Abstract
The present study was conducted to increase the production of Artemia sp. using different supplemental feeds for thirty days. Five treatments were T1 (green water + rice bran), T2 (green water + soybean meal), T3 (green water + oil cake), T4 (green water + formulated feed), and T5 (control, only green water). In formulated feed, there were 40% rice bran, shrimp meal 35%, corn 10%, and oil cake 5%. Artemia spp. was cultured under all conditions. Fifteen experimental aquariums (250 L) were set up with the necessary equipment in the laboratory for measuring the water parameters, biomass, leaching nutrients from supplementary feed, and cysts of Artemia at different life stages. Mechanical aeration and incentives were provided every day in the aquarium for maintaining the optimum level of dissolved oxygen at the initial stage of the experiment. Collection of biomass of cultured Artemia sp. started on day 5 and continued until day 30, and the cyst was collected from day 20 to 30. At baseline, the average wet weight of Artemia spp. was 0.641 g throughout the study aquarium. An average of 3.47 g of wet Artemia was collected from different feedings, with 0.67 g measured in the control condition. Initially, cyst production averaged 0.145 g in the different feed treatments and 0.066 g in the control condition. Finally, cyst production averaged 0.180 g in the treatment with different feeds and 0.086 g without supplementation. Analysis of variance using a one-way test (ANOVA) for population density and biomass of cultured species showed significant differences between the different treatments.
Similar content being viewed by others
Introduction
Artemia spp. is a micro-crustacean popularly known as brine shrimp and an extremely euryhaline species, with standing salinities from 3 to 300 ppt. They can even survive short periods in fresh water, but are unable to reproduce in fresh water. This zooplankton naturally remains in salt lakes where chlorine rich waters are available1. More than 50 different strains of Artemia exist in continents with differences in specific characteristics such as hatching rate, cyst quality, feeding behavior, nauplius size, viability, optimal temperature and salinity range requirements. Some strains are parthenogenetic (only females) but most are zygogenetic (males and females). Artemia has unique two modes of reproduction: ovoviviparous (free-swimming nauplius larvae released from fertilized eggs when conditions are favorable) and oviparous (producing dormant cysts when conditions are unfavorable)2. The most peculiar nature of Artemia is that the dehydrated cyst can be stored in dry and anaerobic conditions, transported, and hatched on demand. Habitually, Artemia is a non-selective filter feeder with high nutritional content, thrives in high-density environments with a variety of water quality variables, including salinity and temperature3.
Bangladesh possesses a vast network of inland water bodies and ranks third globally in capture fisheries and fifth in inland aquaculture. In Bangladesh, aquaculture plays a critical role in global food security and the economy by meeting seafood demand, economic impact, and species conservation. In aquaculture, fish, crustaceans, mollusks, and aquatic plants play a critical role in global food security and the economy. Fish remains the primary source of animal protein for the majority of the population4,5. In this context, Artemia sp. (brine shrimp) plays a crucial role in aquaculture as a live feed, particularly during the larval stages of fish and shrimp. Artemia is an euryhaline, non-selective filter feeder with high nutritional value. It thrives in high-density environments and can tolerate a wide range of salinity levels up to 300 ppt. While Artemia can survive in freshwater, it requires saline conditions to reproduce, typically being oviparous and producing cysts at salinities above 150 ppt6. The unique resilience of dehydrated Artemia cysts, which can remain viable under dry, anaerobic conditions for extended periods, makes them invaluable for hatchery operations. Although Bangladesh does not have natural populations of Artemia, the coastal salt pans provide suitable environments for artificial culture. Local production could reduce dependence on costly imports, enhance the income of salt farmers, and support the sustainable development of the aquaculture industry. Globally, over 85% of aquaculture species utilize Artemia as live feed, highlighting its critical role in larval nutrition and survival. Decapsulated cysts are widely used to rear species such as Penaeus spp., while newly hatched nauplii and adult Artemia serve as highly nutritious feed with protein levels ranging from 40% in nauplii to 60% in adults7 (Mitra et al. 2007). Artemia culture has the potential to generate employment opportunities, provide export revenue, and meet the local demand for quality hatchery feed. It is a key input in nursery systems, where it helps maintain water quality by minimizing feed waste.
Globally, fish farming is expanding. Not all supplemental feeds must provide a balanced diet. Aquaculture production requires supplemental feed since pond food ingredients cannot support quick development. Although some finfish and crustacean species seem to have aquaculture potential, live food availability is one of the major limitations during larval development. Among various live feeds used in fish culture, Artemia cysts are the most widely used feed items8. For the high-density culture of Artemia, non-soluble waste products from crops or the food processing industry (maize, rice bran, soybean pellets, and oil cake) as an excellent feed source. Artemia spp. was fed to aquatic species’ larvae in the 1930s. For the expanding aquaculture sector, Artemia spp. has the potential to be a great food source that might supply high-quality fish and crustacean feed in enough quantities and at the right periods. During the culture period, nutritionally rich Artemia spp. contain protein and lipid levels in the range of 54.9–57.8% and 10.7–11.6%, respectively, at week 3 and 49.4–50.6% and 9.4–10.2% at week 129. About 90% of the commercial Artemia spp. cyst production worldwide comes from a single site, the Great Salt Lake in Utah, USA10.
Over 85% of the world’s cultivated species eat Artemia spp. as live food, making it important in aquaculture. Decapsulated cysts have also been utilized to raise Penaeus marine shrimp. Shrimp and other aquarium and snail water organisms need Artemia spp. Aquatic animals can eat Artemia spp. larvae in nursery settings without polluting the tank. Artemia spp biomass is exported for foreign exchange. Profitable for impoverished salt growers nationwide. It creates public and technocrat jobs11. Efforts are being made to artificially cultivate Artemia by ensuring a suitable environment. Even different types of food are being used9,12. Supplemental feeds boost bacterial growth despite having fewer nutrients than a natural diet. Additionally, Artemia is omnivorous. Along with brood stock nutrition education, supplemental feeding programmes are often provided11. There is no artificial feed formulation to improve Artemia cyst production; however, several supplemental feeds are being used to improve biomass and cyst production. Commercial formulated feeds such as yeast-based powders, microencapsulated diets; agricultural by-products such as rice bran, wheat flour, or soybean meal; enriched live feeds such as microalgae enriched with DHA/EPA, or co-cultures with bacteria, and organic matter inputs such as fermentation-based feeds often used in low-cost systems.
In Bangladesh, laboratory-scale research on Artemia was initially started by Mahmood and Begum13. The production of Artemia cysts in Bangladesh’s coastal saltpans was successful between January 1988 and December 1989. Teachers and graduates from the Institute of Marine Sciences and Fisheries conducted three more successful trials in 1992 and 199414,15,16,17. Even though they have created a method for producing biomass and Artemia cysts, they did not carry out any further research on this kind of thing. Islam et al.12 conducted an indoor culture tank experiment to produce biomass from Artemia. The initial observation yielded a survival percentage of 46.12%. To enhance the survival percentage or increase the production rate of Artemia cysts, more experiments are needed. As Artemia is a significant food source for fish and prawn fry, it must be cultured and its cyst production enhanced by extra feed12,18. The coastal salt plans of Bangladesh are ideal for Artemia spp. artificial culture further highlights the importance of Artemia cysts for sustainable aquaculture12,14,18.
Despite its value, Bangladesh still lacks a standardized artificial feed formulation that can reliably enhance Artemia cyst production. While formulated feeds are essential—especially when pond productivity is insufficient—they remain expensive due to weak regulation and fluctuating import costs. Supplementary feeds are being tested to improve biomass yield and cyst output, especially given Artemia’s omnivorous nature, which allows it to utilize diverse feed types. Algae, which are rich in proteins, carbohydrates, lipids, and vitamins, are frequently used in Artemia diets, either as live microalgae or in encapsulated forms19. Challenges such as natural diet dependency, cyst induction triggers (high salinity, low oxygen, food limitation), unknown micronutrient requirements, more focus on biomass than cysts, limited controlled trials, wild harvest dominance, high production cost, and market acceptance create less awareness about the Artemia cyst production in Bangladesh. In addition to these challenges, knowledge gaps still exist in exploring scientific information on the successful production of Artemia cysts in Bangladesh. To the best of the authors’ knowledge, this is the preliminary study conducted in Bangladesh to produce Artemia cysts using supplementary feeds. This study aims to evaluate the effect of various additional feeds on biomass yield and cyst production of Artemia sp. under controlled culture conditions. It will also reduce Artemia cyst import costs by increasing production in natural beds and enhancing salt manufacturing income.
Materials and methods
Sample collection and brine preparation
Saline water was collected from the Bay of Bengal of Chittagong coastal side, Bangladesh by using plastic container and stored in an experimental tank. Crude salt was purchased from the local wholesale market in Chittagong City, Bangladesh, and stored in plastic containers. Saline water of different gradation was prepared using more reservoir tanks, and allowed to remain static condition for settling the suspended solids and other debris at the bottom of the plastic container. In the present, Artemia cysts used experiment were Red Jungle Brand (USA) that was collected from a commercial hatchery, Coax’s Bazar.
Experimental design and treatments
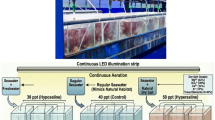
A simplified design of experiment with some modifications was adopted in this study, following Islam et al.8, which involved minimal sophistication. Fifteen experimental aquariums (250 L) were set up in the open yard of the Faculty of Marine Sciences and Fisheries, University of Chittagong, Bangladesh. There was an open space around the experimental aquarium for proper ventilation and light penetration. Mechanical aeration was provided every day in the aquarium to maintain the optimum level of dissolved oxygen during the experiment. A reservoir tank (500 L) was set up and filled with saline water for the exchange of water and reduction of the organic load of the prepared saline water throughout the research. Bottom of reservoir tank (up to 6 inches) was filled up with pebbles stone which act as a filter. An outlet pipe was set at the bottom of the reservoir tank so that filtered water at a rate of 10 L/day. Feeding dose: 1 g/10 L every three days. Thus, saline water was recycled every two or three days, and it was continued for the whole period of the experiment to create a continuous water flow in the tank. Afterwards, 0.5 g of Artemia cyst was provided and cultured in the tank up to 30 days to get biomass and cyst.
Measurement of Artemia biomass and size
Total biomass and weight of cyst were measured by using a microbalance (Electrical Analytical Balance, OSK 11325A). All treatments were conducted in triplicate.
Feed formulation and feeding regime
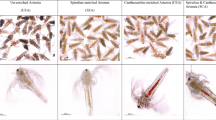
In this research, various combinations of supplementations were tested for their performance on the production of Artemia biomass and the quality of cyst. All experimental aquariums were supplied with “green water” as a natural food source (Scytonematopsis crustacea) once every two days throughout the culture period 104–105 cells/mL every 2 days. The different treatments were maintained as follows: Five treatments were T1 (green water + rice bran), T2 (green water + soybean meal), T3 (green water + oil cake), T4 (green water + formulated feed), and T5 (control, only green water). Only green water treatment was considered as natural treatment. Each treatment had three replicates. In formulated feed there were 40% rice bran, shrimp meal 35%, corn 10% and oil cake 5%. All the single ingredients and even the formulated feed were in powder form. Upon hatching, Nauplii became a pre-adult after 5–7 days. At this stage, feed was being given at a rate of 0.1 g/L, given daily in the form of different single components and formulated feed. The feeding schedule was maintained based on the clarity of the water. If food is applied twice a day, food is continuously broadcast in the culture tanks until the water turns murky.
Proximate composition analysis
The proximate analysis method of different feed ingredients was done (Table 1). Moisture content, crude protein20,21, crude fat22,23and carbohydrate were recorded.
-
(1)
Moisture content (%) = ((Weight of fresh sample − Weight of dry sample) / Weight of fresh sample) × 100.
-
(2)
Crude Protein (%) = Nitrogen × Conversion factor (Conversion factor: 6.25 for animal origin; 5.90 for plant origin).
-
(3)
Nitrogen content (%) = ((Value of HCl × 0.1 × 0.014) / Weight of sample (g)) × 100.
-
(4)
Crude Fat (%) = (Corrected weight of fat / Weight of sample) × 100.
-
(5)
Carbohydrate (%) = 100 − (Moisture + Ash + Protein + Fat).
Preparation of an aquarium
To assess the nutrient depletion from feed ingredients utilized as supplementary feed in Artemia cyst production. The primary productivity of phytoplankton and bacterial biomass contributes to the provision of nutritious sustenance. Nitrite-nitrogen (NO2-N), phosphate-phosphorus (PO4-P), silicate, and ammonium nitrogen are the principal constituents in feed. Following hatching, Artemia nauplii were isolated and introduced into the aquarium via inoculation. Initially, the aquarium was maintained at a salinity of 10 ppt. It increased progressively up to 170 ppt by the conclusion of the experiment. To promote the growth of algae and microbes, the water was enriched with organic matter through the decomposition of supplementary feed (rice bran, soybean, oil cake, maize, and formulated feed) under moderate aeration for three days. Nauplii would reach pre-adulthood five to seven days after hatching.
Measuring nutrient leaching in water
Nutrients leaching from feed ingredients in the culture of Artemia cyst production were evaluated to maintain the quality of the cyst. The primary productivity of phytoplankton and the biomass of bacteria are largely proportional to good nutrients in food. Major micro-nutrients of feed, such as nitrite-nitrogen (NO2-N), phosphate-phosphorus (PO4-P), Silicate, and ammonium nitrogen, were tested. Measurement of Nutrient Leaching in Water. Nutrient leaching from feed ingredients in the Artemia cyst production system was evaluated to monitor water quality and its influence on cyst yield. Water samples were collected at 0 h (immediately after feed addition) and, 24 h intervals to assess the dynamics of nutrient release. The primary productivity of phytoplankton and the biomass of bacteria were considered indirect indicators of nutrient availability, as both are largely proportional to the concentration of essential nutrients in the culture medium. Major dissolved nutrients, including nitrite–nitrogen (NO₂–N), phosphate–phosphorus (PO₄–P), silicate (SiO₂–Si), and ammonium–nitrogen (NH₄–N), were measured following APHA standard methods24. Nutrient concentrations were determined spectrophotometrically using a UV–Vis spectrophotometer (Model: Shimadzu UV-1800 (Shimadzu Corporation, Japan), NO₂–N: diazotization method (540 nm), PO₄–P: ascorbic acid method (880 nm), SiO₂–Si: molybdate blue method (815 nm), NH₄–N: indophenol blue method (640 nm)24,25,26. All analyses were performed in triplicate, and results were expressed in µg/L.
Determination of water parameters
The temperature of water was measured using a centigrade laboratory thermometer having a range from 0 to 110 °C. Dissolved oxygen (DO) was determined following the azide modification of the Winkler method, which is the standard procedure used for dissolved oxygen (DO) measurement27. A digital pH meter (HANNA instruments, Made in Romania, H198107) was used to determine the hydrogen ion concentration of water. Water salinity was determined by a Refractometer (TANAKA, New S-100, Salinity, Japan).
Measuring technique of Artemia biomass and size
Artemia was distributed uniformly throughout the aquarium’s water, and dynamic aeration was performed before sampling. A 100 mL water sample was collected from each replicate aquarium using a random sampling technique to estimate the density and biomass of Artemia spp. A small mesh net (e.g., 250 μm) was used to collect Artemia from the culture. Excess water was gently removed by blotting with absorbent paper. Artemia was weighed by using an electronic analytical microbalance (OSK 11325A). The length of individual Artemia was measured by using a digital microscope and a length scale. The biomass was estimated based on the length–weight relationships for Artemia, followed by Sulistiyarto & Bakrie [18].
Collection and measuring technique of Artemia cyst
Just after the mating period, Artemia gave birth to nauplii and cysts as well. As the salinity was increased gradually in the culture tank, Artemia started to release cysts instead of nauplii at their age of 19–21. As a result, a thin layer of cyst was observed in the surface water of the aquarium, which finally accumulated at the periphery of the aquarium due to wind action. Then the Artemia cyst was collected from the aquarium by adopting the siphoning technique and using various mesh size nets. Then, the cyst is allowed to dilute with freshwater for the separation of waste material from the cyst. The cyst was further allowed to dilute with high saline water (200–250 ppt). At this stage, the heavier substances like sand/mud settled into the bottom, and the majority of the cyst floated over the surface of the water, from where the cyst was collected in a fresh condition. At the final stage, freshwater was added with cyst again, and finally allowed to dry after washing it with 20 ppt saline water. Dried cyst was stored at room temperature to test their hatching performance after a certain period. The hatching rate of dried cysts stored at room temperature was 87.38 ± 0.91%.
Statistical analysis
The data were statistically analyzed using SPSS 20.0 (IBM Corporation, Armonk, NY, USA). Descriptive statistics, including mean and standard deviation (SD), were calculated using MS Excel 2016. A one-way ANOVA was performed to determine the overall significant differences in population parameters, such as cyst production, density, and biomass of Artemia. To examine pairwise differences among the groups, a multivariate post hoc Tukey test was applied. The level of significance was set at 5% (p < 0.05) throughout the analysis, consistent with the results presented. Python with the Matplotlib library was employed for figure panel generation.
Results and discussion
Nutrients leach into the water
In the present study, oil cake, rice bran, soybean meal, maize, and shrimp meal were used as supplementary feed due to low-priced feed sources for the culture of Artemia spp. up to the grown-up phase for the enhancement of Artemia cyst and biomass production. During the study period, supplementary feeds have been used in T1, T2, T3, and T4, respectively, where T5 used only green water. The major leaching nutrients of feed ingredients used as supplementary feed in the culture of Artemia cyst production were observed (Table 2). The leaching of nitrite nitrogen and phosphate phosphorus increased throughout the period for most of the treatments, whereas ammonium nitrogen and silicate silicon increased from day 5 to day 21 and then decreased (Table 2). The data suggested that nutrients leaching from enrichment diets into the water can impact Artemia and the organisms that consume them. Considering the factors like the age and size of the Artemia, the products’ primary benefits are their affordable price, widespread worldwide availability, quality of the feed, and environmental parameters such as temperature and salinity28. Therefore, we provided 0.1 g/L/day of food in the culture aquarium. By influencing Artemia feeds affect the culture conditions that influence the filtration rate, ingestion rate, and assimilation, several factors, such as the larvae’s developmental stage based on the quality of the food provided29.
The protein levels of soybean (SB) and shrimp meal (SM) were high. However, shrimp meal also contained a higher ash level, while soybean had a higher carbohydrate content compared to other products. A formulated feed (FF) was developed to supplement green water, and its composition was comparatively more effective. The average algal density in the treatment aquarium was good. The low density of algae was observed at the initiation of the culture, but an adequate density was achieved after week 2, which enhanced the density of the culture species.
Water quality parameters
Different water quality parameters, i.e., salinity, pH, water temperature, and DO, were measured at a definite time daily.
Three critical factors that significantly impact Artemia populations are salinity, temperature, and feeding. Therefore, the vital indicators of water quality (pH, salinity, temperature, and dissolved oxygen) were maintained during the study period as soon as possible based on the local environment (Fig. 1 and Table S1). The mean ranges of water temperature were 23.0–30.5, 22.0–30.1, 23.6–29.2, 25.0–27.5, and 23.9–28.2 °C, salinity were 10.0–181, 9.80–186, 11.1–185, 12.4–185, and 13.2–183 ppt, DO were 3.50–6.50, 3.20–6.40, 3.20–6.40, 3.00–6.70, and 3.40–6.70, and pH were 7.2–8.0, 6.8–7.9, 7.0–7.7, 7.4–7.8, and 7.5–7.8, respectively for T1, T2, T3, T4, and T5 (Fig. 1 and Table S1). One important factor influencing the generation of Artemia biomass is salinity. In unfavorable environmental circumstances, Artemia often reproduces oviporously; conversely, ovoviporously appears to be favored under satisfactory environments. Suitable for unfavorable environmental circumstances are high salinity, low oxygen levels, temperature, and lack of proper nutrition30. These factors were reported to be, to some extent, effective in the production of dormant cysts of Artemia spp.31.
Temporal variation of water quality parameters in different treatment tanks (T1–T5) during the study period. The panels represent (A) water temperature (°C), (B) salinity (ppt), (C) dissolved oxygen (mg/L), and (D) pH. Each line corresponds to a treatment tank (T1–T5), with distinct colors indicating treatments as shown in the legend.
According to the principle, the present research was undertaken to produce Artemia biomass and cyst in saline water artificially prepared using crude salt, and salinity increased up to 180 ppt Coutteau et al.32 suggested that when producing various Artemia under controlled conditions, water quality parameters should be kept within an ideal range (oxygen above 2 mg/L, salinity between 32 and 65 ppt, pH between 6.5 and 8.0, and temperature between 19 and 25 °C32. On day 19, the Artemia reached adulthood at 20 ppt, and on day 18, it reached 45 ppt 33. Salinity is an important parameter for controlling the growth, survival, and hatching of Artemia spp. Although the Artemia spp. is euryhaline, it is comfortable when exposed to optimum salinity. Although it is rare in waters below 45 ppt, Artemia persists in natural habitats with salinities ranging from 10 to 300 ppt34. Seawater proved to be better for Artemia spp. growth and survival by Soundarapandian and Saravanakumar35, while three salinities were used to cultivate them (freshwater, brackish water, and seawater) for the biomass production of Artemia spp. According to Naegel & Rodriguez36 food becomes a limiting factor at high salinity levels (200–250 ppt), which may be the primary cause of the adults’ Artemia size decrease. Therefore, the salinity range in the present study was maintained at (10–180 ppt) at different stages (Table 3). The salinity ranges were increased artificially with the increase in the age of Artemia spp. Here, various supplementary feeds were applied to keep salinity constant. When cultured in salt ponds, Artemia reached its adult stage in 16–19 days37. When cultured in different salinities, such as freshwater (< 0.5 ppt.), brackish water (0.5–28 ppt.), seawater (28–35 ppt.), or even in brine conditions, Artemia sp. reached its adult stage in 14–20 days35. Artemia attained an adult stage in freshwater on the 20th day, brackish water on the 17th day, and seawater on the 14th. The culture was carried out indoors, and the water was changed every two days. In the present observation, maximum Artemia attained an adult stage in seawater during the 17th day, while the salinity values were 147, 142, 145, 152, and 155 ppt, respectively, in T1, T2, T3, T4, and T5 (Fig. 1 and Table S1). The rate of water exchange matched that of the previously mentioned study. About 60 ppt was found to be the optimal value for the sanfrancisco strain of Artemia sp.’s growth and survival38. In a lab setting, thirteen geological strains of Artemia had high survival over a wide range of salinities (35–100 ppt)39. According to Triantaphyllidis et al.40 after 27 days in culture, a high parthenogenetic population fared well at salinities of 0 ppt to 100 ppt, but less than 50% of the population survived at 35, 40, and 80 ppt. Between 40 and 240 ppt of salinity, Artemia abundances have increased; at salinities greater than 240 ppt, they have decreased. Gilchrist41 demonstrated that Artemia could alter its appearance in response to salinity.
One of the key environmental factors that determines Artemia survival and ability to grow in a given culture is pH. It also has an impact on the metabolism and other physiological functions of Artemia. The pH values of the rearing medium for freshwater, brackish water, and seawater were 7.8–8.5, 8.2–8.9, and 8.5–9.3, respectively35. Neutral to alkaline waters, temperatures below 34 °C, and relatively low O2 levels are the habitats of Artemia42. Naupliar growth slows down and, in adults, the overall appearance deteriorates as pH falls below 7.0. Correa et al.43 suggested that the presence of Artemia in naturally occurring saline ponds can be impacted by changes in water temperature. According to earlier research, the brine lives in bodies of water where the temperature varies between 6 and 35°c44. The average pH value for Artemia is 7.2 to 7.9. Only very low concentrations of less than 2 mg O2/L are recommended concerning dissolved oxygen. Oxygen levels higher than 5 mg/L would result in the individual’s dry weight32. The dissolved oxygen level in this study was considered strictly due to indoor culture and because of mechanical aeration; no significant differences were seen in any of the replications. In the present study, the recorded average dissolved oxygen (mg/L) levels for Artemia were 6–6.5 mg/L. As water temperature directly impacts metabolism, salinity, growth, oxygen consumption, molting, and survival, it is likely the most significant environmental factor in Artemia cultures. Generally speaking, the immune system of crustaceans is impacted by abrupt temperature changes. When the temperature ranged from 25 to 35 °C and the salinity ranged from 42 to 200 ppt, culture studies of Artemia populations showed high values. Reduced temperature would mean slower growth, and more than 30 °C would mean death. The temperature was maintained at 29–32 °C while Soundarapandian and Saravanakumar35 studied the effect of salinity on Artemia biomass production. Hence, the present study was an indoor system, so water temperature was optimum for Artemia growth28.
Growth performance
Biomass and cyst collecting
The collection of Artemia biomass and cyst was centered on the study of cyst and biomass harvest strategies of Islam et al.8 (Table 3).
Growth observation was done by collecting biomass from day 5 and cysts from mature Artemia on day 21. In this study, harvesting occurred every three days. A triangle-shaped scope net (0.12 µm mesh size) and a 10 g precious balance (0.001 g accuracy) were employed to measure submerged population weight. Artemia biomass, density, and individual weight increased on days 5, 13, 20, and 27. Ten Artemia spp. samples were taken from each aquarium, excess water was removed using tissue paper, the samples were weighed, and the average value was computed to estimate the mean individual weight. The maturity rate was assessed using Nguyen et al.45. Day 14 samples were observed. Each sample female was divided into immature and mature based on ovary development.
Enhancement factors
Due to the indoor culture system, the aquarium’s biotic and abiotic elements barely changed. Mean salinity was 119 ± 59.41 ppt, and aquatic depth increased from 0.30 to 0.40 m from the stage. Average water temperature was 25.89 ± 2.27 °C, pH 7.47–7.72, DO 4.91 ± 1.04–5.1 ± 0.99 mg/L throughout the culture period as treatments. After eight months, three replication investigations were directed to quantify cyst quality by determining hatching percentage, population density, and offspring biomass production (F1 cohort). As in the last experiment, each replicate steadily increased salinity from 9.80 to 186 ppt.
Growth rate and survival
Density on days 5, 13, 21, and 29 was estimated to be around 1383–1418, 736–986, 413–821, and 305–622 ind/L, respectively (Table 3). There were no significant differences among treatments (p > 0.05) for density, individual weight, and maturity, whereas the biomass and collection of cysts showed statistically significant differences (p < 0.05) (Table 3, Table S2). There were no significant differences (P > 0.05) among treatment means at day five, with weights ranging from 0.44 to 0.48 mg. The individual weight showed the same on days 13, 20, and 27 (Table 3). Moreover, there was an inclination that Artemia co-feed in T4 or T1 performed better than T2, T3, or T5. However, T4 showed a slight growth difference associated with T3. Elevated levels of nutrients (nitrite nitrogen, ammonium nitrogen, phosphate phosphorus, and silicate silicon) in T4 treatment might be due to the reason for the best performance of Artemia cyst production. The effect was also seen in the maturation rate, as the biomass and density, individual weight, compared to the other four groups that received supplementation feeds (80.20–91.07%), and the control group (75.04%) had a significantly lower percentage.
Total biomass and cyst
The supplementation diets increased biomass production compared to those without Supplementation (only T5 used only green water as the control). Note that co-feeding with formulated diet (T1, T2, T3, and T4) contributed the preeminent performance among the four assessment foods. However, it was not appreciably greater than when the components of the prepared feed were used separately. The cyst collection was started on day 21 and showed the same biomass effect. While the study successfully demonstrates the enhancement of Artemia biomass and cyst production under controlled conditions, it is crucial to consider the practical implications for large-scale aquaculture. Scaling these findings could significantly benefit commercial Artemia hatcheries by improving the cost-efficiency and productivity of cyst yields, which are vital for feeding larval stages of many cultured fish and shrimp species.
For instance, if the optimized conditions (e.g., salinity, feeding regimes, or use of specific microalgae or additives) identified in this study can be reliably reproduced at scale, this could reduce dependency on wild-caught cysts, stabilize supply chains, and potentially lower production costs. However, further trials under semi-intensive and intensive farming systems are recommended to validate the economic and operational feasibility of applying these enhancements in real-world settings. Additionally, these findings could support the development of standardized Artemia production protocols, helping hatcheries optimize both biomass and cyst outputs depending on market demands.
Conclusions
The present study on the Artemia population at the crude salt work has shown that their commercial characteristics are good. This study demonstrated that appropriate biological and nutritional strategies (crude salt pan) enhance the biomass and Artemia cyst yield. The highest biomass yield reached 4.91 g/L for T4 treatment at 29 days, and cyst production peaked at 0.20 cysts/250 L for T4 treatment at 27 days, under optimal salinity conditions. Supplementary feeding with rice bran, soybean meal, oil cake, and maize improved feeding efficiency by 114.6%, contributing to a 2.99 g/L increase in biomass and 0.893 g/250 L improvement in cyst output compared to control diets. These findings underscore the value of using low-cost, locally available feed ingredients to enhance Artemia production on the Bangladeshi coast. As a limitation, this study did not perform cyst hatching (%) and Artemia biomass proximate analyses. For further development, research should focus on the role of beneficial algae and bacterial strains as supplementary feed, as well as optimizing environmental parameters in semi-intensive and large-scale culture systems. These efforts will contribute to sustainable Artemia farming and support hatchery-based aquaculture industries.
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Sorgeloos, P. & Kulasekarapandian, S. Production and use of Artemia in aquaculture. CMFRI Spec. Publ. 15, 1–73. http://eprints.cmfri.org.in/id/eprint/3135 (1984).
Vos, J. & de la Rosa, N. L. Manual on Artemia Production in the Philippines. FAO/UNDP/BFAR Project Manual PHI/75/005/WP, 48 pp. (1980).
Alal, G. W., Olendi, R. & Barasa, J. E. Effect of different salinities on the hatchability and survival of brine shrimp, Artemia salina (Linnaeus) from Malindi. Kenya. African J. Educ. Sci. Technol. 3, 1–9 (2014).
Roy, T. K. et al. A comprehensive assessment of health risks associated with heavy metal through ingestion of two predominant fish species in a developing country. Biol. Trace Element Res. https://doi.org/10.1007/s12011-025-04540-1 (2025).
Rani, S. et al. Occurrence of trace elements in water, fish and shellfish from estuarine and coastal areas of the east coast of Bangladesh, northeast Bay of Bengal: Human health risk implications. Mar. Pollut. Bull. https://doi.org/10.1016/j.marpolbul.2025.117675 (2025).
Nipa, S. A., Hasan, R., Naser, N. & Rashid, H. O. Biometrical Study Of Brine Shrimp (Artemia franciscana) With Special Emphasis On Hatching Efficiency And Hatching Percentage In Tropical Condition At Bangladesh. European Sci. J. 12(18), 123. https://doi.org/10.19044/esj.2016.v12n18p123 (2016).
Mitra, G., Mukhopadhyay, P. K. & Ayyappan, S. Biochemical composition of zooplankton community grown in freshwater earthen ponds: Nutritional implication in nursery rearing of fish larvae and early juveniles. Aquaculture 272, 346–360. https://doi.org/10.1016/j.aquaculture.2007.08.026 (2007).
Islam, M. S., Hossain, M., Molla, R. & Bhuiyan, M. Production of brine shrimp, Artemia salina biomass and cyst in indoor tank using crude salt. J. Chem. 5, 1574–1584 (2015).
Anh, N. T. N., Van Hoa, N., Van Stappen, G. & Sorgeloos, P. Effect of different supplemental feeds on proximate composition and Artemia biomass production in salt ponds. Aquaculture 286, 217–225 (2009).
Van Stappen, G. Artemia. In Lavens, P. & Sorgeloos, P. (eds) Manual on the Production and Use of Live Food for Aquaculture, FAO Fisheries Technical Paper No. 361, 79–106 (FAO, Rome, 1996).
Sorgeloos, P., Dhert, P. & Candreva, P. Use of the brine shrimp, Artemia spp., in marine fish larviculture. Aquaculture 200, 147–159. https://doi.org/10.1016/S0044-8486(01)00698-6 (2001).
Islam, M. S., Kibria, M. M. & Bhuyan, S. Production of Artemia biomass in indoor culture tank in Bangladesh. J. Sci. Res. 11, 101–110. https://doi.org/10.3329/jsr.v11i1.36467 (2019).
Mahmood, N. U. R. U. D. D. I. N. & Begum, B. O. R. H. A. N. A. On hatching of Artemia nauplii under different conditions of temperature and salinity (1978).
Mahmood, N. Final Report, BARC Contact Research Scheme (1990).
Mahmood, N., Haider, S. M. B. & Saikat, S. Q. Final Report, BARC Contract Research Scheme, Bangladesh, 32 pp. (1992).
Mahmood, N., Belal Haider, S. M. & Quaderi Saikat, S. Successful inoculation of Artemia and production of cysts in the coastal saltpans of Bangladesh II (1993).
Mahmood, N., Islam, M. A. & Saikat, S. Q. In Proc. 9th National Zoological Conference, Zoological Society of Bangladesh (1994).
Sulistiyarto, B. & Bakrie, R. Survival, growth, and biomass of brine shrimp (Artemia franciscana) fed with spirulina powder and soybean flour. Int. J. Fish. Aquat. Stud. 12, 13–18 (2024). https://doi.org/10.22271/fish.2024.v12.i1a.2885
Radhakrishnan, S. et al. Outcomes of Surgical Bleb Revision for Complications of Trabeculectomy. Ophthalmology 116, 1713–1718. https://doi.org/10.1016/j.ophtha.2009.04.003 (2009).
Mitchell, P. Chemiosmotic coupling in energy transduction: a logical development of biochemical knowledge. In Membrane Structure and Mechanisms of Biological Energy Transduction 5–24 (Springer, 1972). https://doi.org/10.1007/978-1-4684-2016-6_2
Pearson, T., Galfre, G., Ziegler, A. & Milstein, C. A myeloma hybrid producing antibody specific for an allotypic determinant on “IgD-like” molecules of the mouse. Eur. J. Immunol. 7, 684–690. https://doi.org/10.1002/eji.1830071006 (1977).
Joslyn, M. Methods of Food Analysis (Academic Press, New York, 1950). https://books.google.com.bd/books?id=8b8_f68cyv4C&pg=PP1
Jacobs, M. B. The Chemical Analysis of Foods and Food Products, 2nd edn (Van Nostrand, 1951). https://www.cabdirect.org/cabdirect/abstract/19520401639.
APHA. Standard Methods for the Examination of Water and Wastewater, 23rd edn (American Public Health Association, Washington, DC, 2017).
Lavens, P. & Sorgeloos, P. Manual on the Production and Use of Live Food for Aquaculture. FAO Fisheries Technical Paper No. 361 (FAO, Rome, 1996).
Parsons, T. R., Maita, Y. & Lalli, C. M. A Manual of Chemical and Biological Methods for Seawater Analysis (Pergamon Press, 1984).
Lind, D. L., Geiss, J. & Stettler, W. Solar and terrestrial noble gases in magnetospheric precipitation (1979).
Dobbeleir, J., Adam, N., Bossuyt, E., Bruggeman, E. & Sorgeloos, P. New aspects of the use of inert diets for high density culturing of brine shrimp. Brine Shrimp Artemia 3, 165–174 (1980).
Coutteau, P. & Sorgeloos, P. Feeding of the brine shrimp Artemia on yeast: Effect of mechanical disturbance, animal density, water quality and light intensity. Eur. Aquacult. Soc. Spec. Publ. 10, 344 (1989).
Clegg, J. S. & Trotman, C. N. A. Physiological and biochemical aspects of Artemia ecology. In Artemia: Basic and Applied Biology (eds Abatzopoulos, T. J., Beardmore, J. A., Clegg, J. S. & Sorgeloos, P.) 129–170 (Kluwer Academic Publishers, Dordrecht, 2002). https://link.springer.com/chapter/https://doi.org/10.1007/978-94-017-0791-6_3.
Drinkwater, L. E. & Clegg, J. S. Experimental biology of cyst diapause. In Artemia Biology (eds Browne, R. A., Sorgeloos, P. & Trotman, C. A. N.) 93–117 (CRC Press, Boca Raton, 1991). https://doi.org/10.1201/9781351069892.
Coutteau, P., Dehasque, M., De Wolf, T., Nys, C. & Van Assche, J. Specialty feeds in marine larviculture. Aquacult. Sci. 46, 411–416. https://doi.org/10.11233/aquaculturesci1953.46.411 (1998).
Soniraj, N. Effect of salinity on the life span and reproductive characteristics of brine shrimps in the salt pans at Tuticorin. J. Mar. Biol. Assoc. India 46, 133–140 (2004). http://eprints.cmfri.org.in/id/eprint/2018.
Sorgeloos, P. The use of the brine shrimp Artemia in aquaculture. In The Brine Shrimp Artemia 3, 25–46 (1980).
Soundarapandian, P. & Saravanakumar, G. Effect of different salinities on the survival and growth of Artemia sp. Curr. Res. J. Biol. Sci. 1, 20–22 (2009). .
Naegel, L. C. & Rodríguez-A, S. Ecological observations and biomass proximate composition of the brine shrimp Artemia (Crustacea: Anostraca) from Pichilingue, Baja California Sur. Mexico. Hydrobiologia 486, 185–190. https://doi.org/10.1023/A:1021302918982 (2002).
Kulasekarapandian, S. & Ravichandran, P. Artemia cyst production at Kelambakkam near Chennai. J. Mar. Biol. Assoc. India 45, 166–177 (2003).
Douillet, P. H. Effect of bacteria on the nutrition of the brine shrimp Artemia fed on dried diets. In Artemia Research and Applications: Ecology, Culturing, Use in Aquaculture, Vol. 3 (eds Sorgeloos, P., Bengston, W. & Jasper, E.) 295–308 (University Press, Wettern, Belgium, 1987). https://cir.nii.ac.jp/crid/1573387449033873024.
Vanhaecke, P., Cooreman, A. & Sorgeloos, P. International study on Artemia. XV. Effect of light intensity on hatching rate of Artemia cysts from different geographical origin. Mar. Ecol. Prog. Ser. 5, 111–114 (1981).
Triantaphyllidis, G., Abatzopoulos, T. & Sorgeloos, P. Review of the biogeography of the genus Artemia (Crustacea, Anostraca). J. Biogeogr. 25, 213–226. https://doi.org/10.1046/j.1365-2699.1998.252190.x (1998).
Gilchrist, B. M. Growth and form of the brine shrimp Artemia salina (L). Proc. Zool. Soc. Lond. 134, 221–235 (1960). https://doi.org/10.1111/j.1469-7998.1960.tb05590.x.
Camargo, W. N., Duran, G. C. & Sorgeloos, P. Influence of some physicochemical parameters on Artemia biomass and cyst from the Colombia Caribbean. J. World Aquacult. Soc. 35, 296–305. https://doi.org/10.1111/j.1749-7345.2004.tb01085.x (2004).
Correa, F. et al. Effect of temperature and salinity on the biochemical composition of Artemia franciscana Kellogg 1906, from Araya (Venezuela) and San José (México). Int. Rev. Hydrobiol. 84, 579–585. https://doi.org/10.1002/iroh.199900050 (1999).
Lavens, P. & Sorgeloos, P. Manual on the Production and Use of Live Food for Aquaculture FAO Fisheries Technical Paper No. 361 (FAO, Rome, 1996). https://www.cabdirect.org/cabdirect/abstract/19971409717.
Nguyen, T. N. A., Nguyen, V. H., Van Stappen, G. & Sorgeloos, P. Effect of different supplemental feeds on proximate composition and Artemia biomass production in salt ponds. Aquaculture 286, 217–225. https://doi.org/10.1016/j.aquaculture.2008.09.030 (2009).
Acknowledgements
The authors would like to thank to all the lab members of the Institute of Marine Sciences, University of Chittagong, and the Ministry of Science and Technology, Bangladesh special gratitude to the Department of Agricultural Chemistry, Khulna Agricultural University, Bangladesh for their support to fulfil this work.
Funding
The authors extend their appreciation to the Deanship of Research and Graduate Studies at King Khalid University for funding this work through Large Research Project under grant number RGP2/157/46.
Author information
Authors and Affiliations
Contributions
Author contributions: MHR: Conceptualization and design, Material preparation, Data collection, Methodology, Analysis, and Writing—original draft. TKR : Methodology, Supervised, Writing—reviewed, and edited the manuscript. MWA : Conceptualization, formal analysis, writing—reviewed, and edited the manuscript. MSI : Methodology, Supervised, wrote, reviewed, and edited the manuscript. MSH : Conceptualization and design, Material preparation, writing—reviewed, and edited the manuscript. AAB : Material preparation, Data collection, Formal Analysis, and Writing—original draft. NB : Writing—reviewed and edited the manuscript. SAH : Material preparation, Data collection, Methodology, Analysis, and Writing—original draft. MSI : Material preparation, Data collection, Methodology, Analysis, and Writing—original draft. ZI : Material preparation, Data collection, Methodology, Analysis, and Writing—original draft. AMI : Material preparation, Data collection, Methodology, Analysis, and Writing—original draft. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
I declare that the authors have no competing interests as defined by Springer, or other interests that might be perceived to influence the results and/or discussion reported in this paper.
Dual publication
The results/data/figures in this manuscript have not been published elsewhere, nor are they under consideration by another publisher.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Rahman, M.H., Roy, T.K., Alam, M.W. et al. Enhancement of Artemia biomass and cyst production using supplementary feeds: a preliminary study. Sci Rep 15, 41148 (2025). https://doi.org/10.1038/s41598-025-24882-z
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-24882-z