Abstract
The evaluation of the basic properties and hydrocarbon generation potential of medium-low maturity shale oil serves as a critical link between geological resources and engineering development. This study focuses on JY oil shale, utilizing analytical techniques including vitrinite reflectance (R0), total organic carbon (TOC), thermogravimetric analysis (TGA), and Rock-Eval pyrolysis to systematically characterize its geochemical properties. Based on sample characteristics, an in-situ upgrading simulation system was developed and optimized. By comparing nuclear magnetic resonance (NMR) T1-T2 spectrum and pyrolysis-gas chromatography/mass spectrometry (PY-GC-MS) results before and after thermal cracking, the research further elucidates the mechanism by which thermal cracking influences the occurrence state of organic matter. Results indicate that the JY shale samples have a TOC content ranging from 1.94% to 2.57%, a total hydrocarbon generation potential (S1 + S2) of approximately 21 mg/g, R0 between 0.66% and 1.18%, and a sapropelinite content in kerogen as high as 90.33%. These parameters classify the kerogen as Type0 I, belonging to medium-low maturity source rocks with high organic matter abundance, favorable pyrolysis characteristics, and significant hydrocarbon generation potential, thus qualifying for in-situ upgrading development. Simulation experiments conducted within the temperature range of 400 °C to 600 °C show that under isothermal conditions at 450 °C for 12 h, the organic carbon degradation rate can reach about 40%, and fracturing measures can enhance the in-situ upgrading effect. After thermal cracking, the proportion of kerogen decreases by approximately 10%, the proportion of adsorbed hydrocarbons increases by about 5%, and the proportion of free hydrocarbons increases by roughly 3%. PY-GC-MS analysis further reveals that the proportion of light hydrocarbons increases significantly by about 20%, while the proportion of heavy hydrocarbons decreases by over 20%, validating the conversion sequence of “heavy hydrocarbons → medium hydrocarbons → light hydrocarbons” during thermal cracking. This study methodologically integrates multi-scale analysis with a self-developed simulation system and mechanistically clarifies the evolution pathway of the three-phase state of organic matter and the trend toward lighter hydrocarbon compositions. It provides theoretical and experimental support for assessing the feasibility of in-situ upgrading development and optimizing techniques for medium-low maturity shale oil, offering valuable insights for mitigating development risks.
Similar content being viewed by others
Introduction
Against the backdrop of global energy transition and persistent demand for fossil fuels, the development of unconventional oil and gas resources has emerged as a strategic priority for ensuring energy security and addressing supply-demand imbalances. Unlike conventional resources, unconventional oil and gas—such as shale oil, tight oil, oil sands, and coalbed methane—are characterized by widespread distribution, substantial reserves, and significant development potential. These attributes have elevated their role from a supplementary energy source to a major alternative resource1.
The International Energy Agency (IEA) forecasts that unconventional oil and gas will account for over 50% of the global incremental supply by 20402. Among these resources, shale oil has emerged as a pivotal factor in reshaping the global energy landscape, driven by technological breakthroughs and commercial maturation. Exemplified by the North American “shale revolution,” the United States has witnessed a dramatic surge in shale oil production—from under 500,000 barrels per day in 2005 to more than 9 million barrels per day in 2024—enabled by the widespread adoption of horizontal drilling and hydraulic fracturing. This transformation has not only reinforced U.S. energy independence but also exerted profound impacts on global crude oil markets and geopolitical dynamics.
Globally, technically recoverable shale oil reserves exceed 400 billion barrels, with major deposits located in the United States, China, Russia, and Argentina. China possesses substantial resources, with geological reserves estimated at 200–350 billion barrels and technically recoverable reserves ranging from 35 to 70 billion barrels, primarily distributed across basins in the northwest, southwest, and northern regions. Unlike the high-maturity, brittle shales typical of North American shale oil formations, China’s resources are largely hosted in medium–low maturity or organic-rich mudstones, imparting distinct characteristics and presenting unique development challenges3.
Medium-low maturity shale oil, characterized by vitrinite reflectance (Ro) < 1.3%, exhibits limited thermal evolution of organic matter, resulting in incomplete kerogen cracking and a high content of heavy components (> 40%). At this maturation stage, organic matter has not yet undergone sufficient cracking to generate light crude oil, remaining in the early or initial main oil-generation window. Consequently, conventional fracturing techniques achieve recovery factors of less than 5%, rendering such resources economically unviable under traditional development approaches4,5.
Thermal cracking upgrading enhances medium-low maturity shale oil quality by cleaving macromolecules into lighter hydrocarbons through thermal activation, thereby improving fluidity and recoverability. Primary in-situ technologies include electrical heating, steam/thermal fluid injection, and catalytic cracking6,7,8. A summary of the key features and application status of these technologies is provided in Table 1.
Based on a comprehensive review of the literature and consultations with various universities, research institutions, and companies, this study systematically analyzes nine in-situ upgrading technologies categorized into four types. The analysis covers their application characteristics, technical backgrounds, core mechanisms, and research progress, confirming the overall feasibility of in-situ upgrading for shale oil development.
Geochemical evaluation is crucial for exploring and developing medium–low maturity shale oil, defined as crude oil in shale formations with R0 values between 0.5% and 1.3%. This evaluation primarily focuses on assessing the hydrocarbon generation potential and transformation mechanisms of source rocks, along with characterizing the microscopic occurrence of organic matter. The following sections review the main methodologies and recent advances in this field.
-
(1)
Organic geochemical evaluation of oil shale
Total organic carbon (TOC) serves as a fundamental parameter in the geochemical evaluation of shale, directly determining the material basis for hydrocarbon generation. While shales with TOC values exceeding 2% generally demonstrate economic hydrocarbon potential, accurate assessment requires integration with additional parameters such as kerogen type27.
Kerogen maceral analysis enables the determination of kerogen type index, which serves as a key indicator for predicting hydrocarbon generation trends. Type I kerogen is predominantly oil-prone, Type II generates both oil and wet gas, while Type III is primarily gas-generating. Medium-low maturity shales are typically dominated by Type I–II kerogens, which exhibit higher oil generation efficiency. For instance, in the Bohai Bay Basin, algal organic matter (Type I) demonstrates 3–5 times greater oil generation potential than terrestrial organic matter (Type III)28.
R0 is widely used to classify the thermal evolution stages of shale. The numerical ranges correspond to distinct hydrocarbon generation phases: R0 < 0.5% indicates immaturity; 0.5% < R0 < 0.7% represents low maturity, characterized by initial hydrocarbon generation with predominantly immobile biodegraded oil; 0.7% < R0 < 1.3% corresponds to medium maturity, marking the peak oil generation window with maximum mobile oil content; 1.3% < R0 < 2.0% defines high maturity, producing mainly condensate and wet gas; and R0 > 2.0% signifies over-maturity, generating primarily dry gas and residual carbon27,29.
Organic geochemical analysis quantifies three key parameters—TOC (abundance), kerogen type (I-III), and thermal maturity (R0)-to predict in-situ hydrocarbon generation potential. This assessment provides the geochemical basis for developing in-situ upgrading technologies, with parameter priority following: TOC > kerogen type > maturity31.
-
(2)
Evaluation of hydrocarbon generation potential and transformation mechanism of oil shale
Rock-Eval pyrolysis provides quantitative characterization of organic matter in rocks through programmed temperature heating. This widely used technique yields key parameters including free hydrocarbons (S1), pyrolyzable hydrocarbons (S2), residual carbon (S₃), Tmax (S2 peak temperature), Hydrogen Index (HI), and Oxygen Index (OI), which form the foundation for evaluating source rock potential, determining maturity, and assessing shale oil resources22.
For medium-low maturity shales, S1 reflects present oil content while S2 indicates remaining generative potential. High HI values typically signify favorable oil-prone characteristics22. In the medium-low maturity range, Tmax shows consistent correlation with R0, while HI helps determine organic matter type and generative potential. Recent studies increasingly focus on integrated evaluation models combining multiple Rock-Eval parameters with complementary analytical data for comprehensive resource assessment24.
-
(3)
Microscopic occurrence and characterization of organic matter in shale
Research on the microscopic occurrence and characterization of shale organic matter-particularly the quantitative differentiation between free and adsorbed oil-provides fundamental insights for assessing the recoverable potential of medium-low maturity shale oil and identifying sweet spots.
Some studies have used the low-field nuclear magnetic resonance (NMR) T1-T2 chart proposed by Ausik34 to evaluate shale oil reservoirs in the Qaidam Basin, Songliao Basin, Ordos Basin, and Junggar Basin. In recent years, with the deepening of research on oil and gas mobility evaluation, how to optimize the T1-T2 chart has also become a research hotspot35.
Based on systematic experiments utilizing NMR technology, Wei et al. developed logging evaluation methods for free hydrocarbons and brittleness index in the Fengcheng Formation shale oil reservoirs. Their research demonstrated that two-dimensional NMR spectroscopy effectively distinguishes between movable water, free hydrocarbons, and adsorbed hydrocarbons. Results revealed relatively low free hydrocarbon porosity in the Fengcheng Formation shale layers36.
Based on T1-T2 spectral analysis of shales under varying conditions and pore fluid dynamics, Li38 demonstrated that shale oil in the Gulong Sag (Songliao Basin) primarily exists as bound oil, supplemented by adsorbed hydrocarbons, with limited movable content. In situ oil content is predominantly controlled by total organic carbon(TOC), clay mineral content, and maturity. Both higher TOC and clay mineral fractions, along with increased maturity, correlate with enhanced oil-bearing capacity. Additionally, adsorbed hydrocarbon content shows a declining trend with advancing maturity.
Liu38 investigated the shale of the Triassic Yanchang Formation (7th member) in the Ordos Basin using a combined saturation-centrifugation-NMR experimental approach integrated with the adsorption ratio equation established by Li Junqian39. The study comprehensively analyzed the content, proportion, microscopic distribution, and mobility characteristics of adsorbed versus free shale oil.
Professor Lu’s research team40,41,42,43,44,45 has made significant contributions to characterizing organic matter occurrence in low-medium maturity shale oil. Their multidisciplinary approach integrates advanced analytical techniques to elucidate hydrocarbon occurrence mechanisms and their impact on fluidity. Innovatively employing NMR technology, they quantified movable, semi-movable, and bound oil in nanopores by analyzing fluid relaxation properties. Investigation of oil distribution across varying pore sizes directly revealed the mobility constraints limiting low-medium maturity shale oil production.
This study investigates medium-low maturity shale oil through systematic characterization of JY samples using R0, TOC, thermogravimetric analysis (TGA), and Rock-Eval pyrolysis. A customized in-situ upgrading simulation system was developed based on sample properties. By comparing NMR T1-T2 spectra and Pyrolysis-Gas Chromatography-Mass Spectrometry (PY-GC-MS) data before and after thermal cracking, we elucidate the mechanism of thermal cracking on organic matter occurrence states. These findings provide theoretical and experimental support for optimizing development strategies, assessing in-situ upgrading feasibility, and mitigating risks in medium-low maturity shale oil production.
Basic properties of oil shale
Organic geochemical evaluation of oil shale
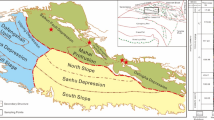
The JY shale samples investigated in this study were obtained from a depression within the Bohai Bay Basin, with burial depths ranging from 3500 to 4000 m. The reservoir exhibits a continuous thickness of 300–500 m, locally reaching over 1000 m, and was deposited during the Sha-1 Member to Dongying Formation period. The samples are dark brown in color and display distinct laminated structures(Fig. 1). Prior to experimentation, all samples were pretreated according to the specific requirements of each analytical method.
Shale samples.
TOC analysis of oil shale
The determination of TOC content in sedimentary rocks was performed using a TOC-Vcph analyzer manufactured by Shimadzu Corporation of Japan. The experimental process was strictly carried out in accordance with the China national standard “Determination of Total Organic Carbon in Sedimentary Rocks” (GB/T 19145 − 2022).
In this standard, the high-temperature combustion oxidation method is used to determine the TOC. The organic carbon content in the sample is calculated based on the amount of CO2 generated, and the results are shown in Table 2.
As can be seen from Table 2, the TOC of oil shale samples ranges from 1.94% to 2.57%, with an average value of 2.39%. Referring to the evaluation criteria for organic matter abundance in Appendix Table 1, it can be determined that the organic matter abundance of JY oil shale samples is at a medium-high level, belonging to the category of “good source rocks and high-quality source rocks”, and having a good material basis for hydrocarbon generation.
Pyrolysis analysis of oil shale
The rock pyrolysis analysis experiment employed the Rock-Eval 6 automatic pyrolysis analyzer produced by Vinci Technologies of France. The experimental process strictly adhered to the national standard “Rock Pyrolysis Analysis” (GB/T 18602 − 2012), obtaining the S1 and S2, and recording the characteristic pyrolysis peak temperature (Tmax). A series of key parameters such as production index (PI), HI, effective carbon (PC), and degradation rate (D) can be derived from TOC, S1, S2, and Tmax, with specific results shown in Table 2.
With reference to the rock evaluation criteria in the Appendix Tables, an analysis of the pyrolysis test data of oil shale samples reveals the following result: The pyrolysis peak temperature of oil shale samples in this block ranges from 435 °C to 455 °C, indicating their good pyrolysis characteristics; their total hydrocarbon generation potential (S1 + S2) is approximately 21 mg/g, a value that shows the oil shale samples have high hydrocarbon generation potential; at the same time, the production index is around 0.60, indicating that they can maintain a stable hydrocarbon production efficiency during pyrolysis; in addition, the degradation rate is about 40%, meaning that a considerable portion of organic matter is effectively converted during pyrolysis. It is worth noting that the HI is greater than 600, and these values further confirm the excellent properties of the oil shale samples as source rocks.
In summary, the oil shale samples in this block belongs to high-quality source rocks of organic matter type I, with high research and development value.
Kerogen analysis of oil shale
Shale kerogen analysis is a key technology for the systematic characterization of insoluble organic matter (kerogen) in shale through chemical and physical methods. The purified kerogen is made into thin sections, and under an optical microscope, kerogen component identification, component content statistics, and reflectance determination are carried out. Conventional indicators mainly include kerogen microscopic examination, kerogen hydrogen-carbon and oxygen-carbon atomic ratios (H/C, O/C), type index (T), HI, etc. The organic matter types of source rocks can be classified through the above indicators.
-
(1)
Maceral analysis of kerogen.
Kerogen in oil shale samples was extracted with reference to the “Method for Separation of Kerogen from Sedimentary Rocks” (GB/T 19144 − 2010), and maceral detection, reflectance detection, and elemental analysis were performed on it. In this study, a ZEISS Axio Scope fluorescent biological microscope was used to observe the morphological structure of kerogen, and the results are shown in Fig. 2; Table 3.
Fig. 2 Photos of Oil shale Kerogen Macerals.
Table 3 Detection results of 1# oil shale Kerogen. According to the identification results of oil shale kerogen macerals, the content of sapropel group in kerogen is as high as 90.33%. The sapropel group is mainly composed of planktonic algal bodies and sapropelic amorphous substances. Such organic substances gradually transform into kerogen after long-term biochemical processes in an anoxic sedimentary environment.
In addition to the sapropel group, the vitrinite group is also an important component of kerogen, with a content of 9.67%. The vitrinite group includes hydrogen-rich vitrinite and normal vitrinite, and such organic substances are mainly derived from terrestrial plants.
According to the classification standard46 of kerogen organic matter types, the kerogen in this oil shale sample is of Type I. This type of kerogen is dominated by the sapropel group, with high hydrocarbon generation potential and oil production capacity. In addition, the hydrocarbon generation process of Type I kerogen is relatively fast, which is conducive to the rapid generation and accumulation of shale oil.
-
(2)
Vitrinite reflectance of kerogen.
Ro is internationally recognized as the most universal indicator for assessing the thermal evolution of organic matter. It reflects the maturity of organic matter by characterizing the evolution of the aromatic structure of vitrinite under thermal conditions, providing irreplaceable reliability in the evaluation of terrestrial and transitional facies source rocks. Currently, Ro is widely used as a key criterion for classifying the thermal evolution stages of shale oil. For specific classification criteria, please refer to Appendix Table 3.
In this study, Ro was analyzed using a UMSP-50 microphotometer. Approximately 50 measurement points were collected, with a deviation of about 0.03%. The experimental procedure followed the Chinese petroleum industry standard “Method for Determination of Ro in Sedimentary Rocks” (SY/T 5124 − 2012).
Table 4 Results of Ro Detection. From the data in Table 4, we can conclude that the Ro of the JY shale samples ranges from 0.66% to 1.18%. According to industry maturity classification standards, these results indicate that the shale oil in this block falls within the low to medium maturity range.
-
(3)
Elemental analysis of oil shale kerogen.
The elemental composition of kerogen directly reflects its potential and capability to generate hydrocarbons. To further understand the hydrocarbon generation potential of JY oil shale, elemental analysis was performed on the kerogen extracted from oil shale samples.
The Vario EL Cube Elemental Analyzer produced by Elementar GmbH (Germany) was employed. The contents of carbon (C), hydrogen (H), nitrogen (N), and sulfur (S) were determined using the combustion method, while the oxygen (O) content was measured via the high-temperature pyrolysis reduction method. The specific procedure refers to Method for Analysis of Carbon, Hydrogen, Oxygen and Nitrogen in Rock Organic Matter( GB/T 19143 − 2017 ).
Classification diagram of kerogen based on elemental analysis.
As can be seen from Table 5, the H/C ratios of kerogen in shale are all greater than 1.5, and the O/C ratios are all less than 0.1, which verifies that the kerogen contained in oil shale is Type I kerogen. (Fig. 3)
The results of organic geochemical evaluation on JY oil shale show that: the TOC of oil shale samples in the study area ranges from 1.94% to 2.57%, indicating good organic matter abundance; the kerogen organic matter type is Type I, and the Ro ranges from 0.66% to 1.18%, suggesting that the source rocks are in the middle-low maturity stage. The pyrolysis peak temperature (Tmax) distributes in the range of 435 °C to 455 °C, showing good pyrolysis performance; the total hydrocarbon generation potential (S1 + S2) is approximately 21 mg/g, indicating that the samples have high hydrocarbon generation potential. Due to the low content of free hydrocarbons and high content of cracked hydrocarbons, it is necessary to consider improving shale oil production in this block through in-situ pyrolysis upgrading technology.
TGA analysis of oil shale
Shale TGA is a key technology that reveals thermal stability, organic matter composition, and hydrocarbon generation behavior by monitoring changes in sample mass with temperature/time.
In this study, the LabsysEvo synchronous thermal analyzer manufactured by Seteram (France) was used to perform TGA on JY 1# oil shale. The main configurations and characteristics of the instrument are as follows: temperature range: room temperature to 1600 °C; heating and cooling rate: 0.01 to 100 K/min; resolution: 0.2 µg /0.02 µg; DSC resolution: 0.4µW; DSC baseline noise: 16µW.
Prior to the experiment, oil shale samples were pretreated to remove impurities such as surface mud. They were dried at a constant temperature of 60 °C for 24 h and ground into powder with a particle size of 80 mesh. During the experiment, both DTG and TG curves were measured simultaneously. The sample weight was controlled at 9.0 ± 0.1 mg throughout the test. For the determination of TG-DTG curves, the initial temperature was 25 °C, the heating rate was 10 °C/min, the reaction atmospheres were nitrogen and air respectively, and the final reaction temperature was 800 °C.
TG/DTG curve of 1# oil shale pyrolysis under varying atmospheres.
The weight loss rates of oil shale samples in the three temperature stages under nitrogen and air atmospheres were calculated respectively, and the results are shown in Figs. 5 and 6.
Thermogravimetric behavior of 1# sample under nitrogen atmosphere.
Thermogravimetric behavior of 1# sample under air atmosphere.
Based on the TG and DTG curves from the TGA of the samples under nitrogen and air atmospheres (Fig. 4), the thermal weight loss of oil shale can be divided into three stages.
Evaporation stage (< 300 °C): The TG curve decreases slowly, with weight loss mainly resulting from the evaporation of free water, interlayer water in clay minerals, and free organic components. The weight loss rate of samples at this stage is below 2%, indicating a low content of free hydrocarbons.
Organic matter pyrolysis stage (300 °C–600 °C): The TG curve shows a significant decrease with rapid weight loss, mainly caused by the pyrolysis of organic matter. Multiple peaks in the DTG curve indicate that this process is a multi-step reaction organic matter first pyrolyzes into asphaltenes, then further generates petroleum, natural gas, and coke. The weight loss rate of samples at this stage ranges from 3% to 10%, suggesting that the content of cracked hydrocarbons is higher than that of free hydrocarbons, with relatively high hydrocarbon generation potential.
Inorganic mineral thermal decomposition stage (> 600 °C): The TG curve decreases rapidly with fast weight loss, mainly resulting from residual carbon oxidation and inorganic mineral thermal decomposition. Based on organic geochemical evaluation (predominantly Type I kerogen, with little difference in DTG curves between air and nitrogen atmospheres), the residual carbon content is low. Thus, the weight loss is mainly caused by the high-temperature decomposition of carbonate rocks such as calcite and dolomite. The weight loss rate of samples at this stage ranges from 10% to 30%.
TGA results show that the main weight loss interval of organic matter is 400 °C-600 °C. Combined with the range of pyrolysis peak temperature (Tmax) of 435 °C-455 °C from pyrolysis analysis, the temperature interval for the in-situ upgrading simulation experiment of JY oil shale can be initially determined as 400 °C-600 °C. To inhibit the oxidation of residual carbon, nitrogen is recommended as the reaction atmosphere.
Optimization of experimental conditions for oil shale pyrolysis
Based on the basic physical and chemical properties of JY oil shale, this study independently developed a multi-functional simulation experimental system for in-situ upgrading of oil shale (Fig. 7). The reaction chamber of this system can withstand a maximum temperature of 800 °C and a pressure of 20 MPa, and the supporting gas-liquid separator can realize the separation and collection of products.
Simulation experimental system for in-situ upgrading of oil shale.
The procedure of the pyrolysis experiment is as follows: (1) Dry the JY oil shale sample at low temperature, weigh it, and then perform NMR testing to obtain the T1-T2 spectrum of the oil shale sample before upgrading, which is used to analyze the occurrence state of organic matter in the oil shale. (2) Place the oil shale sample into the in-situ upgrading simulation experimental system and install the reaction system properly. Before heating, purge with nitrogen for 10 min to remove air from the reactor, then heat to the preset delay temperature at a rate of 10 °C/min. (3) The start of the delay time is when the temperature reaches the delay temperature, and the delay time for each upgrading reaction is set according to the experimental scheme. (4) After the reaction, record the pressure value of the reaction system and allow the system to cool down to near room temperature. (5) After the experiment, collect the products (core, liquid, gas), weigh and measure them, and keep proper records. (6) Perform NMR testing on the upgraded JY oil shale sample to obtain its T1-T2 spectrum, and comparatively analyze the changes in the occurrence state of organic matter in the oil shale. The weight loss rate and cracking rate are obtained by comparing the weight and TOC value of the samples before and after the experiment. The experimental records are shown in Table 6. After the pyrolysis test, the color of the shale became lighter, and small cracks appeared in the cylindrical samples (shown in Fig. 8).
Shale sample after the pyrolysis test.
The simulation results of in-situ upgrading of JY oil shale show that, within the experimental parameters set in this study, the upgrading effect improves gradually with increasing temperature. Notably, when the upgrading temperature exceeds the pyrolysis peak temperature, efficient upgrading at lower temperatures can be achieved by extending the residence time. For example, at 450 °C with a 12-hour residence time, the degradation rate of organic carbon in oil shale reaches approximately 40%, a result consistent with the theoretical degradation rate derived from pyrolysis analysis.
The comparison results show that the upgrading effect of fragmented JY oil shale is significantly better than that of columnar oil shale. Analysis indicates that the larger specific surface area of fragmented shale can promote the uniform dispersion of heat, enhancing the sufficiency of thermal cracking and the efficiency of in-situ upgrading. However, the study found that the presence of water will weaken the upgrading effect to a certain extent. Therefore, for field experiments on in-situ upgrading of JY oil shale, it is recommended to adopt a combined process of waterless fracturing and thermal cracking to avoid the inhibitory effect of water and improve the overall upgrading efficiency.
Analysis of occurrence states of organic matter in oil shale before and after thermal cracking
Studying the occurrence state of organic matter in oil shale is one of the core topics in the field of unconventional oil and gas geology and development. Scholars have developed a variety of research methods, among which NMR for the identification and quantitative evaluation of hydrogen components and the PY-GC-MS method are important means. In this chapter, NMR T1-T2 spectrum scanning and PY-GC-MS testing are performed on Sample 4 and Sample 5 before and after thermal cracking, respectively, to analyze the impact of thermal cracking on the occurrence state of organic matter in oil shale.
Identification of occurrence states of organic matter in oil shale
Based on previous studies of NMR analysis for hydrogen-containing components in oil shale, this work adopted the T1-T2 identification method established by Lu’s team41,42 to develop the classification scheme for hydrogen-containing components in JY shale.
The NMR analysis was performed using a MesoMR23-060 H-I low-field NMR system (Suzhou Niumag Analytical Instruments Co., Ltd.) equipped with an NdFeB permanent magnet. The system maintained a stable magnetic field of 0.52 T at a resonance frequency of 21.36 MHz. A 25 mm detection coil was used with a minimum echo spacing of 0.07 ms and a maximum of 18,000 echoes. T1-T2 spectra were obtained from Shale Sample 4# and 5# before and after pyrolysis (450 °C, 12 h) to track changes in organic matter occurrence states.
According to the positions of signals in the charts (Fig. 9; Table 7), the occurrence forms and abundances of organic matter present in shale can be judged.
Schematic of NMR T1-T2 spectra division scheme for hydrogen-containing components in shale.
T1-T2 spectrum of 4# shale.
T1-T2 spectrum of 5# shale.
The NMR T1-T2 spectra of oil shale samples before and after upgrading (Figs. 10 and 11) show that after upgrading, the kerogen signal weakens, while the signals of adsorbed hydrocarbons and free hydrocarbons are enhanced. The results of quantitative analysis of organic matter in different occurrence states using NMR data are shown in Table 8.
Influence of thermal cracking on the occurrence states of organic matter in shale.
From the analysis results (Fig. 12), after the shale samples were subjected to isothermal pyrolysis at 450 °C for 12 h, the proportion of kerogen decreased by approximately 10% overall, the proportion of adsorbed hydrocarbons remaining in the pores of shale increased by about 5%, and the proportion of free hydrocarbons increased by roughly 3%. Since the experimental environment was an open space, most free hydrocarbons volatilized after the cracking reaction. As shown in Table 6, the mass loss of 11%–14% is mainly caused by the volatilization of free hydrocarbons. Therefore, theoretically, the yield of free hydrocarbons would be higher, and the effect of thermal cracking upgrading would be more significant.
PY-GC-MS analysis of oil shale
To further analyze the influence of the thermal cracking process on the occurrence states of organic matter in oil shale, Samples 4 and 5 were selected for PY-GC-MS analysis.
In the experiment, the analytical instrument used was a GCMS-QP2010Ultra (manufactured by Shimadzu Corporation, Japan), with a pyrolyzer EGA/PY-3030D (manufactured by Frontier Laboratories, Japan) and a UA-5MS chromatographic column (30 m × 0.25 mm × 0.25 μm). Small amounts of oil shale samples before and after thermal cracking were taken respectively, heated to 600 °C through a programmed temperature process, and the pyrolysis products were detected by GC-MS. The pyrolysis-gas chromatography-mass spectrometry spectra and data of oil shale samples before and after thermal cracking were compared and analyzed (Figs. 13 and 14).
PY-GC-MS spectra of 4# shale pre-pyrolysis and post-pyrolysis.
PY-GC-MS spectra of 5# oil shale pre-pyrolysis and post-pyrolysis.
The “peak area normalization method” was used to calculate the proportions of hydrocarbons with different carbon numbers (< C11, C11-C20, > C20). The results are shown in Fig. 15, which illustrates the changes in light hydrocarbons, medium hydrocarbons, and heavy hydrocarbons in the two oil shale samples before and after thermal cracking.
Analysis results for PY-GC-MS oil shale pre-pyrolysis and post-pyrolysis.
As shown in Fig. 15, after thermal cracking, the < C10 components (light hydrocarbons) in both oil shale samples increased significantly. This indicates that the pyrolysis process promoted the conversion of heavy components to light components (< C10), greatly increasing the proportion of low-molecular-weight carbon substances, with the enrichment of light components in Sample 4 being more obvious after conversion. The proportion of C11-C20 (medium hydrocarbons) in both oil shale samples rose slightly; some heavy hydrocarbons (> C20) broke their chains into medium hydrocarbons, and the amount generated exceeded the consumption of medium hydrocarbons further cracked, leading to a slight increase in proportion. The > C20 (heavy hydrocarbons) in both oil shale samples decreased because heavy hydrocarbons (kerogen), as the “raw material” for cracking, broke their chains and converted into light and medium hydrocarbons, resulting in a significant reduction in their proportion.
Therefore, the thermal cracking process of low-to-moderate maturity shale oil involves the conversion sequence of “heavy hydrocarbons → medium hydrocarbons → light hydrocarbons”, where medium hydrocarbons serve as intermediate products that may further convert into light hydrocarbons if the cracking time is extended.
Comparing the two samples, Sample 4 had lower organic matter maturity and higher heavy hydrocarbon content before cracking. After thermal cracking, it showed a greater decrease in heavy hydrocarbons and a larger increase in light hydrocarbons. This is because low-maturity organic matter is more temperature-sensitive, undergoes more complete cleavage, and generates more light hydrocarbons.
Conclusions
This study provides theoretical and experimental support for the efficient development and technical optimization of medium-low maturity shale oil, offering valuable references for assessing the feasibility of in-situ upgrading and mitigating development risks. The main conclusions are as follows:
-
(1)
The oil shale samples from the study area show TOC content of 1.94%-2.57%, pyrolysis peak temperature of 435–455 °C, hydrocarbon generation potential of approximately 21 mg/g, production index of about 0.60, degradation rate of about 40%, Ro of 0.66%-1.18%, and sapropelinite content in kerogen reaching 90.33%. These characteristics confirm Type I kerogen in medium-low maturity source rocks with high organic matter abundance, favorable pyrolysis properties, and significant hydrocarbon generation potential, suitable for in-situ upgrading development.
-
(2)
Thermogravimetric and pyrolysis analyses indicate that under in-situ upgrading simulation at 400–600 °C, specifically under isothermal conditions at 450 °C for 12 h, the organic carbon degradation rate reaches approximately 40%. Fracturing measures can further enhance the in-situ upgrading effect.
-
(3)
NMR analysis reveals that after thermal cracking at 450 °C for 12 h, the kerogen proportion decreases by approximately 10%, adsorbed hydrocarbons increase by about 5%, and free hydrocarbons increase by around 3%, demonstrating significant changes in organic matter occurrence states due to thermal cracking.
-
(4)
Following thermal cracking, light hydrocarbons increase significantly by approximately 20%, medium hydrocarbons increase slightly by about 5%, while heavy hydrocarbons decrease by over 20%, validating the conversion sequence of “heavy hydrocarbons → medium hydrocarbons → light hydrocarbons”. Extended cracking duration may further promote the conversion of medium to light hydrocarbons.
Data availability
To access the measured experimental data, one can contact Dr. Bai Jinmei by contacting email of [baijinmei@cczu.edu.cn](mailto: baijinmei@cczu.edu.cn) .
References
Zou, C. et al. Shale oil and gas revolution and its impact. Acta Petrolei Sinica. 41 (1), 1–12 (2020).
International Energy Agency. World Energy Outlook 2019 (IEA, 2019). https://www.iea.org/reports/world
Zou c et al. Scientific and technological progress, development strategy and policy suggestion regarding shale oil and gas. Acta Petrolei Sinica. 43 (12), 1675–1686 (2022).
He, W. et al. Geological characteristics and exploration breakthroughs of the middle to low mature shale oil of Nenjiang formation in Northern Songliao basin. Acta Petrolei Sinica. 45 (6), 900–913 (2024).
Song, Z. et al. High-pressure phase behavior and mass transfer law of Gulong shale oil and CO2 in Daqing oilfield. Acta Petrolei Sinica 2024, 45(2): 390–402 .
Wang, Y., Meng, X. & Su, J. Pyrolysis kinetics and permeability evolution of low-maturity shale oil. Petroleum Nat. Gas Geol. 40 (3), 678–684 (2019).
Li, H., Chapman, D. N. & Faramarzi, A. Multiphysics modeling of shale oil in-situ upgrading. Energy 295, 126783 (2025).
Li, Y. et al. Evaluation technology and practice of continental shale oil development in China. Pet. Explor. Dev. 49 (5), 955–964 (2022).
He, W. et al. Controlling the in-situ conversion process of oil shale via geochemical methods: A case study on the Fuyu oil shale, China. Fuel Process. Technol. 219, 106876 (2021).
Guo, W. et al. Oil shale in-situ conversion technology triggered by topochemical reaction method and pilot test project in Songliao basin. Acta Petrolei Sinica. 45 (7), 1104–1121 (2024). 1129.
Brandt, A. R. Converting Oil Shale To Liquid Fuels: Energy Inputs and Greenhouse Gas Emissions of the Shell in Situ Conversion Process Vol. 43, 7489–7495 (Environmental Science & Technology, 2008). 19.
Xiong, C. et al. Kinetic model for the pyrolysis of organic matter in oil shale from the Nenjiang Formation, Songliao basin. J. Tsinghua Univ. (Sci Technol). 22, 033 (2025).
Brandt, A. R. Converting oil shale to liquid fuels: energy inputs and greenhouse gas emissions of the shell in situ conversion process. Environ. Sci. Technol. 42 (19), 7489–7495 (2008).
Kang, Z. et al. Pilot test of in-situ steam injection for oil and gas production from oil shale and applicabilitu of multi-mode in-situ thermal recovery technology. Acta Petrolei Sinica. 42 (11), 1458–1468 (2021).
Huang, X. et al. Experimental investigation on micro-fracture evolution and fracture permeability of oil shale heated by water vapor. Energy 277, 127677 (2023).
、Kang, Z. et al. Comparative study of pore structure changes of oil shale based on different heating modes of conduction and convection. Chin. J. Rock Mechan. Eng. 37 (11), 2565–2575 (2018).
Hazra, K. et al. Comparison of Heating Methods for In-situ Oil Shale Extraction. 17th European Symposium on Improved Oil Recovery, cp-342-00038. (2013).
Carter, S. D. & Burnham, R. Comparison of the CCR Process with Other Oil Shale Retorting Technologies. In Oil Shale: A Solution to the Liquid Fuel Dilemma. ACS Symposium Series, 1218:213–235. (2016).
Kang, Z. et al. Microscopic structure evolution and macroscopic mechanical response of oil shale under anhydrous and near/supercritical water pyrolysis. Acta Petrolei Sinica. 46 (4), 801–815 (2025).
Huang, H. et al. A coupled thermo-hydro-mechanical-chemical model for production performance of oil shale reservoirs during in-situ conversion process. Energy 268, 126700 (2023).
Burnham, A. K. Slow Radio-Frequency Processing of Large Oil Shale Volumes To Produce Petroleum-like Shale Oil (Lawrence Livermore National Laboratory Report, 2003). UCRL-ID-155045.
Zhang, Y. et al. Visualization and quantitative study on seepage characteristics of oil shale under microwave radiation. Chin. J. Rock Mechan. Eng. 42 (12), 2919–2931 (2023).
El-Fayoumi, M. A. & Chang, K. A review of In-Situ radio frequency heating technology for enhanced oil recovery from oil shale. Int. J. Eng. Res. Appl. 5 (6), 66–73 (2015).
Wang, H. & Chen, Z. Numerical investigation of the in-situ RF heating process for oil shale recovery. Energy Sci. Eng. 7 (5), 1913–1926 (2019).
Braun, R. L. & Chin, R. C. Comparison of the chemical and physical changes of oil shale during in situ retorting with different heating methods. Ind. Eng. Chem. Res. 26 (3), 427–433 (1987).
Burnham, A. et al. Study of the topochemistry of oil shale pyrolysis. Energy & Fuels, 2013,27(5): 2635–2645. (2013).
Geochemical method for source rock evaluation. Standard of the People’s Republic of China for the Petroleum and Natural Gas Industry, SY/T 5735 – 2019.
Gao, Y. et al. Jump in the structure of type I kerogen revealed from pyrolysis and 13C DP/MAS NMR. Org. Geochem. 112, 105–118 (2017).
Zhang, L. et al. Mechanisms of shale gas generation from typically organic-rich marine shales. Earth Sci. 42 (7), 1092–1106 (2017).
Kuang, L. et al. Key parameters and methods of lacustrine shale oil reservoir characterization. Acta Petrolei Sinica. 42 (1), 1–14 (2021).
Li, J. et al. Evaluating the total oil yield using a single routine Rock-Eval experiment[J]. J. Anal. Appl. Pyrol. 144, 104707 (2019).
Kuang, L. et al. Key parameters and evaluation methods for large-scale production of lacustrine shale oil. Pet. Explor. Dev. 25 (4), 782–791 (2025).
Liu, H. et al. Geochemical Parameters for Evaluating Shale Oil Enrichment and Mobility: A Case Study of Shales in the Bakken Formation, Williston Basin and the Shahejie Formation,Jiyang Depression Vol. 45, 622–636 (Oil & Gas Geology, 2024). 3.
Kausik, R. et al. NMR relaxometry in shale and implications for logging.Petrophysics,2016, 57(4):339–350 .
Venkata, R. et al. An unsupervised learning algorithm to compute fluid volumes from NMR T1-T2 logs in unconventional reservoirs.Petrophysics, 59(5): 617–632. (2018).
Wang, W. et al. Key technology of shale oil sweet spot evaluation and sweet spot type division in Fengcheng formation of Mahu Sag. Earth Sci. 48 (1), 223–234 (2023).
Li, Q. et al. Response characteristics and application of nuclear magnetic resonance T1-T2 spectrum in shale oil reservoirs: a case study of Qingshankou formation in Gulong Sag, Songliao basin. J. Northeast Petroleum Univ. 48 (3), 89–101 (2024).
Liu, F. et al. Quantitative Characterization of Adsorbed and Free Shale Oil Microscopic Distribution Based on Nuclear Magnetic Resonance: a Case Study of Chang 7 Member of Triassic Yanchang Formation in Ordos Basin Vol. 47, 645–658 (Petroleum Geology & Experiment, 2025). 3.
Li, J. et al. Quantitative Evaluation Models of Adsorbed and Free Shale Oil and its Microscopic Occurrence Mechanism Vol. 40, 583–592 (Oil & Gas Geology, 2019). 3.
Wang, M. et al. Occurrence mechanism of lacustrine shale oil in the paleogene Shahejie formation of Jiyang Depression, Bohai Bay Basin, China[J]. Pet. Explor. Dev. 46 (4), 789–802 (2019).
Zhang, P. et al. 1D and 2D nuclear magnetic resonance (NMR) relaxation behaviors of protons in clay, kerogen and oil-bearing shale rocks. Mar. Pet. Geol. 114, 104210 (2020).
Zhang, P. et al. Quantitative characterization of shale pore connectivity and controlling factors using spontaneous imbibition combined with nuclear magnetic resonance T2 and T1-T2. Pet. Sci. 20, 1947–1960 (2023).
Zhang, P. et al. Microscopic occurrence and distribution of oil and water in situ shale: evidence from nuclear magnetic resonance. Pet. Sci. 21, 3675–3691 (2024).
Wang, J. et al. Insights into microscopic oil occurrence characteristics in shales from the paleogene funing formation in Subei Basin, China. Pet. Sci. 22 (1), 55–75 (2025).
Bai, X. et al. Occurrence characteristics and factors that influence shale oil in the jurassic Lianggaoshan Formation, Northeastern Sichuan basin. Mar. Pet. Geol. 171, 107197 (2025).
Geochemical Evaluation Method for Source Rocks. Petroleum and Natural Gas Industry Standard of the People’s Republic of China, SY/T 5735 – 2019.
Acknowledgements
This publication is based upon work supported by CNPC-CZU Innovation Alliance. The author(s) would like to acknowledge the support received under the Research Program 2021DQ06.
Funding
This study received funding from CNPC-CZU Innovation Alliance. The authors would like to acknowledge the support received under the Research Program 2021DQ06.
Author information
Authors and Affiliations
Contributions
Bai Jinmei assumed overall responsibility for the paper’s writing, Qian Kun completed a portion of the experimental work, Wu Xiaojun participated in literature research and data processing, Dou Xiangji was responsible for conceptualizing the paper and reviewing experimental results, while He Yanfeng provided guidance on the paper’s thematic conception, experimental design, and structural framework. All authors reviewed the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Jinmei, B., Kun, Q., Xiaojun, W. et al. Thermal cracking for upgrading medium-low maturity shale oil: evolution of organic matter occurrence. Sci Rep 15, 43054 (2025). https://doi.org/10.1038/s41598-025-27082-x
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-27082-x