Abstract
A novel magnetically active ZnFe2O4@MIL-53(Al)-NH2 composite was prepared for the first time, and its adsorption performance for diclofenac sodium (DS) in solution was investigated systemically. Structural analyses by XRD and FTIR confirmed the successful integration of ZnFe2O4 nanoparticles within the MIL-53(Al)-NH2 framework, while magnetic hysteresis loop measurements revealed excellent magnetic responsiveness, enabling rapid recovery under an external magnetic field within 60 s. The composite exhibited a maximum DS adsorption capacity of 217.85 mg/g at pH 6.44, 298 K, and 60 min contact time, significantly higher than those of most recent benchmarks, including Lignin‑Based Magnetic Nanoparticle (106.4 mg/g), Ni0.5Zn0.5Fe2O4 (52.91 mg/g) and Na-X zeolite (6.68 mg/g). The DS adsorption process is primarily spontaneous and endothermic, dominated by electrostatic attraction, hydrogen bonding, and π–π interactions. The high stability, negligible ion leaching, and efficient magnetic recyclability underscore its potential for scalable wastewater treatment applications. This study provides valuable insights into the mechanism of magnetism-enhanced adsorption and offers a promising pathway for developing multifunctional MOF-based adsorbents for environmental remediation.
Similar content being viewed by others
Introduction
It has been reported that approximately 30% of the global population lacks access to safe drinking water1. Clean water is essential not only for human survival but also for industrial and agricultural activities2,3. However, the persistent release of contaminated effluents into aquatic ecosystems is contributing to rising levels of toxic chemical pollution4,5,6,7. Many studies have reported common toxic pollutants in water, including pharmaceuticals8, antibiotics9, pesticides10, heavy metals11, organic dyes12 and nitroaromatic compound13,14. These contaminants have raised significant concerns in both research and environmental management due to their harmful effects on human health and aquatic microorganisms15,16,17,18. Furthermore, the presence of organic pollutants like dyes and antibiotics in water can diminish light intensity, thereby disrupting the photosynthetic processes of microorganisms and aquatic plants19,20. This leads to a decrease in the biochemical oxygen demand of the water, hindering the decomposition of organic matter by organisms21,22. As a result, monitoring and purifying drinking water is essential to ensure its safety.
A growing number of artificial substances have recently been detected in aquatic ecosystems, which have been categorized as novel pollutants. Pharmaceuticals and personal care products (PPCPs) are prominent examples of emerging contaminants due to their excessive use in safeguarding human health. The accumulation of PPCPs in water bodies poses significant risks to ecological systems23,24. Diclofenac sodium (DS), a prevalent PPCPs in aquatic environments, is commonly found in both drinking and surface waters25. As one of the most frequently prescribed non-steroidal anti-inflammatory drugs (NSAIDs), DS is often used in the treatment of various inflammatory diseases26. However, prolonged or repeated exposure to DS can lead to adverse health effects, including hemodynamic changes and thyroid tumors27. Pharmaceuticals enter the environment primarily through two pathways: untreated wastewater from the pharmaceutical industry and human metabolic byproducts, along with unused medications, found in domestic sewage. Moreover, some municipal wastewater treatment plants fail to adequately remove DS, leading to elevated DS concentrations in water. This highlights the need for further study about wastewater treatment technologies. Simultaneously, the presence of DS in aquatic environments poses toxic risks to aquatic organisms28.
Because of its cost-effectiveness, convenience and reusability, adsorption technology has received increasing attention in wastewater treatment29. Metal-Organic Frameworks (MOFs) are a novel class of hybrid materials composed of metal ions coordinated with organic ligands30. MOFs exhibit high surface area, tunable pore structures and rich active sites, which enhances their capacity for efficiently adsorbing pollutants31. Aluminum is one of the most abundant metals on Earth, ranking third in terms of its content in the Earth’s crust. The MOFs based on aluminum carboxylate coordination chemistry are considered one of the most stable systems within this class of materials32,33. Research on aluminum-based MOFs as adsorbents offers significant practical and scientific value. However, once the adsorbent has adsorbed drugs, its recycling and reuse become challenging34. To address this problem, magnetic compounds have been developed to facilitate the removal of pollutants from solutions. Although many studies have explored the application of aluminum-based MOFs in aquatic environments, there are few reports on the incorporation of magnetic materials. ZnFe2O4 nanomaterials have attracted considerable attention due to their excellent biocompatibility, low toxicity and superior degradability35.
In this work, a novel magnetism induced composite ZnFe2O4@MIL-53(Al)-NH2 was successfully synthesized via an in-situ growth method. Its morphological structure, thermal stability and magnetic properties were systematically characterized. Unlike previously reported magnetic MOFs for DS removal, which often exhibit limited adsorption capacity or poor magnetic recovery, the ZnFe2O4@MIL-53(Al)-NH2 composite combines the high surface area and functional tunability of MOFs with the efficient separability of ZnFe2O4 nanoparticles for the first time. The influence of environmental factors such as pH, salt concentration and temperature on the DS adsorption capacity of ZnFe2O4@MIL-53(Al)-NH2 was assessed. The underlying adsorption mechanisms of DS were further elucidated through kinetic and thermodynamic analyses, as well as X-ray photoelectron spectroscopy (XPS) analysis. This work aims to provide valuable experimental insights for the development of magnetism induced high-performance MOF-based adsorbents for environmental remediation applications.
Materials and methods
Experimental reagents
The reagents in this study were used as received without further purification. The details of reagents used were provided in the Supplementary Information (SI), including 2-amino-terephthalic acid (C8H7NO4), Aluminium chloride hexahydrate (AlCl3·6H2O), N, N-dimethylformamide (DMF), Acetone (C3H6O), Anhydrous zinc chloride (ZnCl2), Ferric chloride hexahydrate (FeCl3·6H2O), Ethanol (C2H6O), Ethylene glycol (EG), Concentrated ammonia water, Tetraethyl silicate (C8H20O4Si), Polyvinyl Pyrrolidone (PVP), Diclofenac Sodium (DS), Potassium hydroxide (NaOH), Hydrochloric acid (HCl), Ultrapure water, as listed in Table S1.
Preparation of MIL-53(Al)-NH2
MIL-53(Al)-NH2 was synthesized through solvothermal method36. In brief, 560 mg NH2-H2BDC and 760 mg AlCl3·6H2O were dissolved in 30 mL DMF under stirring. The resulting solution was transferred to a 100 mL PTFE reactor, and heated at 150 °C for 24 h in an oven. After the reaction was finished, the solid product was placed in 80 mL DMF and refluxed for 8 h The resulting mixture was centrifuged at 1000 rpm for 3 min, followed by three successive washes with 30 mL of acetone each to remove residual impurities. The yellowish powder obtained with an approximate yield of ~ 85%, was labeled as MIL-53(Al)-NH2 after being dried under vacuum at 80 °C for 12 h. Subsequent centrifugation, washing, and drying steps were carried out following the same procedures unless otherwise specified.
Preparation of ZnFe2O4
A mixture containing 40 mL ethylene glycol, 1.4 g ferric chloride hexahydrate, 0.35 g zinc chloride and 3.6 g sodium acetate was prepared and heated in a water bath at 50 °C for 1 h under continuous stirring. The resulting solution was then transferred to a Teflon-lined autoclave and subjected to hydrothermal treatment at 200 °C for 12 h37. The resulting mixture was centrifuged, thoroughly washed, and dried. The product exhibited a yield of approximately ~ 80% and was designated as ZnFe2O4.
Preparation of ZnFe2O4@MIL-53(Al)-NH2
First, 0.35 g ZnFe2O4 was added to a beaker containing 250 mL ethanol and 55 mL water, and then 1 mL concentrated ammonia and 1 mL tetraethyl orthosilicate were introduced. The mixture was stirred at room temperature for 10 h. Subsequently, 0.3 g the resulting product and 1 g polyvinylpyrrolidone were added to 100 mL trichloromethane, and stirred for another 10 h. The mixture was then centrifuged, washed and dried to get the modified ZnFe2O4 for reserve use. 0.0031 mol NH2-BDC and 0.0031 mol AlCl3·6H2O were dissolved in 30 mL DMF under stirring. 0.1 g modified ZnFe2O4 was added to the solution, and the resulting mixture was sonicated for 30 min. The solution was then transferred to the reaction kettle until the reaction was completed for 12 h. The mixture was subsequently centrifuged, washed, and dried to obtain the final product with an approximate yield of ~ 82%, ZnFe2O4@MIL-53(Al)-NH2.
Characterization of materials
The surface structure and physicochemical properties of the samples were evaluated through various characterization and performance study. High-resolution electron microscopy (HRTEM) was employed to observe the surface morphology of the materials, while Fourier transform infrared spectroscopy (FTIR) was used to identify the characteristic functional groups. The crystal structure of the samples was determined via X-ray diffraction (XRD) over a 5° ~ 50° range. The specific surface area and pore size distribution were assessed using the Brunauer-Emmett-Teller (BET) method, and XPS was utilized to analyze the chemical composition of the materials. The ion leaching from the prepared magnetic MOF-based adsorbent during the adsorption process was studied using the inductively coupled plasma optical emission spectrometry (ICP-OES) analysis.
Adsorption experiment
A series of constant-temperature static adsorption experiments were conducted using a temperature-controlled shaking apparatus, and the adsorption mechanism was further investigated. 10 mg adsorbent was added to 50 mL DS solution at a specified concentration to assess the effects of various factors, including adsorption time, salt concentration, and initial pH on the adsorption process. Thermodynamic and kinetic studies were conducted under different initial concentrations and adsorption times. The adsorption capacity was calculated using the following formula37:
where C0 represents the initial concentration of the DS (mg/L), Ce is the concentration of DS at any given time point (t) or equilibrium point (mg/L), V denotes the volume of the DS solution (L), and m is the mass of the ZnFe2O4@MIL-53(Al)-NH2 (g). In this study, all adsorption experiments were conducted in triplicate, and the average values were calculated to ensure accuracy and consistency.
Results and discussion
Characterization of materials
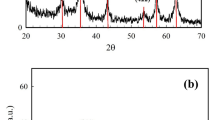
ZnFe2O4@MIL-53(Al)-NH2 was synthesized using the solvothermal method. The crystal structure of the ZnFe2O4@MIL-53(Al)-NH2 composite was characterized by XRD. As shown in Fig. 1, the XRD pattern of ZnFe2O4@MIL-53(Al)-NH2 exhibits five characteristic diffraction peaks at 30.02°, 35.46° and 42.99°, which correspond to the (220), (311) and (400) planes, respectively. Furthermore, after combining ZnFe2O4 with MIL-53(Al)-NH2, the diffraction peaks observed at 8.93°, 10.16°, 15.22°, 17.87°, 20.47° and 27.04° closely match the characteristic peaks of MIL-53(Al)-NH2 reported in the literature38,39,40. This confirms the successful synthesis of the ZnFe2O4@MIL-53(Al)-NH2 composite.
XRD patterns of (a) ZnFe2O4@MIL-53(Al)-NH2, (b) MIL-53(Al)-NH2 obtained, (c) MIL-53(Al)- NH2 standard card (CCDC NO.901254) and (d) ZnFe2O4 standard card (PDF74#2397).
The thermal stability of ZnFe2O4@MIL-53(Al)-NH2 was evaluated through its thermogravimetric curve. Fig. S1 displays the thermogravimetric (TG) analysis along with the corresponding mass change rate of ZnFe2O4@MIL-53(Al)-NH2. From Fig. S1, the mass loss of ZnFe2O4@MIL-53(Al)-NH2 can be categorized into three distinct stages. The first stage, occurring near 100 °C, involves a 19.72% mass loss, attributed to the desorption of adsorbed water. The second stage, within the temperature range of 150 ~ 520 °C, exhibits a 32.84% mass loss, characterized by a rapid and significant reduction in mass due to the decomposition of organic linkers in ZnFe2O4@MIL-53(Al)-NH2, as supported by previous reports on the thermal behavior of similar MOFs41,42. The third stage, observed between 550 and 850 °C, corresponds to the mass loss caused by the further decomposition and collapse of the ZnFe2O4@MIL-53(Al)-NH2 framework at elevated temperatures, suggesting the ideal thermostability of ZnFe2O4@MIL-53(Al)-NH2 obtained.
Figure 2 presents the HRTEM characterization results of ZnFe2O4@MIL-53(Al)-NH2. The HRTEM images reveal that after the incorporation of ZnFe2O4 particles, MIL-53(Al)-NH2 was surrounded by spherical ZnFe2O4 particles, resulting in the formation of a binary composite structure. These ZnFe2O4 spheres did not fully encapsulate MIL-53(Al)-NH2, leaving portions of the ZnFe2O4 exposed. The exposed ZnFe2O4 particles not only confirm their presence within the ZnFe2O4@MIL-53(Al)-NH2 composite but also contribute to surface roughness, thereby providing more active sites available for the DS adsorption. Furthermore, the elemental composition of ZnFe2O4@MIL-53(Al)-NH2, as shown in Fig. S2, reveals the uniform distribution of carbon (red), nitrogen (green), oxygen (blue), iron (purple), aluminum (orange), and zinc (yellow) within the sample.
HRTEM images of ZnFe2O4@MIL-53(Al)-NH2.
To elucidate the interaction mechanism between DS and ZnFe2O4@MIL-53(Al)-NH2 composite, FTIR spectra of MIL-53(Al)-NH2, ZnFe2O4@MIL-53(Al)-NH2 before and after DS adsorption were compared (Fig. 3). In spectrum (a), pristine MIL-53(Al)-NH2 exhibits characteristic bands at 3498.17 cm− 1 and 3381.93 cm− 1, corresponding to the O–H and N–H stretching vibrations, respectively43. In spectrum (b), after the incorporation of ZnFe2O4, these peaks are still observed but with a slight decrease in intensity, indicating minor alterations in the local chemical environment. Following DS adsorption, as shown in spectrum (c), both bands exhibit slight shifts—from 3498.17 to 3494.82 cm− 1, and from 3381.93 to 3386.40 cm− 1, indicating the formation of hydrogen bonds between DS molecules and the –OH/–NH2 functional groups of the framework. The peak at 1669.85 cm− 1 corresponds to C = O stretching of the carboxylate groups in the framework37,44, which shifts to1575.78 cm− 1 after DS adsorption. In spectrum (c), the peaks at 1499.78 and 1336.60 cm− 1 are characteristic of the aromatic skeleton and C = C stretching vibrations of DS36,45,46. These spectral changes indicate π–π interactions between the benzene rings of DS and the organic linkers of MIL-53(Al)-NH2. Furthermore, modifications in the fingerprint region, particularly around 1258.37 and 775.53 cm⁻¹, are indicative of C–N stretching and aromatic C–H bending, and imply possible electrostatic interactions between the carboxylate moieties of DS and the metal sites or other functional groups within the MOF matrix45. ZnFe2O4 provides additional chemically active adsorption sites, where the exposed Zn2+ and Fe3+ centers enhance electrostatic attraction toward the anionic carboxylate (–COO−) groups of DS, while its surface hydroxyls and metal centers facilitate surface complexation and coordination bonding with DS oxygen atoms through inner-sphere interactions20,22. These spectral variations collectively confirm the DS adsorption of ZnFe2O4@MIL-53(Al)-NH2 composite via multiple interaction mechanisms, including hydrogen bonding, π–π stacking, and electrostatic interactions.
FTIR spectra of (a) MIL-53(Al)-NH2, (b) ZnFe2O4@MIL-53(Al)-NH2 before DS adsorption, and (c) ZnFe2O4@MIL-53(Al)-NH2 after DS adsorption.
The Fig. S3 presents the wide-scan XPS spectra of ZnFe2O4@MIL-53(Al)-NH2, and Fig. 4 displays the corresponding XPS spectrum, including high-resolution spectra for C 1s (4a), N 1s (4b), O 1s (4c), Fe 2p (4d), Al 2p (4e), and Zn 2p (4f). The XPS analysis reveals that the C 1s spectrum exhibits a dominant peak at 284.8 eV, corresponding to C = C bond, and the peaks observed at 286.2 and 289.2 eV are attributed to C = O and C–O bonds, respectively47. The N 1s spectrum is deconvoluted into two peaks at 399.5 eV and 400.2 eV, corresponding to –NH2 and C–NH2 bonds, respectively48,49. The O 1s spectrum exhibits three distinct peaks, specifically C = O at 531.0 eV, C–O at 532.5 eV and C–O–C at 533.5 eV50.The Fe 2p spectrum exhibits three peaks at 710.9, 716.4 and 724.5 eV, corresponding to Fe 2p1/2, satellite Fe³⁺, and Fe 2p3/2, respectively51. In the case of Al 2p spectrum, the characteristic Al 2p3/2 peak is observed at 74.9 eV52,53. The Zn 2p spectrum displays two distinct peaks at 1038.53 and 1022.02 eV, which are attributed to the Zn 2p1/2 and Zn 2p3/2, respectively. The aforementioned results indicate the successful synthesis of the ZnFe2O4@MIL-53(Al)-NH254.
XPS spectrum of (a) C 1s, (b) N 1s, (c) O 1s, (d) Fe 2p, (e) Al 2p and (f) Zn 2p about ZnFe2O4@MIL-53(Al)-NH2.
The nitrogen adsorption-desorption isotherms and pore size distributions for both MIL-53(Al)-NH2 and ZnFe2O4@MIL-53(Al)-NH2 are presented in Fig. 5. The two adsorption-desorption isotherms are similar and classified as the type II isotherms. In the low-pressure region (P/P0 < 0.05), rapid gas absorption reaches equilibrium, indicating the presence of micropores. Hysteresis loops observed in the nitrogen adsorption-desorption isotherms at higher pressures suggest that both MIL-53(Al)-NH2 and ZnFe2O4@MIL-53(Al)-NH2 possess mesoporous structures.
(a) N2 adsorption and desorption curve and (b) pore size distribution.
The pore size distribution diagram indicates that the pore sizes of both samples are primarily concentrated in the range of 2 ~ 5 nm. The specific surface area and pore size-related parameters for MIL-53(Al)-NH2 and ZnFe2O4@MIL-53(Al)-NH2 are summarized in Table 1. Upon the incorporation of ZnFe2O4, the specific surface area of the composite decreased slightly, from 349.01 m²/g to 346.51 m²/g. The large specific surface area and appropriate pore size can accommodate more effective binding sites, which is beneficial for the DS adsorption process.
As presented in Fig. 6, the hysteresis loop of ZnFe2O4@MIL-53(Al)-NH2 was measured under a cyclic magnetic field ranging from − 20 to + 20 kOe. The magnetization curve demonstrates unique superparamagnetic behavior, with coercivity and remanence values near zero. Furthermore, the experimental results indicate that the saturation magnetization of ZnFe2O4@MIL-53(Al)-NH2 is 3.95 emu/g, significantly enhancing its recovery efficiency in recycling. With the aid of an external magnetic field, ZnFe2O4@MIL-53(Al)-NH2 was separated easily from the aqueous solution in about 60 s, highlighting its ideal potential for efficient recycling and reuse.
Magnetic hysteresis loop of ZnFe2O4@MIL-53(Al)-NH2.
The ion leaching from the synthesized magnetic MOF-based adsorbent during the adsorption process was studied for evaluating its environmental safety and long-term applicability. The ICP-OES analysis was conducted to quantify the leaching of Zn2+, Fe3+, and Al3+ ions from ZnFe2O4@MIL-53(Al)-NH2 during the DS adsorption process. After the adsorption equilibrium was reached, the supernatant was filtered and analyzed. The concentrations of leached Zn, Fe, and Al ions were found to be below 0.1 mg/L in all cases, which are within acceptable limits and indicate negligible leaching under the experimental conditions. These results demonstrate that the composite exhibits high chemical stability in aqueous media, with minimal risk of secondary contamination. The detailed data are provided in the Table S2.
Effect of pH on adsorption
The pH of the solution significantly influences the chemical properties of the adsorbent surface and the existing form of DS in the solution. At pH values below 4.15, DS exists as a neutral molecule, while in the pH range of 4.15 ~ 10, DS carries a negative charge55. The DS solution with a pH range of 4 ~ 10 was selected for this study. The pH was adjusted using hydrochloric acid or sodium hydroxide solutions as required. Fig. S4 reveals that ZnFe2O4@MIL-53(Al)-NH2 carries a positive charge within the pH range of 4 ~ 10. The DS adsorption capacity of ZnFe2O4@MIL-53(Al)-NH2 decreases as the pH increases, with the maximum uptake (180.02 mg/g) observed at pH 4. This trend is primarily attributed to electrostatic interactions: at pH 4, the strong electrostatic attraction between the negatively charged DS molecules and the positively charged adsorbent surface promotes maximum adsorption. As the pH increases, the surface positive charge diminishes due to deprotonation of functional groups, weakening the electrostatic attraction. Additionally, the increasing concentration of hydroxide ions at higher pH may compete with DS for adsorption sites, further reducing adsorption efficiency. It should be noted that zeta potential at pH 6.44 is below pH = 8, while the adsorption capacity exhibits the opposite trend. This is because electrostatic attraction is not the only mechanism involved in DS adsorption. In addition to electrostatic interactions, π–π stacking between the aromatic rings of DS and the organic linkers of MIL-53(Al)-NH2, as well as hydrogen bonding between –NH2/–OH groups and the carboxylate moieties of DS, also contribute significantly to adsorption. At intermediate pH (around 6.44), these non-electrostatic interactions partially compensate for the reduced surface charge, leading to a relatively higher adsorption capacity than expected from zeta potential alone. Therefore, the adsorption behavior reflects the synergistic contributions of multiple mechanisms rather than being governed exclusively by electrostatic attraction.
Effect of salt concentration on adsorption
To evaluate the effect of salt concentration on the adsorption performance, the DS adsorption of ZnFe2O4@MIL-53(Al)-NH2 was tested at 298 K under NaCl concentrations ranging from 0 ~ 1 mol/L. As shown in Fig. S5, the DS adsorption capacity of ZnFe2O4@MIL-53(Al)-NH2 increases gradually when the salt concentration rises from 0 to 0.01 mol/L. When the salt concentration was in the range of 0.01 ~ 1 mol/L, the DS adsorption amount increases rapidly before transitioning to a slower rate. The positive effect of co-existing ion on the adsorption process indicates the involvement of electrostatic attraction or ion exchange during the DS adsorption process of ZnFe2O4@MIL-53(Al)-NH2. Because this effect was strong thus suggesting that the primary mechanism of DS adsorption was electrostatic attraction, the hydrogen bonding and π-π interactions played a secondary role.
Adsorption isotherms, kinetic and thermodynamic analyses
Isotherm model fitting analysis
The effect of DS concentration on the adsorption performance of ZnFe2O4@MIL-53(Al)-NH2 was examined using the DS solutions with concentrations ranging from 20 to 160 mg/L at three different temperatures including298, 308 and 318 K. As shown in Fig. 7, the DS adsorption capacity of ZnFe2O4@MIL-53(Al)-NH2 increases with the rising temperature (equilibrium uptake Qe from 194.66 mg/g to 338.52 mg/g), demonstrating an endothermic adsorption process. As shown in Table S3, the isothermal data for DS adsorption of ZnFe2O4@MIL-53(Al)-NH2 were analyzed using four adsorption isotherm models: Langmuir, Freundlich, Temkin and Dubinin-Radushkevich. At 298 K, the maximum DS adsorption capacity of ZnFe2O4@MIL-53(Al)-NH2 calculated by the Langmuir model reaches 217.85 mg/g, which is much higher than those of recent adsorbents including LMNA (106.4 mg/g)56, Ni0.5Zn0.5Fe2O4 (52.91 mg/g)57, FZRBC-DES15 (24.33 mg/g)58, Fe-Zn-SSSC (6.33 mg/g)59 and Na-X zeolite (6.68 mg/g)60 as well as many adsorbents listed in Table S4.
Influence of equilibrium temperature on DS adsorption and isothermal curve at pH 6.44.
The Freundlich model offered a better fit to the experimental data than the Langmuir model, as evidenced by consistently higher correlation coefficients across all tested temperatures. This superior performance can be attributed to the underlying assumptions of each model. The Langmuir model is based on the assumption of monolayer adsorption onto a homogeneous surface with uniform adsorption sites and identical adsorption energies. In contrast, the ZnFe2O4@MIL-53(Al)-NH2 composite possesses a heterogeneous surface due to the presence of both metal oxide (ZnFe2O4) and organic linker components, which provide a variety of adsorption sites with different affinities for DS molecules. The Freundlich model accounts for this heterogeneity by allowing for multilayer adsorption and non-uniform distribution of adsorption heat and affinities over the surface. The favorable Freundlich exponent (1/n) values ranging from 0.31 to 0.38 further support the multilayer adsorption mechanism61. In the case of Dubinin-Radushkevich model, the E value is a critical indicator for classifying the adsorption type. Specifically, an E value below 8 kJ/mol signifies physical adsorption, values between 8 and 16 kJ/mol suggest ion exchange processes, and values exceeding 20 kJ/mol are indicative of chemical adsorption. The fitting analysis indicates that the DS adsorption of ZnFe2O4@MIL-53(Al)-NH2 is characterized by multimolecular layer adsorption, primarily governed by physical adsorption, which is in consistent with the aforementioned results.
Kinetic analysis
To evaluate the adsorption rate, the DS solutions at concentrations of 60 mg/L and 80 mg/L were used for adsorption over specified time intervals. The experimental results presented in Fig. 8 indicate that the DS adsorption capacity of ZnFe2O4@MIL-53(Al)-NH2 increases with longer contact time, and is influenced by the initial DS concentration. As the DS concentration rises, the adsorption capacity also increases, which can be attributed to the availability of active sites and the DS providing the driving force for mass transfer. As the adsorption process progresses, DS molecules rapidly occupy the binding sites on the surface of ZnFe2O4@MIL-53(Al)-NH2 leading to a gradual reduction in the number of available adsorption sites. Consequently, the adsorption process approaches equilibrium over time.
Kinetic fitting curves for DS adsorption at 298 K and pH 6.44.
To gain deeper insights into the adsorption mechanism, the kinetic data were analyzed through nonlinear fitting using four models including the Pseudo-first-order, Pseudo-second-order, Elovich, and Intraparticle diffusion models. The mathematical expressions for these models were presented in SI, and the corresponding fitting curves were displayed in Fig. 8. The parameter values fitted were summarized in Table S5. Kinetic analysis revealed that the pseudo-second-order model provided a significantly better fit to the experimental data than the pseudo-first-order model, as evidenced by higher correlation coefficients (R²) and excellent agreement between the calculated (Qe, cal = 291.87 mg/g) and experimental (Qe, exp = 290.11 mg/g) adsorption capacities. This strong consistency highlights the superior suitability of the pseudo-second-order model in describing the adsorption kinetics of DS onto ZnFe2O4@MIL-53(Al)-NH2, suggesting that the rate-limiting step is primarily governed by surface interactions rather than mass transfer from the bulk solution. While the pseudo-second-order model is often associated with chemisorption, in this context, its applicability indicates that the adsorption rate is more dependent on the availability of active sites on the adsorbent surface than on the concentration of DS in the bulk phase62.
To further investigate the adsorption kinetics, the Elovich model was employed to analyze the adsorption behavior of DS on ZnFe2O4@MIL-53(Al)-NH2. As shown in Table S5, the adsorption rate constant (α) was significantly greater than the desorption constant (β), indicating a rapid initial uptake of DS followed by a stabilized adsorption phase. This large α/β ratio suggests that a substantial number of active sites were readily accessible at the beginning of the process, facilitating fast adsorption.
Besides, the fitting results of Intraparticle diffusion model reveal that the DS adsorption process of ZnFe2O4@MIL-53(Al)-NH2 occurs in three distinct stages: surface interaction, intra-particle diffusion, and adsorption equilibrium63. In the initial stage, the concentration gradient of the DS solution acts as the driving force for the adsorption process. This continues until the adsorption on the outer surface of ZnFe2O4@MIL-53(Al)-NH2 reaches saturation, after which DS begins to diffuse into the particles. As the process progresses, the mass transfer resistance increases, resulting in a reduced adsorption rate until the dynamic adsorption-desorption equilibrium is achieved. The Fig. 8 indicates that the fitting curve does not pass through the origin, suggesting that the DS adsorption process is governed by both surface adsorption and intra-particle diffusion effect. Therefore, intra-particle diffusion is not the sole factor determining DS adsorption, other factors also contribute to the adsorption rate.
Thermodynamic analysis
The influence of temperature on DS adsorption was further evaluated by analyzing the corresponding thermodynamic parameters, which were calculated with the formula S11 and S12 in SI. As presented in Table 2, the negative value of ∆G indicates that the DS adsorption of ZnFe2O4@MIL-53(Al)-NH2 occurs spontaneously. As the adsorption temperature increases, the DS adsorption capacity also rises. Additionally, the positive values of ∆H and ∆S suggest that the DS adsorption process is endothermic, and driven by an increase in entropy.
Reusability of ZnFe2O4@MIL-53(Al)-NH2
The reusability of the adsorbent is a key consideration, both from an economic and environmental perspective. The recyclability of ZnFe2O4@MIL-53(Al)-NH2 was examined, as presented in Fig. 9. The ZnFe2O4@MIL-53(Al)-NH2 was recovered through magnetic separation and regenerated using the solution of 0.1 mol/L sodium hydroxide (NaOH) and deionized water as eluents. DS adsorption was carried out over six cycles, and no significant decrease in adsorption capacity was observed. This result suggests that ZnFe2O4@MIL-53(Al)-NH2 exhibits excellent material stability and that the majority of the adsorbed DS can be effectively desorbed. This means that the simple regeneration conditions are sufficient for the expected regeneration of the adsorbent. The excellent magnetism and recyclability make the ZnFe2O4@MIL-53(Al)-NH2 highly promising in practical application such as DS adsorption from wastewater.
Six consecutive adsorption–desorption cycles of ZnFe2O4@MIL-53(Al)-NH2 for the DS removal (C0 = 100 mg/L, T = 298 K, pH = 6.44, m = 100 mg).
Adsorption mechanism study
The DS adsorption of ZnFe2O4@MIL-53(Al)-NH2 involves a combination of mechanisms operating on both the MOF and the ZnFe2O4 phases. MIL-53(Al)-NH2 contributes π–π stacking between the aromatic linker and DS, and hydrogen bonding via –NH2/–OH functional groups, as evidenced by shifts in the N–H/O–H stretching and aromatic C = C vibrations in FTIR spectra. ZnFe2O4 contributes additional chemically active adsorption sites: exposed Zn2+ and Fe3+ centers increase electrostatic attraction toward the anionic –COO⁻ group of DS, while surface hydroxyls and metal centers can form surface complexation/coordination bonds with DS oxygens (inner-sphere interactions), as commonly observed for ferrite and metal-oxide adsorbents7,22. The heterogeneous MOF–ferrite interface further modifies local surface charge and increases site heterogeneity, which explains the superior Freundlich fitting and high adsorption of the composite. These combined interactions (electrostatic attraction, surface complexation, hydrogen bonding, and π–π stacking) produce a synergistic enhancement in adsorption capacity. The pH-dependent adsorption behavior confirms the role of electrostatic interactions, particularly in the pH 4 ~ 10 range where DS exists primarily in anionic form and the MOF surface remains positively charged. This is consistent with the zeta potential data (Fig. S4). The kinetic fitting to the pseudo-second-order model, associated with surface-mediated adsorption, indicates that the rate-limiting step is likely controlled by the interaction at active sites rather than simple diffusion. The three-stage adsorption process seen in the intraparticle diffusion model further confirms that both surface adsorption and pore diffusion contribute to the overall mechanism. Thermodynamic parameters (Table 2) reveal a positive ΔS and negative ΔG, indicating an entropy-driven, spontaneous adsorption process, which favors physical adsorption over chemisorption.
Taken together, the evidence suggests that DS adsorption on ZnFe2O4@MIL-53(Al)-NH2 proceeds primarily via electrostatic attraction, aided by π–π stacking, hydrogen bonding, and pore diffusion, forming multilayer coverage consistent with the Freundlich model. A schematic illustration of this synergistic mechanism is shown in Fig. 10.
Synergetic mechanisms between DS and ZnFe2O4@MIL-53(Al)-NH2.
Conclusion
In this work, a novel magnetism induced adsorbent, ZnFe2O4@MIL-53(Al)-NH2, was prepared and systematically evaluated for its efficacy in DS removal. The material exhibited abundant oxygen- and nitrogen-containing functional groups, contributing to strong interactions with the target pollutant via electrostatic attraction, hydrogen bonding, and π–π stacking. ZnFe2O4@MIL-53(Al)-NH2 demonstrated superior DS adsorption performance (217.85 mg/g at 298 K) compared to several benchmark materials, including LMNA (106.4 mg/g) and Ni0.5Zn0.5Fe2O4 (52.91 mg/g). In addition to its excellent adsorption capacity, the composite achieved rapid magnetic separation (~ 60 s), exhibited negligible ion leaching (< 0.1 mg/L), and retained most of its initial capacity after six reuse cycles. In the future, the scalable synthesis route and modular design of this MOF-based hybrid provide opportunities for tailoring its selectivity toward other emerging contaminants. Further exploration of its integration into advanced water treatment technologies such as magnetic separation systems or continuous-flow reactors could facilitate its translation from laboratory research to real-world applications.
Data availability
All data generated or analysed during this study are included in this published article and its Supplementary Information files.
References
Wan, L. & Wang, H. Control of urban river water pollution is studied based on SMS. Environ. Technol. Innov. 22, 101468. https://doi.org/10.1016/j.eti.2021.101468 (2021).
Krishnamoorthy, L. & Lakshmanan, V. R. Evaluating hydrogeochemistry and heavy metal contamination of groundwater at Ranipet environs: employing multivariate statistics, agricultural indices, and health risk assessment. Environ. Sci. Pollut. Res. 31 (19), 28253–28278. https://doi.org/10.1007/s11356-024-32928-4 (2024).
Bagherzadeh, S. B., Kazemeini, M. & Mahmoodi, N. M. Preparation of novel and highly active magnetic ternary structures (metal-organic framework/cobalt ferrite/graphene oxide) for effective visible-light-driven photocatalytic and photo-Fenton-like degradation of organic contaminants. J. Colloid Interface Sci. 602, 73–94. https://doi.org/10.1016/j.jcis.2021.05.181 (2021).
Kadadou, D., Tizani, L., Alsafar, H. & Hasan, S. W. Analytical methods for determining environmental contaminants of concern in water and wastewater. MethodsX 12, 102582. https://doi.org/10.1016/j.mex.2024.102582 (2024).
Mahmoodi, N. M., Ghadirli, M. M., Hayati, B., Mahmoodi, B. & Rabeie, B. Synthesis of ZIF-8 composite (g-C3N4@ZIF-8/Ag3PO4) as a catalyst for the malachite green and Tetracycline degradation. Inorg. Chem. Commun. 177 https://doi.org/10.1016/j.inoche.2025.114345 (2025).
Mazarji, M. et al. Synthesis, characterization, and enhanced photocatalytic dye degradation: optimizing graphene-based ZnO-CdSe nanocomposites via response surface methodology. J. Alloys Compd. 1010 https://doi.org/10.1016/j.jallcom.2024.177999 (2025).
Rabeie, B., Mahmoodi, N. M. & Mahkam, M. Morphological diversity effect of graphene quantum dot/MIL88A(Fe) composites on dye and pharmaceuticals (tetracycline and doxycycline) removal. J. Environ. Chem. Eng. 10 (5), 108321. https://doi.org/10.1016/j.jece.2022.108321 (2022).
Wydro, U. et al. A review on pharmaceuticals and personal care products residues in the aquatic environment and possibilities for their remediation. Sustainability 16 (1), 169. https://doi.org/10.3390/su16010169 (2023).
Yao, D. et al. The safety of consuming water dropwort used to purify livestock wastewater considering accumulated antibiotics and antibiotic resistance genes. Antibiot. (Basel). 11 (4), 428. https://doi.org/10.3390/antibiotics11040428 (2022).
Spitsov, D., Korobeynikova, E., Grigoriev, A. & Kondratenko, L. Features of Pesticide-Contaminated surface water. Asian J. Water Environ. Pollut. 19 (4), 41–47. https://doi.org/10.3233/ajw220054 (2022).
Feisal, N. A. S., Kamaludin, N. H., Ahmad, M. A. & Tengku Ibrahim, T. N. B. A comprehensive review of nanomaterials for efficient heavy metal ions removal in water treatment. J. Water Process. Eng. 64, 105566. https://doi.org/10.1016/j.jwpe.2024.105566 (2024).
Zhu, S. et al. Simultaneous production of clean water and organic dye from dyeing wastewater by reusable lignin-derived porous carbon. Ind. Crops Prod. 187, 115314. https://doi.org/10.1016/j.indcrop.2022.115314 (2022).
Priya, A. K., Gnanasekaran, L., Rajendran, S., Qin, J. & Vasseghian, Y. Occurrences and removal of pharmaceutical and personal care products from aquatic systems using advanced treatment- A review. Environ. Res. 204(Pt C). 112298. https://doi.org/10.1016/j.envres.2021.112298 (2022).
Chen, R. et al. The plant streptolysin S (SLS)-associated gene B confers nitroaromatic tolerance and detoxification. J. Hazard. Mater. 433, 128779. https://doi.org/10.1016/j.jhazmat.2022.128779 (2022).
Niu, L., Liu, W., Juhasz, A., Chen, J. & Ma, L. Emerging contaminants antibiotic resistance genes and microplastics in the environment: introduction to 21 review articles published in CREST during 2018–2022. Crit. Rev. Environ. Sci. Technol. 52 (23), 4135–4146. https://doi.org/10.1080/10643389.2022.2117847 (2022).
Feng, P. et al. A review on gut remediation of selected environmental contaminants: possible roles of probiotics and gut microbiota. Nutrients 11 (1), 22. https://doi.org/10.3390/nu11010022 (2018).
Shakeel, A. et al. Polymer based nanocomposites: A strategic tool for detection of toxic pollutants in environmental matrices. Chemosphere 303 (1), 134923. https://doi.org/10.1016/j.chemosphere.2022.134923 (2022).
Rabeie, B. et al. Chitosan adorned with ZIF-67 on ZIF-8 biocomposite: A potential LED visible light-assisted photocatalyst for wastewater decontamination. Int. J. Biol. Macromol. 282 (6), 137405. https://doi.org/10.1016/j.ijbiomac.2024.137405 (2024).
Lam, V. T., Ngo, T. C. Q. & Bach, L. G. Facile fabrication of novel NiFe2O4@Carbon composites for enhanced adsorption of emergent antibiotics. Mater. (Basel). 14 (21). https://doi.org/10.3390/ma14216710 (2021).
Rabeie, B., Mahkam, M., Mahmoodi, N. M. & Lan, C. Q. Graphene quantum Dot incorporation in the zeolitic imidazolate framework with sodalite (SOD) topology: synthesis and improving the adsorption ability in liquid phase. J. Environ. Chem. Eng. 9 (6), 106303. https://doi.org/10.1016/j.jece.2021.106303 (2021).
Brillas, E. & Martínez-Huitle, C. A. Decontamination of wastewaters containing synthetic organic dyes by electrochemical methods. An updated review. Appl. Catal. B. 166–167. https://doi.org/10.1016/j.apcatb.2014.11.016 (2015).
Rabeie, B. & Mahmoodi, N. M. Environmentally friendly novel covalent organic framework composites as porous photocatalysts and adsorbents for Tetracycline and dyes (Congo red and methylene Blue) removal: green synthesis, kinetics, regeneration, and removal mechanisms. Appl. Mater. Today. 46, 102884. https://doi.org/10.1016/j.apmt.2025.102884 (2025).
Li, X., Li, L., Tang, L., Mei, J. & Fu, J. Unveiling combined ecotoxicity: interactions and impacts of engineered nanoparticles and PPCPs. Sci. Total Environ. 921, 170746. https://doi.org/10.1016/j.scitotenv.2024.170746 (2024).
Mo, J. et al. What approaches should be used to prioritize pharmaceuticals and personal care products for research on environmental and human health exposure and effects? Environ. Toxicol. Chem. 43 (3), 488–501. https://doi.org/10.1002/etc.5520 (2024).
Granada, J. B. R. et al. Response surface methodology optimization of sodium diclofenac adsorption using activated carbon derived from falcata tree sawdust. Ind. Crops Prod. 221, 119272. https://doi.org/10.1016/j.indcrop.2024.119272 (2024).
Lach, J. & Szymonik, A. Adsorption of diclofenac sodium from aqueous solutions on commercial activated carbons. Desalination Water Treat. 186, 418–429. https://doi.org/10.5004/dwt.2020.25567 (2020).
Collier, A. C. Pharmaceutical contaminants in potable water: potential concerns for pregnant women and children. EcoHealth 4 (2), 164–171. https://doi.org/10.1007/s10393-007-0105-5 (2007).
Contreras-Almazo, I. A. E. & Gonzalez-Renteria, M. Bivalvos, organismos Modelo En El Biomonitoreo Del Riesgo ecotoxicológico de Los antinflamatorios no esteroideos (AINE) Para Los ecosistemas acuáticos. Ecosistemas 31 (2), 2167–2167. https://doi.org/10.7818/ecos.2167 (2022).
Rigueto, C. V. T. et al. Production and environmental applications of gelatin-based composite adsorbents for contaminants removal: a review. Environ. Chem. Lett. 19 (3), 2465–2486. https://doi.org/10.1007/s10311-021-01184-0 (2021).
Fu, L., Yang, Z., Wang, Y., Li, R. & Zhai, J. Construction of Metal-Organic frameworks (MOFs)–Based membranes and their ion transport applications. Small Sci. 1 (2), 2000035. https://doi.org/10.1002/smsc.202000035 (2021).
Otun, K. O., Mukhtar, A., Nafiu, S. A., Bello, I. T. & Abdulsalam, J. Incorporation of redox-activity into metal-organic frameworks for enhanced supercapacitors: A review. J. Energy Storage. 84, 110673. https://doi.org/10.1016/j.est.2024.110673 (2024).
Kang, I. J., Khan, N. A., Haque, E. & Jhung, S. H. Chemical and thermal stability of isotypic metal-organic frameworks: effect of metal ions. Chemistry 17 (23), 6437–6442. https://doi.org/10.1002/chem.201100316 (2011).
Fateeva, A. et al. A water-stable porphyrin-based metal-organic framework active for visible-light photocatalysis. Angew Chem. Int. Ed. Engl. 51 (30), 7440–7444. https://doi.org/10.1002/anie.201202471 (2012).
Kumar, P. et al. Regeneration, degradation, and toxicity effect of mofs: opportunities and challenges. Environ. Res. 176, 108488. https://doi.org/10.1016/j.envres.2019.05.019 (2019).
Moise, S. et al. The cellular magnetic response and biocompatibility of biogenic zinc- and cobalt-doped magnetite nanoparticles. Sci. Rep. 7, 39922. https://doi.org/10.1038/srep39922 (2017).
Cheng, X. et al. Size- and morphology-controlled NH2-MIL-53(Al) prepared in DMF-water mixed solvents. Dalton Trans. 42 (37), 13698–13705. https://doi.org/10.1039/c3dt51322j (2013).
Li, C., Xiong, Z., Zhang, J. & Wu, C. The strengthening role of the amino group in Metal–Organic framework MIL-53 (Al) for methylene blue and malachite green dye adsorption. J. Chem. Eng. Data. 60 (11), 3414–3422. https://doi.org/10.1021/acs.jced.5b00692 (2015).
Mondal, P. & Purkait, M. K. Preparation and characterization of novel green synthesized iron-aluminum nanocomposite and studying its efficiency in fluoride removal. Chemosphere 235, 391–402. https://doi.org/10.1016/j.chemosphere.2019.06.189 (2019).
Mubashir, M., Yeong, Y. F., Chew, T. L. & Lau, K. K. Comparison of Post-Treatment methods on the performance of Hollow fiber membranes containing metal organic framework in gases separation. Ind. Eng. Chem. Res. 58 (17), 7120–7130. https://doi.org/10.1021/acs.iecr.8b05773 (2019).
Xie, D., Ge, X., Qin, W. & Zhang, Y. NH2-MIL-53(Al) for simultaneous removal and detection of fluoride anions. Chin. J. Chem. Phys. 34 (2), 227–237. https://doi.org/10.1063/1674-0068/cjcp2004054 (2021).
Majchrzak-Kucęba, I. & Bukalak-Gaik, D. Regeneration performance of metal–organic frameworks. J. Therm. Anal. Calorim. 125 (3), 1461–1466. https://doi.org/10.1007/s10973-016-5624-2 (2016).
Rehman, T. U. et al. Unveiling the MIL-53(Al) MOF: tuning photoluminescence and structural properties via volatile organic compounds interactions. Nanomaterials (Basel). 14 (5). https://doi.org/10.3390/nano14050388 (2024).
Lu, T. et al. Amino-Functionalized Metal-Organic frameworks Nanoplates-Based energy transfer probe for highly selective fluorescence detection of free Chlorine. Anal. Chem. 88 (6), 3413–3420. https://doi.org/10.1021/acs.analchem.6b00253 (2016).
Zhang, S., Zhou, L. & Chen, M. Amine-functionalized MIL-53(Al) with embedded ruthenium nanoparticles as a highly efficient catalyst for the hydrolytic dehydrogenation of ammonia borane. Royal Soc. Chem. Adv. 8 (22), 12282–12291. https://doi.org/10.1039/c8ra01507d (2018).
Rahimpoor, R., Firoozichahak, A., Alizadeh, S., Serkan, H. & Nematollahi, D. Application of MIL-53(Al)-NH2 as a dispersive Microsolid-Phase extraction material for determination of cyclophosphamide in urine by High-Performance liquid chromatography. ACS Omega. 7 (41), 36643–36652. https://doi.org/10.1021/acsomega.2c04660 (2022).
Feijani, E. A., Mahdavi, H. & Tavasoli, A. Poly(vinylidene fluoride) based mixed matrix membranes comprising metal organic frameworks for gas separation applications. Chem. Eng. Res. Des. 96, 87–102. https://doi.org/10.1016/j.cherd.2015.02.009 (2015).
Guo, S. et al. Construction of MOF/TiO2 nanocomposites with efficient visible-light-driven photocathodic protection. J. Electroanal. Chem. 880, 114915. https://doi.org/10.1016/j.jelechem.2020.114915 (2021).
Ji, C. et al. Mussel-inspired HA@TA-CS/SA biomimetic 3D printed scaffolds with antibacterial activity for bone repair. Front. Bioeng. Biotechnol. 11, 1193605. https://doi.org/10.3389/fbioe.2023.1193605 (2023).
Tran, T. V. et al. Effect of pyrolysis temperature on characteristics and Chloramphenicol adsorption performance of NH2-MIL-53(Al)-derived amine-functionalized porous carbons. Chemosphere 355, 141599. https://doi.org/10.1016/j.chemosphere.2024.141599 (2024).
Yao, H. et al. Metal-organic framework [NH2-MIL-53(Al)] functionalized TiO2 nanotube photoanodes for highly stable and efficient photoelectrochemical cathodic protection of nickel-coated Mg alloy. J. Mater. Sci. Technol. 182, 67–78. https://doi.org/10.1016/j.jmst.2023.09.038 (2024).
Yang, X., Pan, J., Hu, J., Zhao, S. & Cheng, K. MOF-derived La–ZnFe2O4@Fe3O4@carbon magnetic hybrid composite as a highly efficient and recyclable photocatalyst for Mycotoxins degradation. Chem. Eng. J. 467, 143381. https://doi.org/10.1016/j.cej.2023.143381 (2023).
Zhu, C. et al. Ultrathin fluorine-doped TiO2(B) nanosheets-anchored hierarchical cog wheel-shaped NH2-MIL-53(Al) for boosting photocatalytic activity. Chem. Eng. J. 460, 141849. https://doi.org/10.1016/j.cej.2023.141849 (2023).
Zhao, H. et al. Gear-shaped mesoporous NH2-MIL-53(Al)/CdS P-N heterojunctions as efficient visible-light-driven photocatalysts. Appl. Catal. B. 291, 120106. https://doi.org/10.1016/j.apcatb.2021.120106 (2021).
Chen, C., Huang, J. C. & Duh, J. G. Self-template fabrication of multi-scaled ZnFe2O4 microspheres and their excellent lithium-ion storage properties. J. Alloys Compd. 862, 158342. https://doi.org/10.1016/j.jallcom.2020.158342 (2021).
Malakootian, M. & Asadzadeh, S. N. Oxidative removal of Tetracycline by Sono Fenton-like oxidation process in aqueous media. Desalination Water Treat. 193, 392–401. https://doi.org/10.5004/dwt.2020.25810 (2020).
Ye, X., Li, Y., Lin, H., Chen, Y. & Liu, M. Lignin-Based magnetic nanoparticle adsorbent for diclofenac sodium removal: adsorption behavior and mechanisms. J. Polym. Environ. 29 (10), 3401–3411. https://doi.org/10.1007/s10924-021-02127-0 (2021).
Mohammadi, Z., Kelishami, A. R. & Ashrafi, A. Application of Ni0.5Zn0.5Fe2O4 magnetic nanoparticles for diclofenac adsorption: isotherm, kinetic and thermodynamic investigation. Water Sci. Technol. 83 (6), 1265–1277. https://doi.org/10.2166/wst.2021.049 (2021).
Hinsene, H., Bhawawet, N. & Imyim, A. Rice husk Biochar doped with deep eutectic solvent and Fe3O4/ZnO nanoparticles for heavy metal and diclofenac removal from water. Sep. Purif. Technol. 339, 126638. https://doi.org/10.1016/j.seppur.2024.126638 (2024).
Çifçi, D. İ. Efficient removal of paracetamol, diclofenac sodium, and Tetracycline using green synthesized Fe-zn co-doped sunflower seed shells. Sep. Sci. Technol. 60 (1), 21–30. https://doi.org/10.1080/01496395.2024.2418288 (2024).
Medykowska, M., Wiśniewska, M., Szewczuk-Karpisz, K., Panek, R. & Franus, M. Simultaneous removal of inorganic and organic pollutants from multicomponent solutions by the use of zeolitic materials obtained from fly ash waste, Clean Technol. Environ. Policy 25(4) 1133–1148. https://doi.org/10.1007/s10098-022-02429-4. (2022).
Sasaki, T. & Tanaka, S. Magnetic separation of cesium ion using Prussian blue modified magnetite. Chem. Lett. 41 (1), 32–34. https://doi.org/10.1246/cl.2012.32 (2012).
Li, B. et al. Environmentally friendly chitosan/PEI-grafted magnetic gelatin for the highly effective removal of heavy metals from drinking water. Sci. Rep. 7, 43082. https://doi.org/10.1038/srep43082 (2017).
Yu, Y., Wang, C., Guo, X. & Chen, J. P. Modification of carbon derived from sargassum sp. by lanthanum for enhanced adsorption of fluoride. J. Colloid Interface Sci. 441, 113–120. https://doi.org/10.1016/j.jcis.2014.10.039 (2015).
Acknowledgements
This work is supported by the Expert Workstation of Jiang Yong Yunnan Province (202305AF150048), the Central Government Guidance Fund for Local Scientific and Technological Development (202407AB110009) and 2024 Expert grassroots scientific research workstation in Yunnan Province.
Author information
Authors and Affiliations
Contributions
Qicui Wang: Methodology, Validation, Investigation, Writing - review & editing. Juan Yao: Investigation, Characterization. Caixia Fan: Investigation, Characterization. Lanmeng Wei: Writing - review. Xiangfu Gu: Investigation, Validation. Pihui Yang: Methodology, Validation. Yong Liu: Methodology, Validation. Jieli He: Validation, Investigation. Anzhong Peng: Methodology, Supervision, Project administration, Funding acquisition. Kezhen Qi: supervision, Project administration, Resources. Haifeng Li: Methodology, Supervision, Funding acquisition.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Wang, Q., Yao, J., Fan, C. et al. Magnetically active MOF-based adsorbent for efficient diclofenac sodium adsorption. Sci Rep 15, 43323 (2025). https://doi.org/10.1038/s41598-025-27317-x
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-27317-x