Abstract
To investigate the predictive values of plasma levels of cystatin C (CysC), cytokines, and neutrophil gelatinase-associated lipocalin protein (NGAL) for acute kidney injury (AKI) development following cardiopulmonary bypass (CPB) surgery. Clinical data were collected from cardiopulmonary bypass patients at Suzhou Municipal Hospital between May 2022 and April 2024. Plasma NGAL and CysC levels were measured at 12 h and 36 h postoperatively, while Cytokines were measured at 12 h postoperatively. AKI was assessed using Kidney Disease: Improving Global Outcomes (KDIGO) criteria. Receiver operating characteristic (ROC) curves were used to evaluate the predictive value of NGAL and CysC for AKI, and binary logistic regression was used to identify risk factors for postoperative AKI. Among 95 enrolled patients, 42 (44.21%) developed AKI. The area under the ROC curve (AUC) for plasma NGAL was 0.65 (95% CI 0.54–0.77) at 12 h and 0.76 (95% CI 0.67–0.86) at 36 h, with corresponding sensitivities and specificities of 58%/73% and 51%/94%, respectively. The AUCs for plasma CysC at 12 h and 36 h were both 0.77 (95% CI 0.67–0.86 and 0.67–0.87, respectively), with sensitivities/specificities of 56%/88% and 68%/80%. Logistic regression analysis identified CysC-12 h, CysC-36 h, NGAL-12 h, NGAL-36 h, cTnI-36 h, age, and CPB duration as risk factors for postoperative AKI, with hazard ratios (HRs) for CysC-12 h and CysC-36 h of 8.2 (95% CI 2.74–24.55) and 9.51 (95% CI 2.97–30.50). Elevated plasma NGAL and CysC levels are significantly associated with the development of AKI after CPB surgery. These biomarkers may facilitate early identification of patients at high risk for postoperative AKI.
Similar content being viewed by others
Introduction
The incidence of acute kidney injury (AKI) in hospitalized patients exceeds 10%, with over two-thirds requiring further treatment in the intensive care unit (ICU), and highly impacts patient prognosis1. Although renal function gradually recovers within 3–7 days in two-thirds of AKI patients, up to one-third experience unresolved AKI2. Persistent AKI causes severe complications, substantial medical costs, prolonged hospital stays, and increased risks of mortality and chronic renal failure3. Identifying strategies for effective and timely prediction of AKI occurrence is expected to facilitate intervention measures. Although AKI can be diagnosed using plasma creatinine level and urine output criteria, these diagnostic markers do not hold any predictive value.
Cardiac surgery-associated acute kidney injury (CSA-AKI) represents a prevalent perioperative complication that substantially influences patient outcomes by elevating the risk of infections, extending the duration of mechanical ventilation, and contributing to overall morbidity4,5. Furthermore, individuals who experience CSA-AKI are predisposed to an increased risk of major adverse kidney events6, with an estimated 2–5% necessitating renal replacement therapy7,8. Consequently, the early identification of CSA-AKI risk and the deployment of preventive strategies are essential for enhancing patient prognosis.
As a member of the lipocalin family, neutrophil gelatinase-associated lipocalin (NGAL) is widely distributed in various tissues, such as the bronchus, gastrointestinal tract, and renal tubules. NGAL can induce renal progenitor cells to differentiate into renal tubular epithelial cells. Once renal tubules are damaged, the serum level of NGAL rises for repair and regeneration9. It has been proven that NGAL and cystatin C (Cys-C) can predict AKI earlier10. Therefore, this study aimed to explore the predictive values of clinical markers for AKI development following cardiopulmonary bypass surgery.
Materials and methods
Patient selection
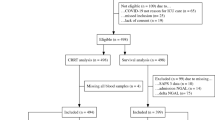
This study was approved by the Ethics Committee of Suzhou Municipal Hospital (IRB numbers KL901511), and informed consent was obtained from all the included patients. All experiments were performed in accordance with relevant guidelines and regulations. Patients who underwent cardiac surgery with cardiopulmonary bypass at Suzhou Municipal Hospital between 1 May 2022 and 30 April 2024 were included. The inclusion criteria were as follows: (1) age ≥ 14 years (any gender); (2) elective cardiac surgery with cardiopulmonary bypass; and (3) normal baseline renal function. The exclusion criteria were (1) history of renal insufficiency or kidney transplantation; (2) concurrent infection; and (3) concurrent malignant tumors or autoimmune diseases. AKI was diagnosed based on the KDIGO definition, and patients were considered to have AKI if they met any of the following criteria: (a) an increase in serum creatinine by ≥ 26.5 μmol/L (≥ 0.3 mg/dL) within 48 h; (b) an increase in serum creatinine to ≥ 1.5 times the baseline value within 7 days.
Data collection
General information
Patient data on age, gender, weight, height, and history of underlying diseases were collected.
Intraoperative details
The duration of cardiopulmonary bypass and aortic cross-clamp time were recorded.
Postoperative data
Postoperative blood test results, lengths of hospital stay and ICU stay, blood purification, and survival rate on postoperative day 30 (D 30) were documented.
Sample collection
Preoperative
Blood samples were collected from patients for baseline testing.
Postoperative
Urine output was measured at 12 h, D 2 and D 3 post-operation. Venous blood samples were collected at 12 h and 36 h post-operation for plasma NGAL and CysC testing. Blood samples collected 12 h post-operation were utilized to measure plasma cytokine levels. All the data points and measurements were sourced from medical records.
Statistical analysis
The SPSS 20.0 software was used for statistical analysis. The distribution of measurement data was determined using the Kolmogorov–Smirnov test. Normally distributed data were expressed as mean ± SD and compared between the two groups by using the t-test. Skewed data were expressed as M (Q1, Q3) and compared between the two groups by using the Mann–Whitney test. Categorical data were expressed as n (%) and compared between the two groups by using the χ2 test. Multivariate logistic regression analysis was utilized to identify factors influencing AKI. The receiver operating characteristic (ROC) curve was plotted to evaluate the predictive efficacies of plasma levels of CysC, NGAL, and tumor necrosis factor (TNF) for AKI. The risk factors for AKI were analysed using binary logistic regression. A p-value < 0.05 was considered to indicate statistical significance.
Results
Comparison of clinical data between the AKI and non-AKI groups
This study included 95 patients who underwent cardiopulmonary bypass surgery, of which 57 were male. There were 42 patients in the AKI group and 53 in the non-AKI group (incidence rate, 44.21%). The demographic characteristics of the two groups are shown in Table 1. In the analysis of clinical characteristics, patients in the AKI group were generally older than those in the non-AKI group (p < 0.05). The duration of CPB was also significantly longer in the AKI group compared with the non-AKI group (p < 0.05). In addition, a higher proportion of patients in the AKI group had coexisting coronary artery disease (CAD). Moreover, combined surgical procedures were more frequently performed in the AKI group than in the non-AKI group.
Subsequently, we further analyzed postoperative clinical parameters, as summarized in Table 2. The differences in plasma NGAL and CysC levels between the two groups at 12 h and 36 h post-operatively, respectively, were statistically significant (p < 0.05). The urine output at 12 h post-operation was significantly lower in the AKI group than in the non-AKI group, and the difference was statistically significant (p < 0.05). The myocardial injury markers in the AKI group, including myoglobin (at 12 h and 36 h) and troponin I (at 36 h), were significantly higher than those in the non-AKI group, and the differences were statistically significant (p < 0.05). The AKI group had a longer ICU stay and a higher risk for renal replacement therapy than the non-AKI group. Although plasma levels of the cytokines IL-6, IL-8, and IL-10 were increased in both groups, the increases were not significantly different between the two groups (p > 0.05).
Predictive values of plasma NGAL and CysC levels for postoperative AKI
The AUCs of plasma CysC levels at 12 h and 36 h postoperatively for predicting AKI were 0.77 (95% CI, 0.67–0.86; 56% sensitivity, 88% specificity) and 0.77 (95% CI, 0.67–0.87; 68% sensitivity, 80% specificity), respectively. Plasma NGAL level at 12 h had an AUC of 0.65 (95% CI, 0.54–0.77), with 58% sensitivity and 73% specificity. NGAL level at 36 h had an AUC of 0.76 (95% CI, 0.67–0.86), with 51% sensitivity and 94% specificity (Fig. 1 and Table 3). In addition, a statistical power analysis was performed for other variables that showed significant differences in Table 2, as summarized in Supplementary Table 1. The AUC for plasma TNF level in predicting postoperative AKI was 0.70 (95% CI, 0.54–0.86) (Supple Table 1).
Receiver operating characteristic curve analyses of plasma levels of CysC and NGAL at 12 h and 36 h postoperatively for predicting acute kidney injury following cardiopulmonary bypass surgery.
To examine factors related to postoperative AKI, we performed univariable regression analyses on variables that showed significant intergroup differences (Table 4). Higher cystatin C levels at 12 h and 36 h postoperatively were linked to the highest hazard ratios (HR = 8.20 and 9.51, respectively), followed by pre-existing hypertension and coronary atherosclerotic heart disease (HR = 4.16 and 3.23, respectively). In addition, age, plasma NGAL levels at 12 h and 36 h, cardiopulmonary bypass duration, and urine output at 12 h after surgery showed notable associations with postoperative AKI.
Discussion
The diagnosis of AKI is primarily based on increased plasma creatinine levels and decreased urine output, both of which have certain limitations. Firstly, increases in creatinine levels can take 24–48 h to become apparent11. Secondly, due to the strong compensatory capacity of the kidneys, plasma creatinine levels do not significantly change until there is ≥ 50% nephron damage12. Furthermore, plasma creatinine level is not a specific marker of AKI as it can be influenced by various confounding factors, such as poor nutritional status and fluid overload13. Similarly, urine output can be reduced by factors such as prolonged fasting, hypovolemia, and pain.
Postoperative AKI is a challenging complication that may occur following cardiopulmonary bypass surgery and impairs patient prognosis. In recent years, many studies have attempted to identify early predictive indicators of AKI, whereby several independent risk factors have been identified. However, the etiology of AKI is generally multifactorial. In our study, advanced age, hypertension, coronary artery disease, and the complexity of surgery (valve replacement combined with coronary artery bypass grafting) were identified as high-risk factors for postoperative AKI. The levels of myocardial injury markers in the postoperative AKI group were significantly higher than those in the non-AKI group, a finding consistent with the results reported by Wen Wei14. In a cohort of 57 patients undergoing cardiopulmonary bypass surgery, Saydam found that ICU stay and hospitalization time were significantly longer in the AKI group (p < 0.05) than in the non-AKI group15. In our study, the incidence of AKI was 44.21%, similar to previously reported rates15,16. Intraoperative factors, such as the duration of cardiopulmonary bypass, were significantly associated with AKI occurrence, consistent with the findings of Lakhal K et al17.
Human NGAL is a 25-kDa glycoprotein belonging to the lipocalin superfamily. It occurs in three molecular forms: a 25-kDa monomer, a 45-kDa homodimer, and a 135-kDa heterodimer complexed with matrix metalloproteinase-9. Damaged renal epithelial cells primarily release the monomeric and heterodimeric forms. Following renal tubular injury, NGAL can be detected within 2–3 h and shows a dynamic dose-dependent response. Studies have found that NGAL has good predictive ability for AKI in critically ill patients, renal transplant patients, and those who have undergone cardiac surgery18. A large multicenter study involving 1219 adult patients found that NGAL is an effective early predictor of AKI development following cardiac surgery, reporting that plasma NGAL level peaked within 6 h post-operation in the patients who developed AKI19,20.
Ho et al. have conducted a study on the abilities of urine and plasma NGAL levels to predict AKI complications following cardiac surgery and estimated the AUCs of these markers as 0.72 and 0.71, respectively21. However, in certain cases, the performance of serum NGAL level can be significantly better. For instance, Srisawat et al. have investigated the abilities of urine and plasma NGAL levels to predict AKI in 206 patients clinically suspected of leptospirosis, finding AUCs of 0.91 and 0.92, respectively21. Gombert demonstrated that an increase in plasma NGAL level is associated with an increase in plasma creatinine level within the preceding 48 h, with significant differences in NGAL levels between AKI patients requiring dialysis and those without AKI (p = 0.0002)22. The highest sensitivity and specificity for plasma NGAL levels after cardiopulmonary bypass were observed 12 h postoperatively (AUC = 0.80; OR = 11.0), with a cut-off value of > 105.37 ng/mL23.
CysC is a 13 kDa non-glycosylated protein belonging to the cystatin superfamily, involved in the intracellular catabolism of peptides. Due to its ability to freely pass through normal glomerular filtration and be completely reabsorbed by proximal tubular cells, CysC has been studied as a marker for glomerular filtration rate. Plasma CysC level is more sensitive to early renal dysfunction than plasma creatinine level, and urinary CysC level may be valuable in detecting renal tubular injury24. In a study involving 85 ICU patients, Herget-Rosenthal reported that changes in plasma CysC level could be used to detect AKI 0.6 day earlier than using plasma creatinine levels, with an AUROC of 0.92–0.9825. In a study involving 72 adult patients undergoing cardiac surgery, Koyner reported that the predictive index of urinary CysC level at 6 h postoperatively had an AUROC of 0.7226. Unlike other biomarkers, postoperative urinary CysC level significantly increased in AKI patients during follow-up, whereas non-AKI patients did not show this increase (p < 0.001). Galić reported that the plasma CysC level measured 12 h after cardiopulmonary bypass was a better predictor of AKI than the level measured at 6 h postoperatively (AUC = 0.96; OR = 130.2), with a cut-off value of ≥ 1.133 mg/L23. In this regard, their results are similar to ours.
Inflammatory responses play a critical role in the pathophysiological progression of AKI. TNFα, a potent proinflammatory cytokine predominantly produced by activated macrophages and monocytes, is one of the key mediators involved in this process. Various stimuli can upregulate the expression of cell adhesion molecules (such as ICAM-1 and VCAM-1), monocyte chemoattractant protein-1, and colony-stimulating factor-1 in different regions of the kidney. This upregulation facilitates the infiltration of monocytes and macrophages into sites of inflammation, further enhancing the release and expression of TNFα. Elevated TNFα levels can induce the generation of reactive oxygen species (ROS), leading to glomerular and tubular injury and ultimately resulting in AKI27. Wang et al. reported that during the transition from AKI to chronic kidney disease (CKD), the persistent presence of Minclehigh neutrophils and macrophages promotes sustained inflammation and TNFα-mediated fibrotic transformation28. Inhibition of TNFα expression has been shown to effectively attenuate inflammatory responses and cellular injury, thereby alleviating the onset and progression of AKI29. Moreover, natural compounds such as coptisine and formononetin have been demonstrated to suppress the expression of TNFα and other inflammatory mediators, mitigating hyperuricemia-induced renal inflammation and tubular injury, and thus exerting renoprotective effects30,31. In the present study, plasma TNFα levels were significantly higher in the AKI group than in the non-AKI group, consistent with the findings of Xu et al.32.
In a study by Larstorp A et al. involving 408 paediatric cardiac-surgery patients, 176 (43%) developed AKI postoperatively20. Among IL-4, IL-5, IL-6 and TNF, IL-8 demonstrated the highest discriminatory ability for predicting AKI progression, with an adjusted AUC of 0.80 (95% CI, 0.69–0.91)33. In the present study, plasma levels of IL-1β, IL-4, and IL-5 were relatively consistent between the AKI and non-AKI groups, with no statistically significant difference observed. Postoperative serum IL-6 and IL-8 levels were increased in both groups, but no differences were noted between the two groups, consistent with the findings of Presta et al.34.
This study is subject to several limitations. Firstly, as a single-center investigation with a relatively small sample size, the possibility of selection bias cannot be entirely ruled out. To mitigate the risk of overfitting, multivariate regression analysis was not conducted. Despite these constraints, this real-world, small-cohort study was designed to assess and validate the diagnostic utility of pertinent biomarkers for postoperative AKI and to identify potential risk factors. Future research involving larger, multicenter cohorts is necessary to substantiate these findings further. Secondly, urinary NGAL levels were not measured in this study, and the dynamic relationship between plasma NGAL levels and the progression of AKI was not explored. Additionally, the earliest biomarker measurement was conducted 12 h postoperatively, potentially missing the earlier diagnostic window for NGAL and CysC. Subsequent research will aim to evaluate these biomarkers at earlier postoperative intervals (e.g., 2 h) and in additional biological specimens, such as urine. Finally, due to the potential influence of various postoperative interventions, such as diuretic administration, on urine output assessment, the KDIGO urine output criteria were not utilized for diagnosing AKI in this study. This decision may have introduced potential bias into our results. Nevertheless, despite these limitations, our findings offer preliminary yet valuable evidence supporting the potential diagnostic utility of specific biomarkers for postoperative AKI. Furthermore, these insights are crucial for informing future large-scale, prospective studies.
Conclusion
Elevated levels of plasma NGAL and CysC following cardiopulmonary bypass surgery are risk factors for the development of AKI and hold predictive value for its occurrence.
Data availability
The dataset(s) supporting the findings of this study are available within the article.
References
Mehta, S. et al. The prognostic importance of duration of AKI: A systematic review and meta-analysis. BMC Nephrol. 19, 91 (2018).
Kellum, J. A., Sileanu, F. E., Bihorac, A., Hoste, E. A. & Chawla, L. S. Recovery after acute kidney injury. Am. J. Respir. Crit. Care Med. 195, 784–791 (2017).
Silver, S. A., Long, J., Zheng, Y. & Chertow, G. M. Cost of Acute Kidney injury in hospitalized patients. J. Hosp. Med. 12, 70–76 (2017).
Hansen, M. K. et al. Acute kidney injury and long-term risk of cardiovascular events after cardiac surgery: A population-based cohort study. J. Cardiothorac. Vasc. Anesth. 29, 617–625 (2015).
Chen, J. J. et al. Long-term outcomes of acute kidney injury after different types of cardiac surgeries: A population-based study. J. Am. Heart Assoc. 10, e019718 (2021).
Molina, A. A. et al. Impact of cardiac surgery associated acute kidney injury on 1-year major adverse kidney events. Front. Nephrol. 3, 1059668 (2023).
O’Neal, J. B., Shaw, A. D. & Billings, F. T. Acute kidney injury following cardiac surgery: Current understanding and future directions. Crit. Care. 20, 187 (2016).
Machado, M. N., Nakazone, M. A. & Maia, L. N. Prognostic value of acute kidney injury after cardiac surgery according to kidney disease: Improving global outcomes definition and staging (KDIGO) criteria. PLoS ONE 9, e98028 (2014).
Kovacevic, L., Lu, H., Kovacevic, N., Thomas, R. & Lakshmanan, Y. Cystatin C, neutrophil gelatinase-associated lipocalin, and lysozyme C: Urinary biomarkers for detection of early kidney dysfunction in children with urolithiasis. Urology 143, 221–226 (2020).
Luo, P., Ao, W., Xiang, D., Wang, J. & Liu, J. Values of serum neutrophil gelatinase-associated lipocalin and cystatin C after percutaneous coronary intervention for early diagnosis of contrast-induced nephropathy. Afr. Health Sci. 23, 593–598 (2023).
Star, R. A. Treatment of acute renal failure. Kidney Int. 54, 1817–1831 (1998).
Decoste, R., Himmelman, J. G. & Grantmyre, J. Acute renal infarct without apparent cause: A case report and review of the literature. CUAJ-Can. Urol. Assoc. J. 9, E237–E239 (2015).
Musso, C. G. et al. Creatinine reabsorption by the aged kidney. Int. Urol. Nephrol. 41, 727–731 (2009).
Wei, W. et al. Predictive value of creatine kinase MB for contrast-induced acute kidney injury among myocardial infarction patients. BMC Cardiovasc. Disord. 21, 337 (2021).
Saydam, O. et al. Emerging biomarker for predicting acute kidney injury after cardiac surgery: Cystatin C. Turk. J. Med. Sci. 48, 1096–1103 (2018).
Ortega-Loubon, C., Fernandez-Molina, M., Carrascal-Hinojal, Y. & Fulquet-Carreras, E. Cardiac surgery-associated acute kidney injury. Ann. Card. Anaesth. 19, 687–698 (2016).
Lakhal, K. et al. Early recognition of cardiac surgery-associated acute kidney injury: lack of added value of TIMP2 IGFBP7 over short-term changes in creatinine (an observational pilot study). BMC Anesthesiol. 21, 244 (2021).
Paragas, N. et al. The Ngal reporter mouse detects the response of the kidney to injury in real time. Nat. Med. 17, 216–222 (2011).
Ghonemy, T. A. & Amro, G. M. Plasma neutrophil gelatinase-associated lipocalin (NGAL) and plasma cystatin C (CysC) as biomarker of acute kidney injury after cardiac surgery. Saudi J. Kidney Dis. Transplant. 25, 582–588 (2014).
Larstorp, A., Salvador, C. L., Svensvik, B. A., Klingenberg, O. & Distante, S. Neutrophil gelatinase-associated lipocalin (NGAL) and cystatin C are early biomarkers of acute kidney injury associated with cardiac surgery. Scand. J. Clin. Lab. Invest. 82, 410–418 (2022).
Ho, J. et al. Urinary, plasma, and serum biomarkers’ utility for predicting acute kidney injury associated with cardiac surgery in adults: A meta-analysis. Am. J. Kidney. Dis. 66, 993–1005 (2015).
Gombert, A. et al. Comparison of urine and serum neutrophil gelatinase-associated lipocalin after open and endovascular thoraco-abdominal aortic surgery and their meaning as indicators of acute kidney injury. Vasa 48, 79–87 (2019).
Galic, S. et al. Early biochemical markers in the assessment of acute kidney injury in children after cardiac surgery. Ther. Apher. Dial. 26, 583–593 (2022).
Coll, E. et al. Serum cystatin C as a new marker for noninvasive estimation of glomerular filtration rate and as a marker for early renal impairment. Am. J. Kidney. Dis. 36, 29–34 (2000).
Herget-Rosenthal, S. et al. Early detection of acute renal failure by serum cystatin C. Kidney Int. 66, 1115–1122 (2004).
Koyner, J. L. et al. Urinary cystatin C as an early biomarker of acute kidney injury following adult cardiothoracic surgery. Kidney Int. 74, 1059–1069 (2008).
Sun, L. & Kanwar, Y. S. Relevance of TNF-alpha in the context of other inflammatory cytokines in the progression of diabetic nephropathy. Kidney Int. 88, 662–665 (2015).
Wang, C. et al. Mincle receptor in macrophage and neutrophil contributes to the unresolved inflammation during the transition from acute kidney injury to chronic kidney disease. Front. Immunol. 15, 1385696 (2024).
Zhang, B. et al. Nephroprotective effects of cardamonin on renal ischemia reperfusion injury/UUO-induced renal fibrosis. J. Agric. Food. Chem. 71, 13284–13303 (2023).
Liu, Y. et al. Coptisine protects against hyperuricemic nephropathy through alleviating inflammation, oxidative stress and mitochondrial apoptosis via PI3K/Akt signaling pathway. Biomed. Pharmacother. 156, 113941 (2022).
Zhang, N. X. et al. Formononetin alleviates ischemic acute kidney injury by regulating macrophage polarization through KLF6/STAT3 pathway. Am. J. Chin. Med. 52, 1487–1505 (2024).
Xu, S. & Tao, D. S. The effect of acute kidney injury on TGF-Beta1 and Smad3 in patients with coronary heart disease after off-pump coronary artery bypass grafting. J. Healthc. Eng. 2021, 1361242 (2021).
Greenberg, J. H. et al. Biomarkers of AKI progression after pediatric cardiac surgery. J. Am. Soc. Nephrol. 29, 1549–1556 (2018).
Presta, P. et al. Antecedent ACE-inhibition, inflammatory response, and cardiac surgery associated acute kidney injury. Rev. Cardiovasc. Med. 22, 207–213 (2021).
Acknowledgements
We thank all patients, their families, and the physicians who participated in this study.
Author information
Authors and Affiliations
Contributions
Zhongying Xu: Investigation, Data curation, Formal analysis, Writing—original draft. Min Zhang: Data curation, Formal analysis. Aixiang Yang: Supervision, Methodology, Review & editing. Weiyi Tao: Conceptualization, Supervision, Methodology, Review & editing.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval and consent to participate
This study was approved by the Ethics Committee of Suzhou Municipal Hospital (IRB numbers KL901511), and informed consent was obtained from all the included patients.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Xu, Z., Zhang, M., Yang, A. et al. Plasma neutrophil gelatinase-associated lipocalin protein and cystatin C as predictive biomarkers for acute kidney injury following cardiopulmonary bypass surgery: a prospective study. Sci Rep 15, 42037 (2025). https://doi.org/10.1038/s41598-025-26230-7
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-26230-7