with similar non-significant results for plasma ApoA4, CD5L, and IBP3. PromarkerD and its constituent protein concentrations, each with some preclinical or epidemiological evidence of an association with DSPN, did not predict incident DSPN over 4 years of follow-up. These data support microangiopathy-specific pathophysiological mechanisms in T2D.
Similar content being viewed by others
Introduction
Distal sensory polyneuropathy (DSPN) develops in up to 50% of people living with diabetes1. It is associated with significant morbidity including falls and fractures, chronic pain, foot ulceration and lower extremity amputation2,3. DSPN can markedly impair quality of life,4 substantially increases health care costs,5 and it is an independent predictor of all-cause mortality6. Although considered largely irreversible,7 there are several potentially modifiable major risk factors including poor glycemic control, dyslipidemia, smoking and hypertension8. Their early and intensive management can prevent or delay the development and/or progression of DSPN9.
There are no validated scores which reliably predict incident DSPN and would thus facilitate timely optimised preventive management strategies. This includes the use of biomarkers,10 but combinations of biomarkers representing distinct aspects of the various pathophysiological processes underlying DSPN might hold promise10. In the case of another major microvascular complication, diabetic kidney disease (DKD), PromarkerD is a biomarker-based blood test that accurately predicts future renal function decline in both type 111 and type 212 diabetes (T2D). This test is based on measurement of the plasma concentrations of three proteins, apolipoprotein A-IV (ApoA4), CD5 antigen-like (CD5L) protein (or apoptosis inhibitor of macrophage) and insulin-like growth factor binding protein 3 (IBP3). A high risk PromarkerD score is associated with raised ApoA4, and low CD5L and IBP3, concentrations13. This pattern reflects the likely direct renal effects of ApoA4 and CD5L,13,14 but the inverse relationship for IBP3 contrasts with the positive association with DKD found in cross-sectional studies13,15.
There is some evidence that the three biomarker proteins measured in the PromarkerD test may have pathophysiological and thus prognostic relevance to DSPN as well as DKD. Transfer of ApoA4 from plasma to neurons may play a role in nerve regeneration,16 while CD5L is involved in neuronal repair through its activity in myelin-loaded macrophages17. Plasma concentrations of IBP3, a key modulator of the neurotrophic effects of insulin-like growth factor 1 (IGF-1),18 are lower in individuals with type 2 diabetes (T2D) and DSPN compared to those without DSPN19. Microvascular complications of diabetes often coexist, reflecting, at least in part, shared risk factors. There are some cross-sectional studies showing a close correlation between DKD and DSPN20,21,22,23. It would, therefore, seem important to ensure that a test such as PromarkerD was specific for DKD and, if not, whether there were single or multiple biomarkers that are prognostically important for both DKD and DSPN.
The aim of this exploratory, secondary analysis was to assess whether PromarkerD, a biomarker test originally developed to predict DKD, and/or its constituent biomarker proteins may also be associated with the risk of developing DSPN, based on the known potential co-occurrence of microvascular complications in community-based adults with T2D.
Materials and methods
Participants and approvals
We utilized data and samples from the longitudinal observational Fremantle Diabetes Study Phase II (FDS2) conducted in a postcode-defined urban community of 157,000 residents surrounding the city of Fremantle in Western Australia (WA). The primary aim of FDS2 was to examine the natural history of diabetes and its consequences in a representative community-based cohort. Details of recruitment, subject characteristics and study procedures have been published24. In brief, 1551 participants with clinically diagnosed T2D were recruited from a postcode-defined urban area surrounding the port of Fremantle between 2008 and 2011. The present study was designed as a sub-group case-control study nested within the FDS2 longitudinal cohort. The South Metropolitan Area Health Service Human Research Ethics Committee approved the FDS2 protocol (reference 07/397) and written informed consent was obtained from each participant. The study protocol conformed to the ethical guidelines of the Declaration of Helsinki.
Clinical assessment
All FDS2 participants underwent a comprehensive baseline and then biennial face-to-face assessments including an interview, detailed questionnaires, a physical examination, and fasting biochemical tests performed in a single nationally accredited laboratory24. Fasting plasma samples were collected at the baseline visit (2008–2011) and stored at − 80 °C until biomarker analysis. All samples tested had no prior freeze-thaw cycles. The physical examination included DSPN defined by the vibration perception threshold (VPT) which was measured as part of the Michigan Neuropathy Screening Instrument (MNSI)25. A detailed review of variables comprising the clinical portion of the MNSI revealed that the VPT was the most reliable26. Although there is no universally endorsed routine clinical tool for detection of DSPN,27 VPT thus appears a simple robust method in observational studies26 and it has also been used in major clinical trials28. For the purposes of the present study, DSPN included participants in whom VPT could be reliably assessed in only one leg due to contralateral amputation or neuropathic findings due to a documented non-diabetes-related cause. DSPN was considered present if the MNSI VPT score was greater than 0.5 out of 2. If VPT was absent, the foot scored 1, if VPT was reduced, the foot scored 0.5, and if VPT was normal then the foot scored 0. The maximum score for both feet was 2.
Laboratory assays
Plasma ApoA4, CD5L and IBP3 concentrations were measured in stored baseline plasma samples by CaptSure™ enzyme linked immunosorbent-assay (TGR BioSciences, an Abcam company, Adelaide, South Australia) as previously described29. PromarkerD scores (0 to 100%) were calculated using these biomarker concentrations and age, serum HDL-cholesterol and the estimated glomerular filtration rate (eGFR) using a proprietary weighted algorithm as described previously12.
Statistical analysis
Statistical analyses were performed in SPSS for Windows (version 29; SPSS Inc., Chicago, IL) and R 4.4.0 with R Studio (v 2024.04.2 + 764). The present case-control study included 80 cases with DSPN at Year 4 and 80 controls without DSPN at Year 4 (Fig. 1). The cases were randomly selected from the 172 available using the computer program SPSS and matched 1:1 by sex and then by age (the closest match, mostly within 1 year) from 433 participants without DSPN at Year 4. Logistic regression modelling was used to determine the ability of PromarkerD and its constituent biomarkers (ApoA4, CD5L and IBP3) to predict incident DSPN. Models used either continuous or categorical variables without any other independent variables (univariate models). Scores and individual biomarker concentrations were log-transformed before use in logistic regression modelling to reduce right-skewness and stabilise variance, improving logistic regression assumptions. Based on previous DKD data for risk of renal function decline over four years, PromarkerD scores were categorized as low (< 10%), moderate (≥ 10%) and high (≥ 20%) risk12. Model performance was evaluated using the area under the receiver operating characteristic curve (ROC AUC), with sensitivity and specificity assessed at the maximum Youden index,30 and at the moderate- and high-risk cutoffs. Odds ratios (ORs) with 95% confidence intervals (CIs) were calculated for continuous PromarkerD scores, PromarkerD risk categories, and individual biomarker concentrations.
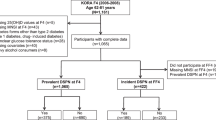
Flow chart showing the process by which two samples each comprising 80 FDS2 participants with T2D were selected for the present analyses.
Results
The 1551 participants with clinically diagnosed T2D were screened for monogenic diabetes and Latent Autoimmune Diabetes of Adults (LADA)31 and those with either diagnosis (n = 121) were excluded from the present analyses. Of the remaining 1430 adults with T2D, 605 had no DSPN at entry and attended for the Year 4 biennial face-to-face assessment (Fig. 1). Two matched sub-groups of 80 participants with or without DSPN at Year 4 were selected from the 172 and 433 participants, respectively, with available data. In addition to the expected similar age and sex distribution, diabetes duration and HbA1c were also similar at baseline (Fig. 1).
Of the 160 FDS2 participants included, 151 had plasma samples available from which valid PromarkerD risk scores were generated. Their mean age was 68.3 years, 55.6% were males, their median diabetes duration was 10.0 years, their mean HbA1c 7.0% (53 mmol/mol), their mean eGFR 79.5 mL/min/1.73m2 and their mean serum HDL-cholesterol 1.3 mmol/L. The geometric mean (SD range) PromarkerD score was 4.7% (0.7%-29.9%), with corresponding concentrations for ApoA4, CD5L and IBP3 of 21.7 (9.8–47.8) ug/mL, 3.5 (1.7-7.0) ug/mL and 2.8 (1.9-4.0) ug/mL, respectively. Of these, 36 (23.8%) were classified as high risk, 31 (20.5%) moderate risk, and 84 (55.6%) low risk. At the Year 4 follow-up, a median of 4.2 years after study entry, 77 (51%) had developed DSPN, reflecting the 1:1 matched case-control study design rather than the incidence in the broader FDS2 cohort of 28% (172/605) (Fig. 1).
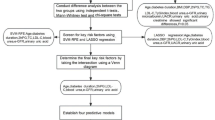
The PromarkerD ROC AUC was 0.53 (95% CI 0.43–0.62; Fig. 2). A post-hoc sample size calculation was performed which indicated that, for 50% developing DSPN, an AUC of 0.85 and a 95% confidence interval, a sample size of 150 would be required for the comparison of the ROC AUC with a null hypothesis value32. This indicates that the present sample size was adequate for clinically meaningful discrimination between those with and without incident DSPN.
ROC for PromarkerD as a predictor of incident DSPN (left hand panel) with a ROC AUC of 0.53. Odds ratios and 95% confidence intervals as horizontal bars (right hand panel) derived from logistic regression models assessing the association between PromarkerD scores (OR 0.97, 0.82–1.16), risk categories (low, moderate, high), and individual plasma biomarkers (ApoA4, CD5L, IGFBP3) with the incidence of DSPN.
At the optimal ROC cut-off by Youden’s index, sensitivity and specificity were 58.4% and 45.1%, respectively. Sensitivity and specificity were 39.0% and 50.0%, respectively, at the moderate risk cut-off, and 20.8% and 73.0% at the high-risk cut-off. The odds ratio (OR) for the continuous PromarkerD score for identifying incident DSPN was 0.97 (95% CI 0.82–1.16, P = 0.76), with similar non-significant results for plasma ApoA4 (1.03 (0.68–1.56)), CD5L (0.93 (0.57–1.48)), and IBP3 (0.90 (0.38–2.11)) concentrations (Fig. 2).
Discussion
The ROC AUCs from the present analyses showed that the PromarkerD test, whether as a continuous variable or subdivided into DKD risk categories, was no better than the play of chance at identifying community-based people living with T2D who developed DSPN during a four-year follow-up period. The PromarkerD test had correspondingly low sensitivity and specificity. Although DKD data were not re-evaluated in this study, previous FDS2 analyses have demonstrated that PromarkerD and its constituent proteins are prognostically specific for renal decline11,12. In contrast, the present findings indicate that PromarkerD does not predict incident DSPN, and prior FDS2 analyses also found no association with incident diabetic retinopathy33. Taken together, these observations suggest that, despite shared risk factors such as a high HbA1c and long diabetes duration,34 individual microvascular complications of type 2 diabetes each have a specific underlying pathophysiology.
The PromarkerD score includes at least two constituent biomarkers (ApoA4 and CD5L) with a relatively strong rationale for inclusion based on renal associations or effects,14,35 even if the relationship between IBP3 and diabetic nephropathy is less clear13. Some preclinical16,17 and cross-sectional epidemiological19 evidence suggests that these three proteins might also have a role in the development of DSPN complicating diabetes. ApoA4 and CD5L could influence neuronal cholesterol transport16 and macrophage function17 during neuronal repair. Although there is a complex relationship between IBP3 and potential positive IGF-1 effects on neuronal integrity,18 low levels of IBP3 and thus perhaps IGF-1 have been associated with DSPN19. Despite these considerations, the present logistic regression modelling shows ORs very close to unity for each biomarker and thus no significant associations with incident DSPN. These data are consistent with the interpretation that PromarkerD and its component plasma proteins are prognostically specific for DKD and are not associated with the development of DSPN or, based on a previous report,33 retinopathy.
Since DKD has been suggested as a risk factor for the development of peripheral neuropathy,36 the present findings support the hypothesis that it is not a major contributor to DSPN complicating T2D. Although cross-sectional studies show a correlation between DKD and DSPN,20,21,22,23 an analysis of incident DSPN data based on VPT at baseline and Year 4 in FDS2 combined with the earlier Fremantle Diabetes Study Phase I (total 1,109 participants) identified an eGFR < 30 mL/min/1.73 m² as a significant independent determinant but milder degrees of renal impairment were not included in the multivariable model26. Those with an eGFR < 30 mL/min/1.73 m2 were a very small group, comprising < 3% of the total sample, suggesting that a contribution of DKD to DSPN complicating T2D occurs late and in relatively few individuals.
Given that mechanisms underlying the initiation and progression of DSPN are likely complication-specific, there is the need for initial proteomic biomarker discovery with well characterised individuals with T2D with or without DSPN rather than adapt an existing test. In a recent publication, an artificial neural network was used to predict VPT-defined DSPN using clinical and laboratory features37 and future proteomic-based risk prediction tools could incorporate this approach to further improve prognostic accuracy.
The present study had some limitations. There could have been selection bias in that healthier residents with diabetes may have participated. We had limited numbers in each randomly selected sub-group but the close proximity of the ROC AUC to 0.5 suggests that a larger sample would not have changed the findings. Similarly, since extremely strong biomarker associations are required for meaningful classification accuracy,38 there is unlikely to have been clinically useful prognostic contributions of PromarkerD and its constituent proteins after adjusting for other variables associated with incident DSPN. The study strengths include evidence that the FDS2 sample is representative of diabetes in an urban Australian setting,24 the long duration follow-up and detailed participant phenotyping.
In conclusion, although the PromarkerD test has demonstrated prognostic utility for DKD in prior studies, it did not identify people with T2D at risk of developing DSPN in the present study. These findings support the hypothesis that distinct microvascular complications of type 2 diabetes require separate predictive tools.
Data availability
The data reported in this study are available from the Corresponding author (TMED) on reasonable request.
References
Hicks, C. W. & Selvin, E. Epidemiology of peripheral neuropathy and lower extremity disease in diabetes. Curr. Diab. Rep. 19, 86. https://doi.org/10.1007/s11892-019-1212-8 (2019).
Hicks, C. W. et al. The association of peripheral neuropathy detected by monofilament testing with risk of falls and fractures in older adults. J. Am. Geriatr. Soc. 71, 1902–1909. https://doi.org/10.1111/jgs.18338 (2023).
McDermott, K. et al. Epidemiology, and disparities in the burden of diabetic foot ulcers. Diabetes Care. 46, 209–221. https://doi.org/10.2337/dci22-0043 (2023).
Perveen, W. et al. Prevalence of peripheral neuropathy, amputation, and quality of life in patients with diabetes mellitus. Sci. Rep. 14, 14430. https://doi.org/10.1038/s41598-024-65495-2 (2024).
Zhao, Y. et al. Prevalence of other diabetes-associated complications and comorbidities and its impact on health care charges among patients with diabetic neuropathy. J. Diabetes Complicat. 24, 9–19. https://doi.org/10.1016/j.jdiacomp.2008.09.001 (2010).
Vagi, O. E. et al. The association between distal symmetric polyneuropathy in diabetes with all-cause mortality—A meta-analysis. Front. Endocrinol. (Lausanne). 14, 1079009. https://doi.org/10.3389/fendo.2023.1079009 (2023).
Zochodne, D. W. The challenges of diabetic polyneuropathy: A brief update. Curr. Opin. Neurol. 32, 666–675. https://doi.org/10.1097/WCO.0000000000000723 (2019).
Tesfaye, S. et al. Prevalence of diabetic peripheral neuropathy and its relation to glycaemic control and potential risk factors: The EURODIAB IDDM complications study. Diabetologia 39, 1377–1384. https://doi.org/10.1007/s001250050586 (1996).
Smith, S. et al. Prevention and management strategies for diabetic neuropathy. Life (Basel). https://doi.org/10.3390/life12081185 (2022).
Chong, Z. Z. & Souayah, N. Crumbling pathogenesis and biomarkers for diabetic peripheral neuropathy. Biomedicines https://doi.org/10.3390/biomedicines13020413 (2025).
Davis, T. M. E. et al. Application of a validated prognostic plasma protein biomarker test for renal decline in type 2 diabetes to type 1 diabetes: The Fremantle diabetes study phase II. Clin. Diabetes Endocrinol. 10, 30. https://doi.org/10.1186/s40842-024-00191-8 (2024).
Peters, K. E. et al. Validation of a protein biomarker test for predicting renal decline in type 2 diabetes: The Fremantle diabetes study phase II. J. Diabetes Complicat. 33, 107406. https://doi.org/10.1016/j.jdiacomp.2019.07.003 (2019).
Peters, K. E. et al. Identification of novel circulating biomarkers predicting rapid decline in renal function in type 2 diabetes: The Fremantle diabetes study phase II. Diabetes Care. 40, 1548–1555. https://doi.org/10.2337/dc17-0911 (2017).
Kronenberg, F. et al. Apolipoprotein A-IV serum concentrations are elevated in patients with mild and moderate renal failure. J. Am. Soc. Nephrol. 13, 461–469. https://doi.org/10.1681/ASN.V132461 (2002).
Narayanan, R. P. et al. IGFBP2 is a biomarker for predicting longitudinal deterioration in renal function in type 2 diabetes. Endocr. Connect. 1, 95–102. https://doi.org/10.1530/EC-12-0053 (2012).
Boyles, J. K., Notterpek, L. M. & Anderson, L. J. Accumulation of apolipoproteins in the regenerating and remyelinating mammalian peripheral nerve. Identification of Apolipoprotein D, Apolipoprotein A-IV, Apolipoprotein E, and Apolipoprotein A-I. J. Biol. Chem. 265, 17805–17815 (1990).
Zheng, B. et al. Unresolved excess accumulation of myelin-derived cholesterol contributes to scar formation after spinal cord injury. Res. (Wash D C. 6, 0135. https://doi.org/10.34133/research.0135 (2023).
Biadgo, B., Tamir, W. & Ambachew, S. Insulin-like growth factor and its therapeutic potential for diabetes complications—Mechanisms and metabolic links: A review. Rev. Diabet. Stud. 16, 24–34. https://doi.org/10.1900/RDS.2020.16.24 (2020).
Karvestedt, L. et al. Peripheral sensory neuropathy associates with micro- or macroangiopathy: Results from a population-based study of type 2 diabetic patients in Sweden. Diabetes Care. 32, 317–322. https://doi.org/10.2337/dc08-1250 (2009).
Lai, Y. R. et al. Correlation between kidney and peripheral nerve functions in type 2 diabetes. QJM 113, 173–180. https://doi.org/10.1093/qjmed/hcz249 (2020).
Savage, S., Estacio, R. O., Jeffers, B. & Schrier, R. W. Urinary albumin excretion as a predictor of diabetic retinopathy, neuropathy, and cardiovascular disease in NIDDM. Diabetes Care. 19, 1243–1248. https://doi.org/10.2337/diacare.19.11.1243 (1996).
Xu, L., Lin, X., Guan, M. & Liu, Y. Correlation between different stages of diabetic nephropathy and neuropathy in patients with T2DM: A cross-sectional controlled study. Diabetes Ther. 9, 2335–2346. https://doi.org/10.1007/s13300-018-0519-9 (2018).
Zhang, Y., Jiang, Y., Shen, X. & Yan, S. Can both normal and mildly abnormal albuminuria and glomerular filtration rate be a danger signal for diabetic peripheral neuropathy in type 2 diabetes mellitus? Neurol. Sci. 38, 1381–1390. https://doi.org/10.1007/s10072-017-2946-1 (2017).
Davis, T. M., Bruce, D. G. & Davis, W. A. Cohort profile: The Fremantle diabetes study. Int. J. Epidemiol. 42, 412–421. https://doi.org/10.1093/ije/dys065 (2013). [doi].
Feldman, E. et al. A practical two-step quantitative clinical and electrophysiological assessment for the diagnosis and staging of diabetic neuropathy. Diabetes Care. 17, 1281–1289 (1994).
Davis, W. A., Hamilton, E. & Davis, T. M. E. Temporal trends in distal symmetric polyneuropathy in type 2 diabetes: The Fremantle diabetes study. J. Clin. Endocrinol. Metab. 109, e1083–e1094. https://doi.org/10.1210/clinem/dgad646 (2024).
Pop-Busui, R. et al. Diabetic neuropathy: A position statement by the American diabetes association. Diabetes Care. 40, 136–154. https://doi.org/10.2337/dc16-2042 (2017).
Gaede, P., Lund-Andersen, H., Parving, H. H. & Pedersen, O. Effect of a multifactorial intervention on mortality in type 2 diabetes. N Engl. J. Med. 358, 580–591. https://doi.org/10.1056/NEJMoa0706245 (2008).
Bringans, S., Peters, K., Casey, T., Ito, J. & Lipscombe, R. The new and the old: Platform cross-validation of immunoaffinity MASS spectrometry versus ELISA for PromarkerD, a predictive test for diabetic kidney disease. Proteomes https://doi.org/10.3390/proteomes8040031 (2020).
Youden, W. J. Index for rating diagnostic tests. Cancer 3, 32–35. https://doi.org/10.1002/1097-0142(1950)3:1%3C32::aid-cncr2820030106%3E3.0.co;2-3 (1950).
Davis, W. A. et al. Prevalence of diabetes in australia: Insights from the Fremantle diabetes study phase II. Intern. Med. J. 48, 803–809. https://doi.org/10.1111/imj.13792 (2018).
Kohn, M. A. & Senyak, J. Sample Size Calculators. (2025). https://www.sample-size.net/
Drinkwater, J. J. et al. Assessment of biomarkers associated with rapid renal decline in the detection of retinopathy and its progression in type 2 diabetes: The Fremantle diabetes study phase II. J. Diabetes Complicat. 35, 107853. https://doi.org/10.1016/j.jdiacomp.2021.107853 (2021).
Crasto, W., Patel, V., Davies, M. J. & Khunti, K. Prevention of microvascular complications of diabetes. Endocrinol. Metab. Clin. North. Am. 50, 431–455. https://doi.org/10.1016/j.ecl.2021.05.005 (2021).
Davis, T. M. E., Peters, K. E. & Lipscombe, R. Apoptosis inhibitor of macrophage and diabetic kidney disease. Cell. Mol. Immunol. 16, 521. https://doi.org/10.1038/s41423-018-0088-x (2019).
Chillon, J. M., Massy, Z. A. & Stengel, B. Neurological complications in chronic kidney disease patients. Nephrol. Dial Transpl. 31, 1606–1614. https://doi.org/10.1093/ndt/gfv315 (2016).
Dubey, V. N., Dave, J. M., Beavis, J. & Coppini, D. V. Predicting diabetic neuropathy risk level using artificial neural network and clinical parameters of subjects with diabetes. J. Diabetes Sci. Technol. 16, 275–281. https://doi.org/10.1177/1932296820965583 (2022).
Pepe, M. S., Janes, H., Longton, G., Leisenring, W. & Newcomb, P. Limitations of the odds ratio in gauging the performance of a diagnostic, prognostic, or screening marker. Am. J. Epidemiol. 159, 882–890. https://doi.org/10.1093/aje/kwh101 (2004).
Acknowledgements
We thank the FDS2 participants and staff for their involvement and PathWest Laboratory Medicine at Fremantle Hospital for laboratory tests. The proteomic analyses were performed, in part, using NCRIS (National Collaborative Research Infrastructure Strategy)-enabled Bioplatforms Australia infrastructure at the Western Australian Proteomics Facility, located at the Harry Perkins Institute for Medical Research.
Author information
Authors and Affiliations
Contributions
Kirsten Peters: Conceptualization, Methodology, Validation, Data curation, Writing – reviewing and editing. Timothy Davis: Conceptualization, Resources, Writing – Final draft, Funding acquisition. Isabella Joubert: Formal analysis, Data curation, Writing - Original draft, Martin Mead: Investigation. Angelo Bautista: Investigation. Wendy Davis: Conceptualization, Methodology, Resources, Data curation, Writing – Reviewing and editing, Funding acquisition.
Corresponding author
Ethics declarations
Competing interests
Proteomics International and the University of Western Australia are beneficiaries of patent PCT/AU2011/001212 which relates to the biomarkers described in this manuscript. TMED, WAD and KEP are named as inventors of this patent. There are no other potential conflicts of interest relevant to this article.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Peters, K., Davis, T.M.E., Joubert, I.A. et al. Assessment of biomarkers associated with renal decline for prediction of incident distal sensory polyneuropathy in type 2 diabetes. Sci Rep 15, 40214 (2025). https://doi.org/10.1038/s41598-025-24110-8
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-24110-8