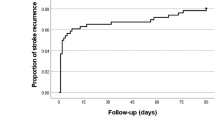
Abstract
Rosacea is a chronic skin disease with unclear causes, involving skin barrier issues and lipid changes. This study analyzed lipid profiles in rosacea patients’ skin surface lipids (SSLs) to explore potential causes. The study included 10 rosacea patients and 10 healthy controls in Beijing. Transepidermal water loss (TEWL) was evaluated to assess the skin barrier function. Liquid chromatography coupled with tandem mass spectrometry (LC-MS/MS) and multivariate data analysis were employed to investigate SSLs alterations. The results showed that rosacea patients had higher TEWL values than healthy controls (16.59 ± 3.95 versus 7.87 ± 2.52, p < 0.01). LC-MS/MS revealed significant differences in the lipidomic profiles and identified 48 species of SSLs that differed between the two groups. Triacylglycerol (TAG) were particularly abundant and varied in rosacea patients, which had 8 down-regulated differential lipids and 28 up-regulated lipids in rosacea patients. TAG, diacylglycerols (DAG), lysophosphatidylcholine (LPC), phosphatidylcholine (PC) were positively correlated with TEWL value (p < 0.05), but FFA were negatively correlated with TEWL value (p < 0.05). The present study indicated that patients with rosacea have impaired skin barrier function and altered SSL composition. Certain SSL species identified in this study may be potential targets for future studies on the pathogenesis and treatment of rosacea.
Similar content being viewed by others
Introduction
Rosacea is a common, chronic disorder that can present with a variety of cutaneous or ocular manifestations. Cutaneous involvement primarily affects the central face, with findings such as persistent centrofacial redness, papules, pustules, flushing, telangiectasia, and phymatous skin changes. The pathways that lead to the development of rosacea are not well understood. Proposed contributing factors include immunity, genetic factors, microorganisms, ultraviolet radiation (UV), neurovascular dysfuction and barrier disfuction1,2,3. Skin surface lipids, primarily composed of ceramides (Cer), fatty acids (FFA), triglycerides (TAG), and cholesterol, originate from sebaceous glands-secreted lipids and epidermal disintegration lipids4. The equilibrium of the skin surface including their relative abundance, composition, molecular organization, dynamics, and intricate interactions, is crucial for maintaining healthy skin5. Consequently, even subtle alterations in the properties or overall profile of skin surface lipids have been associated with the aetiology of various common skin diseases such as atopic dermatitis, psoriasis, xerosis, ichthyosis and acne6. Our clinical observations indicate an elevated level of sebum production on the facial regions of individuals diagnosed with rosacea. However, the role of skin surface lipids in the pathogenesis of rosacea remains underexplored. This study aims at investigating the differences in the lipidome of facial sebumbetween rosacea patients and the healthy controls residing in Beijing area, thereby providing a scientific basis for clinical treatment strategies.
Results
Characteristics of the participants
The clinical characteristics of the patients enrolled in this study and the control group are summarized in Table 1. All participants were recruited between July 2024 and November 2024. Among the 10 rosacea patients (6 with erythematous type and 4 with papulopustular type), 3 were male and 7 were female, with a mean age of 37.70 ± 8.03 years. In the control group of 10 individuals, 3 were male and 7 were female, with a mean age of 37.6 ± 4.30 years. There were no significant differences in age (p = 0.97268) and gender between the two groups (Table 1).
Skin barrier function
TEWL is frequently employed to evaluate the barrier function of human skin. An increase in TEWL generally indicates impairment of the skin barrier7. In this study, rosacea patients exhibited significantly higher TEWL values compared to healthy controls (16.59 ± 3.95 versus 7.87 ± 2.52, p < 0.01) (Table 1). These findings suggest that rosacea is closely linked to dysfunction of the skin barrier8.
SSL profiles
Total lipids were collected from the surface of the left cheek of rosacea patients and healthy controls using the sebutape patches and subsequently analyzed by LC-MS/MS. No significant difference was observed in the relative average content of total lipids between the rosacea group and the healthy control group (P = 0.218) (Fig. 1a). A total of 1082 unique lipids were identified in the lipid extracts. Based on the MS-Dial Lipidomics MSP database, four main classes of identified lipids were classified, and their profiles were characterized in terms of relative abundance: 42.5% glycerolipids (GL), 40.0% glycerophospholipids (GP), 11.7% sphingolipids (SP), and 5.7% fatty acyls (FA) (Fig. 1b). These four main classes were further subdivided into 18 subclasses, with TAG、phosphatidylcholine (PC), sphingomyelin (SM), FFA exhibiting the highest relative abundance within their respective main classes (GL, GP, SP and FA) (Fig. 1c).
Analysis of lipids identified in the lipid extracts. (a) Relative average content of total lipids in samples. (b) The proportion of main classes of identified lipids.FA, fatty acyls; GL, glycerolipids; GP, glycerophospholipids; SP, sphingolipids. (c) The proportion of lipid subclasses in each main class.
Differences in SSLs
Multivariate data analysis of the 1082 identified lipids using PLS-DA demonstrated a distinct separation between rosacea patients and healthy controls (R2 = 0.934, Q2 = 0.822; Fig. 2). These findings suggest that variations in the lipid composition of SSLs may be associated with the pathogenesis of rosacea. Based on the PLS-DA analysis and Q-value (false discovery rate) evaluation, several parameters were employed to identify lipid species with significant differences between the rosacea patients and healthy controls. A total of 48 differential lipids were screened using criteria of VIP value > 1 and p value < 0.05. These included 38 GL (79.2%), 8 GP (16.7%) and 2 FA (4.2%) (Fig. 3a). Compared with healthy controls, rosacea patients exhibited 10 down-regulated and 38 up-regulated differential lipids (Table 2). These 48 SSLs belonged to 5 subclasses (Fig. 3b). TAG, the most abundant and differentially expressed lipids in the GL subclass, showed 8 down-regulated and 28 up-regulated differential lipids in rosacea patients. All 2 diacylglycerols (DAG) in the GL subclass were up-regulated in rosacea patients. In the GP main class, lysophosphatidylcholine (LPC) and PC were up-regulated. While FFA, including docosahexaenoic acid (DHA, FFA 22:6), were all down-regulated in rosacea patients.
PLS-DA score plot of SSL from rosacea patients and healthy controls. SSL profiles of rosacea patients (red dots) and controls (blue dots) are obviously separated.R2 = 0.934, Q2 = 0.822.
Analysis of differential lipids in rosacea patients and healthy controls. (a) The proportion of main classes of differential lipids. (b) The counts of up- or down-regulated differential lipids in rosacea patients versus healthy controls at the subclass level. (c) The heatmap showing the cluster analysis of differential lipids. The color key represrnts the fold change of up- (red) or down-regulated (blue) differential lipids in rosacea patients versus healthy controls. The class represents the group of rosacea patients (patient, the red block) and healthy controls(control, the white block), the green block is used to distinguish different lipids.
Subsequently, cluster analysis was performed for the differential lipids, and the heatmap demonstrated a clear distinction in lipid composition between rosacea patients and healthy controls (Fig. 3c).
Associations between SSL alterations and skin barrier damage
To investigate whether certain SSLs components are associated with the impaired skin barrier function in rosacea patients, the correlations between TEWL and lipids categories were analyzed. The results demonstrated that FFA were negatively correlated with TEWL, whereas TAG, DAG, LPC and PC were positively correlated (Table 3).
Discussion
Rosacea is a common, chronic disorder that can present with a variety of cutaneous or ocular manifestations. The pathogenesis of rosacea is multifactorial, encompassing a range of triggers that elicit both inflammatory and vascular responses. In addition to genetic predispositions, various factors such as microbial agents (including Demodex mites), ultraviolet radiation, dietary influences, neurovascular dysregulation, and psychologicalstress, and immune system dysfunction, have been implicated in its development1,2,3. The stratum corneum lipid matrix, along with sebum- derived lipids from sebaceous glands, forms a highly complex and unique blend of skin surface lipids6. This lipid composition exhibits significant heterogeneity and provides the skin with its essential protective barrier9. Investigations into skin surface lipids in rosacea dates back to the 20th century. However, only one study has specifically examined skin lipidomics in rosacea patients: conducted by Pye et al., this study involved a limited cohort of patients and analyzed lipid components, particularly cholesterol, FFA, TAG, esters, and squalene10. Their findings indicated no differences between rosacea patients and controls, nor between genders or based on disease severity10. Another noteworthy study exploring the skin barrier in rosacea revealed down regulation of the ABCA12 gene, which encodes the lipid transporter ABCA12, a key player in lipid lamellae formation11.
An increasing body of evidence suggests that an abnormal skin barrier structure and function are associated with the etiology and pathological processes of rosacea12,13. In this study, we also reached the same conclusion. It was found that rosacea patients exhibit a reduced skin barrier function compared to asymptomatic control subjects. The relative contents of TAG, DAG, LPC, and PC in rosacea patients were positively correlated with their TEWL, whereas FFA exhibited a negative correlation with TEWL. Impairment of the epidermal barrier function allows external irritants to penetrate the skin, leading to production of proinflammatory mediators such as TSLP (Thymic stromal lymphopoietin), which is an IL-7–like cytokine, IL-4, IL-13, IL-25, and IL-33 14,15. Thus, these increased inflammatory cytokines induce immune cell accumulation, which leads to formation of nitrogen oxide and reactive oxygen species16.
TAG was the most differentially expressed lipids in our study. It is one of the primary components of sebum on the skin surface17. When the synthesis of TAG lipids decreases or their breakdown becomes excessive, transepidermal water loss increases, weakening the skin’s barrier function and making it more susceptible to invasion by harmful external substances, thereby triggering an inflammatory response18,19. Literature reports indicate that locally supplementing or regulating the structure of TAG (such as synthesizing TAG with specific fatty acid chains) can improve the skin barrier, and alleviate the symptoms of acne, atopic dermatitis (AD) and other diseases6,20. This approach is also applicable to rosacea and represents a promising new direction for its treatment. We also observed elevated levels of certain DAGs in rosacea patients, which may be attributed to the hydrolysis of skin surface TAGs or possibly due to increased sebaceous gland secretion.
The FFA in the surface lipids of the skin constitute a critical component of the lipid matrix of the stratum corneum6,21. According to the literature, long-chain FFA play a significant role in maintaining the stability of the skin barrier. The carbon chain length of FFA directly influences the density and hydrophobicity of the lipid layer22. In patients with papulopustular rosacea, an abnormal composition of sebaceous FFA has been observed, characterized by reduced levels of long-chain saturated FFA22. This study, identified a significant reduction in a long-chain FFA with a carbon chain length of 40. This reduction may enhance the fluidity of the lipid layer, thereby compromising the skin barrier function in rosacea22. In our study, the lesional skin surface of rosacea patients likewise exhibited a relative decrease in DHA. Jia et al. further demonstrated in a reconstructed human epidermis model that a 48-hour exposure to 50 µM DHA raised filaggrin and loricrin expression by 1.8- to 2.1-fold, while suppressing TSLP, IL-6 and TNF-α secretion by 30–50%, and significantly reduced TEWL23.
Abnormal levels of LPC in the surface lipids of the skin may contribute to disease progression by modulating keratinocytes function, immune responses, and skin barrier integrity18,24. Elevated LPC levels could potentially exacerbate abnormal hair follicle keratinization and inflammatory responses through alterations in the FFA composition of sebum, such as increased ratios of linoleic acid/linolenic acid18. Furthermore, LPC has been shown to induce T-lymphocyte chemotaxis and thereby help to maintain the chronic inflammation found in psoriatic epidermis25. In another study, it was realized that LPC upregulated IL17, recruited neutrophils more efficiently, and exacerbated DNFB (2,4-Dinitrofluorobenzene)-induced skin inflammation26. Additionally, LPC promotes the inflammatory activation of CD4 + T cells via G protein-coupled receptor G2A signaling, which further compromises epidermal barrier function and exacerbates immune dysregulation27.
PC, a major constituent of cutaneous phospholipids, is abundant in the plasma membranes of keratinocytes and contributes to barrier integrity, lipid signalling and epidermal metabolism28. In this study, we observed for the first time that the levels of phosphatidylcholine (PC) and its hydrolysis product lysophosphatidylcholine (LPC) on the skin surface of patients with rosacea were significantly increased. This is consistent with the neurogenic inflammation, TRPV1 activation, and enhanced activity of phospholipase A2 (PLA2) that are often observed in rosacea 29. Which suggesting that local phospholipid metabolism is active and may participate in inflammation and barrier disorders through pro-inflammatory lipid signaling.It will be necessary in the future to further clarify the dynamic characteristics of PC at different stages of the disease. This might potentially become a lipid biomarker for evaluating the efficacy of the disease treatment.
In conclusion, this study demonstrates that the composition and structure of lipids on the skin surface of rosacea patients differ significantly from those of healthy controls, particularly in the abnormal structure composition of TAG. Moreover, these lipid differences may contribute to the skin barrier dysfunction. Furthermore, these lipid alterations are not only associated with skin barrier impairment but also closely linked to the onset and progression of rosacea. These findings will provide potential new targets for future research into the pathogenesis and treatment of rosacea. Although this study offers valuable insights into the skin surface lipidomics of rosacea, it has certain limitations. Firstly, although LC-MS/MS offers high sensitivity and broad metabolite coverage, the relatively small sample size and all subjects were from Beijing limits statistical power.Future studies with larger cohorts are needed to validate the robustness and reproducibility of the lipidomic alterations identified herein. Secondly, this study employed a cross-sectional design, changes observed in specific lipid species may reflect secondary effects of inflammation rather than primary disease drivers. Longitudinal or interventional studies are warranted to further explore the functional roles of these lipids in disease progression. Thirdly, although efforts were made to control for confounding factors such as sex, age, sampling site, and recent skincare habits, we cannot fully exclude the potential influence of individual lifestyle, diet, or skin microbiota differences on the lipid profiles. Future studies integrating multi-omics approaches, including skin microbiome and host metabolome analyses, may provide a more comprehensive understanding of the cutaneous microenvironment in rosacea.
Methods
Chemicals and reagents
Acetonitrile (ACN), methanol, isopropanol (IPA), and methyl tert-butyl ethe (MTBE) Optima™ LC/MS Grade were obtained from Thermo Fisher Scientific (Waltham, MA, USA). Sebutape was purchased from CuDerm Corporation (Dallas, TX, USA).
Participants
The study was approved by the Ethics Committee of Peking University International Hospital. All procedures adhered to relevant guidelines and regulations, and informed consent was obtained from all participants prior to enrollment. 20 participants from the Beijing area were enrolled in the study, including ten rosacea patients and ten healthy controls. All ten patients were diagnosed as rosacea by two identical dermatologists (refer to “Chinese Guidelines for Diagnosis and Treatment of Rosacea (2021 Edition)”). All 20 participants had not received any treatments or medications that could potentially interfere with the study assessment within the preceding 6 months. The exclusion criterion encompassed topical or oral drugs, physical treatments such as phototherapy, cardiovascular medications, antiepileptic drugs, antibiotics, and antipsychotic drugs. Additionally, none of the participants exhibited local skin lesions, including acne, eczema, melasma, psoriasis or scars, nor did they have any systemic diseases that might influence the skin condition. The study protocol ensured strict matching of participants based on demographic characteristics of sex and age.
TEWL measurement
Both healthy individuals and patients with rosacea were selected for examination. Transepidermal water loss (TEWL) measurements were conducted on the left cheek using a portable VapoMeter (TM300; CK, Cologne, Germany). All tests were conducted in standardized ambient conditions with average humidity of 50% and average temperature of 23 °C. Prior to measurement, participants’ faces were cleaned with warm water without any cosmetics applied. The skin was then allowed to acclimate for 20 min. Subsequently, the detection probe was positioned on the target area, three consecutive readings were obtained from the same site for each participant, which were subsequently averaged.
SSL sampling
Before sample collection, participants were instructed to acclimatize in a controlled environment (room temperature 23 °C and humidity 50%) for 30 min. Sebum was collected from an approximately 4cm2 area at the same site left cheek using Sebutape. Prior to sebum collection, the collection area was wiped with a 5% saline swab and one Sebutape patch was placed on the target site. The Sebutape patch was left in place for 10 min, and then removed to a sterile centrifuge tube using curved forceps. All samples were immediately stored at − 80 °C until further analysis.
Sample preparation
Samples were retrieved from the − 80 °C freezer and transferred to pre-chilled tubes. To each tube, 680µL of a MTBE/methanol/water (400:80:200) extraction solvent was added. Samples were vortex-mixed for 10 min to ensure homogeneity, followed by centrifugation at 3000×g for 15 min to facilitate phase separation. Post-centrifugation, the supernatant (organic phase) was carefully collected, while the lower aqueous phase was discarded. The organic phase lipid extracts were then dried using a low-temperature concentrator (Speed Vac SPD131P; Thermo Fisher Scientific) and stored for subsequent analysis. Prior to mass spectrometry, lyophilized samples were reconstituted in 400 µL of methanol/isopropanol (3:1, v/v) to ensure optimal solubility. Quality control (QC) samples were prepared by pooling aliquots of all study samples to monitor analytical reproducibility, which were analyzed alongside experimental samples using ultra-performance liquid chromatography coupled with quadrupole time-of-flight tandem mass spectrometry (UPLC-QTOF-MS)32.
LC-MS/MS analysis and identification
Chromatographic separation was performed on a Phenomenex Kinetex 1.7 μm EVO C18 column (2.1 × 50 mm, 100 Å; Agilent, USA) under reversed-phase liquid chromatography (RPLC) conditions. The mobile phase system consisted of: (A) 50% acetonitrile-water containing 10 mM ammonium formate, and (B) isopropanol (IPA) with 10% formic acid and 10 mM ammonium formate. A gradient elution program was applied: starting with 90% solvent A, linearly decreasing to 0% A over 11 min, holding at 100% B for 6 min, then immediately re-equilibrating to 90% A and maintaining this condition for 3 min. Mass spectrometry was conducted using a Triple TOF 5600 + orthogonal accelerated time-of-flight mass spectrometer (AB SCIEX, USA) equipped with an electrospray ionization (ESI) source. Data acquisition was performed in both positive and negative ion modes for each chromatographic run. Full-scan mass spectra were collected over an m/z range of 50–1500, with data-dependent acquisition (IDA) mode employed to enhance metabolite coverage. Mass accuracy was ensured by referencing a lock-mass ion throughout the analysis using MS-Dial software (ver. 3.70; 17 April 2019). Metabolite identification was achieved by matching detected ions against the MS-Dial Lipidomics MSP database (http://prime.psc.riken.jp/compms/msdial/main.html)33.
Statistical analysis
Raw mass spectrometry data were processed using MS-Dial (Ver. 3.70), with the Lipidomics MSP database integrated into the software to support peak detection, filtering, and alignment. Following preprocessing, a two-dimensional data matrix was generated, encompassing lipid identifiers, retention times, lipid classes, mass-to-charge ratios (m/z), peak areas, and raw Excel data. Multivariate statistical analysis, including partial least-squares discriminant analysis (PLS-DA), was performed using MetaboAnalyst 5.0 (http://www.metaboanalyst.ca/MetaboAnalyst/) to characterize lipid distributions and identify differential metabolites between senile rosacea patients and healthy controls. PLS-DA models were validated via 10-fold cross-validation with unit variance scaling. Model fitness was evaluated using R² (goodness of fit) and Q² (predictive ability). Between-group comparisons of integrated peak intensities were conducted using a two-tailed Welch’s t-test within MetaboAnalyst 5.0, with statistical significance defined as p < 0.05 and variable importance in projection (VIP) > 1.032.
Data availability
All data generated or analysed during this study are available from thecorrespongding author on reasonable request.
Change history
19 November 2025
The original online version of this Article was revised: In the original version of this Article affiliation 1 was listed as affiliation 2. Additionally, Xiaolei Ma was mentioned twice. The article has been corrected.
References
Maden, S. Rosacea: An overview of its etiological factors, pathogenesis, classification and therapy options. Dermato 3 (4), 241–262 (2023).
Kulkarni, N. N. et al. Innate immune dysfunction in rosacea promotes photosensitivity and vascular adhesion molecule expression. J. Invest. Dermatol. 140, 645–655 (2020).
Addor, F. A. Skin barrier in rosacea. An. Bras. Dermatol. 91 (1), 59–63 (2016).
Pappas, A. Epidermal surface lipids. Dermatoendocrinol 1 (2), 72–76 (2009).
Feng, F. et al. Effects of skin surface lipids on skin health. Asian J. Beauty Cosmetology. 17 (1), 149–155 (2019).
Mijaljica, D., Townley, J. P., Spada, F. & Harrison, I. P. The heterogeneity and complexity of skin surface lipids in human skin health and disease. Prog Lipid Res. 93, 101264 (2024).
Triebl, A., Hartler, J., Trötzmüller, M. & Köfeler, H. C. Lipidomics: prospects from a technological perspective. Biochim. Biophys. Acta Mol. Cell. Bio Lipids. 1862 (8), 740–746 (2017).
Qi, Z., Wang, F., Huang, Y. & Wang, P. Analysis of the correlation between skin barrier function and age in rosacea patients in Qinghai region. J. Cosmet. Dermatol. 23 (3), 999–1003 (2024).
Li, S., Ganguli-Indra, G. & Indra, A. K. Lipidomic analysis of epidermal lipids: a tool to predict progression of inflammatory skin disease in humans. Expert Re Proteom. 13 (5), 451–456 (2016).
Pye, R. J., Meyrick, G. & Burton, J. L. Skin surface lipid composition in rosacea. Br. J. Dermatol. 94 (2), 161–164 (1976).
Medgyesi, B., Dajnoki, Z. & Béke, G. Rosacea is characterized by a profoundly diminished skin barrier. J. Invest. Dermatol. 140 (10), 1938–1950e5 (2020).
Liu, Y. et al. Evaluation of skin barrier function based on skin dermoscopic features in patients with rosacea. Chin. Med. J. (Engl). 136 (15), 1885–1887 (2023).
Chen, C. et al. Exploring the pathogenesis and mechanism-targeted treatments of rosacea: previous Understanding and updates. Biomedicines 11 (8), 2153 (2023).
Camelo, A. et al. IL-33, IL-25, and TSLP induce a distinct phenotypic and activation profile in human type 2 innate lymphoid cells. Blood Adv. 1 (10), 577–589 (2017).
Klonowska, J., Glen, J., Nowicki, R. J. & Trzeciak, M. New cytokines in the pathogenesis of atopic dermatitis-new therapeutic targets. Int. J. Mol. Sci. 19, 3086 (2018).
Mittal, M., Siddiqui, M. R., Tran, K., Reddy, S.P. & Malik, A. B. Reactive oxygen species in inflammation and tissue injury. Antioxid. Redox Signal. 20 (7), 1126–1167 (2014).
Schmidt, M., Binder, H. & Schneider, M. R. The metabolic underpinnings of sebaceous lipogenesis. Commun. Biol. 8 (1), 670. https://doi.org/10.1038/s42003-025-08105-9 (2025). PMID: 40289206; PMCID: PMC12034822.
Pyle, H. J. et al. Saprophytic bacteria and fungi colonize Stearoyl coenzyme-A desaturase-1 knockout skin. Exp. Dermatol. 32 (1), 78–84 (2023).
Cheng, Y. et al. Quantitative lipidomics profiling of skin surface lipids and skin barrier function evaluation in patients with acne vulgaris. Arch. Dermatol. Res. 317 (1), 349 (2025).
Shin, S. H. et al. The therapeutic effect of 1-palmitoyl-2-linoleoyl-3-acetyl-rac-glycerol on chemically induced atopic dermatitis. Sci. Rep. 14 (1), 23402 (2024).
Nădăban, A. et al. The sphingosine and phytosphingosine ceramide ratio in lipid models forming the short periodicity phase: an experimental and molecular simulation study. Langmuir 40 (27), 13794–13809 (2024).
Ní Raghallaigh, S., Bender, K., Lacey, N., Brennan, L. & Powell, F. C. The fatty acid profile of the skin surface lipid layer in papulopustular rosacea. Br. J. Dermatol. 166 (2), 279–287 (2012).
Jia, T., Qiao, W., Yao, Q., Wu, W. & Kaku, K. Treatment with docosahexaenoic acid improves epidermal keratinocyte differentiation and ameliorates inflammation in human keratinocytes and reconstructed human epidermis models. Molecules 24 (17), 3156 (2019).
Ryborg, A. K., Grøn, B. & Kragballe, K. Increased lysophosphatidylcholine content in lesional psoriatic skin. Br. J. Dermatol. 133 (3), 398–402 (1995).
Ryborg, A. K., Deleuran, B., Thestrup-Pedersen, K. & Kragballe, K. Lysophosphatidylcholine: a chemoattractant to human T lymphocytes. Arch. Dermatol. Res. 286, 462–465 (1994).
Song, M. H., Gupta, A., Kim, H. O. & Oh, K. Lysophosphatidylcholine aggravates contact hypersensitivity by promoting neutrophil infiltration and IL17 expression. BMB Rep. 54 (4), 203–208 (2021).
Lysophosphatidylcholine facilitates the pathogenesis of psoriasis through activating keratinocytes and T cells differentiation via glycolysis.
Bouwstra, J. A. & Ponec, M. The skin barrier in healthy and diseased state. Biochim. Biophys. Acta Biomembr. 1758 (12), 2080–2095 (2006).
Xiao, T., Sun, M., Zhao, C. & Kang, J. TRPV1: A promising therapeutic target for skin aging and inflammatory skin diseases. Front. Pharmacol. 14, 1037925 (2023).
Gouin, O. et al. TRPV1 and TRPA1 in cutaneous neurogenic and chronic inflammation: pro-inflammatory response induced by their activation and their sensitization. Protein Cell. 8 (9), 644–661 (2017).
Choi, J. E. & Di Nardo, A. Skin neurogenic inflammation. Semin Immunopathol. 40 (3), 249–259 (2018).
Ma, X. et al. Lipidomics profiling of skin surface lipids in senile pruritus. Lipids Health Dis. 19 (1), 171 (2020).
Tsugawa, H. et al. MS-DIAL: data independent MS/MS Deconvolution for comprehensive metabolome analysis. Nat. Methods. 12 (6), 523–526 (2015).
Funding
The work was supported by research grant from National Natural Science Foundation of China, Grant/Award Number: 82404136 to Xiaolei Ma.
Author information
Authors and Affiliations
Contributions
Xiaolei Ma: Data collection and analysis.Yi Yang: Writing the original draft, reviewing, and editing. Lulu Lu, Na Gao: Liquid chromatography coupled with tandem mass spectrometry (LC-MS/MS) and multivariate data analysis. Zheng Zhao, Xiulian Zhang: Transepidermal water loss detection. Gangwen Han: Clinical cases collection and experimental guidance. The authors read and approved the final manuscript. All the participants consent for publication of their individual details.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval and consent to participate
The study was approved by the Ethics Committee of Peking University International Hospital. All procedures adhered to relevant guidelines and regulations, and informed consent was obtained from all participants prior to enrollment. Informed consent to participate in the study was obtained from each patient and healthy person before enrolling in the study. Clearance No for Ethical approval: 2018-065(BMR).
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Yang, Y., Zhao, Z., Lu, L. et al. Lipidomic profiling of skin surface lipids in a cohort of Chinese patients with rosacea. Sci Rep 15, 39163 (2025). https://doi.org/10.1038/s41598-025-24539-x
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-24539-x