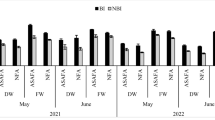Abstract
Drought is a major abiotic stress limiting global maize productivity. In this study, we evaluated the combined application of biochar (BC) and sodium hydrosulfide (NaHS) as a potential strategy for enhancing drought resilience in maize (cv. Gohar-19) under greenhouse conditions. The experiment comprised of two factors i.e., drought stress including S1 (85% ± 5% field capacity), S2 (55% ± 5% field capacity) and S3 (35% ± 5% Field capacity) and various treatments of BC and NaHS i.e., To (control), T1 (3% biochar), T2 (0.05 mmol L− 1 NaHS), T3 (0.1 mmol L− 1 NaHS), T4 (T1 + T2) and T5 (T1 + T3). Results revealed that drought stress significantly reduced growth traits, photosynthetic pigments, photosynthetic rate, stomatal conductance, and leaf water content, while increasing oxidative damage, osmolyte accumulation, and lipid peroxidation. By contrast, the integrated treatment of biochar and NaHS (0.01 mmol L⁻¹) significantly alleviated these negative effects, improving biomass and growth by ~ 20%, increasing photosynthetic performance by more than 50%, and enhancing leaf water content by 38%. Furthermore, this combined treatment reduced stress-related biochemical markers, including superoxide dismutase, peroxidase, malondialdehyde, and hydrogen peroxide, by over 30%, and led to a significant decline in osmolyte and secondary metabolite accumulation. Correlation and principal component analyses confirmed strong associations among growth, physiological, and biochemical parameters, highlighting the synergistic protective role of biochar + NaHS treatment. These findings provide experimental evidence that this integrative approach can improve maize drought tolerance and productivity in maize, supporting its potential application in sustainable agriculture. Field-scale trials and molecular investigations are warranted to validate these findings and elucidate underlying mechanisms of this stress-mitigation effect.
Similar content being viewed by others
Introduction
Drought is considered a major threat to reduce agriculture productivity1 and globally, crop production is severely threatened by drought stress in the context of climate change2,3, through impacting the development and growth of the plant4. Drought stress inhibits photosynthetic activity in plants (by lowering CO2 uptake and the activity of the enzyme ribulose-1,5-bisphosphate carboxylase-oxygenase (RUBISCO)5. It also decreases the chlorophyll contents6, hinders cell division and elongation, decreases the rate of photosynthesis, relative water content, and threw off the equilibrium between antioxidants and reactive oxygen species (ROS). Stress from drought causes an overabundance of ROS, which harms proteins, cell membranes, and nucleic acids7. For maize crop, if plant has to suffer drought stress during the grain filling stage, it can reduce grain yield by 79–81%8. Climate change and hence drought stress will be more severe in future1. Therefor developing climate resilient crops is necessary to meet global food security.
Maize (Zea mays L.) is a third most important crop after wheat and rice that can be grown in a variety of agro-climatic situations9. Farmers prefer this crop because it has the highest grain production potential among all cereals. The crop can be used for grains, fodder, and industrial raw material10. During 2024, It was cultivated on 197 Mha and provided 2853 million tons of food11. In Pakistan, maize is among most important cash crops12. During 2024, Pakistan harvested 1.3 million hectares of maize with estimated production of 11 million tons9.
Application of H2S donors external to the plant, such as sodium hydrosulfide (NaHS), has beneficial impacts on plant respond to different environmental stresses13. Abiotic stress relief mediated by H2S is primarily responsible for mitigating oxidative damage by activating antioxidant enzymatic and non-enzymatic mechanisms. The primary enzyme responsible for plants’ natural production of H2S is known as L-cysteine de-sulfhydrase (LCD), which breaks down cysteine into H2S, pyruvate and ammonium. Researches have proven that H2S is essential to many biological functions that occur throughout plant life, such as stress reactions, root growth, development and seed germination14. Reducing drought effects on plant development through application of biochar (BC) is also highly effective strategy. Biochar, developed via pyrolysis process of organic materials, is distinguished for its higher carbon content and porosity. It serves as a soil conditioner that promotes soil fertility by enhancing water retention, diminishing runoff, and facilitating more effective water absorption by plants during drought conditions15. Due to its alkaline nature and high cation exchange capacity, it is a crucial supplement for the reclamation of soils damaged by salinity. By enhancing soil physiochemical, biological, and chemical processes as well as Na leaching, the application of BC lessened the negative effects of drought stress16. Application of this technique is becoming a popular way to improve agricultural yields, especially in soils that are stressed by environmental factors like salt, drought, or heavy metal contamination.
Many strategies have been put forth in recent years to minimize the negative impacts of drought, including the use of osmo-protectants (synthetic substances), conservation tillage techniques and the development of drought-tolerant varieties. A study was conducted on the genetic dissection of maize to enhance drought tolerance and support trait improvement17. Researchers utilized synthetic bacterial communities derived from xerophytes to strengthen maize resilience under drought stress1. Gene editing techniques are actively used to enhance drought tolerance in maize for developing resilient, high-yielding varieties18. A previous study investigated the effects of drought on maize and discussed a range of management techniques to alleviate its impact19. A previous report highlighted the role of potassium in enhancing drought tolerance in maize20. A study addressed the improvement of drought tolerance in maize through foliar application of zinc and phosphorus, which contribute to enhanced nutrient uptake and stress resistance21. A study demonstrated that the application of biochar mitigated drought stress in maize by improving soil structure, enhancing water retention, and increasing nutrient availability22. Although several approaches such as breeding drought-tolerant varieties, adopting conservation tillage, applying nutrient management, and utilizing gene editing have been developed to enhance maize resilience, the combined application of sodium hydrosulfide (NaHS) and biochar remains largely unexplored. Most existing studies have evaluated these treatments separately, leaving an important gap in understanding their possible synergistic effects. Current study is focused on the combined application of NaHS with biochar to mitigate the adverse effects of drought stress on various traits of maize. The current study aims to: (1) assess the impact of drought stress on maize growth, physio-biochemical properties and quality traits; and (2) evaluate the efficacy of various concentrations of NaHS and biochar in mitigating drought stress in maize.
Materials and methods
A pot experiment was conducted under greenhouse during spring season (April to July, 2024) at research farms of the University of Layyah, Punjab Pakistan. The region is characterized by a hot arid climate, with average maximum summer temperatures approaching 49 °C and average minimum winter temperatures dropping to around 8 °C. Commonly grown local variety Gohar-19 was selected for experimentation. The maize seeds were sown initially in nursery trays and were transplanted to experimental pots after two weeks. To ensure plant availability at each pot, two seedlings per pot were ensured. The pots were filled with sandy loam soil. Each kilogram of soil contained 1.97 g of total nitrogen, 3.38 g of total phosphorous, 13.02 g of total potassium, 93.26 mg alkaline hydro nitrogen contents, 32.14 mg available phosphorous, 51.38 mg available potassium, 13.79 g organic matter and pH = 6.7. The average temperature of greenhouse was in the range of 25–33 °C.
Experimental treatments
The maize plants were treated with three levels of drought stress including S1 (85% ± 5% Field capacity), S2 (55% ± 5% Field capacity) and S3 (35% ± 5% Field capacity). The chemical application included To (control), T1 (3% biochar mixed with pot soil), T2 (0.05 mmol L− 1 NaHS application), T3 (0.1 mmol L− 1 NaHS application), T4 (3% biochar mixed with pot soil + 0.05 mmol L− 1 NaHS application) and T5 (biochar mixed with pot soil + 0.1 mmol L− 1 NaHS application). After 45 days of plantation, drought condition was maintained. Moisture meter was used to ensure drought level continuously. There were three replications for each treatment and each replication has 10 pots for precise data collection. Following factors were selected and determined during experimentation to compare the effect of selected treatments.
Growth and biomass attributes
Plants in random were selected and harvested from experimental pots. The cobs were separated and threshed for grains and whole stalk material was used to record plant height (cm), ear length (cm) and ear diameter (cm), following the methods of20,23.
Physiological/photosynthetic attributes
The leaf chlorophyll fluorescence was recorded before start of pollination24. Hand held chlorophyll fluorimeter was used to find plant leaf fluorescence between 11:30 and 14:00 h. Three leaves of each plant, selected from all study treatments, were covered using clips. The fluorimeter was placed on clip after 20 min of darkness and maximum yield of PSII photochemistry \(\left( {{\raise0.7ex\hbox{${F_{v} }$} \!\mathord{\left/ {\vphantom {{F_{v} } {F_{m} }}}\right.\kern-\nulldelimiterspace} \!\lower0.7ex\hbox{${F_{m} }$}}} \right)\) was recorded.
Chlorophyll and carotenoids were estimated from 18-day-old maize leaves using 80% acetone extracts, with absorbance measured at 663, 645 and 470 nm wavelength as recommended by25. Chlorophyll and carotenoid contents were calculated using Eqs. (1, 2, 3 and 4).
Here, V was volume of purified solution, W was fresh weight, and A663, A645 and A470 were optical absorption wavelengths.
Photosynthetic rates (Pn), stomatal conductance (gs), and intercellular CO2 concentration (Ci) of the maize leaves were recorded using portable photosynthesis system between 10:00 and 11:30 on a clear day. The leaf chamber was set at PAR of 1600 mol m− 2 s− 1), leaf temperature of 25 °C and relative humidity was maintained at 70% as per described method of26.
Water related attribute
Relative water content (RWC) in leaf is an important measure to describe drought stress condition. RWC was calculated using eighteen day old fresh tissue from third leaf. The third leaf from each treatment was cut and immediately placed in sealed plastic bag to avoid any environmental effect. The fresh weight was recorded immediately after leaf transfer to lab. The harvested leaves were soaked in distilled water for 6 to 8 h at ambient temperature and carefully dried with tissue paper. Dry weight was obtained after oven drying at 72 °C for 24 h using weight balance. The RWC was calculated using following Eq. (5)27.
Enzymatic antioxidants activity
The SOD and POD activities were determined utilizing established methodologies documented by28. For POD, 0.5 ml enzyme extract was mixed with buffer substrate (guaiacol and Na3PO4; pH 6.4) and hydrogen peroxide (24 mM). The absorbance was measured twice at one-minute intervals. The polyphenol oxidase activity was determined by following the methods reported by29. The PAL was determined using modified methods suggested by30.
The phenolic contents were calculated using methods described by30. The enzymatic activity of catalase (CAT) was determined following the standard methods given by31. Few modifications in these methods were made as suggested by32. After enzymatic addition to start the reaction, the absorbance at 240 nm was recorded for 2 min with an interval of 20 s.
APX was determined using standard methods of33. The reaction mixture was prepared using enzyme extract (0.1 ml), phosphate buffer (pH 7.0; 50 mM), L-ascorbic acid (0.25 mM), and hydrogen peroxide (1 mM). A spectrometer recorded an increase in absorbance at 290 nm subsequent to ascorbate oxidation. Guaiacol peroxidase was quantified following the approach of34. The enzyme assays were conducted by combining 0.5 ml of 0.1 M K-phosphate buffer (pH 7.5), 0.5 ml of 3.4 mM guaiacol, 0.5 ml of H2O2, and 0.5 ml of enzyme extract in a glass cuvette. The absorbance at 480 nm was quantified by assessing the quantity of oxidized guaiacol.
To measure H2O2, fresh maize leaves (2 g) were ground and mixed into a 4 mL buffer solution (pH 6.8; PBS, 50 mM,), and transferred to a centrifugal tube for centrifugation (10,000× g, 4 °C, 15 min). The supernatant was then added to a mixture of titanium disulfate and sulfuric acid (20% v/v). Absorbance value was recorded at 415 nm and calculate the H2O2 content according to the standard curve as per methodology described by35,36. O2 was also recorded using standard methods of35.
Lipid peroxidation and membrane damage related attributes
Malondialdehyde (MDA) was determined using the methods given by37. The frozen maize leaf tissues were homogenized using 0.1% (w/v basis) trichloroacetic acid. Using spectrophotometer, the absorbance was recorded at 535 nm and was corrected at 600 nm for non-specific turbidity. The MDA (nmol g− 1 FW) was calculated using methods reported by38. 200 mg tissue of eighteen day old fresh tissue was used to calculate electrolyte leakage. Tissue was cut in one cm long strip and was placed in screw cap test tube having 8 ml of deionized and distilled water. The electrolyte leakage was calculated using the standard procedures described by39. The percentage electrolyte leakage was calculated using Eq. (6) as reported by27.
Osmolytes attributes
To measure proline contents in maize leaf, samples (0.5–1 g) from each pot were frozen in liquid nitrogen before determining proline concentration. Afterward, 0.5 g of frozen tissue was homogenized in 10 mL sulfosalicylic acid (3%) and centrifuged for 5 min according to40. Ninhydrin buffer (100 µL of 3% sulfosalicylic acid, 200 µL acidic ninhydrin, 200 µL glacial acetic acid,) and acetic acid (100%) were added to 2 mL of supernatant (v/v/v 1:1:1). The tube was incubated for 60 min at 96 °C. After cooling, the sample was extracted by toluene and the optical density of the upper organic phase was read at 520 nm. The proline concentration was determined using a standard L-proline concentration curve.
Maize leaf soluble sugar contents were determined through standard methods suggested by41. The standard method of42 was followed to determine soluble protein content. For 10 min, 0.5 g leaves were ground in 1.0 ml phosphate buffer (pH 7.0) solution. Then 0.1 ml extract was taken and 1.0 ml solution was prepared by adding distilled water and 1.0 ml of standard reagent [0.75 g Na2CO3, 0.1 N (NaOH) and 0.37 g Na-K tartrate in 40 ml distilled water. The solution was mixed for 10 min and 0.1 ml foline phenol reagent was added. The solution was in incubator for 30 min. The sample absorbance was determined at 650 nm wavelength.
Quality attributes
The ferulic acid was determined following the procedures suggested by30. Soluble sucrose contents were determined following the standard method of43. Sucrose contents were determined using glucose and sucrose curves.
Statistical analysis
Data were analyzed using analysis of variance (ANOVA) to assess treatment effects, and mean comparisons were performed with Tukey’s HSD test at p ≤ 0.05. Statistical analysis were conducted in Minitab (v17), while principal component analysis (PCA), heat maps, and correlation analysis were carried out in R-Studio to visualize multivariate relationships among traits.
Results
Growth and biomass attributes
Drought stress markedly reduced maize growth and biomass, with the greatest decline under severe stress (Fig. 1). Plant height, ear diameter, and ear length decreased compared with controls, whereas NaHS and biochar particularly their combined application (T5) significantly (p ≤ 0.05) restored these traits under drought conditions. Intense drought stress reduced the height of the plant (2.62%), ear diameter (5.10%), ear length (8.92%), wheareas, moderate drought stress decreased the plant height (1.83%), ear diameter (2.36%), ear length (5.37%) as compared to control conditions. Applied biochar + NaHS (0.1 mmol L− 1) of showed the maximum plant height (13.07%), ear diameter (14.59%) and ear length (20.23%) as compared to control under drought stressed conditions (Fig. 1).
Effect of biochar and sodium hydrosulfide (NaHS) on maize growth and biomass under drought stress. Treatments: T0 = control, T1 = 3% biochar mixed with pot soil, T2 = 0.05 mmol L⁻¹ NaHS, T3 = 0.1 mmol L⁻¹ NaHS, T4 = biochar + 0.05 mmol L⁻¹ NaHS, T5 = biochar + 0.1 mmol L⁻¹ NaHS. Stress levels: MDS = moderate drought stress, SDS = severe drought stress. Traits: plant height, ear diameter, and ear length.
Physiological/photosynthetic attributes
Drought stress markedly reduced chlorophyll fluorescence, pigments, carotenoids, photosynthetic rate, and stomatal conductance, while elevating int. CO₂ (Fig. 2). These effects were alleviated by biochar and NaHS, with the combined treatment (T5) showing the greatest improvement in photosynthetic performance (Fig. 2). Severe drought stress proved the maximum decrease in chlorophyll fluorescence (11.65%), chlorophyll a (15.35%), chlorophyll b (24.34%), total chlorophyll (19.44%), photosynthetic rate (10.67%), carotenoids content (16.48%) and stomatal conductance (gs) (12.39%), while, Int. CO2 (5.71%) was higher in comparison to control circumstances. The best level of applied biochar + NaHS (T5) enhanced the chlorophyll fluorescence (61.62%), chlorophyll a (50.28%), chlorophyll b (396.22%), total chlorophyll (138.68%), photosynthetic rate (89.74%), carotenoids content (76.62%) stomatal conductance (gs) (115%) and Int. CO2 (49.40%) as compared to control under drought stressed conditions, respectively.
Effect of biochar and various levels of NaHS on the physiological/photosynthetic traits of maize grown under drought stress. MDS = moderate drought stress; SDS = severe drought stress; To = control, T1 = 3% biochar mixed with pot soil, T2 = 0.05 mmol L− 1 NaHS application, T3 = 0.1 mmol L− 1 NaHS application, T4 = biochar mixed with pot soil + 0.05 mmol L− 1 NaHS application and T5 = biochar mixed with pot soil + 0.1 mmol L− 1 NaHS application.
Water related attribute
Various levels of applied NaHS and biochar significantly improved the leaf water contents in maize over control treatment under the different levels of drought stress (Fig. 3). Severe drought stress proved the maximum decrease in leaf water contents (5.51%) relative to the control group without treatment. Nonetheless, it was observed that applied biochar + NaHS (T5) treatment had a significant effect on enhancing water-related attributes in maize leaves. The best level of applied biochar + NaHS (T5) showed higher leaf water contents (37.81%) as compared to control under drought stressed conditions.
Effect of biochar and various levels of NaHS on the water related traits of maize grown under drought stress. MDS = moderate drought stress; SDS = severe drought stress; To = control, T1 = 3% biochar mixed with pot soil, T2 = 0.05 mmol L− 1 NaHS application, T3 = 0.1 mmol L− 1 NaHS application, T4 = biochar mixed with pot soil + 0.05 mmol L− 1 NaHS application and T5 = biochar mixed with pot soil + 0.1 mmol L− 1 NaHS application.
Enzymatic antioxidants activity
Drought stress elevated antioxidant enzyme activities and ROS markers (H₂O₂, O₂⁻) (Fig. 4), while the combined biochar + NaHS treatment moderated these responses, indicating reduced oxidative stress. Induced severe drought stress increased the SOD activity (17.25%), POD activity (8.71%), PPO activity (53.86%), PAL activity (56.42%), phenolics activity (62.57%), catalase activity (15.55%), APX activity (14.36%), GPX activity (12.91%), H2O2 activity (13.85%) and O2− activity (2.37%) relative to control circumstances. The optimal degree of biochar + NaHS (T5) treatment decreased the SOD activity (54.20%), POD activity (37.84%), PPO activity (56.23%), PAL activity (57.69%), phenolics activity (62.44%), catalase activity (60.58%), APX activity (26.42%), GPX activity (26.41%), H2O2 activity (37.29%) and O2− activity (16.50%) relative to the control group under drought environment.
Effect of biochar and various levels of NaHS on the enzymatic antioxidants activity of maize grown under drought stress. MDS = moderate drought stress; SDS = severe drought stress; To = control, T1 = 3% biochar mixed with pot soil, T2 = 0.05 mmol L− 1 NaHS application, T3 = 0.1 mmol L− 1 NaHS application, T4 = biochar mixed with pot soil + 0.05 mmol L− 1 NaHS application and T5 = biochar mixed with pot soil + 0.1 mmol L− 1 NaHS application.
Lipid peroxidation and membrane damage related attributes
Data on lipid per oxidation and membrane damage attributes of maize plants shown in Fig. 5 exhibited that drought stress increased the lipid per oxidation and membrane damage attributes of maize. Severe drought stress increased the MDA contents (3.54%) and electrolyte leakage (9.34%) as compared to control. However, the addition of NaHS along with biochar (T5) also decreased the MDA contents (32.82%) and electrolyte leakage (57.21%) in comparison with control where no treatment were applied under drought stressed conditions.
Effect of biochar and various levels of NaHS on the lipid peroxidation and membrane damage related traits of maize grown under drought stress. MDS = moderate drought stress; SDS = severe drought stress; To = control, T1 = 3% biochar mixed with pot soil, T2 = 0.05 mmol L− 1 NaHS application, T3 = 0.1 mmol L− 1 NaHS application, T4 = biochar mixed with pot soil + 0.05 mmol L− 1 NaHS application and T5 = biochar mixed with pot soil + 0.1 mmol L− 1 NaHS application.
Osmolytes attributes
The osmolyte characteristics of the drought-stressed maize plants increased linearly (Fig. 6). Under severe drought stress, there was an increased formation of osmolyte characteristics, such as proline (24.01%), soluble sugar (24.26%) and soluble protein (17.77%), compared to control scenarios. The tested level of NaHS (0.1 mmol L− 1) + biochar (T5) decreased the proline (66.26%), soluble sugar (55.94%) and soluble protein (61.57%) levels in maize plants relative to the control group without treatment.
Effect of biochar and various levels of NaHS on the osmolyte traits of maize grown under drought stress. MDS = moderate drought stress; SDS = severe drought stress; To = control, T1 = 3% biochar mixed with pot soil, T2 = 0.05 mmol L− 1 NaHS application, T3 = 0.1 mmol L− 1 NaHS application, T4 = biochar mixed with pot soil + 0.05 mmol L− 1 NaHS application and T5 = biochar mixed with pot soil + 0.1 mmol L− 1 NaHS application.
Quality attributes
Figure 7’s data on maize plant quality attributes demonstrated that drought stress improved the plants’ quality attributes. Severe drought stress increased the feraulic acid (64.50%) and sucrose (18.16%) as compared to control condition. The best level of NaHS (0.1 mmol L− 1) + biochar (T5) decreased the feraulic acid (42.72%) and sucrose (70.20%) levels in maize plants compared to the control group, which received no treatment.
Effect of biochar and various levels of NaHS on the quality traits of maize grown under drought stress. MDS = moderate drought stress; SDS = severe drought stress; To = control, T1 = 3% biochar mixed with pot soil, T2 = 0.05 mmol L− 1 NaHS application, T3 = 0.1 mmol L− 1 NaHS application, T4 = biochar mixed with pot soil + 0.05 mmol L− 1 NaHS application and T5 = biochar mixed with pot soil + 0.1 mmol L− 1 NaHS application.
Correlation analysis
An evident association was observed across all growth, biochemical, lipid peroxidation, reactive oxygen species (ROS) related, enzymatic, and quality-related parameters of maize plants. Physiological characteristics such as chlorophyll and carotenoid contents, water-related attributes, and growth attributes demonstrated negative correlations with enzymatic activities, oxidation of lipids, proline accumulation, and quality traits. Similarly, other attributes showed positive correlation with each other (Fig. 8).
Correlation matrix of various measured attributes of maize by the use of various treatments under drought stress. PH = plant height; ER = ear diameter; EL = ear length; LWC = leaf water contents; SS = soluble sugar; Chl. f = chlorophyll fluorescence; PR = photosynthetic rate; Chl a = chlorophyll a; Chl b = chlorophyll b; Tchl = total chlorophyll; ELL = electrolyte leakage; MDA = malonaldehyde; POD = peroxidase activity; SOD = superoxide dismutase activity; APX = ascorbate activity; CAT = catalase; sucrose; proline; Car = carotenoids; O2- activity; H2O2; SP = soluble protein; GPX; SC = stomatal conductance; Int. CO2; PPO = polyphenol Oxidase; PAL = phenylalanine ammonia-lyase; phenolics; FA = ferulic acid.
Principal component analysis
The principal component analysis (PCA) of the 29 measured variables accounts for 96.1% variability, where Dim1 exhibits 92.2% and Dim2 3.9% contributions for the maize plants. A clear trend of the control and drought-induced stress responses are observed (Fig. 9). The PCA analysis demonstrates clear clusters of variables. Phenolics, FA and PAL have a strong correlation and considerably affect Dim1, but Chl a, Chl b, and Car show a moderate correlation and contribute to the variation in Dim1. Sucrose, O2−, and CAT have a moderate correlation and contribute to the variance in Dim2. Int. CO2, LWC, and SC exert a diminished impact on overall variation.
Principal component analysis figure illustrating the loadings of assessed traits and the contributions of the two principal components (PC1 and PC2). PH = plant height; ER = ear diameter; EL = ear length; LWC = leaf water contents; SS = soluble sugar; Chl.f = chlorophyll fluorescence; PR = photosynthetic rate; Chl a = chlorophyll a; Chl b = chlorophyll b; Tchl = total chlorophyll; ELL = electrolyte leakage; MDA = malonaldehyde; POD = peroxidase activity; SOD = superoxide dismutase activity; APX = ascorbate activity; CAT = catalase; sucrose; proline; Car = carotenoids; O2- activity; H2O2; SP = soluble protein; GPX; SC = stomatal conductance; Int. CO2; PPO = polyphenol Oxidase; PAL = phenylalanine ammonia-lyase; phenolics; FA = ferulic acid.
Heat map
The heatmap illustrates clear grouping patterns among the variables, indicating possible linkages and functional categories. Variables such as Chl a, Chl b and Car display analogous expression patterns, suggesting their possible role in photosynthetic activities. Likewise, H2O2, CAT, and APX exhibit synchronized expression, indicating their involvement in antioxidant defense systems. Phenolics, PAL and FA form a unique cluster, likely linked to secondary metabolism. The color gradient (often red to blue) signifies the comparative expression level of each variable.
Red often signifies strong expression, whereas blue denotes low expression. White or gray signifies a neutral or intermediate expression level (Fig. 10).
Heatmap presenting response of maize plants treated with various treatments under drought stress. PH = plant height; ER = ear diameter; EL = ear length; LWC = leaf water contents; SS = soluble sugar; Chl.f = chlorophyll fluorescence; PR = photosynthetic rate; Chl a = chlorophyll a; Chl b = chlorophyll b; Tchl = total chlorophyll; ELL = electrolyte leakage; MDA = malonaldehyde; POD = peroxidase activity; SOD = superoxide dismutase activity; APX = ascorbate activity; CAT = catalase; sucrose; proline; Car = carotenoids; O2- activity; H2O2; SP = soluble protein; GPX; SC = stomatal conductance; Int. CO2; PPO = polyphenol Oxidase; PAL = phenylalanine ammonia-lyase; phenolics; FA = ferulic acid.
Discussion
Abiotic stress markedly impairs plant growth by disrupting essential physiological processes, resulting in diminished overall growth44. This study explores the potential benefits of using BC and NaHS for the cultivation of maize crop under limited water regimes without affecting their quality. The results of the present study suggested the application of drought stress markedly affected the growth and biomass attributes of the maize crop depending upon the severity of water stress. The application of BC and NaHS, individually or cumulatively, enhanced the growth and development of the maize crop (Fig. 1). Root and shoot biomass, and growth rates show a notable decline in plants in response to inadequate water availability45. Several factors may contribute to this reduction, including decreased water uptake due to diminished soil moisture, oxidative stress-induced damage to root and shoot cells, reallocation of resources to prioritize essential survival over root and shoot improvement, and impaired nutrient absorption46,47. Application of BC and NaHS in a synergistic way alleviates drought induced stress and enhance plant growth and biomass in maize. Biochar alleviates the physical water stress, and NaHS improves internal plant defense mechanism and leads to osmotic adjustment by the accumulation of proline and synthesis of soluble sugars. The possible reason for the improved growth and biomass of maize might be due to the dual action of these treatments that reduces the degradation of photosynthetic pigments, maintains the photosynthetic efficiency that ultimately results in increases the biomass accumulation and plant vigor under abiotic stress48,49. Application of BC increases soil water retention, porosity and cation exchange capacity, thereby assuring sustained water and nutrient availability to plants under drought conditions50. It also helps to improve root growth and stimulate activity of the microbes, involved in nutrient cycling51. As a signaling molecule, NaHS helps to protect the plant from drought stress induced oxidative damage by enhancing plant’s antioxidative defense system52. The sole application of BC makes soil more favorable, while NaHS used together enhances maize physiological resilience by manipulating stress response pathways53,54. That might be the possible reason for improving the drought tolerance in the maize crop. This integrated approach results in better water uptake, more favorable utilization of nutrients and sustained metabolic activity, thereby improving plant growth and biomass even under water limited conditions.
Findings of the current study revealed that all the physiological and photosynthetic attributes of maize were significantly affected by drought stress and overall productivity is reduced (Fig. 2). Limited water supply caused the closure of stomata that hinder the availability of carbon dioxide and ultimately disrupts the Calvin cycle which is important process of photosynthesis55. Limited water regimes further reduces the CO₂ assimilation with the accumulation of reactive oxygen species (ROS) that damage chlorophyll molecules, thylakoid membranes and photosynthetic enzymes56. Under drought stress, the efficiency of electron transport rate decrease result in reduction of the net photosynthetic rate57. Adverse effects on other physiological processes, for example transpiration, and chlorophyll fluorescence, also cause a reduction in overall decrease in the photosynthetic traits that reduces the plant vigor58. From the findings it is obvious that soil addition of BC and foliar applied NaHS application could improve the physiological and photosynthetic of maize under limited water supply. By enhancing soil water retention and availability of nutrients, BC assures a sufficient supply of water and essential nutrients to the plants, and hence stomatal conductance, and photosynthetic activity59. Moreover, addition of BC stimulates the soil microbial functioning and stimulates production of plant growth promoting substances and stress alleviating metabolites60 that might be the possible reason of these improvements. As a supplier of hydrogen sulfide, NaHS is a critical activator for plants’ antioxidative defense mechanisms61. Expression of stress responsive mechanisms also modulated by hydrogen sulfide which leads to gradual improvement of chlorophyll stability and photosynthetic capacity that might be the possible reason for improved photosynthetic and physiological attributes of maize under limited water supply. Cumulative effect of biochar and NaHS enhances the antioxidative defence that in turn suppress oxidative damage to cellular membranes and chloroplasts which ultimately results in improved photosynthetic traits62,63. Together, BC provides suitable environment that is stable, whereas NaHS improves the cellular capacity of the plant to tolerate and recover from stress, that might be the possible reason of improvement of photosynthetic and physiological attributes of maize under drought stress.
Limited water supply caused significant changes in the levels of enzymatic antioxidants, lipid peroxidation, osmolyte and quality variables in maize crop (Figs. 3, 4, 5, 6 and 7). Maize plants under drought stress experience a major metabolic modulation which results in oxidative damage and poor productivity64. Drought stress affect the plant’s antioxidative defense system by the excessive production of ROS, thus limiting the activity of enzymatic antioxidants65. Osmotic imbalances induced by drought stress, and osmoprotective compatible solutes such as proline and soluble sugars are reduced for maintaining cellular turgor and osmoprotection66. When these biochemical disruptions are combined together, there is a decline in the yield and quality attribute of maize. Based on N nutrient concentration, further reduction in photosynthates and impaired translocation of assimilates exacerbate the stress induced decline in productivity and grain quality67. Combined application of BC and NaHS treatment provide a long-term solution to mitigate the negative impacts of drought in maize. Application of BC caused an improvement of water retention and nutrient availability in soil, which results in improved physiological performance owing to a reduction in oxidative stress, via improving soil quality68. In addition, it stimulates accumulation of osmolytes, including proline and soluble sugars, which contribute to osmotic adjustment and stress tolerance69. The application of NaHS enhances the activity of enzymatic antioxidants (SOD, CAT, POD), reduces the level of ROS and maintains the level of lipid peroxidation70. A reduction of MDA levels in maize plants treated with NaHS under drought stress demonstrates similar observations71. Moreover, NaHS has been shown to modulate the expression of stress responsive mechanism including osmolyte biosynthesis that protect cellular structures72, that might be the possible reason for the modulation of enzymatic activities under drought stress. The combined application of BC and NaHS significantly enhance the maize drought stress tolerance by the multiple cumulative mechanisms. The possible reason behind these mechanisms is that BC improves soil physical properties, increases water retention capacity and improves root aeration creating a more favorable rhizospheric environment for nutrient uptake73. At the same time, NaHS is an H2S donor, a key gaseous signaling molecule that is involved in the modulation of stress-dependent pathways74. The resultant decrease in oxidative stress results in lower lipid peroxidation as reflected by lower MDA levels and ensures membrane integrity. So, co-application promotes the accumulation of osmolytes that help maintain cellular turgor during water deficit75,76. Together, biochar and NaHS not only modulate the antioxidative defense system, but they also increase yield and quality attributes by increasing photosynthate production and partitioning mechanism. Improved enzymatic antioxidant activity, reduced lipid peroxidation, and higher yield is also correlated in maize under combined treatments. Significant implications for agriculture and environmental sustainability are provided by the findings of this study. The combined action of BC and NaHS not only mitigates the negative impacts of limited water related effects but also enhances plant growth, resulting in improved overall plant health and increased resistance to limited water supply.
Conclusion
This study demonstrates that drought stress severely compromises maize growth by reducing photosynthetic pigments and leaf water content, while intensifying oxidative damage and osmolyte accumulation. The results demonstrated that combined application of biochar and sodium hydrosulfide (NaHS) at 0.1 mmol L⁻¹ alleviated these negative impacts, leading to improvements in growth traits, photosynthetic activity, and water conservation, along with reductions in excessive antioxidant enzyme activity, lipid peroxidation, and membrane injury. Overall, the findings highlight that integrating biochar with NaHS could serve as an effective approach to strengthen drought tolerance and enhance maize productivity, particularly in water-limited regions. Further investigations at the field level and molecular scale are recommended to validate and deepen the understanding of this synergistic strategy.
Data availability
The data will be provided by the corresponding author on suitable statement.
Change history
28 February 2026
The original online version of this Article was revised: The original version of this Article contained an error in the Funding section. The correct Funding section now reads: “This work was supported and funded by the Deanship of Scientific Research at Imam Mohammad Ibn Saud Islamic University (IMSIU) (grant number IMSIU-DDRSP-2602).”
References
Nourashrafeddin, S. M., Ramandi, A. & Seifi, A. Xerophyte-Derived synthetic bacterial communities enhance maize drought tolerance by increasing plant water use efficiency. J. Plant. Growth Regul. 43, 4135–4150 (2024).
Rasheed, A. et al. A critical review on the improvement of drought stress tolerance in rice (Oryza sativa L). Not Bot. Horti Agrobo. 48, 1756–1788 (2020).
Syed, A., Raza, T., Bhatti, T. T. & Eash, N. S. Climate impacts on the agricultural sector of pakistan: risks and solutions. Environ. Challenges. 6, 100433 (2022).
Waseem, M., Khurshid, T., Abbas, A., Ahmad, I. & Javed, Z. Impact of meteorological drought on agriculture production at different scales in Punjab, Pakistan. J. Water Clim. Change. 13, 113–124 (2022).
Wang, X. et al. Photosynthetic carbon fixation and sucrose metabolism supplemented by weighted gene Co-expression network analysis in response to water stress in rice with overlapping growth stages. Front. Plant. Sci. 13, 864605 (2022).
Yang, Y. et al. Rapid and nondestructive evaluation of wheat chlorophyll under drought stress using hyperspectral imaging. IJMS 24, 5825 (2023).
Hasanuzzaman, M. et al. Regulation of ROS metabolism in plants under environmental stress: A review of recent experimental evidence. IJMS 21, 8695 (2020).
Rad, R. D. et al. Impact of drought stress on biochemical responses, energy, and water productivity on maize forage (Zea Mays L). SN Appl. Sci. 3, 834 (2021).
Naderi, R., Ali, K., Rehman, A., Rasmann, S. & Weyl, P. Estimating the impact on maize production by the weed Parthenium hysterophorus in Pakistan. CABI Agric. Biosci. https://doi.org/10.1186/s43170-024-00217-2 (2024).
Baier, C., Gross, A., Thevs, N. & Glaser, B. Effects of agroforestry on grain yield of maize (Zea Mays L.)—A global meta-analysis. Front. Sustain. Food Syst. 7, 1167686 (2023).
CGIAR. What do we know about the future of maize value chains in a changing climate and agri-food system? (2024).
Safdar, M. E., Tanveer, A., Khaliq, A. & Riaz, M. A. Yield losses in maize (Zea mays) infested with parthenium weed (Parthenium hysterophorus L). Crop Prot. 70, 77–82 (2015).
Zhou, H. et al. Hydrogen sulfide promotes rice drought tolerance via reestablishing redox homeostasis and activation of ABA biosynthesis and signaling. Plant Physiol. Biochem. 155, 213–220 (2020).
Corpas, F. J. & Palma, J. M. H2S signaling in plants and applications in agriculture. J. Adv. Res. 24, 131–137 (2020).
Zonayet, M. et al. Impact of Biochar as a soil conditioner to improve the soil properties of saline soil and productivity of tomato. Sustainability 15, 4832 (2023).
Wu, Y. et al. The critical role of Biochar to mitigate the adverse impacts of drought and salinity stress in plants. Front. Plant. Sci. 14, 1163451 (2023).
Liu, S. & Qin, F. Genetic dissection of maize drought tolerance for trait improvement. Mol. Breed. 41, 8 (2021).
Ma, Z. et al. Maize ZmWRKY71 gene positively regulates drought tolerance through reactive oxygen species homeostasis. Plant Physiol. Biochem. 219, 109399 (2025).
Deribe, H. Review on effects of drought stress on maize Growth, yield and its management strategies. Commun. Soil Sci. Plant Anal. 56, 123–143 (2025).
Gomaa, M. A. et al. Increase maize productivity and water use efficiency through application of potassium silicate under water stress. Sci. Rep. 11, 224 (2021).
Kathirvelan, P. et al. Response of maize (Zea Mays L.) to Foliar-Applied nanoparticles of zinc oxide and manganese oxide under drought stress. Plants 14, 732 (2025).
Naeem, M. B. et al. Improving maize yield and drought tolerance in field conditions through activated Biochar application. Sci. Rep. 14, 25000 (2024).
Igbadun, H. E., Tarimo, A. K. P. R., Salim, B. A. & Mahoo, H. F. Evaluation of selected crop water production functions for an irrigated maize crop. Agric. Water Manage. 94, 1–10 (2007).
Karvar, M., Azari, A., Rahimi, A., Maddah-Hosseini, S. & Ahmadi-Lahijani, M. J. Potassium silicate reduces water consumption, improves drought tolerance, and enhances the productivity of sweet corn (Zea mays) under deficit irrigation. Acta Physiol. Plant. 45, 38 (2023).
Arnon, D. Copper enzymes in isolated chloroplasts: Polyphenoloxidase in Beta vulgaris. Plant. Physiol. 24, 1–15 (1949).
Zhang, R. H., Zhang, X. H., Camberato, J. J. & Xue, J. Q. Photosynthetic performance of maize hybrids to drought stress. Russ. J. Plant Physiol. 62, 788–796 (2015).
Bijanzadeh, E., Barati, V. & Egan, T. P. Foliar application of sodium silicate mitigates drought stressed leaf structure in corn (Zea Mays L). South. Afr. J. Bot. 147, 8–17 (2022).
Wang, J., Zhong, X. M., Lv, X. L., Shi, Z. S. & Li, F. H. Photosynthesis and physiology responses of paired near-isogenic lines in waxy maize (Zea Mays L.) to nicosulfuron. Photosynt 56, 1059–1068 (2018).
Mohammadi, M. & Kazemi, H. Changes in peroxidase and polyphenol oxidase activities in susceptible and resistant wheat heads inoculated with fusarium graminearum and induced resistance. Plant Sci. 162, 491–498 (2002).
Peltonen, S. & Karjalainen, R. Phenylalanine Ammonia-lyase activity in barley after infection with Bipolaris Sorokiniana or treatment with its purified Xylanase. J. Phytopathol. 143, 239–245 (1995).
Chance, B. & Maehly, A. C. [136] Assay of catalases and peroxidases. In Methods in Enzymology Vol. 2 764–775 (Elsevier, 1955).
Yousaf, M. I. et al. Responses of maize hybrids to water stress conditions at different developmental stages: accumulation of reactive oxygen species, activity of enzymatic antioxidants and degradation in kernel quality traits. PeerJ 11, e14983 (2023).
Amako, K., Chen, G. X. & Asada, K. Separate assays specific for ascorbate peroxidase and Guaiacol peroxidase and for the chloroplastic and cytosolic isozymes of ascorbate peroxidase in plants. Plant Cell Physiol. https://doi.org/10.1093/oxfordjournals.pcp.a078621 (1994).
Hammerschmidt, R., Nuckles, E. M. & Kuć, J. Association of enhanced peroxidase activity with induced systemic resistance of cucumber to Colletotrichum lagenarium. Physiol. Plant Pathol. 20, 73–82 (1982).
Talaat, N. B., Shawky, B. T. & Ibrahim, A. S. Alleviation of drought-induced oxidative stress in maize (Zea Mays L.) plants by dual application of 24-epibrassinolide and spermine. Environ. Exp. Bot. 113, 47–58 (2015).
Jiang, Z. et al. Exogenous ABA enhances the antioxidant defense system of maize by regulating the AsA-GSH cycle under drought stress. Sustainability 14, 3071 (2022).
Heath, R. L. & Packer, L. Photoperoxidation in isolated chloroplasts. Arch. Biochem. Biophys. 125, 189–198 (1968).
Ibrahim, M., Khan, A., Anjum, Ali, W. & Akbar, H. Mulching techniques: an approach for offsetting soil moisture deficit and enhancing manure mineralization during maize cultivation. Soil Tillage. Res. 200, 104631 (2020).
Gong, M., Li, Y. J. & Chen, S. Z. Abscisic acid-induced thermotolerance in maize seedlings is mediated by calcium and associated with antioxidant systems. J. Plant Physiol. 153, 488–496 (1998).
Bates, L. S., Waldren, R. P. & Teare, I. D. Rapid determination of free proline for water-stress studies. Plant. Soil. 39, 205–207 (1973).
Van Handel, E. Direct microdetermination of sucrose. Anal. Biochem. 22, 280–283 (1968).
Lowery, O. H., Poesenbrough, N. J., Randall, R. & FAL, A. L. & J. Protein measurement with the Folin phenol reagent. J. Biol. Chem. 193, 265–275 (1951).
Irigoyen, J. J., Einerich, D. W. & Sánchez-Díaz, M. Water stress induced changes in concentrations of proline and total soluble sugars in nodulated alfalfa (Medicago sativa) plants. Physiol. Plant. 84, 55–60 (1992).
Shabbir, R. et al. Combined abiotic stresses: challenges and potential for crop improvement. Agronomy 12, 2795 (2022).
Guasconi, D., Manzoni, S. & Hugelius, G. Climate-dependent responses of root and shoot biomass to drought duration and intensity in grasslands–a meta-analysis. Sci. Total Environ. 903, 166209 (2023).
Chaudhry, S. & Sidhu, G. P. S. Climate change regulated abiotic stress mechanisms in plants: a comprehensive review. Plant. Cell. Rep. 41, 1–31 (2022).
Hu, D. et al. Maize (Zea Mays L.) responses to salt stress in terms of root anatomy, respiration and antioxidative enzyme activity. BMC Plant. Biol. 22, 602 (2022).
Zanganeh, R., Jamei, R. & Rahmani, F. Response of maize plant to sodium hydrosulfide pretreatment under lead stress conditions at early stages of growth. Cereal Res. Commun. 49, 267–276 (2021).
Rahman, S. U. et al. Synergetic effects of potassium and Biochar on morphological, physiological, and biochemical attributes of maize crop grown under different levels of drought stress. BMC Plant. Biol. 25, 402 (2025).
Dengxiao, Z. et al. Combined Biochar and water-retaining agent application increased soil water retention capacity and maize seedling drought resistance in fluvisols. Sci. Total Environ. 907, 167885 (2024).
Abideen, Z. et al. Biochar outperforms Biochar-Compost mix in stimulating ecophysiological responses and enhancing soil fertility under drought conditions. J. Soil. Sci. Plant. Nutr. https://doi.org/10.1007/s42729-024-02073-5 (2024).
Zhao, J. et al. Hydrogen sulfide increases drought tolerance by modulating carbon and nitrogen metabolism in Foxtail millet seedlings. Agronomy 14, 1080 (2024).
Kamali, M. et al. Biochar for soil applications-sustainability aspects, challenges and future prospects. Chem. Eng. J. 428, 131189 (2022).
Liu, H., Chong, P., Liu, Z., Bao, X. & Tan, B. Exogenous hydrogen sulfide improves salt stress tolerance of Reaumuria soongorica seedlings by regulating active oxygen metabolism. PeerJ 11, e15881 (2023).
Zhang, F., Rosental, L., Ji, B., Brotman, Y. & Dai, M. Metabolite-mediated adaptation of crops to drought and the acquisition of tolerance. Plant J. 118, 626–644 (2024).
Rane, J., Singh, A. K., Tiwari, M., Prasad, P. V. V. & Jagadish, S. V. K. Effective use of water in crop plants in dryland agriculture: implications of reactive oxygen species and antioxidative system. Front. Plant. Sci. 12, 778270 (2022).
Bendou, O. et al. Physiological and antioxidant response to different water deficit regimes of flag leaves and ears of wheat grown under combined elevated CO2 and high temperature. Plants 11, 2384 (2022).
Plant Abiotic Stress Physiology: Volume 1: Responses and Adaptations (Apple Academic Press, 2022). https://doi.org/10.1201/9781003180562
Nawaz, F. et al. Biochar amendment in combination with endophytic bacteria stimulates photosynthetic activity and antioxidant enzymes to improve soybean yield under drought stress. J. Soil. Sci. Plant. Nutr. 23, 746–760 (2023).
Mushtaq, T., Bano, A. & Ullah, A. Effects of rhizospheric Microbes, growth Regulators, and Biochar in modulating antioxidant machinery of plants under stress. J. Plant. Growth Regul. https://doi.org/10.1007/s00344-024-11548-9 (2024).
Luo, S. et al. Foliar spraying of NaHS alleviates cucumber salt stress by maintaining N+/K + Balance and activating salt tolerance signaling pathways. Plants 12, 2450 (2023).
Lin, S. et al. Hydrogen sulfide improves photosynthetic efficiency by regulating light energy dissipation and reversible phosphorylation of thylakoid proteins in rice under salt stress. Plant Physiol. Biochem. 224, 109908 (2025).
Gao, C., Qin, J., Tian, Y., Yang, J. & Wang, G. Biochar and soil water synergistically regulating root growth to affect photosynthesis in maize (Zea Mays L). Agronomy 15, 2170 (2025).
Sattar, A. et al. Exogenous application of Strigolactone alleviates drought stress in maize seedlings by regulating the physiological and antioxidants defense mechanisms. Cereal Res. Commun. 50, 263–272 (2022).
Luqman, M. et al. Effect of Strigolactone on growth, photosynthetic efficiency, antioxidant activity, and osmolytes accumulation in different maize (Zea Mays L.) hybrids grown under drought stress. Plant Signal. Behav. 18, 2262795 (2023).
Hussain Wani, S., Singh, B. & Haribhushan, N. Iqbal Mir, J. Compatible solute engineering in plants for abiotic stress Tolerance - Role of Glycine betaine. CG 14, 157–165 (2013).
Teng, Z. et al. Environmental stimuli: A major challenge during grain filling in cereals. IJMS 24, 2255 (2023).
Algethami, J. S., Ibrahim, M., Javed, W., Alosaimi, E. H. & Irshad, M. K. Efficacy of Fe-BC in enhancing growth, photosynthesis, nutrition, and alleviating the toxicity of cd and cr in rapeseed (Brassica Napus L.): A tool for managing the environment and attaining sustainable agriculture. Environ. Technol. Innov. 36, 103789 (2024).
Farouk, S., AL-Huqail, A. A. & El-Gamal, S. M. A. Potential role of biochar and silicon in improving physio-biochemical and yield characteristics of borage plants under different irrigation regimes. Plants 12, 1605 (2023).
Lin, Y. J., Feng, X. H. & Feng, Y. X. Regulation of enzymatic and non-enzymatic antioxidants in rice seedlings against chromium stress through sodium hydrosulfide and sodium Nitroprusside. Environ. Sci. Pollut Res. 30, 25851–25862 (2022).
Oloumi, H., Maleki, M., Habibipour, L. & Lotfi, S. Foliar application of NaHS alleviates cd toxicity in soybean plants through regulation of glutathione metabolism. Plant. Stress. 11, 100363 (2024).
Wang, H. et al. Exogenous hydrogen sulfide increased Nicotiana tabacum L. resistance against drought by the improved photosynthesis and antioxidant system. Sci. Rep. 14, 25534 (2024).
Ahmed, A. F., Fawzy, M., Al-zahrani, M. & Abdelkader, M. Enhancing metabolic processes and water deficit stress tolerance of maize plants through Biochar addition and foliar application of zinc nanoparticles. Russ J. Plant. Physiol. 72, 10 (2025).
Ullah, F. et al. Sodium Nitroprusside and melatonin improve physiological vitality and drought acclimation via synergistically enhancing antioxidant response in dryland maize. J. Plant. Growth Regul. 44, 891–908 (2025).
Faizan, M. et al. Drought stress mitigation with the use of Biochar. In Biochar: A Precious Resource from Biological Waste (eds Singh, S., Srivastava, S. et al.) 193–209 (Springer Nature Singapore, 2025). https://doi.org/10.1007/978-981-95-0425-1_8.
Akin, S. & Kaya, C. Impact of Salicylic acid and sodium hydrosulfide applied singly or in combination on drought tolerance and grain yield in wheat plants. Food Energy Secur. 13, e532 (2024).
Funding
This work was supported and funded by the Deanship of Scientific Research at Imam Mohammad Ibn Saud Islamic University (IMSIU) (grant number IMSIU-DDRSP2602).
Author information
Authors and Affiliations
Contributions
Conceptualization, M.Z.M, K.A., A.E, W.A, Q.Z.; methodology, K.I.; validation, H.M.A.; formal analysis, T.A. A.E, W.A, H.R, M.A.; investigation, M.Z.M., A.E, W.A, H.R, M.A, Ch.S.H.; resources, A.E, M.A.A.; data curation, Ch.S.H.; writing: original draft preparation, M.Z.M., K.I., A.E, W.A, H.R, M.A K.A., Q.Z.; writing: review and editing, M.Z.M., A.E, W.A, H.R, M.A K.A.; funding acquisition,A.E Q.Z. All authors have read and agreed to the published version of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Mansha, M.Z., Elkelish, A., Ikram, K. et al. Synergistic application of biochar and sodium hydrosulfide enhances maize drought tolerance through improved physiological performance and stress mitigation. Sci Rep 15, 43558 (2025). https://doi.org/10.1038/s41598-025-27971-1
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-27971-1