Abstract
Allergic sensitizations to Dermatophagoides pteronyssinus and Dermatophagoides farinae house dust mite (HDM) species are continuously on the rise in Asian countries. Whereas the role of Blomia tropicalis, a mite species widely distributed in South/Southeast Asia, in the development of allergic diseases is well evidenced, the association between Blomia sensitizations and allergic symptoms remains poorly investigated in Malaysia. This cross-sectional study characterized the prevalence of sensitization to the clinically relevant Blomia tropicalis mite in cohorts of 329 subjects from the Klang Valley or East Coast Malaysia and developing allergic symptoms. Total specific IgE to Blomia was assayed by ImmunoCAP, IgE reactivities to recombinant Blo t 2, 5, 7, 21 were determined by ELISA IgE assays, and the association of allergic sensitization with clinical allergic symptoms was further investigated. 55% of the population was sensitized to Blo t, and sensitization to rBlo t 2, 5, 7, and 21 reached 56.9%, 74.6%, 83.4% and 57.5% respectively. rBlo t allergens sensitization was a risk factor for the development of allergic diseases, and high odds ratios observed with AA and AR indicate a strong predictive value for the diagnosis of these allergic diseases (p < 0.05). Higher rBlo t sensitization rates were observed in Klang Valley allergic patients compared to the East Coast (p < 0.05). Our study showed that Blomia sensitizations represent risk factors of allergic diseases in different regions of Malaysia, and Blo t 5 and Blo t 21, Blo t 2 and Blo t 7 can represent major Blomia allergens.
Similar content being viewed by others
Introduction
Allergic diseases are among the most common chronic conditions and are rising substantially worldwide in both developed and developing nations1. It is estimated that around 10% of the global population develops allergic diseases such as allergic asthma (AA), allergic rhinitis (AR), allergic rhinoconjunctivitis (ARC), and atopic dermatitis (AD)1. Whereas the association of allergy development with the sensitization to house dust mite (HDM) species Dermatophagoides pteronyssinus (Der p) and farinae (Der f) is well documented in Asian countries, the contribution of tropical mite Blomia tropicalis remains, by contrast, less investigated1.
To date, 26 Blo t allergens were registered in the World Health Organization – International Union of Immunological Societies (WHO-IUIS) allergen nomenclature database (www.allergen.org; September 23, 2025). Whereas Der p/Der f allergens from group 1, 2 and 23 are classified as major allergens, Blo t 5 and Blo t 21 are regarded as the most important Blomia allergens significantly associated with various allergic diseases, particularly AA and AR in tropical regions like Southeast Asia (SEA)2. Research conducted in Singapore and Malaysia found that 50% of AR patients and 90% of AA children were sensitized to Blo t 53. Similarly, in the Philippines, 80% of allergic individuals sensitized to Blo t extract also showed sensitization to rBlo t 54. Another study in Chiang Mai found that 37.5% of atopic children and 40% of atopic adults had positive SPT reactions to Blo t extracts, however, sensitization to Blo t 5 was observed in only 10% of children and 13.3% of adults5. A comparative study also revealed that Blo t 5 is a more potent sensitizer in Singapore than in Taiwan, likely due to higher exposure levels to Blo t in Singapore6. Gao et al. found that 93% of Singaporean AR patients were sensitized to Blo t 21 by enzyme-linked immunosorbent assay (ELISA) and 95% by skin prick test (SPT) analysis7. Another study involving 253 Singaporean children, 45% and 56% were sensitized to Blo t 5 and Blo t 218. Previous studies showed that the panel of major Blo t allergens is not limited to only Blo t 5 and Blo t 21. Approximately 34% of Singaporean dust-mite allergic individuals exhibited specific IgE binding to recombinant Blo t 2 (rBlo t 2)9, another study reported a higher prevalence, with 54.3% of patients sensitized to Blo t 210. The available data on Blo t 7 showed 24% and 44% of HDM allergic patients are sensitized to this allergen in Thailand and Singapore respectively8.
In contrast, data on the IgE binding frequencies of individual Blo t allergens in Malaysia are scarce. Consequently, the goal of the present study is to characterize the sensitization profile to Blomia allergens among allergic populations from two distinct geographic areas from Malaysia. Moreover, the association between allergic symptoms and Blomia sensitization was analyzed. Finally, the sensitization pattern to four individual Blo t allergens were characterized.
Methods
Study cohort
This cross-sectional study was conducted from August 2023 to January 2024, recruiting 329 patients aged 7–60 years from Pediatric and Ear Nose and Throat (ENT) clinics in the Klang Valley and East Coast hospitals. The Klang Valley sites included Hospital Sultan Abdul Aziz Shah (HSAAS) in Selangor and Hospital Kuala Lumpur (HKL), while the East Coast hospitals were Hospital Raja Perempuan Zainab II (HRPZ II) in Kelantan and Hospital Sultan Zainal Abidin (HoSZA). Participants were allergic sufferers of AA, AR, ARC, or AD diagnosed by clinicians at the respective hospitals. Exclusion criteria employed at the patient recruitment stage included pregnancy, breastfeeding due to ethical and safety concerns, and ongoing allergen immunotherapy. Each participant completed a detailed questionnaire on demographics, pet ownership, cigarette smoke, and the presence of carpets at home. Informed consent was obtained from all participants, with parental or guardian consent required for those under 18 years of age. The research was approved by the Ethics Committee for Research Involving Human Subjects, Universiti Putra Malaysia (JKEUPM-2022-1051) for HSAAS, the Medical Research & Ethics Committee (MREC) (NMRR ID-23–00134-UQB (IIR)) for HKL and HRPZ II, and the Universiti Sultan Zainal Abidin Human Research Ethics Committee (UHREC) (UniSZA.800-1/1/4 Jld.2 (15)) for HoSZA, and the study methods were performed in accordance with the Declaration of Helsinki.
Blomia tropicalis allergen skin Prick test
Skin prick testing (SPT) was performed on a total of 329 clinically allergic individuals who had discontinued the use of long-acting antihistamines for at least one week, short-acting antihistamines, and sympathomimetics for five days. Non-allergic controls were not included, as the study focuses exclusively on allergic individuals. The following compounds were used: Blomia tropicalis extracts (Immunotek, Germany – M608, 150 µg/mL), histamine HCl (MM10-STD-057, 10 mg/mL), and a negative control (MM10-STD-057). A mean wheal diameter of ≥ 3 mm was used as a positive SPT cut-off value.
Blomia tropicalis allergen-specific IgE (sIgE, kUA/L) analysis
Serum Blomia tropicalis-specific IgE levels (kUA/L) were measured in a total of 329 clinically allergic individuals who had undergone SPT using the ImmunoCAP assay (ImmunoCAP-250®, Thermo Fisher Scientific, Sweden) per the manufacturer’s protocol. The threshold of positivity was 0.35 kUA/L and the positive sera were graded into six categories: class 1 (0.35–0.69 kUA/L), class 2 (0.70–3.49 kUA/L), class 3 (3.50–17.49 kUA/L), class 4 (17.50–49.99 kUA/L), class 5 (50.00–99.99 kUA/L), and class 6 (≥ 100.00 kUA/L).
Expression and purification of Recombinant Blomia tropicalis allergens
Blo t 2.0104 (UniProt A6XEN8) was expressed as a His-tagged protein in Shuffle T7 Escherichia coli (E. coli) cells (New England Biolabs Inc.) transformed with recombinant pEt-15b vector (Novagen). When recombinant bacteria culture reached an OD600nm of 0.6, the rBlo t 2 production was induced at 37 °C for 4 h by the addition of 0.1mM Isopropyl β-D-1-thiogalactopyranoside (IPTG). The cells were collected by centrifugation and lyzed by a cell disrupter (Constant, UK). Inclusion bodies were isolated by centrifugation at 3000 rpm for 15 min and solubilized in phosphate buffer saline (PBS), 350mM sodium chloride (NaCl) and 6 M Urea buffer. rBlo t 2 was purified under denaturing conditions using immobilized Ni2 + beads (Ni-High Performance, Cytiva). Bound proteins were eluted by increasing the imidazole concentration in PBS, 350mM NaCl and 6 M Urea. Purified rBlo t 2, from 100 to 250mM imidazole elution steps, was pooled and concentrated by ultrafiltration using 5kD cut-off membrane (Amicon) to get a final volume between 3 and 5 ml. The protein was refolded by dialysis against PBS buffer, using a 3.5 KDa cut-off dialysis bag (ShakeSkinTm Dialysis tubing, Pierce). Refolded soluble rBlo t 2 was collected by centrifugation and stored at −20 C until use. Blo t 5.0101 (O96870), Blo t 7.0101 (A0A286RZM1) and Blo t 21.0101 (A7IZE9) coding sequence was cloned into the pPICZα A expression vector (Invitrogen). Pichia pastoris (P. pastoris) KM71H cells were transformed with the recombinant plasmids for the production and secretion of these Blo t allergens following a previously described protocol. Briefly, individual zeocin-resistant recombinant clones were grown at 30 °C in Buffered glycerol complex medium (BMGY) medium. When the cell density reached an OD600 nm of 6–9, the cultures were pelleted and the cells were resuspended at a final OD600 nm of 2 (rBlo t 21) or 5(rBlo t 5, rBlo t 7) in Buffered methanol complex medium (BMMY) medium containing 1% (rBlo t 7), 2% (rBlo t 5) or 3% (rBlo t 21) methanol (w/v) to trigger Blo t expression at 30 °C. After 24 h, methanol was added to maintain the inducer concentration. Culture supernatants were collected after 48–72 h of induction by centrifugation at 13 000 g for 10 min stored at 4 °C until used. The supernatants were diluted 10 times with MilliQ water, and the pH was adjusted to pH 4. These materials were applied onto an S-Sepharose ion-exchange chromatographic column (Cytiva) equilibrated with 20 mmol/L sodium acetate pH 4. After washing the unbound proteins from the column with the equilibrating buffer, bound proteins were eluted using 20 mmol/L sodium acetate pH 4 containing 100, 200, 300, 500 and 1000 mM NaCl. Fractions containing purified Blo t allergens were pooled and concentrated by ultrafiltration using an Amicon Ultra-5 K membrane (cut-off 5 kD). The purification of each allergen was achieved by a gel filtration step onto a Superdex 75 h column 10/30 (GE Healthcare Lifesciences), equilibrated with PBS pH 7.3. The fractions containing purified rBlo t were pooled and stored at − 20 °C for further analysis. The protein concentration was estimated using the microBCA protein assay kit (Pierce). The purity of each recombinant Blo t allergen was characterized by SDS-PAGE (Supplementary Fig. 1&2). The expected molecular weight of these allergens was 14.5 kDa for rBlo t 2, 13.9 kDa for rBlo t 5, 21.2 kDa for rBlo t 7, and 13.2 kDa for rBlo t 21.
Immunoglobulin E (IgE) reactivity to rBlo t allergens
ELISA plates were coated with 500 ng/well of rBlo t 2, 5, 7, or 21 in 0.1 mol/L sodium carbonate buffer (pH 9.6) and incubated overnight at 4 °C. After washing with PBS containing 0.05% Tween 20 (PBST), wells were blocked with 1% bovine serum albumin (BSA) in PBST for 2 h at 37 °C. Serum samples (at 1:10 dilution in 1% BSA-PBST) were added and incubated at 37 °C for 2 h. After washing again the plates, biotinylated goat anti-human IgE (KPL (Milford, MA, USA) (1:1000 in PBST-BSA) was applied for 1 h at 37 °C, followed by Streptavidin-HRP (BD Pharmingen (San Diego, USA) (1:1000 in PBST-BSA) for another hour. Detection of allergen-IgE complexes was performed using TMB substrate, and the reaction was stopped with a 2 N H2SO4 solution. Optical density (OD) at 450 nm was measured using a Tecan Infinite F50 microplate reader. A total of 149 Blomia-negative sera were used to determine the cut-off values for each recombinant allergen. A positive serum result was defined as an optical density (OD) exceeding the mean OD of these Blomia-negative sera plus two standard deviations. The resulting cut-off values were: rBlo t 2 = 0.27, rBlo t 5 = 0.31, rBlo t 7 = 0.28, and rBlo t 21 = 0.26.
Statistical analysis
Statistical analysis was performed using IBM SPSS Statistics for Windows, Version 29.0. Descriptive statistics summarized demographic data (age, gender, race), with mean and standard deviation calculated for quantitative variables. Logistic regression was used to determine associations between sensitization to Blo t, rBlo t allergens and allergic diseases. Odds ratios (ORs) and 95% confidence intervals (CIs) were calculated. The analysis also examined geographical distribution in the Klang Valley and East Coast of Peninsular Malaysia and assessed correlations between ImmunoCAP class and rBlo t sensitization. A significance level of P < 0.05 was considered statistically significant.
Results
Study cohort
The selected cohort (N = 329) had a gender (female/male) ratio of 69.9/30.1 whereas the mean age of the participants was 30.66 years, ranging from 7 to 60 years. Most participants were Malay (81.5%), followed by Chinese (11.6%), Indian (5.8%), or from other ethnicities such as Bajau and Dusun (1.2%). Participants were categorized into five groups of age (7–12 years-old: children, 13–18-year-old adolescents, 19–30 years-old young adults, 31–50 years-old adults, and ≥ 51 years old: middle-aged and elderly subjects), with the majority being young adults (40.4%) and adults (35.3%). Overall, a higher proportion of patients from the total population suffered from AR (48%), ARC (19.8%), AD (17.1%), and AA (14%), and a detailed demographic characteristic of each cohort is given in Table 1.
Sensitization to Blomia tropicalis by SPT and ImmunoCAP
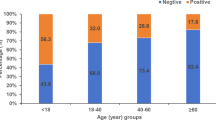
Skin prick testing revealed a Blo t sensitization rate of 51.6% in the total allergic population; sensitization levels were similar in Selangor (47.3%), Kelantan (48.8%), and Terengganu (47.8%) (p > 0.05). However, subjects from Kuala Lumpur had a significantly higher sensitization rate (61.8%) than the participants from other regions (p < 0.05) (Fig. 1a). ImmunoCAP detection of Blo t-specific IgE showed a sensitization rate of 55% in the total population. Regional sensitization rates were 63% (Kuala Lumpur), 56% (Selangor), 53% (Kelantan), and 46% (Terengganu). This indicated a higher prevalence in the Klang Valley (59%) compared to the East Coast (41%). (Fig. 1a). The distribution of ImmunoCAP grade classifications among the allergic population is presented in Supplementary Table 1. A total of 329 samples were assessed, with 45.3% (n = 149) classified as negative (< 0.35 kU/L). Among positive classifications, the highest prevalence was observed in Class 3 (> 3.5–17.5 kU/L), representing 17.9% (n = 59) of the samples, followed by Class 2 (> 0.70–3.5 kU/L) and Class 4 (> 17.5–50 kU/L), each accounting for 12.8% (n = 42). The lowest occurrences were seen in Class 1 (0.35–0.60, N = 21), Class 5 (> 50–100, N = 12), and Class 6 (> 100 kU/L, N = 4), accounting for 6.4%, 3.6%, and 1.2% respectively of the total samples.
(a) Comparative sensitization rates detected by SPT and ImmunoCAP Blot across allergic cohorts (N = 329). The bar graph illustrates the percentage of individuals testing positive for each allergen using two diagnostic methods: SPT Blot (light blue) and ImmunoCAP Blot (dark blue). Data represent only positive cases, highlighting differential detection rates and potential variability in allergen recognition between skin prick testing and in vitro IgE measurement. (b) Distribution of population response to Blomia tropicalis based on concordance and discordance between SPT and ImmunoCAP results. “Concordant” refers to individuals with matching test outcomes (both positive or both negative). “Discordant (SPT⁺/ImmunoCAP⁻)” and “Discordant (SPT⁻/ImmunoCAP⁺)” represent individuals with conflicting test results. Percentages are calculated relative to each diagnostic subgroup and are not additive across categories. This visualization highlights test agreement and discrepancy patterns rather than cumulative population proportions.
Comparison of SPT and IgE-reactivity to Blomia tropicalis
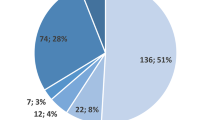
SPT and ImmunoCAP results were not fully concordant among allergic patients. Among 329 cases, 101 (67.8%) were negative in both tests (concordant negative), while 121 (67.2%) were positive in both (concordant positive). However, 48 cases (32.2%) were SPT-positive but ImmunoCAP-negative, and 59 cases (32.8%) were SPT-negative but ImmunoCAP-positive. Overall, 67.5% (222 cases) showed agreement between the two tests, while 32.5% (107 cases) were discordant. Kappa statistical analysis yielded a Kappa value of 0.348 (p = 0.001), indicating a fair level of agreement but highlighting notable discordance (Fig. 1b). Additionally, an ordinal logistic regression analysis was performed to assess the relationship between Blo t ImmunoCAP class and SPT wheal size grade, the model demonstrated a statistically significant association between ImmunoCAP class and wheal size grade with higher likelihood of positive ImmunoCAP value with a higher SPT wheal size (p < 0.05). The estimated regression coefficients and corresponding odds ratios are presented in Supplementary Table 2.
Sensitization patterns to rBlo t allergens
The prevalence of sensitization to rBlo t 2, rBlo t 5, rBlo t 7 and rBlo t 21 reached 56.9%, 74.6%, 83.4% and 57.5% respectively in the Blo t-ImmunoCAP positive subjects (n = 180) and 31.3%, 41%, 45.9, and 31.6% respectively in the total allergic population (n = 329). Of note, two patients (0.6%) were exclusively sensitized to rBlo t 2, nine (2.7%) sensitized to rBlo t 5, whereas nine and one were solely sensitized to rBlo t 7, and rBlo t 21 respectively. Many patients exhibited co-sensitizations to at least two rBlot allergens, 18.2% of the population were sensitized to all four rBlo t allergens (Fig. 2a). Spearman’s rank-order correlation was used to assess the strength and direction of associations among specific IgE levels to rBlo t allergens, and all correlations were statistically significant, indicating consistent co-sensitization patterns among the components. Moderate to strong positive correlations were observed between the allergen components; the strongest correlation was found between rBlo t 5 and rBlo t 7 (ρ (rho) = 0.603, p < 0.001), followed by rBlo t 2 and rBlo t 7 (ρ (rho) = 0.598, p < 0.001). The weakest, though still significant, correlation was between rBlo t 2 and rBlo t 21 (ρ (rho) = 0.415, p < 0.001). These findings suggest that sensitization to one Blo t component is often accompanied by sensitization to others, particularly among rBlo t 2, t 5, and t 7. The heatmap visualizing the Spearman correlation coefficients among rBlo t allergens are presented in Fig. 2b. Sensitization to rBlo t allergens varied across age groups (Fig. 2c), in children (7–12 years), rBlo t 5 is the most prevalent sensitizer (36%), while rBlo t 2 is the least (12%). Among adolescents (13–18 years), sensitization rates increased, with rBlo t 5 (57.1%) and rBlo t 7 (52.4%) being the most immunodominant allergens. High levels of sensitization to rBlo t 7 (51.1%) and rBlo t 5 (45.1%) were maintained in young adults (19–30 years), together with notable IgE binding frequency to rBlo t 2 (40.6%) and rBlo t 21 (31.6%). In adults (31–50 years), rBlo t 7 (45.7%) remained the dominant sensitizer, followed by rBlo t 5 (41.4%) and rBlo t 21 (37.1%). Among those ≥ 50 years, sensitizations to rBlo t 7 (44.1%) and rBlo t 21 (32.4%) are the most prevalent ones followed by rBlo t 5 and rBlo t 2 (29.4% each).
(a) Venn diagram showing the distribution of rBlo t 2, rBlo t 5, rBlo t 7 and rBlo t 21 sensitizations among the study population (N = 329), (b) Spearman correlation matrix of IgE reactivity among rBlo t allergens, (c) Age distribution of sensitization rates to rBlo t allergens in the allergic population, (d) rBlo t sensitization rates among the allergic populations across different cohorts in Malaysia (n = 329), % sensitization in brackets represents Blo t-ImmunoCAP positive cohort (n = 180).
Conversely, sensitization rate to these rBlo t allergens in each study cohort is shown in Fig. 2d. Allergic population for Kuala Lumpur, Selangor, Kelantan, and Terengganu had sensitization rate of 48.3%/45.2%/12.7%/12.9% for rBlo t 2, 36.8%/40.9%/44.3%/42.9% for rBlo t 5, 62.1%/51.6%/37.9%/27.1% for rBlo t 7, and 35.6%/37.6%/27.8%/22.9% for rBlo t 21 while percentages based on Blo t-ImmunoCAP positive was 76.4%/80.8%/23.8%/28.1% for rBlo t 2, 58.2%/73.1%/83.3%/93.8% for rBlo t 5, 98.2%/92.3%/71.4%/59.4% for rBlo t 7, and 56.4%/67.3%/52.4%/50% for rBlo t 21 respectively. Notably, rBlo t 2 is much more prominent in the allergic cohort of Selangor, rBlo t 5 for Terengganu, rBlo t 7 for Kuala Lumpur, and rBlo t 21 in Selangor as well. Allergic individual IgE antibody levels are presented in Fig. 3.
IgE antibody levels were assessed by means of direct ELISA in sera of 329 allergic individuals. Each symbol represents the result obtained with an individual serum. Short horizontal solid lines represent the median value of the group results. The cut-offs are represented by green (0.27 for rBlo t 2 assay), yellow (0.31 for rBlo t 5 assay), blue (0.28 for rBlo t 7), and red (0.26 for rBlo t 21) dashed lines, respectively.
A multinomial logistic regression approach was conducted to examine the geographical variation in the prevalence of sensitization to tested rBlo t. The CI, OR, and p-value was OR = 5.165, CI = (3.013, 8.854), P-value < 0.001 for rBlo t 2; rBlo t 5: OR = 0.776, CI = (0.500, 1.204), P-value = 0.258; rBlo t 7: OR = 2.385, CI = (1.525, 3.730), P-value < 0.001; rBlo t 21: OR = 1.489, CI = (0.930, 2.384), P-value = 0.098 respectively. Collectively, our data evidenced a statistically significant and robust relationship between sensitization to rBlo t 2, rBlo t 7 and the residential region of the patients (p < 0.001), higher sensitization rates to rBlo t 2, and rBlo t 7 being recorded in the Klang Valley (79.4% and 66% respectively) than in the East Coast (20.4% and 34%). In contrast, no significant association was observed between sensitization to rBlo t 5, rBlo t 21 and geographical location (Fig. 4a).
(a) Comparison of % sensitization rates between populations from Klang Valley and East Coast of Malaysia across native and recombinant allergen components (Blo t, rBlo t 2, rBlo t 5, rBlo t 7, rBlo t 21). Statistically significant differences are indicated (***). (b) Distribution of % sensitization rates across allergic disease types: AA (Allergic Asthma), AR (Allergic Rhinitis), ARC (Asthma with Rhinitis), and AD (Atopic Dermatitis) for the same allergen components.
Blo t and rBlo t allergens sensitization as markers for allergic diseases
A multinomial logistic regression analysis was performed to assess Blo t and rBlo t sensitization as a risk factor for the development of allergic disease. Among all the allergic diseases, AA exhibited the strongest relationship with Blo t (OR = 9.442E + 9, CI = 1.615E + 7- 5.519E + 10), suggesting a highly robust effect. Similarly, ARC and AR followed closely, with OR values of 6.853E + 9 and 4.343E + 9, respectively, indicating substantial associations supported by their respective confidence intervals (1.145E + 7 to 4.102E + 10 for ARC and 2.669E + 8 to 7.066E + 10 for AR). AD showed a slightly weaker, yet still significant, association, with an OR of 2.469E + 9 (1.465E + 8-4.162E + 10, (p < 0.001; Fig. 4b). Similarly, AA and AR exhibited the strongest relationship with rBlo t 2, 5, 7 and 21 at a p < 0.001 and the detailed OR and CI values are given in Supplementary Table 3. The consistently high odds ratios observed with AA and AR indicate a strong predictive value, which indicates that sensitization to these rBlo t allergens may help in the diagnosis of these allergic diseases.
Additionally, the analysis of rBlo t sensitization patterns across different allergic diseases reveals distinct trends in the number of allergens to which individuals are sensitized. Among individuals with AA, polysensitization is particularly prominent, with 23.9% showing sensitization to three rBlo t allergens, 19.6% being dual and quadra-sensitized respectively, and monosensitization (4.4%). In contrast, individuals with AR showed the highest rate of quadra-sensitization (21.5%), indicating a broad reactivity to Blomia allergens in this population. Although tri-sensitization is also present (12.7%), dual and mono-sensitization appear less common (11.4% and 6.3% respectively). Patients with ARC had tri-sensitization (18.5%) being the most frequent, while mono, dual and quadra- sensitization was 4.6%, 12.3%, and 15.4% respectively. AD individuals showed a unique sensitization pattern, while it has the highest rate of mono-sensitization (8.8%), it also demonstrates a high rate of quadra-sensitization (21.1%), with 8.8% and 7% reported for mono and dual-sensitization respectively.
rBlo t sensitization and ImmunoCAP grade classification
Figure 5a shows the strength and significance of associations between ImmunoCAP class and rBlo t allergens sensitization. Among the allergens, rBlo t 5 and rBlo t 7 show the strongest and most consistent associations across all ImmunoCAP classes with statistically significant p-values (p < 0.001) in nearly all cases suggesting that rBlo t 5 and rBlo t 7 could serve as key biomarkers for allergic responses in individuals with varying ImmunoCAP levels. In contrast, rBlo t 2 does not exhibit a consistent pattern across all classes but becomes statistically significant in higher ImmunoCAP classes, specifically, in Class 4 & 5. However, in lower ImmunoCAP classes (1, 2, and 3), rBlo t 2 does not show a significant relationship, indicating that its role may be more relevant in severe cases of allergic sensitization. Interestingly, rBlo t 21 consistently fails to show significant associations across all ImmunoCAP classes (p > 0.05), although not statistically significant, its high confidence intervals highlight its major role in allergic sensitization in this study cohort. Additionally, the percentage of recognized rBlo t allergens based on the different ImmunoCAP classes is given in Fig. 5b. Higher ImmunoCAP classes (Class 3 and Class 4) dominate in frequency, with Class 3 (> 3.5–17.5) consistently represents the highest percentage across all rBlo t allergens, ranging from 31% to 35%, while Class 4 (≥ 17.5–50) follows closely with 26% to 32% of cases.
(a) Forest plot of correlations between rBlo t sensitization and ImmunoCAP classification. CI > 1 and p < 0.05 indicates a positive correlation between each Blo t ImmunoCAP class and the likelihood of sensitization to rBlo t allergens, (b) rBlo t sensitization rates among the different ImmunoCAP classes (n = 180).
Discussion
Epidemiological studies on Blo t sensitization in the Malaysian allergic population remain limited, with only three papers reporting prevalence rates between 41–70%3,11,12. The present study found a Blo t sensitization rate of 51.6% by SPT and 55% by ImmunoCAP, compared to other regions, Malaysia’s sensitization rate is lower than those reported in Singapore (91%) and Taiwan (73.3%) but higher than Thailand (37.5% in children, 40% in adults)5,8,13. However, our data confirmed Blomia as an important allergenic source involved in the development of allergic symptoms in Malaysia. The absence of non-allergic controls in the SPT represents a limitation in terms of comparative baseline reactivity; however, the study’s primary objective was to characterize the molecular sensitization profiles to Blomia tropicalisallergens (Blo t 2, 5, 7, and 21) in individuals with clinically confirmed allergic diseases. This targeted design is consistent with previous research that has focused exclusively on allergic populations to delineate clinically relevant sensitization patterns and their association with disease phenotypes. Nonetheless, future studies incorporating non-allergic controls could enhance the robustness of diagnostic thresholds and improve the specificity of component-resolved diagnostics in broader populations14,15.
We observed discordance in sensitization rates to Blo t when comparing SPT and ImmunoCAP. Discrepancies between SPT and serum-specific IgE levels have been previously reported16. A serum tested negative in SPT but positive in ImmunoCAP could be explained by the low quality of the Blomia extracts used for SPT (poor allergenic content) or the age of the participants (SPT wheal size being often smaller in senior individuals)16. The 3 mm cutoff value for positive SPT could not be reliable enough in detecting Blomia-specific IgE by ImmunoCAP. Of note, larger positive SPT threshold values have been positively correlated with Blomia-specific IgE positivity results17.
The major objective of the present study was to examine the molecular sensitization profile in Blomia-sensitized patients from Malaysia. To our knowledge, this is the first report assessing simultaneously the prevalence of sensitization to rBlo t 2, rBlo t 5, rBlo t 7, and rBlo t 21 allergens in Malaysia and the association of these IgE reactivities with allergic diseases. Whereas homogenous preparations of rBlo t 2, rBlo t 5, rBlo t 7 were obtained, two forms of rBlo t 21 were detected by SDS-PAGE (Supplement Fig. 1). This apparent protein heterogeneity could result from the partial N-terminal degradation of rBlo t 21 due to the presence of a disordered N-terminal motif. Blo t 5, sharing an extremely similar three-dimensional (3D) structure with Blo t 21, also contains such an N-terminally disordered motif leading to the N-terminal degradation of rBlo t 5 produced in bacteria18. Moreover, although our rBlo t 5 preparation appears to be homogenous, a previous report showed that purified rBlo t 5 produced in P. pastoris could also be partially degraded (SDS-PAGE migration as a doublet)19. Nevertheless, we speculate that the IgE reactivity to rBlo t 21 is not influenced by this degradation, as the allergenic properties of Blo t 5 were mostly not changed by shortening the N-terminal region18.
High sensitization rates (> 50%) were found for the four tested Blo t allergens, with the exception of those to rBlo t 2 measured in Kelantan and Terenganu (< 30%). Blo t 2 represents a major allergen in Malaysia. The importance of Blo t 2 in Blomia sensitization was previously reported in Brazil and Colombia (~ 54%), whereas this allergen is a weaker sensitizer in Singapore (18%, 34%) and Thailand (34%)2,8,9,20,21. Sensitization rate to rBlo t 5 (74.6%) was comparable with that previously reported in the Singaporean-Malaysian cohort (73.5%)22. Although the IgE reactivity to Blo t 5 did not reach that measured in Taiwan (91.8%) and Singapore (90%), our study confirmed that Blo t 5 is a major Blomia sensitizer in Malaysia3,22. We measured a high sensitization rate (83.4%) to Blo t 7. This allergen remains poorly investigated; previous studies showed that Blo t 7 is not a major trigger of allergic sensitization in Singapore (44%), Thailand (24%), and Brazil (< 15%)2,8,20. Of note, data generated in Colombia and Singapore used a recombinant Blo t 7 produced in E. coli and lacking some internal amino acid sequence20. Further studies are needed to confirm the importance of Blo t 7-specific allergic sensitization in Malaysia. For rBlo t 21, the rate (57.5%) matched with that reported among allergic individuals in Singapore (57%), although higher rates (80–93%) have been reported by SPT and serology test in another Singaporean cohort7,8. Taken together, our CRD data suggest that, together with Blo t 5 and Blo t 21, Blo t 2 and Blo t 7 are major allergens in Malaysia. Additionally, rBlo t 5 and rBlo t 7 emerged as the most significant allergens associated with ImmunoCAP classes, underscoring their role in allergic sensitization. rBlo t 2 was primarily relevant in higher ImmunoCAP classes, while rBlo t 21 showed no significant relationship.
Geographical variation in sensitization rate to rBlo t 2 and rBlo t 7 was also observed, whereas the levels of rBlo t 5 and rBlo t 21 sensitizations are similar in allergic cohorts from the Klang Valley and East Coast of Malaysia. Sensitizations to rBlo t 2 and rBlo t 7 were more prevalent in the Klang Valley. Despite similar temperatures (25–33 °C) and humidity levels (> 75%), the Klang Valley (Kuala Lumpur and Selangor) and East Coast (Kelantan and Terengganu) reflect Malaysia’s environmental and socioeconomic diversity. Kuala Lumpur, the metropolitan city, is highly urbanized and industrialized, with dense populations, elevated pollution levels related to industrial activity, and compact air-conditioned housing. Selangor is more suburban, with larger homes and lower pollutant exposure. In contrast, Kelantan and Terengganu are less urbanized, featuring traditional stilted wooden houses that promote natural cooling and flood protection. These regional differences in urbanization, housing, and environmental exposure shape allergen sensitization patterns and contextualize our findings. This regional variability is consistent with findings from Colombia, Brazil, and Equatorial Africa, where sensitization patterns differ across cities and climates2,23,24. The possible correlation between Blo t 2, Blo t 7 levels in house dust and IgE binding frequencies deserves our attention.
The association of several Der p allergens with AA, AR, and AD was previously evidenced, notably Der p 5, Der p 21, Der p 37, Der p 1, Der p 23, and Der p 11 for AA, Der p 20 for AD, while very few data were produced in the context of Blomia. Our present study evidenced a significant association of sensitizations to rBlo t 2, rBlo t 5, rBlo t 7, and rBlo t 21 with AA, AR, ARC, and AD. In Colombia, IgE responses to Blo t 5/Blo t 21 but not Blo t 2/Blo t 7 were associated with AA, whereas rBlo t 2, rBlo t 5, and rBlo t 21 were the most frequently recognized Blo t allergens in AA from Gabon (equatorial Africa)2,25,26,27,28. Although our results demonstrated a positive association between the four rBlo t allergens and allergic diseases, the consistently high odds ratios observed in individuals with AA and AR suggest that sensitization to the four tested allergens may serve as potential biomarkers for AA and AR, particularly within the Malaysian regions studied. Analysis of rBlo t sensitization patterns reveals distinct allergen reactivity across allergic conditions. Polysensitization is most prominent in AA patients, with tri-sensitization (23.9%) being most common. In AR, quadra-sensitization (21.5%) dominates, suggesting broader allergen recognition. Patients with ARC mostly show tri-sensitization (18.5%). AD presents a unique bimodal pattern, with the highest rates of both mono- (8.8%) and quadra-sensitization (21.1%). These variations highlight the diverse sensitization profiles across allergic diseases, which could inform tailored diagnostic and treatment approaches.
Conclusion
In conclusion, our study showed that Blomia sensitizations represent risk factors of allergic diseases in Malaysia, regional variability exists, where sensitization patterns to Blo t allergens differs across regions of Malaysia. Together with Blo t 5 and Blo t 21, Blo t 2 and Blo t 7 can represent major Blomia allergens in this Southeast Asian country.
Data availability
Data emanating from this study is available from the corresponding author upon request.
Abbreviations
- AA:
-
Allergic asthma
- AD:
-
Atopic dermatitis
- AR:
-
Allergic rhinitis
- ARC:
-
Allergic rhinoconjuctivitis
- Blo t:
-
Blomia tropicalis
- BMGY:
-
Buffered glycerol complex medium
- BMMY:
-
Buffered methanol complex medium
- BSA:
-
Bovine serum albumin
- CI:
-
Confidence interval
- CRD:
-
Component resolved diagnosis
- Der f:
-
Dermatophagoides farinae
- Der p:
-
Dermatophagoides pteronyssinus
- ELISA:
-
Enzyme-linked immunosorbent assay
- ENT:
-
Ear, nose and throat
- HDM:
-
House dust mite
- HKL:
-
Hospital Kuala Lumpur
- HRPZ II:
-
Hospital Raja Perempuan Zainab II
- HSAAS:
-
Hospital Sultan Abdulaziz Shah
- IgE:
-
Immunoglobulin E
- JKEUPM:
-
Jawatankuasa Etika Universiti untuk Penyelidikan Melibatkan Manusia
- MREC:
-
Medical Research and Ethics Committee
- NaCl:
-
Sodium chloride
- OD:
-
Optical density
- OR:
-
Odd ratio
- PBS:
-
Phosphate buffer saline
- PBST:
-
Phosphate buffer saline-Tween
- rBlo t:
-
recombinant Blomia tropicalis
- IPTG:
-
Isopropylβ-D-1-thiogalactopyranoside
- SEA:
-
Southeast Asia
- SPT:
-
Skin prick test
- UHREC:
-
Universiti Sultan Zainal Abidin Human Research Ethics Committee
- UniSZA:
-
Universiti Sultan Zainal Abidin
- WHO-IUIS:
-
World Health Organization International Union of Immunological Societies
References
Gutowska-Ślesik, J., Samoliński, B. & Krzych-Fałta, E. The increase in allergic conditions based on a review of literature. Adv. Dermatol. Allergol. 40 (1), 1–7. https://doi.org/10.5114/ada.2022.119009 (2023).
Zakzuk, J. et al. IgE-mediated sensitization to Blo t 21 and Blo t 5 is associated with asthma in the tropics: A case-control study. J. Investig Allergol. Clin. Immunol. 34 (3), 157–166. https://doi.org/10.18176/jiaci.0892 (2024).
Yeoh, S. M. et al. Sensitization profiles of Malaysian and Singaporean subjects to allergens from dermatophagoides Pteronyssinus and blomia tropicalis. Int. Arch. Allergy Immunol. 132 (3), 215–220 (2003).
Patanindagat, C. Y. et al. Correlation of blomia tropicalis-specific Immunoglobulin epsilon profiles with family history of atopy in a Filipino population. Asia Pac. Allergy. 14 (1), 12–20. https://doi.org/10.5415/apallergy.0000000000000133 (2024).
Trakultivakorn, M. & Nuglor, T. Sensitization to dermatophagoides Pteronyssinus and blomia tropicalis extracts and Recombinant mite allergens in atopic Thai patients. Asian Pac. J. Allergy Immunol. 20 (4), 217–221 (2002).
Kuo, I. C. et al. Sensitization to blomia tropicalis and dermatophagoides pteronyssinus—A comparative study between Singapore and Taiwan. Asian Pac. J. Allergy Immunol. 17 (3), 179–188 (1999).
Gao, Y. F. et al. Identification and characterization of a novel allergen from blomia tropicalis: Blo t 21. J. Allergy Clin. Immunol. 120 (1), 105–112 (2007).
Kidon, M. I. et al. Mite component-specific IgE repertoire and phenotypes of allergic disease in childhood: the tropical perspective. Pediatr. Allergy Immunol. 22 (2), 202–210. https://doi.org/10.1111/j.1399-3038.2010.01094.x (2011).
Reginald, K., Pang, S. L. & Chew, F. T. Blo t 2: group 2 allergen from the dust mite blomia tropicalis. Sci. Rep. 9, 12239. https://doi.org/10.1038/s41598-019-48688-y (2019).
Mondol, E. et al. The allergenic activity of Blo t 2, a blomia tropicalis IgE-binding molecule. Int. J. Mol. Sci. 24 (6), 5543. https://doi.org/10.3390/ijms24065543 (2023).
Nadzrah, Y. S. & AB, Z. Common aeroallergens by skin Prick test among the population in two different regions. Prim. Health Care Open. Access. 5 (3). https://doi.org/10.4172/2167-1079.1000206 (2015).
Lee, H. L., Tang, M. M., Bakhtiar, M. F., Mohamad Yadzir, Z. H. & Johar, A. Sensitization to local seafood allergens in adult patients with atopic dermatitis in Malaysia. Int. Arch. Allergy Immunol. 182 (2), 153–157. https://doi.org/10.1159/000510314 (2021).
Tsai, T. et al. Burden of atopic dermatitis in Asia. J. Dermatol. 46 (10), 825–834. https://doi.org/10.1111/1346-8138.15048 (2019).
Mourao, E. M. M. & Rosario, N. A. Conjunctival provocation test with blomia tropicalis. Front. Allergy. 2, 673462. https://doi.org/10.3389/falgy.2021.673462 (2021).
González-Pérez, R. et al. Molecular allergen profiling of dual mite sensitization in severe allergic rhinitis. J. Investig Allergol. Clin. Immunol. 30 (6), 421–429. https://doi.org/10.18176/jiaci.0439 (2020).
Lee, J-E. et al. Variability of offending allergens of allergic rhinitis according to age: optimization of skin Prick test allergens. Allergy Asthma Immunol. Res. 6 (1), 47–54 (2014).
Kanceljak-Macan, B., Macan, J., Plavec, D., Klepac, T. & Milković-Kraus, S. The 3 mm skin Prick test (SPT) threshold criterion is not reliable for tyrophagus putrescentiae: the re-evaluation of SPT criterion to dust mites. Allergy 57 (12), 1187–1190. https://doi.org/10.1034/j.1398-9995.2002.23730.x (2002).
da Silva, E. S. et al. N-terminal peptide deletion influences immunological and structural features of Blo t 5. Allergy 75 (6), 1503–1507 (2020).
Goh, L. T. et al. Production and purification of Recombinant blomia tropicalis group 5 allergen from Pichia pastoris culture. Biotechnol. Lett. 23, 661–665 (2001).
Soongrung, T. et al. The blomia tropicalis allergen Blo t 7 stimulates innate immune signaling pathways through TLR2. Clin. Exp. Allergy. 48 (4), 464–474. https://doi.org/10.1111/cea.13098 (2018).
Pauli, G. et al. Molecular sensitization profile of asthmatic children in Equatorial Africa. World Allergy Organ. J. 13, 100160 (2020).
Kuo, I. C., Cheong, N., Trakultivakorn, M., Lee, B. W. & Chua, K. Y. An extensive study of human IgE cross-reactivity of Blo t 5 and der p 5. J. Allergy Clin. Immunol. 111 (3), 603–609. https://doi.org/10.1067/mai.2003.167 (2003).
Fernández-Caldas, E. & Lockey, R. F. Blomia tropicalis, a mite whose time has come. Allergy 59 (11), 1161–1164. https://doi.org/10.1111/j.1398-9995.2004.00727.x (2004).
Hussein, A. H. & Elawamy, W. Quantitation of blomia tropicalis allergen Blo t 5 in cereal and cereal-based foods consumed in the nile Delta, Egypt. Am. J. Trop. Med. Hyg. 93 (1), 194–197. https://doi.org/10.4269/ajtmh.14-0837 (2015).
Banerjee, S. et al. Der p 11 is a major allergen for house dust mite-allergic patients suffering from atopic dermatitis. J. Invest. Dermatol. 135 (1), 102–109. https://doi.org/10.1038/jid.2014.271 (2015).
Huang, H. J., Sarzsinszky, E. & Vrtala, S. House dust mite allergy: the importance of house dust mite allergens for diagnosis and immunotherapy. Mol. Immunol. 158, 54–67. https://doi.org/10.1016/j.molimm.2023.04.008 (2023).
Walsemann, T. et al. Specific IgE against the house dust mite allergens der p 5, 20 and 21 influences the phenotype and severity of atopic diseases. Allergy 78 (3), 731–742. https://doi.org/10.1111/all.15553 (2023).
Pauli, G. et al. Frequent IgE recognition of blomia tropicalis allergen molecules in asthmatic children and young adults in Equatorial Africa. Front. Immunol. 14, 1133935. https://doi.org/10.3389/fimmu.2023.1133935 (2023).
Acknowledgements
We sincerely thank all participants for their voluntary involvement in this study. We also appreciate Immunotek, Germany, for providing the Blo t extracts and the Allergy and Immunology Unit at the Institute of Medical Research, Shah Alam, for granting access to the ImmunoCAP Phadia 250 system.
Funding
This work was supported by Universiti Putra Malaysia under Geran Putra Berimpak [GP-GPB/2022/9726000].
Author information
Authors and Affiliations
Contributions
Chau Ling Tham and Alain Jacquet conceived the idea. Chau Ling Tham, Alain Jacquet, and Intan Hakimah Ismail supervised the student (Abosede Temitope Olajide) and contributed to the experimental design. Chau Ling Tham secured the funding. Abosede Temitope Olajide drafted the manuscript. All authors contributed to data collection. Abosede Temitope Olajide performed the statistical analysis and interpreted the results. All authors critically reviewed and approved the final version of the manuscript.
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
Ethical approval was granted by the Ethics Committee for Research Involving Human Subjects, Universiti Putra Malaysia (JKEUPM-2022-1051) for HSAAS, the Medical Research & Ethics Committee (MREC) (NMRR ID-23-00134-UQB (IIR)) for HKL and HRPZ II, and the Universiti Sultan Zainal Abidin Human Research Ethics Committee (UHREC) (UniSZA.800-1/1/4 Jld.2 (15)) for UniSZA.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Olajide, A.T., Ismail, I.H., Zakaria, A.F. et al. IgE-mediated sensitizations to Blomia tropicalis allergens in Malaysian allergic population. Sci Rep 15, 44688 (2025). https://doi.org/10.1038/s41598-025-28726-8
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-28726-8