Abstract
Climate change is accelerating at unprecedented rates, with disproportionate impacts on marine ecosystems. We examined the distributional responses of chum salmon (Oncorhynchus keta), a keystone species, to the recent climatic changes in the North Pacific from 1998 to 2022. We applied a multi-model ensemble approach to examine the spatial and temporal distribution patterns of suitable habitats throughout their seasonal migratory grounds and relate them to recent declines of Japanese chum salmon stocks. Our modelling identified temporal shifts in the locations of feeding and overwintering grounds, driven by increasing ocean temperatures, deteriorating forage conditions, and strengthening wind and marine heatwave intensity. During their feeding migration, the suitable chum habitat in the Bering Sea was reduced but increased in the Arctic. Overwintering habitat patterns further captured an overall decline in the trailing edges of their distribution, accompanied by habitat shifts towards the central North Pacific. Periods of marine heatwaves further coincided with sizable habitat losses. Such habitat displacements potentially affect the Japanese chum salmon stocks, shown by substantial habitat reductions in the Okhotsk and central Bering seas. These findings highlight the exacerbating exposure and vulnerability of the chum salmon populations to recent climatic and productivity changes throughout their marine life history, with concomitant repercussions on their production dynamics and provision of ecosystem services. Thus, requiring climate-adaptive measures such as the readjustment of fishing seasons and quotas according to the changing salmon stocks and habitat conditions, and the improvement of current hatcheries practices to manage and conserve salmon resources.
Similar content being viewed by others
Introduction
Climate change poses ubiquitous impacts on marine ecosystems, altering species distributions and threatening the provision and persistence of many crucial ecosystem services (e.g., goods, services, and cultural benefits)1,2. The ongoing ocean warming and increasing frequency and intensity of extreme events are already triggering abrupt and irreversible ecosystem changes3,4, with disproportionate effects on ecosystem components5. In the North Pacific, for instance, climate change and regional marine heatwaves (MHWs) from 2013 to 2016 and 2018–20226,7 resulted in devastating impacts on marine life, fisheries, and fishing communities8,9,10. For highly migratory species that spend their life cycle across diverse habitats and broad spatial scales, long-term (e.g., global warming) and short-term (e.g., MHWs) climate changes, and human stressors (e.g., overfishing and habitat loss) exert cumulative impacts on the species’ migration patterns, growth, and survival. This is notably true for all anadromous taxa including Pacific salmon (Oncorhynchus spp.), where natural environmental changes and multiple anthropogenic stressors were altering their distribution, production, and population dynamics11,12,13. Hence, an enhanced understanding of the spatial and temporal patterns of climate-driven distribution changes is imperative to better anticipate their ecological consequences, and forge climate-resilient solutions for effective salmon resource management and conservation.
Here, we focus on chum salmon (Oncorhynchus keta) and the potential impacts of the recent climate and environmental changes to their suitable marine habitats in the North Pacific. Chum salmon are the second most abundant, yet the most plentiful by total biomass, and widely distributed species of Pacific salmon14. Unlike many of Pacific salmon, chum salmon spend more of their life cycle in the ocean compared to other Pacific salmon taxa15. Over time, the abundance and catches of O. keta have fluctuated due to the influence of multiple factors such as climate and environmental changes16,17, and hatcheries (artificial hatching and release)18,19. From an ecological perspective, chum salmon, along with other Pacific salmonids, are keystone species affecting nutrient and material transport in the riparian ecosystem20, and are important prey item for many marine predators21. More importantly, chum salmon are integral to the food security, health, economy, and cultural practices of the indigenous peoples and local communities around the North Pacific Rim for millennia22,23.
In Japan, chum salmon supports important commercial fisheries and is the largest chum salmon fishery in the world, comprising more than half of the total harvest in the North Pacific from 1980 to 202324. Japanese chum salmon are largely maintained through hatcheries25. Despite continuous stock enhancement, the number and abundance of the returning Japanese chum salmon sharply declined from 2000 to the present24. Given the significance of chum salmon resources, the recent precipitous declines and historic low levels of the return rates and abundance in Japan and other stocks at the southern limit of their distributions (e.g., British Columbia)12,26,27,28 have raised growing concerns for resource managers and stakeholders. For example, in the southwestern margin of its distributional range (i.e., Korean peninsula), chum salmon abundance has been at low but stable level during the last 25 years24. These production dynamics, however, are in contrast with the increases in productivity reported for the northern stocks of chum salmon (e.g., Russia and Alaska) in recent decades12. Thus, for this study we explored the basin-wide changes in chum salmon habitat across the North Pacific and examined contemporary habitat trends at each putative seasonal migratory grounds for the Japanese chum salmon during their ocean residence. To do this, we constructed a multi-model ensemble to predict seasonal chum salmon habitat distributions, combining species occurrence records and environmental parameters from online repositories and published literature from 1998 to 2022. Such approach permitted us to quantify the relative importance of different environmental and climatic factors in shaping the chum salmon marine habitat and the rate of habitat exposure to recent climatic changes in the region.
While species distribution models (SDMs) have been widely and increasingly used in inferring spatial distributions of marine species29,30,31, the approach has not been intensively utilized for predicting the marine distributions of Pacific salmon. Some recent studies to date have applied statistical spatial models to investigate the distributions and abundance of Pacific salmon species32,33,34,35,36,37. Moreover, these modeling studies have either focused on regional salmon stocks or on the large-scale Pacific salmon abundance-based thermal habitat distributions. We aimed at building into and expanding these model exercises to capture the broad-scale seasonal distributions of the Pacific chum salmon using multiple environmental and climate factors to characterize their suitable marine habitats and responses to ongoing climate changes. While temperature remains one of the most prominent drivers of species distributions and community structure38,39,40, other factors such as food availability27,41, ocean currents42, salinity43, and wind patterns44,45 are important in shaping biodiversity patterns, and regulating species range shifts and mortality rates. Furthermore, multi-model ensemble approach, as implemented in this work, generate robust and more reliable predictions of species distributions than single model46,47.
With this framework, we hope to acquire further insights into the dynamics of suitable chum salmon habitats in the high seas, where our current understanding of the different aspects of their seasonal migrations remains limited. Specifically, we aim to (1) elucidate the spatio-temporal patterns of recent environmental and productivity changes and their impacts on chum salmon distribution throughout its migratory grounds at sea; (2) examine the rate of exposure of suitable chum salmon habitat to these variability; and (3) relate the suitable O. keta habitat changes with declines of Japanese chum salmon abundance in their putative migratory grounds from their ocean entry and return migration over the last 25 years.
Data and methods
Chum salmon occurrence data
Occurrence data of chum salmon were compiled throughout its reported distribution range in the North Pacific from available data repositories and published literature over the last 25 years (1998–2022). Here, we used presence records over catch data in modeling the seasonal habitat distributions of chum salmon to utilize species data from multiple sources, especially in the Okhotsk Sea where survey catch information were publicly unavailable from online databases. We utilized the data from the International Pacific Salmon Data Legacy Database (ver. 1.3; date accessed: 23 October 2024) available from North Pacific Anadromous Fish Commission (https://www.npafc.org/)33. These data were based on salmon research surveys in the North Pacific conducted by Canada, Japan, USA, and Russia, from 1955 to 202333. For these analyses, we used a subset of the data from 1998 to 2022, with surveys conducted using the gillnet, trawl, and longline. We used any location with reported chum salmon catch > 0 as species occurrence. These were supplemented by chum salmon records obtained from the Ocean Biodiversity Information System (OBIS) through the client ‘robis’ ver. 2.11.3 (date accessed: 24 April 2023) and the Global Biodiversity Information Facility (GBIF) using the interface ‘rgbif’ ver. 3.8.1 (date accessed: 24 April 2023) implemented in the R software (ver. 4.4.1). Additional data records from annual summer NOAA bottom trawl surveys in the Eastern and northeastern Bering Sea, Aleutian Island, and Gulf of Alaska were also compiled, accounting for 33% (n = 825) of the total occurrence records used for constructing the summer habitat model (https://www.fisheries.noaa.gov/foss/; date accessed: 24 April 2023). Finally, to supplement the occurrence information in the Okhotsk Sea, western North Pacific, western and central Bering Sea, data from Russian and Japanese Pacific salmon trawl surveys were digitized using the open-source WebPlotDigitizer ver. 3.448 from digital maps in published literature (Table S1). Given the multiple sources of occurrence records utilized for the present analysis, potential spatial, temporal, and sampling biases (e.g., differences in catchability across fishing gears leading to species detection bias) could affect the accuracy of model predictions49,50. Thus, caution should be taken into consideration when using these data to map species distributions over wide geographic scales.
Chum salmon occurrence records from all data sources were then combined seasonally (winter: December-February, Fig. 1a; spring: March-May, Fig. 1b; summer: June-August, Fig. 1c; and autumn: September-November, Fig. 1d) to capture the seasonal migration of Japanese chum salmon in its putative overwintering and feeding grounds in the North Pacific Ocean15,51 (Fig. 1e). We then preprocessed the annual, seasonally collated database to remove duplicated records and inland geographic coordinates. To account for the potential spatial sampling bias in seasonally compiled occurrences often arising from imbalanced surveys and opportunistic records52, we conducted a spatial thinning of monthly species records with a 100-km spatial threshold29 using the ‘thin.algorithm’ function of the ‘spThin’ ver. 0.2 R package53 (Fig. S1).
Japanese Chum salmon migration patterns and commercial catch data
The ocean migration of Japanese chum salmon starts with the ocean entry, where fry migrate in the shallow coastal waters prior to moving offshore into the summer feeding grounds in the southern Okhotsk Sea (Fig. 1e), as indicated by genetic and otolith mark studies15,54. They spend their first winter in the western North Pacific Ocean, and move to the summer feeding grounds in the Bering Sea in spring15,55. Immature and maturing chum salmon subsequently undergo seasonal migrations between the Gulf of Alaska and the Bering Sea during winter and summer, respectively56. Maturing chum salmon (age 3–4) exhibit an earlier intrusion into the Bering Sea than the immature ones (age 1–2) in spring57. Finally, maturing fish undertake the return migration from the Bering Sea to Japan through the western North Pacific in late summer and fall15.
The annual catch data of Japanese chum salmon from 1970 to 2024 were downloaded from the Salmon Research Department of Fisheries Resources Institute, Japan’s Fisheries Research Agency (https://www.fra.go.jp/shigen/salmon/kaiki.html; date accessed: 21 October 2025). For our analysis, we used a 25-year subset of chum salmon commercial catch data (in number) from 1998 to 2022, which were predominantly caught from Hokkaido. These data comprised of total catches within the Japanese territorial waters (at most 12 nautical miles from the coast), mainly targeting salmon returning to their natal rivers using set trap nets, longlines, and gillnets.
Spatial distributions of seasonally-compiled chum salmon occurrences for (a) winter, (b) spring, (c) summer, (d) autumn and (e) map of the North Pacific Ocean showing the migration routes (arrows) and putative overwintering and feeding grounds of Japanese chum salmon15,51,55. The maps were created using GMT 6.3.0. (https://docs.generic-mapping-tools.org/6.3/gmt.html).
Environmental parameters
We compiled a set of environmental parameters in the North Pacific (130°W-110°E; 30°-75°N) over the same 25-year period as for the occurrence data (1998–2022) that included physical and productivity variables. The physical variables comprised sea surface temperature (SST), marine heatwave (MHW) category, sea surface salinity (SSS), mixed layer depth (MLD), zonal sea water velocity (U), meridional sea water velocity (V), ocean current velocity (VEL), and sea surface wind speed (WS). The productivity variables included chlorophyll-a (CHL) concentration and zooplankton (ZOOP) biomass (see Table S2 for details on the data sets and sources of all predictor variables). This suite of environmental variables was selected based on their reported direct and indirect influences on chum salmon distribution at different marine life stages15,58. In particular, the inclusion of the MHW category as one of the covariates attempts to examine the impact of extreme events to chum salmon suitable marine habitat. All environmental data were resampled to a common 0.25° x 0.25° spatial grid using a bilinear interpolation method in ‘resample’ function of the ‘raster’ R package version 3.6–32. We then computed the seasonal averages for each environmental parameter to match the seasonally-compiled chum salmon occurrences, which we used for the construction of the seasonal-based species distribution models (see next section). All the data preprocessing and computations of seasonal averages were conducted using the R software version 4.5.059.
Species distribution models
Data processing for the development of seasonal species distribution models comprised the extraction of environmental predictor values at all locations with the seasonally compiled chum salmon occurrences available for each year during the 25-year period. We also generated the pseudo-absence data to supplement the true absences for each available year and season when chum salmon occurrences were recorded. To do this, we implemented a spatially constrained pseudo-absence points selection60 where we used a 100-km buffer zone around each presence record, matching the distance used for the spatial thinning of occurrence records, and randomly selected 10,000 pseudo-absences outside of the buffer area. We then, combined all the annual pseudo-absences (10,000 x n years with presence data, per season) and randomly selected the pseudo-absences (10,000 – true absences; Table S3) from the data pool61 for constructing each seasonal model. Pseudo-absences represent the range of environmental conditions available within the study area. Together with the occurrence data, they are used by model algorithms to fit the statistical species-environment relationships used to make model predictions61.
Once the input data for species distribution models were prepared, we constructed the seasonal models over the overwintering (winter and spring) and feeding (summer and autumn) migrations of chum salmon at sea. Prior to running the final seasonal models, we first checked for collinearity between the environmental parameters by season to exclude highly correlated variables (R > 0.70; Fig. S2)62. We then run each seasonal model using the twelve single algorithms implemented in the ‘biomod2’ version 4.2-6-263 R package to select the three most important variables for constructing the final multi-model ensembles. These models include regression (generalized additive model, generalized linear model, flexible discriminant analysis, multiple adaptive regression splines), decision trees (classification tree analysis, random forest), and machine learning algorithms (artificial neural network, generalized boosting model, maximum entropy, extreme gradient boosting training). Each model estimates an index of habitat suitability (hereafter, HSI) by statistically relating species records and environmental layers64, reflecting spatial and temporal variations in the habitat quality for chum salmon. The seasonal models were built using eight (physical variables) and ten (physical and productivity variables) environmental factors during the overwintering (winter and spring) and feeding (summer and autumn) migrations, respectively. Here, the productivity variables were excluded from the overwintering models as overwintering chum salmon feed less and rely primarily on their energy stores during these periods65,66. Nonetheless, we also ran the winter and spring habitat models with the full set of uncorrelated environmental covariates to examine the contributions of productivity variables to chum salmon suitable habitat (Table S4). Habitat predictions from these models did not vary significantly from those obtained from models without the productivity variables (Figs S3-S4), suggesting their minimal contributions to chum salmon habitat during winter and spring. Using the results of the preliminary model runs, we then constructed the final models using the three most important variables for each season (Table S4), to generate the parsimonious model that retain the most informative environmental variables to chum salmon marine habitat. In the final seasonal models, eleven single algorithms (excluding generalized linear model due to convergence failure) were developed (see Table S5 for details of modeling options implemented for each algorithm) and the multi-model seasonal ensemble of best-performing models were generated. For each seasonal model, we apportioned the occurrences into 80% training and 20% testing datasets, with three random-based cross-validation runs producing a total of 33 models. The multi-model ensembles based on the mean, committee average (where HSI predictions are expressed in terms of the percent of agreement across the selected models), median, and weighted mean of the best-performing single models were subsequently generated for each season using the Boyce index threshold derived from the model validation (testing datasets; Table S6). The threshold was computed as the average of the three highest values of Boyce index obtained from the cross-validation runs of each single model. The Boyce index is a presence-only performance metric that measures how much do the model predictions differ from the random distributions of observed occurrences across the prediction gradients67. Its values can range from − 1 to + 1, with positive values indicating that presences are more frequent than expected by chance in areas with higher predicted habitat suitability. Values close to zero suggest that suitability predictions are no better than random, and negative values indicate counter predictions (i.e., presences are more frequent in areas with lower predicted habitat suitability)68.
The model performance of the seasonal multi-model ensembles was further evaluated using other additional metrics including the true skill statistic (TSS), area under the receiver operating characteristic (ROC) curve (AUC) and the Cohen’s kappa (hereafter referred to as kappa). TSS index is computed based on the components of the confusion matrix capturing matches and mismatches between the observations and model predictions69 and is independent of prevalence70. TSS ranges from − 1 to + 1, where − 1 and + 1 indicates incorrect and correct predictions, respectively, and 0 suggests random predictions. The AUC metric assesses the capacity of the model to distinguish between presence and absence and is independent of the classification threshold71. The AUC index ranges from 0 to 1, where values closer to 1 indicate perfect discrimination and scores of 0.5 or less suggest low discrimination power72. In contrast, the kappa is a threshold- and prevalence-dependent metric that normalizes the overall accuracy of the model predictions by the accuracy expected to occur by chance70. The kappa statistic values range from − 1 to + 1, where + 1 represents a perfect agreement and values of zero or less indicate a performance no better than random73.
Temporal trends and bioclimatic velocity
To examine the long-term changes in seasonal environmental conditions and suitable marine habitat of chum salmon over the 25-year period across the North Pacific Ocean, we computed the pixel-wise temporal trends of the most influential environmental parameters and model-derived potential habitat using the ‘tempTrend’ function of the VoCC R package74. We also explored the rate of exposure of chum salmon to recent climate and productivity changes in its marine habitats. To examine this, we calculated the bioclimatic velocity75,76 over the 25-year period as the ratio between the temporal trend and spatial gradient in the multi-model ensemble predictions of habitat suitability using the ‘gVOCC’ function. We truncated the low values of spatial gradient of habitat suitability at 0.001 HSI/km to avoid very low gradients generating near infinite velocities74,76.
Results
Model predictive performance and importance of environmental parameters
The multi-model ensembles developed seasonally from the suite of best performing single algorithm models to predict suitable habitat distributions for chum salmon in the North Pacific showed modest to high predictive performance (Table S7). Selected single algorithm models based on the Boyce index for each season varied (Table S2), and generally included machine-learning algorithms (e.g., maximum entropy, MAXENT; and random forest, RF). Across the multi-model ensembles, all seasonal models gave high values for all the performance metrics, except for the spring model that registered modest values (Table S7). The differences in model predictive skills are reflective of the disparity in the number and spatial coverage of available species records used to fit the models. Across these four multi-model ensembles, the weighted mean model outperformed the rest of the algorithms based on computed performance metrics. More importantly, using a multi-model ensemble approach resulted in an overall gain of predictive performance for the seasonal models, with highest improvement in spring model (18% increase in Boyce index).
The relative importance of each environmental parameters averaged over the 12 single algorithm models captured the significant influence of sea surface temperature (SST; 33–60%) for all seasonal models (Table S3). Nonetheless, zooplankton biomass (ZOOP), wind speed (WS), marine heatwave (MHW), and sea surface salinity (SSS) showed modest contributions to potential chum salmon habitat. Model-derived ranges of environmental preferences from each seasonal model also reflected the differences across seasons (Fig. 2; Fig. S5). In particular, high suitable chum salmon habitats in winter were situated in areas with SST and WS ranging from 3.2 to 9.2 °C and 1.8–10.4 m/s (Fig. 2a, left panel), and SST and MLD from 6.2 to 9.2 °C and 61.4–79.5 m (Fig. 2a, right panel). In spring, however, the high suitable chum salmon habitats were located within narrower ranges of SST (4.3–6.4 °C) and WS (8.9–9.6 m/s) relative to winter (Fig. 2b, left panel). This is concomitant with moderately high suitable habitat in areas with no to moderate MHW (Fig. 2b, right panel). During summer, the high chum salmon habitats shifted to warmer and productive waters, with SST and ZOOP ranging from 7.4 to 13.7 °C and 4.9–9.7 g/m2, respectively (Fig. 2c, left panel). Interestingly, suitable habitats were captured during the season with moderate to strong MHW, yet SST remained within its preferred range (Fig. 2c, right panel). In autumn, the preferred SST and ZOOP ranged from 4.5 to 8.2 °C, 3.1–7.2 g/m2 (Fig. 2d, left panel), and SST and SSS from 4.5 to 6.9 °C and 27.8–31.9 ppt (Fig. 2d, right panel). In general, during the migratory phases of chum salmon in its overwintering and feeding grounds, contractions in the preferred environmental ranges were consistently evident towards the latter parts of seasonal migrations (spring and autumn, respectively).
Bivariate response curves of the most important variables for seasonal multi-model weighted mean ensemble for (a) winter, (b) spring, (c) summer, and (d) autumn. SST ranges are plotted in the x-axes against the second (left panels) and third (right panels) influential variables to suitable chum salmon habitat.
Spatial patterns and temporal environmental trends
Spatial distributions of temporal trends over the 25-year period for environmental parameters in the North Pacific showed strong seasonal differences and highlighted regions with the largest changes over time (Fig. 3; Fig. S6). For instance, during the overwintering migration from winter to spring, extensive warming trends were observed throughout the study area (Fig. 3a,b, left panels). The maximum warming trend (0.16 °C/yr) was observed in the western North Pacific (WNP, 140°E-180°) in winter, accompanied by extensive warming in the Central North Pacific (CNP, 180°-160°E), extending throughout the Gulf of Alaska (GOA) and the Eastern North Pacific (ENP, 160°E-120°E; Fig. 3a, left panel). Temporal trends of winter WS showed a pronounced zonal contrast characterized by the increasing sea surface winds in the WNP and weakening in the CNP and ENP (Fig. 3a, middle panel). This is accompanied with pronounced deepening of the MLD in WNP, extending further in the CNP (Fig. 3a, right panel). In spring, the extent of warming in the WNP contracted, albeit, sustained warming signals in the GOA and ENP prevailed (Fig. 3b, left panel). Temporal trends of WS in spring, however, reflected a decline in WS and pattern reversal in the GOA and ENP relative to winter (Fig. 3b, middle panel). Further, observed increases in the MHW strength category emerged (Fig. 3b, right panel), collocated in areas exhibiting moderate to high warming trends over the last 25 years.
Spatial distributions of the temporal trends of the most influential environmental predictors of suitable chum salmon habitat during overwintering periods (sea surface temperature, SST; wind speed, WS) from (a) winter to (b) spring, and feeding periods (SST; zooplankton biomass, ZOOP) from (c) summer to (d) autumn across the 25-year period (1998–2022). The maps were created using GMT 6.3.0. (https://docs.generic-mapping-tools.org/6.3/gmt.html).
During the chum salmon seasonal feeding migrations in summer and autumn, 60–61% of the geographic domain (130°E-110°W; 30°-70°N) showed SST increases over time, with pronounced warming trends located in chum salmon feeding grounds in the Okhotsk Sea and Bering Sea (Fig. 3c,d, left panels). During the summer (autumn) period, the zonal averages of SST trends in the Okhotsk Sea and Bering Sea showed that the areas have warmed by as much as 0.04 (0.05) °C/yr and 0.08 (0.05) °C/yr, respectively. In particular, the maximum warming trend in summer (0.26 °C/yr) was recorded in the Bering Sea (Fig. 3c, left panel). Further, the strong warming trends observed in summer and autumn coincided with conspicuous declines in the zooplankton biomass (Fig. 3c-d, middle panels) over the chum salmon feeding areas in the Okhotsk Sea (-0.06 g/m2/yr) and Bering Sea (-0.03 g/m2/yr). In summer, the MHW strength category showed increasing trends in the WBS and the ENP (Fig. 3c, right panel). In autumn, a regional freshening was observed in the eastern Bering Sea and GOA over the last 25 years (Fig. 3d, right panel), likely in response to the increase in the freshwater discharge from surrounding glaciated and non-glaciated watersheds, and changes in precipitation patterns associated with warming in recent decades77,78.
Chum salmon suitable habitat exposure
The pace at which suitable chum salmon habitat varied in response to climate and productivity changes in the North Pacific over the 25-year period highlighted distinct spatial and seasonal patterns (Fig. 4). In winter, the suitable overwintering habitat of chum salmon in the WNP (139°W-180° and 35°-56°N) has declined at an average rate of − 1.26 ± 2.55 km/yr (mean ±1 standard deviation, SD), accompanied by increases in CNP (180°-160°E and 35°-56°N; 0.99 ± 3.21 km/yr) and northern part of GOA (1.33 ± 3.71 km/yr; 160°-120°E and ≥ 48°N) (Fig. 4a). In spring, the northern waters of the WNP and western Bering Sea (WBS) showed increasing suitable habitat, whereas waters in the GOA recorded pronounced declines, with rates higher than the winter period (Fig. 4b). During the feeding migrations of chum salmon in summer and autumn, massive declines in suitable habitat were observed throughout the North Pacific, with noticeable increases in the northern waters of the Bering-Chukchi Seas and Okhotsk Sea (Fig. 4c,d). In summer, the feeding habitat of chum salmon declined in the central Bering Sea whilst increased in near-coast waters of the Okhotsk Sea, northeastern Bering and Chukchi seas (Fig. 4c). In autumn, declines in chum salmon feeding habitat were confined in the western Bering Sea, and southern waters of the Aleutian Islands, with slight decreases in the Okhotsk Sea and Kuril Islands (Fig. 4d). These were further coupled with increases in the chum salmon feeding habitat in the Chukchi Sea and northern waters of the Okhotsk Sea.
Spatial distributions of bioclimatic velocity of suitable chum salmon habitat during its overwintering (a) winter (December-February), (b) spring (March-May) and feeding migrations (c) summer (June-August), and (d) autumn (September-November) over the last 25 years (1998–2022). The maps were created using GMT 6.3.0. (https://docs.generic-mapping-tools.org/6.3/gmt.html).
Suitable habitat changes in Japanese Chum salmon migratory grounds
In the putative oceanic habitats of the Japanese chum salmon across the North Pacific (Fig. 5a), pronounced declines in abundance (Fig. 5b), and fluctuations in habitat suitability index (HSI) across different regions over the 25-year period were observed (Fig. 5c-f). In particular, during its ocean entry in coastal waters in early summer and subsequent movement into offshore areas of the Okhotsk Sea from late summer to autumn, annual averaged suitable chum salmon habitat showed inter-annual variability, characterized by a significant increasing (decreasing) trend in suitable habitat trend in summer (autumn) (Fig. 5c). During its first overwintering migration in the WNP, however, no significant trends in suitable chum salmon habitat were observed in winter and spring (Fig. 5d). As Japanese chum salmon undergo their feeding migration in the Bering Sea, significant declines in summer and autumn habitat were observed over the 25-year period (Fig. 5e). During their overwintering migration in the GOA in winter and spring, strong inter-annual fluctuations, albeit, no significant trends in suitable habitat were observed over the 25-year period (Fig. 5f).
(a) Map of the putative oceanic habitats for the Japanese chum salmon in the North Pacific (represented by the black boxes15,55, and (b) its annual catches from 1998 to 2022, noting the significant declining trend and years with highest (light red; 2004) and lowest (blue; 2021) values. The time-series of average annual habitat suitability index (HSI) during its feeding and overwintering migrations were computed for the (c) Okhotsk Sea, (d) Western North Pacific, (e) Central Bering Sea, and (f) Gulf of Alaska from 1998 to 2022. Trend lines of seasons with significant trends over time were overlain in dashed lines. The map in (a) was created using GMT 6.3.0. (https://docs.generic-mapping-tools.org/6.3/gmt.html).
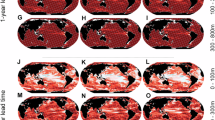
Spatial distributions of chum salmon habitat across the different seasons (Figs. S7–S10) and representative brood years of the Japanese chum salmon under contrasting levels of catches showed inter-annual differences in the North Pacific and putative migratory grounds (Fig. 6a-b). In particular, tracking the spatial distributions of suitable habitat of Japanese chum salmon for the brood year (BY) 2001, recording the highest catch (2004), showed generally higher HSI across the migratory grounds (Fig. 6a) relative to BY 2018 (Fig. 6b), marking the lowest catch over the 25-year period. Discernable changes in chum salmon habitat between the years of high (2004) and low (2021) catches were primarily observed in their feeding grounds in the Central Bering Sea. The latter showed comparatively less favorable habitat coinciding with the recent period of marine heatwaves (2018–2021).
Spatial distributions of suitable chum habitat based on the multi-model weighted mean ensemble for representative years of (a) highest (2004) and (b) lowest (2021) catches of Japanese chum salmon across the duration of its ocean residence. The putative overwintering and feeding grounds (black polygons15,55 are overlain on the maps. Years in red are periods of marine heatwaves. The maps were created using GMT 6.3.0. (https://docs.generic-mapping-tools.org/6.3/gmt.html).
Discussion
Our analyses provide perspectives on spatial and temporal distributions of the chum salmon throughout their migratory grounds in the North Pacific over the last 25 years elucidated from results of multi-ensemble species distribution models. This modeling approach enabled us to examine the relative effects of key environmental parameters to suitable chum salmon habitat across spatio-temporal scales. With recent availability of compiled Pacific salmon data from various national and international surveys33,79, along with publicly-available repositories for species occurrences, climate and environmental datasets, we were able to broadly explore the basin-scale distributions of chum salmon habitat and their response to rapid climatic changes in recent decades. Our findings highlighted the significant changes across the different seasons and regions of the North Pacific, mainly driven by fluctuations in sea surface temperature, wind, MLD, food availability, MHW, and salinity. The impacts of warming ocean temperatures have often been linked to declines (increases) of many Pacific salmon stocks at the trailing (leading) edges of their distribution12,36,80,81,82. These are consistent with the results of our analysis. Indeed, the unprecedented ocean warming over the last two decades resulted in contraction (expansion) of suitable chum salmon habitat at their southern (northern) ranges during their overwintering and feeding migrations.
During the overwintering migration, the formation of chum salmon habitat in the North Pacific was also regulated by sea surface winds, where moderate winter winds (5.7–10.7 m/s) created highly suitable habitat. While previous studies examining the effects of scalar winds on Pacific salmon reported an overall positive relationship with growth in late spring and summer58,83,84,85, the increased turbulence from strong winds in winter has negative impacts on growth and behavior of Pacific and Atlantic salmon85,86. Notably increasing trends of winter winds in many areas of the North Pacific over the last 25 years could be detrimental to chum salmon survival, especially in the western North Pacific. In spring, strong winds could affect the chum salmon growth and abundance when wind mixing disrupts the stratification of water column, potentially delaying spring bloom58. On one hand, high zooplankton biomass favor formation of suitable chum salmon habitat as it undergoes its seasonal feeding migration in summer and autumn. The declines in summer zooplankton biomass in the Okhotsk Sea and Bering Sea over time suggest poor feeding conditions for chum salmon, with potential negative consequences on the energy allocated for growth and storage. In the Eastern Bering Sea, for instance, the low prey availability and quality in recent years of unprecedented warming events led to poor body conditions and low energy stores for juvenile chum salmon, increasing their vulnerability and overwintering mortality16,87. In particular, cold and warm years in the Eastern Bering Sea were characterized by a sharp contrast in zooplankton abundance and composition, marked with the presence and absence of large copepods and euphausiids, respectively88. This pattern in zooplankton composition during cold years was also observed in the Okhotsk Sea89. Similarly, the recent significant increases in pink salmon biomass in the Bering Sea from Asian and North American stocks are likely to compound the dire impacts of limited food availability on salmonids and other fish species26,90,91.
The redistribution of suitable chum salmon habitat throughout their distribution range identifies critical and emerging regions of habitat loss and gain, with important ramifications on vulnerability and resilience of different chum salmon stocks under ongoing climate change. In particular, recent ocean warming is exacerbated by the increased frequency and severity of extreme events such as marine heatwaves (MHWs), bringing about marked biological impacts that threaten the integrity of the marine ecosystems8,92. Our modeling results also captured the negative impacts of the largest 2013–2015 MHW on record93 to chum salmon overwintering habitat in the GOA, characterized by the significant habitat contraction in their southern limits of their distribution and zonal shift in central North Pacific. This multi-year prolonged warming resulted to declines and biogeographic shifts of many fisheries stocks8,94. In contrast, the rapid poleward expansions of suitable chum salmon habitat in summer and autumn predicted from our models were in agreement with recent documented influx of chum salmon in the Canadian Arctic in response to warming and sea ice loss95,96. Our findings also aligned with the reported impacts of warmer ocean temperatures, MHWs, and sea ice loss to the thermal suitability across the migration corridors of western Alaska chum salmon in the Bering Sea and GOA, potentially leading to poor conditions and poleward shift97. The northward shifts of sub-Arctic species to the Arctic waters are increasingly reported and raised diverse ecological, social, and economic concerns29,98,99,100,101. For Pacific salmon, the multifaceted impacts of ongoing climate changes have far-reaching and cascading consequences for the communities dependent on them and various ecosystem services they provide96,102.
Spatial and temporal patterns of predicted habitat across its putative migratory grounds provide insights into potential drivers of recent declines in the abundance of the Japanese chum salmon. Despite the lack of significant trends in the suitable habitat of Japanese chum salmon during their ocean entry in the coastal and offshore waters of the Okhotsk Sea, an overall basin-wide decline in the summer suitable habitat emerged. This is consistent with reported shrinking and shifting regions of the adaptable and optimal growth temperatures for chum salmon in the Okhotsk Sea in recent decades12,26. Our results also highlighted the significant decrease in suitable habitat for the Japanese chum salmon during their first overwintering migration in the WNP, suggesting that the unfavorable overwintering conditions could have resulted in the low survival rates in recent decades. The cumulative impacts of poor marine habitats for Japanese chum salmon during its early ocean life stages may have contributed to the recent declines in their abundance, coinciding to a period when the size-dependent mortality rates are highest103. Further deterioration of summer and autumn suitable habitats of the Japanese chum salmon in their feeding grounds in the CBS could have amplified the declines in abundance. In recent decades, primary productivity and zooplankton community composition in the Bering Sea have fluctuated in response to the rapid warming and loss of seasonal sea ice104,105,106. These changes resulted in poor foraging conditions for chum salmon16,87 and other fish species107,108. With the low food availability for growth and energy storage in the preceding summer and autumn seasons, warming temperatures in their overwintering grounds in the GOA could adversely impact the survival of Japanese chum salmon. This is further supported by recent observations of poor body condition and low-lipid content of Japanese chum salmon samples collected in the GOA51. Albeit the availability of suitable habitats in the early marine life stages remained crucial to Japanese chum salmon productivity, shrinking suitable feeding habitats in the Bering Sea could further create bottlenecks that are likely to constrain their future productivity.
While our current modeling exercise provided seasonal and basin-wide distributions of chum salmon in recent decades, our analyses exclude the potential effects of non-habitat factors such as biotic interactions and hatcheries impacts on changes in suitable chum salmon habitat and abundance. For instance, recent studies of co-migratory salmonids reported the competitive effects of pink salmon on chum salmon stocks in the ocean, where the former outcompeted the latter26,91,109. Increasing abundance of pink salmon in the ocean was also linked to declines in the body size and productivity of sockeye salmon90,110 and declines in the catches of Chinook salmon111. The high abundance, elevated consumption rates, rapid growth, and generalist food habits of pink salmon afford them a competitive advantage over other salmonids112,113,114. Similarly, large releases of hatchery salmonids elicited negative density-dependent responses on inter-mingling salmon stocks, causing adverse genetic effects on diversity, productivity and abundance115. Hence, given the negative effects of warming on ocean productivity and over reliance on hatchery populations, the increasing intra- and inter-species competitions for space and prey resources will continue to impact the growth and dynamics of Pacific salmon80,115,116. Future studies using species distribution model should incorporate information or indices of species interactions and hatchery releases to better characterize the spatio-temporal changes in chum salmon habitat.
Another caveat from our current analyses emerged from the used of all occurrence data available for chum salmon at a species level to build the SDMs, while interpreting the model predictions within the context of the putative migration grounds of the Japanese stock. Potential differences in the environmental preferences and habitat and non-habitat drivers across chum salmon populations in the North Pacific could solicit contrasting responses. Hence, considering genetic and lineage-level distribution models could improve habitat predictions117. Finally, sustained high seas surveys especially during the challenging winter and spring periods will be increasingly important for providing more occurrence and abundance datasets to improve species distribution models and obtain more robust patterns of shifts in chum salmon suitable habitat across their overwintering migration grounds in the North Pacific. Availability of chum salmon data in the Okhotsk Sea during the summer season could further improve and reduce the uncertainty of habitat predictions throughout the distributional range of their feeding grounds.
Conclusion
Our findings add to the accumulating evidence of the adverse climate-driven impacts on chum salmon populations and provide insights into concomitant changes in their broad-scale habitat distributions in the North Pacific. In particular, we captured the significant trends of increasing ocean temperatures and declining productivity in the last two decades, along with an increasing frequency of extreme events. As extreme events are becoming more prevalent, more studies on their ecosystem impacts are warranted. Given the potential of marine heatwaves to exacerbate climate change impacts on fisheries8, more studies on their ecosystem impacts on chum salmon stocks are needed. For example, such conditions are known to have resulted in recent shifts and contractions of the seasonal marine chum salmon habitat distribution while increasing the exposure and vulnerability to ongoing climate and environmental changes16,26,80,96. Thus, escalating the volatility of the ecosystem services they provide. Our analyses shed further light into the recent decline of Japanese chum salmon, potentially driven by the deterioration of their overwintering and feeding habitats in the North Pacific. With the exacerbating climatic changes combined with other pervasive anthropogenic stressors, understanding the spatial and temporal redistributions of suitable chum salmon habitat within and beyond their known distribution range is crucial for designing adaptive ecosystem-based management to conserve the shifting and declining salmon resources20,80.
Data Availability
Data sources and their details used for the analyses are available in the Supplementary materials. Presence records for chum salmon are publicly available from the International Pacific Salmon Data Legacy Database (https://www.npafc.org/), R clients for Ocean Biodiversity Information System (OBIS; ‘robis’, https://rdrr.io/cran/robis/man/robis.html) and the Global Biodiversity Information Facility (GBIF; ‘rgbif’, https://cran.r-project.org/web/packages/rgbif/index.html), and NOAA summer bottom trawl surveys (https://www.fisheries.noaa.gov/foss/). The annual catch data of Japanese chum salmon between 1970 and 2024 were downloaded from Japan’s Fisheries Research Agency (https://www.fra.go.jp/shigen/salmon/kaiki.html). Daily data of sea surface temperature were publicly available and downloaded from the NOAA Physical Sciences Laboratory (https://psl.noaa.gov/data/gridded/data.noaa.oisst.v2.highres.html). The zooplankton biomass data, monthly chlorophyll-a concentration, salinity, and ocean currents from biogeochemical models were downloaded from the Copernicus Marine Environment Monitoring Service (CMEMS; https://resources.marine.copernicus.eu/). The daily-resolved marine heatwave (MHW) category data were available online from the NOAA coral reef watch (https://coralreefwatch.noaa.gov/product/marine_heatwave/) and the monthly sea surface wind speed from NOAA NCEI Blended Science Quality data were available from NOAA website (https://coastwatch.noaa.gov/erddap/griddap/noaacwBlendedWindsMonthly.graph).
Data availability
Data sources and their details used for the analyses are available in the Supplementary materials. Presence records for chum salmon are publicly available from the International Pacific Salmon Data Legacy Database (https://www.npafc.org/), R clients for Ocean Biodiversity Information System (OBIS; ‘robis’, https://rdrr.io/cran/robis/man/robis.html) and the Global Biodiversity Information Facility (GBIF; ‘rgbif’, https://cran.r-project.org/web/packages/rgbif/index.html), and NOAA summer bottom trawl surveys (https://www.fisheries.noaa.gov/foss/). The annual catch data of Japanese chum salmon between 1970 and 2024 were downloaded from the Salmon Research Department of Fisheries Resources Institute, Japan’s Fisheries Research Agency (https://www.fra.go.jp/shigen/salmon/kaiki.html). Daily data of sea surface temperature were publicly available and downloaded from the NOAA Physical Sciences Laboratory (https://psl.noaa.gov/data/gridded/data.noaa.oisst.v2.highres.html). The zooplankton biomass data, monthly chlorophyll-a concentration, salinity, and ocean currents from biogeochemical models were downloaded from the Copernicus Marine Environment Monitoring Service (CMEMS; https://resources.marine.copernicus.eu/). The daily-resolved marine heatwave (MHW) category data were available online from the NOAA coral reef watch (https://coralreefwatch.noaa.gov/product/marine_heatwave/) and the monthly sea surface wind speed from NOAA NCEI Blended Science Quality data were available from NOAA website (https://coastwatch.noaa.gov/erddap/griddap/noaacwBlendedWindsMonthly.graph).
References
Doney, S. C. et al. Climate change impacts on marine ecosystems. Annual Rev. Mar. Sci. 4, 11–37. https://doi.org/10.1146/annurev-marine-041911-111611 (2012).
García Molinos, J., Lawler, J. J., Alabia, I. D. & Olden, J. D. In Reference Module in Earth Systems and Environmental Sciences (Elsevier, 2025).
Heinze, C. et al. The quiet crossing of ocean tipping points. Proc. Natl. Acad. Sci. USA 118, e2008478118. https://doi.org/10.1073/pnas.2008478118 (2021).
Lee, H. et al. IPCC, 2023: Climate Change 2023: Synthesis Report, Summary for Policymakers. Contribution of Working Groups I, II and III to the Sixth Assessment Report of the Intergovernmental Panel on Climate Change (eds Core Writing Team, Lee, H. & Romero, J.) (IPCC, 2023).
Venegas, R. M., Acevedo, J. & Treml, E. A. Three decades of ocean warming impacts on marine ecosystems: A review and perspective. Deep Sea Res. Part II. 212, 105318. https://doi.org/10.1016/j.dsr2.2023.105318 (2023).
Hauri, C. et al. More than marine heatwaves: A new regime of Heat, Acidity, and low oxygen compound extreme events in the Gulf of Alaska. AGU Adv. 5, e2023AV001039. https://doi.org/10.1029/2023AV001039 (2024).
Zhao, Y. & Yu, J. Y. Two marine heatwave (MHW) variants under a basinwide MHW conditioning mode in the North Pacific and their Atlantic associations. J. Clim. 36, 8657–8674. https://doi.org/10.1175/JCLI-D-23-0156.1 (2023).
Cheung, W. W. L. & Frölicher, T. L. Marine heatwaves exacerbate climate change impacts for fisheries in the Northeast Pacific. Sci. Rep. 10, 6678. https://doi.org/10.1038/s41598-020-63650-z (2020).
Free, C. M. et al. Impact of the 2014–2016 marine heatwave on US and Canada West Coast fisheries: surprises and lessons from key case studies. Fish Fish. 24, 652–674. https://doi.org/10.1111/faf.12753 (2023).
Welch, H. et al. Impacts of marine heatwaves on top predator distributions are variable but predictable. Nat. Commun. 14, 5188. https://doi.org/10.1038/s41467-023-40849-y (2023).
Cline, T. J., Ohlberger, J. & Schindler, D. E. Effects of warming climate and competition in the ocean for life-histories of Pacific salmon. Nat. Ecol. Evol. 3, 935–942. https://doi.org/10.1038/s41559-019-0901-7 (2019).
Kaeriyama, M. Warming climate impacts on production dynamics of Southern populations of Pacific salmon in the North Pacific ocean. Fish. Oceanogr. 32, 121–132. https://doi.org/10.1111/fog.12598 (2023).
Schoen, E. R. et al. Future of Pacific salmon in the face of environmental change: lessons from one of the world’s remaining productive salmon regions. Fisheries 42, 538–553. https://doi.org/10.1080/03632415.2017.1374251 (2017).
Salo, E. O. in In Pacific Salmon Life Histories (eds Groot, C. & Margolis, L.) (University of British Columbia, 1991).
Urawa, S., Beacham, T. D., Fukuwaka, M. & Kaeriyama, M. in The ocean ecology of Pacific salmon and trout (ed R. J. Beamish) (American Fisheries Society, 2018).
Farley, E. V. Jr. et al. Critical periods in the marine life history of juvenile Western Alaska Chum salmon in a changing climate. Mar. Ecol. Prog. Ser. 726, 149–160 (2024).
Kaeriyama, M. Dynamics on distribution, production, and biological interactions of Pacific salmon in the changing climate of the North Pacific ocean. North. Pac. Anadromous Fish. Comm. Tech. Rep. 17, 102–106 (2021).
Miyakoshi, Y., Nagata, M., Kitada, S. & Kaeriyama, M. Historical and current hatchery programs and management of Chum salmon in Hokkaido, Northern Japan. Rev. Fish. Sci. 21, 469–479. https://doi.org/10.1080/10641262.2013.836446 (2013).
Morita, K. et al. A review of Pacific salmon hatchery programmes on Hokkaido Island, Japan. ICES J. Mar. Sci. 63, 1353–1363. https://doi.org/10.1016/j.icesjms.2006.03.024 (2006).
Kaeriyama, M. & Sakaguchi, I. Ecosystem-based sustainable management of Chum salmon in japan’s warming climate. Mar. Policy. 157, 105842. https://doi.org/10.1016/j.marpol.2023.105842 (2023). https://doi.org/https://doi.org/
Okado, J. & Hasegawa, K. Exploring predators of Pacific salmon throughout their life history: the case of Japanese chum, pink, and Masu salmon. Rev. Fish. Biol. Fisheries. 34, 895–917. https://doi.org/10.1007/s11160-024-09858-y (2024).
Atlas, W. I. et al. Indigenous systems of management for culturally and ecologically resilient pacific salmon (Oncorhynchus spp.) fisheries. BioScience 71, 186–204 (2021). https://doi.org/10.1093/biosci/biaa144
Sutton, M. Q. The Fishing Link: Salmonids and the Initial Peopling of the Americas. PaleoAmerica 3, 231–259 (2017). https://doi.org/10.1080/20555563.2017.1331084
NPAFC. NPAFC Pacific Salmonid Catch Statistics (Updated June 2024) (North Pacific Anadromous Fish Commission, 2024).
Kitada, S. Lessons from Japan marine stock enhancement and sea ranching programmes over 100 years. Reviews Aquaculture. 12, 1944–1969. https://doi.org/10.1111/raq.12418 (2020). https://doi.org/https://
Kaeriyama, M., Alabia, I. D. & Urawa, S. Production trend of Hokkaido Chum salmon estimated by multivariable models incorporating environmental factors and biological interactions in the North Pacific ocean. N Pac. Anadr Fish. Comm. Tech. Rep. 23, 34–39. https://doi.org/10.23849/npafctr23/4bb8ty (2025).
Chang, Y. L. K., Honda, K. & Morita, K. Beyond lethal temperatures: factors behind the disappearance of Chum salmon from their Southern margins under climate change. PLOS ONE. 20, e0330957. https://doi.org/10.1371/journal.pone.0330957 (2025).
Beamish, R. The need to see a bigger picture to understand the ups and downs of Pacific salmon abundances. ICES J. Mar. Sci. 79, 1005–1014. https://doi.org/10.1093/icesjms/fsac036 (2022).
Alabia, I. D., García Molinos, J., Hirata, T., Mueter, F. J. & David, C. L. Pan-Arctic marine biodiversity and species co-occurrence patterns under recent climate. Sci. Rep. 13, 4076. https://doi.org/10.1038/s41598-023-30943-y (2023).
Robinson, N. M., Nelson, W. A., Costello, M. J., Sutherland, J. E. & Lundquist, C. J. A systematic review of Marine-Based species distribution models (SDMs) with recommendations for best practice. Front. Mar. Sci. 4 (2017).
Rodrigues, L. et al. d. S. Modelling the distribution of marine fishery resources: Where are we? Fish Fish. 24, 159–175 (2023). https://doi.org/10.1111/faf.12716
Freshwater, C., Anderson, S. C. & King, J. Model-based indices of juvenile Pacific salmon abundance highlight species-specific seasonal distributions and impacts of changes to survey design. Fish. Res. 277, 107063. https://doi.org/10.1016/j.fishres.2024.107063 (2024).
Langan, J. A., Cunningham, C. J., Watson, J. T. & McKinnell, S. Opening the black box: new insights into the role of temperature in the marine distributions of Pacific salmon. Fish Fish. 25, 551–568. https://doi.org/10.1111/faf.12825 (2024).
Somov, A., Farley, E. V., Yasumiishi, E. M. & McPhee, M. V. Comparison of juvenile Pacific salmon abundance, distribution, and body condition between Western and Eastern Bering sea using Spatiotemporal models. Fish. Res. 278, 107086. https://doi.org/10.1016/j.fishres.2024.107086 (2024).
Hart, L. K. G. et al. Species distribution models estimate time-varying juvenile salmon distributions in the north- and southeastern Bering sea. Can. J. Fish. Aquat. Sci. 82, 1–13. https://doi.org/10.1139/cjfas-2024-0137 (2025).
Shelton, A. O. et al. Redistribution of salmon populations in the Northeast Pacific ocean in response to climate. Fish Fish. 22, 503–517. https://doi.org/10.1111/faf.12530 (2021).
Jensen, A. J. et al. Modeling ocean distributions and abundances of natural- and hatchery-origin Chinook salmon stocks with integrated genetic and tagging data. PeerJ 11, e16487. https://doi.org/10.7717/peerj.16487 (2023).
Rutterford, L. A., Simpson, S. D., Bogstad, B., Devine, J. A. & Genner, M. J. Sea temperature is the primary driver of recent and predicted fish community structure across Northeast Atlantic shelf seas. Glob. Change Biol. 29, 2510–2521. https://doi.org/10.1111/gcb.16633 (2023).
Sunday, J. M., Bates, A. E. & Dulvy, N. K. Thermal tolerance and the global redistribution of animals. Nat. Clim. Change. 2, 686–690 (2012). https://doi.org/http://www.nature.com/nclimate/journal/v2/n9/abs/nclimate1539.html#supplementary-information
Waldock, C., Dornelas, M. & Bates, A. E. Temperature-Driven biodiversity change: disentangling space and time. BioScience biy096-biy096 https://doi.org/10.1093/biosci/biy096 (2018).
Danovaro, R. Understanding marine biodiversity patterns and drivers: the fall of Icarus. Mar. Ecol. n/a. (e12814). https://doi.org/10.1111/maec.12814 (2024).
García Molinos, J. et al. Climate, currents and species traits contribute to early stages of marine species redistribution. Commun. Biology. 5, 1329. https://doi.org/10.1038/s42003-022-04273-0 (2022).
Smyth, K. & Elliott, M. in Stressors in the Marine Environment: Physiological and Ecological Responses; Societal implications (eds Martin, S. & Nia, W.) (Oxford University Press, 2016).
Friedland, K. D. et al. Variation in wind and piscivorous predator fields affecting the survival of Atlantic salmon, Salmo salar, in the Gulf of Maine. Fish. Manag. Ecol. 19, 22–35. https://doi.org/10.1111/j.1365-2400.2011.00814.x (2012).
Peterman, R. M. & Bradford, M. J. Wind speed and mortality rate of a marine Fish, the Northern anchovy (Engraulis mordax). Science 235, 354–356. https://doi.org/10.1126/science.235.4786.354 (1987).
Araújo, M. B. & New, M. Ensemble forecasting of species distributions. Trends Ecol. Evol. 22, 42–47. https://doi.org/10.1016/j.tree.2006.09.010 (2007). https://doi.org/http://dx.doi.org/
Hao, T., Elith, J., Lahoz-Monfort, J. J. & Guillera-Arroita, G. Testing whether ensemble modelling is advantageous for maximising predictive performance of species distribution models. Ecography 43, 549–558. https://doi.org/10.1111/ecog.04890 (2020).
Rohatgi, A. WebPlotDigitizer user manual version 3.4. 1–18 (2014).
Boakes, E. H. et al. Distorted views of biodiversity: Spatial and Temporal bias in species occurrence data. PLoS Biol. 8, e1000385. https://doi.org/10.1371/journal.pbio.1000385 (2010).
Moudrý, V. et al. Optimising occurrence data in species distribution models: sample size, positional uncertainty, and sampling bias matter. Ecography 2024 (e07294). https://doi.org/10.1111/ecog.07294 (2024).
Urawa, S., Beacham, T., Sutherland, B. & Sato, S. Winter distribution of Chum salmon in the Gulf of alaska: a review. N Pac. Anadr Fish. Comm. Tech. Rep. 18, 93–97 (2022).
Baker, D. J., Maclean, I. M. D. & Gaston, K. J. Effective strategies for correcting Spatial sampling bias in species distribution models without independent test data. Divers. Distrib. 30, e13802. https://doi.org/10.1111/ddi.13802 (2024).
Aiello-Lammens, M. E., Boria, R. A., Radosavljevic, A., Vilela, B. & Anderson, R. P. SpThin: an R package for Spatial thinning of species occurrence records for use in ecological niche models. Ecography 38, 541–545. https://doi.org/10.1111/ecog.01132 (2015).
Urawa, S., Seiki, J. & Kawana, M. Origins of juvenile Chum salmon caught in the Southwestern Okhotsk sea during the fall of 2000. Bull. Natl. Salmon. Resour. Cent. 8, 9–16 (2006).
Kitada, S., Myers, K. W. & Kishino, H. Hatcheries to high seas: climate change connections to salmon marine survival. Ecol. Evol. 15 (e71504). https://doi.org/10.1002/ece3.71504 (2025).
Beacham, T. D. et al. Stock origins of Chum salmon (Oncorhynchus keta) in the Gulf of Alaska during winter as estimated with microsatellites. N Pac. Anadr Fish. Comm. Bull. 5, 15–23 (2009).
Nagasawa, T. & Azumaya, T. Distribution and CPUE trends in Pacific salmon, especially Sockeye salmon in the Bering sea and adjacent waters from 1972 to the mid 2000s. N Pac. Anadr Fish. Comm. Bull. 5, 1–13 (2009).
Kohan, M. L., Mueter, F. J., Orsi, J. A. & McPhee, M. V. Variation in size, condition, and abundance of juvenile Chum salmon (Oncorhynchus keta) in relation to marine factors in Southeast Alaska. Deep Sea Res. Part II. 165, 340–347. https://doi.org/10.1016/j.dsr2.2017.09.005 (2019).
R: A Language and Environment for Statistical Computing v. 4.5.0. (R Foundation for Statistical Computing, 2025).
Senay, S. D., Worner, S. P. & Ikeda, T. Novel Three-Step Pseudo-Absence selection technique for improved species distribution modelling. PLOS ONE. 8, e71218. https://doi.org/10.1371/journal.pone.0071218 (2013).
Barbet-Massin, M., Jiguet, F., Albert, C. H. & Thuiller, W. Selecting pseudo-absences for species distribution models: how, where and how many? Methods Ecol. Evol. 3, 327–338. https://doi.org/10.1111/j.2041-210X.2011.00172.x (2012).
Dormann, C. F. et al. Collinearity: a review of methods to deal with it and a simulation study evaluating their performance. Ecography 36, 27–46. https://doi.org/10.1111/j.1600-0587.2012.07348.x (2013). https://doi.org/.
Thuiller, W. et al. biomod2: Ensemble Platform for Species Distribution Modeling. R package version 4.2-6-2 (2025).
Elith, J. & Leathwick, J. R. Species distribution models: ecological explanation and prediction across space and time. Annu. Rev. Ecol. Evol. Syst. 40, 677–697 (2009).
Hurst, T. P. Causes and consequences of winter mortality in fishes. J. Fish Biol. 71, 315–345. https://doi.org/10.1111/j.1095-8649.2007.01596.x (2007). https://doi.org/https://
Waters, C. D. et al. Winter condition and trophic status of Pacific salmon in the Gulf of Alaska. N. Pac. Anadr. Fish comm. Tech. Rep. 18, 106–111. https://doi.org/10.23849/npafctr18/106.111 (2022).
Boyce, M. S., Vernier, P. R., Nielsen, S. E. & Schmiegelow, F. K. A. Evaluating resource selection functions. Ecol. Model. 157, 281–300. https://doi.org/10.1016/S0304-3800(02)00200-4 (2002).
Hirzel, A. H., Le Lay, G., Helfer, V., Randin, C. & Guisan, A. Evaluating the ability of habitat suitability models to predict species presences. Ecol. Model. 199, 142–152. https://doi.org/10.1016/j.ecolmodel.2006.05.017 (2006).
Fielding, A. H. & Bell, J. F. A review of methods for the assessment of prediction errors in conservation presence/absence models. Environ. Conserv. 24, 38–49 (1997). https://doi.org/doi:null
Allouche, O., Tsoar, A. & Kadmon, R. Assessing the accuracy of species distribution models: prevalence, kappa and the true skill statistic (TSS). J. Appl. Ecol. 43, 1223–1232. https://doi.org/10.1111/j.1365-2664.2006.01214.x (2006).
Sofaer, H. R., Hoeting, J. A. & Jarnevich, C. S. The area under the precision-recall curve as a performance metric for rare binary events. Methods Ecol. Evol. 10, 565–577. https://doi.org/10.1111/2041-210X.13140 (2019).
Jiménez-Valverde, A. Insights into the area under the receiver operating characteristic curve (AUC) as a discrimination measure in species distribution modelling. Glob. Ecol. Biogeogr. 21, 498–507. https://doi.org/10.1111/j.1466-8238.2011.00683.x (2012).
Cohen, J. A. Coefficient of agreement for nominal scales. Educ. Psychol. Meas. 20, 37–46. https://doi.org/10.1177/001316446002000104 (1960).
García Molinos, J., Schoeman, D. S., Brown, C. J., Burrows, M. T. & VoCC An r package for calculating the velocity of climate change and related Climatic metrics. Methods Ecol. Evol. 10, 2195–2202. https://doi.org/10.1111/2041-210X.13295 (2019).
Serra-Diaz, J. M. et al. Bioclimatic velocity: the Pace of species exposure to climate change. Divers. Distrib. 20, 169–180. https://doi.org/10.1111/ddi.12131 (2014).
Alabia, I. D. et al. Distribution shifts of marine taxa in the Pacific Arctic under contemporary climate changes. Divers. Distrib. 24, 1583–1597. https://doi.org/10.1111/ddi.12788 (2018).
Reister, I., Danielson, S. & Aguilar-Islas, A. Perspectives on Northern Gulf of Alaska salinity field structure, freshwater pathways, and controlling mechanisms. Prog. Oceanogr. 229, 103373. https://doi.org/10.1016/j.pocean.2024.103373 (2024).
Royer, T. C. & Grosch, C. E. Ocean warming and freshening in the Northern Gulf of Alaska. Geophys. Res. Lett. 33 https://doi.org/10.1029/2006GL026767 (2006).
McKinnell, S. & Langan, J. A. The international Pacific salmon data Legacy; historical sampling of salmon in the ocean. Bull. No. 7, 77–92 (2024).
Connors, B., Ruggerone, G. T. & Irvine, J. R. Adapting management of Pacific salmon to a warming and more crowded ocean. ICES J. Mar. Sci. fsae135 https://doi.org/10.1093/icesjms/fsae135 (2024).
Kaeriyama, M., Seo, H. & Qin, Y. Effect of global warming on the life history and population dynamics of Japanese Chum salmon. Fish. Sci. 80, 251–260. https://doi.org/10.1007/s12562-013-0693-7 (2014).
Springer, A. M. & van Vliet, G. B. Climate change, Pink salmon, and the nexus between bottom-up and top-down forcing in the Subarctic Pacific ocean and Bering sea. Proc. Natl. Acad. Sci. 111, E1880–E1888. https://doi.org/10.1073/pnas.1319089111 (2014).
Agler, B. A., Ruggerone, G. T., Wilson, L. I. & Mueter, F. J. Historical growth of Bristol Bay and Yukon River, Alaska Chum salmon (Oncorhynchus keta) in relation to climate and inter- and intraspecific competition. Deep Sea Res. Part II. 94, 165–177. https://doi.org/10.1016/j.dsr2.2013.03.028 (2013).
Mueter, F. J., Peterman, R. M. & Pyper, B. J. Opposite effects of ocean temperature on survival rates of 120 stocks of Pacific salmon (Oncorhynchus spp.) in Northern and Southern areas. Can. J. Fish. Aquat. Sci. 59, 456–463. https://doi.org/10.1139/f02-020 (2002).
Wells, B. K., Grimes, C. B. & Waldvogel, J. B. Quantifying the effects of wind, upwelling, curl, sea surface temperature and sea level height on growth and maturation of a California Chinook salmon (Oncorhynchus tshawytscha) population. Fish. Oceanogr. 16, 363–382. https://doi.org/10.1111/j.1365-2419.2007.00437.x (2007).
Barbier, E., Oppedal, F. & Hvas, M. Atlantic salmon in chronic turbulence: effects on growth, behaviour, welfare, and stress. Aquaculture 582, 740550. https://doi.org/10.1016/j.aquaculture.2024.740550 (2024).
Wechter, M. E., Beckman, B. R., Iii, A., Beaudreau, A. G., McPhee, M. V. & A. H. & Growth and condition of juvenile Chum and Pink salmon in the Northeastern Bering sea. Deep Sea Res. Part II. 135, 145–155. https://doi.org/10.1016/j.dsr2.2016.06.001 (2017).
Stabeno, P. J. et al. Comparison of warm and cold years on the southeastern Bering sea shelf and some implications for the ecosystem. Deep Sea Res. Part II. 65–70, 31–45. https://doi.org/10.1016/j.dsr2.2012.02.020 (2012). https://doi.org/http:
Kim, S. T. A review of the sea of Okhotsk ecosystem response to the climate with special emphasis on fish populations. ICES J. Mar. Sci. 69, 1123–1133. https://doi.org/10.1093/icesjms/fss107 (2012).
Rand, P. S. & Ruggerone, G. T. Biennial patterns in Alaskan Sockeye salmon ocean growth are associated with Pink salmon abundance in the Gulf of Alaska and the Bering sea. ICES J. Mar. Sci. 81, 701–709. https://doi.org/10.1093/icesjms/fsae022 (2024).
Ruggerone, G. T. et al. From diatoms to killer whales: impacts of Pink salmon on North Pacific ecosystems. Mar. Ecol. Prog. Ser. 719, 1–40 (2023).
Smith, K. E. et al. Biological impacts of marine heatwaves. Annual Rev. Mar. Sci. 15, 119–145. https://doi.org/10.1146/annurev-marine-032122-121437 (2023). https://doi.org/https://doi.org/
Bond, N. A., Cronin, M. F., Freeland, H. & Mantua, N. Causes and impacts of the 2014 warm anomaly in the NE Pacific. Geophys. Res. Lett. 42, 3414–3420. https://doi.org/10.1002/2015GL063306 (2015).
Peterson Williams, M. J., Gisclair, R., Cerny-Chipman, B., LeVine, E., Peterson, T. & M. & The heat is on: Gulf of Alaska Pacific Cod and climate-ready fisheries. ICES J. Mar. Sci. 79, 573–583. https://doi.org/10.1093/icesjms/fsab032 (2022).
Dunmall, K. M. et al. First juvenile Chum salmon confirms successful reproduction for Pacific salmon in the North American Arctic. Can. J. Fish. Aquat. Sci. 79, 703–707. https://doi.org/10.1139/cjfas-2022-0006 (2022).
Dunmall, K. M. et al. Pacific salmon in the Canadian Arctic highlight a range-expansion pathway for sub-Arctic fishes. Glob. Change Biol. 30, e17353 (2024). https://doi.org/10.1111/gcb.17353
Lemagie, E., Farley, E., Langan, J. A. & Stabeno, P. J. Mapping suitable thermal migration corridors for Western Alaska Chum salmon in the North Pacific. Deep Sea Res. Part I. 222, 104531. https://doi.org/10.1016/j.dsr.2025.104531 (2025).
Alabia, I. D., Garcia Molinos, J., Hirata, T., Narita, D. & Hirawake, T. Future redistribution of fishery resources suggests biological and economic trade-offs according to the severity of the emission scenario. PLOS ONE. 19, e0304718. https://doi.org/10.1371/journal.pone.0304718 (2024).
Dunmall, K. M. et al. Invading and range-expanding Pink salmon inform management actions for marine species on the move. ICES J. Mar. Sci. (fsae199). https://doi.org/10.1093/icesjms/fsae199 (2025).
Huntington, H. P. et al. Societal implications of a changing Arctic ocean. Ambio 51, 298–306. https://doi.org/10.1007/s13280-021-01601-2 (2022).
Levine, R. M. et al. Climate-driven shifts in pelagic fish distributions in a rapidly changing Pacific Arctic. Deep Sea Res. Part II. 208, 105244. https://doi.org/10.1016/j.dsr2.2022.105244 (2023).
Mantua, J. N. Shifting patterns in Pacific climate, West Coast salmon survival rates, and increased volatility in ecosystem services. Proc. Natl. Acad. Sci. 112, 10823–10824. https://doi.org/10.1073/pnas.1513511112 (2015).
Beamish, R. J. & Mahnken, C. A critical size and period hypothesis to explain natural regulation of salmon abundance and the linkage to climate and climate change. Prog. Oceanogr. 49, 423–437. https://doi.org/10.1016/S0079-6611(01)00034-9 (2001).
Eisner, L. B. et al. Seasonal, interannual, and Spatial patterns of community composition over the Eastern Bering sea shelf in cold years. Part I: zooplankton. ICES J. Mar. Sci. 75, 72–86. https://doi.org/10.1093/icesjms/fsx156 (2018).
Frey, K. E. & NOAA Arctic Report Card. : Arctic Ocean Primary Productivity: The Response of Marine Algae to Climate Warming and Sea Ice Decline. (2023). https://doi.org/10.25923/nb05-8w13
Kimmel, D. G., Eisner, L. B. & Pinchuk, A. I. The Northern Bering sea zooplankton community response to variability in sea ice: evidence from a series of warm and cold periods. Mar. Ecol. Prog. Ser. 705, 21–42 (2023).
Duffy-Anderson, J. T. et al. Responses of the Northern Bering sea and southeastern Bering sea pelagic ecosystems following Record-Breaking low winter sea ice. Geophys. Res. Lett. 46, 9833–9842. https://doi.org/10.1029/2019GL083396 (2019).
Hunt, G. L. Jr., Yasumiishi, E. M., Eisner, L. B., Stabeno, P. J. & Decker, M. B. Climate warming and the loss of sea ice: the impact of sea-ice variability on the southeastern Bering sea pelagic ecosystem. ICES J. Mar. Sci. 79, 937–953. https://doi.org/10.1093/icesjms/fsaa206 (2022).
Litz, M. N. C. et al. Competition with odd-year Pink salmon in the ocean affects natural populations of Chum salmon from Washington. Mar. Ecol. Prog. Ser. 663, 179–195 (2021).
Ohlberger, J., Cline, T. J., Schindler, D. E. & Lewis, B. Declines in body size of sockeye salmon associated with increased competition in the ocean. Proc. R. Soc. B 290, 20222248. https://doi.org/10.1098/rspb.2022.2248 (2023).
Ruggerone, G. T., Irvine, J. R. & Connors, B. Are there too many salmon in the North Pacific ocean? NPAFC News Letter. 51, 38–43 (2022).
Graham, C., Pakhomov, E. A. & Hunt, B. P. V. Meta-Analysis of salmon trophic ecology reveals Spatial and interspecies dynamics across the North Pacific ocean. Front. Mar. Sci. 8 https://doi.org/10.3389/fmars.2021.618884 (2021).
Qin, Y. & Kaeriyama, M. Feeding habits and trophic levels of Pacific salmon (Oncorhynchus spp.) in the North Pacific ocean. North. Pac. Anadromous Fish. Comm. Bull. 6, 469–481 (2016).
Ruggerone, G. T. & Nielsen, J. L. Evidence for competitive dominance of Pink salmon (Oncorhynchus gorbuscha) over other salmonids in the North Pacific ocean. Rev. Fish. Biol. Fisheries. 14, 371–390. https://doi.org/10.1007/s11160-004-6927-0 (2004).
McMillan, J. R. et al. A global synthesis of peer-reviewed research on the effects of hatchery salmonids on wild salmonids. Fish. Manag. Ecol. 30, 446–463. https://doi.org/10.1111/fme.12643 (2023).
Alen, B. W. V. Hatchery salmon and ecological overshoot. Aquaculture Fish. Fisheries. 5, e70103. https://doi.org/10.1002/aff2.70103 (2025). https://doi.org/
Zhang, Z. et al. Lineage-level distribution models lead to more realistic climate change predictions for a threatened crayfish. Divers. Distrib. 27, 684–695. https://doi.org/10.1111/ddi.13225 (2021).
Acknowledgements
This work is supported by Japan’s national programs on Arctic research, Arctic Challenge for Sustainability II (ArCS II; Grant No. JPMXD1420318865), ArCS III (Grant No. JPMXD1720251001) funded by Japan’s Ministry of Education, Culture, Sports, Science and Technology (MEXT), EO-RA3 (Grant No. RA3MAF006), and EO-RA4 (Grant No. ER4MAF008), funded by Japan Aerospace Exploration Agency (JAXA). The authors sincerely thank the different agencies that provided the environmental datasets and all the scientists, fishery and hatchery managers, and fishers who contributed to the collection of species data used for analyses.
Author information
Authors and Affiliations
Contributions
I.D.A., S.-I. S., and M.K. conceptualized the ideas for this study. I.D. A. performed the analyses and wrote the first draft of the manuscript. All the authors contributed to the discussions and provided comments and edits on the earlier versions of the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Alabia, I.D., Saitoh, SI., García Molinos, J. et al. Climate-driven shifts in marine habitat explain recent declines of Japanese Chum salmon. Sci Rep 15, 41747 (2025). https://doi.org/10.1038/s41598-025-26397-z
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-26397-z