Abstract
This study aimed to evaluate the impact of unilateral biportal endoscopy (UBE) assisted interbody fusion on the functional recovery of patients diagnosed with lumbar disc herniation (LDH). From October 2021 to October 2022, 132 patients with LDH were selected as the research objects. The clinical data were retrospectively analyzed. According to the different surgical methods, LDH patients were divided into experimental group and control group (n = 66). The control group underwent conventional posterior lumbar open interbody fusion surgery, while the experimental group received interbody fusion surgery assisted by UBE. The clinical efficacy and perioperative indicators (operation time, total blood loss, 48 h postoperative drainage volume, and length of hospital stay) were recorded and compared between the two groups. The lumbar spine imaging parameters (lumbar lordosis index, posterior disc height, intervertebral space height, and Cobb Angle) before and after treatment were compared between the two groups. Lumbar function was evaluated using the Japanese Orthopedic Association (JOA) score, the Visual Analogue Scale (VAS), and the Oswestry Disability Index (ODI). Quality of life was assessed using the World Health Organization Quality of Life Scale (WHO-QOL-BREF). The incidence of complications after treatment was compared between the two groups. Multivariate logistic regression analysis was employed to identify influencing factors. In terms of low back pain improvement, the effective rate of the experimental group was 96.97%, which was significantly higher than 80.30% of the control group (P < 0.01). In terms of leg pain improvement, the effective rate of the experimental group was 95.45%, which was significantly higher than 75.76% of the control group (P < 0.01). The operation time and length of hospital stay in the experimental group were shorter than those in the control group (P < 0.001). The total blood loss and 48 h postoperative drainage volume in the experimental group were significantly less than those in the control group (P < 0.001). The lumbar lordosis index and posterior disc height of the two groups after operation were significantly higher than those before operation (P < 0.05). The intervertebral space height and Cobb Angle of the two groups after operation were significantly lower than those before operation (P < 0.05). The improvement of lumbar imaging parameters in the experimental group after operation was significantly better than that in the control group (P < 0.01). The JOA scores of the two groups after operation were significantly higher than those before operation, and the VAS scores of low back pain and leg pain and ODI scores were significantly lower than those before operation (P < 0.05). The improvement of the above indexes in the experimental group after operation was significantly better than that in the control group (P < 0.01). The incidence of complications in the experimental group was significantly lower than that in the control group (P < 0.01). The WHOQOL-BREF scores of the two groups after surgery were significantly higher than those before surgery (P < 0.05). The improvement of WHOQOL-BREF score in the experimental group after surgery was significantly better than that in the control group (P < 0.001). Multivariate Logistic regression analysis showed that UBE-assisted interbody fusion was an independent protective factor for good clinical efficacy in patients with LDH after adjusting the covariates of clinical symptom distribution, trauma history and Modic changes (P < 0.05). UBE interbody fusion has certain clinical advantages in the treatment of LDH. Compared with conventional open surgery, UBE interbody fusion has less bleeding and faster postoperative recovery. Moreover, UBE interbody fusion can relieve the symptoms of low back pain, promote the recovery of lumbar function and improve the quality of life. UBE interbody fusion is a safe and feasible choice in clinical treatment.
Similar content being viewed by others
Introduction
Lumbar disc herniation (LDH) is a disease caused by degeneration of the intervertebral disc, rupture of the annulus fibrosus, and stimulation or compression of the spinal nerve root and cauda equina nerve by protrusion of the nucleus pulposus. LDH is one of the common and important causes for low back and leg pain. Age, inactivity, and long periods of sitting can result in the occurrence of LDH. Physical therapy, traction, rest and medication therapy can often improve symptoms in patients with symptomatic LDH. Surgery should be considered if there is no improvement after more than six weeks to three months of physical and medicinal therapy1,2. Additionally, patients with severe disease, leg skin numbness, hypoesthesia, bowel and urine dysfunction, recurrent in the same segment, instability, bilateral foraminal stenosis, unilateral foraminal stenosis with high collapse of intervertebral disc and other symptoms, should choose surgical treatment in time3. Transforaminal lumbar interbody fusion is a widely used lumbar fusion procedure. However, due to the damage to the posterior structure of the spine and prolonged nerve traction during the operation, some patients may have intractable low back pain, which impairs the effectiveness of treatment4. Spinal surgeons both domestically and overseas have continued to investigate minimally invasive procedures in recent years. Unilateral biportal endoscopy (UBE) has been progressively incorporated into clinical treatment as a result of the gradual advancement of spinal endoscopic technology. UBE is a new minimally invasive spine endoscopic technique, which is performed through two channels (one visual channel and one operating channel). The decompression of central spinal canal, bilateral nerve roots and lateral recess stenosis can be completed under the assistance of arthroscopy. UBE is a technique that can be performed under the microscope a technique of interbody fusion5,6. When compared to open surgery, UBE is a less invasive procedure that offers reduced trauma, bleeding, and infection rates. These benefits contribute to the patients’ continued physical recovery7. UBE interbody fusion is one of the minimally invasive interbody fusion methods used in clinical practice in recent years., UBE-assisted interbody fusion uses two independent channels to treat lumbar degenerative diseases with less damage to paravertebral muscles. This study made the assumption that the therapy of LDH might still be affected by this method.
The purpose of this study was to investigate the effectiveness of UBE assisted interbody fusion in treating LDH and the influence on the functional recovery of patients. The clinical effect of UBE interbody fusion and conventional posterior open lumbar interbody fusion was analyzed and compared. The advantages of UBE interbody fusion in the treatment of LDH were evaluated, which provided evidence for clinical treatment. This work has significant practical implications for raising the standard of LDH treatment and enhancing patient quality of life.
Materials and methods
General materials
Propensity score matching
This study was a retrospective analysis. The research subjects were 168 patients with LDH who were initially admitted to our hospital from October 2021 to October 2022. All clinical data were retrospectively extracted from the hospital’s electronic medical record system and follow-up records. To control confounding bias, we used Propensity Score Matching (PSM) for inter-group balancing.
The specific methods for propensity score matching were as follows: Covariate selection and designation: The propensity score model included the following covariates, all of which were common confounding factors in clinical practice: age (continuous variable, unit: years), BMI (continuous variable, unit: kg/m²), disease duration (continuous variable, unit: years), lesion segment (categorical variable: L4-5 or L5-S1), and degree of spinal canal stenosis (categorical variable: mild, moderate, severe, based on MRI-measured lateral recess width).
Matching method: The 1:1 nearest neighbor matching method was adopted, the caliper width was set to 0.2, and there was no replacement matching to ensure the maximum similarity between matching groups.
Matching process and sample size: After PSM matching in the initial 186 patients, 132 patients were finally included (66 in the experimental group and 66 in the control group), and 54 patients were excluded due to unmatched or covariates exceeding the common support area. After matching, the sample sizes of the two groups were balanced, meeting the requirements of statistical analysis.
Balance diagnosis: The balance of covariates before and after matching was evaluated by Standardized Mean Difference (SMD). After matching, the SMD of all covariates was less than 0.1, indicating a good balance of covariates between groups (Table 1). The changes in SMD before and after matching were visually presented through the Love plot (covariate balance plot) (Fig. 1).
Balance Plot of covariates before and after propensity score matching (Love plot). The surgical method a patient should undergo was mainly determined by the attending physician based on a comprehensive consideration of the patient’s specific condition (such as the segment of the lesion, the type of protrusion, the degree of spinal stenosis, previous surgical history, etc.), the patient’s wishes, the medical conditions at that time, and the maturity of the technology. Although no prospective efficacy analysis was conducted in advance, post hoc analysis using G*Power 3.1 software showed that: When α = 0.05 and β = 0.2 (efficacy = 80%), the sample size of 66 cases in each group was sufficiently effective in detecting differences in perioperative indicators (such as blood loss, operation time) and functional scores (such as VAS, ODI) (effect size > 0.8).
Common support assessment: The propensity score distribution showed that there was sufficient overlap between the experimental group and the control group in the common support area. After matching, the propensity score distributions of the two groups were similar, and no individuals with extreme propensity scores occurred, indicating a good matching effect.
The research objects were 132 patients with LDH admitted to our hospital from October 2021 to October 2022. All clinical data were retrospectively extracted from the hospital’s electronic medical record system and follow-up records. The general data selection process was shown in Fig. 2. The clinical data of these subjects were retrospectively analyzed. According to the time of surgery and technical application, patients who underwent UBE interbody fusion were included in the experimental group. Patients who underwent conventional posterior open lumbar interbody fusion during the same period or earlier were included in the control group by propensity score matching (1:1). The choice of surgical procedure was determined by the attending physician according to the patient’s specific condition (such as lesion segment, herniation type, degree of spinal stenosis, previous surgical history, etc.), patient’s willingness, medical conditions at that time, technical maturity and other factors. A prospective Power analysis was not performed. However, a post hoc analysis using G*Power 3.1 software showed that a sample size of 66 patients per group had sufficient power (effect size > 0.8) to detect differences in perioperative indicators (e.g., blood loss, operation time) and functional scores (e.g., VAS, ODI) with α = 0.05 and β = 0.2 (power = 80%). The experimental group consisted of 66 cases of patients, 35 males and 31 females. The ages of the patients ranged from 38 to 80 years old, with a median age of 59.0 ± 14.5 years. Their average body mass index (BMI) was 25.0 ± 2.5 kg/m2, with a range of 18 to 30 kg/m2. In the experimental group, the disease course lasted an average of 2.0 ± 0.5 years, ranging from 1 to 3 years. Within the experimental group, there were 30 cases of patients with L4 − 5 segment and 36 cases of patients with L5-S1. The control group consisted of 66 cases of patients, 40 males and 26 females. The ages of the patients ranged from 39 to 81 years old, with a median age of 60.0 ± 15.0 years. Their average BMI was 26.0 ± 2.5 kg/m2, with a range of 17 to 33 kg/m2. In the control group, the disease course lasted an average of 2.3 ± 1.0 years, ranging from 0.5 to 4 years. Within the control group, there were 32 cases of patients with L4 − 5 segment and 34 cases of patients with L5-S1. There was no statistically significant difference in age, gender, BMI, and course of disease between two groups (P > 0.05).
The inclusion process of the general data of 132 patients.
Inclusion criteria for the experimental group: (1) The clinical symptoms of patients were aligned with the diagnosis of LDH in the Surgery (the 3rd Edition)8; (2) Imaging diagnosis (CT and MRI) was performed to confirm the diagnosis. Imaging results (CT and MRI) showed that the lesion segment was limited to a single level of L4-5 or L5-S1; (3) BMI ≤ 30 kg/m² (To avoid paravertebral muscle thickness affecting access establishment); (4) The degree of spinal canal stenosis was mild to moderate (lateral recess stenosis < 3 mm without severe central spinal canal stenosis); (5) No severe spinal instability (vertebral slip < 3 mm on hyperextension/flexion radiographs); (6) Conservative treatment for more than 6 months was ineffective or recurrent, combined with spinal instability, severe spinal stenosis, recurrent herniation or severe loss of intervertebral disc height. Decompression surgery alone was difficult to achieve long-term efficacy, thus fusion surgery was necessary to restore spinal stability, relieve nerve compression and prevent disease progression. Inclusion criteria for the control group: (1) Multi-segment disease can be combined (but in this study, all patients were single-segment, matching the experimental group); (2) There were no strict BMI restrictions (the mean BMI of the control group was 26.0 ± 2.5 kg/m² in this study, which was similar to that of the experimental group); (3) Combined with moderate to severe spinal stenosis or mild spinal instability (slip 3–5 mm).
Exclusion criteria: Patients with a history of lumbar disc surgery; Patients had structural variations of the lumbar spine, including congenital sacral lumbarization, sacral lumbarization, pedicle dysplasia (such as pedicle absence and abnormal morphology), vertebral insufficiency (such as hemivertebra, butterfly vertebra), spina bifida, etc. The above variations were diagnosed by preoperative lumbar CT three-dimensional reconstruction; Patients with severe spinal stenosis, scoliosis and spinal tumors; Patients with aberrant mental states; Patients with intervertebral foramen or intervertebral disc herniation outside of the intervertebral foramen; Patients with central spinal stenosis; Patients with other spinal diseases, such as spinal tumors and osteoporotic fractures; Patients with abnormal coagulation routine or long-term oral anticoagulants prior to surgery; Patients with incomplete information; Patients who had poor general health and cannot endure surgery. The inclusion criteria, exclusion criteria and observation indicators (such as perioperative parameters, imaging parameters, functional scores, etc.) of this study were prospectively formulated based on clinical routine and previous studies to standardize the screening and extraction of retrospective data and ensure data consistency.
To reduce the selection bias that may be introduced by the retrospective single-center design, the following measures were taken:
Standardized inclusion/exclusion criteria: Patients in the two groups were strictly screened according to unified criteria (such as single-segment disease, failure to respond to conservative treatment for ≥ 6 months). Data were extracted through structured search of the electronic medical record system, and cases with incomplete data or failure to meet the criteria were excluded to ensure the homogeneity of the enrolled population.
Unified surgical team and evaluation criteria: All operations were performed by the same group of surgeons with more than 5 years of spinal surgery experience to avoid the influence of technical differences between the operators. The efficacy was evaluated using standardized scales (VAS, ODI, JOA), which were double-blind judged by two attending physicians of the non-surgical team to reduce subjective bias.
Transparent grouping: The grouping was based on the promotion stage of surgical techniques (open surgery was the main stage from 2021.10 to 2022.3, and UBE was gradually promoted from 2022.4 to 2022.10), rather than subjective choice. The risks and benefits of the two procedures were fully informed to the patients before operation. The informed consent of the patients was combined to avoid preferential allocation of doctors.
Methods
Preoperative preparation: All patients completed relevant preoperative examinations, including blood routine, electrolytes, liver and kidney function, coagulation function, infectious diseases, urine routine, blood pressure monitoring, lower limb veins, electrocardiogram and cardiac ultrasound examination, chest X-ray, lumbar spine anteroposterior and lateral X-ray, lumbar spine hyperextension and flexion lateral X-ray, and lumbar spine MRI examination. The patient’s past medical history was clarified. The recent history of oral reserpine, aspirin and other perioperative contraindications was excluded. Before the operation, relevant education was carried out. The complications related to the operation were communicated with the patients and their families, and the possible risks were informed. The consent of the patients and their families was obtained, and the informed consent was signed. Routine blood preparation was carried out before the operation, and the patient was informed to fast water for 12 h. All patient surgeries were performed by the same team of experienced attending physicians.
The choice of surgical method was based on the following factors: (1) Anatomical considerations: Patients with single-segment disease and without severe scoliosis or spondylolisthesis were more suitable for UBE interbody fusion. Patients with multi-segment disease, severe spinal instability or complex anatomical structure (such as pedicle variation) were more likely to be treated with open surgery. (2) Surgeon assessments: The patients were judged by the same group of attending doctors with more than 5 years of experience in spinal surgery according to the anatomical display under intraoperative fluoroscopy and the feasibility of operation. (3) Patient selection: After fully informing the patients about the trauma, recovery time and risks of the two surgical methods, the decision was made according to the patient’s preference for minimally invasive treatment.
Patients in the control group received the conventional posterior lumbar open lumbar interbody fusion surgery. After being given general anesthesia, the patient was positioned prone. Their chest and abdomen were cushioned and spread with conventional disinfection and sterile sheet. An incision about 6 cm long was created in the posterior median incision at the 4–5 lumbar vertebrae. The supraspinous ligament in the deep fascia was then retained when the skin was cut open. The paraspinal muscles were severed along the supraspinous ligament to the lateral facet joints. A pedicle screw was placed on both sides of L4 and L5. C-arm fluoroscopy was used to observe the screw positions. A portion of the zygapophysial joint on the right side of the waist 4/5 was excised to expose the dural sac and nerve root after confirming that the position was satisfactory. Subsequently, the adhesion surrounding the nerve roots was peeled and the intervertebral disc in the lumbar 4/5 space was removed. During exploration, a scraping procedure was carried out until no intervertebral disc tissue remained. After implanting autologous bone particles into the patient’s intervertebral space, an interbody fusion cage made of matched autologous bone was put in place. An iron rod was used to join and fasten the pedicle screws on both sides. After decompression of the intervertebral space, a polyetheretherketone (PEEK) Cage was implanted. The size selection criteria were the same as those of the experimental group (height 10–12 mm, width 16–18 mm), and the type of cage was the same as that of the experimental group (sagittal lordosis Angle 5°). Autologous bone particles were implanted to ensure that the material, shape and size of the cage were not statistically different between the two groups. A C-arm fluoroscopy was necessary to check the implant’s location once more after fixation. Following confirmation, the incision required to be periodically cleaned and hemostasis had to be carried out. Layer-by-layer suturing was done after covering the dura mater with gelatin sponge and positioning a drainage tube on one side of the incision. The surgical instruments and dressings used were counted. The incision can be closed layer by layer only after the count was correct and the patient’s wound was not actively bleeding.
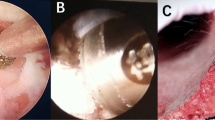
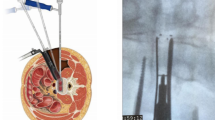
Patients in the experimental group received UBE-assisted interbody fusion surgery. After being given general anesthesia, the patient was positioned prone. The surgical site was identified using C-arm fluoroscopy by projecting the pedicle’s body surface on both sides of the waists 4 and 5. Standard cleaning and sterile sheeting were then carried out. Based on the indicated points, lateral puncture of the pedicle was done. Fluoroscopic examination of the puncture needle’s location during the procedure was necessary to determine whether it was at the vertebral arch’s pedicle. Following confirmation, the guide wire could be placed. The operation and observation channels could be built by making a double incision along the left side to introduce the skin expander. It was necessary to insert a 30° arthroscope into the observation channel after connecting the light source. Following a clean removal of the surrounding soft tissue in the work channel, adequate hemostasis was achieved with the use of a radiofrequency knife. The field of vision was exposed to the lower edge of the L4 vertebral plate and the higher edge of the L5 vertebral plate. To expose the dura mater and nerve roots in the visual field, the lumbar 4 lamina was removed from the ligamentum flavum insertion point, the upper edge of the lumbar 5 lamina and part of the facet joints of the lumbar 4/5 were removed, and the ligamentum flavum was also removed. Subsequently, protruding lumbar 4/5 intervertebral discs, as well as compressed posterior dura mater and nerve roots could be observed. The nerve roots were pulled apart using nerve stripping ions. The intervertebral disc was removed. The intervertebral space was processed to the upper and lower endplates. The bone graft bed was prepared. The protruding nucleus pulposus was retrieved with nucleus pulposus forceps. Hemostasis and intervertebral disc ablation were carried out with a radiofrequency knife, and the endplate was scraped off. After the intervertebral space was processed, the polyetheretherketone (PEEK) Cage was implanted. The size of the cage was selected according to the height (10–12 mm) and width (16–18 mm) of the intervertebral space measured by CT before operation. The cage was built with autologous bone, and the cage was high in anterior and low in posterior (sagittal lordosis Angle 5°) to maintain the physiological lordosis of the lumbar spine. Radiofrequency ablation hemostasis could be carried out once more when the nerve root had relaxed and the dura mater pulsation was found to be normal. After implanting autologous bone particles into the patient’s intervertebral space, an interbody fusion cage made of matched autologous bone was put in place. The arthroscopy could be removed once the fusion device was properly positioned during the fluoroscopy. The pedicle screw was then screwed along the guide wire, the round rod was positioned, and the screw plug was tightened and secured. The instruments and dressings could be counted once the C-arm fluoroscopy had confirmed the internal fixture’s position and proper length. A drainage tube could be inserted once it was determined that no mistakes had been made and the patient’s incision was not actively bleeding. Disinfection was performed again, and the incision was sutured. Patients in both groups were followed up for 3 months. The operation diagram was shown in Fig. 3. Lumbar spine images before and after surgery were shown in Figs. 4 and 5.
Schematic of the operation of UBE-assisted interbody fusion.
Images of the lumbar spine before surgery in a patient with LDH.
Images of the lumbar spine after surgery in a patient with LDH.
Postoperative treatment: After the operation, patients were instructed to exercise their lower limbs properly in bed to prevent thrombosis. Low-dose hormone (dexamethasone sodium phosphate injection, 5 mg, i.v, once daily for 3 days) was given to reduce nerve root edema. Its intended use was to reduce the edema of perineural tissue by inhibiting inflammatory response, relieve the pain and numbness caused by nerve root compression, and promote the early recovery of nerve function. Patients received antibiotics to avoid infection. The postoperative incision drainage volume was recorded. The drainage tube was withdrawn if the drainage volume was less than 30 mL. Patients were provided braces to enable them to get out of bed properly, based on the extubation time. Blood routine, electrolytes, erythrocyte sedimentation rate, C-reactive protein and other indicators were regularly detected after operation. The patient was treated with non-steroidal anti-inflammatory drugs, neurotrophic drugs, and blood-activating drugs after operation. Lumbar MRI and lumbar anteroposterior and lateral X-ray were reexamined timely after operation. Stitches were removed according to the healing of the incision. Patients were reexamined at the outpatient clinic in three months and a year after being discharged. Lumbar MRI and lumbar anteroposterior and lateral examinations were performed. Patients were routinely contacted by telephone or WeChat for follow-up.
Outcome measures
(1) Treatment effectiveness: Following the procedure, the clinical efficacy was assessed three months later. The study was conducted by two attending physicians (non-surgical team) in a blinded manner, combining the following three aspects: (1) Subjective symptoms of patients (relief of low back pain and leg pain was recorded by structured interview); (2) Medication use (postoperative analgesic prescription records); (3) Daily function (referring to the item “limitation of daily activities” in ODI score). Marked effectiveness represented that the patient’s low back and leg pain disappears, and did not need to start analgesic treatment, which almost not affect the patient’s normal life. Effectiveness represented that the patient’s waist and leg pain was relieved, but the patient needed to take painkillers at night to treat so that the patient can sleep and rest. Invalid represented that the patient’s back and leg pain did not improve, the patient cannot sleep, and had adverse effects on the patient’s work and daily life. Effective rate = cases of (marked effectiveness + effectiveness)/total number of cases * 100%.
(2) Perioperative situation: The surgical time, total blood loss, postoperative drainage volume at 48 h, and hospitalization time of patients in the two groups were recorded.
(3) Lumbar imaging parameters: The lumbar curvature index, posterior edge height of intervertebral discs, intervertebral space height (defined as the vertical distance from the line connecting the midpoints of the posterior edge of the vertebral body, unit: mm), Cobb angle (unit: °) of patients in the two groups were measured using X-ray films before and 3 months after surgery.
(4) Lumbar function: The degree of low back pain was assessed using the visual analogue scale (VAS) before and 3 months after surgery, with a total of 10 points. The specific procedures were as follows: Using a horizontal marker, the patient’s level of pain was recorded. One end of the horizontal line had the number 0 to indicate no pain, and the other end had the number 10 to indicate extreme pain. Patients’ levels of pain were represented by several locations, where lower scores corresponded to less pain and higher scores to more pain. The functional recovery of patients was evaluated using the Lumbar Oswestry Dysfunction Index (ODI) before and 3 months after operation. Higher score indicated poorer recovery of the patient’s physical function, and vice versa. Based on the patient’s functional state, the Japanese Orthopedic Association (JOA) score took into account constraints on subjective symptoms, clinical signs, and daily activity limitations, with a total score of 29 points. A lower score indicated a more pronounced level of patient dysfunction, and vice versa.
(5) Comparison of complications: The percentage of complications of patients was recorded and calculated before and 3 months after operation using three different indicators, including dural tear, nerve root injury, and wound infection. Muscle atrophy was evaluated by measuring the cross-sectional area (CSA) of bilateral multifidus and erector spinae muscles by lumbar MRI at 3 months after operation, and comparing with the preoperative CSA of corresponding muscles. When the postoperative CSA was decreased by ≥ 15% compared with the preoperative CSA, muscle atrophy was defined. MRI examination at 3 months after operation showed that the CSA reduction of bilateral paraspinal muscles was less than 15% in both groups, so there was no muscle atrophy.
(6) World Health Organization Quality of Life Scale (WHO-QOL-BREF): The quality of life of patients was evaluated before and 3 months after operation. There were 24 items on this scale, each with 1 ~ 5 points. The score was calculated using a range of factors from good to poor, including seven items totaling 35 points in the physiological domain, six items totaling 30 points in the psychological domain, eight entries totaling 40 points in the environmental domain, and three items totaling 15 points in the social relations domain. Higher scores indicated better quality of life.
Statistical analysis
SPSS24.0 statistical software was used to analyze the data. The measurement data were expressed as mean ± standard deviation and compared using t-test. Cohen’s d and 95% confidence interval (CI) were used to represent the effect size of the difference between the two groups. The enumeration data were expressed as percentages and compared using χ 2 test. OR and 95% CI were used to represent the effects of the difference between the two groups. Multivariate logistic regression was used to analyze the influencing factors of clinical efficacy in patients with LDH. The difference was statistically significant when P < 0.05.
Results
Comparison of treatment effects
The effective rate of surgery in the experimental group was 97.0%, which was higher than that in the control group with 80.3% in terms of low back pain improvement (P < 0.01; OR = 7.9 [95%CI: 1.7, 36.3], Table 1). In terms of leg pain improvement, the effective rate of surgery in the experimental group was 95.5%, which was higher than that in the control group with 75.8% (P < 0.01; OR = 6.7 [95%CI: 1.9, 24.4], Tables 2 and 3).
Comparison of perioperative conditions
Patients in the experimental group had shorter operative time and hospital stay (P < 0.001; 0.8 (95%CI: 0.6, 1.2); 1.15 (95%CI 1.0, 1.9)), and less total blood loss and postoperative 48-h drainage as compared with these in the control group (P < 0.001). Effect size Cohen’s d values were 1.1 (95%CI 0.9, 1.3) and 0.9 (95%CI 0.7, 1.1), respectively (Table 4; Fig. 6).
Comparison of perioperative conditions. A: Comparison of surgical time; B: Comparison of total blood loss; C: Comparison of drainage volume 48 h after surgery; D: Comparison of hospitalization time. Note: #P < 0.05 compared with the control group.
Comparison of lumbar imaging parameters
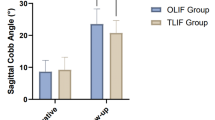
There was no statistically significant difference in preoperative lumbar imaging parameters between the two groups (P > 0.05). The lumbar curvature index and posterior margin height of intervertebral discs in both groups after surgery were significantly higher than those before surgery, while the intervertebral space height (reflecting the objective changes in the space after surgical decompression and fusion device implantation) and Cobb Angle (segmental lortosis Angle) were significantly lower than those before surgery (P < 0.05). After surgery, the improvement of lumbar imaging parameters was more obvious in the experimental group than these in the control group (P < 0.01). The effect size Cohen’s d values were 1.1 (95%CI 1.0, 1.3), 1.3 (95%CI 1.1, 2.3), 1.1 (95%CI 1.1, 1.4) and 1.1 (95%CI 1.1, 2.4), respectively (Tables 5 and 6; Fig. 7).
The lumbar spine imaging parameters were compared between the two groups. A: Comparison of lumbar curvature index; B: Comparison of intervertebral space height; C: Comparison of posterior edge height of intervertebral discs; D: Comparison of Cobb angle. Note: *P < 0.05 compared with the same group before surgery, #P < 0.05 compared with the experimental group after surgery.
Comparison of JOA score, pain score, and ODI score
There was no statistically significant difference in preoperative JOA score, VAS score of low back pain and leg pain, and ODI score between the two groups (P > 0.05). After surgery, the JOA score was elevated, the VAS score of low back pain and leg pain and ODI score were decreased in the both two groups (P < 0.05). The improvement in JOA score, VAS score of low back pain and leg pain and ODI score was more obvious in the experimental group than that in the control group (P < 0.01). The effect size Cohen’s d values were 0.9 (95%CI 0.7, 1.5), 1.1 (95%CI 1.0, 2.1), 2.7 (95%CI 1.5, 4.4) and 1.8 (95%CI 1.6, 2.1), respectively (Tables 7 and 8; Fig. 8).
Comparison of JOA score, pain score and ODI score. A: Comparison of JOA scores; B: Comparison of lower back and leg pain score; C: Comparison of VAS scores of leg pain; D: Comparison of ODI scores. Note: *P < 0.05 compared with the same group before surgery, #P < 0.05 compared with the experimental group after surgery.
Comparison of complications
There was no muscle atrophy in the two groups. The incidence of complications in the experimental group was 3.03%, which was much lower than that in the control group with 16.7% (P < 0.01, OR = 6.4, 95% CI: 3.6–30.1, Table 9).
Comparison of the quality of life
There was no significant difference in WHOQOL-BREF score between the two groups before surgery (P > 0.05). The WHOQOL-BREF score of the two groups after surgery was higher than that before surgery (P < 0.05), and the improvement of WHOQOL-BREF score in the experimental group after surgery was significantly better than that of the control group (P < 0.001). The effect size Cohen’s d values were 2.7 (95%CI 2.4, 3.0), 1.9 (95%CI 1.4, 2.1), 2.7 (95%CI 2.0, 3.9) and 1.4 (95%CI 1.1, 1.8), respectively (Tables 10 and 11).
Single factor analysis of factors affecting clinical curative effect of patients with LDH
Among 132 patients with lumbago and leg pain, the surgery was effective for 113 patients and 19 patients were ineffective. There was a significant difference in the distribution of clinical symptoms, history of trauma, Modic changes and surgical methods between the effective group and the ineffective group (P > 0.05, Table 12).
Multivariate logistic regression analysis of the influencing factors of clinical efficacy in patients with LDH
The clinical efficacy (effective = 0, ineffective = 1) of patients with major lumbar disc surgery was used as the dependent variable, and the statistically significant indicators in Table 12 were served as the independent variables for multivariate logistic regression analysis. The results showed that UBE-assisted interbody fusion surgery was an independent protective factor for good clinical efficacy in patients with LDH after adjusting the covariates of clinical symptom distribution, trauma history and Modic changes (OR = 0.760, 95%CI: 0.618–0.936, P < 0.05, Table 13). The interaction test between trauma history and surgical method showed that the interaction term was not statistically significant (B = 0.192, P = 0.241; OR = 1.212, 95%CI: 0.881–1.667).
Subgroup analysis: In patients without trauma history, the effective rate was 98.1% in the experimental group and 82.1% in the open surgery group (P < 0.001). In patients with a history of trauma, the effective rate was 83.3% in the experimental group and 60.0% in the open surgery group (P = 0.317).
Discussion
For LDH, a wide range of therapies are available, including some conservative methods such as physical therapy, massage and acupuncture. However, not every patient responds well to conservative therapy, and the appropriate treatments should to be selected based on the patient’s condition. Especially for patients with more serious low back pain and leg pain that has seriously affected daily life, it is necessary to seek medical treatment in time, and choose the appropriate surgical treatment according to the doctor’s advice, so as to relieve the disease9,10. Nowadays, minimally invasive surgery has steadily replaced open surgery as the primary surgical treatment option. Although conventional lumbar fusion can also play a certain therapeutic effect, open surgery is required. Therefore, the patient’s trauma is large and the postoperative recovery time is slow, which is not the best choice for the patient. People’s needs for surgical operations are evolving along with the ongoing development of living standards. In addition to hoping to treat the disease directly, they also want to reduce trauma and scarring from the surgical incision. Endoscopy has become increasingly common in clinical therapy because to advancements in medical technology. Its benefits include minimum trauma, quick recovery, and low cost11,12. UBE is a new endoscopic assisted lumbar decompression procedure that combines endoscopy and open surgery. In the portal layout, UBE technique was used with 5 mm observation + 15 mm working two independent channels, 10–15 mm apart. Single-port percutaneous endoscopic transforaminal lumbar interbody fusion has only one 10–12 mm channel, and the field of view overlaps with the operating space. UBE decompression and lumbar interbody fusion are both UBE techniques, but the channels are more inclined to the lateral approach. In terms of instrument use, the UBE technique is compatible with conventional open surgical instruments. Percutaneous endoscopic transforaminal lumbar interbody fusion relies on dedicated endoscopic instruments, which limits operational flexibility13. In terms of the workflow of fusion, the unilateral dual-channel endoscopic technique is consistent with the double-channel decompression of the spinal canal and then the intervertebral space. Percutaneous endoscopic transforaminal lumbar interbody fusion is difficult to implant cage due to the limitation of single channel and step-by-step decompression. In terms of indications, UBE technique is suitable for single-segment lumbar disc herniation with mild to moderate spinal stenosis. Percutaneous endoscopic transforaminal lumbar interbody fusion is more suitable for simple disc herniation. UBE decompression and lumbar interbody fusion is mainly used for lateral interbody fusion, which is suitable for cases with intervertebral foramen stenosis14,15. UBE can be used in conjunction with conventional lumbar fusion surgery after it has been applied to LDH. Combining established notions from spine surgery with newer ideas from endoscopic surgery can enhance surgical results and better serve patients. However, evidence suggests that microdiscectomy for recurrent LDH can lower back and nerve root VAS pain scores and improve JOA scores16. Additionally, it has been proposed that, in comparison to endoscopic spine surgery, minimally invasive spine surgery can enhance the neural sulcus and spinal canal areas of lumbar vertebral segments in lumbar degenerative diseases17.
Relevant results demonstrate that UBE promotes early postoperative recovery and lowers the risk of surgery in patients receiving precise, minimally invasive care18,19. Many forms of LDH, cervical spondylotic radiculopathy, mild lumbar spondylolisthesis, lumbar spinal stenosis, recurrence of intervertebral disc herniation, and spinal revision can all be treated with UBE. Indirect decompression of degenerative lumbar spondylolisthesis by lumbar interbody fusion is suggested by Nikaido T et al20. as a successful treatment for pain and disability. In addition, it significantly improved disc height, disc height and area, and the effect of segmental lordosis on radiological outcomes when compared to the posterior approach. In comparison to the control group, the experimental group showed significantly higher treatment efficacy rates, shorter surgical and inpatient stays, lower rates of total blood loss, and postoperative drainage volume after 48 h in this study. Consequently, these results suggested that UBE-assisted interbody fusion had some clinical application value compared with conventional open surgery in the scope of cases in this study. This may be due to the fact that the treatment process was completed with the assistance of endoscopy, which can combine endoscopy with traditional spinal surgery to ensure the adequacy of decompression and improve the safety of cage implantation. This can not only expand the application range of endoscopic surgery, but also reduce the amount of intraoperative bleeding. Furthermore, there was no need for a lengthy suture because the minimally invasive wound was small. As a result, the operation took less time, which helped patients recuperate and further reduced the length of time they had to stay in the hospital21. In the treatment of severe intervertebral disc herniation, unilateral dual-channel endoscopy has the advantages of lower recurrence rate, lower residual rate of nucleus pulposus, shorter learning curve and better field of vision than single-channel endoscopy, which is worthy of promotion in primary hospitals22. Additional research indicates that UBE offers a broad operating space, a clear and wide field of vision, relatively low surgical equipment needs, and a convenient and flexible operation. These features have been shown to have high clinical efficacy in the treatment of lumbar spinal stenosis and LDH23. According to a different study, UBE clearly benefits patients’ comfort and pain management when treating lumbar disc herniation24. Contrary to the findings of our current study, UBE has the disadvantages of increased total blood loss, intraoperative and occult blood loss, longer surgical time, and longer hospital stay when compared to percutaneous endoscopic lumbar discectomy. The patients who underwent traditional posterior lumbar open lumbar interbody fusion surgery were included in this study as the control group, and the observation of results may have been influenced in part by the use of different surgical controls. The following benefits of UBE-assisted interbody fusion are evident from the study’s findings: The minimal amount of paravertebral musculature that is eliminated can optimize the structural integrity of the spine and guarantee the stability of the spine following surgery25; The procedure is performed under continuous flushing. Radiofrequency electrodes can be used to control bleeding even in cases of peridural sac microvascular bleeding and nerve damage during surgery. Thus, this significantly prevent wound contamination and postoperative infection and expedite recovery; Unlike single-port endoscopic techniques, which use a single rigid working tube (such as an 8–10 mm diameter sleeve), unilateral dual-channel endoscopic techniques operate through two separate flexible channels (the viewing channel is a 5 mm endoscopic sheath, and the working channel is a 15 mm soft tissue expander). There is no rigid tube to limit the range of motion of the instrument, so the decompression is more extensive. It can cover the central spinal canal, lateral recess and intervertebral foramen area. At the same time, with the clear field of view of 30° arthroscopy, the nerves in the target area of the spinal canal can be carefully explored, released and decompressing to achieve sufficient decompression26.
The study’s findings also demonstrated that, following surgery, the experimental group’s improvement in lumbar imaging parameters was noticeably greater than that of the control group. The experimental group’s intervertebral space height and Cobb angle were considerably lower than those of the control group. This indicated that UBE interbody fusion can improve the maintenance and support of intervertebral space during the follow-up period of this study. The reason may be that this operation has less influence on the structure of the spine and can effectively maintain the stability of the lumbar spine after surgery. The interlaminar approach, which is the most often utilized surgical technique for UBE, creates a possible gap between the multifidus muscle and the spinous process with the aid of the C-arm’s location. In this study, the height of the intervertebral space measured after the surgery (defined as the vertical distance from the line connecting the midpoints of the posterior edge of the vertebral body) decreased. This result was different from the conventional understanding that “fusion device implantation increases the intervertebral space”. The main reason for this lied in the differences in surgical strategies: Before the surgery, the pathological posterior margin height of the lesion segment often increased due to intervertebral disc degeneration and protrusion. The surgery aimed to restore the physiological curvature of the lumbar spine (Cobb Angle) and the effective volume of the intervertebral foramen by removing the protruding intervertebral disc tissue, scraping the cartilage endplate and preparing the bone graft bed for the pre-high and post-low type of intervertebral fusion Cage (PEEK Cage, sagittal lordosis Angle 5°), rather than simply pursuing the maintenance of the posterior margin height of the intervertebral space. Therefore, the reduction in the posterior margin height after the surgery reflected the correction from the pathological protrusion state to the functional anatomical position, and it was one of the expected results of the surgery to achieve the goals of nerve decompression and spinal sequence reconstruction. Following the procedure, the experimental group’s WHOQOL-BREF and JOA scores significantly improved, but their VAS and ODI scores fell significantly short of those of the control group. It is possible to draw the conclusion that individuals with LDH may benefit from UBE-assisted interbody fusion surgery in terms of improved lumbar function, decreased discomfort, and increased mobility. Two tiny incisions, one for the working channel measuring 15 mm and the other for the observation channel measuring 5 mm, are all that are needed for UBE-assisted interbody fusion. Minimal surgical incision size lowers the possibility of a difficult recovery after surgery. The entire decompression fusion is done endoscopically throughout the decompression and fusion period. On the whole, this greatly decreases the damage of the skin, subcutaneous tissue and paravertebral muscles, reduces the related complications, and also creates favorable conditions for the patients to recover faster after surgery27,28. Postoperative discomfort in patients is greatly reduced by the reduction in surgical incision and blood loss. With exercise, patients can get out of bed as soon as possible and progressively regain their physical function, which improves their prognosis and quality of life and has favorable implications for their return to work and daily life. However, some patients continue to experience post-operative residual symptoms such leg and waist pain and stiffness, which lowers their quality of life. It was thought in this study that the goal of LDH surgery was to relieve radicular pain and nerve compression brought on by disc herniation. However, symptoms resulting from muscle problems in the lower back and legs persisted, necessitating the need for physical therapy in order to improve lumbar spine stability and alleviate pain and other post-operative symptoms. Researchers observed that the ODI scores of patients undergoing both conventional posterior lumbar interbody fusion and UBE improved one year following surgery, and the ODI scores of the UBE group improved more significantly one week after surgery29. Heo et al30. treat bilateral LDH with UBE technology, and the examination of postoperative efficacy reveals that all postoperative patients have a significant improvement in the VAS and ODI scores for leg pain, indicating good efficacy of the surgery. In conclusion, UBE surgery approach provides good treatment outcomes and considerable advantages in the treatment of lumbar degenerative diseases. The study’s findings also demonstrated that the distribution of clinical symptoms, the history of trauma, and the presence of Modic changes were independent risk factors for the clinical efficacy of patients with LDH. Consequently, interventions tailored to address these risk factors could enhance the efficacy of surgical treatment. Although confounding factors such as a history of trauma were controlled for by means of multivariate regression and interaction analyses, patients with a history of trauma may have been preferentially assigned to the open-surgery group because of complex factors such as tissue adhesion. However, subgroup analysis showed that even in patients with a history of trauma, the treatment effect in the experimental group still showed a trend of superiority (83.3% vs. 60.0%), and the main conclusion was supported by the large sample size in the subgroup without a history of trauma (P < 0.001).
In general, under the conditions of this study, UBE-assisted interbody fusion in the treatment of LDH has the characteristics of less bleeding and faster postoperative recovery. UBE-assisted interbody fusion has a positive effect on relieving symptoms and improving lumbar function and quality of life. UBE-assisted interbody fusion can provide a reference for clinical treatment, but it needs to be selected according to the specific conditions of patients. Furthermore, although we adopted propensity score matching to control for known confounding factors, there were temporal sequence differences in patient groups (earlier in the open surgery group and later in the UBE assisted intervertebral fusion group), and there might be calendar time bias and unmeasured confounding factors. This might be one of the reasons for the significantly higher response rate in the UBE assisted intervertebral fusion group. Future research should adopt methods such as prospective design or instrumental variable analysis to further control such biases. Meanwhile, although the definition of the effective rate combined subjective symptoms and some functional indicators, the verified minimum clinically important difference (MCID) threshold (such as a VAS reduction of ≥ 2 points or an ODI improvement of ≥ 10 points) was not adopted as the judgment criterion. Subsequent studies will incorporate MCID into the efficacy evaluation system to enhance the comparability and clinical significance of the results. Intervertebral fusion is the core efficacy index, woperationhich should be judged by CT and X-ray film combined with Suk criteria at 6–12 months after surgery. In this study, the follow-up was only 3 months, which could not be accurately evaluated. In the future, the follow-up should be extended to more than 1 year, and the success rate of fusion should be evaluated by CT to verify the reliability of long-term bone fusion. The study content did not compare the time of discectomy and final version preparation. In the future, more comprehensive multicenter prospective studies may be needed to further explore the advantages and disadvantages of unilateral dual-tract technique in the treatment of various spinal diseases.
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Benzakour, T., Igoumenou, V., Mavrogenis, A. F. & Benzakour, A. Current concepts for lumbar disc herniation. Int Orthop. Apr. 43 (4), 841–851 (2019).
Rogerson, A., Aidlen, J. & Jenis, L. G. Persistent radiculopathy after surgical treatment for lumbar disc herniation: causes and treatment options. Int Orthop. Apr. 43 (4), 969–973 (2019).
Rickers, K. W., Pedersen, P. H., Tvedebrink, T. & Eiskjær, S. P. Comparison of interventions for lumbar disc herniation: a systematic review with network meta-analysis. Spine J. Oct. 21 (10), 1750–1762 (2021).
Jain, M. et al. Effect of approach based lumbar interbody fusion on sagittal spinopelvic parameters and functional outcomes: comparison between oblique lumbar interbody fusion (OLIF) and transforaminal lumbar interbody fusion (TLIF). Indian J. Orthop. Jan. 59 (1), 40–46 (2025).
Chu, P. L. et al. Global and current research trends of unilateral biportal Endoscopy/Biportal endoscopic spinal surgery in the treatment of lumbar degenerative diseases: A bibliometric and visualization study. Orthop Surg. Apr. 14 (4), 635–643 (2022).
Park, M. K. et al. Unilateral biportal endoscopy for decompression of extraforaminal stenosis at the lumbosacral junction: surgical techniques and clinical outcomes. Neurospine Dec. 18 (4), 871–879 (2021).
Zheng, B. et al. Efficacy and safety of unilateral biportal endoscopy versus other spine surgery: A systematic review and meta-analysis. Front. Surg. 9, 911914 (2022).
Cosamalón-Gan, I. et al. Inflammation in the intervertebral disc herniation. Neurocirugia (Astur: Engl. Ed) Jan-Feb. 32 (1), 21–35 (2021).
Roiha, M., Marjamaa, J., Siironen, J. & Koski-Palkén, A. Inferior long-term outcomes after surgery for lumbar disc herniation in patients with prior lumbar spine surgery. Acta Neurochir. (Wien) Jan. 24 (1), 32 (2024).
Beall, D. P. et al. Treatment gaps and emerging therapies in lumbar disc herniation. Pain Physician Sep. 27 (7), 401–413 (2024).
Aygun, H. & Abdulshafi, K. Unilateral biportal endoscopy versus tubular microendoscopy in management of single level degenerative lumbar Canal stenosis: A prospective study. Clin. Spine Surg. 07 (1), E323–e328 (2021).
Kim, S. K., Kang, S. S., Hong, Y. H., Park, S. W. & Lee, S. C. Clinical comparison of unilateral biportal endoscopic technique versus open microdiscectomy for single-level lumbar discectomy: a multicenter, retrospective analysis. J Orthop. Surg. Res. Jan. 31 (1), 22 (2018).
Chen, C., Liu, C., Wu, Z. & Su, J. How I do it? Uniportal full-endoscopic transforaminal lumbar interbody fusion with a complete reduction for L5 isthmic grade 2 spondylolisthesis. Acta Neurochir. (Wien) Dec. 165 (12), 3969–3974 (2023).
Fan, Z. et al. Unilateral biportal endoscopic lumbar interbody fusion (ULIF) versus endoscopic transforaminal lumbar interbody fusion (Endo-TLIF) in the treatment of lumbar spinal stenosis along with intervertebral disc herniation: a retrospective analysis. BMC Musculoskelet. Disord Feb. 29 (1), 186 (2024).
Wang, T. et al. Complete removal of intraspinal extradural mass with unilateral biportal endoscopy. Front. Surg. 9, 1033856 (2022).
Ahsan, M. K. et al. Lumbar revision microdiscectomy in patients with recurrent lumbar disc herniation: A single-center prospective series. Surg. Neurol. Int. 11, 404 (2020).
Patel K, Harikar MM, Venkataram T, et al. Is Minimally Invasive Spinal Surgery Superior to Endoscopic Spine Surgery in Postoperative Radiologic Outcomes of Lumbar Spine Degenerative Disease? A Systematic Review. J Neurol Surg A Cent Eur Neurosurg. Aug 2 2023.
Lee, C. K. & Kim, I. Commentary on unilateral biportal endoscopy for decompression of extraforaminal stenosis at the lumbosacral junction: surgical techniques and clinical outcomes. Neurospine Dec. 18 (4), 880–881 (2021).
Gatam, A. R. et al. Unilateral biportal endoscopic lumbar interbody fusion: A technical note and an outcome comparison with the conventional minimally invasive fusion. Orthop. Res. Rev. 13, 229–239 (2021).
Nikaido, T. & Konno, S. I. Usefulness of lateral lumbar interbody fusion combined with indirect decompression for degenerative lumbar spondylolisthesis: A systematic review. Medicina (Kaunas) Mar. 29(4), 492 (2022).
Tian, D. S., Zhu, B. & Jing, J. H. [Application expansion of unilateral biportal endoscopy in spine surgery and its related issues]. Zhonghua Yi Xue Za Zhi Nov. 8 (41), 3241–3245 (2022).
Xie, X., Zhang, G. & Liu, N. Clinical effect of unilateral biportal endoscopy in the treatment of lumbar diseases: a systematic review and meta-analysis. Wideochir Inne Tech. Maloinwazyjne Mar. 17 (1), 61–68 (2022).
Heo, D. H., Lee, D. C. & Park, C. K. Comparative analysis of three types of minimally invasive decompressive surgery for lumbar central stenosis: biportal endoscopy, uniportal endoscopy, and microsurgery. Neurosurg. Focus. 05 (1), E9 (2019).
Lin, G. X. et al. A systematic review of unilateral biportal endoscopic spinal surgery: preliminary clinical results and complications. World Neurosurg. May. 125, 425–432 (2019).
Yu, Q. et al. Unilateral biportal endoscopic transforaminal lumbar interbody fusion versus conventional interbody fusion for the treatment of degenerative lumbar spine disease: a systematic review and meta-analysis. BMC Musculoskelet. Disord Oct. 24 (1), 838 (2023).
Wang, X. et al. A single-arm retrospective study of the clinical efficacy of unilateral biportal endoscopic transforaminal lumbar interbody fusion for lumbar spinal stenosis. Front. Surg. 9, 1062451 (2022).
Park, M. K., Park, S. A., Son, S. K., Park, W. W. & Choi, S. H. Clinical and radiological outcomes of unilateral biportal endoscopic lumbar interbody fusion (ULIF) compared with conventional posterior lumbar interbody fusion (PLIF): 1-year follow-up. Neurosurg Rev. Sep. 42 (3), 753–761 (2019).
Kim, J. E. & Choi, D. J. Unilateral biportal endoscopic decompression by 30° endoscopy in lumbar spinal stenosis: technical note and preliminary report. J Orthop. Jun. 15 (2), 366–371 (2018).
Min, W. K., Kim, J. E., Choi, D. J., Park, E. J. & Heo, J. Clinical and radiological outcomes between biportal endoscopic decompression and microscopic decompression in lumbar spinal stenosis. J Orthop. Sci. May. 25 (3), 371–378 (2020).
Heo, D. H., Lee, N., Park, C. W., Kim, H. S. & Chung, H. J. Endoscopic unilateral laminotomy with bilateral discectomy using biportal endoscopic approach: technical report and preliminary clinical results. World Neurosurg. 137, 31–37 (2020).
Funding
This study was supported by Quzhou City Guiding Science and Technology Research Project (No. 2022033).
Author information
Authors and Affiliations
Contributions
Yunzhong Zhan: Conceptualization, Formal analysis, Writing- Reviewing and Editing. Fan Yang: Resources, Software. Zhou Ye and HuaFei Liu: Investigation, Methodology. YiChen Wu and WeiCheng Huang: Validation, Visualization. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval and consent to participate
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. This study was approved by the Ethics Committee of The Quzhou Affiliated Hospital of Wenzhou Medical University (Approve No. 2021-013). The study was conducted in compliance with pertinent standards and regulations, and participants gave their informed consent before beginning.
Consent to participate
Written informed consent to participate was obtained from all of the participants in the study.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Zhan, Y., Yang, F., Ye, Z. et al. Comparative analysis of the efficacy and functional recovery of unilateral biportal endoscopy-assisted lumbar interbody fusion for the treatment of lumbar disc herniation. Sci Rep 15, 41924 (2025). https://doi.org/10.1038/s41598-025-25838-z
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-25838-z