Abstract
The safety of daptomycin in infants under 1 year old remains uncertain. This study aimed to identify signals potentially associated with daptomycin use in this special population based on the Food and Drug Administration adverse event reporting system (FAERS). Real-world pharmacovigilance research was conducted on the FAERS database from Q1 2004 to Q2 2024. Disproportionality analyses, including reporting odds ratio (ROR), proportional reporting ratio (PRR), multi-item gamma Poisson shrinker (MGPS), and Bayesian confidence propagation neural network (BCPNN), were used to assess the strength of AEs signals for daptomycin. A subgroup analysis was conducted on the data from the past 5 years to further explore the association between AEs and daptomycin. A total of 96 AEs of daptomycin as the primary suspected drug used in infants under 1 year old were reported, and 8 risk signals involving 3 system organ classes(SOCs) were identified as simultaneously meeting the 4 algorithms. The strongest associations were observed for hepatobiliary disorders (ROR 8.54), renal and urinary disorders (ROR 4.55), infections and infestations (ROR 3.68) in SOCs, and the top 4 signal intensities of AEs were hepatic cytolysis (ROR 297.93), drug reaction with eosinophilia and systemic symptoms (DRESS) (ROR 92.54), acute kidney injury (ROR 20.99), hypotension (ROR 14.10). In the subgroup analysis, 4 positive risk signals for AEs were identified, which were distributed across 3 SOCs. Among these, hepatobiliary disorders (ROR 10.98) topped the list. The strongest associations were observed for hepatic cytolysis (ROR 226.08), DRESS (ROR 110.04) in positive risk signals. The results highlight the new daptomycin AEs signals in younger infants and provide vital risk monitoring and identification support. In our analysis, hepatic cytolysis and DRESS were the most significant risk signals, and no positive signals related to the nervous system were observed. Additionally, due to the limitations of the FAERS, the correlation between AEs and daptomycin warrants cautious interpretation, and further explorations in the future are required.
Similar content being viewed by others
Introduction
Daptomycin, a novel cyclic lipopeptide antibiotic, has been approved for the treatment of complicated skin and skin structure infection (cSSSI), Staphylococcus aureus bloodstream infections (S. aureus bacteremia) in adults and pediatric patients aged 1–17 years, and right-sided infective endocarditis in adults1. In vitro studies have demonstrated that the effect of daptomycin on cell membranes is concentration-dependent. It causes bacterial membrane depolarization by promoting potassium ion efflux and arrests DNA, RNA, and protein synthesis, resulting in bacterial cell death2,3. Therefore, daptomycin possesses rapid bactericidal activity and a very low potential for the development of resistance4. It is effective against most gram-positive organisms, including Staphylococcus and Enterococcus species resistant to vancomycin, linezolid, and quinupristin/dalfopristin5.
cSSSI and S. aureus bacteremia caused by Staphylococcus aureus, including methicillin-resistant Staphylococcus aureus (MRSA), are common and challenging infections among children worldwide6,7,8. Studies conducted from 2015 to 2019 reported a total of 2747 cases of infections caused by gram-positive bacteria in children aged 17 years old and younger, specifically with skin and skin structure infections. Staphylococcus aureus was identified as the primary pathogen across all age groups, with MRSA being most prevalent in the group of children aged 1 year old or younger9.
Daptomycin, vancomycin, and linezolid are commonly used to treat MRSA infection in pediatric patients. Pharmacokinetic (PK) studies of daptomycin have been reported in infants 1–17 years of age with proven or suspected gram-positive infection10,11,12,13,14,15. However, there is very little relevant data regarding the use of daptomycin in infants under 1 year old. The safety of daptomycin in treating gram-positive infections has not been systematically and comprehensively evaluated in younger infants. Besides, daptomycin is excreted unchanged by the kidney16,17, studies have shown that children have a greater renal clearance of daptomycin than adults11,18, which means that children need a larger dose of daptomycin than adults for the treatment of positive bacterial infections, which undoubtedly makes clinicians concerned about the safety of large doses of daptomycin in children at younger ages.
Daptomycin for injection is not recommended for pediatric patients under 1 year old due to the risk of potential effects on muscular, neuromuscular, and/or nervous systems (either peripheral and/or central) observed in neonatal dogs. However, daptomycin is a trade-off choice to save the infant’s life when infected with multidrug-resistant gram-positive bacteria such as vancomycin-resistant staphylococci.
In contrast to the data available for adults, there is limited information to determine the efficacy and safety of daptomycin in infants. No systematic analyses of infants under 1 year old have been published to date, and there is a lack of large-scale clinical trials, with only scattered case reports available11,19,20. Therefore, it is necessary to comprehensively summarize the AEs of daptomycin in infants under 1 year old based on real-world data to identify statistical associations between daptomycin exposure and AEs in younger infants. Our research team used data mining algorithms to extract and analyze daptomycin AEs based on the FAERS database to address the lack of research in this area.
FAERS is the most comprehensive global database for spontaneously reported adverse drug reactions21. It monitors and evaluates the safety of drugs after they are marketed by collecting and analyzing AE reports, helping the FDA identify potential drug safety issues and take timely measures to protect public health22. The FAERS database receives reports from multiple sources, including drug manufacturers, healthcare professionals, and consumers. These reports are coded using Medical Dictionary for Regulatory Activities (MedDRA) and include information on drug-related AEs, medication errors, and product quality complaints, among other details, with updates made quarterly22. Researchers have extensively employed data mining algorithms to conduct post-marketing safety surveillance and re-evaluate medications using FAERS databases23,24,25,26. The FAERS database contains some AEs associated with the use of daptomycin in infants under 1 year old from around the world over a period of more than a decade. We used data mining techniques to analyze this data in order to identify statistical associations between daptomycin exposure and AEs in younger infants and to provide clinicians with information beyond scattered case reports. However, FAERS also has some limitations, such as unclear causality, voluntary reporting, and incompleteness, which need to be overcome through further research and verification27. Therefore, our team published a related study using pharmacokinetic modeling techniques to simulate the pharmacokinetic disposition process of different doses of daptomycin in infants under 1 year old28, which complements this study, and together provide valuable reference for reasonable clinical decision-making on daptomycin in infants under 1 year old.
Methods
Data source and collection
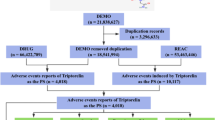
The data for this study were obtained from the FAERS database. The FAERS database, maintained by the FDA, is a public repository designed for the collection, used to evaluate and monitor AEs reports for marketed drugs, biological products, and medical devices21. The FAERS database was opened to the public in 2004. Daptomycin was first marketed in the United States in September 2003. Therefore, we downloaded the American Standard Code for Information Interchange (ASCII) files from the database, covering the period from its inception in 2004 up to the second quarter of 2024. These ASCII codes encompassed patient demographic and administrative information (DEMO), adverse event coding (REAC), drug/biological information (DRUG), and other relevant data. Signal mining was conducted exclusively on AEs where daptomycin was the primary suspected drug in infants under 1 year old, regardless of gender or nationality.
Standardization of data analysis
Daptomycin was chosen as the study drug, and duplicate reports were excluded. Only the most recent report based on the date was retained for data with identical case IDs. In this study, the drug names were standardized using the Medex_UIMA_1.8.3 system. Preferred terms (PTs) for AEs associated with daptomycin were aligned using the Medical Dictionary for Regulatory Activities version 25.0 (MedDRA 25.0). System Organ Classes (SOCs) corresponding to these PTs were also listed.
Data mining and statistical analysis
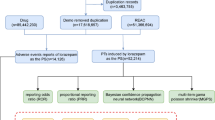
Disproportionality analysis is widely used for AEs signal detection, relying on the ratio of the observed number of AEs associated with a particular drug compared to the expected number or the number caused by other drugs. If this measured ratio is significantly large, indicating disproportionality, it suggests a potential association between the suspect drug and the AEs rather than being due to chance factors. This type of analysis includes metrics such as the reporting odds ratios (ROR), proportional reporting ratios (PRR), Bayesian confidence propagation neural network (BCPNN), and empirical Bayesian geometric mean (EBGM)29.
ROR have excellent abilities for discovering risk signals with high reliability and sensitivity30, which can estimate relative risks, reducing bias31. This study mainly utilized the ROR for AEs signal detection. An ROR value of 3 or higher indicates a positive risk signal, suggesting an association between the target drug and the AEs. Moreover, a higher ROR value indicates a stronger signal and a more robust association.
The formulas and screening criteria of the above 4 signal detection methods are expressed below. All data related to daptomycin were processed and analyzed using the R software (version 4.3.2).
Algorithms | Equation | Criteria |
|---|---|---|
ROR | \(ROR = \frac{ad}{{bc}}\) \(95\% CL = {\text{eln}}(ROR) \pm 1.96\sqrt {(\frac{1}{a} + \frac{1}{b} + \frac{1}{c} + \frac{1}{d})}\) | a ≥ 3; the lower limit of 95%CL > 1 |
PRR | \(PRR = \frac{a/(a + b)}{{c/(c + d)}}\) \({\chi 2} = e\ln (PRR) \pm 1.96\sqrt {(\frac{1}{a} - \frac{1}{a + b} + \frac{1}{c} - \frac{1}{c + d}} )\) | a ≥ 3; χ2 > 1 |
MGPS | \(EBGM = a\left( {a + b + c + \left. d \right)} \right./\left( {\left( {a + c} \right)\left( {a + b} \right)} \right)\) \(EBGM05 = e^{{\ln \left( {EBGM} \right) - 1.64\left( {\frac{1}{a} + \frac{1}{b} + \frac{1}{c} + \frac{1}{d}} \right)^{{0.5}} }}\) | a > 0; EBGM05 > 2 |
BCPNN | \(IC = {\text{log}}_{2} \frac{{p\left( {x,y} \right)}}{p\left( x \right)p\left( y \right)} = \log_{2} \frac{{a\left( {a + b + c + d} \right)}}{{\left( {a + b} \right)\left( {a + c} \right)}}\) \(IC025 = E\left( {IC} \right) - 2\sqrt {V\left( {IC} \right)}\) | a ≥ 3; IC025 > 0 |
Results
Basic characteristics of daptomycin-related AEs in younger infants
From the database’s inception in 2004 to the second quarter of 2024, after removing duplicate reports, the FAERS database collected a total of 65,083,961 drug records and 17,956,652 spontaneous reports involving 52,454,963 adverse reaction reports. Among these, daptomycin was identified as the primary suspected drug affecting the infants under 1 year old and accounting for 96 AEs in 40 reports.
Among the AEs reports associated with the use of daptomycin in infants under 1 year old, the number of case reports was highest during the 2021-2024.06 period (50.0%). More AEs were reported for females than males, with a median age of approximately 7.5 days and a median weight of approximately 7.8 kg. The majority of the reports were submitted by Physician (42.5%). The countries with the higher number of reported cases were the United States (35.0%), France (20.0%), Japan (15.0%), and Italy (12.5%). Nearly half of the reports (45.0%) do not specify the route of administration, but daptomycin is currently only approved for intravenous administration and is not available in oral or other formulations, so the administration routes not reported can be understood as intravenous administration. The primary indications of daptomycin in the infants under 1 year old were infection (37.5%), bacteremia (22.5%), endocarditis (15.0%), pneumonia (2.5%), skin infection (2.5%), abscess (2.5%), joint infection (2.5%), and other/unreported (15.0%), there are basically infection-related. In terms of clinical outcomes, AEs leading to hospitalization were the most common (37.5%), followed by death (32.5%) and other or unknown outcomes (32.5%). Detailed information on AEs reports for infants under 1 year old use is listed in Table 1.
Signal detection and analysis at the SOCs level
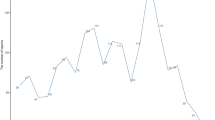
Analysis of the reports that identified daptomycin as the primary suspected drug for AEs revealed 16 associated SOCs. Among them, the 3 most frequently reported systems were “injury, poisoning and procedural complications” (ROR 1.30), “general disorders and administration site conditions” (ROR 1.29) and “hepatobiliary disorders” (ROR 8.54). However, further analysis of the signals using the ROR method revealed that the signal strength for the 3 most common systems was not the strongest. Upon analyzing the signals, positive risk signals were identified for the following SOCs: “hepatobiliary disorders” (ROR 8.54), “renal and urinary disorders” (ROR 4.55), and “infections and infestations” (ROR 3.68), the signal strengths of these 3 systems were substantial. Detailed information for the number of reports for the SOCs and results of the 4 signaling calculations, including ROR, are presented in Table 2, and the forest of AEs under SOCs ranked by ROR signal strength is shown in Fig. 1.
AEs signal analysis of PTs level under each SOCs for daptomycin use in infants under 1 year old based on the ROR.
Signal detection and analysis at the PTs level
We conducted screening at the PTs level to identify clinical symptoms or diseases with a reported count of at least 3 cases and positive risk signals. 9 PTs were identified: “hepatic cytolysis” (ROR 297.93), “drug reaction with eosinophilia and systemic symptoms” (ROR 92.54), “acute kidney injury” (ROR 20.99), “hypotension” (ROR 14.10), “no adverse event” (ROR 9.48), “product use in unapproved indication” (ROR 8.81), “respiratory failure” (ROR 6.55), “product use issue” (ROR 6.00) and “off-label use” (ROR 2.86). Detailed information is provided in Tables 3 and 4. The distribution of the number of AEs reported according to PTs level is shown in Fig. 2, and the Venn diagram of PTs level AEs screened according to four algorithms is shown in Fig. 3.
The distribution of the number of AEs reported according to PTs level.
The Venn diagram of PTs level AEs screened according to four algorithms including ROR, PRR, MGPS and BCPNN.
Sensitivity analysis
Over the past 5 years, a subgroup analysis was conducted on AEs involving daptomycin as the primary suspected drug in infants under 1 year old, involving 14 SOCs. The 3 most frequently reported systems were “injury, poisoning, and procedural complications” (ROR 1.36), “hepatobiliary disorders” (ROR 10.98) and “general disorders and administration site conditions” (ROR 0.98). However, based on the ROR algorithm for assessing signal strength, the SOCs with the top 3 strongest signal intensity were “hepatobiliary disorders” (ROR 10.98), “blood and lymphatic system disorders” (ROR 3.35) and “renal and urinary disorders” (ROR 2.07). Detailed information is provided in Table S1. We screened at the PTs level to identify AEs with at least 3 reported cases and positive risk signals, totaling 4 types, as shown in Table S3. The most frequently reported was “hepatic cytolysis” (ROR 226.08), followed by “drug reaction with eosinophilia and systemic symptoms” (ROR 110.04), “product use in unapproved indication” (ROR 5.83) and “product use issue” (ROR 5.23). Table S2 ranks AEs at the SOCs and PTs levels according to signal strength using the ROR algorithm.
Discussion
Invasive infections caused by gram-positive bacteria, especially drug-resistant infections such as methicillin-resistant Staphylococcus aureus (MRSA) and vancomycin-resistant Enterococcus faecium (VRE), are not uncommon in the pediatric population and challenging to treat32,33,34,35. Infants diagnosed with MRSA infections are typically treated with vancomycin36,37,38. However, vancomycin therapy requires therapeutic drug monitoring and may contribute to renal insult, which is not optimal for infants under 1 year old. In this setting, daptomycin is an attractive therapeutic alternative to vancomycin for treating MRSA or VRE39,40.
Small studies have begun to demonstrate the safety, efficacy, and pharmacokinetics of daptomycin in children, but there are limited data for daptomycin in younger infants and neonates11,12,19,20,41,42,43,44,45. Studies show that daptomycin clearance (CL) in this population of young infants was similar to the CL observed in 2–6 year old children and greater than the CL observed in older children and adults10,46. In this case, a higher dose is required. In addition, the FDA label cautions that daptomycin for injection is not recommended in pediatric patients younger than 1 year of age due to the risk of potential effects on muscular, neuromuscular, and/or nervous systems (either peripheral and/or central) observed in neonatal dogs.
Clinicians are typically cautious about using daptomycin in infants under 1 year old due to potential safety risks and insufficient data. In instances of severe MRSA infections, where no other effective treatment options are available, it is essential to obtain strict informed consent and ensure close monitoring of the patient. Consequently, this study is the first comprehensive exploration of AEs reports associated with daptomycin exposure in infants under 1 year old based on the FAERS database. Rather than inferring causality, the identified signals offer clinicians descriptive data to weigh potential benefits against observed risk signals when standard therapies are unavailable.
MedDRA was used to classify the daptomycin AEs signals. According to the ROR algorithm(a ≥ 3, the lower limit of the 95% CI of the ROR ≥ 1), the SOCs that included more signals were “hepatobiliary disorders”, “renal and urinary disorders”, “infections and infestations”, “vascular disorders”. In addition to fulfilling the signal screening criteria of the ROR algorithm, the top 3 signals also conform to the criteria of MHRA (PRR), BCPNN, and MGPS. This compliance across multiple algorithms significantly strengthens the statistical signals and enhances their credibility. Clinicians need to pay attention to the disproportionately reported AEs, especially the hepatobiliary system that not listed in the FDA label.
Further analysis of AEs at PTs level showed that 9 kinds of AEs meet the signal screening criteria of the ROR algorithm, and the first 8 kinds in the table are also combined with the other 3 algorithms. Among them, the top 4 signals are “hepatic cytolysis”, “drug reaction with eosinophilia and systemic symptoms (DRESS)”, “acute kidney injury (AKI)” and “hypotension”.
Although the ROR algorithm is most commonly used to evaluate the strength and possible correlation of AEs signals, some ROR confidence intervals are excessively wide due to the small sample size, increasing the uncertainty of analysis results. The absolute number of reports may be very low for rare adverse reactions, further widening the confidence interval. In order to make the research results more reliable, we added 3 other algorithms on the basis of the ROR algorithm, including PRR, MGPS and BCPNN. It can be seen from Table 4 that according to the criteria of the algorithms, the results and relevance ranking of the 4 algorithms are consistent. By integrating these diverse algorithms, we aimed to enhance the robustness and reliability of our findings, mitigating the limitations imposed by small sample sizes and wide confidence intervals.
DRESS and AKI are known adverse drug reactions on the FDA label, while “hepatic cytolysis” and “hypotension” are not mentioned in the warnings and precautions of the manual. However, clinical trial experience has reported abnormal liver function tests and hypotension in adult cSSSI patients who received daptomycin 4 mg/kg for injection.
Previously published data mining for AEs signals of daptomycin based on FAERS indicated that “antimicrobial susceptibility test resistant”, “eosinophilic pneumonia”, “staphylococcal bacteraemia”, and “endocarditis” were the top 4 AEs with the strongest signal risk in the top 20 reported cases47. “antimicrobial susceptibility test resistant”, “staphylococcal bacteraemia”, and “endocarditis” may be related to antibiotic abuse, failure to select appropriate drugs according to drug sensitivity, or the dose chosen is too small. “eosinophilic pneumonia” may be related to the direct damage and inflammation of alveolar epithelial cells caused by daptomycin by binding to pulmonary surfactant48.
Unlike previous reports, “hepatic cytolysis” and “hypotension” are risk signals for AEs of daptomycin in infants under 1 year old, both of which have not been reported in previous daptomycin signal mining study.
Since a large proportion of reports were submitted between 2021 and 2024, which may reflect improved completeness or reporting quality, we selected data from the last 5 years as a subgroup for sensitivity analysis. In the subgroup analysis, we analyzed AEs data from the use of daptomycin in infants under 1 year old over the past 5 years. Compared to the overall analysis, 2 SOCs were excluded: “metabolism and nutrition disorders” and “gastrointestinal disorders”. The SOC with the highest number of reports and the SOC with the highest signal strength in the subgroup analysis were consistent with the results of the overall analysis, namely “injury, poisoning, and procedural complications” and “hepatobiliary disorders”. At the PTs level, the number of AE types in the subgroup analysis was reduced by 5 compared to the overall analysis, specifically “acute kidney injury”, “hypotension”, “no adverse event”, “product use issue”, and “off-label use”. The most frequently reported AE and the AE with the highest signal strength were consistent with the overall analysis, both being “hepatic cytolysis”. This indicated that the analysis result of “hepatic cytolysis” as the AE with the highest signal strength and the most frequently reported was robust and reliable. Similarly, “drug reaction with eosinophilia and systemic symptoms” as the AE with the second-highest signal strength in both the subgroup analysis and the overall analysis, also warrants attention.
Hepatic cytolysis is the deterioration or destruction of liver cells, often signaled by elevated levels of liver enzymes in the bloodstream. Liver enzymes such as alanine transaminase (ALT) and aspartate transaminase (AST), which are usually confined within liver cells, are released into the blood when these cells are damaged or destroyed. Liver injury caused by daptomycin is uncommon, studies have shown that daptomycin is primarily eliminated by the kidney, the non-renal elimination does not involve cytochrome P450 enzymes, and does not inhibit or induce any of the key cytochrome P450 isoenzymes49,50, subjects with moderate hepatic impairment receiving daptomycin do not require an adjustment in dose regimen51. However, in clinical trials assessing daptomycin for treating complicated skin and skin structure infections, 3% of the participants exhibited abnormal results in liver function tests. In most instances, elevations in serum aminotransferase levels occur in 2% to 6% of patients receiving daptomycin, are generally mild-to-moderate, asymptomatic and self-limited. This indicator anomaly can occur anywhere between 2 and 14 days after initiation of daptomycin therapy, and frequently resolving when interruption of daptomycin52. Case reports of possible liver injury from daptomycin have been reported, typically accompanied by severe muscle injury with marked CK elevations19,46,53,54,55, some scholars believe that the mild-to-moderate serum aminotransferase elevations that occur during daptomycin therapy may represent muscle rather than liver injury and are likely due to direct toxicity to muscle. There are also isolated cases involving daptomycin induced hepatotoxicity in the absence of CK elevation, AKI, myositis56, this indicates that there is an alternative mechanism for daptomycin related liver injury. To our knowledge, this has not been described at the cellular level52. Among the many pharmaceuticals implicated in drug-induced liver injury (DILI), anti-antimicrobials are more frequently identified as suspect drug class57.
Liver injury caused by daptomycin may be related to mitochondrial dysfunction, which can increase reactive oxygen species, impair fatty acid oxidation and decrease cellular ATP production to result in cell necrosis58. Additionally, reactive metabolites of daptomycin may bind to intracellular proteins to create covalent adducts, disrupting protein homeostasis and cellular function, causing cellular stress and potentially leading to cellular damage59,60,61.
Data suggest that the incidence of pediatric DILI and the drugs involved are not similar to those encountered in adult medicine, possibly because clinical detection or evaluation is often based on liver biomarkers and their trends, such as liver ALT, AST, bilirubin, γ-glutamyltransferase, and alkaline phosphatase62. However, in neonates, these indicators have their limitations. For example, hyperbilirubinemia is very common in neonates because the liver is not fully mature and the ability to convert and excrete bilirubin is weak. Similarly, because metabolic bone disease is quite common in preterm infants, alkaline phosphatase is correspondingly elevated63. Data from a study of 1,860 newborns showed that liver enzyme activity was higher in very preterm infants than in late-gestational age newborns64. Therefore, the current analysis cannot determine the causal relationship between hepatolysis, a high-signal adverse event, and daptomycin, and can only provide some warning.
The incidence of hypotension caused by daptomycin is rare. That occurred in 2.4% of adult patients in the daptomycin for injection treatment phase 3 cSSSI trials mentioned in the FDA label, as far as we know, there are no other detailed reports. Hypotension can occur as a complication of the disease, such as shock from a severe infection, more research is needed to clarify its relationship with daptomycin.
DRESS and AKI were two of the top 3 signal-rated AEs in this study, which were known to the manual and reported in previous studies65,66,67,68,69,70,71.
DRESS is characterized by maculopapular rash, fever, lymphadenopathy, and eosinophilia. The liver was the most commonly involved internal organ, followed by the kidneys72,73,74,75. Antibiotics are well-recognized culprits of DRESS with multiple studies suggesting a rate of 15% to 74% where β-lactams and vancomycin are the most common drugs, and daptomycin has also been reported65. The median latency for antibiotics was 21.5 days, and systemic corticosteroids was the main treatment modality76,77. The pathogenesis of DRESS is still incompletely clear, it is generally believed to be an interplay of genetics, dysregulated CD8þ and Th2 T-cell immune responses, polymorphism of detoxification enzymes, and viral reactivation78,79.
AKI describes a sudden loss of kidney function that is determined on the basis of increased serum creatinine levels and reduced urinary output80. Multiple mechanisms are involved to explain how certain antibiotics can cause AKI. Most notably chemotherapeutics (such as cisplatin) and antimicrobials (such as amphotericin or aminoglycosides), have direct chemical nephrotoxicity81. Nephrotoxicity of daptomycin is rarely reported, and it is even considered a substitute for kidney damage caused by vancomycin use39,40. Crucially, while not all drugs that impact kidney function are nephrotoxic, any medication that diminishes renal performance can exacerbate AKI and result in AEs. Thus, limiting exposure to such drugs as much as possible is imperative82.
The most common daptomycin AEs in other analysis was “increased CPK”, “eosinophilic pneumonia”, and “rhabdomyolysis”, all have been reported in the literature47. However, in our analysis, “off-label use”, “hepatic cytolysis” and “hypotension” are the most common AEs of daptomycin that are not listed on the drug labels included. As observed in the case reports available, the administration of daptomycin in infants under 1 year of age has resulted in various outcomes, including elevation of both CK and liver enzymes46,54,55,83, abnormalities in only one of these indicators83,84,85,86, or no AEs at all, even with doses reaching up to 12–15 mg/kg10,12,84,87,88,89,90,91,92,93. Neither our analysis nor the case reports found any neurological abnormalities, nerve conduction velocity measurement was performed in the case report, and no neurological symptom or sign was detected in any infant, which was found to be normal89. In addition to case reports in the target population, no neurologic AEs were reported in clinical trials in the infants, which is consistent with the findings of clinical trials in the pediatric population6,10,11,53,55,93. In contrast, clinical trials in adults have reported neurologic disturbances, including decreased nerve conduction velocities, sensory abnormalities, and Bell’s palsy, suggesting the presence of peripheral neuropathy94,95. However, the majority of its symptoms are self-limiting and resolve without the need to discontinue the drug. Noteworthy, infants are unable to communicate symptoms verbally, and often in the arms, it may therefore be not easy to detect the presence of any neurologic AEs11. Besides, the long-term effects of drugs on the developing brain may not become apparent until later in life, requiring long-term monitoring of drugs that affect the central nervous system. However, it is burdensome and expensive to follow children and their families for years after discharge, so long-term studies are not easy to conduct96,97.
While hepatotoxicity and myotoxicity are rare AEs associated with daptomycin therapy, they can result in severe and potentially fatal outcomes52. Clinicians should monitor liver function tests, renal function, and CK level in patients receiving daptomycin treatment and remain vigilant about these possible AEs.
Multiple studies have shown that infants and young children have a higher clearance of daptomycin, and larger dose are required to achieve antimicrobial efficacy11,18. Furthermore, published literatures indicated that administering reasonably high doses was safe for infants and young children; however, it is recommended that the dosage should not exceed 15 mg/kg. A female infant born at 29 weeks complicated by sepsis, was treated by daptomycin up to 15 mg/kg. During this period the patient exhibited moderately increased values of transaminases, while renal functiontests and CPK values were not affected90. Relevant PK analysis was observed CPK was one of the important safety parameters, and the risk of myotoxicity was significantly increased at trough concentrations exceeded 24.3 mg/L98,99. It is recommended to conduct liver and kidney function tests 1–2 times a week for infants under 1 year of age receiving daptomycin, and to conduct therapeutical drug monitoring after the third dose to ensure that the valley concentration is not higher than 24.3 mg/L to reduce the risk of using daptomycin in this special population.
There are several limitations of this study. Given that the FAERS database relies on spontaneous reporting, it is susceptible to data omissions and loss of information. First of all, in this study’s early data processing stage, if the reporter did not fill in the patient’s age, the report would not be included in this study so that the sample size would be smaller. Secondly, due to the absence of key time points such as the start date of administration, end date of administration, and date of the AEs onset, it is impossible further to analyze the potential relationship between AEs and time. Similarly, the lack of weight reporting also affected the relationship between AEs and weight in the further analysis. In addition, 15% of the outcomes in our data analysis were “other serious” and 17.5% were “unreported”, which had a certain impact on our research results. However, the primary focus of this study is on AEs reported with daptomycin as the primary suspected drug, rather than the relationship between daptomycin and the outcome. This is because outcome is associated with more confounding factors, such as disease progression and treatment regimens, and it is not equate to the medical severity grading of the AEs themselves100. Furthermore, although 45% of the administration routes are unknown in our data analysis, it is well known that daptomycin is currently only available in intravenous formulations. This is because daptomycin has a large molecular weight and extremely low oral bioavailability, making it impossible to be effectively absorbed through the intestines. As a result, the 45% of unknown administration routes can be assumed to be intravenous administration. It is important to note that differences in the nature and number of AEs between preterm and full-term infants could not be analyzed due to lack of information to distinguish between preterm and preterm births. Moreover, the association between drug and AEs reported in FAERS can be obscured by comorbidities and concurrent medications, making it impossible to definitively establish a causal relationship through this system alone. Additionally, the sample size of this study is relatively small, the research results should be interpreted with caution and require validation through larger clinical trials.
Conclusion
This study suggests a potential association between daptomycin used in infants under 1 year old and AEs such as hepatic cytolysis, DRESS. The high-signal AEs of daptomycin in infants under 1 year of age differ from the most common daptomycin AEs in other analysis, and these differences need to be monitored in clinical treatment. It is important to monitor both liver function tests, renal function and CK level in younger infants treated with daptomycin. A more comprehensive understanding of the AEs associated with daptomycin in younger infants was evaluated using the FAERS database based on real-world data in our analysis, providing insights to support rational clinical decision-making.
Data availability
The corresponding authors are willing to provide the data related to this manuscript upon reasonable request.
References
Heidary, M. et al. Daptomycin. J. Antimicrob. Chemother. 73, 1–11 (2018).
Hawkey, P. M. Pre-clinical experience with daptomycin. J. Antimicrob. Chemother. 62 (Suppl 3), iii7–14 (2008).
Kanafani, Z. A. & Corey, G. R. Daptomycin: a rapidly bactericidal lipopeptide for the treatment of Gram-positive infections. Expert Rev. anti-infective Therapy. 5, 177–184 (2007).
Sader, H. S., Farrell, D. J., Flamm, R. K. & Jones, R. N. Daptomycin activity tested against 164457 bacterial isolates from hospitalised patients: summary of 8 years of a worldwide surveillance programme (2005–2012). Int. J. Antimicrob. Agents. 43, 465–469 (2014).
Streit, J. M., Jones, R. N. & Sader, H. S. Daptomycin activity and spectrum: a worldwide sample of 6737 clinical Gram-positive organisms. The J. Antimicrob. Chemotherapy 53, 2369 (2004).
Iwata, S., Koyama, H. & Murata, Y. Efficacy and safety of daptomycin in Japanese pediatric participants with complicated skin and soft tissue infections or bacteremia caused by gram-positive Cocci. J. Infect. Chemotherapy: Official J. Japan Soc. Chemother. 28, 406–412 (2022).
Cobos-Carrascosa, E. et al. Staphylococcus aureus bacteremia in children: changes during eighteen years. Pediatr. Infect. Dis. J. 34, 1329–1334 (2015).
Spaulding, A. B. et al. Epidemiology of bloodstream infections in hospitalized children in the united States, 2009–2016. Clin. Infect. Diseases: Official Publication Infect. Dis. Soc. Am. 69, 995–1002 (2019).
Carvalhaes, C. G. et al. Activity of tedizolid and comparator agents against Gram-positive isolates causing skin and skin structure infections in pediatric patients in united States hospitals (2015–2019). Pediatr. Infect. Dis. J. 41, 731–735 (2022).
Abdel-Rahman, S. M. et al. Single-dose pharmacokinetics of daptomycin in children with suspected or proved gram-positive infections. Pediatr. Infect. Dis. J. 27, 330–334 (2008).
Bradley, J. S., Benziger, D., Bokesch, P. & Jacobs, R. Single-dose pharmacokinetics of daptomycin in pediatric patients 3–24 months of age. Pediatr. Infect. Dis. J. 33, 936–939 (2014).
Cohen-Wolkowiez, M., Watt, K. M., Hornik, C. P., Benjamin, D. K. Jr. & Smith, P. B. Pharmacokinetics and tolerability of single-dose daptomycin in young infants. Pediatr. Infect. Dis. J. 31, 935–937 (2012).
Ishii, M., Orito, Y., Shiomi, M., Wrishko, R. E. & Yoshitsugu, H. Pharmacokinetics of intravenous daptomycin in Japanese pediatric patients: pharmacokinetic comparisons supporting dosing recommendations in Japanese pediatric patients. J. Infect. Chemotherapy: Official J. Japan Soc. Chemother. 29, 592–598 (2023).
Maurille, C. et al. Pharmacokinetics and safety of daptomycin administered subcutaneously in healthy volunteers: a single-blinded randomized crossover trial. J. Antimicrob. Chemother. 79, 3016–3022 (2024).
Olney, K. B., Howard, J. I. & Burgess, D. S. Daptomycin dose optimization in pediatric Staphylococcus aureus bacteremia: a Pharmacokinetic/Pharmacodynamic investigation. J. Clin. Pharmacol. 64, 860–865 (2024).
Zhang, Y. et al. Assessment of the impact of renal impairment on systemic exposure of new molecular entities: evaluation of recent new drug applications. Clin. Pharmacol. Ther. 85, 305–311 (2009).
Tally, F. P. et al. Daptomycin: a novel agent for Gram-positive infections. Expert Opin. Investig. Drugs. 8, 1223–1238 (1999).
Persha, H. et al. Real-World clinical characteristics and outcomes with daptomycin use in pediatric patients: a retrospective case series. Antibiotics (Basel Switzerland) 13, 4526 (2024).
Asfour, S. S., Asfour, R. S., Khalil, T. M. & Al-Mouqdad, M. M. The use of daptomycin in the treatment of persistent Coagulase-Negative Staphylococcal sepsis in premature infants: a case series. J. Pediatr. Pharmacol. Therapeutics: JPPT : Official J. PPAG. 26, 92–98 (2021).
Mohzari, Y. et al. Safety and efficacy of daptomycin in neonates with Coagulase-Negative staphylococci: case series analysis. Antibiotics (Basel Switzerland) 10, 1452 (2021).
Sakaeda, T., Tamon, A., Kadoyama, K. & Okuno, Y. Data mining of the public version of the FDA adverse event reporting system. Int. J. Med. Sci. 10, 796–803 (2013).
Administration, U. S. F. & a., D. FDA Adverse Event Reporting System (FAERS) Public Dashboard (2023). https://www.fda.gov/drugs/fdas-adverse-event-reporting-system-faers/fda-adverse-event-reporting-system-faers-public-dashboard.
Sakaeda, T., Kadoyama, K., Minami, K. & Okuno, Y. Commonality of drug-associated adverse events detected by 4 commonly used data mining algorithms. Int. J. Med. Sci. 11, 461–465 (2014).
Zhao, Y., Fei, L. & Duan, Y. Movement disorders related to antidiabetic medications: a real-world pharmacovigilance study. Prog. Neuro-psychopharmacol. Biol. Psychiatry. 135, 111128 (2024).
Wang, Z. et al. Exploring the correlation between cardiovascular adverse events and antidepressant use: a retrospective pharmacovigilance analysis based on the FDA adverse event reporting system database. J. Affect. Disord. 367, 96–108 (2024).
Hua, M. et al. Abemaciclib increases the risk of venous thromboembolism in breast cancer: integrate meta-analysis, pharmacovigilance database analysis, and in vitro validation. Cancer Treat. Rev. 130, 102827 (2024).
Chedid, V., Vijayvargiya, P. & Camilleri, M. Advantages and limitations of the federal adverse events reporting system in assessing adverse event reporting for Eluxadoline. Clin. Gastroenterol. Hepatol.: Off. Clin. Pract. J. Am. Gastroenterol. Assoc. 16, 336–338 (2018).
Ye, L., Zhou, H., Guo, G., Chen, M. & Zhang, J. Physiologically-based Pharmacokinetic modeling to predict the exposure and to assess pharmacodynamics of daptomycin in infants within 1 year old. Eur. J. Pharm. Sciences: Official J. Eur. Federation Pharm. Sci. 208, 107058 (2025).
Fusaroli, M., Raschi, E., Poluzzi, E. & Hauben, M. The evolving role of disproportionality analysis in pharmacovigilance. Exp. Opin. Drug Saf. 23, 981–994 (2024).
Hauben, M. & Zhou, X. Quantitative methods in pharmacovigilance: focus on signal detection. Drug Saf. 26, 159–186 (2003).
Ooba, N. & Kubota, K. Selected control events and reporting odds ratio in signal detection methodology. Pharmacoepidemiol. Drug Saf. 19, 1159–1165 (2010).
Rallis, D. et al. Molecular epidemiology clinical Manifestations, decolonization Strategies, and treatment options of Methicillin-Resistant Staphylococcus aureus infection in neonates. Pathogens (Basel Switzerland) 14, 4523 (2025).
Dong, Y., Glaser, K. & Speer, C. P. New threats from an old foe: Methicillin-Resistant Staphylococcus aureus infections in neonates. Neonatology 114, 127–134 (2018).
Ward, J. et al. Bloodstream infections in infants and children with congenital heart disease undergoing cardiac surgery. Am. J. Crit. Care: Official Publication Am. Association Critical-Care Nurses. 32, 157–165 (2023).
Furtado, I. et al. Enterococcus faecium and Enterococcus faecalis in blood of newborns with suspected nosocomial infection. Rev. Inst. Med. Trop. Sao Paulo. 56, 77–80 (2014).
Hill, L. F. et al. Optimised versus standard dosing of Vancomycin in infants with Gram-positive sepsis (NeoVanc): a multicentre, randomised, open-label, phase 2b, non-inferiority trial. Lancet Child. Adolesc. Health. 6, 49–59 (2022).
Hill, L. F. et al. An optimised dosing regimen versus a standard dosing regimen of Vancomycin for the treatment of late onset sepsis due to Gram-positive microorganisms in neonates and infants aged less than 90 days (NeoVanc): study protocol for a randomised controlled trial. Trials 21, 329 (2020).
Plan, O. et al. Continuous-infusion Vancomycin therapy for preterm neonates with suspected or documented Gram-positive infections: a new dosage schedule. Arch. Dis. Child. Fetal Neonatal Ed. 93, F418–421 (2008).
Burgess, L. D. & Drew, R. H. Comparison of the incidence of vancomycin-induced nephrotoxicity in hospitalized patients with and without concomitant piperacillin-tazobactam. Pharmacotherapy 34, 670–676 (2014).
Moenster, R. P. et al. Acute renal failure associated with Vancomycin and β-lactams for the treatment of osteomyelitis in diabetics: piperacillin-tazobactam as compared with cefepime. Clin. Microbiol. Infection: Official Publication Eur. Soc. Clin. Microbiol. Infect. Dis. 20, O384–389 (2014).
Principi, N., Caironi, M., Venturini, F., Pani, L. & Esposito, S. Daptomycin in paediatrics: current knowledge and the need for future research. J. Antimicrob. Chemother. 70, 643–648 (2015).
Karageorgos, S. A., Miligkos, M., Dakoutrou, M. & Tsioutis, C. Clinical Effectiveness, safety Profile, and pharmacokinetics of daptomycin in pediatric patients: A systematic review. J. Pediatr. Infect. Dis. Soc. 5, 446–457 (2016).
Dvorchik, B. & Damphousse, D. Single-dose pharmacokinetics of daptomycin in young and geriatric volunteers. J. Clin. Pharmacol. 44, 612–620 (2004).
Dvorchik, B. H., Brazier, D., DeBruin, M. F. & Arbeit, R. D. Daptomycin pharmacokinetics and safety following administration of escalating doses once daily to healthy subjects. Antimicrob. Agents Chemother. 47, 1318–1323 (2003).
Arbeit, R. D., Maki, D., Tally, F. P., Campanaro, E. & Eisenstein, B. I. The safety and efficacy of daptomycin for the treatment of complicated skin and skin-structure infections. Clin. Infect. Diseases: Official Publication Infect. Dis. Soc. Am. 38, 1673–1681 (2004).
Vonasek, B. J., Samuel, A. M., Henderson, S. L., Strayer, J. R. & Bogenschutz, M. C. Safety and treatment outcomes of infants and children treated with daptomycin: Six-Year experience from a pediatric academic medical center. Clin. Pediatrics 2024, 99228241242186 (2024).
Chen, J. J., Huo, X. C., Wang, S. X., Wang, F. & Zhao, Q. Data mining for adverse drug reaction signals of daptomycin based on real-world data: a disproportionality analysis of the US food and drug administration adverse event reporting system. Int. J. Clin. Pharm. 44, 1351–1360 (2022).
Hagiya, H. et al. Myopathy and eosinophilic pneumonia coincidentally induced by treatment with daptomycin. Intern. Med. (Tokyo, Japan). 54, 525–529 (2015).
Gregoire, N. et al. Clinical pharmacokinetics of daptomycin. Clin. Pharmacokinet. 60, 271–281 (2021).
Oleson, F. B., Berman, C. L. & Li, A. P. An evaluation of the P450 Inhibition and induction potential of daptomycin in primary human hepatocytes. Chemico-Biol. Interact. 150, 137–147 (2004).
Dvorchik, B. Moderate liver impairment has no influence on daptomycin pharmacokinetics. J. Clin. Pharmacol. 44, 715–722 (2004).
Janda, A. & Jogendra, M. R. D. A case report and literature review of daptomycin-induced liver injury. IDCases 14, e00452 (2018).
Bradley, J. et al. Daptomycin for complicated skin infections: a randomized trial. Pediatrics 139, 4523 (2017).
Bradley, J. S. et al. Daptomycin for pediatric Gram-Positive acute hematogenous osteomyelitis. Pediatr. Infect. Dis. J. 39, 814–823 (2020).
Arrieta, A. C. et al. Randomized multicenter study comparing safety and efficacy of daptomycin versus Standard-of-care in pediatric patients with Staphylococcal bacteremia. Pediatr. Infect. Dis. J. 37, 893–900 (2018).
Bohm, N. et al. Case report and cohort analysis of drug-induced liver injury associated with daptomycin. Antimicrob. Agents Chemother. 58, 4902–4903 (2014).
Chen, M. et al. DILIrank: the largest reference drug list ranked by the risk for developing drug-induced liver injury in humans. Drug Discovery Today. 21, 648–653 (2016).
Mihajlovic, M. & Vinken, M. Mitochondria as the target of hepatotoxicity and Drug-Induced liver injury: molecular mechanisms and detection methods. Int. J. Mol. Sci. 23, 15526 (2022).
Yuan, L. & Kaplowitz, N. Mechanisms of drug-induced liver injury. Clin. Liver Dis. 17, 507–518 (2013). vii.
Andrade, R. J. et al. Drug-induced liver injury. Nat. Rev. Dis. Primers 5, 58 (2019).
Chen, M., Suzuki, A., Borlak, J., Andrade, R. J. & Lucena, M. I. Drug-induced liver injury: interactions between drug properties and host factors. J. Hepatol. 63, 503–514 (2015).
Shi, Q. et al. Drug-Induced liver injury in children: clinical Observations, animal Models, and regulatory status. Int. J. Toxicol. 36, 365–379 (2017).
Victor, S., Dickinson, H. & Turner, M. A. Plasma aminotransferase concentrations in preterm infants. Arch. Dis. Child. Fetal Neonatal Ed. 96, F144–145 (2011).
Rayyan, M., Devlieger, H., Jochum, F. & Allegaert, K. Short-term use of parenteral nutrition with a lipid emulsion containing a mixture of soybean oil, Olive oil, medium-chain triglycerides, and fish oil: a randomized double-blind study in preterm infants. JPEN J. Parenter. Enteral. Nutr. 36, 81s–94s (2012).
Streifel, A. C., Varley, C. D., Ham, Y., Sikka, M. K. & Lewis, J. S. 2 nd. The challenge of antibiotic selection in prosthetic joint infections due to Corynebacterium striatum: a case report. BMC Infect. Dis. 22, 290 (2022).
Nishimura, Y. Daptomycin-Related rhabdomyolysis complicated by severe hyperkalemia and acute kidney injury. Cureus 14, e29764 (2022).
Patek, T. M., Teng, C., Kennedy, K. E., Alvarez, C. A. & Frei, C. R. Comparing acute kidney injury reports among antibiotics: a pharmacovigilance study of the FDA adverse event reporting system (FAERS). Drug Saf. 43, 17–22 (2020).
Kazory, A., Dibadj, K. & Weiner, I. D. Rhabdomyolysis and acute renal failure in a patient treated with daptomycin. J. Antimicrob. Chemother. 57, 578–579 (2006).
Abraham, G., Finkelberg, D. & Spooner, L. M. Daptomycin-induced acute renal and hepatic toxicity without rhabdomyolysis. Annals Pharmacotherapy. 42, 719–721 (2008).
Odero, R. O., Cleveland, K. O. & Gelfand, M. S. Rhabdomyolysis and acute renal failure associated with the co-administration of daptomycin and an HMG-CoA reductase inhibitor. J. Antimicrob. Chemother. 63, 1299–1300 (2009).
Ramírez-Martín, R., Pérez-Rodríguez, P., Rico-Nieto, A. & Mauleón-Ladrero, C. Early onset rhabdomyolysis and acute renal failure associated with the administration of daptomycin. A case report and literature review. Revista Esp. De Geriatria Y Gerontologia. 53, 362–363 (2018).
Kardaun, S. H. et al. Drug reaction with eosinophilia and systemic symptoms (DRESS): an original multisystem adverse drug reaction. Results from the prospective RegiSCAR study. Br. J. Dermatol. 169, 1071–1080 (2013).
Soria, A. et al. Drug reaction with eosinophilia and systemic symptoms May occur within 2 weeks of drug exposure: a retrospective study. J. Am. Acad. Dermatol. 82, 606–611 (2020).
Husain, Z., Reddy, B. Y. & Schwartz, R. A. DRESS syndrome: part I. Clinical perspectives. J. Am. Acad. Dermatol. 68, 693e691–693e614 (2013). quiz 706 – 698.
Awad, A., Goh, M. S. & Trubiano, J. A. Drug reaction with eosinophilia and systemic symptoms: a systematic review. J. Allergy Clin. Immunol. Pract. 11, 1856–1868 (2023).
Wolfson, A. R. et al. Drug reaction with eosinophilia and systemic symptoms (DRESS) syndrome identified in the electronic health record allergy module. J. Allergy Clin. Immunol. Pract. 7, 633–640 (2019).
Blumenthal, K. G., Patil, S. U. & Long, A. A. The importance of vancomycin in drug rash with eosinophilia and systemic symptoms (DRESS) syndrome. Allergy Asthma Proc. 33, 165–171 (2012).
Pirmohamed, M. et al. Carbamazepine-hypersensitivity: assessment of clinical and in vitro chemical cross-reactivity with phenytoin and oxcarbazepine. Br. J. Clin. Pharmacol. 32, 741–749 (1991).
Musette, P. & Janela, B. New insights into drug reaction with eosinophilia and systemic symptoms pathophysiology. Front. Med. 4, 179 (2017).
Kellum, J. A. et al. Acute kidney injury. Nat. Reviews Disease Primers. 7, 52 (2021).
Costa, Silva, V. T., Marçal, L. J. & Burdmann, E. A. Risk factors for Vancomycin nephrotoxicity: still a matter of debate*. Crit. Care Med. 42, 2635–2636 (2014).
Ostermann, M. et al. Controversies in acute kidney injury: conclusions from a kidney disease: improving global outcomes (KDIGO) conference. Kidney Int. 98, 294–309 (2020).
Asfour, S. S., Aljobair, F., Abdelrahim, A. & Al-Mouqdad, M. M. High Alanine aminotransaminase associated with daptomycin use in a premature infant. J. mother. child. 25, 65–68 (2021).
Cohen-Wolkowiez, M., Smith, P. B., Benjamin, D. K. Jr., Fowler, V. G. Jr. & Wade, K. C. Daptomycin use in infants: report of two cases with peak and trough drug concentrations. J. Perinatology: Official J. Calif. Perinat. Association. 28, 233–234 (2008).
Beneri, C. A., Nicolau, D. P., Seiden, H. S. & Rubin, L. G. Successful treatment of a neonate with persistent vancomycin-resistant enterococcal bacteremia with a daptomycin-containing regimen. Infect. Drug Resist. 1, 9–11 (2008).
Gkoufa, A., Goutas, D., Sakellariou, S. & Cholongitas, E. A case of Daptomycin-Induced acute cholestatic hepatic injury with fatal outcomes. Am. J. Ther. 29, e671–e673 (2022).
Shigeta, H., Tanaka, K., Hoshina, T. & Kusuhara, K. Successful daptomycin therapy for a premature neonate with left-sided infective endocarditis caused by Staphylococcus epidermidis. Pediatr. Neonatol. 62, 456–457 (2021).
Chan, J. I., Noor, A., Clauss, C., Aggarwal, R. & Nayak, A. Methicillin-Resistant Staphylococcus aureus endovascular infection in a neonate: prolonged, Safe, and effective use of daptomycin and Enoxaparin. J. Pediatr. Pharmacol. Therapeutics: JPPT : Official J. PPAG. 25, 68–74 (2020).
Syrogiannopoulos, G. A., Michoula, A. N., Petinaki, E. & Grivea, I. N. Daptomycin use in children: experience with various types of infection and age groups. Pediatr. Infect. Dis. J. 36, 962–966 (2017).
Antachopoulos, C. et al. Serum levels of daptomycin in pediatric patients. Infection 40, 367–371 (2012).
Ardura, M. I. et al. Daptomycin therapy for invasive Gram-positive bacterial infections in children. Pediatr. Infect. Dis. J. 26, 1128–1132 (2007).
Hussain, A., Kairamkonda, V. & Jenkins, D. R. Successful treatment of meticillin-resistant Staphylococcus aureus bacteraemia in a neonate using daptomycin. J. Med. Microbiol. 60, 381–383 (2011).
Abdel-Rahman, S. M. et al. Single-dose pharmacokinetics and tolerability of daptomycin 8 to 10 mg/kg in children aged 2 to 6 years with suspected or proved Gram-positive infections. Pediatr. Infect. Dis. J. 30, 712–714 (2011).
Byren, I. et al. Randomized controlled trial of the safety and efficacy of daptomycin versus standard-of-care therapy for management of patients with osteomyelitis associated with prosthetic devices undergoing two-stage revision arthroplasty. Antimicrob. Agents Chemother. 56, 5626–5632 (2012).
Fowler, V. G. Jr. et al. Daptomycin versus standard therapy for bacteremia and endocarditis caused by Staphylococcus aureus. N. Engl. J. Med. 355, 653–665 (2006).
Marlow, N. et al. Assessment of long-term neurodevelopmental outcome following trials of medicinal products in newborn infants. Pediatr. Res. 86, 567–572 (2019).
Marlow, N. Measuring neurodevelopmental outcome in neonatal trials: a continuing and increasing challenge. Arch. Dis. Childhood Fetal Neonatal Ed. 98, F554-558 (2013).
Samura, M. et al. Population Pharmacokinetic analysis and dosing optimization based on unbound daptomycin concentration and Cystatin C in Nonobese elderly patients with hypoalbuminemia and chronic kidney disease. Pharm. Res. 38, 1041–1055 (2021).
Tally, F. P. & DeBruin, M. F. Development of daptomycin for gram-positive infections. J. Antimicrob. Chemother. 46, 523–526 (2000).
Kumar, A. The newly available FAERS public dashboard: implications for health care professionals. Hosp. Pharm. 54, 75–77 (2019).
Acknowledgements
The authors would like to thank the China Pharmacist Association Clinical Practice Research Capability Empowerment Program - Specialized Training Program on Bleeding and Coagulation Disorders.
Funding
This work has been supported by the Fujian Natural Science Foundation project (2022J011037).
Author information
Authors and Affiliations
Contributions
L.Y. and G.G. contributed equally to the research and are considered co-first authors. L.Y., G.G. and J. Z. were involved in designing the study. L.Y., H.Z., M.C. and Z.F. took charge of collecting and analyzing the data. L.Y. authored the initial draft of the manuscript, while J.Z. provided the necessary revisions. All authors contributed to the article and approved the submitted version.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Ye, L., Guo, G., Zhou, H. et al. A pharmacovigilance study of daptomycin use in infants under 1 year old based on the FDA adverse event reporting system. Sci Rep 15, 40632 (2025). https://doi.org/10.1038/s41598-025-24217-y
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-24217-y
Keywords
This article is cited by
-
Daptomycin use in infants: safety signals identified
Reactions Weekly (2025)