Abstract
Serum biomarkers of neuronal and glial damage gained significant attention in recent years for their diagnostic and prognostic value in acute stroke. In this study, we explored the potential of spot urine testing as a practical, non-invasive alternative. Using ultrasensitive single-molecule arrays, we assessed levels of glial fibrillary acidic protein (GFAP), neurofilament light chain (NfL), ubiquitin carboxy-terminal hydrolase L1 (UCH-L1), and total tau (t-tau) in urine and blood samples from hospitalized patients with acute ischemic stroke (AIS) and intracerebral hemorrhage (ICH) within 96 h after symptom onset. The results were correlated with mortality rates, tissue damage extent and modified Rankin Scale (mRS) scores at discharge and after three months. Significant variables were included into an multivariate regression model with confounder correction (NIHSS at admission, age at onset and serum creatinine). As a result, absolute concentrations of urine GFAP, NfL and t-tau were significantly higher in patients with in-hospital death or death at 3 months follow-up compared to patients reaching a mRS ≤ 3. Using multivariate regressions models, urine GFAP demonstrated the strongest predictive value across all biomarkers for in-hospital functional outcome, which was superior to the respective GFAP serum concentrations, whereas NfL in serum was the only significantly predictive biomarker for 3 months functional outcome. For radiological outcomes, urine GFAP concentrations were closely linked with ischemic lesion size (ρ = 0.42, p = 0.01). In conclusion, the measurement of neuronal and glial biomarkers in urine is feasible and may enhance the prediction of functional and imaging outcomes.
Similar content being viewed by others
Introduction
The glial and neuronal cell death due to intracerebral hemorrhage (ICH) or acute ischemic stroke (AIS) leads to the release of neuronal and glial biomarkers into blood1,2. As such, glial fibrillary acidic protein (GFAP), neurofilament light chain (NfL), ubiquitin carboxy-terminal hydrolase L1 (UCH-L1), and tau levels in particular increase in serum following acute brain damage3,4,5,6,7. Previous studies have already shown a positive correlation of these biomarkers in blood with the functional outcome and the extent of tissue damage and the feasibility (for GFAP when performed in the early phase) to even differentiate between etiologies (ICH vs. AIS)6,8,9,10. While the measurement of biomarkers in blood has widely been evaluated for this particular purpose over the last decade, there are no reliable data about the use of urine as a non-invasive alternative. In a previous study, we demonstrated that GFAP, NfL, UCH-L1, and total tau (t-tau) could be non-invasively detected in urine from healthy volunteers and patients with acute neuronal and glial damage, providing a diagonstic value in differentiating patients with acute brain damage from healthy subjects11. To date, it has not been investigated whether the measurement of these biomarkers in urine is also suitable for predicting mortality and functional outcome or its association with the extent of tissue damage. In this regard, the advantage of a measurement in urine is its non-invasiveness, which can be particularily helpful in structurally weaker regions. In addition, urine as an ultrafiltrate of the blood with a possible accumulation of proteins could have a different, perhaps even superior diagnostic or predictive value to a measurement in serum. The aim of this study was therefore to evaluate the correlation and predictive value of GFAP, NfL, UCH-L1 and t-tau levels in urine with the neurological outcome and the imaging based tissue damage. Furthermore, we aimed to investigate whether a distinction can be made between the respective etiologies (ICH vs. AIS).
Methods
Study design
This prospective monocentric study was performed between October 2019 and January 2021 at the Department of Neurology of the University Hospital Frankfurt, Germany.
Inclusion criteria were as follows: 1. confirmation of AIS or ICH by cerebral magnetic resonance imaging or computed tomography; 2. age > 18 years; and 3. blood and urine collection within 96 h after symptom onset or “last seen well”. Exclusion criteria were: 1. transient ischemic attack, ischemic stroke or ICH within the last 3 months; 2. brain tumor; 3. traumatic brain injury within the last 3 months; and 4. lack of informed consent from the patient, legal representative, or relatives.
Clinical baseline and outcome parameters
Clinical parameters were captured as follows: diagnosis of AIS or ICH according to International Statistical Classification of Diseases and Related Health Problems 10th revision (2021), age, sex, National Institutes of Health Stroke Scale (NIHSS) score at admission and discharge, serum and urine creatinine, stroke etiology, individual pre-existing conditions and risk factors and the pre-stroke modified Rankin Scale (pre-mRS). For patients with AIS, the mode of recanalization (if performed) was registered (i.e. intravenouse thrombolysis or mechanical thrombectomy). The main outcome measures were the in-hospital mortality, the mRS at discharge and, if available, at 3 months following the index event. The extent of tissue damage (lesion volume) was detected in the follow-up cerebral imaging at 24 h (or if follow-up scans were unavailable in the initial imaging) and evaluated by an expierenced neuroradiologist by manual volumetry on a radiologic workstation (Centricity RIS-i 7 Viewer; GE Healthcare, Chicago, USA) using axial diffusion-weighted MR images (b = 1000 mm/s2, slice thickness 5 mm,) or axial computed tomographic images (slice thickness 5 mm), respectively. For patients who underwent mechanical thrombectomy, the reperfusion result was also recorded using the thrombolysis in cerebral infarction (TICI) score.
Blood and urine sampling and measurements
Serum samples were drawn by venous puncture, while the concomitant urine samples were obtained as midstream spot urine or, if this was not possible, from the puncture chamber of a transurethral catheter and collected in a urine tube. The time point of collection within 96 h after symptom onset or “last seen well” was recorded. The 96-h time frame was determined based on previously reported kinetics of serum GFAP, aiming to measure the estimated serum peak of biomarkers during the early subacute phase12,13. After obtaining serum and urine samples, the tubes were centrifuged and the supernatant was stored in Eppendorf tubes at − 80 °C; sample collection and freezing was carried out within 4 h after collection. The Simoa® Human Neurology 4-Plex A and B (N4PA, N4PB) assay (a digital immunoassay for the quantitative determination of NfL, t-tau, GFAP and UCH-L1 in human serum, plasma and cerebrospinal fluid) was used to analyze all samples. Incorrect measurements, such as saturated samples, were excluded from the following analysis. Values below the limit of detection were considered to be 0.0 pg/ml and included in the analysis. This was the case for 5 patients in the serum UCH-L1 measurements and for one patient in the urine GFAP measurement. A sensitivity analysis excluding these patients can be found in the supplemental material. Because urine collection was performed as spot urine, dilution correction was calculated using a biomarker–creatinine ratio in addition to the absolute urine concentration (biomarker/creatinine in urine [mg/dl]/[mg/dl])11,14.
Statistical analysis
Statistical analysis was performed using GraphPad Prism Version 10.2.2, GNU R Version 4.1.1 as well as IBM SPSS® (IBM Corp. Released 2021. IBM SPSS Statistics for Macintosh, Version 29.0. Armonk, NY: IBM Corp). Ordinal and non-normal continuous data is depicted as median with 25th and 75th percentiles (Q1–Q3), normally distributed continuous data is depicted as mean ± standard error of the mean (SEM). Frequencies of nominal data are depicted as percentages. Depending on the normal distribution and level of measurement, baseline variables and biomarker levels were compared between two outcome groups using the t test, the Mann–Whitney U test, the χ2 test, or if more than to groups were compared using the Kruskal–Wallis test. A p-value < 0.05 indicated statistical significance.
A multivariate ordinal regression model was applied to predict the mRS at discharge and at 3 months. Candidate covariates were identified through Spearman correlation and univariate regression analyses of serum and urine biomarkers. Variables with a p-value < 0.05 were considered for inclusion in the multivariate analysis. Each biomarker was tested in a separate model as the primary variable of interest, with adjustment for NIHSS on admission, age, and serum creatinine. Model fit of the multivariate regression models were evaluated using the Chi-square (χ2) statistic from the likelihood ratio test, with higher χ2 values indicating a greater contribution to model fit, allowing direct comparison of explanatory power across biomarkers. To control for multiple testing, p-values were adjusted using the Benjamini–Hochberg false discovery rate (FDR) procedure and reported as q-values. The same analytical strategy was applied to infarct size and hemorrhage volume (ml), using linear regression models. In addition, receiver operating characteristic (ROC) analysis was performed to determine cutoff values of serum and urine biomarkers for in-hospital mortality and 3-month mortality. For each ROC analysis, the area under the curve (AUC), sensitivity (Sn), and specificity (Sp) were reported.
Standard protocol approvals, registrations, and patient consents
The study protocol was approved by the institutional review board of the Goethe-University Frankfurt (236/05). All procedures involving human participants were conducted in accordance with the ethical standards of the institutional review board and with the 1964 Declaration of Helsinki and its later amendments. Written informed consent was obtained from all patients or their authorized representatives before the samples were included in the study.
Results
Study population
Blood and urine samples were collected from 74 patients. One patient was excluded who was initially mistakenly treated as a stroke, but the final diagnosis was an epileptic seizure with Todd’s paresis. In 4 patients, the measurements failed and no values (neither from the urine nor the blood) could be obtained. In two patients written informed consent was missing. In one patient, the final diagnosis was a transient ischemic attack, leaving 66 patients to the combined patient analysis. Two patients with AIS presented with parenchymal hematoma type PH2 according to the Heidelberg bleeding classification15. In one patient, the infarctions were caused by a sinus thrombosis. This patient also presented with congestion hemorrhages. These three patients were excluded from the analysis according to the stroke types due to uncertain group assignment (ICH vs. AIS, Fig. 1). From 6 patients no data on the mRS at 3 months was available (lost to follow-up rate: 8.96%). Overall, 10 patients (15.2%) of the combined patient group analysis died within the first 7 days (defined as in-hospital mortality), and 5 more (7.5%) between discharge and the follow-up at 3 months.
Study flow chart. AIS acute ischemic stroke, ICH intracerebral hemorrhage.
Characteristics according to the respective stroke subtype (AIS vs. ICH)
Our cohort consisted of 48 patients with AIS (2 patients were excluded from this subgroup analysis due to parenchymal hematoma) and 15 patients with ICH. The median age of the patients suffering an ICH was significantly lower compared to patients with AIS (62 years [53–71 years] in the ICH group vs. 77 years [66–83 years] in the AIS group, p = 0.02). The AIS group showed a higher average initial serum creatinine level, while the ICH group tended to be more severely affected with a higher NIHSS at admission and at discharge, although these differences were not statistically significant.
The median infarct size was 36 ml (2–104 ml), the median ICH volume was 128 ml (38–294). We found a trend towards a worse functional outcome measured by mRS at discharge in patients with ICH (median mRS at discharge 5 (4–5) vs. 4 (2– 5) in the AIS group, p = 0.1) as well as in mortality, however this was not statistically significant. Time from symptom onset to sample collection did not show a significant difference between AIS and ICH (Table 1).
Median serum levels were lower in the ICH patients than in the AIS group for NfL: Δ = 10.31 pg/ml, p = 0.24; UCH-L1: Δ = 3.95 pg/ml, p = 0.56 and t-tau: Δ = 0.12 pg/ml, p = 0.5, while GFAP showed an increased median level in ICH compared to AIS patients (Δ = 1803 pg/ml, p = 0.45). Urine median levels were approximately the same for all biomarkers across both groups. However, all the differences mentioned were not statistically significant (supplemental Fig. S1). The corresponding comparisons using the biomarker-creatinine ratios (biomarker-CRs) can be found in the supplemental material (supplemental Fig. S2).
Differences in mRS at discharge and after 3 months
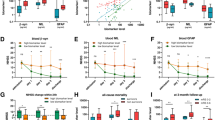
We stratified the combined patient group according to their functional status at discharge and after 3 months by using the mRS categories at least fair outcome (mRS 0–3), poor to very poor outcome (mRS 4–5) and dead (mRS 6)16. We found a steady increase in the urine concentrations of GFAP, NfL and t-tau when stratified to the respective mRS categories with significant differences between patients with at least fair outcome and dead patients for NfL, t-tau and GFAP (GFAP: Δ 13.45, p = 0.02; Δ 15.95, p = 0.02; NfL: Δ 22.7, p = 0.009; Δ 20.4, p = 0.004; t-tau: Δ 20.2, p = 0.03, Δ 18.8, p = 0.01 at discharge and after 3 months, respectively; Fig. 2) and a reversed pattern for UCH-L1 with higher levels in the patients with at least fair outcome compared to patients with poor to very poor outcome and dead patients for both time points (at discharge and after 3 months). A detailed description of the single biomarker levels can be found in the supplemental material.
(A–C) GFAP, NfL and t-tau urine concentration according to mRS category at discharge. (D–F) GFAP, NfL and t-tau urine concentration according to mRS category at 3 months. Tukey box-and-whisker-plot.
Correlation and regression analysis of the functional outcome
A multivariate ordinal regression model was used to predict the mRS at discharge and at 3 months. In an initial Spearman correlation and univariate ordinal regression analysis to determine suitable covariates, serum and urine levels of GFAP, NfL, and t-tau were significantly associated with the mRS at discharge and at 3 months, whereas the NfL-CR was associated only with the mRS at discharge. A detailed summary is provided in Table 2, and correlations with functional outcomes for the individual patient groups (ICH and AIS) are presented in the supplemental material (Supplemental Table S1).
The significant variables were entered into multivariable ordinal regression models with the mRS at discharge or at 3 months as the dependent variable, adjusted for confounders (NIHSS at admission, age at onset, and serum creatinine concentration). For the mRS at discharge, urinary GFAP was the most predictive covariate (Exp(B) [95% CI]: 1.005 [1.00–1.01]); however, it did not remain significant after correction for multiple testing (p = 0.049, q = 0.07). Urinary GFAP also showed a higher χ2 compared to serum GFAP (χ2 urine: 4.9, p = 0.027; χ2 serum: 1.08, p = 0.30). Serum and urinary NfL as well as urinary t-tau were not significantly associated with the mRS at discharge. For the mRS at 3 months, serum NfL was the only significant covariate (Exp(B) [95% CI]: 1.02 [1.00–1.04]; p = 0.02, q = 0.04) (Table 3). Across all multivariable ordinal regression models, NIHSS was the strongest predictor, showing consistently higher χ2 values and Exp(B) compared to any serum or urine biomarker. In a final regression model including both GFAP and NfL in serum and urine, urinary GFAP remained the most predictive biomarker for mRS at discharge, whereas serum NfL remained the most predictive for mRS at 3 months (Table 3).
Correlation and regression analysis of infarct size and hematoma volume
In the AIS patient group, urine and serum GFAP, serum UCH-L1 and serum t-tau correlated best with the ischemic lesion size (in ml as determined by volumetry, Table 4). In a subsequent linear regression model with correction to age at onset, NIHSS at admission and creatinine in serum, only GFAP in urine remained significant with an Exp(B) [95%-CI] of 1.56 [1.19–2.05] (p-value: 0.001, q-value: 0.007) (Table 5). The dependency between the GFAP concentration in urine and the infarct size is visualized using as a scatterplot (Fig. 3).
Scatterplot between the GFAP concentration (pg/ml) in urine and the infarct size in mL after acute ischemic stroke.
In the ICH group, a Spearman correlation revealed a significant, positive correlation between ICH volume and GFAP in serum (Spearman ρ: 0.75, q-value: 0.02) (Table 4). However, after inclusion into a multivariate linear regression model with NIHSS at admission, age at onset and creatinine in serum, only NIHSS at admission remained the most strongest and independent predictors of the ICH volume (Exp(B) 7.0 [1.84–26.68], q-value: 0.02) (Table 5).
Prediction of mortality
In the ROC curve analysis of the individual urine and serum biomarker levels for the prediction of in-hospital mortality (Table 6), we identified NfL in urine as well as tau in serum and urine as significant markers. For the prediction of mortality after 3 months, GFAP, NfL and t-tau in urine were significant predictors. In a combined ROC analysis with all significant biomarkers, NfL in urine as well as t-tau in urine and serum remained significant with an AUC of 0.77 [0.6–0.94] (p = 0.003) for NfL in urine, 0.79 [0.6–0.94] (p = 0.042) for t-tau in urine and 0.70 [0.51–0.9] (p = 0.042) for t-tau in serum. For the prediction of mortality after 3 months, only NfL in urine remained significant with an AUC of 0.74 [0.58–0.90] (p = 0.009) (Table 6). The ROC curves are visualized in Fig. 4.
ROC curve analysis of significant urine biomarkers for the prediction of in-hospital mortality (left) and mortality at 3 months (right).
Discussion
Our study offers insights into the utility of urine-based biomarkers for predicting outcomes following acute cerebral damage. Specifically, our findings suggest that NfL, t-tau and GFAP, when detected in urine, could serve as valuable indicators of neurological and tissue outcomes. To our knowledge, this is the first study to analyse the functional outcome after AIS or ICH based on neuronal or glial biomarkers in urine.
Among the urinary biomarkers, absolute GFAP concentration showed the strongest predictive value for mRS at discharge and, to a lesser extent, at 3 months following AIS or ICH. Its performance was superior to serum GFAP within the same time frame, as indicated by higher χ2 values for model fit (Table 3). In contrast, although NfL correlated significantly with mRS at both time points, these associations did not remain significant after adjustment for confounders and were inferior to urinary GFAP for predicting in-hospital- and 3-month functional outcomes. Regarding serum biomarkers, only serum NfL remained a significant predictor of 3-month mRS, where it outperformed urinary NfL concentrations.
When models were adjusted for established risk factors, NIHSS at admission remained the strongest predictor, surpassing all biomarker measurements. Nevertheless, urinary GFAP contributed additional and independent predictive value for in-hospital functional outcome, while serum NfL independently predicted 3-month outcome (Table 3). For in-hospital mortality, urinary NfL as well as serum and urinary t-tau were significant predictors, whereas only urinary NfL remained significant for 3-month mortality (Table 6).
Surprisingly, the serum biomarker concentrations only showed a weak correlation with the in-hospital mortality and mortality after 3-months, which especially after inclusion into a multivariate regression model remained in-significant for all biomarkers and did not add additional value for the prediction of functional outcomes. A possible explanation might be the time of sample acquisition, which allowed later times compared to similar studies8,17,18. Furthermore, using the biomarker-creatinine ratio (which is supposed to be a more valid and accurate method as the values are corrected for urine dilution19), did not provide additional predictive value compared to the absolute urine biomarker concentrations. A possible explanation is that absolute biomarker concentrations in urine may increase in the presence of glomerular impairment or highly concentrated urine (e.g., due to dehydration or renal failure), both of which are also associated with poorer functional outcomes after ischemic stroke20. In such cases, elevated absolute urine biomarker levels might reflect not only the extent of brain injury but also acute kidney failure. Adjusting for these factors through a biomarker-to-creatinine ratio may therefore diminish the predictive value of absolute concentrations. To partially account for this, we included serum creatinine levels in the multivariate regression model; however, the exact contribution of acute renal failure was not fully evaluated in our study.
In patients with AIS, GFAP in urine showed the best correlation with the ischemic lesion size and is in line with previous data about GFAP in serum as a marker of astroglial injury9 to assess tissue damage in brain injuries4,21. In this context, the detectability in urine11 represents an intriguing finding, which adds to the existing literature about GFAP following astrocytic injury22,23 and could assist clinicians in assessing tissue-specific outcomes following AIS.
We did not find significant differences of the absolute urine biomarker concentrations between the respective underlying etiologies, which strengthens our approach of a composite group of AIS and ICH for the statistical outcome analysis. On the downside, a distinction between ICH and AIS could not be made based solely on the urine biomarker determination alone within the chosen timeframe of sample collection. In particular, a differentiation between ICH and AIS is especially possible within the first 6 h after symptom onset with a subsequent alignment of the serum levels between the etiologies24. In our study, only a minority of our blood and serum samples were drawn within 24 h after symptom onset, none within 6 h. In this context, the estimated serum peak, converging between AIS and ICH in the chosen timeframe, was selected to improve prognostic assessment, accepting a lower threshold to distinguish between the two entities. It is worth noting, that, as expected, there was a strong correlation between GFAP in serum and ICH volume, but contrary to the correlation in AIS this could not be identified in urine. Besides the lower number of cases, a different release of GFAP into blood and subsequently into urine could be assumed as possible explanations for this finding.
In clinical practice, the assessment of stroke severity und thus outcome prediction can be challenging in the subacute phase following an acute stroke as concomitant extracerebral diseases and systemic (such as infections) or cerebral complications (such as epileptic seizures) may contribute to a worse outcome. Urine biomarkers offer advantages in terms of accessibility, safety, and the ability to conduct repeated assessments without invasive procedures. This could be particularly beneficial in populations where blood draws are challenging, such as pediatric or elderly patients as well as regions with poor medical infrastructure. Despite the sensitivity and specificity of urine biomarkers in this study were moderate, they were overall superior to the respective predictive value of serum in our cohort. However, prior publications, showed sligthly higher AUC values for predicting short term mortality and functional outcomes (ranging from 0.78 to 0.88 for GFAP, 0.681–0.812 for NfL, 0.81–0.76 for t-tau and 0.86–0.89 for UCH-L1)17,25,26,27,28,29. In our study, the AUC values for predicting mortality (ranging from 0.68 to 0.74 for GFAP, NfL, and t-tau) suggest that urine biomarkers can provide valuable information, but they may not yet be a standalone diagnostic tool. Given the moderate sensitivity and specificity observed, urine biomarkers may be best used in conjunction with clinical assessments and imaging, rather than as a replacement for them. With ongoing advances in imaging techniques, many new imaging biomarkers have gained attention in recent years. These have been shown to correlate with clinical and tissue outcomes and can assist in determining the penumbra, thereby influencing treatment decisions30,31. The combination of imaging and classical serum biomarkers is already used in other areas, and will be increasingly applied in in stroke, yielding enhanced diagnostic and prognostic accuracy32,33,34.
The variability in biomarker levels among patients and across different types of brain injury highlights the complexity of interpreting urine biomarkers. Furthermore, pre-existing neurological diseases, acute infections, kidney function, and concomitant medication can all influence biomarker levels in acute stroke patients. Future studies should thus also explore the potential confounding factors that may affect the excretion of these biomarkers in urine, such as renal function, hydration status, and age. A more comprehensive understanding of the dynamics of these biomarkers in urine, including their stability, half-life, and elimination pathways, will be essential for optimizing their use in clinical practice. Additionally, longitudinal studies tracking urine biomarker levels over time could provide insights into the temporal progression of cerebral damage and recovery. A more detailed assessment of biomarker kinetics would therefore ideally require measurements at several time points, such as 24, 48, 72, and 96 h this would help to assess whether a sustained increase might correlate with clinical severity.
In addition, the data was collected within 96 h during hospitalization and therefore were performed outside the normal time window for treatment decisions. The benefit of urine biomarker measurements in the hyperacute phase to direct therapeutic approaches therefore remains unclear and requires further investigation in the future. In this case, it would be of particular interest to what extent urine biomarkers can be used to differentiate between AIS and ICH, or whether there is a correlation with the early extent of infarction or ICH.
A weakness of this study is the relatively small group size, especially in the evaluations for lesion size (tissue outcome), as the ICH group was generally much smaller, resulting in a limited statistical power. However, given that we found significant and plausible effects for many of the parameters of interest, this pilot study offers a foundation for larger studies.
Our study evaluated neuro-glial biomarkers in serum and urine within 96 h of symptom onset for predicting in-hospital and 3-month functional outcomes in patients with AIS or ICH. Among the biomarkers, urinary GFAP showed the strongest predictive value for in-hospital functional outcome, which was higher than the respective serum concentrations and remained independently associated with outcome alongside NIHSS at admission. While urinary NfL showed a trend toward additional predictive value for 3-month outcome, this effect was stronger and statistically significant for serum NfL. For mortality prediction, urinary NfL was the only biomarker consistently associated with both in-hospital and 3-month outcomes. In conclusion, urinary GFAP and NfL may serve as potential non-invasive biomarkers for prognostic assessment in acute stroke, possibly providing additional information beyond established clinical risk factors.
Data availability
Data is provided within the manuscript or supplementary information files. Source data will be made available upon reasonable request to the corresponding author (Dr. Kohlhase, kohlhase@med.uni-frankfurt.de).
References
Montellano, F. A. et al. Role of blood-based biomarkers in ischemic stroke prognosis. Stroke 52, 543–551 (2021).
Khalil, M. et al. Neurofilaments as biomarkers in neurological disorders—Towards clinical application. Nat. Rev. Neurol. 20, 269–287 (2024).
Pekny, M. et al. Neurofilament light chain (Nfl) in blood—A biomarker predicting unfavourable outcome in the acute phase and improvement in the late phase after stroke. Cells https://doi.org/10.3390/cells10061537 (2021).
Ren, C. et al. Assessment of serum UCH-L1 and GFAP in acute stroke patients. Sci. Rep. 6, 1–9 (2016).
Bitsch, A. et al. Serum tau protein level as a marker of axonal damage in acute ischemic stroke. Eur. Neurol. 47, 45–51 (2002).
Luger, S. et al. Diagnostic accuracy of glial fibrillary acidic protein and ubiquitin carboxy-terminal hydrolase-L1 serum concentrations for differentiating acute intracerebral hemorrhage from ischemic stroke. Neurocrit. Care 33, 39–48 (2020).
Stanne, T. M. et al. Association of plasma brain-derived tau with functional outcome after ischemic stroke. Neurology 102, e209129 (2024).
Luger, S. et al. Role of S100B serum concentration as a surrogate outcome parameter after mechanical thrombectomy. Neurology 97, e2185–e2194 (2021).
Abdelhak, A. et al. Blood GFAP as an emerging biomarker in brain and spinal cord disorders. Nat. Rev. Neurol. 18, 158–172 (2022).
Kamtchum-Tatuene, J. & Jickling, G. C. Blood biomarkers for stroke diagnosis and management. Neuromol. Med. 21, 344–368 (2019).
Kohlhase, K. et al. Brain-specific biomarkers in urine as a non-invasive approach to monitor neuronal and glial damage. Eur. J. Neurol. 30, 729–740 (2023).
Herrmann, M., Vos, P., Wunderlich, M. T., de Bruijn, C. H. M. M. & Lamers, K. J. B. Release of glial tissue-specific proteins after acute stroke. Stroke 31, 2670–2677 (2000).
Wunderlich, M. T., Wallesch, C. W. & Goertler, M. Release of glial fibrillary acidic protein is related to the neurovascular status in acute ischemic stroke. Eur. J. Neurol. 13, 1118–1123 (2006).
Schwab, S. J., Christensen, R. L., Dougherty, K. & Klahr, S. Quantitation of proteinuria by the use of protein-to-creatinine ratios in single urine samples. Arch. Intern. Med. 147, 943–944 (1987).
von Kummer, R. et al. The Heidelberg bleeding classification. Stroke 46, 2981–2986 (2015).
Saver, J. L. et al. Standardized nomenclature for modified Rankin scale global disability outcomes: Consensus recommendations from stroke therapy academic industry roundtable XI. Stroke 52, 3054–3062 (2021).
Uphaus, T. et al. NfL (neurofilament light chain) levels as a predictive marker for long-term outcome after ischemic stroke. Stroke 50, 3077–3084 (2019).
Ferrari, F. et al. Quantification and prospective evaluation of serum NfL and GFAP as blood-derived biomarkers of outcome in acute ischemic stroke patients. J. Cereb. Blood Flow Metab. 43, 1601–1611 (2023).
Waikar, S. S., Sabbisetti, V. S. & Bonventre, J. V. Normalization of urinary biomarkers to creatinine during changes in glomerular filtration rate. Kidney Int. 78, 486–494 (2010).
El Husseini, N. et al. Association of kidney function with 30-day and 1-year poststroke mortality and hospital readmission. Stroke 49, 2896–2903 (2018).
Barba, L. et al. Serum β-synuclein, neurofilament light chain and glial fibrillary acidic protein as prognostic biomarkers in moderate-to-severe acute ischemic stroke. Sci. Rep. 13, 20941 (2023).
Vollmuth, C. et al. Incremental value of serum neurofilament light chain and glial fibrillary acidic protein as blood-based biomarkers for predicting functional outcome in severe acute ischemic stroke. Eur. Stroke J. 9, 751–762 (2024).
Meier, S. et al. Serum glial fibrillary acidic protein compared with neurofilament light chain as a biomarker for disease progression in multiple sclerosis. JAMA Neurol. 80, 287–297 (2023).
Luger, S. et al. Glial fibrillary acidic protein serum levels distinguish between intracerebral hemorrhage and cerebral ischemia in the early phase of stroke. Clin. Chem. 63, 377–385 (2017).
Liu, G. & Geng, J. Glial fibrillary acidic protein as a prognostic marker of acute ischemic stroke. Hum. Exp. Toxicol. 37, 1048–1053 (2018).
Rattanawong, W., Ongphichetmetha, T., Hemachudha, T. & Thanapornsangsuth, P. Neurofilament light is associated with clinical outcome and hemorrhagic transformation in moderate to severe ischemic stroke. J. Cent. Nerv. Syst. Dis. 15, 11795735221147212 (2023).
Correia, M. et al. Early plasma biomarker dynamic profiles are associated with acute ischemic stroke outcomes. Eur. J. Neurol. 29, 1630–1642 (2022).
Korley, F. K. et al. Prognostic value of day-of-injury plasma GFAP and UCH-L1 concentrations for predicting functional recovery after traumatic brain injury in patients from the US TRACK-TBI cohort: An observational cohort study. Lancet Neurol. 21, 803–813 (2022).
Wu, J. et al. Plasma neurofilament light chain: A biomarker predicting severity in patients with acute ischemic stroke. Medicine (United States) 101, E29692 (2022).
Uchida, Y. et al. Longitudinal changes in iron and myelination within ischemic lesions associate with neurological outcomes: A pilot study. Stroke 55, 1041–1050 (2024).
Uchida, Y. et al. Penumbra detection with oxygen extraction fraction using magnetic susceptibility in patients with acute ischemic stroke. Front. Neurol. 13, 752450 (2022).
Wu, M. et al. A study on structural imaging changes and serum neurofilament light chain (NfL) levels in individuals with white matter hyperintensities, combining imaging techniques with biomarker analysis. IBRO Neurosci. Rep. 18, 794–802 (2025).
Huang, X.-H. et al. Association of serum neurofilament light and glial fibrillary acidic protein with cerebral small vessel disease in nondemented middle-to-old aged population. Transl. Stroke Res. https://doi.org/10.1007/s12975-025-01359-9 (2025).
Prinse, F. A. M. et al. Investigating the role of neuroinflammation and brain clearance in frontotemporal lobar degeneration using 7T MRI and fluid biomarkers: Protocol for a cross-sectional study in a tertiary care setting. BMJ Open 15, e102668 (2025).
Acknowledgements
We thank Dr. Franziska Frank, Dr. Christian Wilmes and Dr. Kimberly Koerbel for the help with the sample collections.
Funding
Open Access funding enabled and organized by Projekt DEAL.
Author information
Authors and Affiliations
Contributions
FL and KK conceived the idea and designed the study. MR evaluated and exctracted the imaging data. FL and DTM processed the data, performed the statistical analysis and designed the figures. FL drafted the manuscript. CF, CG and JHS aided in interpreting the results and made critical revision of the manuscript for important intellectual content. All authors discussed the results and commented on the manuscript. KK supervised the work.
Corresponding author
Ethics declarations
Competing interests
Prof. Foerch holds the patent for using GFAP for identification of intracerebral hemorrhage (US20150247867) and further reports speaker honoraria and honoraria for participating in advisory boards from Abbott, Alexion, Bristol Myers Squibb, Novartis, Teva, Merck, Sanofi Genzyme, and Roche. The university hospital Frankfurt received a research grant from Merck Healthcare Germany GmbH, Weiterstadt, Germany, an affiliate of Merck KGaA (CrossRef Funder ID: 10.13039/100009945) for the development and validation of brain-specific biomarkers in urine. K. Kohlhase and C. Foerch were responsible for the financial distribution of the research grant. However, the measurements presented in this manuscript were not directly supported or funded by this research grant and therefore had no influence on the study design, data acquisition, data analysis, interpretation of results, or manuscript preparation. Dr. Bohmann reports speaker honoraria from Laerdal, AstraZeneca, Bristol-Myers-Squibb, Pfizer, Medtronic and grants and personal fees from Alexion, AstraZeneca, Stryker Neurovascular, Boehringer Ingelheim. Dr. Schaefer reports travel reimbursements by Bayer AG outside of the submitted work. The other authors report no conflicts.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Lieschke, F., Marggrander, D.T., Rauch, M. et al. The use of brain-specific biomarkers in urine for prediction of neurological outcome and extent of tissue damage following stroke. Sci Rep 15, 43089 (2025). https://doi.org/10.1038/s41598-025-28115-1
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-28115-1