Abstract
Bismuth-containing compounds have attracted considerable interest in electrochemical energy storage owing to their distinctive layered structures and favorable redox features. However, systematic studies on bismuth oxybromide (BiOBr) as an electrode materials for supercapacitors remain limited, despite its promising layered architecture that an facilitate ion transport and offer abundant active sites. In this study, we report the solvothermal preparation of BiOBr nanostructures with a unique curved sheet-like morphology, specifically designed to improve electrochemical activity. The fabricated BiOBr electrode demonstrated a high capacity of 84.6 mAh/g at 1.1 A/g, along with notable cycling stability. Furthermore, the assembled symmetric supercapacitor exhibited a notable energy density (Ed) of 10 Wh/kg at a power density (Pd) of 750 W/kg, while retaining 98% of its original capacitance over 3000 continuous runs. These findings highlight the potential of BiOBr-based electrodes as effective and durable materials for next-generation supercapacitor technologies.
Similar content being viewed by others
Introduction
The growing global emphasis on sustainable and high-performance energy storage has driven significant interest in developing materials that can simultaneously deliver high power and energy densities1. Supercapacitors (SCs), known for their rapid charge–discharge ability, long operational lifetime, and superior power density (Pd), have become promising alternatives to bridge the performance gap between traditional capacitors and rechargeable batteries1,2. However, their relatively low energy density continues to limit widespread applications, motivating research into innovative electrode materials and structural designs to improve energy storage efficiency.
Among the various electrode materials explored, transition metal-based compounds, particularly bismuth-containing systems, have attracted increasing research interest owing to their multivalent oxidation states, rich redox activity, and strong electrochemical stability3. Bismuth oxybromide (BiOBr), in particular, has emerged as a capable candidate for energy-storage because of its tetragonal matlockite-type layered structure, in which alternating [Bi2O2]2+ and Br⁻ layers facilitate rapid ion transport and provide abundant active sites for redox reactions4,5. Recent studies indicate that controlling the morphology, crystallinity, and defect distribution of BiOBr nanostructures can substantially improve their specific capacitance, rate capability, and cycling durability4,5.
In this context, nanosheet architectures have become especially valuable for designing high-performance electrodes due to their ultrathin geometry and enlarged surface exposure. Such two-dimensional frameworks shorten ion-diffusion distances, increase the density of accessible active sites, and promote intimate contact between the electrode and electrolyte, thereby accelerating charge-transfer kinetics and improving long-term capacitance retention6,7. For example, nanosheets based on layered double hydroxides (LDHs) and transition-metal oxyhalides have demonstrated enhanced conductivity and faster ion migration compared to bulk materials, largely attributed to their large lateral dimensions and atomic-level thinness8,9. The inherent properties of nanosheets, flexible stacking, mechanical robustness, and efficient electron pathways, make them well-suited for high-power, long-life supercapacitor systems8,9. In the present work, the curved BiOBr nanosheet morphology is engineered to exploit these advantages, providing a large surface-to-volume ratio, minimized diffusion barriers, and efficient charge transport characteristics that collectively enhance both Ed and Pd capability in the developed symmetric supercapacitor device.
The controlled synthesis of BiOBr nanostructures has attracted significant devotion in recent years owing to their tunable morphology and promising electrochemical behavior3,10. Among various approaches, hydrothermal synthesis has proven to be particularly effective for producing BiOBr with adjustable crystallinity and microstructure. For instance, Kumari et al.11 described a one-pot hydrothermal route for the preparation of nanostructured bismuth fluoride (BiF3) designed for symmetric supercapacitor applications, achieving a high capacitance of 717.3 F/g at 1 A/g along with notable cycling durability. Although their study focused on BiF3, the results highlight the general suitability of hydrothermal techniques for developing bismuth-based nanostructures with enhanced charge-storage capability. Recent progress in electrode material engineering has further expanded the performance of bismuth compounds in aqueous systems. Qin et al.12 introduced a halogen-doping strategy by incorporating iodine into the BiOBr lattice, yielding a BiOBr0.87I0.13 composition. The introduction of iodine optimized the (110) crystal orientation, promoting direct ion diffusion channels and stronger interactions with hydroxide ions. As a result, the altered electrode revealed an impressive capacity of 187.6 mAh/g at 1 A/g and preserved 153.1 mAh/g at 4 A/g, demonstrating admirable rate capability and reversibility. This finding emphasizes that halogen modification can effectively tune the interlayer spacing and electronic conductivity of BiOBr-type materials, offering new possibilities for high-efficiency energy storage. Similarly, Thanur et al.13 developed a BiOCl electrode that displayed remarkable electrochemical properties, with a capacity of 501 C/g at 0.5 A/g and 80% retention over 2,500 runs. When employed in a symmetric device, the BiOCl-based system delivered an Ed of 22 Wh/kg at 773 W/kg, demonstrating the practical feasibility of Bi-based nanostructures for advanced supercapacitor configurations.
In spite of these advancements, challenges persist in optimizing the electrochemical performance of BiOBr-based supercapacitors. Issues such as limited electrical conductivity, structural degradation during cycling, and scalability of synthesis methods need to be addressed. Strategies to overcome these challenges include the incorporation of conductive additives, the exploration of novel synthesis routes to enhance the structural integrity and electrochemical features of BiOBr nanostructures. This investigation is intended to contribute to the ongoing efforts in refining the performance of BiOBr-based supercapacitors by investigating the solvothermal synthesis of curved sheet-like BiOBr nanostructures and evaluating their electrochemical properties in symmetric supercapacitor configurations. The vision grown from this research are anticipated to advance the understanding of BiOBr nanostructures in energy storage uses and inform the development of high-performance supercapacitor platforms.
Experimental section
Synthesis of BiOBr nanosheets
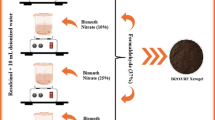
BiOBr nanosheets were synthesized via a solvothermal method involving ammonium bromide (NH4Br) as the bromide source. Initially, 8 mM of Bi(NO3)3 5H2O was added in 100 mL of ethylene glycol, preheated to 120 °C. After stirring the solution for 30 min, 30 mM of NH4Br was introduced, and the mixture was further stirred. This was followed by an additional 30 min of vigorous stirring at ambient temperature. Subsequently, 300 mL of isopropanol was added to the mixture. The resulting clear solution was shifted into Teflon-lined stainless-steel autoclaves and exposed to a solvothermal reaction at 170 °C for 15 h. The resulting yellow product was collected, thoroughly washed with ethanol, and dried at 80 °C overnight.
Electrochemical testing
Electrochemical performance was evaluated using a Biologic SP-200 system configured with a standard three-electrode setup. The prepared BiOBr sample, coated onto a conductive substrate, acted as the working electrode. A Hg/HgO electrode served as the reference electrode and a platinum mesh functioned as the counter electrode. CV was performed from − 1.0 to 0.6 V with scan rates ranging from 2 to 200 mV/s. GCD measurements were also conducted in 1 M KOH over the same potential window (vs. Hg/HgO) at several current densities. EIS was verified at open-circuit potential in the frequency range of 200 kHz to 100 mHz. All tests were carried out in 1 M KOH electrolyte. The mass of the electrode is 1.1 mg.
Preparation of gel electrolyte (PVA/KOH)
The alkaline gel electrolyte was made by mixing 3 g of poly(vinyl alcohol) (PVA) in 24 mL of deionized water under vigorous stirring at 90 °C until a transparent viscous solution was obtained after about 1 h. Separately, 3 g of KOH was mixed in 6 mL of H2O and then gradually mixed to the PVA solution under constant stirring until a homogeneous gel formed. The mixture was permitted to cool to room temperature before use.
Symmetric device fabrication
For an aqueous symmetric supercapacitor, the BiOBr was used as both positive and negative electrodes. The wide potential of 0–1.5 V was applied for the two-electrode system and gel electrolyte was used as the electrolyte. The working electrode was fabricated by combining of 80 wt% active material, 10 wt% carbon black, and 10 wt% PVDF in NMP to form a homogeneous slurry, which was then uniformly coated onto a 12 mm-diameter nickel foam substrate. The paste was then coated on the respective electrodes, and heated at 80 °C for 12 h. Specific capacity (Cs) in mAh/g from GCD curves can also be calculated as
where, ‘I’ is current intensity and ‘∆t’ is discharge time (t) of the experimental charge–discharge curve.
Further Ed (Wh/kg) and Pd (W/kg) of the electrode were recorded from the GCD profiles using the following equations:
Characterization
The phase composition of the synthesized materials was examined by XRD on a PANalytical X’pert Pro MRD instrument equipped with Cu Kα radiation (λ = 1.5406 Å). Surface morphology was characterized by FE-SEM (model 4800) operated at an acceleration voltage of 10 kV. Chemical states and elemental distribution were probed using XPS instrument (ThermoFisher). For high-resolution TEM, the powders were ultrasonically dispersed in ethanol and then drop-cast onto a 400-mesh copper grid (3.5 mm diameter). Imaging was carried out using a Hitachi H-7600 TEM operated at an applied voltage of 120 kV.
Results and discussion
XRD study was employed to explore the crystal structure of the fabricated BiOBr nanosheet, and the results confirmed the successful formation of a highly crystalline tetragonal phase. Figure 1b depicts the XRD profile of BiOBr. The diffraction peaks observed in the XRD pattern correspond well with the standard card JCPDS No. 09–0393, which is characteristic of tetragonal bismuth oxybromide (BiOBr)14. Notably, the most intense diffraction peak appears around 2θ ⁓ 31.7°, which is indexed to the (102) plane. This dominant orientation suggests a preferred growth direction of the BiOBr nanosheets along the (102) crystallographic plane. Additional prominent peaks at approximately 2θ = 10.8°, 25.1°, 32.2°, 46.1°, and 57.1° are associated with the (001), (101), (110), (200), and (212) planes, individually, indicating the polycrystalline nature of the sample with well-defined structural order14,15. The sharpness and intensity of the diffraction peaks imply good crystallinity of the synthesized material16. Moreover, no secondary phases or impurity peaks were detected, further confirming the phase purity of the BiOBr nanosheets. Using the most intense diffraction peak corresponding to the (102) plane at 31.8°, the average crystallite size was determined to be around 28 nm. The relatively small crystallite size facilitates a high surface-to-volume ratio of the nanosheets.
(a) Schematic representation of BiOBr nanosheets synthesis procedure; and (b) XRD pattern of BiOBr nanosheets.
The surface morphology of the synthesized BiOBr was examined with FESEM, as presented in Fig. 2a-d. The micrographs reveal a hierarchical architecture composed of densely packed nanosheets and microplate-like structures. From Fig. 2a, b, the BiOBr material appears to consist of interconnected flake-like particles forming flower-like assemblies. These nanosheet structures are uniformly distributed with no visible agglomeration, suggesting a well-dispersed morphology ideal for electrolyte accessibility in electrochemical uses. At higher magnification (Fig. 2c, d), the ultrathin nature of the nanosheets and highlights their curved edges and layered stacking. The thinness and uniformity of these sheets are clearly visible, and the layered structure of BiOBr is evident. This nanostructured morphology is beneficial for charge transfer, as it offers short diffusion paths and an abundance of electroactive sites.
(a–e) FESEM images of BiOBr at different magnifications, revealing nanosheet morphology; (f–h) elemental mapping images for Bi, Br, and O, respectively; (i) composite elemental distribution map; (j) EDX spectrum confirming the presence of Bi, Br, and O; and (k) quantitative elemental composition of BiOBr based on EDX analysis.
Figure 2e displays the high-resolution FESEM micrograph of the BiOBr particles, revealing aggregated nanosheets with a flake-like morphology. To investigate the uniformity of elemental dispersion across these nanosheets, elemental mapping was conducted. Figure 2f-h correspond to the elemental mappings of bismuth (Bi), bromine (Br), and oxygen (O), respectively. The distribution maps confirm that all three elements are homogeneously spread throughout the scanned region, indicating the successful and uniform synthesis of BiOBr without any observable phase segregation or elemental clustering. Additionally, the color-coded elemental overlay (Fig. 2i) visually reinforces the elemental homogeneity in the selected spectrum region. The compositional analysis (Fig. 2j) further supports this observation, showing that the sample consists of 60.39 wt% Bi, 25.17 wt% Br, and 14.44 wt% O. The atomic percentages are calculated as 19.18% for Bi, 20.90% for Br, and 59.92% for O, closely aligning with the stoichiometric ratio expected for BiOBr. The absence of impurities and the balanced distribution of Bi, Br, and O validate the high purity and phase consistency of the synthesized BiOBr nanomaterial.
Figure 3a-f illustrates the HRTEM images of the BiOBr, providing detailed insight into the structural characteristics and crystallinity of the synthesized BiOBr nanosheets. Figure 3a-c captured at progressively higher magnifications (50–10 nm scales) clearly reveal the ultrathin, sheet-like morphology of the BiOBr nanostructures. These images highlight the presence of transparent and overlapping nanosheets, suggesting their two-dimensional nature and high surface area, an advantageous trait for charge transfer and ion diffusion in energy storage or photocatalytic systems. Figure 3d, fine lattice fringes begin to emerge, indicating the presence of well-crystallized regions within the sheets. Moving to Fig. 3e, these lattice fringes are more distinctly resolved. A measured interplanar spacing of approximately 0.28 nm is evident, which links to the (101) crystallographic plane of tetragonal BiOBr, affirming the crystalline integrity of the synthesized material. Furthermore, the SAED image in Fig. 3f shows distinct concentric diffraction rings accompanied by bright spots, which can be indexed to the (110), (001), and (101) planes of BiOBr14,16. These features provide clear evidence of the polycrystalline character of the nanosheets, consistent with the XRD findings. The distribution and sharpness of the rings reflect the high degree of structural order within the sample. Figure 3h illustrates the raw HRTEM fringe pattern, which clearly shows periodic lattice fringes aligned in a parallel fashion, signifying the crystalline nature of the material. The interplanar distance between these fringes, measured along the line scan, is approximately 0.28 nm, as highlighted in Fig. 3k. This spacing corresponds to the (101) crystallographic plane of tetragonal BiOBr, consistent with previous XRD findings14. The corresponding intensity profile (Fig. 3g) extracted from the HRTEM fringes further confirms the regular spacing between lattice planes. The peaks in the intensity profile reflect periodic changes in image contrast caused by the interference of electron waves diffracted by the atomic planes, validating the presence of a well-defined (101) lattice orientation. Figure 3i displays an FFT result, where atomic columns are visualized as bright spots, confirming high crystallinity at the atomic scale. Complementing this, the FFT pattern (Fig. 3j) exhibits sharp and symmetrical diffraction spots, further verifying the single-crystalline domain and its well-ordered atomic arrangement. Overall, the fringe analysis strongly supports the presence of highly ordered, crystalline BiOBr with distinct (101) lattice planes.
(a–c) Low- and medium-resolution TEM images of BiOBr nanosheets, revealing their ultrathin layered morphology; (d–e) HRTEM images highlighting clear lattice fringes with an interplanar spacing of 0.28 nm, corresponding to the (101) plane; (f) SAED pattern displaying distinct diffraction rings indicative of the polycrystalline nature; (g) Intensity profile extracted from HRTEM, validating the measured d-spacing; (h) Filtered HRTEM image showing periodic lattice structure; (i) FFT spot pattern derived from the HRTEM region; (j) IFFT image used for precise fringe visualization; (k) Reconfirmation of 0.28 nm spacing from the inverse FFT image, consistent with the (101) plane of BiOBr.
The full XPS survey scan of the BiOBr nanosheets is displayed in Fig. 4a. The high-resolution band of Bi 4f. (Fig. 4b) reveals two distinct peaks situated at 159.2 eV and 164.5 eV, which are related to Bi 4f7/2 and Bi 4f5/2, correspondingly17. These values are characteristic of the Bi3+ oxidation state, affirming the trivalent mode of bismuth in the BiOBr matrix. Similarly, the Br 3d spectrum (Fig. 4c) depicts two symmetric peaks positioned at approximately 68.48 eV and 69.6 eV, allotted to Br 3d5/2 and Br 3d3/2, respectively18. These binding energies indicate the presence of Br⁻, consistent with the chemical structure of BiOBr. The O 1 s spectrum of the BiOBr nanosheets (Fig. 4d) was deconvoluted into three components centered at approximately 530.2, 531.2, and 532.7 eV. The main feature at ~ 530.2 eV related to lattice oxygen (O2−) bonded within the [Bi2O2]2+ layers of the BiOBr crystal structure, consistent with previously reported Bi-O environments in BiOX compounds17,18,19,20,21. The additional component at ~ 531.2 eV arises from surface-adsorbed oxygen species linked with oxygen vacancies or defect sites, indicating the presence of moderate lattice distortion and active surface centers. The weak high-binding-energy shoulder at ~ 532.7 eV is assigned to chemisorbed hydroxyl groups or adsorbed water molecules. The relative peak intensities ) reveal a balanced combination of structural integrity and surface reactivity16,22. These findings confirm that the solvothermally synthesized BiOBr nanosheets possess a chemically uniform yet defect-rich surface, enabling efficient electrochemical activity and contributing to the high capacitance and cycling stability of the symmetric supercapacitor device.
XPS spectra (a) survey scan, (b) Bi4f., (c) Br 3d and (d) O1s of BiOBr nanosheets.
The electrochemical characteristics of the synthesized BiOBr nanosheets were thoroughly evaluated using a 3-electrode setup in 1 M KOH electrolyte, as depicted in Fig. 5a-d. Figure 5a illustrates the cyclic voltammetry curves recorded at several scan rates extending from 0.5 to 200 mV/s. The CV profiles display distinct redox peaks, indicating a faradaic charge storage mechanism governed by reversible redox reactions of Bi3+/Bi0 or BiOBr surface transformations23. With increasing scan rates, the redox peak currents rise steadily without significant distortion in shape, suggesting good rate capability and robust electrochemical kinetics10,23. Figure 5b presents the GCD curves of the BiOBr electrode recorded at several current densities ranging from 1.1 to 8.8 A/g. The curves exhibit non-linear charge–discharge characteristics, confirming the pseudocapacitive nature of the material. A gradual decrease in discharge time with increasing current density is observed, which can be qualified to restricted ion diffusion and polarization effects at higher rates. Nevertheless, the retention of a substantial portion of the capacitance even at elevated current densities reflects the notable rate capability and efficient electron/ion transport within the BiOBr framework. Unlike the ideal triangular profiles typical of purely capacitive systems, the BiOBr electrode displays several distinctive features: (i) a small instantaneous voltage drop at the onset of discharge (iR drop) that becomes more pronounced at higher currents, indicating finite internal resistance due to contact and electrolyte contributions; (ii) a mild curvature and subtle shoulder in the mid-potential region, suggestive of surface redox reactions coupled with double-layer formation; and (iii) increased deviation from linearity at high currents, implying diffusion-controlled ion transport within the nanosheet layers.
Electrochemical performances: (a) CV profiles, (b) GCD curves, (c) specific capacity vs current density and (d) EIS spectra of BiOBr nanosheets.
In Fig. 5c, the specific capacity plotted against current density reveals that the BiOBr electrode delivers a high initial specific capacity (e.g., 84.6 mAh/g at 1.1 A/g), which gradually decreases with increasing current but remains substantial, indicating commendable capacity retention and structural stability under variable operative conditions. The calculated capacity of BiOBr nanostructure is 84.6, 53.5, 45.22, 41.5, and 32.7 mAh/g at 1.1, 2.2, 3.3, 4.4, and 8.8 A/g, respectively. Table 1 provides a comparison between previously reported Bi-based electrodes and the BiOBr nanosheets studied in this work, showing that the nanosheets deliver a reasonable electrochemical capacity relative to other Bi-based materials. For instance, Vadivel et al.24 solvothermally synthesized BiOXCO3-5 attained a highest capacitance of 645 F/g at 1 A/g, highlighting its notable electrochemical activity. Similarly, William et al.5 reported that the BiFeO3/Bi2Fe4O9 electrode delivered a highest capacity of 445 C/g at 2 mA/cm2, maintaining 82% of its initial capacity over 2,000 cycles, signifying notable electrochemical stability.
Figure 5d confirms the EIS spectra of the BiOBr electrode, showing a small semicircle at large frequency region and a straight line at low frequency region. The extracted values for solution resistance (Rs), charge-transfer resistance (Rct), and Warbug impedance (W) are approximately 1.55 Ω, 10.44 Ω, and 18.06 Ω.s−1/2 respectively. This indicates low charge transfer resistance and efficient ion diffusion, confirming good electrical conductivity and favorable capacitive behavior4,10.
The charge storage mechanism of the BiOBr electrode was further investigated by analyzing the connection between peak current vs scan rate. As shown in Fig. 6a, the log–log plot of peak current vs scan rate yields a slope, which is much closer to 0.5. This result suggests that the electrochemical response of BiOBr is largely diffusion-controlled, characteristic of battery-type behavior, rather than being dominated by surface-limited capacitive processes. To provide a more quantitative insight, Dunn’s method was employed to separate the total current into capacitive and diffusion-controlled contributions (Fig. 6b). At low scan rates, the charge storage is primarily diffusion-driven, as ions have adequate time to penetrate the bulk or defect regions of BiOBr. However, as the scan rate rises, the capacitive contribution gradually rises, indicating the growing role of fast surface redox reactions on the nanosheet surface. A representative CV curve at 1 mV/s, shown in Fig. 6c, further illustrates this decomposition. The diffusion-controlled portion aligns closely with the experimental profile, confirming the validity of the analysis. diffusion contributions suggests that the layered BiOBr nanosheet morphology not only provides abundant surface sites for rapid ion adsorption/desorption but also facilitates short-path OH⁻ diffusion into near-surface regions. Such a hybrid storage mechanism accounts for the enhanced electrochemical performance of BiOBr-based electrodes.
(a) log (peak current) versus log (scan rate) plot for BiOBr electrode, (b) Capacitive and diffusive contributions at different scan rates, (c) Representative CV curve at 1 mV/s with separated total and diffusive contribution, and (d) plausible electrochemical mechanism of BiOBr nanosheets.
The FESEM and TEM analyses reveal that the BiOBr sample consists of thin, sheet-like domains arranged in a stacked configuration. Layered nanosheet architectures are frequently reported to be beneficial for electrochemical electrodes because the exposed sheet faces and edges provide abundant electrochemically active interfaces and the loose inter-layer spacing offers unobstructed pathways for ion migration during cycling25,26. As illustrated in Fig. 6d, the BiOBr nanosheets participate in reversible surface redox reactions at the electrode–electrolyte boundary during charge–discharge. The short transport paths within the thin lamellae facilitate rapid electron transfer along the sheet plane, while the inter-sheet voids function as ion-buffering channels, supporting the insertion and extraction of OH⁻ without significant structural strain. These structural attributes collectively aid in suppressing polarization, accelerating interfacial kinetics, and sustaining charge storage over repeated cycles effects that align with previously reported behavior of layered bismuth oxyhalide systems25,26.
Figure 7a illustrates the schematic design of a symmetric supercapacitor fabricated using BiOBr nanosheets as both positive and negative electrodes (i.e., BiOBr//BiOBr), operating in a gel electrolyte with K+ and OH− ions mediating the charge storage process22. The CV curves in Fig. 7b reveal that the device maintains stable redox behavior across a range of potential windows (0.4–1.5 V), with the 1.5 V window demonstrating the most pronounced area under the curve, indicating enhanced charge storage. Figure 7c presents the CV response at numerous scan rates (2–500 mV/s), where the near-symmetric shape at higher scan rates confirms the device’s favorable rate capability and electrochemical reversibility. The GCD profiles shown in Fig. 7d at diverse current densities (1–8 A/g) display non-linear charge/discharge behavior, suggesting diffusive contributions. Additionally, the plot in Fig. 7e shows the capacitance decreasing with increased current densities, a typical trend due to limited ion diffusion at higher currents. The BiOBr//BiOBr symmetric device demonstrates specific capacitance values of 32, 14.67, 8.2, 6, 4.2, and 3 F/g when tested at 1, 2, 3, 4, 6, and 8 A/g, respectively.
(a) Schematic illustration of the symmetric BiOBr//BiOBr supercapacitor device; (b) CV curves recorded at various operating voltages; (c) CV curves at different scan rates; (d) GCD curves at varying current densities; and (e) plot of specific capacitance versus current density for the BiOBr//BiOBr device.
The Ragone plot (Fig. 8a) presents a graphical comparison between Ed (Wh/kg) and Pd (W/kg) for the symmetric BiOBr//BiOBr supercapacitor device. As observed, the device achieves a commendable Ed at low power output, maintaining its charge retention effectively. The BiOBr//BiOBr symmetric supercapacitor device delivers energy densities of 10, 4.6, 2.6, 1.9, 1.3, and 0.95 Wh/kg at corresponding power outputs of 750, 1500, 2250, 3000, 4500, and 6000 W/kg, respectively. As the power output increases, a gradual decline in Ed is observed, reflecting the expected behavior of electrochemical capacitors under high-rate conditions. This inverse relationship underscores the device’s strong rate capability, enabling it to sustain high power delivery with a manageable reduction in stored energy. These results reflect a competitive improvement in energy storage performance when compared with earlier reports17,27,28,29,30,31,32,33,34,35,36,37,38,39,40,41,42,43,44,45,46,47,48. Compared to representative symmetric devices (Table 2), our BiOBr//BiOBr device offers one of the candidates of longevity and moderate energy/power density, positioning it favorably among symmetric aqueous-electrolyte systems. For instance, Kumari et al.11 recorded an Ed of only 0.17 Wh/kg at 344 W/kg for a BiF3//BiF3 device. In another study, Karnam et al.37 achieved 20.8 Wh/kg at 4.1 kW/kg using a Bi2WO6-based symmetric configuration. Similarly, Shinde et al.38 reported that a BiO-polyaniline symmetric system offered an Ed of 79.42 Wh/kg at 583.5 W/kg. These comparisons highlight the balanced and enhanced performance of the BiOBr-based system, particularly in maintaining Ed across a wide range of power outputs. Complementing the Ragone plot, the 3D bar graph (Fig. 8b) offers a comprehensive visualization by incorporating discharge time (s) alongside energy and power densities. This tri-dimensional analysis provides deeper insight into the real-time operation of the device. Longer discharge durations (48 s) at lower power densities (750 W/kg) correlate with higher energy storage (10 Wh/kg), confirming efficient electrochemical behavior. As the Pd increases to 6000 W/kg, the Ed decreases to 0.95 Wh/kg, illustrating the typical trade-off between energy and power in a symmetric device. The gradual reduction in discharge time with increasing Pd emphasizes the fast-charge capability, reinforcing the device’s potential for applications requiring rapid energy delivery without substantial performance loss.
(a) Ragone plot illustrating the relationship between energy density and power density; (b) 3D plot showing energy density, power density, and discharge time; (c) cycling stability of the BiOBr//BiOBr device over multiple charge–discharge cycles; and (d) EIS spectrum of the device with an inset highlighting the high-frequency region.
The enduring electrochemical durability of the BiOBr//BiOBr symmetric device was evaluated through extended cycling at a constant current density. As shown in Fig. 8c, the device retained nearly 98% of its original capacitance over 3000 GCD runs, demonstrating notable cyclic stability and superior structural integrity. Additionally, the coulombic efficiency remained consistently high throughout the entire test, indicating efficient charge storage and minimal energy loss during repeated cycles. EIS analysis (Fig. 8d) provides valuable information on the charge-transport behavior of the BiOBr//BiOBr symmetric device. The fitted parameters show a Rs value of 10.82 Ω, Rct value of 2.641 Ω, and W is 404.3 Ω.s−1/2, indicating rapid ion migration and efficient electron conduction throughout the electrode–electrolyte interface. In the Nyquist graph, a small semicircular arc appears in the high-frequency zone, reflecting minimal charge-transfer resistance, followed by a nearly vertical line at low frequencies, which is characteristic of ideal capacitive behavior. Overall, these observations confirm that the BiOBr-based symmetric supercapacitor possesses notable electrochemical reversibility and low internal resistance.
Conclusion
In summary, BiOBr nanosheets have been effectively synthesized and systematically investigated as electrode materials for high-performance supercapacitors. Structural analysis confirmed their crystalline nature and purity, with XRD patterns aligning with the standard JCPDS card. Morphological evaluations using FESEM and HRTEM revealed well-defined nanosheet architectures, providing a high surface area favorable for electrochemical activity. Elemental mapping and EDX analysis confirmed the uniform distribution of Bi, O, and Br throughout the sample. XPS analysis further validated the chemical conditions and bonding environments of the constituent elements, affirming the material’s integrity. Electrochemical assessments in a 1 M KOH electrolyte demonstrated notable redox behavior, a high capacity of 84.6 mAh/g at 1.1 A/g with robust rate capability. The symmetric BiOBr//BiOBr device showcased remarkable energy-storage performance (10 Wh/kg at 750 W/kg), including high cyclic stability with nearly 98% retention over 3000 cycles and low internal resistance as confirmed by EIS studies. Overall, this study establishes BiOBr nanosheets as a promising and efficient electrode material for next-generation symmetric supercapacitors, combining structural stability, favorable charge transport, and long-term operational durability.
Data availability
The data provided for the results presented in this study are available through the corresponding author upon reasonable request.
References
Devi, N. & Ray, S. S. Performance of bismuth-based materials for supercapacitor applications: A review. Mater. Today Commun. 25, 101691 (2020).
Ramasamy, B., Manoharan, K., Raman, K. & Sundaram, R. Blooming flower-like BiOBr decorated RGO nanocomposite as an advanced electrode material for high-performance supercapattery applications. Mater. Sci. Eng. B 300, 117108 (2024).
Deng, L., Ma, Z. & Liu, Z.-H. Battery-type graphene/BiOBr composite for high-performance asymmetrical supercapacitor. J. Alloys Compd. 812, 152087 (2020).
Sivasubramanian, P. D. et al. A review on bismuth-based nanocomposites for energy and environmental applications. Chemosphere 307, 135652 (2022).
Tian, H. et al. Oxygen-vacancy-rich ultrathin BiOBr nanosheets for high-performance supercapacitor electrodes. Inorg. Chem. Commun. 118, 108018 (2020).
Bojarajan, A. K. et al. Advances in two-dimensional materials for supercapacitor applications: From metal carbides to metal borides and beyond. Renew. Sustain. Energy Rev. 226, 116278 (2026).
Sun, M.-Q. et al. Antifungal effects of BiOBr nanosheets carrying surfactant cetyltrimethylammonium bromide. J. Biomed. Res. 32, 380–388 (2018).
Gicha, B. B. et al. Transition metal-based 2D layered double hydroxide nanosheets: Design strategies and applications in oxygen evolution reaction. Nanomaterials 11, 1388 (2021).
Di, J., Xia, J., Li, H., Guo, S. & Dai, S. Bismuth oxyhalide layered materials for energy and environmental applications. Nano Energy 41, 172–192 (2017).
Devarayapalli, K. C., Vengamamba, P., Vattikuti, S. V. P. & Shim, J. 3D flower-like BiOBr prepared via solvothermal method for methanol oxidation and supercapacitor application. Chem. Pap. 76, 259–265 (2022).
Kumari, P. et al. One-pot synthesis of nanostructured bismuth fluoride tailored for symmetric supercapacitor performance. J. Mater. Sci. Mater. Electron. 35, 1034 (2024).
Qin, T. et al. Solid-solution iodine levers the interlayer storage of BiOBr electrode via optimizing the preferred crystal orientation for aqueous batteries. J. Power Sources 613, 234814 (2024).
Thakur, Y. S. et al. Synthesis of 3D rice-like BiOCl battery-type electrode material and evaluation of their electrochemical performance in a symmetrical supercapacitor device. Mater. Sci. Semicond. Process. 177, 108376 (2024).
Xing, H. et al. Preparation of BiOBr by solvothermal routes with different solvents and their photocatalytic activity. J. Renew. Sustain. Energy 7, 063120 (2015).
Hou, L., Niu, Y., Yang, F., Ge, F. & Yuan, C. Facile solvothermal synthesis of hollow BiOBr submicrospheres with enhanced visible-light-responsive photocatalytic performance. J. Anal. Methods Chem. 2020, 3058621 (2020).
Vattikuti, S. V. P. et al. Morphology-dependent photocatalytic and photoelectrochemical performance of bismuth oxybromide crystals applied to malachite green dye degradation. Colloids Surf. A 655, 130267 (2022).
Cai, Z., Zhong, J., Li, J. & Jin, H. Oxygen vacancies enriched BiOBr with boosted photocatalytic behaviors. Inorg. Chem. Commun. 126, 108450 (2021).
Devarayapalli, K. C. et al. In-situ Pt nanoparticles decorated BiOBr heterostructure for enhanced visible light-based photocatalytic activity: Synergistic effect. Chemosphere 298, 134125 (2022).
Hu, X., Zhao, M. & Zhang, R. New insight into visible-light-driven photocatalytic activity of Ag-loaded and oxygen vacancy-containing BiOBr(OV)/BiOI0.08 microspheres. Materials 17, 6297 (2024).
Peng, Y., Kan, P., Zhang, Q. & Zhou, Y. Oxygen vacancy enhanced photoreduction of Cr(VI) on few-layer BiOBr nanosheets. Catalysts 9, 558 (2019).
Li, G., Wang, X., Zhang, L. & Zhu, C. Electronic structures and optical properties of BiOBr/BiOI heterojunction with an oxygen vacancy. Chem. Phys. 549, 111264 (2021).
Zhang, X. et al. Self-accelerating photocharge separation in BiOBr ultrathin nanosheets for boosting photoreversible color switching. Chem. Eng. J. 428, 131235 (2022).
Arora, Y. et al. Nanostructured MoS₂/BiVO₄ composites for energy storage applications. Sci. Rep. 6, 36294 (2016).
Vattikuti, S. V. P. et al. Facile synthesis of ultrathin Bi(OH)SO₄·H₂O nanosheets and battery-like electrode for symmetric supercapacitors. J. Alloys Compd. 936, 168186 (2023).
Chen, C. J., Hu, P., Hu, X. L., Mei, Y. N. & Huang, Y. H. Chem. Commun. 51, 2798 (2015).
Gujar, T. P., Shinde, V. R., Lokhande, C. D. & Han, S.-H. Electrosynthesis of Bi₂O₃ thin films and their use in electrochemical supercapacitors. J. Power Sources 161, 1479–1485 (2006).
Li, W. et al. “Pillararene-based supramolecular polymers”. Chem. Commun. 55, 6507 (2019).
Li, Y. et al. Facile fabrication of novel Ti₃C₂Tₓ-supported fallen leaf-like Bi₂S₃ nanopieces with enhanced symmetrical supercapacitor performance. Electrochim. Acta 366, 137406 (2021).
Pooladi, M. & Zerafat, M. M. Three-dimensional flowerlike, two-dimensional sheet-like, and one-dimensional rod-like mesoporous Bi₂O₃ nanostructures for high performance asymmetric supercapacitors. J. Phys. Chem. Solids 184, 112343 (2023).
Xia, Q. X. et al. Bismuth oxychloride/MXene symmetric supercapacitor with high volumetric energy density. Electrochim. Acta 271, 351–360 (2018).
Deng, L. et al. Battery-type graphene/BiOBr composite for high-performance asymmetrical supercapacitor. J. Alloys Compd. 812, 152087 (2020).
Devarayapalli, K. C. et al. 3D flower-like BiOBr prepared via solvothermal method for methanol oxidation and supercapacitor application. Chem. Pap. 76, 259–265 (2022).
Vadivel, S. et al. A robust approach for designing a bismuth high-entropy material (BiOXCO₃) as a novel electrode for supercapacitor applications. Mater. Res. Bull. 184, 113225 (2025).
Johnson, J. W., Manohara Babu, I. & Muralidharan, G. Spherical bismuth iron oxide nanostructures as battery-type negative electrode for supercapacitor applications. J. Electroanal. Chem. 978, 118869 (2025).
Li, L. et al. Ni(OH)₂ nanosheets grown on porous hybrid g-C₃N₄/RGO network as high-performance supercapacitor electrode. Sci. Rep. 7, 43413 (2017).
Liu, J. et al. Nanosheet-interwoven structures and ion–electron decoupling storage enable Fe₁₋ₓS fast ion transport in Li⁺/Na⁺/K⁺ batteries. Nano Energy 131, 110266 (2024).
Manickavasakam, K. et al. Nanoarchitectonic marvels: Pioneering one-pot synthesis of Bi₂WO₆ nanostructures for breakthrough in symmetric supercapacitor innovation. ACS Appl. Energy Mater. 7, 5490–5500 (2024).
Shinde, P. V., Dutta, D. P., Chaskar, M. G. & Mane, R. S. High-performance aqueous supercapacitors with bismuth oxide/polyaniline composite electrodes and extended voltage window. J. Energy Storage 100, 113617 (2024).
Lin, Y.-H. et al. Evaluation of polymer gel electrolytes for use in MnO₂ symmetric flexible electrochemical supercapacitors. Polymers 15, 3438 (2023).
Khalid, S., Cao, C., Wang, L. & Zhu, Y. Microwave-assisted synthesis of porous NiCo₂O₄ microspheres: Application as high-performance asymmetric and symmetric supercapacitors. Sci. Rep. 6, 22699 (2016).
Sanger, A. et al. Silicon carbide nanocauliflowers for symmetric supercapacitor devices. Ind. Eng. Chem. Res. 55, 9452–9458 (2016).
Babu, C. R., Avani, A. V. & Anila, E. I. Optimization of rGO–MoO₃ nanocomposite electrode to fabricate an aqueous symmetric supercapacitor with enhanced performance. Int. J. Hydrogen Energy 109, 242–253 (2025).
Jirankalagi, S. N. et al. Enhanced performance of 1D ZnMn₂O₄ nanofibers for symmetric supercapacitor device. Inorg. Chem. Commun. 174, 114057 (2025).
Issar, S. et al. Li-salt assisted high-performance bimetallic titanium vanadium nitride-based symmetric supercapacitor device for energy storage application. Electrochim. Acta 535, 146636 (2025).
Pise, S. et al. Design and fabrication of gold nanoparticles decorated SSM@NiCo₂O₄ as a binder-free electrode for solid-state symmetric supercapacitor application. J. Energy Storage 114, 115965 (2025).
Kanwal, E. et al. Synergistic effects of CuO–Cr₂O₃ heterostructures fabricated via spray pyrolysis for high-performance symmetric supercapacitors. Inorg. Chem. Commun. 181, 115188 (2025).
Rajkumar, S. et al. One-step facile synthesis of Ba₂V₂O₇ nanostructures for electrochemical behaviour improvement of symmetric supercapacitor. J. Ind. Eng. Chem. 151, 762–772 (2025).
Naik, P. S. et al. Binderless synthesis of hierarchical marigold flower-like NiCo₂O₄ films for high-performance symmetric supercapacitor. J. Energy Storage 86, 111302 (2024).
Acknowledgements
The author Sivarama Krishna Lakkaboyana thanks Vel Tech Rangarajan Dr. Sagunthala R&D Institute of Science and Technology for providing the RDF-Seed Money and Seed Fund. The author, Reem Alreshidi, extends her appreciation to the Deanship of Scientific Research at Northern Border University, Arar, KSA, for funding this research work through the project number "NBU-FFR-2025-3547-03".
Funding
The research received no external funding.
Author information
Authors and Affiliations
Contributions
PSB, RMNK, SD, and JL: Formal analysis, conducting experiments, Writing and editing, Methodology, and reviewing; wrote the main manuscript text and prepared figures. LSRK, SK, SP, and RA: Formal analysis, reviewing and editing. All authors reviewed the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Babu, P.S., Kalla, R.M.N., Doraswamy, S. et al. Solvothermal synthesis and electrochemical performance of BiOBr nanosheets for symmetric supercapacitor devices. Sci Rep 15, 45787 (2025). https://doi.org/10.1038/s41598-025-28432-5
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-28432-5