Abstract
Chronic myeloproliferative neoplasms are characterized by clonal myeloid expansion driven by activating mutations in the JAK2 pathway and chronic inflammation. The aim was to investigate the contribution of circulating inflammatory mediators to the abnormalities in the megakaryocytic lineage characteristic of MF and ET. Plasma samples from 30 MF and 28 ET patients were incubated with normal cord-blood CD34 + progenitors and megakaryo/thrombopoiesis was evaluated. MF plasma increased megakaryocyte output, which was attenuated in sequential samples from ruxolitinib-treated patients. JAK1/2, MAPK and NF-kB inhibitors reverted this effect, revealing the concomitant involvement of all three pathways. Elevated levels of circulating IL-1β and IL-6 correlated with megakaryocyte output, which was reverted by blocking antibodies, indicating this phenotype is partly driven by these inflammatory cytokines. Instead, ET plasma promoted enhanced proplatelet formation, which was coupled with increased NFE2 and Bcl-xL expression. Elevated levels of circulating RANTES correlated with ET plasma-induced proplatelet formation, which was partially reverted by RANTES receptor CCR5 antagonist Maraviroc, indicating RANTES is involved in this process. These findings indicate that, in addition to clonal mutations, extrinsic inflammatory mediators play a direct role in MF and ET megakaryocyte abnormalities. The distinct cytokine profile could potentially be useful for the development of targeted therapies.
Similar content being viewed by others
Introduction
Megakaryopoiesis begins with the differentiation of a hematopoietic stem cell followed by megakaryocyte (MK) lineage proliferation, polyploidization and maturation. This process is strictly regulated by transcription factors, such as RUNX1, GATA-1 and FLI-1, and cytokines, such as thrombopoietin and IL-61,2. In normal hematopoiesis, megakaryocyte progenitors have a low proliferation rate and megakaryocytes (MK) are relatively rare cells in healthy bone marrow. Thrombopoiesis is the final step of MK maturation leading to proplatelet formation (PPF), where transcription factor NFE2, and certain chemokines, such as RANTES, are involved3. Alterations in megakaryopoiesis and thrombopoiesis have been described in several disorders, including myeloproliferative neoplasms (MPN).
Myelofibrosis (MF) and essential thrombocythemia (ET) are MPN belonging to the Philadelphia chromosome-negative (Phi-) group, which are characterized by abnormal proliferation of megakaryocytic progenitors and chronic inflammation. These diseases are closely related and share driver mutations, JAK2V617F, CALR and MPL4. These mutations induce the constitutive activation of the JAK/STAT signaling pathway, which gives the hematopoietic mutated clone a proliferative advantage. MF is associated with the worst outcome among MPNs. Although there is marked expansion of the megakaryocytic lineage in MF, MK are dysplastic, immature and promote bone marrow fibrosis through the release of profibrotic and proinflammatory factors into the microenvironment5,6. Patients with ET present thrombocytosis and hyperproliferation of large, mature MK that display exacerbated PPF with increased number of bifurcations and tips in culture7. Chronic inflammation is a key feature of MPN that contributes to disease pathogenesis. High levels of a spectrum of inflammatory cytokines, including IL-1β and IL-6, are found in circulation8. The cytokine profile is variable, both between MF and ET, as well as among individuals within each pathology9. This inflammatory state is more pronounced in MF than in ET and is associated with MF disease progression and worse outcome8,10. JAK1/2 inhibitor ruxolitinib improves constitutional symptoms and reduces splenomegaly in MF patients11,12,13. The efficacy of this drug is largely due to the improvement in the cytokine profile rather than to direct action on the neoplastic clone14. However, ruxolitinib treatment for prolonged periods of time was not able to completely revert plasma cytokines levels to normal values, suggesting that, in addition to JAK/STAT hyperactivation, other signaling pathways are involved15. In this regard, enhanced activation of NF-κΒ and MAPK was shown in MF hematopoietic stem cells16.
The effect of proinflammatory cytokines on megakaryopoiesis has been previously demonstrated. In addition to the well-recognized role of IL-6, IL-1β has been shown to exert a direct stimulating effect on megakaryopoiesis by activating NF-κΒ and MAPK16,17,18. In addition, elevated IL-1β and IL-6 in several inflammatory diseases trigger thrombopoietin production, which, in turn, promotes megakaryopoiesis in vivo19. However, it remains to be established whether elevated cytokine levels play a role in megakaryocytic abnormalities displayed by ET and MF patients and whether inflammatory signaling pathways, such as NF-κΒ and ΜAPΚ, could be involved in the response of MK to inflammation in these conditions.
Given the central role of MK in these neoplasms, the predominat role of inflammation and the phenotypic differences between MF and ET, the aim of this study was to assess the effect of soluble inflammatory mediators present in the plasma of MF and ET patients on the megakaryocytic lineage and identify the intracellular pathways involved.
Results
Soluble factors in the plasma of MF patients induce exacerbated MK proliferation
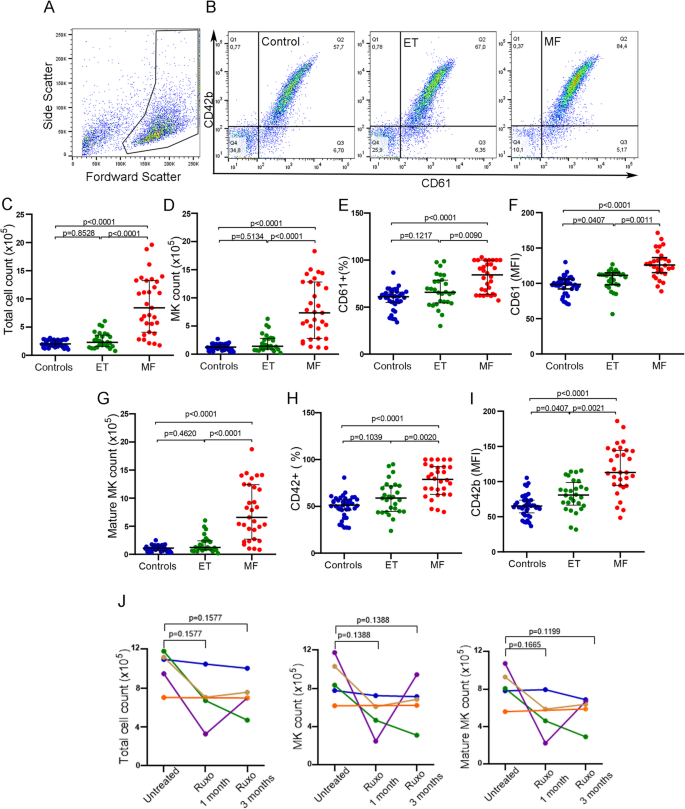
To evaluate the effect of soluble factors present in the plasma of MPN patients on megakaryopoiesis, plasma samples from untreated MF or ET patients were incubated with normal cord blood-derived CD34 + progenitors and cultured during 12 days under MK differentiation conditions. Total cell number was counted and the number of MK and mature MK (MMK) were calculated using the percentage of CD61+ (MK marker) and CD42+ (maturation marker) events obtained by flow cytometry. An example of MK selection strategy is shown (Fig. 1A, B). Plasma from MF patients induced an increase in total cell, MK and MMK number compared to control plasma. This increase was not observed with plasma from ET patients, which behaved similarly to the control group (Kruskal–Wallis followed by Dunn’s multiple comparisons test; Fig. 1C–I). Moreover, expression levels of CD61 and CD42b were also increased following incubation with MF compared to ET and control plasma, as evidenced by higher mean fluoresce intensity (Figs. 1F, I). We then analyzed the size and complexity of MKs incubated with plasma using the forward and side scatter data of the CD61 + population obtained by flow cytometry. CD34 + progenitors incubated with MF plasma exhibited normal size (FSC), but higher complexity (SSC) compared to MK incubated with control and ET samples (Fig. S1). Overall, these results indicate that soluble factors present in MF but not ET plasma lead to enhanced MK output and maturation.
Effect of myeloproliferative neoplasm plasma on CD34 + progenitors. Umbilical CD34 + progenitor cells were cultured in the presence of 10% plasma from normal controls (C, n = 40), essential thrombocythemia (ET, n = 28) or myelofibrosis (MF, n = 30) patients and TPO during 12 days to evaluate megakaryopoiesis. Cells were labeled with CD61-FITC and CD42b-PE antibodies and evaluated by flow cytometry. Cells were selected by forward scatter (FSC) versus side scatter (SSC) and analyzed by CD61-FITC and CD42b-PE expression to determine megakaryocyte (MK) percentage and maturation, respectively. (A) An example of selected gate for MK analysis is shown. (B) Representative analysis of MK output in cultures incubated with Control, ET and MF plasma analyzed by CD61 and CD42b staining is shown. (C) Total cell number (Neubauer chamber), (D) MK (CD61 + events), (E) Percentage of CD61 + events, (F) CD61 median florescence intensity, (G) Mature MK (CD61/CD42b + events) number, (H) Percentage of CD61/CD42 + events and (I) CD42b median florescence intensity were plotted. Kruskal–Wallis followed by Dunn’s multiple comparisons test. Bars and error bars indicate the median with interquartile range. (J) Effect of Ruxolitinib on MF plasma-induced megakaryopoiesis. Megakaryopoiesis was evaluated on CD34 + progenitors in the presence of sequential plasma samples obtained from MF patients without treatment and during treatment with ruxolitinib (at 1 and 3 months) (n = 5). Total cell, MK and mature MK count are shown. Friedman followed by Dunn’s multiple comparisons test. Bars and error bars indicate median with interquartile range. All experiments were carried out in triplicate in three independent umbilical cord blood samples.
Plasma from ruxolitinib-treated MF patients decrease MK numbers
Treatment with ruxolitinib leads to clinical improvement in MF patients, at least in part by reducing the levels of circulating inflammatory cytokines. As an approach to evaluate whether the effect of plasma factors on megakaryopoiesis could be due to elevated inflammatory cytokines present in MF circulation, we evaluated serial samples from 5 MF patients before treatment and at 1 and 3 months after starting treatment with ruxolitinib, using the above described system. Three patients showed a decrease in the total number of cells, MK and MMK at 1 and 3 months of treatment, compared to pre-treatment samples from the same patients, although without reaching statistical significance (Friedman followed by Dunn’s multiple comparisons test), in accordance with improvement of constitutional symptoms (Fig. 1J). These data suggest that the inflammatory cytokines present in patient plasma could be responsible, at least in part, for enhanced MK output induced by MF samples. On the other hand, MK numbers induced by incubation with the remaining two patient samples was not ameliorated during ruxolitinib treatment. Interestingly, these two samples belonged to patients whose clinical response to ruxolitinib was suboptimal or absent (orange and blue lines, Fig. 1J).
Influence of patient clinical and molecular features on abnormal megakaryopoiesis induced by MF plasma
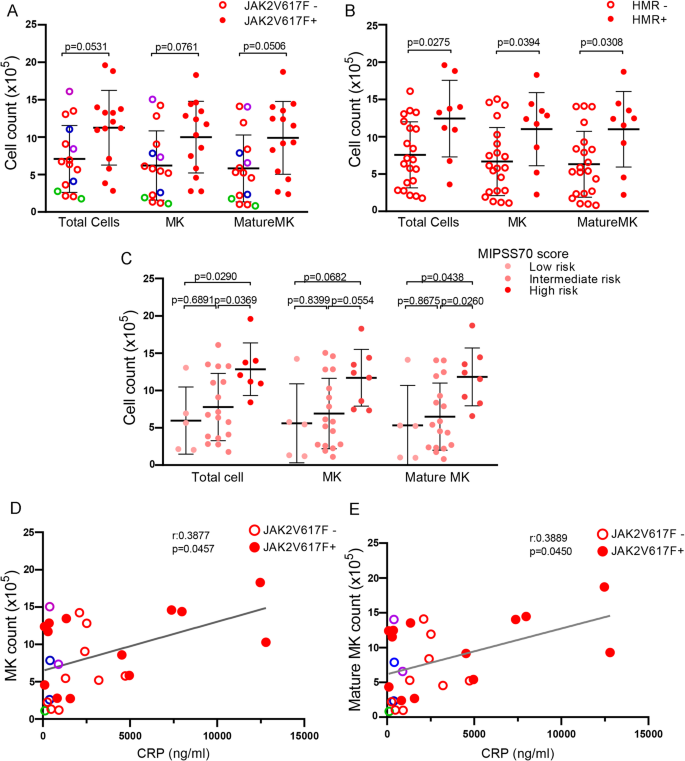
To analyze the influence of driver mutations on soluble factors present in samples that stimulated megakaryopoiesis, MF patients were divided according to their mutational status into JAK2V617F- and JAK2V617F+. A trend towards increased total cell, MK and MMK number was induced by plasma from MF patients carrying the JAK2V617F mutation (Student-t test with Welch´s correction; Fig. 2A), suggesting a relationship among the presence of the JAK2V617F mutation, plasma factors and the increase in cell number.
Relationship between clinical features and altered megakaryopoiesis induced by MF plasma. Myelofibrosis (MF) patients were grouped by mutational status in (A) JAK2V617F+ (n = 14)/JAK2V617F- (n = 16), including CALR1 (n = 10, red), CALR2 (n = 2, green), MPL (n = 2, violet) and triple-negative (n = 2, blue) patients or (B) by high-molecular risk (HMR) mutations positive (n = 9) including ASXL1 (n = 8), U2AF1 (n = 1), SRSF2 (n = 1, coexisting with ASLX1) or negative (n = 21). Total cell, megakaryocyte (MK, CD61+) and mature MK (CD61/CD42+) count are shown. Student-t test with Welch´s correction, bars and error bars indicate mean and standard deviation (SD). (C) MF patients stratified into low (n = 5), intermediate (n = 17), and high (n = 8) risk groups according to MIPSS70 score. Total cell, MK (CD61+) and mature MK (CD61/CD42+) count are shown. One-way ANOVA followed by Tukey´s multiple comparison test. Bars and error bars indicate the mean and SD. Correlation between C-reactive protein (CRP) levels and (D) MK or (E) mature MK number are shown in JAK2V617F+ (n = 13)/JAK2V617F- (n = 14), including CALR1 (n = 9, red), CALR2 (n = 1, green), MPL (n = 2, violet) and triple-negative (n = 2, blue) patients, Pearson correlation.
Plasma from patients harboring HMR mutations, including ASXL1, SRSF2 and U2AF1, induced significantly higher total cell, MK and MMK count (Student-t test with Welch´s correction; Fig. 2B). Megakaryopoiesis was also influenced by the degree of disease progression, as patients in higher risk groups, as stratified according to the Mutation-Enhanced International Prognostic Score System (MIPSS70), displayed higher MK output than those classified into lower risk groups (One-way ANOVA followed by Tukey´s multiple comparison test; Fig. 2C).
C-Reactive Protein (CRP), an inflammatory marker, was elevated in this cohort (data not shown), as reported20. Correlation between CRP and both MK and MMK numbers was found (Pearson correlation, Fig. 2D–E), mirroring the effect of MF plasma on megakaryopoiesis and suggesting a direct relationship between the inflammatory state and MK output.
Plasma from MF induces its effect on MK precursors
To assess whether the stimulatory effect of soluble factors occurs at the level of CD34 + progenitors or is exerted on immature MK, CD34 + cells were cultured during 12 days and patient plasma was added since day 1, as described in the previous assays, or at day 6 of culture when progenitors were more differentiated under MK culture conditions. Addition of MF plasma at day 6 of culture yielded an increase in total cell, MK and MMK number, to a similar extent as that observed when samples were added since day 1 (p = ns, Wilcoxon test; Figure S2). These results suggest that the stimulating effect of MF plasma factors can be exerted on cells already committed to the MK lineage, and imply that at least some of the cytokines implicated in this process are directly involved in the regulation of megakaryopoiesis. Of note, an increase in MK count was evident when addition of plasma from healthy subjects was delayed until day 6 of culture (Wilcoxon test), suggesting that steady-state plasma factors may have an inhibitory effect on MK differentiation. In contrast to MF, plasma from ET patients behaved similarly to control plasma, increasing total cell count when plasma was added at day 6 compared to day 1 (Fig. S2).
Plasma from MF upregulate Cyclin D1 expression
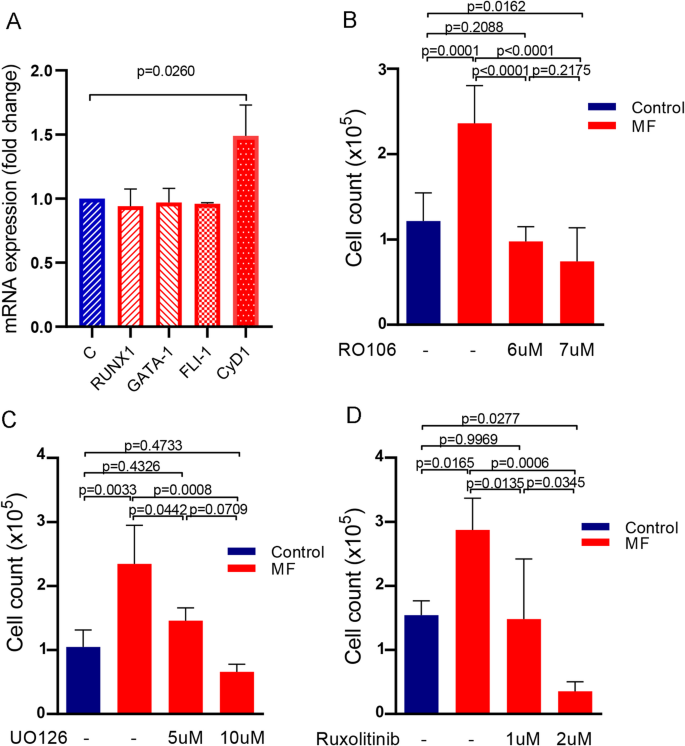
To study the mechanisms involved in MK stimulation, expression levels of MK transcription factors RUNX1, GATA-1 and FLI-1, main genes involved in megakaryocytic differentiation, were assessed by qPCR. To this end, CD34 + progenitors were incubated in the presence of MF or control plasma during 9 days followed by mRNA extraction. No significant increase was found respect to control, indicating that exacerbated megakaryopoiesis is not due to deregulation of these transcription factors (Mann-Whitney test; Fig. 3A). Then, we investigated the possible role of cell cycle alterations by focusing on cyclin D1, which participates in G1 to S phase transition, and showed increased expression levels of this cell cycle regulator, suggesting it could be involved on this process (Mann-Whitney test; Fig. 3A).
Mechanisms involved in the regulation of megakaryocyte proliferation by myelofibrosis plasma. (A) RNA expression of transcription factors involved in megakaryopoiesis. CD34 + progenitor cells were cultured in the presence of 10% plasma of myelofibrosis (MF) patients during 9 days, and after that, RNA was obtained (n = 6). Expression levels of RUNX1, GATA-1, FLI-1 and Cyclin D1 were quantified using GAPDH as housekeeping gene by qPCR. Fold-change with respect to the control (C) was plotted, Mann-Whitney test. Bars and error bars indicate the median with interquartile range. (B–D) Inhibition of inflammatory signaling pathways. CD34 + progenitors were cultured in StemSpan supplemented with TPO during 6 days. At day 6 of culture, megakaryocyte (MK) were treated with (B) RO106 (NF-κΒ inhibitor), (C) UO126 (MAPK inhibitor) or (D) Ruxolitinib (JAK1/2 inhibitor) during 1 h, then 10% MF plasma was added (C = 3; MF = 3). Cells were counted at day 12 of culture. Repeated-measures one-way ANOVA followed by Tukey´s multiple comparison test. Bars and error bars indicate the mean and SD.
NF-κΒ and MAPK pathways are involved in MK stimulation induced by MF plasma
Previous data indicate that cyclin D1 could be regulated through stimulation of the MAPK and NF-kB pathway by inflammatory cytokines21,22,23. To deepen into the mechanisms underlying enhanced MK number triggered by MF plasma, we focused on inflammatory signaling networks. In addition to the JAK2 pathway, NF-κΒ and MAPK have been shown hyperactivated in MF patients15,16. To this end, inhibitors of the NF-κΒ (RO106), MAPK (UO126) and JAK1/2 (ruxolitinib) pathways were added to cell culture at day 6. After 90 min, MF plasma was added to the culture system and cells were counted at day 12. Addition of each of these inhibitors was able to revert the increase in cell counts triggered by MF plasma, indicating that exacerbated cell proliferation induced by MF plasma is dependent on all of these signaling pathways (MF plasma with inhibitors vs. control plasma, p = ns, One-way ANOVA followed by Tukey´s multiple comparison test; Fig. 3B–D). As expected, all three inhibitors decreased MK proliferation in cells incubated with control plasma (Fig. S3).
IL-1β and IL-6 participate in MF plasma-induced MK stimulation
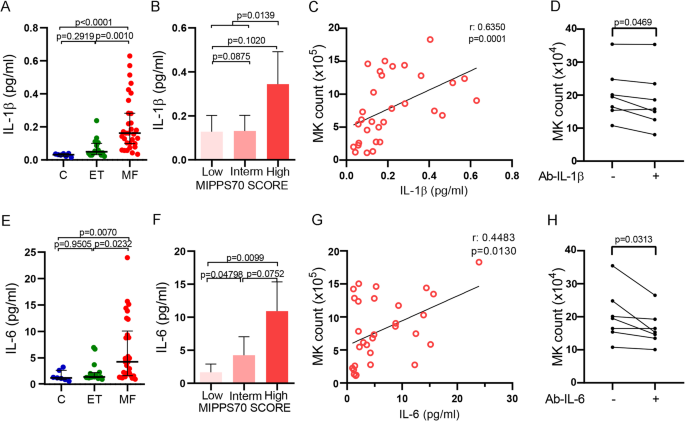
NF-κB and MAPK are important mediators in the inflammatory pathways which are activated mainly by proinflammatory cytokines, including IL-1β and IL-6, respectively16,17,18. Considering their important role in the development of the megakaryocytic lineage, plasma levels of IL-1β and IL-6 were measured in patients with MF and ET. Circulating IL-1β and IL-6 were elevated in our MF cohort, while levels in ET were similar to the control group (Kruskal–Wallis followed by Dunn’s multiple comparisons test; Fig. 4A, E). Indeed, a close association was found between plasma levels of these cytokines and MF plasma-induced MK output (Spearman Correlation; Fig. 4C, G), suggesting their role in this observation. Of interest, levels of both cytokines were higher in patients stratified into higher MIPSS70 scores (Kruskal–Wallis followed by Dunn’s multiple comparisons or Mann Whitney test for IL1β, low + intermediate vs. high risk group; Fig. 4B, F), in accordance with the increased MK output induced in vitro by plasma samples from patients belonging to this risk group. To determine the contribution of these cytokines, specific blocking anti-IL-1β or anti-IL-6 antibodies were added to the culture system prior to incubating MK with MF plasma. A representative dose-response curve of both blocking antibodies is shown (Fig. S4) and further experiments were carried out using 50 µg/ml of each antibody. Inhibition of MK output was observed with both blocking antibodies, further reinforcing the participation of IL-1β and IL-6 in this phenotype (Wilcoxon test; Fig. 4D, H).
Relationship between IL-1β and IL-6 and megakaryocyte output. Plasma levels of (A) IL-1β and (E) IL-6 in myelofibrosis (MF, n = 30), essential thrombocythemia (ET, n = 16) and control (n = 10) samples quantified by ELISA, were plotted. Kruskal–Wallis followed by Dunn’s multiple comparisons test. Bars and error bars indicate the median and interquartile range. MF patients were grouped according to the MIPSS70 score and levels of plasma (B) IL-1β and (F) IL-6 were plotted. Kruskal–Wallis followed by Dunn’s multiple comparisons or Mann Whitney test (IL1β, low + intermediate vs. high risk group). Bars and error bars indicate the median with interquartile range. Spearman correlation between (C) IL-1β or (G) IL-6 and megakaryocyte (MK) count. Inhibition of megakaryopoiesis in MF plasma samples. IL-1β and IL-6 were blocked in plasma using specific antibodies prior to the addition of MF plasma samples to day 6 MK culture. At day 12, cells were counted in a Neubauer chamber. MF samples (n = 7) were incubated with blocking (D) IL-1β or (H) IL-6 (50 µg, +) antibodies and compared to the same plasma samples without previous incubation with the corresponding antibody (-). Wilcoxon test.
Soluble factors present in ET plasma induce exacerbated PPF from normal MK
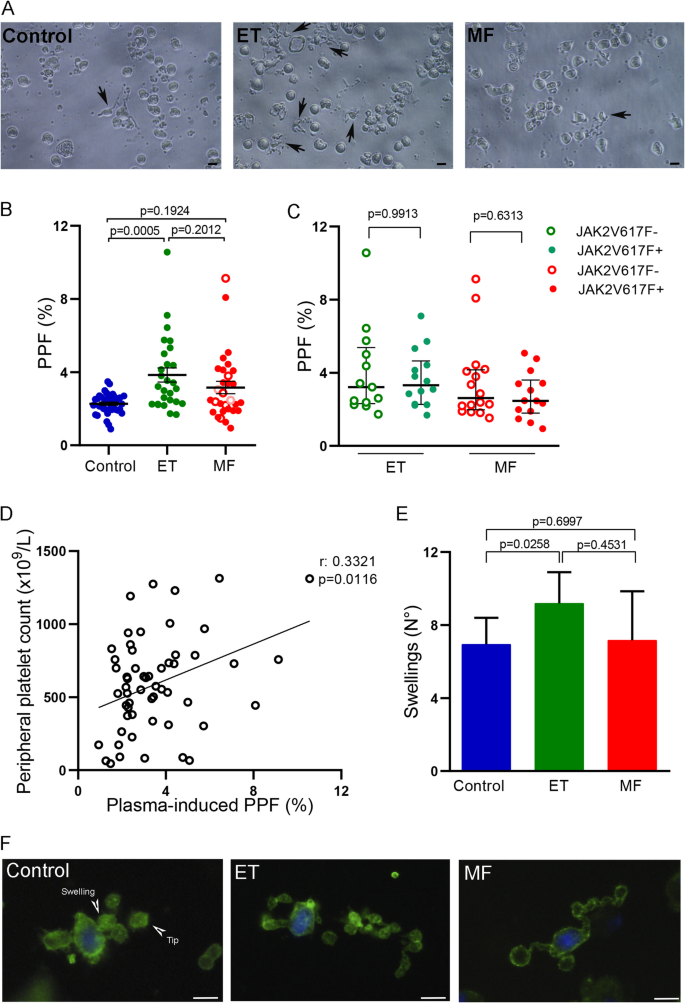
To study the effect of soluble mediators from MF and ET patients on thrombopoiesis, we assessed the ability of plasma to induce PPF from MMK. To this end, CD34 + progenitors were cultured during 12 days in MK differentiation conditions, in the absence of plasma. Then, 10% of MF and ET patient plasma was added to the culture and incubated until day 15. Representative examples of MK forming proplatelets are shown (Fig. 5A). A significant increase in PPF was induced by ET patient plasma compared to control group (Kruskal–Wallis followed by Dunn’s multiple comparisons test; Fig. 5B), without differences between JAK2V617F-positive and negative patients (Mann-Whitney test; Fig. 5C). Although no significant increase in PPF was found in the presence of plasma from the overall MF cohort (Fig. 5B), samples from 11 (37%) of 30 MF patients induced exacerbated PPF (higher than mean plus 2SD of control). Of the 11 samples with increased PPF, two are post-ET MF and 9 primary MF. Interestingly, the ability of MPN plasma to induce PPF correlated with platelet counts in patient peripheral blood (Spearman Correlation; Fig. 5D). To analyze proplatelet morphology, the number of swellings and tips contained in each proplatelet were counted. Proplatelet processes displayed an increased number of swellings and tips when MK were incubated in the presence of ET samples respect to control and MF group (Kruskal–Wallis followed by Dunn’s multiple comparisons test; Fig. 5E). A representative example of control, ET and MF proplatelet-bearing MK is shown (Fig. 5F).
Proplatelet formation in the presence of plasma from myeloproliferative neoplasms patients. Cord blood-derived day 12 megakaryocyte (MK) were incubated with 10% plasma during 72 hs. (A) Example of a MK producing proplatelets (inverted microscope) in the presence of control, essential thrombocythemia (ET) and myelofibrosis (MF) samples. The arrows indicate MK producing proplatelets. (B) Proplatelet processes were counted in an inverted microscope and proplatelets-bearing MK (PPF) number was calculated and referred to total cell count (Neubauer chamber). Results are expressed as percentage of PPF (Control, n = 40; ET, n = 28; MF, n = 30). Red empty circles represent post-ET MF, pink empty circles represent post-PV MF, red filled circles represent primary MF. Kruskal–Wallis followed by Dunn’s multiple comparisons test. Bars and error bars indicate the median with interquartile range. The experiments were carried out in triplicate in independent umbilical cord blood samples. (C) Percentage of plasma-induced PPF, according to the JAK2V617F mutational status of the myeloproliferative neoplasms (MPN) patients (p = ns, Mann-Whitney test). (D) Correlation between plasma-induced PPF in culture and peripheral platelet count in MPN patients (Spearman correlation). (E) Proplatelet morphology in the presence of plasma samples. MK were seeded in fibrinogen-coated glass during 72 hs, incubated in the presence of control (n = 6), ET (n = 6) or MF (n = 6) plasma. Then, cells were labeled with Hoechst (nucleus, blue) and CD61-FITC antibody (green). Number of swelling and tips per proplatelet process were counted in at least 50 MK. Kruskal–Wallis followed by Dunn’s multiple comparisons test. Bars and error bars indicate the median with interquartile range. (F) Representative example of proplatelet-bearing MK incubated with control, ET and MF samples. Swelling and tip are pointed out. Scale bars, 20 μm.
RANTES and NFE2 are involved thrombopoiesis stimulation induced by ET plasma
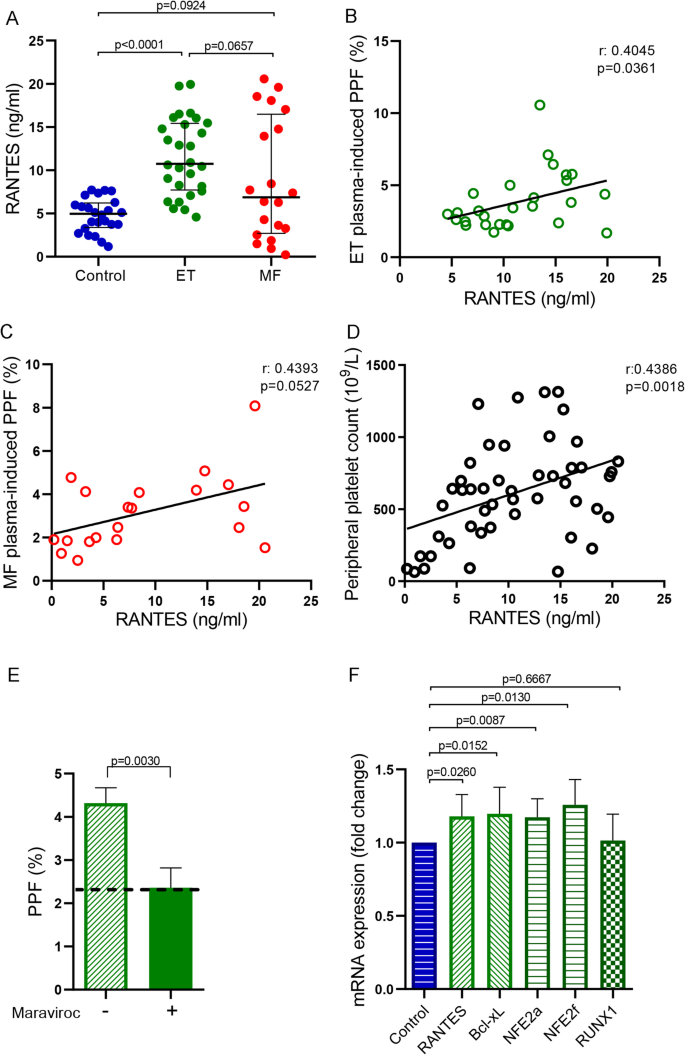
Although there is limited information about the extracellular factors that regulate PPF, RANTES (CCL5) has been recently shown to be involved in this process3. To identify possible factors underlying the increase in PPF induced by ET plasma, we measured circulating levels of RANTES and found it elevated in ET patients compared to control (Kruskal–Wallis followed by Dunn’s multiple comparisons test; Fig. 6A). Indeed, RANTES levels displayed a close relationship with ET plasma-induced PPF in vitro (Spearman Correlation; Fig. 6B). Patients with MF showed variable levels of plasma RANTES, ranging from normal to elevated, which also directly correlated with PPF in culture (Spearman Correlation; Fig. 6C). Overall, the association between RANTES levels and PPF in both ET and MF suggests that RANTES is implicated in the alteration in PPF observed, independently of the specific MPN subtype. Of note, circulating RANTES levels also correlated with peripheral blood platelet count in MPN patients (Spearman Correlation; Fig. 6D).
Regulation of thrombopoiesis by essential thrombocythemia plasma. (A) Plasma RANTES was quantified in controls (n = 24), essential thrombocythemia (ET, n = 28) and myelofibrosis (MF, n = 20) patients by ELISA. Kruskal–Wallis followed by Dunn’s multiple comparisons test. Bars and error bars indicate the median with interquartile range. Correlation between RANTES levels and (B) ET or (C) MF plasma-induced proplatelet formation (PPF) (Spearman correlation). (D) Correlation between plasma RANTES and peripheral platelet count in myeloproliferative neoplasms patients (Spearman correlation). (E) Umbilical CD34 + progenitor cells cultured in StemSpan, and TPO (10 ng/ml) were treated on day 12 during 1 h with Maraviroc (100 nM) and incubated with 10% ET plasma (n = 3) during 72 h. PPF were counted in an inverted microscope and the percentage was calculated relative to total cell count (Neubauer chamber). Percentage of PPF was plotted. Dotted line represent mean control plasma value. Paired t-test. Bars and error bars indicate mean and SD values. (F) mRNA expression of transcription factors involved in thrombopoiesis. Mature megakaryocytes were cultured in the presence of 10% plasma of ET patients during 48hs and then, mRNA was obtained (n = 6). RANTES, Bcl-xL, NFE2a, NFE2f and RUNX1 gene expression levels were assessed by qPCR using GAPDH as housekeeping gene. Mann-Whitney test. Bars and error bars indicate the median with interquartile range.
To corroborate the effect of RANTES in ET plasma-induced enhanced PPF, MMK were pretreated with Maraviroc, a specific antagonist of RANTES receptor (CCR5), prior to the addition of ET plasma. Maraviroc was able to revert the increase in ET plasma-induced PPF, further reinforcing the participation of RANTES in this phenotype (Paired t-test; Fig. 6E). An inhibition in PPF was also observed when MMK were treated with Maraviroc and control plasma (Fig. S5).
To assess whether patient plasma induce RANTES deregulation in MMKs, we evaluated its expression in these cells incubated with ET samples. Interestingly, RANTES expression was upregulated in normal MK incubated with ET plasma, raising the possibility that circulating factors present in ET could stimulate RANTES MK synthesis in vivo, potentially contributing to elevated levels of RANTES in patient circulation (Mann-Whitney test; Fig. 6F).
Considering that RANTES was shown to regulate platelet production by apoptosis inhibition, we assessed the expression of Bcl-xL, an antiapoptotic factor known to play an essential role in thrombopoiesis3,24. Interestingly, we found increased Bcl-xL expression in MK incubated with ET plasma samples that induced an increase in PPF in culture (Mann-Whitney test; Fig. 6F), raising the possibility that higher RANTES levels present in patient plasma could contribute to Bcl-xL upregulation.
NFE2 is the main transcription factor involved in PPF. An increase of NFE2 expression was found in MK incubated with these ET samples, whereas RUNX1 expression, which also regulates PPF, was normal, suggesting that NFE2 but not RUNX1 could be involved in the increase in thrombopoiesis (Mann-Whitney test, Fig. 6F).
Discussion
MPN are characterized by exacerbated growth of myeloid lineages and overproduction of mature blood cells. Driver mutations trigger the disease, although inflammation has been recently recognized to provide a proper environment for its development25. Megakaryocytic expansion is a prominent feature in both ET and MF. While ET MK are pleomorphic mature large cells which produce large numbers of elaborated proplatelets and platelets, MF patients display marked megakaryocytic hyperplasia leading to the formation of close clusters of atypical MK which release excessive amounts of profibrotic growth factors. Here we show that soluble factors present in plasma from MF patients contribute directly to enhanced proliferation of megakaryocytic precursors, while plasma from ET promote exacerbated PPF, indicating that extrinsic inflammatory mediators differentially contribute to megakaryocytic abnormalities that characterize each of these disorders.
In this work, we used CD34 + progenitors from the umbilical cord as an easily available source of MK. Cord blood (CB) and peripheral blood (PB) from adult donors are alternative sources of CD34 + hematopoietic progenitors for MK generation in vitro. Differences and similarities have been demonstrated in MKs obtained from CB compared to those from adult PB under TPO stimulation. MK output derived from CB CD34 + cells has been shown to be significantly higher than that from adult PB donors, indicating a proliferative phenotype in newborn MK committed progenitors26. Furthermore, once fully differentiated, these CB-derived MKs reach lower levels of ploidy and, consequently, produce fewer platelets than their adult counterparts27. However, both types respond to TPO, express similar levels of CD42b and exhibit alpha granules, demonstrating that CB-MKs do reach maturity27. Consequently, this system represents a suitable and extensively utilized experimental model for the study of MK biology and proplatelet formation.
As an approach to identify the soluble mediators present in MF circulation that trigger the increase in MK output observed and considering that JAK2-positive patients display higher levels of inflammatory cytokines8, we compared the effect of plasma samples from JAK2V617F-positive and -negative MF patients. Higher MK numbers were shown in the presence of plasma from patients harbouring the JAK2 mutation, suggesting that proinflammatory factors present in their plasma may contribute to this finding. Then, we evaluated the behavior of normal hematopoietic progenitors cultured in the presence of longitudinal MF samples obtained before and during treatment with the JAK1/2 inhibitor ruxolitinib, which has been shown to decrease circulating inflammatory cytokines, improving patients’ constitutional symptoms12. Ruxolitinib-treated samples ameliorated the increase in MK output induced by paired samples obtained before treatment, although without reaching control values. These results reinforce the participation of proinflammatory cytokines in the proliferative effect observed and point to the involvement of the JAK1/2 signaling pathway in this process. Our in vitro model is based on cord blood-derived MK. The effect of MPN plasma on patient primary cells, which display hyperactivated JAK2 signalling, and the potential influence of different intrinsic driver mutations would be worth of further studies. To further investigate the mechanisms involved in the effect of MF plasma on megakaryopoiesis, we targeted the JAK1/2, as well as the NF-κΒ and MAPK pathways, which are involved in inflammation and cell proliferation and have been shown to be hyperactivated in MF15,16. Normalization of cell number by blocking each of these three pathways with specific inhibitors suggests that, as shown for control samples, increased megakaryopoiesis driven by MF plasma relies on these signaling molecules. The participation of NF-κΒ and MAPK in MF pathogenesis has been previously described by Fisher and colleagues, who showed that pharmacological inhibition of these pathways ameliorated inflammatory cytokine production from mononuclear cells from these patients15,16. Altogether, these results suggest NF-κΒ and MAPK pathways could be attractive therapeutic targets to be combined with ruxolitinib15. It has been described that NF-κB and MAPK activation may lead to cellular division through increased expression of cyclin D121,22,23. We found high levels of cyclin D1 expression in MF plasma-stimulated MK, which may support the role of these pathways in cytokine-mediated cell proliferation. However, further studies would be necessary to elucidate the connection of cyclin D1 with this process.
As mentioned above, both MF and ET present a chronic inflammatory state. This state is caused by the increase of numerous inflammatory cytokines, some of which are decreased by treatment with JAK2 inhibitors. Among these cytokines, IL-1β and IL-6 have well-established effects on megakaryopoiesis15. Considering that IL-1β and IL-6 are able to stimulate the NF-κΒ and MAPK signaling pathways, respectively16,17,18, we then focused on the possible role of these inflammatory cytokines on the effects of MF plasma samples on MK proliferation. An increase in circulating levels of both IL-1β and IL-6 was found in our MF cohort. Interestingly, plasmatic IL-1β and IL-6 levels tightly correlated with plasma-induced MK number. Patients with advanced disease displayed higher levels of circulating IL-1β and IL-6 and plasma samples from these patients triggered higher degree of MK output in our in vitro assay, revealing a direct relationship among the presence of inflammatory cytokines in circulation, patients risk status and MK output. Moreover, MF plasma was able to stimulate the growth of MK precursors, which is consistent with the fact that these cytokines act directly on the megakaryocytic lineage. This effect was partially reverted by blocking IL-1β and IL-6 with specific antibodies, further supporting the participation of these soluble mediators on megakaryocytic overgrowth, although the contribution of other inflammatory mediators cannot be ruled out. Interesting a therapeutic approach could rely on targeting inflammatory cytokines. Currently, IL-1β receptor antagonist (Anakinra) and IL-6 blocking antibody (Tocilizumab) are used in the treatment of autoimmune diseases, such as rheumatoid arthritis28,29. In accordance with the pivotal role of IL-1β in MF, preclinical models show that anti-IL-1β antibody ameliorates myelofibrosis in JAK2V617F mice in combination with ruxolitinib30,31. Our results showing that blockade of IL-1β and IL-6 in vitro decreases MK overproduction stimulated by patient plasma, provides additional evidence for the potential benefit of this intervention.
As mentioned, plasma from MF and ET behaved differently regarding their effect on normal megakaryopoiesis and thrombopoiesis, with MF samples stimulating the former process whereas ET plasma did not modify MK output but rather exacerbated PPF. While IL-1β and IL-6 are likely mediators of the increased MF plasma-induced MK proliferation, there is scarce information in the literature regarding the extracellular factors that regulate steady-state proplatelet formation. Machlus et al. have demonstrated the involvement of RANTES in this process showing that this chemokine binds to its receptor in the MK (CCR5) and that its effect is blocked by antagonizing CCR5 with Maraviroc3. Then, RANTES, emerges as an attractive candidate to explain the effect of ET plasma on thrombopoiesis. We found increased levels of this chemokine in ET plasma, that was tightly associated with PPF in culture. Reversion of the increased PPF by the specific antagonist of RANTES receptor, Maraviroc, supported this hypothesis. In addition, the increase in RANTES mRNA levels in normal MK incubated with ET plasma, suggests the contribution of these cells to elevated plasma levels of this chemokine found in patients. Likewise, RANTES has been shown to stimulate the production of proplatelets by inhibiting apoptosis3. We found increased expression of Bcl-xL, an anti-apoptotic molecule of relevance in platelet formation, which could represent a link between high RANTES levels and enhanced thrombopoiesis.
In addition to the regulation of proplatelet formation by RANTES, the hematopoietic transcription factor NFE2 plays a role in the pathophysiology of MPN as most patients have increased NFE2 levels, and elevated NFE2 expression recapitulates MPN hallmarks in vitro and in vivo32,33,34,35. The molecular mechanism causing elevated NFE2 levels in MPN patients remains unclear. Our findings suggest that soluble factors present in ET plasma could, directly or indirectly, stimulate NFE2 expression in MK, which could also contribute to enhanced PPF. Further work would be required to definitively establish the role of NFE2 in this process. Although in contrast to ET, plasma from the overall MF cohort did not significantly enhance thrombopoiesis, higher PPF was observed in a subset of MF patients. This samples also display increased RANTES levels, thus mimicking the behavior of ET plasma. The positive correlation shown between plasma RANTES levels and platelet count in peripheral blood from the entire MPN population further implicates its participation in this process.
Previous studies have shown consistency in the levels of several inflammatory mediators between peripheral blood and bone marrow samples from MPN patients36, suggesting that the observed increase in megakaryopoiesis and proplatelet formation driven by MPN plasma in this study would be likely relevant within the bone marrow microenvironment. Furthermore, it is well-establish that circulating factors are able to reach the bone marrow compartment and regulate multiple aspects of hematopoiesis and platelet production37, further supporting this possibility. Nonetheless, the potential contribution of other local factors in the bone marrow niche cannot be excluded.
Treatment options for MPNs are actively expanding. Ruxolitinib and other JAK2 inhibitors provide substantial clinical benefit for MF patients, although response duration may be limited in a substantial proportion of patients13. Amelioration of constitutional symptoms and spleen reduction induced by Ruxolitinib correlate with a decrease in the levels of inflammatory cytokines. However, recent data and our own studies suggest that regulation of cytokine levels by JAK inhibitors is not enough by itself to fully abort this aberrant inflammatory cytokine production15,20. Keohane et al. showed a significant decrease of cytokines levels after the first month of treatment with ruxolitinib but a further moderate rise after six months of treatment38. This finding highlights that, in addition of JAK2, other deregulated signaling pathways and molecular mediators participate in MPN inflammation. In view of the biologic and clinical importance of cancer-related inflammation, it is reasonable to consider targeting simultaneously clonal hematopoiesis and the accompanying inflammatory status. New agents, alone or in combination with ruxolitinib, are currently under investigation, including the bromodomain and extraterminal domain (BET) inhibitor pelabresib, which downregulates NF-κΒ signaling and has been shown to elicit robust clinical responses39. Our in vitro results using an NF-κΒ inhibitor demonstrate its participation, as well as that of MAPK and JAK1/2 pathways, in exacerbated MK proliferation, reinforcing the potential benefit of combined therapies in the treatment of these patients.
In conclusion, this work highlights the importance of the effect of cytokines in the phenotype of MF and ET, emphasizing the crucial contribution of inflammation to the pathogenesis of these neoplasms. Furthermore, our results reveal that, in addition to intrinsic clonal abnormalities, MPN inflammatory signals differentially contribute to exacerbated megakaryocyte proliferation in MF and PPF in ET, which represent central features in these disorders. Further insight into these mechanisms could contribute to generating the bases for optimizing new therapeutic strategies.
Methods
Patients
Fifty-eight patients with MPN (MF = 30, ET = 28) diagnosed according to the 2016 WHO criteria were included (median age: 56 years, range: 19–89)40. Patient characteristics are summarized in Table 1. Patients were not under cytoreductive treatment. Paired samples from consecutive patients who started Ruxolitinib by indication of the treating physician during the course of this study and had a baseline sample available were analyzed at baseline, before ruxolitinib was started, and at 1 month and 3 months of treatment. Forty healthy individuals were studied as controls (median age: 45 years, range 25–83). The study was approved by the Institutional Ethics Committee and patients and controls signed an informed consent in accordance with the Declaration of Helsinki. Clinical and laboratory parameters were collected by chart review. Driver mutations including JAK2V617F, MPL and CALR, and high molecular risk (HMR) mutations in ASXL1, IDH1/2, SRSF2 and U2AF1 were assessed as described20.
Plasma samples
Blood samples were collected with sodium citrate (3.8%). Plasma was obtained by two centrifugation steps at 3000 rpm for 15 min and recalcified with 25 mmol/l CaCl2 for 2 h at 37 °C to allow the removal of fibrin and prevent clotting during culture incubation.
Purification of CD34 + cells
Umbilical cord blood from normal pregnancies and deliveries was obtained from Hospital Materno-Infantil Dr. Gianantonio, Buenos Aires, Argentina. Low-density mononuclear cells were obtained by Ficoll Hypaque gradient (SIGMA Aldrich). CD34 + cells were purified using a magnetic cell-sorting system (Miltenyi Biotec) as described41.
Evaluation of megakaryopoiesis
CD34 + cells were cultured under megakaryocytic differentiation conditions in StemSpan medium (Stemcell Technologies) supplemented with 25 ng/ml Thrombopoietin (TPO) and 10 ng/ml Interleukin 6 (IL-6) (Miltenyi) with the addition of 10% patients or control plasma. Medium, TPO (10 ng/ml) and plasma of CD34 + cells were refreshed at day 5 and day 9 of culture. After incubation at 37 °C in a humidified atmosphere of 5% CO2 for 12 days, cells were counted and analyzed by flow cytometry (FACSCanto II, BD Biosciences) using anti-human CD61-FITC conjugated and anti-human CD42b-PE conjugated (BD Biosystems) to determinate MK differentiation and maturation. The parameters used for sample acquisition are as follows (voltage): FSC 286; SSC 420; FITC 290 log; PE 300 log; compensation (spectral overlap) FL2/FL1:50, FL1/FL2: 0.10. Post-acquisition data was analyzed using FlowJo software (Turku Centre for Biotechnology, University of Turku, Finland). All samples were processed by triplicate using different cord blood samples.
Expression of transcription factors
RNA was extracted from MK incubated with patient or control plasma using Trizol reagent (Life technologies) and reverse transcribed using SuperScript II Reverse Transcriptase (Thermo Fisher Scientific). cDNA was analyzed by qPCR using SYBR® Green (Life Technologies) in a CFX96 Connect Real-Time PCR Detection System (BioRad). Samples were run in triplicate and relative expression gene was determined by the comparative CT method (ΔΔ Ct) using glyceraldehyde 3-phosphate dehydrogenase (GAPDH) as housekeeping gene.
Forward and reverse primer sequences:
RUNX1: GAGGCAATGGATCCCAGGTA, CTCCCTGAACCACTCCACTG;
GATA-1: CTGGGATCACACTGAGCTTGC, GATTAACCTGGGCTGGTGGTT;
FLI-1: CACCACCCTCTACAACACGGA, TTG GTCGGTGTGGGAGGTT;
Cyclin D1: CAAATGTGTGCAGAAGGAGGT, CTCGCACTTCTGTTCCTCGC;
NFE2a: CCTGCTGTGACTCCACCACA, GCCAGAGTCTGGTCCAGGTTC;
NFE2f: TGACTCTGCCTTTAGCCAGGA, CCAGATGGCTCTAGAAACCTT;
RANTES: TCCTCATTGCTACTGCCCTCT, GGCAATGTAGGCAAAGCAGCA;
Bcl-xL: AAAGGAAACTTGACAGAGGA, GGATCTTTATTTCATGAGGCA;
GAPDH: ATCATCAGCAATGCCTCCTGC, GGGCCATCCACAGTCTTCTG.
Signal transduction pathways on megakaryocyte precursors
CD34 + progenitors were cultured for 6 days under megakaryocytic differentiation conditions without the addition of plasma. Then, the participation of JAK1/2, MAPK and NF-kB pathways was assessed with specific inhibitors: Ruxolitinib (JAK1/2, 1–2 μm, Selleckchem), U0126 (5–10 μm, MAPK, Biomol Gmbh) or RO106-9920 (6–7 μm, NF-kB, Tocris Bioscience) incubated for 90 min prior to the addition of plasma. Cells were counted at day 12 of culture.
Quantification of IL-1β, IL-6, C-reactive protein (CRP) and RANTES
Plasma IL-1β and IL-6 were measured by a Quantikine HS enzyme-linked immunosorbent assay (ELISA, R&D Systems). Plasma C-reactive protein (CRP) concentration was measured by human C-reactive Protein Quantikine ELISA (R&D Systems) as previously described20,42. RANTES levels were measured by ELISA MAX (TM Deluxe Set Human CCL5, Biolegend). In each case, the recommendations of the manufacturer were followed.
Inhibition of IL-1β and IL-6 on megakaryocyte precursors
CD34 + progenitors were cultured for 6 days under megakaryocytic differentiation conditions without the addition of plasma. Plasma IL-1β and IL-6 were blocked by specific antibodies (2.5–100 ug/ml, R&D Systems) 1 h at 37 °C before the addition into the culture (1 × 104cells/well). Cells were counted at day 12 of culture.
Evaluation of MK producing proplatelets
CD34 + progenitors were cultured during 12 days in megakaryocytic differentiation conditions. Then, mature MKs were incubated with the addition of 10% plasma. After 72 h, the total number of proplatelet-bearing MKs (PPF) was counted under an inverted microscope using a 32x objective, and the percentage PPF was calculated on the basis of the viable cell count. All samples were processed in triplicate at least using different cord blood samples. In some experiments, Maraviroc (100 nM, Sigma Aldrich) was added before plasma samples.
Morphological analysis of proplatelets
Mature MKs (day 12 of culture) were seeded on glass coverslips previously coated with 200 µg/ml fibrinogen (SIGMA Aldrich). Cells were incubated for 72 h with 10% plasma from patients or controls. Then, cells were fixed with 1% paraformaldehyde for 20 min at RT, permeabilized with 0.2 Triton X-100 for 6 min, and blocked with PBS containing 10% fetal bovine serum (FBS) (Internegocios, Argentina) and human albumin (1% p/v, Sigma Aldrich). Cells were labelled with anti CD61-FITC in PBS for 1 h and nuclear staining was performed with Hoechst (Sigma Aldrich) for 4 min at RT. Samples were mounted in VECTAShield (Vecta Laboratories) and analyzed under an epifluorescent microscope using a 40x objective (Carl Zeiss, Microimaging Inc.). Microphotographs were obtained using a digital camera (Cannon Power Shot G6, Tokyo, Japan). For proplatelet analysis, the number of swellings and tips on each proplatelet-bearing MK was counted. At least 50 proplatelet-bearing MKs were analyzed.
Statistical analysis
Data were tested for Gaussian distribution. For comparison between two groups, Student-t or Paired-t test; Mann-Whitney test or Wilcoxon matched-pairs signed rank test were used. For comparison among multiple groups One-way ANOVA followed by Tukey´s multiple comparison test, Kruskal–Wallis or Friedman followed by Dunn’s multiple comparison test were used. For correlations, Pearson´s r or Spearman´s r coefficients were used according to the data distribution. Data were presented as mean ± standard deviation or median with interquartile range. P < 0.05 was considered significant. The GraphPad Prism 8.0.1 (La Jolla, CA) software was applied.
Data availability
The raw data supporting the conclusions of this article will be made available by the authors, without undue reservation.
References
Noh, J. Y. Megakaryopoiesis and Platelet Biology: Roles of transcription factors and emerging clinical implications. Int. J. Mol. Sci. 22, 9615. https://doi.org/10.3390/ijms22179615 (2021).
Yu, M. & Cantor, A. B. Megakaryopoiesis and thrombopoiesis: An update on cytokines and lineage surface markers. Methods Mol. Biol. 788, 291–303. https://doi.org/10.1007/978-1-61779-307-3_20 (2012).
Machlus, K. R. et al. CCL5 derived from platelets increases megakaryocyte proplatelet formation. Blood 127, 921–926. https://doi.org/10.1182/blood-2015-05-644583 (2016).
Mascarenhas, J. O., Orazi, A., Bhalla, K. N., Champlin, R. E., Harrison, C. & Hoffman, R. Advances in myelofibrosis: A clinical case approach. Haematologica 98, 1499–1509 (2013).
Ciurea, S. O. et al. Pivotal contributions of megakaryocytes to the biology of idiopathic myelofibrosis. Blood 110, 986–993. https://doi.org/10.1182/blood-2006-12-064626 (2007).
Melo-Cardenas, J., Migliaccio, A. R. & Crispino, J. D. The role of megakaryocytes in myelofibrosis. Hematol. Oncol. Clin. North. Am. 35, 191–203. https://doi.org/10.1016/j.hoc.2020.11.004 (2021).
Balduini, A. et al. In vitro megakaryocyte differentiation and proplatelet formation in Ph-negative classical myeloproliferative neoplasms: Distinct patterns in the different clinical phenotypes. PLoS One 6, e21015 (2011).
Tefferi, A. et al. Circulating Interleukin (IL)-8, IL-2R, IL-12, and IL-15 levels are independently prognostic in primary myelofibrosis: A comprehensive cytokine profiling study. J. Clin. Oncol. 29, 1356e1363 (2011).
Mondet, J., Hussein, K. & Mossuz, P. Circulating cytokine levels as markers of inflammation in Philadelphia negative myeloproliferative neoplasms: Diagnostic and prognostic interest. Mediat. Inflamm. 2015, 670580. https://doi.org/10.1155/2015/670580 (2015).
Fisher, D. A. C., Fowles, J. S., Zhou, A. & Oh, S. T. Inflammatory pathophysiology as a contributor to myeloproliferative neoplasms. Front. Immunol. 12, 683401. https://doi.org/10.3389/fimmu.2021.683401 (2021).
Vannucchi, A. M. et al. Abnormalities of GATA-1 in megakaryocytes from patients with idiopathic myelofibrosis. Am. J. Pathol. 167, 849–858. https://doi.org/10.1016/S0002-9440(10)62056-1 (2005).
Vannucchi, A. M. et al. A pooled analysis of overall survival in COMFORT-I and COMFORT-II, 2 randomized phase 3 trials of ruxolitinib for the treatment of myelofibrosis. Haematologica 100, 1139–1145 (2015).
Verstovsek, S. et al. Long-term survival in patients treated with ruxolitinib for myelofibrosis: COMFORT-I and -II pooled analyses. J. Hematol. Oncol. 10, 156. https://doi.org/10.1186/s13045-017-0527-7 (2017).
Mesa, R. A. Ruxolitinib a selective JAK1 and JAK2 inhibitor for the treatment of myeloproliferative neoplasms and psoriasis. IDrugs 13, 394–403 (2010).
Fisher, D. A. C. et al. Cytokine production in myelofibrosis exhibits differential responsiveness to JAKSTAT, MAP Kinase, and NFkappaB signaling. Leukemia 33, 1978–1995. https://doi.org/10.1038/s41375-019-0379-y (2019).
Fisher, D. A. C. et al. Mass cytometry analysis reveals hyperactive NF kappa B signaling in myelofibrosis and secondary acute myeloid leukemia. Leukemia 31, 1962–1974. https://doi.org/10.1038/leu.2016.377 (2017).
Quesenberry, P. J. et al. Multifactor stimulation of megakaryocytopoiesis: Effects of Interleukin 6. Exp. Hematol. 19, 35–41 (1991). PMID: 1703492.
Beaulieu, L. M. et al. Interleukin 1 receptor 1 and interleukin 1β regulate megakaryocyte maturation, platelet activation, and transcript profile during inflammation in mice and humans. https://doi.org/10.1161/ATVBAHA.113.302700 (2014).
Couldwell, G. & Machlus, K. R. Modulation of megakaryopoiesis and platelet production during inflammation. Thromb. Res. 179, 114–120. https://doi.org/10.1016/j.thromres.2019.05.008 (2019).
De Luca, G. et al. High cell-free DNA is associated with disease progression, inflammasome activation and elevated levels of inflammasome-related cytokine IL-18 in patients with myelofibrosis. Front. Immunol. 16, 14:1161832. https://doi.org/10.3389/fimmu.2023.1161832 (2023).
Balmanno, K. & Cook, S. J. Sustained MAP kinase activation is required for the expression of cyclin D1, p21Cip1 and a subset of AP-1 proteins in CCL39 cells. Oncogene 18, 3085–3097 (1999).
Matsumura, I. et al. Transcriptional regulation of the Cyclin D1 promoter by STAT5: Its involvement in cytokine-dependent growth of hematopoietic cells. EMBO J. 8, 1367–1377 (1999).
Hinz, M. et al. NF-kappaB function in growth control: Regulation of Cyclin D1 expression and G0/G1-to-S-phase transition. Mol. Cell. Biol. 19, 2690–2698. https://doi.org/10.1128/MCB.19.4.2690 (1999).
Debrincat, M. A. et al. BCL-2 is dispensable for thrombopoiesis and platelet survival. Cell. Death Dis. 6, e1721. https://doi.org/10.1038/cddis.2015.97 (2015).
Allegra, A. et al. Synergic crosstalk between Inflammation, oxidative Stress, and genomic alterations in BCR-ABL-negative myeloproliferative neoplasm. Antioxidants 9, 1037. https://doi.org/10.3390/antiox9111037 (2020).
Liu, Z. J. et al. Developmental differences in megakaryocytopoiesis are associated with up-regulated TPO signaling through mTOR and elevated GATA-1 levels in neonatal megakaryocytes. Blood 117, 4106–4117 (2011).
Mattia, G. et al. Different ploidy levels of megakaryocytes generated from peripheral or cord blood CD34 + cells are correlated with different levels of platelet release. Blood 99, 888–897 (2002).
Fleischmann, R. M. et al. Anakinra, a Recombinant human interleukin-1 receptor antagonist (r-metHuIL-1ra), in patients with rheumatoid arthritis: A large, international, multicenter, placebo-controlled trial. Arthritis Rheum. 48, 927–934. https://doi.org/10.1002/art.10870 (2003).
Oldfield, V., Dhillon, S. & Plosker, G. L. Tocilizumab: A review of its use in the management of rheumatoid arthritis. Drugs 69, 609–632. https://doi.org/10.2165/00003495-200969050-00007 (2009).
Rai, S. et al. Inhibition of interleukin-1β reduces myelofibrosis and osteosclerosis in mice with JAK2-V617F driven myeloproliferative neoplasm. Nat. Commun. 13, 5346. https://doi.org/10.1038/s41467-022-32927-4 (2022).
Rahman, M. F. et al. Interleukin-1 contributes to clonal expansion and progression of bone marrow fibrosis in JAK2V617F-induced myeloproliferative neoplasm. Nat. Commun. 13, 5347. https://doi.org/10.1038/s41467-022-32928-3 (2022).
Goerttler, P. S. et al. Gene expression profiling in polycythaemia vera: Overexpression of transcription factor NF-E2. Br. J. Hematol. 129, 138–150 (2005).
Wang, W., Schwemmers, S., Hexner, E. O. & Pahl, H. L. AML1 is overexpressed in patients with myeloproliferative neoplasms and mediates JAK2V617F-independent overexpression of NF-E2. Blood 116, 254–266 (2010).
Kaufmann, K. B. et al. A novel murine model of myeloproliferative disorders generated by overexpression of the transcription factor NF-E2. J. Exp. Med. 209, 35–50 (2012).
Bogeska, R. & Pahl, H. L. Elevated nuclear factor erythroid-2 levels promote epo-independent erythroid maturation and recapitulate the hematopoietic stem cell and common myeloid progenitor expansion observed in polycythemia Vera patients. Stem Cells Transl Med. 2, 112–117 (2013).
Chen, P. et al. Cytokine consistency between bone marrow and peripheral blood in patients with Philadelphia-Negative myeloproliferative neoplasms. Front. Med. (Lausanne). https://doi.org/10.3389/fmed.2021.598182 (2021).
Decker, M., Leslie, J., Liu, Q. & Ding, L. Hepatic thrombopoietin is required for bone marrow hematopoietic stem cell maintenance. Science 360, 106–110 (2018).
Keohane, C. et al. JAK Inhibition induces silencing of T helper cytokine secretion and a profound reduction in T regulatory cells. Br. J. Haematol. 171, 60–73. https://doi.org/10.1111/bjh.13519 (2015).
Rampal, R. K. et al. Pelabresib plus ruxolitinib for JAK inhibitor-naive myelofibrosis: A randomized phase 3 trial. Nat. Med. https://doi.org/10.1038/s41591-025-03572-3 (2025).
Arber, D. A. et al. The 2016 revision to the world health organization classification of myeloid neoplasms and acute leukemia. Blood 127, 2391–2405 (2016).
Balduini, A. et al. Adhesive receptors, extracellularproteins, and myosin-IIa orchestrate proplatelet formation by HumanMK. J. Thromb. Haemost. 6, 1900–1907 (2008).
De Luca, G. et al. Elevated levels of damage-associated molecular patterns HMGB1 and S100A8/A9 coupled with toll-like receptor-triggered monocyte activation are associated with inflammation in patients with myelofibrosis. Front. Immunol. 25, 15:1365015. https://doi.org/10.3389/fimmu.2024.1365015 (2024).
Acknowledgements
The authors are grateful to Fernanda Rubio and Monica Costas, Laboratorio Apoptosis Molecular, IDIM-UBA-CONICET, for collaboration with antibodies; Paola D’atri from IMEX-CONICET for collaboration with inhibitors; Laura Abojer, Head of Departament of Obstetric from the Hospital Materno-Infantil Dr Gianantonio, Lic. Soledad Guerra, nurse supervisor and all the nurse team, for collection of cord blood; and Marina Khoury for statistical advice. The authors wish to thank the Fundación Natalí Dafne Flexer for continuous support.
Funding
This work was supported by the Consejo Nacional de Investigaciones Cientı́ficas y Técnicas (CONICET) Proyectos Unidades Ejecutoras 2017 (grant number: PUE 0055, 2017), Agencia Nacional de Promoción Cientı́fica y Tecnológica (ANPCyT) (grant number: 2021-00449) and the Fondation Nelia et Amadeo Barletta.
Author information
Authors and Affiliations
Contributions
D.Yañuk: Investigation, Methodology, Formal analysis, Writing—review & editing. A.Cellucci: Methodology, Writing—review. R.Marta: Investigation, Methodology, Writing—review & editing. A.Discianni Lupi, G.De Luca, A.Glembotsky: Methodology, Writing—review. M.Castro Rios: Investigation, Writing—review. B.Moiraghi: Investigation, Writing—review. F.Sackmann: Investigation, Writing—review. V.Cortes Guerrieri: Investigation, Writing—review. P.Heller: Conceptualization, Methodology, Funding acquisition, Writing—review & editing. N.Goette: Conceptualization, Investigation, Methodology, Formal analysis, Supervision, Writing—review & editing. P.Lev: Conceptualization, Investigation, Methodology, Formal analysis, Funding acquisition, Supervision, Writing—original draft.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Yañuk, D.B., Cellucci, A.S., Marta, R.F. et al. Inflammatory mediators differentially regulate megakaryopoiesis and thrombopoiesis in myelofibrosis and essential thrombocythemia. Sci Rep 15, 43716 (2025). https://doi.org/10.1038/s41598-025-27594-6
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-27594-6