Abstract
The motion of ferrofluid droplet on hydrophobic surface has attracted significant research interests due to its potential applications in biomedicine, sensor technologies, and oil–water separation processes. This study investigates ferrofluid droplet impact on a hydrophobic surface and analyzes how ferromagnetic particles in the droplet affect the interaction characteristics of droplet in terms of spreading, retraction, and the formation of secondary droplets during rebound cycles. The results reveal that introducing a magnetic field reduces the droplet spreading factor by approximately 20%. Additionally, increasing ferro particles concentrations to 0.05% (wt%) gives rise to a significant reduction in the spreading factor. The effect on the droplet rebound height is also noteworthy: for a lower particle concentration (0.005%, wt%), the restitution coefficient decreases by 15.5%. However, at high weight concentration (0.05%), this decrease is as much as 69%, which indicates a strong dependency on particle concentration. The analysis shows that the force generated under magnetic field, which is around 2.28 × 10−5 N, is substantially greater than the capillary force, which is ~ 2.69 × 10−6 N, leading to the attachment of droplets at magnet when the spacing from the magnet to droplet becomes small. During the rebound, the droplet experiences momentum dissipation, which results in the break-off of smaller “newborn” droplets. This phenomenon is primarily governed by the interfacial force (∼ 1.47 × 10−4 N) due to the particles and the surrounding liquid that is larger than the magnetic force itself. The newborn droplets tend to have a higher concentration of particles and are prone to sticking on the magnet.
Similar content being viewed by others
Introduction
Ferromagnetic particles are typically composed of ferromagnetic materials, characterized by a high magnetic susceptibility and the ability to retain a strong magnetic moment even after the elimination of a magnetic field. Commonly used ferromagnetic materials are mainly iron (Fe), cobalt (Co), and nickel (Ni)1. These elements possess a high number of unpaired electrons in their atomic or molecular orbitals, contributing to the development of magnetic moments. Ferromagnetic particles are employed in different applications because of their unique magnetic properties. Some key applications include Storage Devices in computers, Magnetic Resonance Imaging (MRI) and Electromagnetic Devices. Ferrofluids are colloidal suspensions of nanoscale ferromagnetic particles in a liquid carrier, typically composed of oil or water. Ferrofluids were first used for the intention of making a suitable liquid fuel propelling the rockets in a zero-gravity environment2. The ferromagnetic particles, often made of iron oxide or other magnetic materials, exhibit superparamagnetism, meaning they behave as individual magnetic dipoles with no overall magnetic moment without applied external field. The unique characteristic of ferrofluids lies in their responsiveness to external magnetic fields, causing the nanoparticles to align and produce a remarkable increase in the fluid’s magnetization. The stability of ferrofluids is maintained by a surfactant coating on the nanoparticles, preventing their agglomeration and ensuring a uniform distribution within the liquid carrier. This combination of nanoscale ferromagnetic particles and colloidal stability gives ferrofluids their distinctive properties, allowing them to be manipulated and controlled with precision.
Altering ferrofluids dynamic behavior by applying magnetic field is one of the interesting areas of research for various applications. Ferrofluids are used in diverse applications across several fields. In technology and engineering, they are used in seals and bearings to eliminate friction and wear in rotating machinery, enhancing overall system performance3. In the field of medical diagnostics, ferrofluids serve as contrast agents in MRI, improving the appearance of biological tissues and aiding in disease identification. Additionally, ferrofluids have been employed in various emerging technologies, such as microfluidics and lab-on-a-chip devices that unique magnetic properties gives rise to accurate control and manipulation of fluids at the microscale3. The versatility of ferrofluids continues to drive innovation, with ongoing research exploring new applications in fields ranging from robotics and electronics to biomedicine and environmental science. The dispersion of droplets holds significance in various industrial contexts, notably in the spray cooling procedure, due to its effectiveness in extracting heat from solid surfaces. This method is commonly employed in electronic cooling and other applications with high heat flux4. In cooling by the droplet spraying, as they dispersed across the surface, either undergo evaporation or form a finite size liquid film because of the heat of evaporation. This results in the release of substantial energy at lower temperature. The ultimate configuration of the droplet remains vital in determining the efficacy of the substrate coating5. Therefore, a detailed understanding of the intricate behavior involved in droplet spreading is indispensable for achieving a proficient and regulated deposition of droplets on designated substrates.
The characteristics of droplet impact are primarily influenced by dynamic settings and liquid properties. When the force created due to pressure exceeds the surface tension, it can result in splashing or droplet break-up on the impacted surface6. This phenomenon typically occurs when the Weber number surpasses a critical splash threshold. The influence of Weber and gravitational Bond numbers (Bo) on the bouncing time, maximum spreading time, maximum spreading ratio, maximum elongation ratio, and maximum restitution were investigated previously7. The effect of Weber number on the impacting droplet characteristics is more pronounced for bouncing time, maximum spreading time and maximum spreading ratio. The formation ferro droplets on surfaces becomes important in the magnetic field. From this end, the neck evolution during the formation of a ferrofluid droplet due to a capillary influence was investigated earlier and in the proximity of the droplet detachment, the droplet pinch-off process was observed8. In biomedical contexts, droplet splashing is particularly concerning, as it can lead to the spread of contaminated fluids across the medically treated surfaces. The Weber number, which is \({\text{We}} = \frac{{\uprho {\text{V}}^{2} {\text{R}}}}{\upgamma }\), where ρ is density, V is velocity, R is droplet radius, and γ is the surface tension, influences droplet behavior. Splashing is often triggered by the broadening of the droplet rim, which can initiate at a Weber number as low as 209. In many biomedical applications, droplet splashing is a significant mechanism for spreading and transporting infectious fluids within tide-controlled units10. The behavior resulting in rim expansion at low Weber numbers can be managed by modifying the force balance at droplet. Introducing forces, like dielectrophoretic forces through electrowetting, can be used to manipulate the splashing and rebound behavior of impacting droplets, thereby helping to control fluid dispersion11.
Extensive research has been conducted to analyze the behavior of droplets impacting hydrophobic and hydrophilic surfaces. Two main factors—namely, the Weber number and the hydrophobic nature of the surface play a crucial role in determining whether a droplet will rebound. This effect is even more pronounced for droplets impacting curved surfaces like glass spheres. When droplets hit such curved surfaces, variations in the Weber number significantly influence the height and dynamics of droplet rebound and the possibility of break-off. Additionally, as the curvature of the surface increases, the velocity at which the droplet becomes pinned to the surface decreases, further affecting its movement12. For heterogeneous surfaces where the wetting properties are non-uniform, droplet impact behavior is influenced by the relative size of hydrophobic patches compared to the droplet size. When the hydrophobic patch size increases relative to droplet size, droplet spreading is enhanced at lower Weber numbers. In these scenarios, surface adhesion is reduced, and the droplet is less likely to break apart upon impact13. Moreover, the topography and texture of the surface can significantly alter the behavior of droplets on hydrophobized surfaces. When the droplet fluid penetrates textured features, it tends to slow down the spreading and retraction phases14. The presence of pinched-off fluid within the texture gaps can also affect the rebound height of the droplet15. However, when surfaces are designed with evenly spaced micro-post arrays that have isotropic hydrophobic properties, the distortions in droplet spreading and retraction are minimized. This design prevents fluid from becoming trapped within the surface textures, thereby eliminating the unwanted pinching-off phenomenon, and ensuring a more uniform droplet rebound16. The interfacial properties of ferrofluid droplets on hydrophobic surfaces behave differently compared to conventional liquid droplets when exposed to a magnetic field. This distinction arises due to the presence of magnetic particles within the ferrofluid, which alter the interfacial tension. As a result, the size of the droplet’s meniscus varies due to magnetic field, affecting the contact angle. These variations become more pronounced when the surface texture profile is irregular17. The impact dynamics of ferrofluid droplet changes on soft surface, such as PDMS surfaces. In this case, the magnetic, viscous, inertial, and interfacial energies non-trivially affect the equilibrium shape of the impacting ferrofluid droplet and collective role of these forces ensures the ferrofluid droplet exhibits different equilibrium shapes18. The non-uniform force field gradient created causes the formation of a hump-like structure inside the left bulge of the droplet, where the magnet is located close by, while altering the flow field in this region. This in turn leads to the asymmetric splitting of the droplet into two sister droplets19. Moreover, the dynamics of self-healing of near-surface dips due to the Moses Effect (deformation of the diamagnetic liquid/vapor interface by the magnetic field) was considered by Bormashenko20. He indicated that the surface tension of liquids essentially governed the formation of near-surface dips due to the presence of magnetic fields. The droplet impact dynamics and its effect on the droplet fluid rheological properties change when silica nanoparticles together with polymetric additives included in the droplet fluid. In this case, transition from “rebound” to “stick” occurs, due to increase of the friction force of the nanoparticles and polymer aggregation with the substrate, while demonstrating the increase of the sliding angle with impact velocity21. Additionally, the wetting state of droplets can be altered by adjusting the magnetic field strength. For instance, the droplet’s wetting behavior can be altered commencing the Cassie-Baxter state to the Wenzel state under a strong magnetic field. This transition generates inertial forces that can be harnessed to move droplets across surfaces22. The magnetic field also influences ferrofluid droplet motion during rolling. Although both forces due to external field and gravitation can cause deformation, the droplet’s rear edge deforms differently under magnetic influence23. In applications such as oil–gas separation, ferrofluid droplets are useful for encapsulating and removing unwanted materials from surfaces. The presence of semi-fluorinated ligands enables nanoparticles to blend seamlessly with oil, enhancing oil droplets motion when influenced by a magnetic force24.
Studying the impact of droplets, formed by ferrofluids, on solid surfaces holds crucial importance in diverse scientific and technological fields. This investigation provides insights into the complex dynamics of surface tension, magnetic forces, and substrate properties which are essential for optimizing many processes including inkjet printing, microfluidics, and spray coating. In microelectronics, it enables the development of precise patterning techniques, while in biomedical applications, it contributes to the design of targeted drug delivery systems. Furthermore, the understanding gained from ferrofluid droplet impact on solid surfaces has implications for environmental science, offering potential solutions for oil spill cleanup. Ultimately, this multidisciplinary research not only advances fundamental knowledge but also opens avenues for innovative technologies with broad applications across industries. While previous studies have explored the impact and spreading behavior of ferrofluid droplets on hydrophobic surfaces, they have not thoroughly examined the dynamic responses of these droplets, such as rebounding, break-off, and rates of spreading and contraction25,26. The current study addresses this gap by investigating how the impact height and volume of ferrofluid droplets affect their dynamic behavior under the magnetic field. In order to conduct the research, high-speed imaging was employed to capture the droplet dynamics in detail, and tracking software was used to analyze the recorded footage, allowing precise measurement of droplet characteristics after impact. Additionally, the study evaluates the role of varying ferro particles concentrations on impact behavior towards quantifying the pinning forces.
Experiment
A glass substrate measuring 60 × 20 × 2 mm3 (length × width × thickness) underwent hydrophobization in this study. Silica particles of about 30 nm sizes were synthesized employing tetraethyl orthosilicate (TEOS), isobutytrimethoxysilane (OTES), ethanol, and ammonium hydroxide solutions in line with the previous practice27. Using the process of dip-coating, glass surfaces were coated with the particles. Droplet contact angles and hysteresis were measured (Kyowa, model DM 501), while SEM was facilitated for micro-graphing of the coated substrates. The surface wetting characteristics were evaluated using a goniometer (Data-Physics, Model: OCA11). Additionally, the surface roughness was quantified through an atomic force microscopy (AFM) line scan.
Ferrofluid, consisting of average size of 50 nm ferro particles (Fe3O4, Sigma Aldrich) in water, was prepared with different concentrations of ferro particles having weight ratios of 0.005% (wt), 0.01%wt, and 0.05% (wt). The mixture underwent ultrasonic shaking for 15 h and before dispensing onto the hydrophobic surface; an additional 30 min of shaking ensured homogeneous ferro particle distribution. Moreover, the suspended ferro particles were prone to sedimentation due to van der Waals, gravitation, buoyancy, and magnetic forces, the stability of colloidal suspension needs to be assured through zeta potential assessment. The zeta potential was measured as − 30 mV, which demonstrates suspension of particles in the mixture28. Additional tests were conducted towards further assessing ferro particles suspension. The findings reveal that particle suspension in water was observed to be stable over 500 h. Two different droplet volumes are considered in the experiments including 20 µL, and 40 µL. Moreover, experiments utilized a strong magnet (NdFeB, K&J Magnetics Inc., USA) having a maximum magnetic strength of 11.806 × 105 A/m. A computer-controlled fixture positioned the magnet beside the surface hydrophobized, and a schematic view and image depicted the setup shown in Fig. 1. High-speed recordings (5000 fps, 1280 × 800 pixels, 14 µm × 14 µm pixel size) captured the impact of ferrofluid droplets which is analyzed with a tracker program. The uncertainty was determined using the flowing equation5:
where: \({\mu }_{e}\) is the velocity of droplet on impact, n is the velocity repeats, and p(x) is the function related to the probability distribution. By assessing the small peaks with reference to probability function, the standard uncertainty was found to be 2.2%. Experiments are carried out in the laboratory environment at 300 K temperature, 1 atm pressure, and 75% humidity.
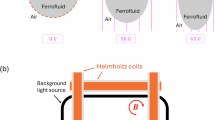
Illustration of impacting Ferro droplet on surface prepared with presence of magnetic field.
Results and discussion
This section explores the complex behavior of ferrofluid droplets on hydrophobic surfaces, emphasizing the interaction between droplet dynamics and external magnetic fields. A thorough analysis is presented to understand the influence of surface characteristics and magnetic forces on phenomena such as spreading, retraction, and rebound. The findings aim to provide a comprehensive understanding of the underlying physical mechanisms governing these interactions. Moreover, in order to relatively assess and validated present results for the droplet impact characteristics on hydrophobic surface, such as dmax/do (where is the maximum diameter of droplet after spreading period and do is the droplet diameter prior to impact), tests are extended to include the comparison of dmax/do measured from the present study with those reported in the early study29. Figure 2 shows dmax/do variation with the Weber number for water droplet. The findings revel that both results agree and discrepancies are with the error limits. It should be noted that the experimental error bars were not provided in the early study; therefore, the error bars are only included for the data points of the current study.
Comparison of droplet maximum diameter ratio with Weber number measured from this study and presented in the early study29.
Hydrophobized surface
Hydrophobized surface exhibits particles with approximately 30 nm sizes, some of which are piled, forming Nano-peaks. The peaks have the maximum value in the order of 150 nm27,30. Furthermore, the roughness (Ra) is approximately 110 nm, with Nano-size cavities formed between the peaks, covering about 30% of the projected area. Contact angle measurement employs the precision shape analysis method, where a view of the droplet from a Goniometer screen is utilized, and a mathematical function is accommodated to resemble the droplet circumference. A three-phase contour is identified in the image, typically appearing as a rake intersecting the function developed in horizontal direction. However, slight inclination may be observed in some instances, prompting the introduction of an angle of correction (αBL) in Eq. (2)
In this context, \(\Delta y\) represents the perpendicular distance between the line of horizon and the mid-point of the derived function, while R indicates the curvature radius. Measurement error, assessed through 15 repetitions, is approximately ± 2°. Figure 3 shows the contact angle measurements for droplets at different conditions. The contact angle droplet stands at ~ 150° ± 2°, with a hysteresis of 2° ± 1°. Additionally, slight variations in contact angles occur with changes in ferro particles concentrations in the droplet fluid; for instance, it measures 150° for zero concentration and 145° for a concentration of 0.05 wt%. This may occur because of the change of the droplet fluid properties by mixing of nanosized ferro particles, such as surface tension increases, i.e. \(\frac{{\gamma }_{m}}{{\gamma }_{w}}\) = 1.025, where \({\gamma }_{m}\) is surface tension of ferro particles mixed droplet fluid (0.05 wt%) and \({\gamma }_{w}\) is surface tension of water31. It was reported that the contact angle change signifies as the particle size reduces few nanometers32. However, external magnetic fields affect the droplet’s contact angle significantly, i.e. it reduces notably, as shown Fig. 5 i.e. it reduces to 137° for a concentration of 0.05 wt% after applying a magnetic field, which is in line with the early study33. Placing the hydrophobic sample beside the magnet gives rise to contact angle and hysteresis change due to the side magnetic effect. Inside the droplet, the ferro particles accumulate toward the magnet side due to the magnetic attraction, which alter the droplet shape and increase the hysteresis to reach 7°. Given that contact angle alteration correlates with magnetic field strength, there arises a need to formulate the equivalent fluid surface tension. Moreover, as the droplet becomes close to the magnet the Moses effect influence the droplet deformation and repulsion, which is more pronounced under a strong magnetic field20 In the current study, the magnetic flux intensity gradient (dB/dx, x is the normal distance away from the magnet) is in the order of 233 mT/m (Fig. 5). The simplified formulation of magnetic force acting on per unit volume of a droplet is \({F}_{mag-vol}=\frac{\chi }{{\mu }_{o}}B(x)\frac{dB(x)}{x}\), here \(\chi\) is the magnetic susceptibility, \({\mu }_{o}\) is magnetic permeability of free space (\(4\pi \times\) 10−7 H/m). The magnetic force acting on 30 µL droplet located 5 mm away from the magnet is \({F}_{mag}\sim\) 5.34 \(\times\) 10−4 N, which is larger than the droplet gravitational force (\(\sim\) 2.94 \(\times\) 10−4 N) under the free fall. In this case, droplet attaches the magnet surface.
Wetting analysis of ferro droplets at different Ferro particles concentration (A) pure water droplet, (B) water droplet with ferro particles concentration of 0.05 wt%, and (C) water with ferro particles concentration of 0.05 wt% under magnetic influence.
Magnetic effect on impacting droplet
The magnetic effect is related to the concentration of particles and the distance from the magnet to droplet. As the ferro droplet approaches the substrate, it gains acceleration due to the increase in the magnetic force. This force is contingent upon both the particle concentrations and the magnitude of the impact height. To ensure adequate wetting and coverage of the ferro particle surface by the fluid, the spreading coefficient of the fluid must maintain a value exceeding unity.
where: \({\gamma }_{p}\) is the surface free energy of particles (\(\sim 1.15\times {10}^{3} \; \text{mJ}/{\text{m}}^{2}\))34, \(\gamma\) is the fluid surface tension with a value of 0.0738 N/m, and \({\gamma }_{p-w}\) is the interfacial force with a value of \(\sim 0 .126\times {10}^{3} \; \text{mJ}/{\text{m}}^{2}\)31,35,36. The spreading factor becomes positive and around 946 \(\text{mJ}/{\text{m}}^{2}\) , therefore droplet wets the ferro particles. The interfacial force strength created on particles by droplet liquid plays a crucial role in retaining these particles within the liquid, since the magnetic force, which is exerted on the particles. When the magnetic influence surpasses the interfacial force, the particles may detach from the liquid, potentially leading to their impact on the hydrophobic substrate prior to the droplet itself. Figure 4 illustrates the magnetic flux intensity in relation to horizontal distance, as determined from the measurements. It is observed that the magnetic effect diminishes when the horizontal space increases away from the magnet.
Magnetic flux density variation parallel to droplet impact direction.
The force created due to magnetic field and acting on a particle within the liquid can be mathematically represented as23.
where: \(\phi\) represents the volumetric particle concentration, calculated as (\(\text{\% }wt \frac{({\rho }_{c}+{\rho }_{w})}{{\rho }_{c}}\)), \({M}_{s}\) denotes the saturated magnetization per unit volume, H signifies strength of magnetic field strength, \({m}_{\text{o}}\) stands for particle magnetic moment, \({K}_{B}\) is the Boltzmann constant, T denotes temperature, S represents spacing from magnet to droplet, \(L \left[\frac{{m}_{O}H}{{K}_{B}T}\right] =\text{coth}\left(\frac{{m}_{O}H}{{K}_{B}T}\right)-\frac{1}{\left[\frac{{m}_{O}H}{{K}_{B}T}\right]})\) is the Langevin function, and FR is the retention force due to gravity and surface tension along the s-direction. The Langevin function is approximately equal to unity due to the large order of the term (\({10}^{4}\)). Moreover, at high temperatures, ferro-liquid droplet magnetic properties change while influencing the magnetic force strength37 i.e. \(M(T)\propto (1-\frac{T}{{T}_{c}}{)}^{\beta }\), where M(T) is magnetization at temperature T, Tc is Curie temperature, and β is exponent (β \(\approx\) 0.33). The ferro particle size has influenced on the magnetic properties change at elevated temperatures. In this case, nano-size Ferro particles (Fe3O4 for particle size \(<\) 20 nm), the Curie temperature decreases with increasing particle size38. On the other hand, the magnetic field strength at room temperature can be determined via utilizing specific values such as \({\rho }_{c}\) = 5170.4 kg/m3, \({\rho }_{w}\)= 1000 kg/m3, \({M}_{s}\) = 6.6 × 10−3 mT for the particles, and 0.005 wt% for concentration39, magnetic field strength of H = 6.95 × 104 A/m, S = 20 mm of spacing from magnet to droplet, and a 20 μL volume of droplet. After assuming retention force is negligibly small as compared to magnetic force, the magnetic force is calculated to be \(\approx 2.3 \times {10}^{-5}\text{ N}\). When the ferrofluid droplet approaches the magnet, this force increases significantly due to the reduced distance (S) in the equation. Assuming the droplet impact (s) equals to diameter of droplet (4.67 mm) before the impact, the magnetic field strength becomes H = 7.22 × 104 A/m, resulting in a magnetic force influencing the particles is ~ 6.8 × 10−5 N. Conversely, the capillary force (\({F}_{\gamma }\)) required to maintain the ferro particles in the liquid has been previously formulated40.
Here, \({m}_{p}\) represents the mass of particles, γp−w is interfacial tension between particles (\(\sim\) 0.125 N m−1), and \({\rho }_{c}\) is ferro particle density (5170.4 kg/m3). For 0.05% (wt%) concentration in a droplet of 20 μL volume, the mass of the clustered particles is approximately 1.5 × 10−6 kg. Considering the interfacial tension at liquid-particle interface as 0.126 N/m40, interfacial force (Fγ) computed from Eq. (5) yields to \(\sim\) 1.47 × 10−4 N. Meanwhile, the force due to magnetic effect on these particles, as determined from Eq. (4), is approximately 6.8 × 10−5 N. Consequently, interfacial force surpasses the force acting on the clustered particles due to magnetic field, ensuring their retention within the droplet fluid rather than their separation and attraction towards the magnet. Furthermore, the particles tend to gather toward magnet direction inside the droplet under the combined influence of magnetic, gravitational, and buoyancy forces. To assess the relative significance of the magnetic force on particles at the droplet prior to impact, ratio of the magnetic force to sum of magnetic, gravitational, and buoyancy forces is evaluated. For 0.05% (wt%) concentration in a 20 μL droplet liquid, with the magnetic force at approximately 6.8 × 10−5 N, the gravitational force almost 1.472 × 10−5 N, and the buoyancy force at approximately 2.9011 × 10−7 N, the calculated ratio exceeds 0.8. Consequently, the magnetic force exerts a greater influence than the capillary force on the particles gathering at bottom of droplet liquid prior to impact, as per Eqs. (3)–(5).
However, to detach the particles from the water, the magnitude of the magnetic force must exceed the surface tension of the ferrofluid, as represented in Eq. 5. The minimum distance (S) required to prevent the magnetic particles from contacting the liquid is formulated in Eq. 8. During the droplet’s descent, these forces influence the falling profile of droplets with different ferrofluid concentrations.
Critical distance (Sc)
To assess the critical distance (Sc), at which the particles are detached from the droplet liquid to magnet surface, a new force ratio is introduced in terms of externally applied magnetics force (\({F}_{m}\)) over the gravitational force (\({F}_{g}\)). The force ratio can be expressed as in a new number, which yields:
where \(EB\) is the new number, which becomes:
where, md is the total droplet mass prior to ferro particles pulled-off from the droplet by the magnet and it consists of mass of the droplet fluid (\({m}_{d-l}\)) and mass of the ferro particles (\({m}_{p}\)), i.e.: \({m}_{d}={m}_{d-l}+{m}_{p}\). The new number (EB) variation with the distance from magnet to droplet gives rise to determine the critical distance Sc. Figure 5 shows the variation of EB with the normalized distance S/So, where So is the distance from the magnet surface as the droplet location at the initial position where the impact height is maximum. It appears that increasing S/So lowers the new number (EB) gradually. The minimum normalized distance ratio is the same as the distance corresponding to ferro particles pulled off (Sc) over So (Sc/So). Therefore, using the experimental data for S, the critical force ratio (EBc) for particles to be pulled off becomes in the order of 4500 and the critical distance ratio yields about 0.026. Hence, the ferro particles cannot be pulled off by the magnet at any distance ratio less than 0.026. Using the values in Eq. 8 results in Sc/So < 0.3351; hence the value estimated from Eq. 10 satisfies the condition stated in Eq. 8 for Sc, i.e. Sc/So = 0.026 < 0.3351.
Ratio of magnetic force to gravitational force with normalized distance between the droplet and magnet surface.
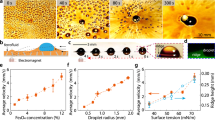
Figure 6 illustrates the optical images of ferro droplets at various ferro particles concentrations prior to impact. In Fig. 6a, the impact of a ferro droplet without a magnet is shown. Here, the ferro particles settle at the bottom of the droplet due to gravity, resulting in a straight falling profile since there is no magnetic force to alter the droplet’s shape or falling trajectory. Conversely, Fig. 6b,c demonstrate the ferrofluid droplet impact profiles in the presence of a magnetic field. The magnetic force causes the particles within the liquid to move in the side of the magnet (as indicated by the small arrows beside the droplets), elongating the droplet as it is attracted by the magnet. Additionally, the spacing from magnet to droplet (s) significantly affects the impact profile. As the droplet closes to the magnet, the magnetic flux increases. The particle detachment occurs when the magnetic force surpasses the capillary force. This interaction highlights the critical role of magnetic force in altering the dynamics of ferro droplet impact and deformation.
the optical images of ferro droplets at various ferro particles concentrations prior to impact: (a) no magnetic field applied, (b) 0.01 wt% concentration under magnetic effect % and (c) 0.05 wt% concentration under magnetic effect.
Figure 7 presents the complete impact profiles of ferro droplets with varying concentrations and sizes at different distances from the magnet. The figure tracks the droplet position reference to the magnet during free fall before impacting on the surface. Notably, at a small spacing of 5 mm, the droplets adhere to the magnet due to magnetic attraction, which pulls the particles towards the magnet. This effect is particularly evident in droplets with higher ferro particles concentrations. For instance, Fig. 7b, which depicts a droplet with a size of 40 µL, shows a more pronounced approach towards the magnet compared to Fig. 6a with 20 µL droplet size and the 0.05 wt% concentration. The increased ferro particles concentration enhances the magnetic susceptibility of the droplet, thereby increasing its tendency to be pulled towards the magnet (Eq. 4). This behavior underscores the significant consequence of both the magnetic field strength and the ferrofluid concentration on the impact dynamics and rebound characteristics of the droplets.
Impact profiles of ferro droplets with varying concentrations at different distances from the magnet. During free fall, the location of droplet becomes close to the magnet, which depends on the ferro particle concentrations.
Figure 8 displays high-speed images capturing different stages of the droplet impact. Due to the magnet’s attraction of ferro particles, the impacting droplet undergoes both rebounding and side movement. However, the droplet experiences fewer jumps, and the nature and amplitude oscillation are different compared to a droplet without magnetic attraction. Consequently, the magnetic force suppresses particle kinetic energy in rebound cycle, particularly noticeable with higher concentration ferro particles (0.05% wt). Moreover, the magnetic force affects the shape of the impacting droplet, causing particles to cluster at the side of the droplet near the magnetic flux direction (Fig. 8c). This clustering occurs as the droplet approaches the magnet, with the droplet liquid spreading after impact while the clustered particles accumulate to the side due to the strong force acting on them. Additionally, as the droplet becomes close to the magnet, a magnetic field strength in terms of magnetic flux intensity increases (Fig. 4). This can create a localized magnetic potential that attracts the droplet due to droplet magnetic susceptibility. This attractive force, due to the Moses affect, acts in addition to surface tension and substrate interactions while effectively altering the inertial-capillary dynamics that govern droplet spreading and retraction41,42. Hence, increasing particle concentrations elongate the spreading length to the magnet side, changing the droplet shape and pushing the impacted droplet to move toward the magnet, i.e. the suppression effect of magnetic field on the lateral expansion of the droplet, accumulates the kinetic energy in the vertical direction, which was also observed and reported in the early study43. Furthermore, the magnetic force (where droplet is located at the impacted surface), which is ~ 6.8 × 10−5 N exceeds the capillary force, which is ~ 2.69 × 10−6 N, causing the fluid to detach from the impact surface and separate from the pinned ferro particles during rebounding, as depicted in Fig. 8c. It is worth mentioning that after a droplet impacts and retracts on a hydrophobic or superhydrophobic surface, the momentum of retraction causes a central upward rebound to form. This rebound can undergo necking due to capillary instabilities (like the Rayleigh–Plateau instability), eventually causing break-off of a satellite droplet44. Interfacial tension across the droplet fluid and particles (~ 1.47 × 10−4 N) exceeds the magnetic force (~ 6.8 × 10−5 N) acting on the clustered ferro particles, ensuring the strong interfacial force facilitates the breakoff of the droplet in rebound cycle, while generating a small droplet, having a high particle concentration, to pin at the magnet. This breakoff leads to an initiation of momentum resulting in droplets rolling on the hydrophobic surface toward the magnet. In terms of Weber and Ohnesorge number, the break off is observed at We \(\ge\) 20 and Oh \(\ge\) 0.18 × 10−2 for ferro particle concentration 0.05 wt%. The height of the droplet in rebound cycle correlates with the restitution coefficient (e), which is related to the ratio of rebound height to impact height \((\sqrt{\frac{{h}_{R}}{{h}_{I}}})\), where \({h}_{R}\) is rebound height and \({h}_{I}\) is droplet free-fall height before impact.
where: \({V}_{d-B1}\) is pre-impact velocity, \({V}_{d-1}\) is the initial droplet velocity, equal to zero, \({V}_{d-1A}\) is droplet velocity immediately after impact, and \({V}_{d-1S}\) is droplet velocity after impact, equal to zero.
High-speed photographs representing different stages of the droplet impact at different concentrations: (a) 0.005 wt%, (b) 0.010 wt% and (c) 0.050 wt%.
Figure 9 illustrates the restitution coefficient of the droplet as a function of the Weber number (We), which is defined as:
Variation of restitution coefficient for different particle concentrations.
Here ρ represents liquid density, \({V}_{d}\) is velocity of free-falling droplet, and \({D}_{d}\) is equivalent diameter of droplet. Various concentrations of particles are examined for comparison of the restitution coefficient. The data reveals the restitution coefficient inversely proportional to the Weber number, indicative of reduced energy transfer efficiency upon impact. Moreover, higher concentrations of ferro particles lead to a significant reduction in the restitution coefficient. This trend suggests that increasing ferro particles concentrations substantially influences the rebound height of the droplet due to the pronounced magnetic force exerted on the particles. On the other hand, the presence of ferro particles can alter the surface tension of the droplet; in which case, the equivalent surface tension under the magnetic influence can be considered, which yields45:
Here, \(\chi\) represents magnetic susceptibility, \(N\) is demagnetizing factor, BoM is magnetic Bond number (ratio of magnetic force over the surface tension force), s is surface area factor (\(s=\frac{{\text{sin}}^{-1}r}{r}+p\) where \(r=\sqrt{1-\frac{{b}_{0}^{2}}{{a}_{0}^{2}}}\) is the eccentricity and \(p=\frac{{b}_{0}}{{a}_{0}}\) is droplet aspect ratio, ao is droplet initial major axis, and bo is minor droplet axis). To simplify the arrangements, ferrofluid droplets can be considered to be deformed into an oval geometry. Inserting initial magnetic susceptibility \(\chi = 1.2\)46, demagnetizing factor \(N\)= 0.547 and considering \(\frac{{b}_{0}}{{a}_{0}}=0.7\), BoM ~ 0.1 (due to low concentration of Ferro particles), hence, \(\frac{{\gamma }_{m}}{\gamma }\) yields ~ 1.025, which results in 2.5% variation of surface tension. Increase in surface tension causes reduction in the Weber number while influencing the droplet roundness during rebounding. It is worth mentioning that increasing ferro particles concentration enhances the magnetic dipoles for a given droplet volume. This in turn modifies the magnetic susceptibility, i.e. \(\chi \propto {C}_{m}\), where Cm is the volume concentration. On the other hand, high concentration gives rise to magnetic clustering, which influence magnetic response of the droplet. Since the ferro particle concentrations incorporated in the present study is low, omittable change of susceptibility occurs48,49,50. Moreover, surface tension, shear, and gravitational forces influence the droplet motion on the impacted surface while influencing the droplet rebound height. The ratio of shear to surface tension forces as described by Ohnesorge number (\(Oh=\mu /\sqrt{\rho a\gamma }\), here μ is viscosity, ρ is the density, and a is the droplet size) and the ratio of droplet weight over the surface tension force as described by the Bond number (\(Bo=\frac{\Delta \rho g{a}^{2}}{\gamma }\), here \(\Delta \rho\) is density difference and g is the gravitational acceleration) remain important describing the droplet behavior. In addition, the Capillary number (\(Ca=\frac{\mu {V}_{d}}{\gamma }\)) become important for the relative effect of viscous drag forces reference to surface tension force. The Capillary number can be written in terms of Ohnesorge and Weber number, i.e. \(Ca=Oh.\sqrt{We}\). Ohnesorge number (Oh) takes the values in the range of ~ 0.26 × 10−2–0.16 × 10−2, Bond number varies within ~ 0.5–3.5, Weber number values changes within ~ 8–28, and Capillary number takes the values within ~ 1.4–18.5 for droplet at the impacted surface. Hence droplet evolves in a disk shape during expansion over impacted surface without suffering breakoff because of higher surface tension force than the shear force created inside droplet fluid, i.e. Ohnesorge number remains low. Gravitational force contributes to droplet deformation over the surface during the impact, since the gravitational force remains larger than the surface tension force. On the other hand, the droplet may pin at a point where spreading cycle ends due to the capillary effects, which is the case where the impacted surface has large size porous structures51. Since the average surface roughness of the impacted surface is in the order of 75 nm, the droplet pinning due to porous capillary force can be negligible. However, rough surfaces such as porous media, the impacting surface characteristics, such as porosity, pore size, and capillary absorption need to be considered in the analysis of the droplet dynamics52,53.
Figure 10 illustrates the droplet interaction duration (contact time) over the substrate surface versus the Weber number for different concentrations. The contact time is normalized by the capillary time (\({t}_{c}=\sqrt{\frac{\rho {R}^{3}}{\gamma }}\), here ρ is the droplet fluid density, R is the droplet radius before impact, \(\gamma\) is the surface tension). The contact duration represents the droplet stay on the impacted substrate surface prior to splashing (rebound). It includes both spreading and retraction phases and the contact time is expressed relative to the capillary time for a 20 μL volume of droplet (tc ~ 0.014 s). Figure 11 displays the spreading factor, defined by the ratio of maximum droplet diameter to initial droplet diameter, plotted against the Weber number at different concentrations of particles. Both interaction duration and spreading exhibit related patterns: as the Weber number increases, the droplet spreads more on the surface, resulting in a longer contact duration. In general, depending on the droplet Reynolds number, two regimes may occur during the droplet impact, namely inertial regime (Re ≥ 230) and viscous regime (Re ≤ 0.7)54,55. The Reynolds number is in the range of 250 in the current study, which falls in the inertial regime and the impact force exhibits self-similarity. In this case, the pressure on the impacted wall becomes the highest at the droplet center and an annular jet develops during spreading and pressure may vary from center to the droplet edge almost linearly while influencing the maximum droplet spread diameter56. However, increasing the ferro particles concentrations strengthens the magnetic force acting on the particles, causing them to become pinned to the hydrophobic surface and disrupting the fluid flow within the spreading droplet. The droplet experiences shear over the substrate surface and drag by air, but these effects on the momentum loss are consistent across all ferro particles concentrations, as the droplet size and area of wetting remain unchanged. This momentum loss results in a lower rebound height, which decreases further with higher particle concentrations due to stronger magnetic forces. For a Weber number of 13 and a ferro particles concentrations of 0.05%, the contact time is reduced by about 36% compared to a droplet without ferro particles; for a Weber number of 23, it is reduced by 33.3%. The spread factor, shown in Fig. 11, decreases by about 25% at a Weber number of 13 and 22% at 23. This reduction is different for contact time and the spreading factor, with contact time showing a larger decrease. This indicates that particle pinning has a greater impact on contact duration than on droplet spreading. Thus, ferro particle pinning not only lowers spread factor and lessens contact period. but has a more pronounced effect on contact duration. The ferro particles are packed toward the magnet, and interfacial forces between particles and liquid changes spreading on the surface. These forces act like a coil, pulling the fluid back toward the magnet during spreading and shortening the retraction phase. Meanwhile, the shear force (\({F}_{s}\)) is related to \({F}_{s}=\underset{0}{\overset{{R}_{d}}{\int }}\mu \frac{d{U}_{d}(r)}{dn}2\pi rdr\), where μ is viscosity, \({U}_{d}\) is velocity in droplet, r is variable radius, n is distance normal to surface, and \({R}_{d}\) is the equivalent radius across wetted surface. After assuming a circular wetted area, the shear force is found to be at around 6.12 × 10−7 N. In comparison, the interfacial force of the particles, approximately 1.47 × 10−4 N, is significantly larger than the force due to shear.
Droplet contact duration variation on hydrophobic substrate for different concentrations. Contact duration is normalized by capillary duration (0.014 s).
Spreading factor variation for different concentrations.
Conclusion
The behavior of a ferrofluid droplet impacting a hydrophobic surface under a magnetic field is analyzed, focusing on the droplet’s dynamics before and after impact. Altering particles concentration changes the internal magnetic effect inside droplet. This causes deformation of the droplet shape before impact. As the concentrations increase, the magnetic force becomes stronger, causing the ferro particles to form a cluster and accumulate at the magnet side of the droplet. The droplet then assumes an asymmetrical, oval shape because the magnetic force surpasses the gravitational force. Despite this distortion, the droplet remains intact during free fall because the interfacial force created, which is ∼ 1.47 × 10−4 N is larger than force resulted by the magnetic field, which is ∼ 2.28 × 10−5 N. After impact, the spreading and retraction behaviors of the ferrofluid droplet differ from those of a standard droplet without ferro particles. As the concentration increases, the spreading factor decreases. The average reduction in spreading factor is around 20%, highlighting the effect of magnetic forces on the droplet’s dynamics. The change in the restitution coefficient, which measures the efficiency of energy transfer during impact, is about 15.5% at a low ferro particles concentration (0.005 wt%) and jumps to 69% at a higher concentration (0.05 wt%). This indicates that as ferro particles concentrations rises, the reduction in the coefficient of restitution becomes significant and follows a nonlinear pattern. Moreover, the interfacial force is much greater than the force created due to capillary effect, causing the droplet to split. A new number is introduced assessing the critical spacing (minimum spacing) between the droplet free-fall location and the magnet at which the ferro-particles breakoff from the droplet. As the concentration increases and the new number attains large values, the critical break-off distance reduces. The variation of droplet trajectory under the magnetic influence changes as the new number reduces. As a result, part of the rebounding droplet detaches, creating a smaller “newborn” droplet. This new droplet moves away toward the magnet and pinned to the magnet. This separation process reduces the momentum of the detached droplet, altering its rebound behavior. This demonstrates that the magnetic and interfacial forces significantly impact the overall rebound dynamics, altering both the droplet’s shape and motion during the impact and rebound phases.
Data availability
The datasets generated during the current study are available from the corresponding author on request.
References
Kim, Y. S. & Kim, Y. H. Application of ferro-cobalt magnetic fluid for oil sealing. J. Magnet. Magnet. Mater. 267(1), 105–110 (2003).
Kole, M. & Khandekar, S. Engineering applications of ferrofluids: A review. J. Magn. Magn. Mater. 537, 168222 (2021).
Guo, H. & Barnard, A. S. Thermodynamic modelling of nanomorphologies of hematite and goethite. J. Mater. Chem. 21(31), 11566–11577 (2011).
Ali, A. M., Yahya, N. & Qureshi, S. Interactions of ferro-nanoparticles (hematite and magnetite) with reservoir sandstone: implications for surface adsorption and interfacial tension reduction. Petrol. Sci. 17, 1037–1055 (2020).
Bhattacharya, S., Charonko, J. J. & Vlachos, P. P. Particle image velocimetry (PIV) uncertainty quantification using moment of correlation (MC) plane. Meas. Sci. Technol. 29, 115301 (2018).
Riboux, G. & Gordillo, J. M. Experiments of drops impacting a smooth solid surface: a model of the critical impact speed for drop splashing. Phys. Rev. Lett. 113, 24507 (2014).
Akbari, M. J., Bijarchi, M. A. & Shafii, M. B. Experimental investigation on the bouncing dynamics of a liquid marble during the impact on a hydrophilic surface. J. Coll. Interface Sci. 662, 637–652 (2024).
Bijarchi, M. A., Favakeh, A., Mohammadi, K., Akbari, A. & Shafii, M. B. Ferrofluid droplet breakup process and neck evolution under steady and pulse-width modulated magnetic fields. J. Mol. Liq. 343, 117536 (2021).
Quintero, E. S., Riboux, G. & Gordillo, J. M. Splashing of droplets impacting superhydrophobic substrates. J. Fluid Mech. 870, 175–188 (2019).
Bourouiba, L. The fluid dynamics of disease transmission. Ann. Rev. Fluid Mech. 53, 473–508 (2021).
Quetzeri-Santiago, M. A., Castrejón-Pita, J. R. & Castrejón-Pita, A. A. Scientific reports controlling droplet splashing and bouncing by dielectrowetting. Sci. Rep. 11, 1–8 (2021).
Atherton, S. et al. Drop impact behaviour on alternately hydrophobic and hydrophilic layered bead packs. Chem. Eng. Res. Des. 110, 200–208 (2016).
Xu, J., Chen, Y. & Xie, J. Non-dimensional numerical study of droplet impacting on heterogeneous hydrophilicity/hydrophobicity surface. Int. J. Heat Mass Transf. 116, 951–968 (2018).
Abubakar, A. A., Yilbas, B. S. & Al-Qahtani, H. A water droplet impact on a hydrophobic soft surface. J. Fluids Eng. 143, 13 (2021).
Kooij, S. A. et al. Sprays from droplets impacting a mesh. J. Fluid Mech. 871, 489–509 (2019).
Chen, L., Xiao, Z., Chan, P. C. H. & Lee, Y. K. Static and dynamic characterization of robust superhydrophobic surfaces built from nano-flowers on silicon micro-post arrays. J. Micromech. Microeng. 20, 105001 (2010).
Guba, S., Horváth, B. & Szalai, I. Examination of contact angles of magnetic fluid droplets on different surfaces in uniform magnetic field. J. Magn. Magn. Mater. 498, 166181 (2020).
Shyam, S., Banerjee, U., Mondal, P. K. & Mitra, S. K. Impact dynamics of ferrofluid droplet on a PDMS substrate under the influence of magnetic field. Colloids Surf., A 661, 130911 (2023).
Shyam, S., Dhapola, B. & Mondal, P. K. Magnetofluidic-based controlled droplet breakup: Effect of non-uniform force field. J. Fluid Mech. 944, A51 (2022).
Bormashenko, E. Moses effect: Physics and applications. Adv. Coll. Interface. Sci. 269, 1–6 (2019).
Zang, D., Wang, X., Geng, X., Zhang, Y. & Chen, Y. Impact dynamics of droplets with silica nanoparticles and polymer additives. Soft Matter 9(2), 394–400 (2013).
Poesio, P. & Wang, E. N. Resonance induced wetting state transition of a ferrofluid droplet on superhydrophobic surfaces. Exp. Therm. Fluid Sci. 57, 353–357 (2014).
Asakura, H. et al. Deformation and motion by gravity and magnetic field of a droplet of water-based magnetic fluid on a hydrophobic surface. Appl. Surf. Sci. 253, 3098–3102 (2007).
Alp, G., Alp, E. & Aydogan, N. Magnetic liquid marbles to facilitate rapid manipulation of the oil phase: Synergistic effect of semifluorinated ligand and catanionic surfactant mixtures. Colloids Surf. A Physicochem. Eng. Asp. 585, 124051 (2020).
Zhou, J. & Jing, D. Effects of vertical magnetic field on impact dynamics of ferrofluid droplet onto a rigid substrate. Phys. Rev. Fluids 4, 83602 (2019).
Ahmed, A., Fleck, B. A. & Waghmare, P. R. Maximum spreading of a ferrofluid droplet under the effect of magnetic field. Phys. Fluids 30, 77102 (2018).
Abubakar, A. A., Yilbas, B. S., Al-Qahtani, H. & Alzaydi, A. Ammar, Liquid droplet impact on a sonically excited thin membrane. Soft Matter 18, 1443 (2022).
Salari, E., Peyghambarzadeh, S. M., Sarafraz, M. M., Hormozi, F. & Nikkhah, V. Thermal behavior of aqueous iron oxide nano-fluid as a coolant on a flat disc heater under the pool boiling condition. Heat Mass Transfer 53, 265–275 (2017).
Ahmed, A., Fleck, B. A. & Waghmare, P. R. Maximum spreading of a ferrofluid droplet under the effect of magnetic field. Phys. Fluids 30, 077102. https://doi.org/10.1063/1.5032113 (2018).
Yilbas, B. S. et al. Dynamics of a water droplet on a hydrophobic inclined surface: influence of droplet size and surface inclination angle on droplet rolling. RSC Adv. 777, 48806–48818 (2017).
Yilbas, B. S., Hassan, G., Al-Sharafi, A., Abubakar, A. A. & Al-Qahtani, H. Dynamics of ferro fluid droplet impact on hydrophobic mesh surface. RSC Adv. 15, 27167–27176 (2025).
Vafaei, S. et al. Effect of nanoparticles on sessile droplet contact angle. Nanotechnology 17, 2523 (2006).
Zhou, Q., Ristenpart, W. D. & Stroeve, P. Magnetically induced decrease in droplet contact angle on nanostructured surfaces. Langmuir 27, 11747–11751 (2011).
Guo, H. & Barnard, A. S. Thermodynamic modelling of nanomorphologies of hematite and goethite. J. Mater. Chem. 21, 11566–11577 (2011).
Yilbas, B. S. et al. Sliding and rolling motion of a ferro-liquid droplet on the hydrophobic surface under magnetic influence. Langmuir 38, 3925–3935 (2022).
Ali, A. M., Yahya, N. & Qureshi, S. Interactions of ferro-nanoparticles (hematite and magnetite) with reservoir sandstone: implications for surface adsorption and interfacial tension reduction. Pet. Sci. 17, 1037–1055 (2020).
Cullity, B. D., & Graham, C. D. Introduction to magnetic materials. In The physical principles of magnetism, (Morrish, A. H., ed. 2001) pp. 696 (John Wiley & Sons, 2011).
Kodama, R. H. Magnetic nanoparticles. J. Magn. Magn. Mater. 200, 359–372 (1999).
A. Elements, Iron (II,III) oxide nanoparticles /nanopowder, U.S. Regist. Trademark. © 1997–2022 Am. Elem. (n.d.). https://www.americanelements.com/iron-ii-iii-o xide-nanoparticles-nanopowder-1317-61-9 (2021).
Shikida, M., Takayanagi, K., Honda, H., Ito, H. & Sato, K. Development of an enzymatic reaction device using magnetic bead-cluster handling. J. Micromech. Microeng. 16, 1875 (2006).
Chu, L., Li, W., Zhan, Y. & Amirfazli, A. Magnetically responsive superhydrophobic surfaces with droplet manipulation capability. Adv. Eng. Mater. 25, 2201352 (2023).
Yang, C., Lei, Wu. & Li, G. Magnetically responsive superhydrophobic surface: in situ reversible switching of water droplet wettability and adhesion for droplet manipulation. ACS Appl. Mater. Interfaces. 10, 20150–20158 (2018).
Liu, X., Dai, Y., Zhao, H., Li, C. & Peng, Yu. Dynamics of ferrofluid droplet impact on slippery liquid-infused porous surfaces. Langmuir 41, 14807–14816 (2025).
Yarin, A. L. Drop impact dynamics: splashing, spreading, receding, bouncing. Annu. Rev. Fluid Mech. 38, 159–192 (2006).
Ahmed, A., Fleck, B. A. & Waghmare, P. R. Maximum spreading of a ferrofluid droplet under the effect of magnetic field. Phys. Fluids. 30, 77102 (2018).
Bai, K. et al. Effective magnetic susceptibility of suspensions of ferromagnetic particles. J. Appl. Phys 124, 123901 (2018).
Franklin, T. A. Ferrofluid flow phenomena. (2003).
Zahn, M. Magnetic fluid and nanoparticle applications to nanotechnology. J. Nanopart. Res. 3, 73–78 (2001).
Neuringer, J. L. & Rosensweig, R. E. Ferrohydrodynamics. Phys. Fluids 7, 1927–1937 (1964).
Odenbach, S. Magnetoviscous effects in ferrofluids (Springer, Berlin, Heidelberg, 2002).
Lee, J. B., Derome, D. & Carmeliet, J. Drop impact on natural porous stones. J. Colloid Interface Sci. 469, 147–156. https://doi.org/10.1016/j.jcis.2016.02.008 (2016).
Hapgood, K. P., Litster, J. D., Biggs, S. R. & Howes, T. Drop penetration into porous powder beds. J. Colloid Interface Sci. 253, 353–366 (2002).
Zhang, F., Shen, Z., Li, H., Li, X. & Yuan, P. Dynamics of drops on hygroscopic ionic liquid aqueous solution-wetted porous surfaces. Int. Commun. Heat Mass Transfer 139, 106495. https://doi.org/10.1016/j.icheatmasstransfer.2022.106495 (2022).
Zhang, B. et al. Experimental studies on the effect of Reynolds and weber numbers on the impact forces of low-speed droplets colliding with a solid surface. Exp. Fluids 58(9), 1–12. https://doi.org/10.1007/s00348-017-2413 (2017).
Gordillo, L., Sun, T. P. & Cheng, X. Dynamics of drop impact on solid surfaces: evolution of impact force and self-similar spreading. J. Fluid Mech. 840, 190–214. https://doi.org/10.1017/jfm.2017.901 (2018).
Lv, Q., Li, J., Guo, P., Zhang, B. & Tang, P. Effect of Reynolds number on impact force and collision process of a low-velocity droplet colliding with a wall carrying an equal-mass deposited droplet. Int. J. Multiph. Flow 163, 104432. https://doi.org/10.1016/j.ijmultiphaseflow.2023.104432 (2023).
Acknowledgements
The authors acknowledge the support of the Interdisciplinary Research Center for Sustainable Energy Systems (IRC-SES), King Fahd University of Petroleum and Minerals (KFUPM), and King Abdullah City for Atomic and Renewable Energy (K.A. CARE) to accomplish this work.
Author information
Authors and Affiliations
Contributions
Bekir Sami Yilbas: Methodology, Visualization, Investigation, Writing—original draft, review & editing. Abba Abdulhamid Abubakar: Methodology, Investigation, Writing—original draft. Ghassan Hassan: Methodology, Writing—review & editing. Hussain Al-Qahtani: Investigation, Writing—review & editing. Abdullah Al-Sharafi: Investigation, Writing—review & editing.
Corresponding author
Ethics declarations
Competing interests
The author declares that there is no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Hassan, G., Yilbas, B.S., Abubakar, A.A. et al. Impact of ferrofluid droplets on hydrophobic surfaces with presence of magnetic fields. Sci Rep 15, 43621 (2025). https://doi.org/10.1038/s41598-025-27534-4
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-27534-4