Abstract
Vonoprazan, a novel and potent acid suppressant, has recently been incorporated into Helicobacter pylori eradication regimens. However, the data on combination of vonoprazan with levofloxacin remains scarce. This study represents one of the first evaluations comparing 7-day and 14-day once-daily regimens containing vonoprazan, levofloxacin, clarithromycin-MR, and bismuth for H. pylori eradication in regions characterized by high clarithromycin and metronidazole resistance. Between March 2022 and December 2023, patients presenting with dyspepsia who underwent upper gastrointestinal endoscopy at a tertiary care hospital in Thailand were recruited. Those testing positive for H. pylori infection were randomly assigned to receive either a 7-day or 14-day once-daily regimen comprising vonoprazan 40 mg, clarithromycin-MR 1 g, levofloxacin 500 mg, and bismuth subsalicylate 1,048 mg. CYP3A4 polymorphism testing and antimicrobial susceptibility assessment (using either the Epsilometer test or the GenoType® HelicoDR assay) were performed. Eradication was confirmed by a negative ¹³C-urea breath test conducted no sooner than four weeks after completing therapy. A total of 312 dyspeptic patients who underwent EGD were screened, and 102 (32.7%) were diagnosed with H. pylori infection. The study enrolled and randomized these patients into two treatment groups: 51 received the 7-day once-daily vonoprazan-based bismuth therapy, and 51 received the 14-day regimen. Baseline characteristics were similar between the two groups, except for a higher prevalence of hypertension in the 7-day regimen group (49% vs. 27.5%, p = 0.025). The eradication rates for the 7-day regimen were 88.2% (45/51) in the intention-to-treat (ITT) analysis and 89.6% (43/48) in the per-protocol (PP) analysis. Similarly, the 14-day regimen achieved eradication rates of 88.2% (45/51) in the ITT analysis and 93.8% (45/48) in the PP analysis. The risk difference for eradication in the PP analysis was 4.17% (95% CI: -6.86 to 15.19, p = 0.71). Adverse events were comparable between the two regimens, occurring in 35.3% (18/51) of patients in the 7-day regimen and 38.8% (19/51) in the 14-day regimen (p = 0.130). The most frequently reported adverse event was black stool (35.3% vs. 38.8%, p = 0.130). Other adverse events included nausea and vomiting (15.7% vs. 22.5%, p = 0.742), bitter taste (17.7% vs. 18.4%, p = 1.000), and dizziness (7.8% vs. 6.1%, p = 0.113). All adverse events resolved spontaneously without requiring medical intervention, and no serious adverse events were reported. The 14-day regimen demonstrated excellent eradication rate (> 90%) regardless of CYP3A4 polymorphisms. These findings suggest that vonoprazan, levofloxacin, clarithromycin-MR, and bismuth for 14 days is a promising alternative first-line treatment option, particularly in regions with high clarithromycin and metronidazole resistance.
Similar content being viewed by others
Introduction
Helicobacter pylori (H. pylori) infection is a key risk factor in gastric carcinogenesis, which is a complex interplay between microbial, host, and environmental factors1,2,3. A recent systematic review and meta-analysis revealed a 15.9% decline in the global prevalence of H. pylori infection among adults over the past three decades4. In Southeast Asia, a similar decreasing trend in prevalence has been observed; however, this is paralleled by a rising trend in antibiotic resistance4,5,6. This downward trend has been accompanied by a corresponding decrease in the incidence of gastric cancer, both globally and in regions where H. pylori prevalence has significantly declined4,7. However, gastric cancer remains one of the most lethal malignancies worldwide, making effective eradication of H. pylori in era of drug resistance a critical area of research8.
This emphasizes how crucial it is to eradicate H. pylori in order to lower the risk of gastric cancer. The success of treatment has been severely hampered by the growing incidence of antibiotic resistance in recent decades, especially to metronidazole and clarithromycin, which has severely reduced the efficacy of eradication regimens9,10. The current American College of Gastroenterology (ACG) guidelines recommend bismuth quadruple therapy (BQT) as the first-line treatment for H. pylori infection in treatment-naïve patients11. While optimized BQT remains the preferred regimen, its complexity poses a significant barrier to compliance. The requirement to take four medications multiple times a day from separate pill bottles presents a significant barrier to patient adherence, which directly impacts eradication rates12.
The ability of H. pylori to survive and proliferate is closely linked to intragastric pH. The bacterium actively replicates in a near-neutral environment (pH 6–7), whereas exposure to acidic conditions (pH 3–6) induces a transition into its coccoid form, which is more resistant to antibiotic treatment13. Consequently, maintaining stable and effective gastric acid suppression is a key determinant of successful H. pylori eradication14. Vonoprazan, a potassium-competitive acid blocker (P-CAB), has demonstrated significant efficacy in managing acid-related disorders, including H. pylori treatment15,16. Its potent and sustained acid suppression has established it as a promising candidate for H. pylori eradication, particularly in combination with amoxicillin for treatment-naïve patients, leading to its recommendation as a suggested regimen in the latest ACG guideline11. However, eradication rates for dual vonoprazan-amoxicillin remain suboptimal in Southeast Asia, which may be attributed to the high prevalence of CYP3A4 extensive metabolizers and a greater proportion of individuals with CYP3A5 *1/*1 and *1/*3 genotypes17.
We hypothesized that integrating bismuth subsalicylate with vonoprazan in a once-daily quadruple therapy regimen could enhance eradication rates. This pioneer study aims to evaluate the efficacy of 7-day and 14-day once-daily vonoprazan, levofloxacin, clarithromycin-MR, and bismuth for H. pylori eradication in regions with high clarithromycin and metronidazole resistance.
Materials and methods
Study design and participants
This prospective, single-center, open-label randomized trial aimed to evaluate the efficacy and tolerability of once-daily vonoprazan-based bismuth therapy as a first-line treatment for H. pylori eradication in patients with non-ulcer dyspepsia. The study was conducted over a 14-month enrollment period at a tertiary care center in Thailand between March 3, 2022, and December 31, 2023. Eligible participants were adults aged 18 to 75 years who underwent esophagogastroduodenoscopy (EGD) at the Gastroenterology Unit, Faculty of Medicine, Thammasat University Hospital. Participants were required to have a diagnosis of non-ulcer dyspepsia and confirmed H. pylori infection, with symptoms warranting EGD based on the criteria established by the Gastroenterological Association of Thailand18. All enrolled patients received the study regimen as first-line treatment. Written informed consent was obtained from all participants prior to enrollment.
Patients were excluded if they had active upper gastrointestinal bleeding at the time of enrollment, had received antibiotics or other medications affecting H. pylori within one month before the study, or had contraindications to gastric biopsy such as coagulopathy or bleeding disorders. Individuals with a history of hypersensitivity to any study medications or prior H. pylori eradication therapy were also excluded. Furthermore, patients with severe comorbidities, including end-stage renal disease, end-stage liver cirrhosis, immunodeficiency disorders, advanced malignancy, severe cerebrovascular disease with functional dependence, QTc prolongation, significant cardiac arrhythmias, or those on anticoagulants such as warfarin or direct oral anticoagulants (DOACs), were not eligible to participate. Additional exclusion criteria included a history of gastric surgery, refusal to participate, and concurrent use of medications known to have significant drug interactions with study medications. Participants retained the right to withdraw from the study at any time without compromising their standard medical care.
This study was approved by The Human Research Ethics Committee of Thammasat University with approval number MTU-EC-IM-6–313/64 and was conducted according to good clinical practice guidelines and the Declaration of Helsinki. Written informed consent was obtained before enrollment. There were no changes in the protocol after trial commencement. This study has a data monitoring committee. This trial is registered with Thaiclinicaltrials.org, TCTR20240809001. Date of first registration: 01/11/2022.
Funding declaration
This study was supported by the Thailand Science Research and Innovation Fundamental Fund 2025, the Gastroenterological Association of Thailand (GAT), and the Center of Excellence in Digestive Diseases, Thammasat University.
Treatment regimens
The once daily vonoprazan-based bismuth therapy regimen comprised of once-daily administration of vonoprazan (40 mg), bismuth subsalicylate (1024 mg), clarithromycin MR (1 g), and levofloxacin (500 mg) for both the 7-day and 14-day treatment durations. To ensure that all medications could be administered once daily, clarithromycin was used in its modified-release (MR) formulation. The MR formulation is distinct from the extended-release version and has previously been evaluated in clinical trials related to H. pylori eradication17. In addition, previous clinical trial have demonstrated that vonoprazan 20 mg once daily provides a more potent and sustained acid-inhibitory effect compared with 2–4 times the standard daily dose of rabeprazole. In this study, however, we employed vonoprazan 40 mg once daily to further substantiate acid suppression19.
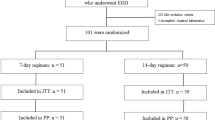
All medications were taken after meals. The efficacy of H. pylori eradication was assessed four weeks post-treatment using the ¹³C-urea breath test (UBT). To ensure adherence, patients were systematically questioned about their medication intake during scheduled follow-up visits, with nonadherence defined as consumption of less than 90% of the prescribed doses. Treatment-related adverse events were characterized as any occurrence resulting in death, hospitalization, disability, congenital anomaly, or requiring medical intervention during the treatment period. Serious adverse events were further categorized as any adverse effect significantly impairing the patient’s daily activities or necessitating hospitalization. Additionally, all adverse events were documented and monitored during follow-up visits at the outpatient clinic, with structured physician-led assessments. The study’s design and CONSORT diagram are illustrated in Fig. 1.
CONSORT flow diagram of this study.
Outcomes
This study aimed to evaluate and compare the efficacy and tolerability of 7-day versus 14-day once-daily vonoprazan-based bismuth therapy as a first-line treatment for H. pylori eradication in patients with non-ulcer dyspepsia. The primary outcome was the eradication rate of H. pylori between the two treatment regimens, assessed using the ¹³C-urea breath test at four weeks post-treatment. Eradication rates were analyzed using both intention-to-treat (ITT) and per-protocol (PP) approaches to provide a comprehensive evaluation of treatment efficacy. Secondary outcomes included an assessment of treatment-related adverse events, as well as the role of CYP3A4 genetic polymorphisms in influencing H. pylori eradication outcomes. Additionally, the study examined the effectiveness of vonoprazan-based bismuth therapy in patients infected with clarithromycin-resistant, metronidazole-resistant, and dual-resistant H. pylori strains. All secondary outcomes were analyzed using ITT analysis among participants with available data.
Diagnosis of H. pylori infection and CYP3A4 genetic polymorphisms
Esophagogastroduodenoscopy (EGD) was performed using an Olympus gastroscope (GIF-HQ190) with dual focus and the EVIS EXERA III system. Gastric biopsy samples were obtained from both the antrum and corpus in accordance with the Sydney protocol. These samples were utilized for rapid urease testing (RUT, Prontodyne®), histopathological examination, H. pylori culture, and antibiotic susceptibility testing using either the Epsilometer test (E-test) or the GenoType® HelicoDR assay. H. pylori infection was defined by a positive bacterial culture or by concordant positive results from both RUT and histology. Additionally, 3 mL of venous blood was collected from each participant for DNA extraction to analyze CYP3A4 genetic polymorphisms. Genotyping was performed using the polymerase chain reaction-restriction fragment length polymorphism (PCR-RFLP) technique with reagents from New England Biolabs (NEB), USA. CYP3A4 genotypes *1/*1 and *1/*1B were categorized as rapid metabolizers, whereas *1B/*1B was classified as an ultrarapid metabolizer.
Randomization and masking
Participants were randomized into one of two treatment groups using a web-based randomization system. The allocation process employed computer-generated random sequences as illustrated in Fig. 1. However, concealment of the randomization sequence was not performed. Informed consent was also obtained for individual patients.
Statistical analysis
A prior study reported an expected H. pylori eradication rate of 76% for a 7-day bismuth-containing quadruple therapy regimen and 96% for a 14-day regimen20. Based on these data, a sample size of 100 patients (50 per group) was calculated, assuming an 80% power, a two-sided alpha of 0.05, and a 10% anticipated loss to follow-up. For statistical analysis, demographic characteristics, eradication rates, and adverse event frequencies were assessed using Student’s t-test or the chi-square test, depending on data distribution. H. pylori eradication rates were evaluated through both intention-to-treat (ITT) and per-protocol (PP) analyses. Statistical significance was set at p < 0.05, and all statistical analyses were performed using SPSS version 28.0 (IBM, Armonk, NY, USA).
Results
Baseline characteristics
Between January 1, 2022, and March 31, 2023, a total of 312 dyspeptic patients who underwent esophagogastroduodenoscopy (EGD) were screened for eligibility. Among them, 102 patients (32.7%) were diagnosed with H. pylori infection, comprising 41 men and 61 women, with a mean age of 57.2 ± 11.6 years. All H. pylori-positive patients were subsequently enrolled in the study and randomized into two treatment groups: 51 patients received a 7-day once-daily vonoprazan-based bismuth therapy, while another 51 patients underwent the 14-day once-daily vonoprazan-based bismuth therapy. Baseline demographic characteristics, including sex distribution, history of smoking, and alcohol consumption, did not show any statistically significant differences between the two groups except for hypertension that participant in 7-day regimen had higher proportion of participants with hypertension (49% vs. 27.5%, p-value = 0.025). Six patients were excluded from the final analysis: four patients (two from each treatment arm) were lost to follow-up, while two patients (one from each arm) discontinued treatment due to minor adverse effects, such as nausea and bitter taste. A summary of the baseline demographic data is demonstrated in Table 1.
Outcomes
The eradication rates for the 7-day vonoprazan-based bismuth therapy regimen were 88.2% (45/51) in the intention-to-treat (ITT) analysis and 89.6% (43/48) in the per-protocol (PP) analysis. Similarly, the 14-day regimen achieved eradication rates of 88.2% (45/51) in the ITT analysis and 93.8% (45/48) in the PP analysis. The risk difference for eradication in the PP analysis was 4.17% (95% CI: −6.86 to 15.19, p = 0.71), as demonstrated in Fig. 2.
Eradication rates according to the treatment regimen.
Antibiotic susceptibility testing was performed on eight patients, with resistance detected for clarithromycin in 25% (2/8), metronidazole in 50% (4/8), and levofloxacin in 75% (6/8) of cases. No resistance was observed for amoxicillin or tetracycline. Among patients receiving the 7-day vonoprazan-based bismuth therapy regimen, one exhibited clarithromycin resistance, three were resistant to metronidazole, and one had levofloxacin resistance. Despite this, all patients in the 7-day regimen achieved successful eradication. In contrast, treatment failure was observed in three patients receiving the 14-day regimen. Among these, one patient exhibited resistance to clarithromycin, metronidazole, and levofloxacin, another had metronidazole resistance but demonstrated poor adherence, and the third had levofloxacin resistance. In both treatment arms, one patient was identified with multidrug resistance to clarithromycin, metronidazole, and levofloxacin.
The overall eradication rates were 100% (3/3) for the 7-day regimen and 40% (2/5) for the 14-day regimen. Regarding the CYP3A4 genetic polymorphisms, the CYP3A4 genotype accounted for 100% of the rapid metabolizers.
The incidence of adverse events was comparable between the two treatment regimens, occurring in 35.3% (18/51) of patients in the 7-day regimen and 38.8% (19/51) in the 14-day regimen (p = 0.130). The most frequently reported adverse event in both groups was black stool, observed in 35.3% vs. 38.8% of patients in the 7-day vs. 14-day regimens, respectively (p = 0.130). Other commonly reported adverse events included nausea and vomiting (15.7% vs. 22.5%, p = 0.742), bitter taste (17.7% vs. 18.4%, p = 1.000), and dizziness (7.8% vs. 6.1%, p = 0.113).
All adverse events resolved spontaneously without the need for medical intervention. No serious adverse events were reported throughout the study period. Details of adverse events are summarized in Table 2.
Discussion
Successful eradication of H. pylori is influenced by multiple factors, including antibiotic resistance, gastric acid suppression, and patient adherence to treatment. Globally, antibiotic resistance has been rising, particularly against metronidazole and clarithromycin, both of which are key components in first-line H. pylori treatment according to various international guidelines. This growing resistance has significantly reduced the efficacy of conventional triple therapy in several regions, including Thailand, highlighting the urgent need for an alternative eradication regimen6,10,11,21. Vonoprazan, a potassium-competitive acid blocker (P-CAB), a novel class of acid-suppressing agents with superior potency in increasing intragastric pH compared to proton pump inhibitors (PPIs)15. Despite its promising pharmacological profile, our previous study demonstrated that both 14-day vonoprazan-based dual therapy and 14-day vonoprazan-based triple therapy failed to achieve satisfactory eradication rates17. However, another study showed that a 14-day vonoprazan-based bismuth quadruple therapy regimen yielded excellent eradication rates. Nonetheless, the regimen’s complexity—requiring twice-daily vonoprazan and four-times-daily tetracycline posed adherence challenges22. To address this, we developed a simplified once-daily vonoprazan-based bismuth therapy regimen aimed at improving compliance while maintaining high eradication efficacy.
Our study demonstrated that 14-day once-daily vonoprazan-based bismuth therapy regimens achieved excellent eradication rates. These findings are comparable to our previous study, which utilized a 14-day vonoprazan-based quadruple regimen, and align with results from a similar study conducted in China23,24. However, the regimen used in the Chinese study consisted of vonoprazan 20 mg once daily, combined with amoxicillin 1000 mg, furazolidone 100 mg, and colloidal bismuth 200 mg, each administered twice daily24. While this regimen achieved high eradication rates, its complexity and the limited availability of furazolidone restrict its widespread clinical application. A previous meta-analysis demonstrated that in patients with clarithromycin-resistant H. pylori infections, vonoprazan-based therapies achieved an eradication rate of 81% (76%–85%), significantly outperforming PPI-based regimens (76% vs. 40%; RD = 0.33, 95% CI = 0.24–0.43)24. Consistent with these findings, our study reported treatment failure in only three patients receiving the 14-day vonoprazan-based bismuth therapy regimen. Among them, one patient exhibited resistance to clarithromycin, metronidazole, and levofloxacin, another had metronidazole resistance coupled with poor adherence, and the third had levofloxacin resistance. In contrast, all patients in the 7-day regimen group successfully achieved H. pylori eradication. These results suggest that a once-daily vonoprazan-based regimen may offer a promising strategy to overcome the challenge of rising clarithromycin resistance.
Current standard guidelines recommend PCAB-clarithromycin triple therapy over PPI-clarithromycin triple therapy for treatment-naïve H. pylori-infected patients with unknown clarithromycin susceptibility21. Given the increasing prevalence of clarithromycin resistance, our study supports the potential role of vonoprazan-based regimens as an effective alternative in high-resistance regions.
Furthermore, genetic testing for CYP3A4 polymorphisms in our study demonstrated that 100% of participants were classified as rapid metabolizers. Despite this, the once-daily vonoprazan-based regimen remained highly effective, further supporting its clinical utility regardless of CYP3A4 metabolism status.
Our study had few limitations. As a single-center, open-label clinical trial, the potential for bias was unavoidable. To strengthen the validity of these findings, further multicenter, double-blind trials are warranted. Furthermore, limited availability of clinical data on the use of potassium-competitive acid blockers (P-CABs) in Europe. While vonoprazan and other P-CABs have been extensively investigated and widely used across several Asian countries17,22,23,24, data from Western populations remain scarce. However, emerging studies from the United States are beginning to expand the evidence base16, reflecting growing clinical interest and experience with this newer class of acid suppressants.
Additionally, incorporating a comparator group receiving standard bismuth-based quadruple therapy would provide valuable comparative data for future research. Another limitation was the limited number of patients with positive antibiotic susceptibility testing. The H. pylori isolation rate varies significantly ranging from 2.1% to 76.4% due to multiple factors and methodological differences across laboratories, which may have influenced the generalizability of our findings25,26. Consequently, the number of cases in antibiotic resistance and CYP3A4/5 genotype information was limited, reducing the statistical power of subgroup analyses. Nonetheless, we consider these data meaningful, as they provide preliminary insights and may serve as a foundation for future studies with larger sample sizes. Despite these limitations, our study had several notable strengths. It was the first randomized controlled trial to evaluate a simplified once-daily vonoprazan-based bismuth quadruple therapy as a first-line treatment for H. pylori eradication. Furthermore, since this study was conducted in a country with high clarithromycin and metronidazole resistance rates, the findings are especially relevant to regions facing similar antibiotic resistance challenges. Notably, our study explored the potential influence of CYP3A4 polymorphisms on eradication rate, providing valuable insights into the roles of genetics polymorphism and treatment response. Future studies should be designed to compare 10-day and 14-day regimens in a non-inferiority manner to better reflect current international recommendations.
In conclusion, this study demonstrated that 14-day once-daily vonoprazan-based levofloxacin bismuth therapy achieved high eradication rates for H. pylori, without major adverse event. Furthermore, the 14-day regimen had achieved excellent eradication rate (> 90%) regardless of CYP3A4 polymorphisms and clarithromycin resistance. Given the rising prevalence of antibiotic-resistant H. pylori strains, these findings highlight the potential of vonoprazan-based bismuth therapy as a promising alternative first-line eradication strategy.
Data availability
The deidentified datasets collected and analyzed during this study are available from the corresponding authors upon reasonable request.
References
Amieva, M. & Peek, R. M. Jr. Pathobiology of Helicobacter pylori-Induced gastric cancer. Gastroenterology 150, 64–78. https://doi.org/10.1053/j.gastro.2015.09.004 (2016).
Nabavi-Rad, A. et al. The interaction between autophagy, Helicobacter pylori, and gut microbiota in gastric carcinogenesis. Trends Microbiol. 31, 1024–1043. https://doi.org/10.1016/j.tim.2023.04.001 (2023).
Ahn, H. J. & Lee, D. S. Helicobacter pylori in gastric carcinogenesis. World J. Gastrointest. Oncol. 7, 455–465. https://doi.org/10.4251/wjgo.v7.i12.455 (2015).
Chen, Y. C. et al. Global Prevalence of Helicobacter pylori Infection and Incidence of Gastric Cancer Between 1980 and 2022. Gastroenterology 166, 605–619 https://doi.org/10.1053/j.gastro.2023.12.022 (2024).
Li, Y. et al. Global prevalence of Helicobacter pylori infection between 1980 and 2022: a systematic review and meta-analysis. Lancet Gastroenterol. Hepatol. 8, 553–564. https://doi.org/10.1016/s2468-1253(23)00070-5 (2023).
Bongkotvirawan, P. et al. Sa1375 Epidemiology and antibiotic resistance pattern of helicobacter pylori infection in south east asia countries: a systematic review with meta-analysis. Gastroenterology 166 (S-435). https://doi.org/10.1016/S0016-5085(24)01477-X (2024).
Shirani, M. et al. The global prevalence of gastric cancer in Helicobacter pylori-infected individuals: a systematic review and meta-analysis. BMC Infect. Dis. 23, 543. https://doi.org/10.1186/s12879-023-08504-5 (2023).
Bray, F. et al. Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 74, 229–263. https://doi.org/10.3322/caac.21834 (2024).
Bujanda, L. et al. Effectiveness of Helicobacter pylori treatments according to antibiotic resistance. Am. J. Gastroenterol. 119, 646–654. https://doi.org/10.14309/ajg.0000000000002600 (2024).
Hong, T. C. et al. Primary antibiotic resistance of Helicobacter pylori in the Asia-Pacific region between 1990 and 2022: an updated systematic review and meta-analysis. Lancet Gastroenterol. Hepatol. 9, 56–67. https://doi.org/10.1016/s2468-1253(23)00281-9 (2024).
Chey, W. D. et al. ACG clinical guideline: treatment of Helicobacter pylori infection. Am. J. Gastroenterol. 119, 1730–1753. https://doi.org/10.14309/ajg.0000000000002968 (2024).
Huguet, J. M. et al. Role of compliance in Helicobacter pylori eradication treatment: results of the European registry on H. pylori management. United Eur. Gastroenterol. J. 12, 691–704. https://doi.org/10.1002/ueg2.12569 (2024).
Ierardi, E. et al. Optimizing proton pump inhibitors in Helicobacter pylori treatment: old and new tricks to improve effectiveness. World J. Gastroenterol. 25, 5097–5104. https://doi.org/10.3748/wjg.v25.i34.5097 (2019).
Ke, H. et al. The appropriate cutoff gastric pH value for Helicobacter pylori eradication with bismuth-based quadruple therapy. Helicobacter 26 (e12768). https://doi.org/10.1111/hel.12768 (2021).
Patel, A., Laine, L., Moayyedi, P. & Wu, J. A. G. A. Clinical practice update on integrating Potassium-Competitive acid blockers into clinical practice: expert review. Gastroenterology 167, 1228–1238. https://doi.org/10.1053/j.gastro.2024.06.038 (2024).
Chey, W. D. et al. Vonoprazan triple and dual therapy for Helicobacter pylori infection in the united States and europe: randomized clinical trial. Gastroenterology 163, 608–619. https://doi.org/10.1053/j.gastro.2022.05.055 (2022).
Ratana-Amornpin, S. et al. Pilot studies of vonoprazan-containing Helicobacter pylori eradication therapy suggest Thailand May be more similar to the US than Japan. Helicobacter 28, e13019. https://doi.org/10.1111/hel.13019 (2023).
Pittayanon, R. et al. Thail. Dyspepsia Guidelines: 2018 J Neurogastroenterol Motil 25, 15–26 https://doi.org/10.5056/jnm18081 (2019).
Takeuchi, T. et al. Randomised trial of acid Inhibition by Vonoprazan 10/20 mg once daily vs Rabeprazole 10/20 mg twice daily in healthy Japanese volunteers (SAMURAI pH study). Aliment. Pharmacol. Ther. 51, 534–543. https://doi.org/10.1111/apt.15641 (2020).
Gan, H. Y. et al. Efficacy of two different dosages of Levofloxacin in curing Helicobacter pylori infection: A Prospective, Single-Center, randomized clinical trial. Sci. Rep. 8, 9045. https://doi.org/10.1038/s41598-018-27482-2 (2018).
Malfertheiner, P. et al. Management of Helicobacter pylori infection: the Maastricht VI/Florence consensus report. Gut https://doi.org/10.1136/gutjnl-2022-327745 (2022).
Tungtrongchitr, N. et al. Fourteen-day vonoprazan-based bismuth quadruple therapy for H. pylori eradication in an area with high clarithromycin and Levofloxacin resistance: a prospective randomized study (VQ-HP trial). Sci. Rep. 14, 8986. https://doi.org/10.1038/s41598-024-59621-3 (2024).
Lu, F., Xu, W., Shi, X., Yu, H. & Qi, X. 14-Day Vonoprazan-Based bismuth quadruple therapy for Treatment-Naïve patients with Helicobacter pylori infection: A retrospective comparative study. Int. J. Gen. Med. 16, 4279–4281. https://doi.org/10.2147/ijgm.S427450 (2023).
Lu, L. et al. Quadruple therapy with Vonoprazan 20 mg daily as a first-line treatment for Helicobacter pylori infection: A single-center, open-label, noninferiority, randomized controlled trial. Helicobacter 28, e12940. https://doi.org/10.1111/hel.12940 (2023).
Yin, Y., He, L. H. & Zhang, J. Z. Successful isolation of Helicobacter pylori after prolonged incubation from a patient with failed eradication therapy. World J. Gastroenterol. 15, 1528–1529. https://doi.org/10.3748/wjg.15.1528 (2009).
Sukri, A., Lopes, B. S. & Hanafiah, A. The emergence of Multidrug-Resistant Helicobacter pylori in Southeast asia: A systematic review on the trends and intervention strategies using antimicrobial peptides. Antibiot. (Basel). 10 https://doi.org/10.3390/antibiotics10091061 (2021).
Acknowledgements
This study was supported by the Thailand Science Research and Innovation Fundamental Fund 2025, the Gastroenterological Association of Thailand (GAT), and the Center of Excellence in Digestive Diseases, Thammasat University.
Author information
Authors and Affiliations
Contributions
Conceptualization, S.S., P.B. and R.K.V.; methodology, S.S. and R.K.V.; software, P.B.; validation, P.B., S.C., B.P. S.Si., P.Bh., N.I, P.G., A.W., N.A., V.V., P.S., V.M., M.M., Y.Y., P.K. and R.K.V.; formal analysis, P.B. and S.S.; investigation, S.S.; data curation, S.S, writing—original draft, S.S.; writing—review and editing, P.B. and R.K.V.; supervision, R.K.V.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Sukkamolsantiporn, S., Bongkotvirawan, P., Chonprasertsuk, S. et al. Efficacy of fourteen-day once-daily vonoprazan-based quadruple therapy for Helicobacter pylori in high clarithromycin-resistance regions (ONCE-VONO Trial). Sci Rep 15, 44105 (2025). https://doi.org/10.1038/s41598-025-27876-z
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-27876-z