Abstract
This study aimed to compare the safety and efficacy of two target-controlled infusion (TCI) concentrations of propofol for sedation during cranial functional magnetic resonance imaging (fMRI) in children with autism spectrum disorder (ASD). A total of 120 preschool children with ASD scheduled for fMRI between January 2021 and July 2023 were enrolled and randomized into two groups: a low-concentration group (2.0 µg/mL, n = 60) and a high-concentration group (2.5 µg/mL, n = 60). Vital signs, sedation parameters, and adverse events were meticulously recorded to assess outcomes. The first-time sedation success rate was high and comparable between the 2.0 group and the 2.5 group (91.67% vs. 96.67%, p = 0.439). However, the 2.5 group demonstrated a significant reduction in mean systolic blood pressure (p = 0.001). While the overall incidence of adverse events was not significantly different, respiratory and circulatory depression events were less frequent in the 2.0 group. Furthermore, the 2.0 group required a significantly lower maintenance dose of propofol (p < 0.001), experienced a shorter recovery time (p = 0.042), and had a smoother emergence profile. In conclusion, a propofol TCI concentration of 2.0 µg/mL is effective and safe for fMRI sedation in children with ASD and is clinically preferable to 2.5 µg/mL due to a more stable hemodynamic profile, reduced drug requirement, and faster, smoother recovery.
Trial registration: ChiCTR2100050071.
Similar content being viewed by others
Introduction
Autism spectrum disorder (ASD), referred to as autism in this study, is a complex neurodevelopmental condition that typically manifests in early childhood. Globally, the prevalence of autism is approximately 0.76%, with reported rates in China ranging between 0.3% and 1%1,2. Autism is characterized by deficits in social communication, restricted interests, repetitive behaviors, and various challenges affecting learning, communication, and daily life3. Despite extensive research, the exact etiology of autism remains unclear, although it is widely believed to involve a combination of genetic, environmental, and neurobiological factors4.
Functional magnetic resonance imaging (fMRI) has become an invaluable tool for exploring the neural mechanisms underlying autism-related functional abnormalities5. However, performing fMRI in children with autism presents unique challenges. These children often face heightened anxiety, sensory sensitivities, and difficulty following behavioral instructions, such as remaining still during scanning6. Movement during imaging can compromise data quality, making sedation or anesthesia necessary to ensure successful examinations. While sedation can help mitigate these challenges, existing research on sedation in children with autism is limited, primarily consisting of case reports and small observational studies7,8,9. This lack of standardized guidelines underscores the need for evidence-based protocols tailored to this population.
Propofol, a widely used anesthetic agent, is favored for its rapid onset, predictable recovery profile, and suitability for target-controlled infusion (TCI)10. However, the optimal propofol concentration for achieving effective sedation with minimal risks in children with autism undergoing fMRI remains unclear. To address this gap, our study evaluated the safety, efficacy, and hemodynamic stability of propofol-induced intravenous deep sedation in 120 preschool children with autism. By comparing different TCI concentration levels, we aimed to provide insights into the best sedation practices for this vulnerable population, contributing to improved management strategies in pediatric neuroimaging.
Materials and methods
Ethical considerations
The study protocol received approval from the Ethics Committee of the Children’s Hospital of Chongqing Medical University (IRB #139/2021, approved on April 27, 2021) and was prospectively registered in the Chinese Clinical Trial Registry (Registration No.: ChiCTR2100050071, registered on August 16, 2021). Enrollment of the first participant began on September 6, 2021. Conducted at the Children’s Hospital of Chongqing Medical University, the study adhered to the ethical standards outlined in the Helsinki Declaration. Written informed consent was obtained from the parents or legal guardians of all patients before enrollment.
Subjects
From September 2021 to July 2023, we enrolled children with ASD from our hospital’s neurology and pediatrics clinics who required fMRI due to language or behavioral issues. Inclusion criteria were a diagnosis of autism according to the Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition (DSM-5), age between 2 and 6 years, right-handedness, no significant findings on routine MRI, and American Society of Anesthesiologists (ASA) physical status I–II. Exclusion criteria included ASD with fragile X syndrome, childhood disintegrative disorder, or Rett syndrome; significant head trauma or other neurological conditions; and lack of parental consent.
Anesthesia management and data collection
All children were required to fast for 6–8 h and refrain from drinking water for 2 h before the examination. Upon arrival at the observation room, two attending anesthesiologists established a peripheral venous access and collected baseline information (gender, age, weight, body mass index (BMI), ASA classification). Prior to the examination, one anesthesiologist administered an intravenous induction of propofol at a dose of 3–4 mg/kg. The examination began when the bispectral index (BIS) reached 50–70 and the Ramsay sedation score (Table S1) was ≥ 5, with vital signs continuously monitored11,12. A target-controlled infusion pump (TCI, TCI-V, VERYARK, Guangxi) was placed outside the MRI room in the observation area, and the infusion was connected to the child via an extended infusion line. The child’s age, height, and weight were input into the system, and the Paedfusor model was used to set the target-controlled infusion of propofol to achieve an effect-site concentration. Children were divided into two groups based on the target effect-site concentration of propofol: the 2.0 µg/mL group (60 children) and the 2.5 µg/mL group (60 children).
During the examination, MR-compatible monitoring equipment was used to monitor oxygen saturation (SpO2), heart rate (HR), respiratory rate (RR), end-tidal carbon dioxide (ETCO2), mean systolic blood pressure (mSBP), and mean diastolic blood pressure (mDBP). ETCO2 was continuously monitored via a nasal cannula-type CO2 monitoring tube.2An ETCO2 level > 50 mmHg indicated hypercapnia, while RR falling 20% below baseline signaled respiratory depression. Upon observing these conditions, drug administration was immediately halted, and jaw-thrust maneuver was initiated to provide assisted ventilation. Vital signs, including SpO2, HR, RR, ETCO2, mSBP, mDBP, and Ramsay sedation scores, were recorded every 5 min. If any movement or a Ramsay sedation score below 5 occurred, an additional dose of propofol (0.5–1 mg/kg) was administered13. Another anesthesiologist was responsible for monitoring and recording the child’s vital signs, anesthesia time, examination duration, recovery time, maintenance dose of propofol, adverse reactions, and movement status. Following the examination, the child was transferred to the recovery area for emergence from anesthesia and was only discharged when the Sedation Agitation Scale (SAS, Table S2) score reached 3–4 points under parental supervision14.
Outcomes
The primary outcome measure of the study was the proportion of first successful sedation, defined as the ratio of cases in which no additional propofol was required during the examination to the total number of cases. Successful sedation was defined as obtaining high-quality MRI images after sedation. The secondary outcome measures included vital signs during the examination (HR, RR, mSBP, mDBP, SpO2, ETCO2), incidence of adverse events (tachycardia, bradycardia, hypotension, hypertension, hypoxemia, hypercapnia, respiratory depression), and sedation parameters (BIS, propofol dose, anesthesia duration, examination duration, recovery time, SAS Score, Ramsay Sedation Score).
Statistical analysis
Randomization was performed using computer-generated random number sequences with allocation concealment achieved through opaque, sealed envelopes. Participants were allocated in a 1:1 ratio to either the 2.0 µg/mL group or the 2.5 µg/mL group. Continuous variables are presented as mean ± standard deviation (SD) or median [IQR], while categorical variables are presented as numbers (%). The normality of the data was assessed using the Shapiro-Wilk test, and based on the results, either independent t-tests or Mann-Whitney U tests were used for analysis. Proportions were compared using chi-square tests or Fisher’s exact test. All p-values were two-sided, and p < 0.05 was considered statistically significant. All analyses were performed using R.
Results
Demographic characteristics
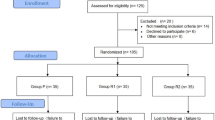
A total of 125 children with ASD were initially recruited for this study. Due to intracranial structural abnormalities in 4 children and refusal to participate by 1 family, 120 children were ultimately included, with the enrollment process shown in Fig. 1. Based on the TCI effect-site concentration settings, they were divided into two groups (2.0 group and 2.5 group), with 60 children in each group. There were no statistically significant differences between the two groups in terms of gender, age, weight, height, BMI, or ASA classification (Table 1).
Flow diagram of the inclusion process for children with autism spectrum disorder (ASD).
Primary outcome measures
Regarding the primary outcome, the first-time sedation success rate was 91.67% (n = 55) in the 2.0 group and 96.67% (n = 58) in the 2.5 group, with no statistically significant difference between the two groups (p = 0.439). The detailed results are presented in Table 2.
Secondary outcome measures
The mean systolic blood pressure in the 2.5 group was lower than that in the 2.0 group (2.0 group vs. 2.5 group; 96.11 [94.25,102] vs. 93.54 [87.81,96.92]; median [IQR]; p = 0.001). The trend charts of vital signs for both groups are shown in Fig. 2. There was no statistically significant difference in the incidence of adverse events between the two groups, although the frequency of respiratory and circulatory depression was lower in the 2.0 group. The 2.0 group demonstrated a higher mean BIS during sedation (59.92 ± 3.52 vs. 56.21 ± 3.58; mean ± SD; p < 0.001), required a lower maintenance dose of propofol (10.6 [9.39, 12.28] vs. 13.26 [12.07, 14.74]; median [IQR]; p < 0.001), had a shorter recovery time (22 [15.75, 25] vs. 24 [18, 31.25]; median [IQR]; p = 0.042), and experienced a smoother emergence profile. Detailed data are presented in Tables 2 and 3.
Trend chart of vital sign changes in children with autism spectrum disorder during functional magnetic resonance imaging (fMRI). (a) Heart Rate (HR); (b) Mean Arterial Pressure (MAP); (c) Respiratory Rate (RR); (d) Oxygen Saturation (SpO2); (e) End-Tidal Carbon Dioxide (ETCO2); (f) Bispectral Index (BIS).
Discussion
This study systematically compared the safety and efficacy of two different TCI concentrations of propofol for sedation during cranial fMRI in children with ASD. To the best of our knowledge, this is the first prospective study to directly compare effect-site concentrations of 2.0 µg/mL versus 2.5 µg/mL propofol for fMRI sedation in children with ASD.
Although fMRI offers a unique perspective for elucidating the neural mechanisms of ASD15,16, its application in the pediatric population is severely limited by challenges such as loud noise, prolonged scanning times, and the confined environment of the procedure6. Sedation is crucial for the successful completion of the examination; however, no standardized sedation protocol currently exists for this specific scenario. Previous research has indicated that sedative agents may interfere with the blood oxygen level-dependent (BOLD) signal through effects on neurovascular coupling, thereby constraining the image quality and interpretive validity of fMRI17. Consequently, exploring sedation strategies that simultaneously ensure high-quality imaging and patient safety is of paramount importance. Several prior studies provide a rationale for the use of propofol in this field: Bernal et al. demonstrated its position as the sedative of choice for pediatric fMRI17; Zhou et al. confirmed the efficacy and recovery advantages of its use, both alone and in combination with dexmedetomidine18; particularly importantly, Tang et al. found that while propofol suppresses responses to auditory stimuli in certain cortical areas, its impact on the primary auditory cortex is limited19, preserving the potential for detecting key neural activity signals in fMRI studies. Based on this evidence, this study focused on the refined adjustment of propofol TCI concentrations, aiming to provide an optimized sedation option for clinical practice.
In this study, we used propofol for induction and maintained sedation with TCI. TCI ensures stable drug concentrations at the effect site or in plasma, minimizing fluctuations and maintaining consistent sedation depth20. The effective TCI concentration required for deep sedation typically ranges from 2.0 to 3.0 µg/mL21. Based on this concentration range, the study further compared the clinical efficacy of two target concentrations (2.0 µg/mL vs. 2.5 µg/mL). Our findings indicate that both TCI concentrations of propofol provided effective and reliable sedation for fMRI. Objective monitoring via BIS confirmed a dose-dependent sedative effect, with the lower concentration group maintaining a significantly lighter yet adequate plane of sedation. This electrophysiological profile aligns with the superior clinical outcomes observed in the 2.0 µg/mL group, which included faster recovery times and a lower total propofol requirement. Furthermore, a trend toward a higher incidence of self-limiting respiratory events was noted at the higher concentration, reinforcing the dose-dependent nature of propofol’s side effects. This is consistent with the findings of Dueck et al.22, who reported dose-dependent attenuation of auditory cortex activation, highlighting propofol’s broader influence on central processing. Therefore, by integrating the BIS-correlated sedation depth with the enhanced recovery and safety profile, we recommend a TCI concentration of 2.0 µg/mL as the optimal balance for preserving BOLD signal integrity while minimizing patient risk.
In the 2.5 µg/mL group, one child with ASD exhibited emergence agitation during recovery (SAS score: 7), accompanied by aggressive behavior. Intranasal dexmedetomidine (3 µg/kg) was administered, leading to resolution without further complications. Martini noted that preschool children are at higher risk for emergence agitation, particularly in unfamiliar environments23. In our study, ASD children experienced heightened difficulty being separated from parents in such settings, increasing their susceptibility to agitation during recovery. Special attention should be given to this risk when sedating ASD children. Moreover, the sedation needs of ASD children may vary significantly due to the impact of ASD on the central nervous system. Elmorsy found that ASD may alter sedative drug requirements24. In our study, one child with a Childhood Autism Rating Scale (CARS) score of 36 (severe ASD) required a significantly higher propofol dose. CARS scores can be used to assess ASD severity and guide individualized sedation strategies25,26. Tailoring protocols based on ASD severity may improve sedation outcomes, enhance MRI success rates, and reduce the risk of adverse effects, such as respiratory depression (e.g., hypoxemia and hypercapnia).
Despite offering valuable insights, our study has limitations. The small sample size and single-center design may limit the generalizability of the findings. Future multicenter, randomized controlled trials are needed to validate our results. Additionally, while this study focused on propofol monotherapy, the sedation needs of ASD children may vary. Future research should explore the efficacy of combining propofol with other sedatives, particularly for mitigating emergence agitation and other potential adverse effects.
Conclusion
This is the first study to systematically compare two propofol target-controlled infusion concentrations (2.0 µg/mL vs. 2.5 µg/mL) for efficacy and safety during fMRI in children with autism spectrum disorder. By jointly assessing scan success, emergence time, drug consumption and adverse events, we demonstrate that the lower concentration achieves highly effective sedation while allowing faster recovery, lower doses and a better safety profile, providing direct evidence for individualized sedation in this special population.
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
References
Elsabbagh, M. et al. Global prevalence of autism and other pervasive developmental disorders. Autism Research: Official J. Int. Soc. Autism Res. 5 (3), 160–179 (2012).
Wang, F. et al. The prevalence of autism spectrum disorders in china: a comprehensive meta-analysis. Int. J. Biol. Sci. 14 (7), 717–725 (2018).
Sanchack, K. E. & Thomas, C. A. Autism spectrum disorder: primary care principles. Am. Family Phys. 94 (12), 972–979 (2016).
Hirota, T. & King, B. H. Autism Spectr. Disorder: Rev. JAMA, 329(2), 157–168 (2023).
Li, D., Karnath, H. O. & Xu, X. Candidate biomarkers in children with autism spectrum disorder: A review of MRI studies. Neurosci. Bull. 33 (2), 219–237 (2017).
Schulte-Uentrop, L. & Goepfert, M. S. Anaesthesia or sedation for MRI in children. Curr. Opin. Anaesthesiol. 23 (4), 513–517 (2010).
Abulebda, K., Louer, R., Lutfi, R. & Ahmed, S. S. A comparison of safety and efficacy of Dexmedetomidine and Propofol in children with autism and autism spectrum disorders undergoing magnetic resonance imaging. J. Autism Dev. Disord. 48 (9), 3127–3132 (2018).
Kamat, P. P. et al. Outpatient procedural sedation of patients with autism spectrum disorders for magnetic resonance imaging of the brain using Propofol. J. Child. Neurol. 33 (5), 313–319 (2018).
Lubisch, N., Roskos, R. & Berkenbosch, J. W. Dexmedetomidine for procedural sedation in children with autism and other behavior disorders. Pediatr. Neurol. 41 (2), 88–94 (2009).
Mu, J., Jiang, T., Xu, X. B., Yuen, V. M. & Irwin, M. G. Comparison of target-controlled infusion and manual infusion for Propofol anaesthesia in children. Br. J. Anaesth. 120 (5), 1049–1055 (2018).
Powers, K. S. et al. Bispectral index as a guide for Titration of Propofol during procedural sedation among children. Pediatrics 115 (6), 1666–1674 (2005).
Chen, S. C. et al. Application of bispectral index system (BIS) monitor to ambulatory pediatric dental patients under intravenous deep sedation. Diagnostics (Basel Switzerland). 13 (10), 1789 (2023).
Ramsay, M. A., Savege, T. M., Simpson, B. R. & Goodwin, R. Controlled sedation with alphaxalone-alphadolone. Br. Med. J. 2 (5920), 656–659 (1974).
Riker, R. R., Picard, J. T. & Fraser, G. L. Prospective evaluation of the Sedation-Agitation scale for adult critically ill patients. Crit. Care Med. 27 (7), 1325–1329 (1999).
Ismail, M. M. et al. Studying autism spectrum disorder with structural and diffusion magnetic resonance imaging: A survey. Front. Hum. Neurosci. 10, 211 (2016).
Rafiee, F., Habibabadi, R., Motaghi, R., Yousem, M., Yousem, I. J. & D. M., & Brain MRI in autism spectrum disorder: narrative review and recent advances. J. Magn. Reson. Imaging: JMRI. 55 (6), 1613–1624 (2022).
Bernal, B., Grossman, S., Gonzalez, R. & Altman, N. FMRI under sedation: what is the best choice in children? J. Clin. Med. Res. 4 (6), 363–370 (2012).
Zhou, Q., Shen, L., Zhang, X., Li, J. & Tang, Y. Dexmedetomidine versus Propofol on the sedation of pediatric patients during magnetic resonance imaging (MRI) scanning: a meta-analysis of current studies. Oncotarget 8 (60), 102468–102473 (2017).
Tang, C. Y. & Ramani, R. fMRI and anesthesia. Int. Anesthesiol. Clin. 54 (1), 129–142 (2016).
Struys, M. M. et al. The history of Target-Controlled infusion. Anesth. Analg. 122 (1), 56–69 (2016).
Anderson, B. J. & Bagshaw, O. Practicalities of total intravenous anesthesia and Target-controlled infusion in children. Anesthesiology 131 (1), 164–185 (2019).
Dueck, M. H. et al. Propofol attenuates responses of the auditory cortex to acoustic stimulation in a dose-dependent manner: a FMRI study. Acta Anaesthesiol. Scand. 49 (6), 784–791 (2005).
Martini, D. R. Commentary: the diagnosis of delirium in pediatric patients. J. Am. Acad. Child Adolesc. Psychiatry. 44 (4), 395–398 (2005).
Elmorsy, S. A., Soliman, G. F., Rashed, L. A. & Elgendy, H. Dexmedetomidine and Propofol sedation requirements in an autistic rat model. Korean J. Anesthesiology. 72 (2), 169–177 (2019).
Schopler, E., Reichler, R. J., DeVellis, R. F. & Daly, K. Toward objective classification of childhood autism: childhood autism rating scale (CARS). J. Autism Dev. Disord. 10 (1), 91–103 (1980).
Moon, S. J. et al. Accuracy of the childhood autism rating scale: a systematic review and meta-analysis. Dev. Med. Child Neurol. 61 (9), 1030–1038 (2019).
Acknowledgements
We thank the research staff of the Department of Radiology for their assistance in completing this study.
Funding
This work was supported by Chongqing municipal medical scientific research project (Major project) (2023DBXM003); and Chongqing Science and Technology Bureau’s Technical Innovation and Application Development (General Project) (CSTB2022TIAD-GPX0007).
Author information
Authors and Affiliations
Contributions
All authors contributed to the conception and design of the study. Material preparation, data collection, and analysis were performed by Lan Liu, Xue Ru, Min Du, Shaoqing Zhang, and Haisu Li. The initial draft of the manuscript was written by Haisu Li, and Jianxia Liu, Ying Xu, and Zudan Xie reviewed previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval
The study protocol received approval from the Ethics Committee of the Children’s Hospital of Chongqing Medical University (IRB #139/2021, approved on April 27, 2021) and was prospectively registered in the Chinese Clinical Trial Registry (Registration: No. ChiCTR2100050071, registered on August 16, 2021).
Consent to participate
In accordance with the Declaration of Helsinki, written informed consent was obtained from the parents of all participants.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Li, H., Du, M., Ru, X. et al. Comparison of different propofol target-controlled infusion concentrations in children with autism spectrum disorder undergoing magnetic resonance imaging. Sci Rep 15, 42706 (2025). https://doi.org/10.1038/s41598-025-26741-3
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-26741-3