Abstract
To determine the critical blink frequency threshold for maintaining tear film stability in young participants with dry eye symptoms. In this two-phase study, 45 young participants (aged 19–28 years) with dry eye symptoms (Ocular Surface Disease Index, OSDI ≥ 13) underwent spontaneous blink measurements (Phase 1) and controlled blink experiments (Phase 2: spontaneous, 10 blinks/min, and 20 blinks/min). Tear meniscus height (TMH), non-invasive tear break-up time (BUT), bulbar redness (BR), and Standard Patient Evaluation of Eye Dryness (SANDE) scores were obtained and analyzed. Spontaneous blink frequency (27.75 ± 14.43 blinks/min) positively correlated with TMH (r = 0.48, p < 0.01). Reducing blink frequency to 10 blinks/min significantly worsened TMH (p = 0.0002), BUT (p < 0.0001), BR (p < 0.0001), and SANDE scores (p < 0.0001) compared to spontaneous blinking. No differences were found between spontaneous blinking and controlled 20 blinks/min. A blink frequency threshold of ≥ 20 blinks/min is critical for stabilizing tear film in young participants with dry eye symptoms. Interventions targeting blink frequency may mitigate symptoms exacerbated by digital device use.
Similar content being viewed by others
Introduction
Dry eye is a chronic ocular surface disease caused by abnormalities in the quality, quantity, and dynamics of tears, leading to tear film instability or imbalance of the ocular surface microenvironment, which may be accompanied by inflammatory responses, tissue damage, and neural dysfunction, resulting in various ocular discomfort symptoms and/or visual impairment1. Dry eye has become a global public health issue, with approximately 5% to 34% of global population affected2,3. Chinese populations are considered a high-risk group, with a nation-wide symptom-based prevalence estimated of 31.4% and even higher than 50% in certain areas4,5. A crucial factor influencing tear film stability is the blink threshold, which refers to the minimum number of blinks required to maintain optimal tear distribution and ocular surface lubrication. Currently, there is a considerable number of dry eye studies focused on individuals over 40 years of age, but studies on the young population are very limited6. Exploring blink dynamics, including blink threshold, could offer valuable insights into the development and management of dry eye in younger people.
With changes of lifestyle and popularization of digital devices, the risk of dry eye in the young population may be gradually increasing7. Although studies have shown that reduced blink frequency due to video terminal use may exacerbate dry eye symptoms8, systematic researches on how different blink frequencies affect dry eye symptoms and signs are limited. This gap makes it difficult to implement evidence-based interventions. Since no studies have clarified the level at which reduced blink frequency significantly worsens symptoms, we conducted a two-phase study to assess the impact of specific blink frequencies on dry eye symptoms and core signs(tear film break-up time (BUT)9; tear meniscus height (TMH)10; bulbar redness(BR)11) to determine the “threshold” of blink frequency below which tear film function will be severely impaired. The findings aim to provide new perspectives and scientific evidence for the management and prevention of dry eye as a chronic condition, particularly identifying the blink frequency threshold, which could offer scientific support for the development of a new generation of eye protection features in electronic products.
Methods
This study is a cross-sectional observational study conducted in accordance with the principles of the Helsinki Declaration and has been approved by the Ethics Committee of the Fudan University Eye, Ear, Nose, and Throat Hospital (Approval No: 2024016).
Participants
Participants were recruited through the internet and posters posted in nearby hospitals, schools, and communities. At enrollment, participants were required to meet the following criteria: (1) aged between 14 and 35 years, (2) presence of dry eye symptoms defined in Ocular Surface Disease Index (OSDI) with a total OSDI score ≥ 13, (3) the absence of significant corneal damage, which is defined as a corneal fluorescein staining score < 1 under slit-lamp examination. According to the diagnostic criteria put forward by TFOS DEWS III12,13,14, these subjects were symptomatic but could not be diagnosed as dry eye patients. The age range was selected based on the definition of youth in "Chinese Medium and Long-Term Youth Development Plan (2016–2025)," which specifies the age range of 14–35 years. Participants were excluded if they had: (1) a history of eye surgery or trauma, (2) a history of ocular medication use, (3) any ocular disease (including ocular allergies), (4) autoimmune diseases (such as Parkinson’s disease, diabetes), (5) use of medications that could affect blinking, or (6) were pregnant. Prior to the experiment, all participants were fully informed, provided written informed consent, and were instructed not to wear contact lenses or makeup on the day of the experiment.
The sample size was determined based on previous studies with similar designs investigating blink dynamics and tear film stability, which typically included 30–50 participants15,16. Considering comparable methodologies and populations, 45 participants were recruited in this study to ensure adequate statistical power.
Procedure
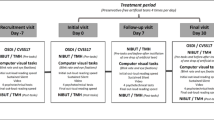
The study workflow is shown in Fig. 1.
This figure illustrates the two-phase methodology employed in our study, as detailed in the Methods section. In phase 1, spontaneous blinking parameters were measured using eye-tracking glasses while participants freely conversed with researchers. Blink frequency and intervals were calculated, and participants completed a demographic questionnaire and underwent dominant eye determination. In phase 2, three experimental blinking conditions were tested: spontaneous blinking (Sp group), instructed blinking at 10 times per minute (10 group), and instructed blinking at 20 times per minute (20 group). Blinking in the instructed groups was controlled via auditory cues, and the order of conditions was randomized to minimize bias. Each experimental condition lasted 30 min, with a 30-min washout period between sessions. Monitoring was conducted in real time using an eye tracker and direct observation by researchers. Primary ocular surface parameters were measured immediately after each condition to compare the effects of blinking frequencies in participants with dry eye symptoms.
In phase one, spontaneous blinking parameters were measured with eye-tracking glasses while participants freely have a talk with researchers. Blink frequency and interval were calculated using customized software, and participants completed a demographic questionnaire and dominant eye determination. All assessments were binocular. In phase two, three conditions were tested: spontaneous blinking (Sp group), 10 blinks per minute (10 group), and 20 blinks per minute (20 group), with auditory cues to control blinking. Each condition lasted 30 min, with a 30-min washout period between them. Monitoring was done via real-time eye tracking and researcher observation. Participants underwent all conditions in a randomized order, and primary ocular surface parameters were measured after each condition to compare the effects of different blink frequencies.
Each ocular surface parameter was measured three times per eye within the same session. Between non-invasive image acquisitions, participants were instructed to blink naturally for a few seconds (3–5 s) to re-establish tear film stability before the next acquisition. On the day of non-contact ocular examinations, no fluorescein or invasive procedures were performed, and they were conducted on another day to avoid the potential interference of fluorescein on the tear film dynamics17,18.
Measurement of blinking
Spontaneous blinking parameters, including spontaneous blink frequency and interval, were measured using an eye tracking device (aSee Glasses Scientific Edition, 7invensun) Before each use, a three-point calibration was performed, with successful calibration defined as achieving a score of 100 for both eyes. The eyes’positions in the eye-tracking display had to be vertically centered, with a ring of light points visible around both pupils. For consistent measurements, the position of the nose clip was fixed for each participant; otherwise, recalibration was required. During data collection, participants avoided direct exposure to sunlight or bright lights. Participants were seated on adjustable chairs with armrests and headrests for stability. The eye-tracking glasses recorded data at a resolution of 1920 × 1080@30fps, with a sampling rate of 240 Hz and a latency of < 10 ms (Fig. 2). The aSee Studio eye-tracking software detected blinks based on changes in eyelid closure, pupil occlusion, and the transitions in preceding and subsequent data points.
The eyeglass-mounted eye-tracking device (aSee Glasses Scientific Edition, 7invensun) features: (A) high-speed eye cameras integrated into the glasses and (B) the eye camera view, displaying the wearer’s eye (bottom right). The device is connected to a laptop for real-time monitoring, as shown in the figure, which depicts the workstation setup for the eyeglass-mounted eye-tracking device. The individual shown is a research staff member who provided written informed consent for publication of the identifiable image.
Tear film function
Dry eye questionnaire
Two questionnaires were used in this study. In the first phase of patient enrollment and baseline data collection, we used the OSDI questionnaire to classify dry eye symptoms into different severity levels. In the second phase, to more accurately reflect the participants’ subjective sensations, we selected the simpler and more targeted SANDE questionnaire to assess the impact of different blink frequencies on their subjective feelings.
The OSDI score is calculated as (sum of all scores * 100) / (total number of items * 4), with a score range from 0 to 100. A score of 0–12 indicates no dry eye symptoms, while 13–22, 23–32, and 33–100 refer to mild, moderate and severe dry eye symptoms, respectively.
The Standard Patient Evaluation of Eye Dryness (SANDE) questionnaire was used, which consisted of two questions:"How often do your eyes feel dry and/or irritated?"and “How severe do you feel your symptoms of dryness and/or irritation are?”Both questions are assessed using a 100-mm horizontal Visual Analog Scale (VAS). For each patient, they mark their perceived symptom frequency and severity on the two lines, and the marked positions are measured in millimeters. In the data analysis, the frequency and severity scores of dry eye symptoms were separately calculated and compared to assess changes in different dimensions of the symptoms19,20,21.
Tear film evaluation
Oculus Keratograph (5 M, Germany) was used to evaluate the parameters of tear film function including BUT, TMH, BR, tear film lipid layer and meibomian dropout score. BUT, TMH, BR were selected as primary dry eye parameters in this study.
Before capturing the tear meniscus images, all subjects were instructed to blink normally. Then TMH measurement was performed at the 6 O’ clock position between the cornea and the lower eyelid, The TMH was measured as the distance from the darker edge of the lower eyelid to the upper border of the tear meniscus reflex line22. BUT which was composed of the first break-up time (FBUT) and average break-up time (ABUT) was measured to record the time (in seconds) from a blink to the appearance of irregularities in the tear film. Shorter times indicate greater tear film instability. The conjunctival hyperemia was automatically analyzed using the R-scan function of the ocular surface analyzer. The focus was set on the corneal surface, with the diffuse grid overlaying the cornea. Subjects were instructed to keep their eyes wide open (scanning area ≥ 4 mm2). After the scan, the bulbar redness index was automatically generated.
The quality of tear film lipid layer was classified into four categories based on its color appearance: multicolored, red-green, blue-gray, and gray-white, with the sequence indicating a gradual decline in lipid layer quality. Typically, a normal lipid layer exhibits a rich multicolored appearance with slow movement. In contrast, a thinner lipid layer shows progressively weaker colors, transitioning from red-green to blue-gray, and ultimately to gray-white, accompanied by significantly increased flow23.
Meibomian gland dropout was evaluated along with an assessment of gland morphology. The scoring criteria for gland loss were as follows: no loss scored 0; loss proportion < 1/3 was classified as mild loss, scoring 1; loss proportion 1/3–2/3 was classified as moderate loss, scoring 2; and loss proportion > 2/3 was classified as severe loss, scoring 3. The morphology of the meibomian glands was categorized into four types: normal, tortuous, scanty, and dilated24,25.
The Schirmer I test measures the length of wetting of paper strips placed in the inferior cul de sac of the lower eyelid in mm/5 min, with shorter lengths indicating less tear production26,27,28.
Corneal fluorescein staining score is commonly assessed under slitlamp biomicroscopy with the assistance of fluorescein staining. The cornea is divided into four quadrants (upper, lower, nasal, and temporal) and a 12-point scale was used, with each quadrant scored from 0 to 3. A score of 0 indicates no staining; a score of 1 is given for 1 to 30 punctate stains; a score of 2 is given for more than 30 punctate stains that have not fused; and a score of 3 is given when punctate staining merges, with the presence of filaments or ulcers29.
Data analysis
Statistical analysis was performed using GraphPad Prism (version 10.1.1 270). The Kolmogorov–Smirnov test, histograms, and Q–Q plots were used to assess the normality of the data. Results are presented as mean ± standard deviation. Spearman’s rank correlation coefficient (ρ) was applied for non-normally distributed data. P values were adjusted for multiple comparisons using the Bonferroni correction. For normally distributed data, comparisons between three subgroups were conducted using a one-way ANOVA, while the Friedman test was used for non-normally distributed data. All tests were two-tailed, with significance set at p < 0.05.
Results
A total of 45 individuals were enrolled in the study, with an average age of 23.64 ± 2.27 (19–28) years old. The demographic information of the participants is shown in Table 1.
Phase 1 Spontaneous blink measurement
The data for spontaneous blink parameters (blink rate, blink interval), dry eye symptoms, tear film, and meibomian gland parameters measured in the first phase are shown in Table 2. The average blink rate of the participants was 27.75 ± 14.43 (blinks/min), and the blink interval was 3.04 ± 2.64 (s). There was a significant correlation between blink rate and TMH(Fig. 3) (r = 0.4785; P = 0.0009), indicating that higher blink rates were associated with higher TMH. There was no correlation between blink rate and gender, age, or BUT.
Associations between tear meniscus height (Oculus Keratograph V.5) and spontaneous blink frequency.
Phase 2 Assess the impact of blink frequency
Impact of blink frequency on ocular surface parameters in both eyes and dominant eye
TMH comparison
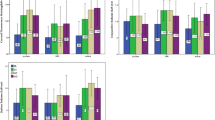
The TMH in both eyes of the Sp group was significantly higher than that of the 10 group (P = 0.0002; P = 0.0024) (Table 3, Fig. 4). In the comparison of the dominant eye TMH, the Sp group and the 20 group had significantly higher TMH than the 10 group, with notable differences (P = 0.0080; P = 0.0027) (Table 3, Fig. 4). No significant difference was observed between the Sp group and the 20 group.
Comparison of (A) TMH, (B) FBUT, (C) ABUT, (D) BR, (E) SANDE frequency, (F) SANDE severity, (G) dominant-eye FBUT, (H) dominant-eye ABUT, (I) dominant-eye TMH, and (J) dominant-eye BR among the Sp, 10, and 20 groups in both eyes and the dominant eye.
FBUT comparison
The FBUT in both eyes of the Sp group and the 20 group was significantly higher than that of the 10 group (P < 0.0001; P = 0.0139) (Table 3, Fig. 4). In the comparison of the dominant eye FBUT, the Sp group had significantly higher FBUT than the 10 group (P = 0.0095), with no difference observed between the Sp group and the 20 group (Table 3, Fig. 4).
ABUT comparison
The ABUT in both eyes of the Sp group and the 20 group was significantly higher than that of the 10 group (P < 0.0001; P = 0.0219) (Table 3, Fig. 4). In the analysis of the dominant eye, the Sp group had significantly higher ABUT than the 10 group (P < 0.0001) (Table 3, Fig. 4).
BR comparison
The BR score in both eyes of the Sp group and the 20 group was significantly lower than that of the 10 group (P < 0.0001; P = 0.0079) (Table 3, Fig. 4). In the analysis of the dominant eye, the Sp group had a significantly lower BR score than the 10 group (P = 0.0002) (Table 3, Fig. 4). There was no significant difference between the Sp group and the 20 group.
SANDE score comparison
Comparing the SANDE dry eye symptom frequency and severity scores, both the Sp group and the 20 group had significantly lower frequency and severity scores in both eyes compared to the 10 group (frequency: P < 0.0001; P = 0.0005; severity: P < 0.0001; P = 0.0015) (Table 3, Fig. 4). There was no significant difference in dry eye frequency and severity between the Sp group and the 20 group.
Impact of blink frequency on ocular surface parameters in participants with different levels of dry eye symptoms
In participants with mild dry eye symptoms, TMH, ABUT, BR, and SANDE frequency and severity scores were significantly higher in the spontaneous (Sp) group than those in the 10 blinks/min group (P = 0.0356, P < 0.0001, P = 0.0006, and P < 0.0001, respectively) (Table 4, Fig. 5). In participants with moderate symptoms, ABUT was significantly higher in the Sp group than in the 10 blinks/min group (P = 0.0030) (Table 4, Fig. 5), while the other parameters did not present significant differences. In participants with severe symptoms, TMH, FBUT, ABUT, and BR were significantly greater in the Sp group compared to both the 10 and 20 blinks/min groups (vs 10 group: P = 0.0008, P = 0.0068, P < 0.0001, and P < 0.0001; vs 20 group: P = 0.0296, P = 0.0047, P = 0.0004, and P = 0.0137). SANDE frequency and severity scores were significantly lower in the Sp group compared to the 10 blinks/min group (p = 0.0021 and p = 0.0036) (Table 4, Fig. 5).
Comparison of (A) Mild-eye TMH, (B) Mild-eye FBUT, (C) Mild-eye ABUT, (D) Mild-eye BR, (E) Mild-eye SANDE frequency, (F) Mild-eye SANDE severity, (G) Moderate-eye TMH, (H) Moderate-eye FBUT, (I) Moderate-eye ABUT, (J) Moderate-eye BR, (K) Moderate-eye SANDE frequency, (L) Moderate-eye SANDE severity, (M) Severe-eye TMH, (N) Severe-eye FBUT, (O) Severe-eye ABUT, (P) Severe-eye BR, (Q) Severe-eye SANDE frequency, and (R) Severe-eye SANDE severity among the Sp, 10, and 20 groups across different levels of dry eye symptom severity.
Discussion
Our study is the first to examine the impact of different blinking frequencies on ocular surface parameters in young individuals with dry eye symptoms. Unlike previous studies in which direct observation or video recording methods that require stable head positions were used30,31,32,33,34,35, the current study employed a wearable eye-tracking device that allowed free head and eye movement, enabling more accurate blink measurements. The average spontaneous blink frequency of participants with dry eye symptoms was 27.44 blinks/min in our study, which was within the range reported previously36.
Although previous studies did not show significant correlations between blink frequency or blink interval and ocular surface symptoms or signs37,38,39, our findings revealed that a higher blink frequency (20 blinks/min) was beneficial to maintain tear film stability, being consistent with previous studies40. Further analysis revealed that increasing blink frequency from 10 blinks/min to 20 blinks/min helped reduce ocular discomfort caused by dry eye and stabilize tear function. Previous study have shown that higher blink rates are associated with increased tear volume and tear meniscus height41. Our study suggested that within the normal range of blink frequency, appropriately increasing the blink rate can help maintain tear volume and distribution. Digital terminal users, the majority of whom were young people, are at higher risk of reduced spontaneous blink frequency and compromised tear film stability, highlighting the need to manage blink frequency to prevent the worsening of dry eye symptoms42.
Our study also identified a potential “threshold effect” of blink frequency on dry eye symptoms. Using spontaneous blink frequency as the baseline, when the blink frequency was below 20 blinks/min, tear film parameters showed significant abnormalities, particularly in TMH, BUT, and BR. This suggests that low-frequency blinking leads to insufficient tear film renewal, exacerbating tear evaporation and instability. In contrast, when the blink frequency reached 20 blinks/min, dry eye symptoms were significantly alleviated, especially in TMH and the frequency and severity of dry eye episodes. This indicates that 20 blinks/min is a critical blink frequency threshold for improving dry eye symptoms. Below this frequency, tear film stability is compromised, and further increases beyond 20 blinks/min do not significantly enhance symptom improvement. These findings provide new insights into blink frequency management in individuals with dry eye symptoms and offer practical guidance for preventing and alleviating ocular surface discomfort.
When comparing participants with different levels of dry eye symptoms, forced low-frequency blinking(10 blinks per minute) led to a decrease in the BUT across all severity levels, particularly in mild and severe cases. However, increasing blink frequency within the normal range significantly improved TMH, especially among participants with mild and severe dry eye symptoms. This increase in blink frequency helps to distribute tears more evenly, reducing evaporation and dryness, which can alleviate symptoms in mild cases and enhance tear distribution in severe cases. Additionally, forced low-frequency blinking(10 blinks per minute) significantly raised SANDE scores, suggesting that a reduced blink frequency worsens the frequency and severity of dry eye symptoms. These findings emphasize the importance of increasing blink frequency as a non-invasive strategy for managing ocular surface discomfort. While parameters like BR showed no significant differences, the observed trends indicate that larger sample sizes or longer intervention periods might yield more pronounced effects. Overall, these results highlight the significance of blink frequency in daily life, particularly for individuals with dry eye symptoms associated with prolonged screen use.
This study has several limitations. First, the small sample size limits the generalizability of the findings, so future research should include larger samples. Second, although participants showed improvements in tear function, the SANDE questionnaire did not reveal a significant correlation between increased blink frequency and improved dry eye symptoms, suggesting a discrepancy between subjective assessments and objective measures. This underscores the need for comprehensive evaluation of both clinical indicators and patient-reported outcomes in clinical practice. Third, participants were aware that their blinking behavior was being monitored in the current study, which might have led to conscious control and alterations in natural blinking patterns. The use of auditory cues to regulate blinking frequency may also differ from spontaneous blinking dynamics, potentially influencing the results. These factors could have affected the findings and should be considered when interpreting the data. Future studies should employ less noticeable monitoring techniques to minimize behavioral bias, as suggested by Acet (2024), who proposed non-noticeable methods for assessing blink dynamics43. Finally, the study was conducted under controlled laboratory conditions, which may not fully imitate real-life environments influenced by factors such as humidity, air quality, and screen distance. Future research should explore the effects of blink frequency and tear film stability under more natural and ecologically valid conditions.
Conclusion
This study examined the impact of blink frequency on ocular surface parameters in participants with dry eye symptoms and identified a potential “blink threshold” for maintaining tear film stability. The results show that increasing blink frequency significantly improves tear meniscus height and tear film stability, highlighting the role of blinking in maintaining tear film function and alleviating dry eye symptoms. Future research with a larger sample size is needed to explore the relationship between blinking behavior and dry eye symptoms. Moderately increasing blink frequency may be an effective strategy for improving tear film health in individuals with dry eye symptoms, supporting the development of eye protection strategies.
Data availability
The data that support the findings of this study may be requested from the corresponding author.
References
Committee of Cross-Straits Medicine Exchange Association, Dry Eye Group of Chinese Ophthalmologist Association. [Chinese expert consensus on meibomian gland dysfunction: Definition and classification]. Zhonghua Yan Ke Za Zhi 59(4), 256–261. https://doi.org/10.3760/cma.j.cn112142-20230114-00023 (2023).
Stapleton, F., Velez, F. G., Lau, C. & Wolffsohn, J. S. Dry eye disease in the young: A narrative review. Ocul Surf 31, 11–20. https://doi.org/10.1016/j.jtos.2023.12.001 (2024).
Messmer, E. M. The pathophysiology, diagnosis, and treatment of dry eye disease. Dtsch Arztebl Int. 112(5), 71–81. https://doi.org/10.3238/arztebl.2015.0071 (2015).
Li, X. et al. Prevalence and associated risk factors of dry eye disease in Hotan, Xinjiang: A cross-sectional study. BMC Ophthalmol. 23(1), 214. https://doi.org/10.1186/s12886-023-02955-9 (2023).
Zhang, H. et al. Epidemiological characteristics of dry eye disease in asian and asian female populations: A database-driven descriptive study. J. Curr. Ophthalmol. 36(2), 159–167. https://doi.org/10.4103/joco.joco_46_24 (2024).
Stapleton, F. et al. TFOS DEWS II epidemiology report. Ocul Surf. 15(3), 334–365. https://doi.org/10.1016/j.jtos.2017.05.003 (2017).
Muntz, A. et al. Extended screen time and dry eye in youth. Cont. Lens Anterior Eye 45(5), 101541. https://doi.org/10.1016/j.clae.2021.101541 (2022).
Fjaervoll, K. et al. Review on the possible pathophysiological mechanisms underlying visual display terminal-associated dry eye disease. Acta Ophthalmol. 100(8), 861–877. https://doi.org/10.1111/aos.15150 (2022).
Pierce, C. W. Dry Eye. New Engl. J. Med. 379(11), e19. https://doi.org/10.1056/NEJMc1808906 (2018).
Zhou, N. et al. Enterovirus A71 infection-induced dry eye-like symptoms by damaging the lacrimal glands. Front. Cell Infect Microbiol. 14, 1340075. https://doi.org/10.3389/fcimb.2024.1340075 (2024).
Zhao, J. et al. Anterior segment inflammation and its association with dry eye parameters following myopic SMILE and FS-LASIK. Ann. Med. 55(1), 689–695. https://doi.org/10.1080/07853890.2023.2181388 (2023).
Perez, V. L. et al. TFOS DEWS III: Executive summary. Am. J. Ophthalmol. https://doi.org/10.1016/j.ajo.2025.09.035 (2025).
Stapleton, F. et al. TFOS DEWS III: Digest. Am. J. Ophthalmol. 279, 451–553. https://doi.org/10.1016/j.ajo.2025.05.040 (2025).
Wolffsohn, J. S. et al. TFOS DEWS III: Diagnostic methodology. Am. J. Ophthalmol. 279, 387–450. https://doi.org/10.1016/j.ajo.2025.05.033 (2025).
Chidi-Egboka, N. C., Jalbert, I., Wagner, P. & Golebiowski, B. Blinking and normal ocular surface in school-aged children and the effects of age and screen time. Br. J. Ophthalmol. 107(11), 1613–1620. https://doi.org/10.1136/bjo-2022-321645 (2023).
Himebaugh, N. L., Begley, C. G., Bradley, A. & Wilkinson, J. A. Blinking and tear break-up during four visual tasks. Optom. Vis Sci. 86(2), E106–E114. https://doi.org/10.1097/OPX.0b013e318194e962 (2009).
Acet, Y. & Dağ, Y. Changes caused by fluorescein in the tear film evaluated with hybrid break-up time test as a new method - Part Two: Its effect on breakup locations and other quantitative values. Photodiagn. Photodyn. Ther. 43, 103651. https://doi.org/10.1016/j.pdpdt.2023.103651 (2023).
Hwang, H. B. et al. Easy and effective test to evaluate tear-film stability for self-diagnosis of dry eye syndrome: Blinking tolerance time (BTT). BMC Ophthalmol. 20(1), 438. https://doi.org/10.1186/s12886-020-01686-5 (2020).
Gulati, A. et al. Validation and repeatability of a short questionnaire for dry eye syndrome. Am J Ophthalmol. 142(1), 125–131. https://doi.org/10.1016/j.ajo.2006.02.038 (2006).
Schaumberg, D. A. et al. Development and validation of a short global dry eye symptom index. Ocul Surf. 5(1), 50–57. https://doi.org/10.1016/s1542-0124(12)70053-8 (2007).
Amparo, F., Schaumberg, D. A. & Dana, R. Comparison of two questionnaires for dry eye symptom assessment: The ocular surface disease index and the symptom assessment in dry eye. Ophthalmology 122(7), 1498–1503. https://doi.org/10.1016/j.ophtha.2015.02.037 (2015).
Chen, M., Wei, A., Xu, J., Zhou, X. & Hong, J. Application of keratograph and fourier-domain optical coherence tomography in measurements of tear meniscus height. J Clin Med 11(5), 1343. https://doi.org/10.3390/jcm11051343 (2022).
Gokul, A., Wang, M. T. M. & Craig, J. P. Tear lipid supplement prophylaxis against dry eye in adverse environments. Cont. Lens Anterior Eye. 41(1), 97–100. https://doi.org/10.1016/j.clae.2017.09.013 (2018).
Feng, Y., Gao, Z., Feng, K., Qu, H. & Hong, J. Meibomian gland dropout in patients with dry eye disease in China. Curr Eye Res. 39(10), 965–972. https://doi.org/10.3109/02713683.2014.891748 (2014).
Kiyat, P., Palamar, M., Gerceker Turk, B. & Yagci, A. Evaluation of dry eye and Meibomian gland dysfunction in female androgenetic alopecia patients. Int Ophthalmol. 41(11), 3749–3757. https://doi.org/10.1007/s10792-021-01946-3 (2021).
Bitton, E. & Wittich, W. Influence of eye position on the Schirmer tear test. Cont Lens Anterior Eye. 37(4), 257–261. https://doi.org/10.1016/j.clae.2013.11.011 (2014).
Wang, X. et al. Rear 4-min Schirmer test, a modified indicator of Schirmer test in diagnosing dry eye. Sci Rep. 12(1), 6272. https://doi.org/10.1038/s41598-022-09791-9 (2022).
Yu, K., Bunya, V., Maguire, M., Asbell, P. & Ying, G. S. Systemic conditions associated with severity of dry eye signs and symptoms in the dry eye assessment and management study. Ophthalmology 128(10), 1384–1392. https://doi.org/10.1016/j.ophtha.2021.03.030 (2021).
Committee of Cross-Straits Medicine Exchange Association, Dry Eye Group of Chinese Ophthalmologist Association. [Chinese expert consensus on meibomian gland dysfunction: Diagnosis and management 2023]. Zhonghua Yan Ke Za Zhi 59(11), 880–887. https://doi.org/10.3760/cma.j.cn112142-20230822-00054 (2023).
Zametkin, A. J., Stevens, J. R. & Pittman, R. Ontogeny of spontaneous blinking and of habituation of the blink reflex. Ann Neurol. 5(5), 453–457. https://doi.org/10.1002/ana.410050509 (1979).
Doughty, M. J. Consideration of three types of spontaneous eyeblink activity in normal humans: During reading and video display terminal use, in primary gaze, and while in conversation. Optom Vis Sci. 78(10), 712–725. https://doi.org/10.1097/00006324-200110000-00011 (2001).
Kim, J. S., Wang, M. T. M. & Craig, J. P. Exploring the Asian ethnic predisposition to dry eye disease in a pediatric population. Ocul Surf. 17(1), 70–77. https://doi.org/10.1016/j.jtos.2018.09.003 (2019).
Bentivoglio, A. R. et al. Analysis of blink rate patterns in normal subjects. Mov Disord. 12(6), 1028–1034. https://doi.org/10.1002/mds.870120629 (1997).
Caplan, R., Guthrie, D. & Komo, S. Blink rate in children with attention-deficit-hyperactivity disorder. Biol Psychiatry. 39(12), 1032–1038. https://doi.org/10.1016/0006-3223(95)00315-0 (1996).
Salman, M. S. & Liu, L. Spontaneous blink rates in children during different types of eye movements. Can J Neurol Sci. 40(5), 717–721. https://doi.org/10.1017/s0317167100014979 (2013).
Doughty, M. J. Consideration of three types of spontaneous eyeblink activity in normal humans: During reading and video display terminal use, in primary gaze, and while in conversation. Optom Vis Sci. 78(10), 712–725 (2001).
Golebiowski, B., Long, J., Harrison, K., Lee, A. & Chidi-Egboka, N. Asper L 2020 Smartphone Use and Effects on Tear Film, Blinking and Binocular Vision. Curr Eye Res. 45(4), 428–434. https://doi.org/10.1080/02713683.2019.1663542 (2020).
Chu, C. A., Rosenfield, M. & Portello, J. K. Blink patterns: Reading from a computer screen versus hard copy. Optom Vis Sci. 91(3), 297–302. https://doi.org/10.1097/opx.0000000000000157 (2014).
Chidi-Egboka, N. C., Jalbert, I., Chen, J., Briggs, N. E. & Golebiowski, B. Blink rate measured in situ decreases while reading from printed text or digital devices, regardless of task duration, difficulty, or viewing distance. Invest Ophthalmol Vis Sci. 64(2), 14. https://doi.org/10.1167/iovs.64.2.14 (2023).
Himebaugh, N. L., Begley, C. G., Bradley, A. & Wilkinson, J. A. Blinking and tear break-up during four visual tasks. Optom. Vis. Sci. 86(2), E106–E114. https://doi.org/10.1097/OPX.0b013e318194e962 (2009).
Palakuru, J. R., Wang, J. & Aquavella, J. V. Effect of blinking on tear dynamics. Invest Ophthalmol Vis Sci. 48(7), 3032–3037. https://doi.org/10.1167/iovs.06-1507 (2007).
Jaiswal, S. et al. Ocular and visual discomfort associated with smartphones, tablets and computers: What we do and do not know. Clin Exp Optom. 102(5), 463–477. https://doi.org/10.1111/cxo.12851 (2019).
Acet, Y. Assessing blink dynamics with non-noticeable method. Cont Lens Anterior Eye. 47(6), 102277. https://doi.org/10.1016/j.clae.2024.102277 (2024).
Acknowledgements
The authors express their gratitude to the ophthalmology team of the Eye & ENT Hospital of Fudan University for providing clinical facilities and venue support. Special thanks go to Pei Yang and Shuyun Zhou from the Dry Eye Center for their technical assistance. The authors sincerely appreciate the support from Professor Jiaxu Hong’s research group, including faculty members and fellow researchers, throughout various stages of this study. They also extend their appreciation to the research team at the Chinese Academy of Medical Sciences and Peking Union Medical College, with special thanks to Professor Dingguan and colleagues for their assistance with variance analysis. Additionally, the authors acknowledge the support from China Glory Terminal Co., Ltd. in terms of manpower and funding. Finally, they are grateful for the financial support provided by various funding agencies.
Funding
This work was supported by the National Science Fund for Distinguished Young Scholars (Grant No. 82425015), the National Natural Science Foundation of China (Grant No. 82171102, 82471042), the National Key Research and Development Program of China (Grant No. 2023YFA0915000), the “Dawn” Program of Shanghai Municipal Education Commission (Grant No. 24SG11), the Shanghai Science and Technology Innovation Action Plan for Cell and Gene Therapy (Grant No. 24J22800500), the Shanghai Science and Technology Innovation Action Plan for Advanced Materials (Grant No. 24CL2900802), the Shanghai Municipal Commission of Health (Grant No. 20254Z0019), the Shanghai High-Quality Development Plan for Science and Technology Industry (Grant No. 25SF1907000), and the National Natural Science Foundation of Shanghai Science and Technology Committee (24ZR1409600)
Author information
Authors and Affiliations
Contributions
Yawei Chai: Responsible for experiment design and execution, data collection and analysis, drafting the initial manuscript, and revising the manuscript. Mingrui Cheng: Participated in experiment design and execution, responsible for data collection, processing, and analysis, and contributed to manuscript revision. Xinhua Liu: Participated in experiment execution and data collection, and contributed to manuscript revision. Qihua Le: Supervised the experiment, participated in data analysis, and was responsible for revising and optimizing the figures and tables in the manuscript. Jiaxu Hong: Responsible for the overall planning, design, and implementation of the study, supervising and coordinating the research process, ensuring the integrity and accuracy of the study, revising the manuscript, and providing financial and technical support. Xingyu Zhu, Yiteng Lu, and Yuqing Wu: Provided suggestions for revising the figures and tables in the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval
This study involved human participants and was approved by the Ethics Committee of Fudan University Eye & ENT Hospital (Approval No.: 2024016). Informed consent was obtained from all participants before their participation in the study.
Consent for publication
Written informed consent for publication of the identifiable image of the research staff member appearing in Fig. 2 was obtained.
Informed consent
Consent obtained directly from patient(s).
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Chai, Y., Cheng, M., Liu, X. et al. Identification of a blink frequency threshold for maintaining tear film stability in young adults with dry eye symptoms. Sci Rep 15, 42321 (2025). https://doi.org/10.1038/s41598-025-26424-z
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-26424-z