Abstract
Severe acute pancreatitis (SAP) is associated with high mortality, and pulmonary complications worsen prognosis. However, risk stratification tools for patients with SAP and pneumonia remain underdeveloped. We developed a predictive model for mortality in these patients. A predictive model for early mortality in SAP patients with pneumonia was developed and validated. The training cohort consisted of patients admitted to Xiangya Hospital between April 2017 and May 2021, while external validation used the Medical Information Mart for Intensive Care-IV database. The primary endpoint was 30-day mortality. Predictors were identified using least absolute shrinkage and selection operator regression and incorporated into a nomogram. A total of 220 patients were included, with 30-day mortality of 22.7%. Six predictors were identified: BUN, RDW, age, SBP, HCT, and WBC. Multivariable analysis confirmed BUN and age as independent risk factors and SBP as a protective factor. The nomogram demonstrated good discrimination in the training cohort and moderate discrimination in external validation. The model performed comparably to SOFA and APACHE II scores and outperformed disease-specific scores.We developed and validated a novel prediction model for 30-day mortality in SAP patients with pneumonia, with potential for aiding early risk stratification and clinical decision-making.
Similar content being viewed by others
Introduction
Acute pancreatitis (AP), an inflammatory disorder of the exocrine pancreas, is a common gastrointestinal disease with increasing incidence. Although typically self-limiting within 1 week, approximately 20% of patients progress to severe state. According to 2012 Revised Atlanta criteria, AP is classified as mild AP, moderately severe AP, or severe AP (SAP) based on organ failure extent. SAP involves persistent single or multiple organ failure, with 15–20% mortality rate1,2.
Respiratory complications are the most common organ-related complications in patients with SAP, and they occur in more than 50% of cases3. Acute respiratory failure is the primary cause of early mortality in patients with SAP4,5. Pulmonary manifestations include oedema, atelectasis, pneumonia, and critical conditions such as acute hypoxaemic respiratory failure and acute respiratory distress syndrome (ARDS)6. Pneumonia, a frequent early complication2,6,7, is detectable using chest computed tomography (CT), which is a key modality for pulmonary assessment8.
Prognostic models tailored specifically for patients with SAP and pneumonia remain underdeveloped, as previous studies have predominantly addressed general SAP cohorts9,10 or isolated complications (e.g., ARDS11 and sinistral portal hypertension12, with insufficient attention to the broader clinical entity of SAP complicated by pneumonia. Thus, we retrospectively reviewed the clinical records of patients with SAP and pulmonary disease using physiologic and biochemical variables collected within the 24 h of intensive care admission to develop a predictive model for mortality.
Method
Participants
We conducted a retrospective study of 1,608 consecutive patients with acute pancreatitis (AP) admitted to Xiangya Hospital, Central South University, between April 2017 and May 2021. Among them, 220 patients with severe acute pancreatitis (SAP) complicated by pneumonia, diagnosed according to the 2012 Revised Atlanta Criteria13, constituted the training cohort.
AP was diagnosed based on the presence of at least one of the following: (1) characteristic acute abdominal pain; (2) serum amylase and/or lipase levels ≥ 3-fold the upper limit of normal; (3) characteristic imaging findings on contrast-enhanced CT, magnetic resonance imaging, or ultrasonography. SAP was defined by persistent organ failure lasting > 48 h and a Modified Marshall Score (MMS) ≥ 2, ascertained at admission.
Pneumonia was diagnosed within 24 h of admission based on new radiographic infiltrates on chest X-ray or CT alongside clinical signs. Pneumonia was classified as:
-
Infectious Pneumonia: Required are characteristic symptoms with radiographic evidence, plus either confirmed microbiological aetiology or a substantial response to antimicrobial therapy.
-
Systemic inflammatory response syndrome (SIRS)-associated Pneumonia14: Required is (1) radiographic evidence of pneumonia, (2) meeting ≥ 2 SIRS criteria (Temperature > 38 °C or < 36 °C; Heart rate > 90 bpm; Respiratory rate > 20 breaths/min, PaCO₂ <32 mmHg, or mechanical ventilation; Leukocyte count > 12 × 10⁹/L or < 4 × 10⁹/L, or > 10% immature neutrophils), and (3) exclusion of other causes of infiltrates (e.g., heart failure, pulmonary embolism, and malignancy) through clinical, laboratory, and imaging assessment.
To ensure diagnostic consistency, all cases were independently reviewed by two intensivists, with disagreements resolved by a third senior clinician.
Exclusion criteria were: age < 18 years, indeterminate pneumonia classification, and pregnancy.
The external validation cohort comprised patients from the Medical Information Mart for Intensive Care (MIMIC)-IV database (v3.1)15,16, involving adults (≥ 18 years) with AP and pneumonia identified using ICD codes (Supplementary Table S1 online), yielding 190 eligible cases. Data access was authorised for author M.D. (ID: 9932040). The use of MIMIC-IV was approved by the institutional review boards of Beth Israel Deaconess Medical Centre and the Massachusetts Institute of Technology, which granted a waiver of informed consent due to the de-identified nature of the data.
Endpoint
The primary endpoint was 30-day all-cause mortality after admission. Survival status at 30 days was determined from medical records or via telephone follow-up for patients discharged earlier.
Clinical data collection
Covariates were selected a priori based on previous literature17,18,19 and subject and methodologic expertise. Baseline clinical data representing the first measurements obtained within 24 h of admission were analysed. In this retrospective study, demographic characteristics (age, sex, and aetiology), behavioural history (smoking and alcohol consumption), comorbidities (cardiovascular disease [CVD], cerebrovascular disease [CeVD], pulmonary disease, diabetes mellitus, and hypertension), vital signs (body temperature, systolic blood pressure [SBP], diastolic blood pressure [DBP], heart rate, and respiratory rate), arterial blood gas parameters (pH, partial pressure of arterial oxygen [PaO₂], PaCO₂, and lactate [Lac]), complete blood count indices (haematocrit [HCT], white blood cell count [WBC], platelet count [PLT], red blood cell distribution width [RDW], and haemoglobin concentration [HB]), serum biochemistry profiles (potassium [K+], sodium [Na+], calcium [Ca2+], albumin [ALB], total bilirubin [TBIL], alanine aminotransferase [ALT], aspartate aminotransferase [AST], lactate dehydrogenase [LDH], blood urea nitrogen [BUN], creatinine [CREA], and glucose [GLU]), coagulation metrics (prothrombin time [PT], activated partial thromboplastin time [APTT], and international normalised ratio [INR]), and composite clinical severity scores (oxygenation index [OI], Acute Physiology and Chronic Health Evaluation II [APACHE II], Sequential Organ Failure Assessment [SOFA], MMS, and Bedside Index of Severity in Acute Pancreatitis [BISAP]) were extracted.
Statistical analysis
Variables with > 15% missing data were excluded from the analysis. For datasets exhibiting 5–15% missing values, multiple imputations derived plausible values from the observed distribution patterns. Missing values constituting < 5% of the data were replaced with the arithmetic mean. Detailed information regarding missing data patterns and cohort characteristics before and after imputation are provided in Supplementary Tables S2 and S3 online.
Continuous variables with an approximately normal distribution are expressed as mean (standard deviation), whereas non-normally distributed data are expressed as median (interquartile range). Categorical variables are presented as counts (percentages). Inter-group comparisons were conducted using unpaired t-tests or Mann–Whitney U tests for continuous variables and chi-square or Fisher’s exact tests for categorical variables, with statistical significance defined at P < 0.05.
Variable selection was performed using the least absolute shrinkage and selection operator (LASSO) regression model with minimum log lambda, coupled with 10-fold cross-validation to reduce the risk of overfitting. Additional details are available in the Supplementary Materials. LASSO regression was used to identify candidate predictors, followed by univariate logistic regression screening for covariates with P < 0.05. Significant variables were subsequently incorporated into a multivariate logistic regression model using backward stepwise elimination. A nomogram was developed based on the final predictors.
The discriminative ability of the model was evaluated against established scoring systems (e.g., APACHE II) using receiver operating characteristic (ROC) curve analysis. Calibration curves and the Hosmer–Lemeshow test were used to quantify prediction accuracy, whereas decision curve analysis (DCA) was used to assess clinical utility. ROC and calibration curve analyses were also conducted to evaluate the performance heterogeneity of the model, with stratification by admission year/season and pneumonia classification. A sensitivity analysis was conducted by excluding patients who died within 48 h of admission and excluded participants with missing values to verify the robustness of the associations.
All statistical analyses were conducted using R (version 3.3.1).
Ethics committee approval and consent to participate
Ethical clearance for this study (Certification Code 201912477) was issued by the Xiangya Hospital Central South University Research Ethics Committee, confirming the adherence to the Helsinki Declaration. Written consent documentation was obtained from all participants or their legally authorised guardians. Legal guardians provided informed consent on behalf of patients in cases where the patients were unable to provide consent themselves due to the severity of their condition (e.g., unconsciousness, impaired decision-making capacity, or critical illness). The data collection and research resource derivation from the MIMIC database were reviewed by the Institutional Review Board of Beth Israel Deaconess Medical Centre that issued a retrospective waiver of informed consent and authorised the data sharing initiative.
Results
Baseline characteristics of patients with SAP and pneumonia
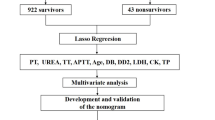
Figure 1 illustrates the enrolment process of 220 patients with SAP and pneumonia in the Xiangya training cohort. Table 1 presents the baseline characteristics of the survival (n = 170, 77.3%) and mortality (n = 50, 22.7%) groups after stratification by 30-day mortality rates. Patients in the mortality group were significantly older, more likely to be non-smokers and non-alcohol consumers, and experienced fewer days of hospitalisation and exhibited lower SBP and DBP, higher PaO2 and Lac but lower pH, higher RDW but lower HB, HCT, and RBC count, higher K+, Na+, BUN, and CREA levels but lower ALB levels, higher INR, and longer APTT. As presented in Table 2, patients in the mortality group exhibited significantly higher APACHE II and SOFA scores but lower Glasgow Coma Scale scores than those in the survival group. MMS and BISAP scores did not differ significantly between groups. There were significant differences in pneumonia type distribution and admission season. Sex distribution, incidence of hypertension and other comorbidities, and use of mechanical ventilation did not differ significantly between groups.
Flowchart of the study. SIRS: Systemic Inflammatory Response Syndrome; SAP: Severe Acute Pancreatitis; ROC: Receiver Operating Characteristic.
Significant predictors of outcomes of patients with SAP and pneumonia
Twenty-nine candidate predictors were initially selected using baseline analysis (Table 1, P < 0.05) and literature11,20. LASSO regression with 10-fold cross-validation (P < 0.10 threshold, L1se criterion) was used for screening. Supplementary Figure S1 and Table S4 online present this process. Six non-zero coefficient variables from the training cohort were assessed: BUN, RDW, age, SBP, HCT, and WBC. Univariate analysis (Table 3) demonstrated BUN and age as significant risk factors and SBP and HCT as protective factors against 30-day mortality in SAP patients with pneumonia. RDW exhibited borderline significance (CI approaching null value), and WBC showed no significant association with 30-day mortality. Consequently, multivariate analysis incorporated BUN, age, SBP, and HCT, with the final model incorporating BUN (β = 0.09, OR = 1.10, 95% CI: 1.06–1.14, P < 0.001), age (β = 0.03, OR = 1.03, 95% CI: 1.01–1.06, P = 0.022), SBP (β = − 0.02, OR = 0.98, 95% CI: 0.97–0.99, P = 0.037), and HCT (β = − 0.04, OR = 0.96, 95% CI: 0.92–1.01, P = 0.159) (Table 3).
Nomogram of outcomes of patients with SAP and pneumonia
A nomogram predicting 30-day mortality in SAP patients with pneumonia was developed using significant predictors from multivariate logistic regression. Each variable was assigned weighted points according to its contribution (Supplementary Fig. S2 online). The total score, derived by summing individual variable points, demonstrated direct linear association with mortality risk within 30 days after admission. To evaluate model adaptability, stratified analyses were conducted on the training cohort based on pneumonia aetiology, admission year, and admission season. The predictive model demonstrated superior performance in patients with infectious pneumonia [AUC(Area Under Curve) = 0.87], those admitted between April 2017 and December 2019 (AUC = 0.88), and those admitted during spring and summer (AUC = 0.90) (Supplementary Fig. S3 online).
Assessment and validation of the nomogram
A comparison of baseline characteristics between the Xiangya training and MIMIC-IV validation cohorts is summarised in Table 4. The Xiangya training cohort exhibited higher DBP, ALB, Na+, pH, and smoking prevalence, whereas the MIMIC-IV cohort had higher age, RDW, Lac, AST, Ca2+, proportion of women, CVD/CeVD/pulmonary disease prevalence, and mechanical ventilation rate. There were no significant differences in alcohol use, hospitalisation duration, 30-day mortality, other laboratory parameters, or comorbidities between groups.
Based on ROC and calibration curves of the training set, the nomogram demonstrated excellent discrimination (AUC = 0.83), with the validation set exhibiting moderate discrimination (AUC = 0.75) (Fig. 2). Our model’s AUC (0.83) was comparable to SOFA (0.83) and APACHE II (0.83) but indicated significantly higher discrimination than pancreatitis-specific scores (MMS: 0.57; BISAP: 0.56) in the training set (Supplementary Fig. S4A online). Our model’s AUC (0.75) indicated significantly higher discrimination than other scores [BISAP: 0.683; SOFA: 0.63; APS III (Acute Physiology and Chronic Health Evaluation III) : 0.663] in the validation set (Supplementary Fig. S4B online).
Receiver operating characteristic (ROC) curves of the nomogram model in the training cohort and validation cohort.
The model’s calibration was assessed in Supplementary Figure S5A-B and Table S5 online. In the external validation cohort, the Hosmer-Lemeshow test yielded a non-significant p-value of 0.5219. However, detailed visual and quantitative analysis of the calibration plot revealed a more nuanced picture. The model’s predicted probabilities ranged from 0.1 to 0.6. Within this range, systematic miscalibration was identified, where the model overestimated risk for patients at lower and upper ends of the predicted probability spectrum but underestimated risk for those in the middle range (approximately 30–50%).
This finding was quantitatively confirmed by a maximum calibration error (Emax) of 0.374, indicating substantial absolute deviation between predicted and observed outcomes at the worst-calibrated point. The Brier score, measuring overall predictive accuracy, was 0.146 (Supplementary Table S5 online).
Despite these calibration issues, decision curve analysis demonstrated that the model could provide positive net benefit across clinically relevant threshold probabilities (approximately 15% to 45%) when compared to “treat-all” or “treat-none” strategies (Supplementary Fig. S5C and D online).
Sensitivity analysis was performed by excluding patients who died within 48 h of admission and those with missing values to assess the robustness of identified associations. The odds ratios and AUC values demonstrated high consistency between original and sensitivity analyses, indicating stable model performance (Supplementary Table S6 online).
Discussion
This study focused on patients with SAP complicated by pneumonia—a high-risk subpopulation. Pulmonary complications are among the most frequent and severe manifestations of SAP, with incidence rates of approximately 75%21. This specificity is clinically relevant, since pneumonia in patients with SAP may arise from distinct mechanisms and require tailored management. Imaging findings of pneumonitis indicate severe disease and poor prognosis8. Pneumonia considerably affects SAP clinical outcomes22. The proposed pathophysiological mechanism involves localised pancreatic inflammation progressing to systemic inflammation and inducing SIRS. Circulating inflammatory cytokines traversing the pulmonary vasculature can induce alveolar damage. This is followed by a compensatory anti-inflammatory response syndrome, leading to immunosuppression and increased susceptibility to secondary infections23,24.
Various models have been developed to predict and manage organ dysfunction in SAP and AP, with APACHE-II score, Ranson’s criteria, CT severity index (CTSI), and BISAP widely applied for prognostic assessment in AP17. However, these scores have substantial limitations. Ranson’s criteria are temporally constrained, requiring 48 h for full assessment, while CTSI depends on CT imaging, limiting its utility in acute settings. Although numerous predictive models exist for SAP, only a few target mortality in patients with SAP complicated by pneumonia—a high-risk subgroup requiring early identification. This novel prediction model addresses these gaps by providing distinct clinical utility, including early, parsimonious, and specific risk stratification in emergency department triage or resource-limited settings where rapid, simple risk stratification is critical. Our model incorporates four routine parameters, enabling mortality risk assessment within hours of admission, compared with the more complex APACHE-II (18 items) and SOFA (6 items). Contextually, external MIMIC-IV cohort validation demonstrated that our model is simpler and more discriminative than established scores. Our model achieved an AUC of 0.75, outperforming APS III, BISAP, and SOFA scores. This evidence moves beyond simplicity, establishing that our disease-specific model more accurately predicts mortality in patients with SAP complicated by pneumonia than general organ dysfunction scores.
To construct a nomogram for predicting individual mortality risk in patients with SAP complicated by pneumonia, LASSO regression was used for variable selection, followed by logistic regression for model development. This enhanced the developed model’s clinical applicability and practical utility. Four predictors (age, SBP, HCT, and BUN) were incorporated to establish the nomogram. Logistic regression identified BUN and age as considerable independent risk factors, and SBP as an independent protective factor.
BUN level at admission is a valuable disease severity and outcome predictor in patients with SAP25,26. Consequently, an elevated level at admission is an integrative marker of hypovolaemia, renal hypoperfusion, and SIRS, portending multi-organ dysfunction risk27,28,29,30. This parameter’s established role in predicting pneumonia outcomes further supports its inclusion31,32. Although HCT was not significant in the final multivariable model (P = 0.159), it contributed unique predictive information, evidenced by a decrease in model discriminative ability when removed (Supplementary Fig. S6A online). It provides a reliable measure of haemoconcentration and oxygen delivery, with fewer confounders than other inflammatory markers. HCT’s prognostic value extends across both conditions: a recognised severity marker in AP33,34 and an independent predictor of long-term survival in pneumococcal pneumonia35. The inverse relationship between lower SBP and higher mortality identifies hypotension as a critical marker of physiological decompensation and distributive shock in patients with SAP complicated by pneumonia, rather than suggesting that hypertension is protective. This finding aligns with established literature, recognising low SBP as a harbinger of severe complications in AP and high-risk pneumonia20,36,37,38. The model’s predictive utility centres on low SBP, effectively identifying patients in circulatory failure. Advanced age is a non-modifiable risk factor associated with increased mortality in patients with AP/SAP39,40,41,42. This vulnerability stems from diminished physiological reserve and greater comorbidities43,44, impairing compensatory responses to the massive fluid shifts and inflammatory insults typical of SAP. Older patients experience more severe disease45 and are more susceptible to the spiral of organ failure.
Excluding respiratory variables (e.g., PaO₂) from the final model, despite their clinical relevance, suggests that systemic physiological derangement primarily drives mortality in patients with SAP complicated by pneumonia. The LASSO algorithm excluded PaO₂ due to redundant predictive value within the multivariate context; its forced inclusion reduced the model’s discriminative ability (Supplementary Fig. S6B online). This aligns with severity tools, including CURB-65, which also omit PaO₂, suggesting respiratory-specific variables are not essential for predicting outcome in complex, systemically ill cohorts. Future studies should validate this interplay between systemic and respiratory factors.
Most SAP predictive models focus on overall mortality or complications. Xu et al. showed that AdaBoost predicts multi-organ failure best (AUC = 0.826)46, while another study developed nomograms for predicting ARDS with AUCs > 0.7511. Gutierrez et al. (n = 516)47 and Han et al. (n = 850)48 developed models based on general SAP cohorts, compared with our study’s focus on subpopulation mortality. To our knowledge, this is the first nomogram developed to predict mortality in patients with SAP complicated by pneumonia. The model demonstrated excellent performance in the training cohort and acceptable discrimination in the external MIMIC-IV validation cohort, indicating preserved baseline risk stratification ability in out-of-distribution data. The observed discriminatory performance attenuation likely reflects inherent differences in data distribution and clinical protocols between the MIMIC database and our single-centre cohort.
Moreover, the AUC reduction during external validation was expected, providing insights into the model’s generalisability. The MIMIC-IV cohort represents a distinct, more severe clinical phenotype, characterised by older age, higher comorbidity burden, and greater use of mechanical ventilation. This ‘case-mix shift’ plausibly explains the anticipated decline in model discrimination. The features derived from a younger, hypertensive training cohort demonstrated limited generalisability when applied to an older, hypotensive population. Mortality was driven by acute inflammatory insult from SAP in the derivation cohort, but stemmed from a complex interplay between acute SAP-related injury, underlying chronic organ dysfunction, and prolonged intensive care unit (ICU) stay-related complications in the MIMIC cohort. Since our model is built on acute physiological markers, it is inherently less sensitive to chronic and iatrogenic risk factors, explaining the predicted AUC decline. Renal function (BUN) and HCT distributions were similar between cohorts, with slightly lower average BUN in the validation set, suggesting that ‘greater severity’ is inconsistent across all clinical dimensions. Supplementary Figure S7 online shows the subgroup analysis results for age and SBP. Therefore, the AUC decline likely reflects the model’s limited generalisability to the combination of advanced age and hypotension, rather than non-specific elevations in illness severity. This insight can guide future model optimisation strategies.
External validation also revealed critical calibration limitations. Although the Hosmer–Lemeshow test was not significant (P = 0.5219), the validation set’s calibration plotindicates serious miscalibration with high clinical risk. This pattern indicates that the model systematically underestimates risk for medium-probability patients—leading to undertreatment—and overestimates risk for others. Notably, this finding was quantitatively supported by a high maximum calibration error (Emax = 0.374), indicating non-trivial local deviations between predicted and observed outcomes. This miscalibration pattern carries substantial clinical risk: underestimation and overestimation may lead to undertreatment and prompt unnecessary resource allocation, respectively. The most probable explanation is the substantial case-mix differences between cohorts. Our model was developed on a population with a specific risk profile; however, its inherent risk estimates become inaccurate when applied to the older, more comorbid, and mechanically ventilated MIMIC-IV cohort. Although discriminative ability was preserved (AUC = 0.75), indicating retained utility for risk stratification, the model’s absolute risk estimates proved unreliable. Therefore, the model, in its current form, is unsuitable for direct clinical application in MIMIC-like populations. This external validation’s value lies in clarifying the extent and nature of performance degradation in divergent clinical settings rather than in demonstrating perfect performance. Recalibration and domain adaptation are essential next steps. Our findings underscore that applying this model to new populations requires mandatory recalibration to correct systematic biases and ensure accurate absolute risk estimation. Local recalibration is also recommended before implementation, with further generalisability optimisation planned through prospective multicentre validation.
DCA demonstrated the model’s clinical utility, showing a positive net benefit within the 15–45% risk threshold range when compared with default strategies. This supports its application in guiding clinical decisions—including ICU admission or intensified monitoring—for patients with a predicted mortality risk at or above the recommended 30% threshold. Although net benefit decreased in the validation cohort, the model consistently outperformed the ‘treat-all’ approach across this clinically relevant range.
Our analysis revealed that the model’s predictive performance was better for infectious pneumonia than for SIRS-associated pneumonia, indicating considerable subtype-dependent variation. Temporal subgroup analysis showed maintained performance in pre-pandemic patients with moderate decline during the pandemic period. Seasonal analysis further suggested slightly better performance in spring-summer than in autumn-winter. These findings underscore how aetiological and temporal factors influence prognostic accuracy, warranting further investigation into pneumonia classification interactions.
The endpoint of 30-day all-cause mortality was selected due to the high short-term mortality risk characteristic of SAP complicated by pneumonia. This timeframe captures the majority of deaths directly attributable to the disease process49,50, while minimising the potential influence of unrelated long-term events. The use of all-cause mortality helps mitigate adjudication bias, a considerable concern in retrospective studies where precise cause-of-death determination can be subjective and inconsistent. We acknowledge that this approach may include a few deaths not directly caused by SAP or pneumonia, constituting a study limitation. However, given the studied conditions’ acuity and severity, most deaths within this period are likely related to the index illness or its complications. Future studies should consider applying competing risk models to better distinguish between SAP-pneumonia-related deaths and unrelated causes, thereby refining prognostic accuracy.
Our study revealed some counterintuitive findings. Non-survivors had a lower prevalence of smoking and alcohol use history, alongside higher PaO₂ levels at admission. While these findings require cautious interpretation and further validation, we postulate several possibilities. Regarding smoking and alcohol use history, we posit that a simple binary assessment (yes/no) may fail to capture deeper contextual information, leading to seemingly paradoxical outcomes. Future studies should incorporate more detailed investigations, including duration, frequency, and quantity of tobacco and alcohol use, to draw more reliable conclusions. Regarding higher PaO₂ levels observed in non-survivors, survivor bias may represent a plausible explanation. The higher PaO₂ levels in non-survivors could be a marker of more aggressive early respiratory support rather than better intrinsic lung function. Furthermore, single-time-point measurement within 24 h may not capture the dynamic progression of respiratory failure. These hypotheses highlight the complexity of SAP complicated by pneumonia and underscore the need for dynamic monitoring in future studies.
This study has some other constraints beyond the abovementioned limitations. First, incorporating pneumonia into established SAP modelling frameworks represents only an incremental improvement. Second, the modest sample size (n = 220) was only partially offset by external validation. Third, potential treatment-related confounders exist in model-incorporated predictors. Fourth, although data were stratified by admission year, the coronavirus disease 2019 pandemic disruptions were not explicitly analysed. Fifth, seasonal patterns were only partially captured and pathogen-specific variations were unexamined. Sixth, electronic health record-derived data are susceptible to coding inaccuracies, documentation gaps, and systematic measurement bias. Finally, non-routine biomarkers (e.g., interleukin-6) and variables with > 15% missingness (e.g., amylase/lipase) were omitted, raising the risk of unmeasured confounding.
Conclusions
We have developed and validated a novel, parsimonious prediction model for 30-day mortality specifically for SAP patients with pneumonia. It possesses potential for aiding early triage and decision-making in acute care settings. We propose a risk threshold of 30% to identify high-risk patients who may benefit from escalated care. Nevertheless, external prospective validation and further assessment of its impact on clinical outcomes are necessary before it can be recommended for routine clinical use.
Data availability
Owing to ethical restrictions protecting patient confidentiality, the research datasets analysed in this study are not openly accessible. However, qualified researchers may request access to de-identified data by contacting the corresponding author, subject to approval from the Institutional Review Board at the Central South University’s Xiangya Hospital. The MIMIC-IV critical care database is publicly accessible via PhysioNet (https://physionet.org/content/mimiciv/3.1/).
Abbreviations
- ALB:
-
Albumin
- APACHE II:
-
Acute Physiology and Chronic Health Evaluation II
- APTT:
-
Activated Partial Thromboplastin Time
- AST:
-
Aspartate Amino Transferase
- BISAP:
-
Bedside Index for Severity in Acute Pancreatitis
- BUN:
-
Blood Urea Nitrogen
- Ca2+:
-
Calcium Concentration
- CVD:
-
Cardiovascular Diseases
- CI:
-
Confidence Interval
- CREA:
-
Creatinine
- CT:
-
Computed Tomography
- CTSI:
-
Computed Tomography Severity Index
- CeVD:
-
Cerebrovascular Disease
- DBP:
-
Diastolic Blood Pressure
- DM:
-
Diabetes Mellitus
- HB:
-
Hemoglobin
- HCT:
-
Hematocrit
- HR:
-
Heart Rate
- GCS:
-
Glasgow Coma Scale
- GLU:
-
Glucose
- HCT:
-
Haematocrit
- HGB:
-
Haemoglobin
- ICU:
-
Intensive Care Unit
- INR:
-
International Normalised Ratio
- Jan.:
-
January
- K+:
-
Potassium Concentration
- Lac:
-
Lactic Acid
- LDH:
-
Lactate dehydrogenase
- MMS:
-
Modified Marshall Score
- Na+:
-
Sodium Concentration
- OI:
-
Oxygen Index
- OR:
-
Odds Ratio
- PaCO2:
-
Artery Carbon Dioxide Partial Pressure
- PaO2:
-
Arterial Oxygen Partial Pressure
- PDW:
-
Platelet Distribution Width
- PLT:
-
Platelets
- PT:
-
Plasma Prothrombin Time
- RBC:
-
Red Blood Cells
- RDW:
-
Red Blood Cell Distribution Width
- RR:
-
Respiratory Rate
- SBP:
-
Systolic Blood Pressure
- SIRS:
-
Systemic Inflammatory Response Syndrome
- SOFA:
-
Sequential Organ Failure Assessment
- TBIL:
-
Total Bilirubin
- Temp:
-
Temperature
- WBC:
-
White Blood Cell
References
Xiao, A. Y. et al. Global incidence and mortality of pancreatic diseases: a systematic review, meta-analysis, and meta-regression of population-based cohort studies. Lancet Gastroenterol. Hepatol. 1, 45–55 (2016).
Poropat, G. et al. Prevention of infectious complications in acute pancreatitis: results of a single-center, randomized, controlled trial. Pancreas 48, 1056–1060 (2019).
Pintado, M. C. et al. New Atlanta classification of acute pancreatitis in intensive care unit: complications and prognosis. Eur. J. Intern. Med. 30, 82–87 (2016).
Akbarshahi, H., Rosendahl, A. H., Westergren-Thorsson, G. & Andersson, R. Acute lung injury in acute pancreatitis–awaiting the big leap. Respir Med. 106, 1199–1210 (2012).
Dombernowsky, T., Kristensen, M. Ø., Rysgaard, S., Gluud, L. L. & Novovic, S. Risk factors for and impact of respiratory failure on mortality in the early phase of acute pancreatitis. Pancreatology 16, 756–760 (2016).
Iyer, H., Elhence, A., Mittal, S., Madan, K. & Garg, P. K. Pulmonary complications of acute pancreatitis. Expert Rev. Respir Med. 14, 209–217 (2020).
Kumar, P., Gupta, P. & Rana, S. Thoracic complications of pancreatitis. JGH Open. 3, 71–79 (2019).
Peng, R. et al. Chest computed tomography semi-quantitative pleural effusion and pulmonary consolidation are early predictors of acute pancreatitis severity. Quant. Imaging Med. Surg. 10, 451–463 (2020).
Xu, C., Wang, J., Jin, X., Yuan, Y. & Lu, G. Establishment of a predictive model for outcomes in patients with severe acute pancreatitis by nucleated red blood cells combined with Charlson complication index and APACHE II score. Turk. J. Gastroenterol. 31, 936–941 (2020).
Chen, Y., Li, Q., Ma, L., Cai, Z. & Zhou, J. Development and internal validation of a practical model to predict 30 days mortality of severe acute pancreatitis patients. Ann. Med. 55, 2236648. https://doi.org/10.1080/07853890.2023.2236648 (2023).
Li, Y. L. et al. Development and external validation of models to predict acute respiratory distress syndrome related to severe acute pancreatitis. World J. Gastroenterol. 28, 2123–2136 (2022).
Xiao, M., An, Y., Di, Y. & Cui, Y. Analysis of risk factors and construction of a predictive model for severe acute pancreatitis complicated by sinistral portal hypertension. Front. Physiol. 16, 1512144. https://doi.org/10.3389/fphys.2025.1512144 (2025).
Banks, P. A. et al. Classification of acute pancreatitis–2012: revision of the Atlanta classification and definitions by international consensus. Gut 62, 102–111 (2013).
Parnell, G. P. et al. A distinct influenza infection signature in the blood transcriptome of patients with severe community-acquired pneumonia. Crit. Care. 16, R157. https://doi.org/10.1186/cc11477 (2012).
Johnson, A. et al. MIMIC-IV (version 3.1). PhysioNet RRID. https://doi.org/10.13026/kpb9-mt58 (2024). SCR_007345.
Johnson, A. E. W. et al. MIMIC-IV, a freely accessible electronic health record dataset. Sci. Data. 10, 1. https://doi.org/10.1038/s41597-022-01899-x (2023).
Di, M. Y. et al. Prediction models of mortality in acute pancreatitis in adults: a systematic review. Ann. Intern. Med. 165, 482–490 (2016).
Ueda, T. et al. Simple scoring system for the prediction of the prognosis of severe acute pancreatitis. Surgery 141, 51–58 (2007).
Harrison, D. A., D’Amico, G. & Singer, M. The pancreatitis outcome prediction (POP) score: a new prognostic index for patients with severe acute pancreatitis. Crit. Care Med. 35, 1703–1708 (2007).
Zhu, X. G. et al. Development and validation of a nomogram for predicting survival in patients with acute pancreatitis. World J. Emerg. Med. 14, 44–48 (2023).
Browne, G. W. & Pitchumoni, C. S. Pathophysiology of pulmonary complications of acute pancreatitis. World J. Gastroenterol. 12, 7087–7096 (2006).
Kim, Y. J. et al. Analysis of factors influencing survival in patients with severe acute pancreatitis. Scand. J. Gastroenterol. 52, 904–908 (2017).
Mentula, P. et al. Plasma anti-inflammatory cytokines and monocyte human leucocyte antigen-DR expression in patients with acute pancreatitis. Scand. J. Gastroenterol. 39, 178–187 (2004).
Beger, H. G., Bittner, R., Block, S. & Büchler, M. Bacterial contamination of pancreatic necrosis. A prospective clinical study. Gastroenterology 91, 433–438 (1986).
Dai, M., Fan, Y., Pan, P. & Tan, Y. Blood Urea nitrogen as a prognostic marker in severe acute pancreatitis. Dis. Markers. 2022 (7785497). https://doi.org/10.1155/2022/7785497 (2022).
Lin, S. et al. Blood Urea nitrogen as a predictor of severe acute pancreatitis based on the revised Atlanta criteria: timing of measurement and cutoff points. Can. J. Gastroenterol. Hepatol. 2017 (9592831). https://doi.org/10.1155/2017/9592831 (2017).
Whitcomb, D. C. et al. Angiopoietin-2, a regulator of vascular permeability in inflammation, is associated with persistent organ failure in patients with acute pancreatitis from the united States and Germany. Am. J. Gastroenterol. 105, 2287–2292 (2010).
Ghim, M. et al. Visualization of three pathways for macromolecule transport across cultured endothelium and their modification by flow. Am. J. Physiol. Heart Circ. Physiol. 313, H959–H973 (2017).
Biyik, Z. et al. The role of the BUN/albumin ratio in predicting poor clinical outcomes in patients with acute pancreatitis. Niger J. Clin. Pract. 28, 360–366 (2025).
Wan, Y. et al. J-shaped association between blood Urea nitrogen-to-creatinine ratio and mortality in critically ill patients with acute pancreatitis: a retrospective cohort study using the MIMIC-IV database. Eur. J. Med. Res. 30, 185. https://doi.org/10.1186/s40001-025-02430-9 (2025).
Cheng, A. et al. Diagnostic performance of initial blood Urea nitrogen combined with D-dimer levels for predicting in-hospital mortality in COVID-19 patients. Int. J. Antimicrob. Agents. 56, 106110. https://doi.org/10.1016/j.ijantimicag.2020.106110 (2020).
Ryu, S. et al. Utility of the blood Urea nitrogen to serum albumin ratio as a prognostic factor of mortality in aspiration pneumonia patients. Am. J. Emerg. Med. 43, 175–179 (2021).
Acehan, F. et al. Revisiting the Ranson score in acute pancreatitis: is the drop in hematocrit a worrisome sign? J. Hepatobiliary Pancreat. Sci. 30, 315–324 (2023).
Zou, L. J., Ruan, H. & Li, Y. S. Nonlinear association between hematocrit levels and short-term all-cause mortality in ICU patients with acute pancreatitis: insights from a retrospective cohort study. BMC Gastroenterol. 25, 186. https://doi.org/10.1186/s12876-025-03764-8 (2025).
Ruiz, L. A. et al. Factors influencing long-term survival after hospitalization with Pneumococcal pneumonia. J. Infect. 79, 542–549 (2019).
Thandassery, R. B. et al. Hypotension in the first week of acute pancreatitis and APACHE II score predict development of infected pancreatic necrosis. Dig. Dis. Sci. 60, 537–542 (2015).
Chalmers, J. D., Singanayagam, A. & Hill, A. T. Systolic blood pressure is superior to other haemodynamic predictors of outcome in community-acquired pneumonia. Thorax 63, 698–702 (2008).
Fine, M. J. et al. Prognosis and outcomes of patients with community-acquired pneumonia. A meta-analysis. JAMA 275, 134–141 (1996).
He, F. et al. Factors predicting the severity of acute pancreatitis in elderly patients. Aging Clin. Exp. Res. 33, 183–192 (2021).
Husu, H. L., Leppäniemi, A. K., Lehtonen, T. M., Puolakkainen, P. A. & Mentula, P. J. Short- and long-term survival after severe acute pancreatitis: a retrospective 17 years’ cohort study from a single center. J. Crit. Care. 53, 81–86 (2019).
Yasuda, H. et al. Etiology and mortality in severe acute pancreatitis: a multicenter study in Japan. Pancreatology 20, 307–317 (2020).
de Beaux, A. C., Palmer, K. R. & Carter, D. C. Factors influencing morbidity and mortality in acute pancreatitis; an analysis of 279 cases. Gut 37, 121–126 (1995).
Baeza-Zapata, A. A., García-Compeán, D. & Jaquez-Quintana, J. O. Collaborators. Acute pancreatitis in elderly patients. Gastroenterology 161, 1736–1740 (2021).
Wang, Q. et al. The clinical characteristics and outcome of elderly patients with acute pancreatitis. Pancreas 51, 1284–1291 (2022).
Tenner, S. et al. Relationship of necrosis to organ failure in severe acute pancreatitis. Gastroenterology 113, 899–903 (1997).
Xu, F. et al. Prediction of multiple organ failure complicated by moderately severe or severe acute pancreatitis based on machine learning: a multicenter cohort study. Mediators Inflamm. 2021 (5525118). https://doi.org/10.1155/2021/5525118 (2021).
Barrera Gutierrez, J. C. et al. Severe acute pancreatitis prediction: a model derived from a prospective registry cohort. Cureus 15, e46809. https://doi.org/10.7759/cureus.46809 (2023).
Han, D. et al. A novel nomogram for predicting survival in patients with severe acute pancreatitis: an analysis based on the large MIMIC-III clinical database. Emerg. Med. Int. 2021 (9190908). https://doi.org/10.1155/2021/9190908 (2021).
Schepers, N. J. et al. Impact of characteristics of organ failure and infected necrosis on mortality in necrotising pancreatitis. Gut 68, 1044–1051 (2019).
Postma, D. F. et al. Antibiotic treatment strategies for community-acquired pneumonia in adults. N Engl. J. Med. 372, 1312–1323 (2015).
Acknowledgements
We would like to thank Editage (www.editage.cn) for English language editing.
Funding
This study was supported in part by the Natural Science Foundation of Changsha (No. kq2403004), the Key R&D Program of Hunan Province (No. 2022SK2038), the Natural Science Foundation of Hunan Province of China (No. 2023JJ30930), the Project Program of Central South University Graduate Education Teaching Reform (No. 2022JGB025), the Research Project on Teaching Reform of Ordinary Colleges and Universities in Hunan Province (No. 2022JGYB037), the Scientific Research Program of the FuRong Laboratory (No. 2023SK2101), and the Scientific Research Project Topics of the Hunan Provincial Health Commission (No. B202302068083).
Author information
Authors and Affiliations
Contributions
Conception and design: M.D. and P.P.; Acquisition of data: Y.F. and M.D.; Analysis: Y.T. and M.D.; Drafting the manuscript for important intellectual content: M.D.; Revised the manuscript: Y.T. and P.P.; Final approval of the version submitted for publication: P.P and B.Y. All authors reviewed the manuscript. The authors declare no conflicts of interest regarding the publication of this article.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics committee approval and consent to participate
Ethical clearance for this study (Certification Code 201912477) was issued by the Xiangya Hospital Central South University Research Ethics Committee, confirming the adherence to the Helsinki Declaration. Written consent documentation was obtained from all participants or their legally authorised guardians. Legal guardians provided informed consent on behalf of patients in cases where the patients were unable to provide consent themselves due to the severity of their condition (e.g., unconsciousness, impaired decision-making capacity, or critical illness). The data collection and research resource derivation from the MIMIC database were reviewed by the Institutional Review Board of Beth Israel Deaconess Medical Centre that issued a retrospective waiver of informed consent and authorised the data sharing initiative.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Dai, M., Tan, Y., Fan, Y. et al. Development and validation of a nomogram for predicting 30-day mortality in patients with severe acute pancreatitis and pneumonia. Sci Rep 15, 42135 (2025). https://doi.org/10.1038/s41598-025-26223-6
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-26223-6