Abstract
Faunal habitat selection, or the disproportionate use of available resources, is closely linked to habitat composition and configuration across a seascape. However, the drivers of habitat selection operate across multiple scales and require a hierarchical approach to study. This study combines acoustic telemetry, field survey data, remote sensing, and machine learning to investigate the multi-scale (seascape and patch) habitat selection of spotted seatrout (Cynoscion nebulosus) in Florida Bay, Everglades National Park, USA. Spotted seatrout responded to both scales, as there were three patch-scale (Halodule cover, standard deviation of submerged aquatic vegetation (SAV) cover, and SAV species richness) and one seascape-scale (patch density) predictor in the top four. However, responses were scale-specific, exhibiting logistic responses to seascape-level variables and optimal (specific-range) responses to patch-level characteristics. This study highlights the importance of investigating habitat selection across multiple scales as climate change alters not only species ranges, but local seascapes as well.
Similar content being viewed by others
Introduction
Faunal habitat selection, or the disproportionate use of available conditions or resources due to perceived risks and rewards1, is a vital ecological process, expected to operate at multiple levels of spatial organization2,3. At the broadest level, populations of organisms select their ecological range (first-order selection) based on species-specific metabolic constraints as well as the availability and amount of essential habitat needed to survive2. Within this ecological range, individuals select their home range (second-order selection) based on habitat requirements, as well as other density-dependent ecological processes such as competition2,4,5. Lastly, third-order habitat selection of an individual refers to the use of various habitat patches, and particular features of those patches, within their home range (e.g., central place foraging, the use of core vs. edges and corridors2,6,7,8). Third-order habitat selection operates over multiple spatial and temporal scales dependent upon the state of the organism, as well as life history requirements.
Though scale is often cited as a central theme in ecology9, research focusing on multi-scale habitat selection has been lacking and forms a considerable knowledge gap. For example, McGarigal et al.10 found that of 859 movement ecology studies (both terrestrial and aquatic) published between 2009 and 2014, only 20% examined habitat selection across multiple scales. In their review, the authors confirmed the importance of scale in habitat selection but cautioned researchers about the difference between ‘level’ and ‘scale’. They put forth that many studies attempting to measure habitat selection over multiple scales do not use the correct framework or do not optimize the variables; however, they identified certain studies that appropriately utilize the multi-scale framework. One example is Leblond et al.11, who used GPS positions of woodland caribou (Rangifer tarandus caribou) to investigate landscape and local habitat selection across seasons. This study stresses that not addressing the landscape context within habitat selection studies will lead to biased inferences due to how landscapes constrain the choices available to the organisms.
In marine systems, third-order habitat selection is studied through the lens of seascape ecology, or the study of patch configuration and composition of seagrasses, coral reefs, and other marine ecosystems12,13. In these seascapes, third-order habitat selection can be conceptualized as occurring within two primary spatial contexts14,15: the patch scale and the surrounding spatially heterogeneous mosaic (hereafter, the seascape scale12,16). The patch scale refers to a discrete, relatively homogeneous unit of habitat17 typically evaluated at fine spatial extents (e.g., 0.1–10 m2) and can be characterized by biotic and abiotic structural features (e.g., seagrass density, rugosity, species richness14,17,18). In contrast, the seascape scale is the broader matrix of habitats within which patches are embedded. This seascape scale is usually measured using configurational metrics such as patch size, shape, connectivity, and other aspects of spatial arrangement19,20,21.
The patch-matrix structure of marine systems is known to govern multiple ecological processes16 and is expected to strongly influence multi-scale habitat selection as animals move through seascapes tracking resources, risks, and conditions22,23. For example, prior studies have linked marine faunal recruitment, density, movement, and assemblage structure to within-patch compositional attributes, such as the density of submerged aquatic vegetation (SAV24,25,26), canopy height27,28, and vegetation species identity29,30,31,32. At the seascape scale, previous research has highlighted the importance of seascape configuration, including patch size33,34,35, edge characteristics34,36,37,38, and spatial proximity to adjacent habitats39,40,41. However, few studies have attempted to examine habitat selection at multiple scales of spatial organization. An exception is Pittman et al.20, who show that faunal density was positively related with seagrass cover at both patch and seascape scales, but the relation was linear at the seascape scale and showed a threshold at the patch scale (a sharp decline below 20% seagrass cover). In lieu of multi-scale studies, reviews that compare single scale studies show that seascape-faunal interactions are distinct at the patch vs. seascape scales. For instance, Yeager et al.42 found that fragmentation has a positive effect on faunal biomass at the seascape scale but no effect at the patch scale, and Yarnell et al.43 showed that edge effects at the patch scale were more influential on fish biomass than fragmentation at the seascape scale.
Residential species inhabiting shallow coastal habitats, such as seagrass meadows, provide an ideal opportunity to map and investigate the contextual nature of multi-scale habitat selection. One such species is spotted seatrout (Cynoscion nebulosus; hereafter seatrout), a coastal mesoconsumer that exhibits a strong affinity for seagrass habitats and relatively small home ranges44,45. These traits, coupled with their high site fidelity46,47, make seatrout particularly well-suited to further evaluate how habitat selection varies across the patch and seascape scale. In this study, we examined single level, multi-scale habitat selection10 in seatrout using Resource Selection Functions (RSFs) derived from random forest (RF) models applied to passive acoustic telemetry data48 and a suite of field-based and remotely sensed habitat metrics. By single level, multi-scale habitat selection, following McGarigal et al.10, we refer to applying a single modeling approach to a hierarchically-structured habitat. We hypothesize that (1) seatrout habitat selection is related to both patch and seascape scale characteristics but will respond more to patch scale variables due to the importance of edge effects43 and foraging strategy (ambush predators49); and (2) seatrout habitat selection is stronger in areas with high spatial heterogeneity (e.g., greater diversity of SAV species and higher patchiness50,51).
Methods
Study site: Florida Bay, Everglades National Park (ENP)
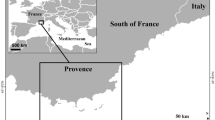
Florida Bay, the largest estuary in Florida (2200 km2), lies at the southern end of the Florida peninsula within Everglades National Park (ENP; Fig. 1) and supports a highly valuable recreational fishery that contributed $439 million and over 4100 jobs to the local economy in 2015–201652. The bay consists of shallow basins, mud banks, mangrove islands, and tidal channels, with seagrass—primarily Thalassia testudinum (turtle grass; hereafter Thalassia)—dominating up to 95% of the benthos53. However, anthropogenic water management practices, including upstream channelization and impoundment, have reduced freshwater inflows and intensified Florida Bay’s naturally high water residence times, leading to periods of hypersalinity, prolonged anoxia, and recurrent algal blooms53,54,55,56,57,58. These conditions have made the northcentral portion of the bay particularly vulnerable to large-scale seagrass die-offs, with major events recorded in 1987–1991 and again in 201559,60,61. Although the bay recovered by 2010 after the first event, the more recent die-off has left the northcentral region in a state of recovery. This state of recovery is characterized by pioneer species such as Halodule wrightii (hereafter Halodule) and calcareous green algae replacing the bare benthos prior to transitioning back toward climax communities dominated by Thalassia61,62,63. Rankin Basin in northcentral Florida Bay was selected as the focal site for this study because it was heavily impacted by the 2015 die-off, is undergoing active recovery, serves as a key fishing location, and has long-term seagrass monitoring (Fig. 1).
Map of acoustic receiver array in Rankin Basin, Florida Bay, Everglades National Park (ENP). In the insert, green shading represents ENP, blue shading Florida Bay, and the red box represents the area covered by the main map. Light blue on the main map represents Rankin Basin. Acoustic receiver locations (a mix of Innovasea VR2W and VR2Tx) are indicated by dark blue dots on the main map.
Study species: spotted seatrout
Spotted seatrout is a popular sportfish throughout all of its range, from Massachusetts to Florida and throughout the Gulf of Mexico45,49. In fact, they are the top saltwater recreational fishery, with 54 million fish caught each year64. In southwest Florida, seatrout are a top 4 targeted fish species in Everglades National Park, with approximately 4.19 million fish caught and released alive and 457,000 harvested65,66,67. Seatrout are estuary-dependent species, spending their entire life within a single estuarine system, resulting in each estuary in Florida harboring a unique subpopulation45,68,69. However, they are highly mobile within their natal estuary due to foraging and spawning needs46,47,50,70.
Within these estuaries, seatrout have been shown to be reliant on seagrass throughout their life cycle, feeding on seagrass-associated species throughout their ontogeny44,71,72. Previous studies have primarily used catch data to examine seatrout habitat associations45; however, recent studies have investigated seatrout distribution, movement and habitat use using acoustic telemetry (e.g., examining temperature effects45,50 and spawning activity46,70,73,74. For example, Moulton et al.46 used fine-scale acoustic telemetry to investigate habitat partitioning between seatrout and another estuarine fish, Red Drum (Sciaenops ocellatus). Seatrout used deeper water than Red Drum and forage over seagrass during the day. Unknowns remain, however, on habitat selection by seatrout, particularly in relation to the effects of seagrass die-off events.
Mapping habitat variables
Habitat variables were mapped at the patch and seascape scale to characterize the structural features likely to form seatrout habitat. Patch scale variables were collected in the field and included percent cover of total SAV, percent cover of Thalassia and Halodule, standard deviation of percent cover, and number of SAV plant species. These variables were mapped annually (n = 3: 2020–2022) using a series of kriging models75 predicted at 75 m to match the average 70% detection range of the acoustic array. The kriging interpolation maps were evaluated based on cross-validation and semivariogram comparison with the best-fit model selected using lowest root-mean square error, resulting in the selection of the ordinary kriging maps. Mapping was performed in the Geostatistical Wizard extension of ArcGIS Pro. The data informing the kriging came from two sources. The first included SAV surveys conducted around each receiver, which measured percent cover, standard deviation of percent cover, and SAV species richness. The second source was data provided by the Fisheries Habitat Assessment Program (FHAP) of the Florida Fish and Wildlife Conservation Commission (FWC), which consisted of percent cover of Thalassia and Halodule63(see Supplemental Methods for detailed descriptions of both approaches). Standard errors of the total SAV cover maps ranged between 8.94 and 12.4% cover, standard deviation of total SAV cover maps ranged between 3.92 and 5.79% cover, number of species maps ranged between 0.26 and 0.46 species, Thalassia cover maps ranged between 4.12 and 5.4% cover, and Halodule cover maps ranged between 3.29 and 6.43% cover (Table S1). Because our study was conducted during SAV recovery, ranges of seagrass cover are at the low end (max Thalassia cover = 33%, max Halodule cover = 20%; Fig. S1). Thus, our study measures selection within limited ranges of SAV cover. Future studies should investigate higher cover ranges and selection in areas with denser SAV.
Seascape scale variables were quantified by mapping SAV using aerial imagery collected by ENP in 2021 and calculating spatial pattern metrics on the resulting SAV map. A binary classification scheme was used to represent the total cover of SAV consisting of dense SAV (> 25% cover) and sparse SAV/bare sediment (0–25% cover) due to a spectrally significant threshold at 25% cover76,77,78,79,80,81. The binary class SAV map was created using a RF classifier trained with data obtained through photointerpretation and had an overall accuracy of 97.2% (Figs. 2, S2, S3). Eight spatial pattern metrics (Percentage of Landscape, Edge Density, Patch Density, Division, Mean Radius of Gyration, Area-eighted Mean Perimeter-to-area Ratio, Mean Shape, and Mean Patch Size) were then calculated on the dense SAV class using the landscapemetrics package in R82. However, only four metrics were retained for the final RSF model due to correlation between variables as well as the accuracy of the RSF model. These metrics consisted of: Percentage of Landscape (PLAND) which quantifies the amount of dense SAV within an area, Edge Density (ED) which measures the amount of edge within an area, Patch Density (PD) which quantifies the amount of patches within an area, and Division (DIV) which measures the probability that an adjacent pixel is part of the same patch (Table 1). These four spatial pattern metrics were chosen because they represent a range of seascape measurements and have been used in previous papers measuring seascape complexity (see15,81). General Additive Models (GAMs) were then run using the mgcv package in R83 to determine relationships between habitat variables.
Workflow of the field and analytical methods used in this study. Seatrout were acoustically tagged and released in Rankin Basin. SAV surveys were conducted at each receiver each year, consisting of 20 quadrat surveys. SAV survey data as well as data from FHAP and aerial imagery were used to map habitat variables. COAs were calculated from acoustic detections in 60-min time bins then grouped by individual, daily period, and season to randomly distribute pseudo-absence points in a 1:1 ratio with the COAs. Habitat data were extracted from the habitat maps at each COA and pseudo-absence point then used to train a random forest (RF) model using a 70% training—30% testing data split. Predictor importance was determined using the decrease in accuracy method, while marginal effects were investigated with partial dependency plots.
Acoustic telemetry
We used acoustic telemetry to track the habitat selection of seatrout in Rankin Basin using a gridded array of 29 omnidirectional receivers (models VR2W and VR2Tx; Innovasea) from 2020 to 2022 (Fig. 1). A grid design was chosen because it facilitates the relatively fine-scale study of space use, home range estimation of fish, and habitat selection within the array84,85. Extensive range testing determined that the detection range of acoustic receivers deployed at a depth of 1 m within this system was 75 m (see supplemental methods for details on deployment and range testing). Seatrout (n = 151) were caught throughout the array deployment area by hook-and-line with artificial lures. Upon capture, fish were measured and those > 300 mm total length were tagged with Innovasea V9-2L tags (2 settings: low power with random delay of 60-90 s, n = 127; high power with a random delay of 20-40 s, n = 24). Fish deemed large enough to accept a tag were weighed and tagged using a protocol approved by the FIU Institutional Animal Care and Use Committee (IACUC-18–062) in accordance with guidelines set forth by the American Veterinary Medical Association to reduce mortality and pain (Fig. 2).
Raw acoustic telemetry data consisting of a time stamp, receiver ID, and tag ID were loaded into R and filtered to only include individuals tagged in this study. Detection data were then filtered to remove false detections (one detection of a single individual across the array in a 2-h period86) and deceased individuals (individuals that were recorded constantly by one receiver for 24 h, n = 1). Short-term centers of activity (COAs) were then calculated to estimate spatial locations of fish detected away from exact receiver locations87. This method provides an average position of a fish based on the weighted means of the number of detections on each receiver over a given amount of time87. Using the VTRACK package in R88, COAs were calculated using a 60-min time bin (the minimum time interval used by48) and individuals with less than 50 COAs over their detection history were removed. Pseudo-absence points were created by grouping the COAs by individual, diel period (night, dawn, day, dusk; Table S2) and season (dry, early wet, wet, early dry; Table S2), summing the number of COAs in each group, and then randomly placing pseudo-absence points throughout the acoustic array. Pseudo-absence points were added until the ratio of COAs to pseudo-absence points was 1:1, which is the suggested ratio for machine learning models89 (Fig. 2).
Habitat selection: resource selection functions
Relative habitat selection of seatrout was predicted using RSFs. RSFs determine habitat selection by using spatiotemporal records of animal occurrence to evaluate relationships between space use and habitat characteristics relative to the available habitat90,91. This method operates under a use/availability framework, where space use is associated with a positive presence (such as a COA), which is then compared to a random sampling of background points (pseudo-absences) across the available environmental conditions48. RSFs are viable to study habitat selection over the correlation of individual presence and habitat variables because all the available habitat is considered in the modeling process, resulting in the investigation of selection. However, this framework does not allow for the calculation of habitat preference, as only experiments can investigate preference per se92,93. All analyses were conducted in R version 4.2.294.
Habitat variable data for all COAs and pseudo-absence points were extracted from the habitat maps. For patch scale variables, COAs and pseudo-absence points were grouped by year, and habitat variable values were extracted at each point from the interpolated habitat rasters based on year. Seascape scale variables were quantified within a 75 m radius of every COA and pseudo-absence point (Fig. 2). RF models95 were then used to apply RSFs to investigate relative habitat selection of seatrout using the ranger package96 and executed in the mlr package in R97. The RF model was trained using 500 trees (subset with replacement) with 70% of the data. The remaining 30% of the data were used for model validation. To increase the accuracy of RF models, hyperparameter optimization was performed to determine the ideal number of predictors within each tree (mtry), the fraction of observations that should be used to train each tree (sample.fraction), and the number of observations the terminal node of each tree should have (min.node.size48). The hyperparameter tuning was performed using the tuneParams() function in the mlr package (Fig. 2).
Accuracy of the RF model was determined by using the trained model to predict across the 30% validation dataset, resulting in an overall accuracy of the RF model of 98% (Fig. S4). To identify the most important habitat variables driving seatrout habitat selection, predictor importance was calculated using the permutation importance method in the iml package in R98. This method assesses the increase in the prediction error of the model when each variable is removed95. Partial dependency plots were then constructed for model predictors to determine the marginal effect of the covariates on the predicted outcome (presence/absence of seatrout) using the pdp package in R99. These plots illustrate how each variable impacts the probability of presence across the range of its values. The RF model was then used to investigate the interaction between predictors (Fig. 2).
Results
Acoustic telemetry data summary
Out of 151 tagged seatrout, 58 individuals had at least 50 60-min COAs calculated and thus were retained for analysis. The longest duration of tracking of an individual fish was 534 days, while the shortest tracked duration was 4 days (Table S3). A total of 338,294 acoustic detections of seatrout were used to create 21,971 COAs after pre-processing the acoustic data and filtering for the minimum number of COAs (Fig. S5). Of these, 4,264 COAs were calculated for 2020, 5,722 for 2021, and 11,985 for 2022. An average of 406 COAs were calculated per individual seatrout (range: 51–3,438).
Predictor importance
Habitat selection was influenced by both patch and surrounding seascape-scale characteristics based on our model results, supporting our hypothesis that both scales of habitat would influence seatrout habitat selection (hypothesis 1). The most important predictor of seatrout habitat selection within Rankin Basin was the total cover of Halodule (a patch-scale variable; Fig. 3). The second most important predictor was patch density (a seascape-scale variable). Two other patch-scale variables were among the top four predictors: standard deviation of total SAV cover and number of SAV species. The least important variable was year (Fig. 3).
Relative predictor importance within the random forest model. Values were calculated using the mean decrease in accuracy method. Predictors are color coded by scale (green = patch, blue = seascape), and shading indicates importance.
Marginal effects of the top 4 predictors
Partial dependency plots were used to investigate the relative selection strength of the top four predictors (when other predictors were held constant). Here a higher marginal effect (ŷ) indicated a higher relative selection (Fig. 4). The general selection strength patterns differed between patch and seascape-scale variables. Patch-scale selection displayed an optimal pattern whereas seascape-scale selection exhibited a logistic pattern in which the probability of selection eventually plateaued. At the patch scale, the probability of seatrout selection increased with increasing Halodule cover until it reached a maximum at 5.5% total cover. Selection then decreased with higher levels of Halodule cover (up to 20%, Fig. 4a). For patch density, ŷ values increased with increasing patch density up to 2,750 patches per hectare, where probability of seatrout selection was high and remained relatively stable up to patch density values > 9,000 patches per hectare (Fig. 4b). Relative selection for standard deviation of total SAV cover exhibited a bimodal relationship, with higher relative selection at both the lowest and the highest values (< 10 and > 25 SD units; Fig. 4c). The probability of seatrout selection was trimodal when considering the number of SAV species, with higher values at 1.5 and 2.75, and with values above 3.5 having the highest probability of presence, whereas values of 2.2 resulted in the lowest probability of presence (Fig. 4d). These results support our hypothesis that seatrout select for areas of high spatial heterogeneity (hypothesis 2). Other partial dependency plots are presented in Fig. S6.
Partial dependency plots of the top 4 predictors. The marginal effect represents the relative selection strength for each given predictor when all other predictors are held constant. Higher ŷ values indicate a higher relative selection. Color coded by level (green = patch, blue = seascape).
Interaction between predictors
Interactions among the top three predictors (Halodule cover, patch density, and standard deviation of total cover) were investigated by utilizing the RF model to predict across the range of values of the patch-scale variables (0–25 for Halodule cover and 0–30 for standard deviation of total cover), while varying the seascape-scale variable (patch density; Fig. 5). Relative habitat selection for seatrout was highest for lower values of Halodule cover and a lower standard deviation of total cover in patchy SAV habitats. In contrast, when the seascape was a continuous bed (patch density = 1), seatrout selected for higher levels of Halodule cover and higher standard deviation of total cover (Fig. 5).
Stacked plot visualizing the interaction between the top three predictors in the random forest model. Patch level variables Halodule cover and standard deviation of cover are represented by the x- and y-axes. Seascape variable patch density is represented by the z-axis. Darker values represent higher relative habitat selection.
Discussion
Habitat selection for highly mobile marine species is a multi-scale process that requires the consideration of both seascape (broad-scale) and patch (fine-scale) habitat variables10,100. Combining remote sensing, acoustic telemetry, and machine learning, our study investigated what drives seatrout habitat selection at the patch and seascape scale in an area recovering from a large-scale disturbance (i.e., a 2015 seagrass die-off). Based on 21,971 COAs calculated from 338,294 acoustic detections over a 3-year period, we found that seatrout third-order habitat selection (e.g., patch selection for foraging) in Florida Bay is influenced by habitat characteristics varying at multiple spatial scales. Of the top four predictors of seatrout habitat selection in SAV meadows, three were patch (Halodule cover, standard deviation of SAV cover, and SAV species richness) and one was seascape scale (patch density). At the patch scale, seatrout exhibited an optimal response, illustrating a specific range of habitat values influencing habitat selection The most important predictor of seatrout habitat selection at the patch scale was the total cover of Halodule within a SAV patch, with seatrout being most likely to select areas containing 5.5% cover. Values above and below that value resulted in a lower selection probability, with a minimum selection at the highest level of Halodule cover (20%). At the seascape scale, seatrout exhibited a logistic response to patch density, where seatrout selected SAV areas with a minimum level of complexity (i.e., above 3,000 patches per hectare). Furthermore, seatrout were most likely to select patches of consistently low Halodule cover with the only exception being when the habitat was not fragmented, and then selection was for continuous meadows of high and variable Halodule cover.
Scale is a crucial aspect of ecology, as individuals experience the environment across a range of scales based on their perception of the landscape4,9. Our study found that seatrout make decisions on habitat selection utilizing information from two scales, which has been observed across different levels of terrestrial mobile organisms. For example, Rather et al.101 found that tigers in India avoided areas dominated by Sal trees at the broad scale and utilized areas where the dominant slope was to the south at the fine scale. In a nature reserve in China, Ma et al.102 found that cranes selected habitat variables at different scales depending on the time of day. In seascape ecology, studies investigating multi-scale drivers of faunal abundance and biomass have highlighted both seascape and patch scale dynamics as critical factors influencing habitat selection. For example, Pittman et al.20 found a linear relationship between faunal density and seagrass cover at the seascape scale but a threshold relationship at 20% seagrass cover at the patch scale. However, multi-scale habitat selection studies on mobile marine fauna using movement are lacking13.
Habitat selection at the seascape scale
All seascape scale predictors exhibited a logistic response, where the relationship either starts out with a positive or negative trend but levels out at a certain value (see Figs. 5b, S3). Of these relationships, the most important to seatrout habitat selection was seascape patchiness. Seatrout selected for patchier seascapes, with their selection probability increasing linearly as continuous habitats fragmented into patches, peaking at a maximum patch density of 3000 patches per hectare. Beyond this value, seatrout selection probability remained high and stable. Logistic relationships in animal-seascape relationships are relatively common, having been found for habitat connectivity103,104, distance from a key habitat (nursery41, shelf edge105, reef106, land48), and amount of habitat42. For instance, Griffin et al.48 found that lemon shark habitat selection in St. Croix had a negative logistic relationship with distance from shore while Yeager et al.42 found that fish diversity had a positive logistic relationship with the amount of dense seagrass in the seascape.
The greater habitat selection with higher patch density may be due to the benefits of edge habitat in seascapes, which is also supported by the higher relative habitat selection at higher standard deviations in total cover. In seagrass ecosystems, edge habitat has been found to increase secondary production, leading to a higher abundance of prey items for seatrout37,107. Total fish density and foraging efficiency were also found to increase along patch edges43. Patch edges may also benefit seatrout due to their foraging strategy. Seatrout are ambush predators and therefore can utilize patch edges to sit and wait for prey items to exit the safety of SAV cover49. Seatrout in Texas, USA, utilized seagrass habitat during the day and bare sediment at night, which is hypothesized as a mechanism to reduce predation risk from piscivores such as Bottlenose Dolphins46 (Tursiops truncates). Due to the presence of dolphins in Florida Bay, seatrout may be utilizing areas with a certain level of patchiness to take advantage of the matrix of bare sediment for nocturnal foraging. While temporal analysis was not performed in this study, future investigation of these data will include temporal drivers of habitat selection and space use to better tease out these effects.
Habitat selection at the patch scale
While logistic responses were observed at the seascape scale, they were rare for the patch-scale variables. All but one (cover of Thalassia) patch- scale variables considered in our analysis showed an optimal response. Optimal responses result from a species selecting for one or more ‘optimum’ values across a continuous environmental gradient108,109. These optima vary across species, and can be influenced by various factors such as metabolism (e.g., temperature110), predation risk (e.g., water column turbidity in planktivorous fishes111, and foraging efficiency (e.g., short vs. long foraging trips by nesting seabirds112). In seagrass systems, optimal relationships between within-patch variables and fauna have been previously reported. For example, Belgrad et al.113 found that peak pinfish abundance was associated with high Thalassia cover and low drift biomass. In our study, seatrout selected for a single optimum Halodule cover, but for multiple optima for standard deviation of cover and number of species.
Cover of Halodule was the single most important predictor of seatrout habitat selection of in Rankin Basin. Seatrout exhibited a maximum relative habitat selection in areas where Halodule covered approximately 5% of the benthos. In seatrout, previous work has shown a preference for seagrass habitat relative to oyster and saltmarsh habitats46 and for seagrass when spawning70 (vs. open water habitat), but no previous work has determined preference of seagrass species nor density. Halodule has been found to be an important habitat for primary and secondary consumers as well as preferred habitat of pink shrimp (Farfantepenaeus duorarum), a common diet item for seatrout32,114,115. Thus, the presence of any Halodule may increase prey availability within the SAV patch. Furthermore, the sparse cover of Halodule selected for by seatrout is also indicative of multi-species SAV beds. We found that the maximum number of SAV species occurred throughout our study system when Halodule cover was 5% (Fig. S7). Mixed seagrass beds may increase faunal density and diversity due to species-specific relationships with certain SAV species (e.g., Pinfish prefer Thalassia over Halodule116,117, Blue crabs prefer Halodule over Thalassia and Syringodium filiforme32). Therefore, higher SAV diversity may increase faunal diversity by facilitating many species to inhabit the same seagrass patch.
Interaction of scales
Seatrout in this study utilized decision-making at multiple scales to select for habitat variables, leading to interactions across scales that support foraging theory. When investigating how the interaction of the top three predictors (Halodule cover, standard deviation of cover, and patch density) impacted habitat selection, we found two maximal habitat preferences depending on the seascape structure. When patch density was equal to one (representing one continuous dense SAV patch), seatrout selected for areas categorized by high standard deviation of cover and a range of Halodule cover. However, seatrout selected for areas categorized by low Halodule cover and standard deviation of cover once patch density was increased. This pattern illustrates a conservation of foraging strategy across seascape complexity, wherein seatrout select for habitat edges to increase foraging efficiency43. Overall, seatrout preferred higher patch densities because edges are apparent throughout the seascape. However, when patches, and therefore edges, are not present (one continuous meadow), the higher standard deviation of cover might create “pseudo-edges” (canopy openness) where SAV densities vary across the seascape while staying dense (> 25% cover). Therefore, seatrout might use these “pseudo-edges” as a replacement to patch edges to ambush prey. Future work should compare the energy costs and fitness benefits across patchy and continuous habitats to determine the difference between using real edges and “pseudo-edges” for foraging.
Conclusion
Our study found that a seagrass-associated mesoconsumer exhibited different habitat selection trends across hierarchical scales of spatial organization. Seatrout utilized the seascape scale to find a minimum number of patches (logistic response) and then selected for specific ranges of patch scale characteristics such as Halodule cover (optimal response). This is the first study to look at multi-scale habitat selection using movement in a seascape and emphasizes that the response of selection is different across scales. We encourage future studies to examine the shapes of these relationships to provide more insight into habitat preference of individual species. This is especially crucial for conserving species into the future, as climate change will not only cause species range shifts but shifts in local habitats as well. Therefore, to fully understand the impacts of climate change on organisms, we need to consider how habitat selection is occurring at multiple scales.
Data availability
Data and code will be made available upon request to the corresponding author (JR, jrodeman@fiu.edu).
References
Mayor, S. J., Schneider, D. C., Schaefer, J. A. & Mahoney, S. P. Habitat selection at multiple scales. Ecoscience 16, 238–247 (2009).
Johnson, D. H. The comparison of usage and availability measurements for evaluating resource preference. Ecology 61, 65–71 (1980).
Van Moorter, B., Rolandsen, C. M., Basille, M. & Gaillard, J. M. Movement is the glue connecting home ranges and habitat selection. J. Anim. Eco. 85, 21–31 (2016).
Wiens, J. A. Population responses to patchy environments. Annu. Rev. Ecol. Syst. 7, 81–120 (1976).
Pittman, S. J. & McAlpine, C. A. Movements of marine fish and decapod crustaceans: Process, theory and application. In Advances in Marine Biology 205–294 (Elsevier, 2003).
Kacelnik, A. & Houston, A. I. Some effects of energy costs on foraging strategies. Anim. Behav. 32, 609–614 (1984).
Chetkiewicz, C.-L.B., St. Clair, C. C. & Boyce, M. S. Corridors for conservation: Integrating pattern and process. Annu. Rev. Ecol. Evol. Syst. 37, 317–342 (2006).
Smith, T. M. et al. Edge effects in patchy seagrass landscapes: The role of predation in determining fish distribution. J. Exp. Mar. Biol. Ecol. 399, 8–16 (2011).
Levin, S. A. The problem of pattern and scale in ecology: The Robert H. MacArthur award lecture. Ecology 73, 1943–1967 (1992).
McGarigal, K. et al. Multi-scale habitat selection modeling: A review and outlook. Land. Ecol. 31, 1161–1175 (2016).
Leblond, M. et al. Assessing the influence of resource covariates at multiple spatial scales: An application to forest-dwelling caribou faced with intensive human activity. Land. Ecol. 26, 1433–1446 (2011).
Pittman, S. J., Kneib, R. T. & Simenstad, C. A. Practicing coastal seascape ecology. Mar. Ecol. Prog. Ser. 427, 187–190 (2011).
Pittman, S. J. et al. Seascape ecology: Identifying research priorities for an emerging ocean sustainability science. Mar. Ecol. Prog. Ser. 663, 1–29 (2021).
Jackson, E. L., Santos, R. O. & Pittman, S. J. Seascape patch dynamics. In Seascape Ecology 153–188 (Wiley, 2017).
Santos, R. O., Lirman, D., Pittman, S. J. & Serafy, J. E. Spatial patterns of seagrasses and salinity regimes interact to structure marine faunal assemblages in a subtropical bay. Mar. Ecol. Prog. Ser. 594, 21–38 (2017).
Pittman, S. J. Seascape Ecology (Wiley, 2017).
Costa, B., Walker, B. K. & Dijkstra, J. A. Mapping and quantifying seascape patterns. In Seascape Ecology 27–56 (Wiley, 2017).
Santos, R. O., Lirman, D. & Serafy, J. E. Quantifying freshwater-induced fragmentation of submerged aquatic vegetation communities using a multi-scale landscape ecology approach. Mar. Ecol. Prog. Ser. 427, 233–246 (2011).
Wiens, J. A., Chr, N., Van Horne, B. & Ims, R. A. Ecological mechanisms and landscape ecology. Oikos 66, 369–380 (1993).
Pittman, S. J., McAlpine, C. A. & Pittman, K. M. Linking fish and prawns to their environment: A hierarchical landscape approach. Mar. Ecol. Prog. Ser. 283, 233–254 (2004).
Tonetti, V. et al. Landscape heterogeneity: Concepts, quantification, challenges and future perspectives. Environ. Conserv. 50(2), 83–92 (2023).
Matthiopoulos, J. et al. Establishing the link between habitat selection and animal population dynamics. Ecol. Monogr. 85(3), 413–436 (2015).
Abrahms, B. et al. Emerging perspectives on resource tracking and animal movement ecology. Trends Ecol. Evol. 36(4), 308–320 (2021).
Bell, J. & Westoby, M. Variation in seagrass height and density over a wide spatial scale: Effects on common fish and decapods. J. Exp. Mar. Biol. Ecol. 104, 275–295 (1986).
McCloskey, R. M. & Unsworth, R. K. F. Decreasing seagrass density negatively influences associated fauna. PeerJ https://doi.org/10.7717/peerj.1053 (2015).
Pullen, M., Gerber, D., Thomsen, M. S. & Flanagan, S. P. Seasonal dynamics of faunal diversity and population ecology in an estuarine seagrass bed. ESCO 45, 2578–2591 (2022).
Connolly, R. M. & Butler, A. J. The effects of altering seagrass canopy height on small, motile invertebrates of shallow Mediterranean embayments. Mar. Ecol. 17, 637–652 (1996).
Gullström, M., Bodin, M., Nilsson, P. G. & Öhman, M. C. Seagrass structural complexity and landscape configuration as determinants of tropical fish assemblage composition. Mar. Ecol. Prog. Ser. 363, 241–255 (2008).
Rooker, J. R., Holt, S. A., Soto, M. A. & Joan Holt, G. Postsettlement patterns of habitat use by sciaenid fishes in subtropical seagrass meadows. Estuaries. 21(2), 318–327 (1998).
Hyndes, G. A., Kendrick, A. J., MacArthur, L. D. & Stewart, E. Differences in the species- and size-composition of fish assemblages in three distinct seagrass habitats with differing plant and meadow structure. Mar. Biol. 142, 1195–1206 (2003).
Flaherty-Walia, K. E., Matheson, R. E. Jr. & Paperno, R. Juvenile spotted seatrout (Cynoscion nebulosus) habitat use in an eastern Gulf of Mexico estuary: The effects of seagrass bed architecture, seagrass species composition, and varying degrees of freshwater influence. ESCO. 38(1), 353–366 (2015).
Ray, B. R., Johnson, M. W., Cammarata, K. & Smee, D. L. Changes in seagrass species composition in Northwestern Gulf of Mexico Estuaries: Effects on associated seagrass fauna. PLoS ONE 9, e107751 (2014).
Bell, S. S. et al. Faunal response to fragmentation in seagrass habitats: Implications for seagrass conservation. Biol. Conserv. 100, 115–123 (2001).
Hovel, K. A. & Lipcius, R. N. Habitat fragmentation in a seagrass landscape: Patch size and complexity control blue crab survival. Ecology 82, 1814–1829 (2001).
Jelbart, J. E., Ross, P. M. & Connolly, R. M. Patterns of small fish distributions in seagrass beds in a temperate Australian estuary. J. Mar. Biol. Assoc. U. K. 87, 1297–1307 (2007).
Smith, T. M., Hindell, J. S., Jenkins, G. P. & Connolly, R. M. Edge effects on fish associated with seagrass and sand patches. Mar. Ecol. Prog. Ser. 359, 203–213 (2008).
Macreadie, P. I. et al. Resource distribution influences positive edge effects in in a seagrass fish. Ecology 91, 2013–2021 (2010).
Moore, E. C. & Hovel, K. A. Relative influence of habitat complexity and proximity to patch edges on seagrass epifaunal communities. Oikos 119, 1299–1311 (2010).
Dorenbosch, M., van Riel, M. C., Nagelkerken, I. & van der Velde, G. The relationship of reef fish densities to the proximity of mangrove and seagrass nurseries. Estuar. Coast. Shelf Sci. 60, 37–48 (2004).
Davis, J. P. et al. Seagrass corridors and tidal state modify how fish use habitats on intertidal coral reef flats. Mar. Ecol. Prog. Ser. 581, 135–147 (2017).
Berkström, C. et al. Thresholds in seascape connectivity: The spatial arrangement of nursery habitats structure fish communities on nearby reefs. Ecography 43, 882–896 (2020).
Yeager, L. A. et al. Threshold effects of habitat fragmentation on fish diversity at landscapes scales. Ecology 97, 2157–2166 (2016).
Yarnall, A. H., Byers, J. E., Yeager, L. A. & Fodrie, F. J. Comparing edge and fragmentation effects within seagrass communities: A meta-analysis. Ecology 103, e3603 (2022).
Powell, A. B. et al. Growth, mortality, and hatchdate distributions of larval and juvenile spotted seatrout (Cynoscion nebulosus) in Florida Bay, Everglades National Park. Fish. Bull. 102, 142–155 (2004).
TinHan, T. C. et al. Linking habitat use and trophic ecology of spotted seatrout (Cynoscion nebulosus) on a restored oyster reef in a subtropical estuary. ESCO. 41, 1793–1805 (2018).
Moulton, D. L. et al. Habitat partitioning and seasonal movement of red drum and spotted seatrout. ESCO. 40, 905–916 (2017).
Reyier, E. A. et al. Residency and dispersal of three sportfish species from a coastal marine reserve: Insights from a regional-scale acoustic telemetry network. Glob. Ecol. Conserv. 23, e01057 (2020).
Griffin, L. P. et al. A novel framework to predict relative habitat selection in aquatic systems: Applying machine learning and resource selection functions to acoustic telemetry data from multiple shark species. Front. Mar. Sci. 8, 631262 (2021).
Bortone, S. A. Biology of the Spotted Seatrout (CRC Press, 2002).
Callihan, J. L. Spatial ecology of adult spotted seatrout, Cynoscion nebulosus, in Louisiana coastal waters (2011).
Payne, L. M. Evaluation of large-scale movement patterns of spotted seatrout (Cynoscion nebulosus) using acoustic telemetry (2011).
Stainback, G. A., Fedler, T., Davis, S. E. III. & Birendra, K. C. Recreational fishing in Florida Bay: Economic significance and angler perspectives. Tour. Mar. Environ. 14, 89–105 (2019).
Fourqurean, J. W. & Robblee, M. B. Florida Bay: A history of recent ecological changes. Estuaries 22, 345 (1999).
Nuttle, W. K. et al. Influence of net freshwater supply on salinity in Florida Bay. Water Resour. Res. 36, 1805–1822 (2000).
Rudnick, D. T., Ortner, P. B., Browder, J. A. & Davis, S. M. A conceptual ecological model of Florida Bay. Wetlands 25, 870–883 (2005).
Kelble, C. R. et al. Salinity patterns of Florida Bay. Estuar. Coast. Shelf Sci. 71, 318–334 (2007).
Madden, C. J. et al. Ecological indicators for assessing and communicating seagrass status and trends in Florida Bay. Ecol. Ind. 9, S68–S82 (2009).
Briceño, H. O. & Boyer, J. N. Climatic controls on phytoplankton biomass in a sub-tropical estuary, Florida Bay, USA. ESCO. 33, 541–553 (2010).
Robblee, M. et al. Mass mortality of the tropical seagrass Thalassia testudinum in Florida Bay (USA). Mar. Ecol. Prog. Ser. 71, 297–299 (1991).
Hall, M., Furman, B., Merello, M. & Durako, M. Recurrence of Thalassia testudinum seagrass die-off in Florida Bay, USA: Initial observations. Mar. Ecol. Prog. Ser. 560, 243–249 (2016).
Hall, M. O., Bell, S. S., Furman, B. T. & Durako, M. J. Natural recovery of a marine foundation species emerges decades after landscape-scale mortality. Sci. Rep. 11, 6973 (2021).
Zieman, J. C. The ecology of the seagrasses of South Florida: A community profile. Department of the Interior, U.S. Fish and Wildlife Service (1982).
Furman, B. T. & Jarvis, J. C. South Florida fisheries habitat assessment program annual report to South Florida Water Management District. South Florida Water Management District (2022).
National Marine Fisheries Service. Fisheries of the United States, 2020. U.S. Department of Commerce, NOAA Current Fishery Statistics No. 2020 (2022).
Osborne, J., Schmidt, T. W. & Kalafarski, J. Year 2005 Annual Marine Fisheries Report. Everglades National Park (2006).
Santos, R. O., Rehage, J. S., Boucek, R. & Osborne, J. Shift in recreational fishing catches as a function of an extreme cold event. Ecosphere. 7, e01335 (2016).
Muller, R. & Addis, D. An Update Assessment of the Status of Spotted Seatrout in Florida Waters Through 2017 (Fish and Wildlife Research Institute, St. Petersburg, 2019).
Weinstein, M. P. & Yerger, R. W. Electrophoretic investigation of subpopulations of the spotted seatrout, Cynoscion nebulosus, in the Gulf of Mexico and Atlantic coast of Florida. Comp. Biochem. Physiol. 54B, 97–102 (1976).
Mazzotti, F. J. et al. Stressor-response model for the spotted seatrout (Cynoscion nebulosus). EDIS. 2008, 3 (2008).
Boucek, R. E. et al. More than just a spawning location: Examining fine scale space use of two estuarine fish species at a spawning aggregation site. Front. Mar. Sci. 4, 355 (2017).
Rutherford, E. S., Schmidt, T. W. & Tilmant, J. T. Early life history of spotted seatrout (Cynoscion nebulosus) and gray snapper (Lutjanus griseus) in Florida Bay, Everglades National Park, Florida. Bull. Mar. Sci. 44(1), 49–64 (1989).
Hettler, W. F. Jr. Food habits of juveniles of spotted seatrout and gray snapper in Western Florida Bay. Bull. Mar. Sci. 44, 155–162 (1989).
Callihan, J. L., Cowan, J. H. & Harbison, M. D. Sex differences in residency of adult spotted seatrout in a Louisiana Estuary. Mar. Coast. Fish. 5, 79–92 (2013).
Zarada, K. et al. Estimating site-specific spawning parameters for a spawning aggregation: An example with spotted seatrout. Mar. Ecol. Prog. Ser. 624, 117–129 (2019).
Weihe, G., Chamberlain, R. H., Sabol, B. M. & Doering, P. H. Mapping submerged aquatic vegetation with GIS in the Caloosahatchee Estuary: Evaluation of different interpolation methods. Mar. Geod. 22, 69–91 (1999).
Meyer, C. A. & Pu, R. Seagrass resource assessment using remote sensing methods in St. Joseph Sound and Clearwater Harbor, Florida, USA. Environ. Monit. Assess. 184, 1131–1143 (2012).
Pu, R., Bell, S. & Meyer, C. Mapping and assessing seagrass bed changes in Central Florida’s west coast using multitemporal Landsat TM imagery. Estuar. Coast. Shelf Sci. 149, 68–79 (2014).
Blakey, T., Melesse, A. & Hall, M. O. Supervised classification of benthic reflectance in shallow subtropical waters using a generalized pixel-based classifier across a time series. Remote Sens. 7, 5098–5116 (2015).
Santos, R. O. et al. Implications of macroalgae blooms to the spatial structure of seagrass seascapes: The case of the Anadyomene spp. (Chlorophyta) bloom in Biscayne Bay, Florida. Mar. Poll. Bull. 150, 110742 (2020).
Kovacs, E. M. et al. Cloud processing for simultaneous mapping of seagrass meadows in optically complex and varied water. Remote Sens. 14, 609 (2022).
Rodemann, J. R. et al. Response of submerged aquatic vegetation seascapes to a large-scale seagrass die-off: A case study in Florida Bay. Estuar. Coast. Shelf Sci. 318, 109221 (2025).
Hesselbarth, M. H. K. et al. landscapemetrics: An open-source R tool to calculate landscape metrics. Ecography 42, 1648–1657 (2019).
Wood, S. & Wood, M. S. Package ‘mgcv’. R Package Version 1(29), 729 (2015).
Heupel, M. R., Semmens, J. M. & Hobday, A. J. Automated acoustic tracking of aquatic animals: Scales, design and deployment of listening station arrays. Mar. Fresh. Res. 57, 1–13 (2006).
Brownscombe, J. W. et al. Conducting and interpreting fish telemetry studies: Considerations for researchers and resource managers. Rev. Fish Biol. Fish. 29, 369–400 (2019).
Simpfendorfer, C. A. et al. Ghosts in the data: False detections in VEMCO pulse position modulation acoustic telemetry monitoring equipment. Anim. Biotel. 3, 1–10 (2015).
Simpfendorfer, C. A., Heupel, M. R. & Hueter, R. E. Estimation of short-term centers of activity from an array of omnidirectional hydrophones and its use in studying animal movements. Can. J. Fish. Aquat. Sci. 59, 23–32 (2002).
Campbell, H. A. et al. V-Track: Software for analysing and visualising animal movement from acoustic telemetry detections. Mar. Freshwater Res. 63, 815–820 (2012).
Barbet-Massin, M., Jiguet, F., Albert, C. H. & Thuiller, W. Selecting pseudo-absences for species distribution models: How, where and how many?. Methods Ecol. Evol. 3, 327–338 (2012).
Boyce, M. S., Vernier, P. R., Nielsen, S. E. & Schmiegelow, F. K. A. Evaluating resource selection functions. Ecol. Model. 157, 281–300 (2002).
Manly, B. F. et al. Resource Selection by Animals: Statistical Design and Analysis for Field Studies (Springer, 2007).
Underwood, A. J., Chapman, M. G. & Crowe, T. P. Identifying and understanding ecological preferences for habitat or prey. J. Exp. Mar. Bio. Ecol. 300(1–2), 161–187 (2004).
Underwood, A. J. & Clarke, K. R. Solving some statistical problems in analyses of experiments on choices of food and on associations with habitat. J. Exp. Mar. Bio. Ecol. 318(2), 227–237 (2005).
R Core Team. R: A Language and Environment for Statistical Computing. Vienna, Austria: R Foundation for Statistical Computing Available at: https://www.R-project.org/ (2023).
Breiman, L. Random forests. Mach. Learn. 45, 5–32 (2001).
Wright, M. N., Wager, S., & Probst, P. Package ‘ranger’. Version-11, 2. (2019).
Bischl, B. et al. mlr: Machine learning in R. J. Mach. Learn. Res. 17(170), 1–5 (2016).
Molnar, C., Casalicchio, G. & Bischl, B. Iml: An R package for interpretable machine learning. J. Open Source Softw. 3(26), 786 (2018).
Greenwell, B. M. pdp: An R package for constructing partial dependence plots. R J. 9, 421–436 (2017).
Brownscombe, J. W. et al. Applications of telemetry to fish habitat science and management. Can. J. Fish. Aquat. Sci. 79, 1347–1359 (2022).
Rather, T. A., Kumar, S. & Khan, J. A. Multi-scale habitat modelling and predicting change in the distribution of tiger and leopard using random forest algorithm. Sci. Rep. 10(1), 11473 (2020).
Ma, T. et al. Multi-scale habitat selection modeling using combinatorial optimization of environmental covariates: A case study on nature reserve of red-crowned cranes. Ecol. Ind. 154, 110488 (2023).
Berkström, C. et al. Exploring ‘knowns’ and ‘unknowns’ in tropical seascape connectivity with insights from East African coral reefs. Estuar. Coast. Shelf Sci. 107, 1–21 (2012).
Caldwell, I. R. & Gergel, S. E. Thresholds in seascape connectivity: Influence of mobility, habitat distribution, and current strength on fish movement. Land. Ecol. 28, 1937–1948 (2013).
Pittman, S. J. & Brown, K. A. Multi-scale approach for predicting fish species distributions across coral reef seascapes. PLoS ONE 6, e20583 (2011).
Eggertsen, L. et al. Where the grass is greenest in seagrass seascapes depends on life history and simple species traits of fish. Estuar. Coast. Shelf Sci. 266, 107738 (2022).
Bologna, P. A. X. & Heck, K. L. Impact of habitat edges on density and secondary production of seagrass-associated fauna. Estuaries 25, 1033–1044 (2002).
Ter Braak, C. J. Unimodal Models to Relate Species to Environment (Wageningen University and Research, 1987).
Anderson, M. J., Walsh, D. C., Sweatman, W. L. & Punnett, A. J. Non-linear models of species’ responses to environmental and spatial gradients. Ecol. Lett. 25(12), 2739–2752 (2022).
Uiterwaal, S. F. & DeLong, J. P. Functional responses are maximized at intermediate temperatures. Ecology 101(4), e02975 (2020).
Pangle, K. L. et al. Context-dependent planktivory: Interacting effects of turbidity and predation risk on adaptive foraging. Ecosphere. 3(12), 1–18 (2012).
Chaurand, T. & Weimerskirch, H. The regular alternation of short and long foraging trips in the blue petrel Halobaena caerulea: A previously undescribed strategy of food provisioning in a pelagic seabird. J. Anim. Ecol. 63, 275–282 (1994).
Belgrad, B. A. et al. Environmental drivers of seagrass-associated nekton abundance across the northern Gulf of Mexico. ESCO. 44(8), 2279–2290 (2021).
Zink, I. C., Browder, J. A., Lirman, D. & Serafy, J. E. Pink shrimp Farfantepenaeus duorarum spatiotemporal abundance trends along an urban, subtropical shoreline slated for restoration. PLoS ONE 13(11), e0198539 (2018).
James, W. R. et al. A review of the potential impacts of commercial inshore pink shrimp fisheries on the recreational flats fishery in Biscayne Bay, FL, USA. Environ. Biol. Fishes 106(2), 349–360 (2023).
Jordan, F. et al. Risk of predation affects habitat selection by the pinfish Lagodon rhomboides (Linnaeus). J. Exp. Mar. Bio Ecol. 208(1–2), 45–56 (1997).
Rudershausen, P. J., Locascio, J. V. & Rojas, L. M. A comparison of macroepifauna among vegetated and unvegetated habitats in a south Florida estuary using a passive sampling gear. Gulf Mex. Sci. 21(2), 2 (2003).
Acknowledgements
We would like to thank the editors and the reviewers for their time and effort for reviewing the manuscript. We would like to thank FWRI for contributing seagrass cover data and Everglades National Park for contributing aerial imagery of Florida Bay. This is contribution #2071 from the Institute of Environment at Florida International University. We would also like to thank CREST CAChE and the FCE LTER for their support.
Funding
This project was funded by Task Agreement P23AC00798 under Master Cooperative Agreement P18AC00974 between the National Park Service and Florida International University.
Author information
Authors and Affiliations
Contributions
JRR, MW, WRJ, ROS, SJP, DG, and JSR conceptualized project. JRR and BTF collected and entered field data. JRR mapped habitat variables. JRR, MW, WRJ, ROS, LPG, and SVC performed statistical analyses. JRR, MW, WRJ, ROS, JSR, and SVC conceptualized paper and figures. JRR, SVC, and JSR prepared first draft. MW performed major edits on first draft. All authors contributed to the manuscript preparation.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Rodemann, J., White, M., James, W.R. et al. Hierarchical habitat selection by a predatory fish in a patchy seascape. Sci Rep 15, 43443 (2025). https://doi.org/10.1038/s41598-025-27322-0
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-27322-0