Abstract
Retinal ganglion cells detect changes in illuminance. An increase and a decrease in illuminance levels induce ON and OFF responses, respectively. This segregation into two major types of visual responses is carried out through the lateral geniculate nucleus (LGN) to the primary visual cortex area V1. In area V1, these responses are partially merged on single neurons, although domains of ON and OFF responses are still found in layer IV, which receives LGN afferents, and in layers II-III, which have no direct inputs from the LGN. However, it is unclear whether feedback circuits in area V1 can process ON and OFF responses independently. Here, we demonstrate that optogenetic local activation of layer IV–VI pyramidal neurons modulates the receptive field (RF) area of ON and OFF responses independently. Although the RF area was reduced for both ON and OFF responses (77.9% of control, Q1 = 56.0%, Q3 = 93.1%, and 60.7% of control, Q1 = 45.1%, Q3 = 92.2%, respectively; n = 60), there was no correlation between these changes in single neurons (Spearman’s rank correlation coefficient ρ = 0.144, p > 0.27, n = 60). In contrast, the ON and OFF response amplitude changes correlated in single neurons (ρ = 0.486, p < 0.0003, n = 60), and the median amplitude change was similar for both types of responses (79.6% and 84.6%, respectively, for OFF and ON responses). These results can be explained by assuming that modulation of the response amplitude is largely dependent on changes in the membrane properties of a neuron, while the RF area is modulated mainly in feedback loops presynaptically.
Similar content being viewed by others
Introduction
Retina is tuned to detect changes in illuminance and retinal ganglion cells (RGCs) mainly react to changes in illuminance levels, though sustained responses are also found in a fraction of cells1. Since illuminance levels can either increase or decrease, two major types of responses are distinguished. ON responses are produced by an increase in illuminance while OFF responses occur after a decrease in illuminance levels2. Although recent studies have indicated that more than 40 different types of RGCs exist, they can be grouped into two main types: ON and OFF RGCs1,3. Thus, starting from the retina, ON and OFF responses are processed in parallel. This segregation of the ON and OFF pathways continues to the thalamic dorsal lateral geniculate nucleus (dLGN), a major target of retinal RGC axons in mammals, and then persists up to the primary visual cortex (area V1), where domains of prevalently ON and OFF responses produced by the dLGN axon terminals are found in cats, ferrets and mice4,5,6,7. At least in some species, these area V1 ON and OFF domains are found not only in cortical layer IV, which receives afferents from the dLGN, but also in layers II-III, which have no direct thalamic inputs, alternatively, these inputs may be much weaker8. This segregation of cortical inputs shapes the structure of receptive fields and may contribute to directional and orientation tuning in the cortex9, suggesting segregation of ON and OFF pathways in local V1 feedforward circuits, at least from layer IV to layers II–III. However, in addition to these feedforward links, V1 neurons have extensive feedback connections: local circuits connecting pyramidal neurons via V1 interneurons, and extra-cortical circuits that go back from V1 to the dLGN and to other subcortical visual centers, such as the superior colliculus (SC)10,11. Neurons in the dLGN send their axons back to layer IV of V1 and these afferents form the above mentioned ON and OFF domains in V14. Thus, these feedback connections, from V1 (mainly layer VI) to the dLGN and then back to V1, may affect ON and OFF responses in a selective way; in other words, these feedback connections may modulate ON and OFF response properties independently of each other. Moreover, it is argued that a large fraction of cortico-cortical connections may actually be made through the cortico-thalamic loop12; therefore, cortico-cortical connections maybe also segregated to ON and OFF response pathways. Nevertheless, we are not aware of evidence that would support such segregation to ON and OFF pathways in the area V1 feedback connections. We recently showed that local optogenetic stimulation of layer IV–VI pyramidal neurons in V1 can modulate ON response properties via feedback connections that involve local interneurons13. Preliminary results indicated that OFF responses were also modulated during pyramidal neuron stimulation.
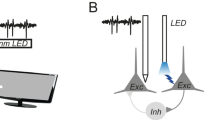
Schematics of the experimental procedures. (A) Visual response receptive fields were studied in vivo during recordings in anesthetized rats viewing visual stimuli on a monitor. (B) To modulate visual responses, optogenetic stimulation of excitatory pyramidal neurons was employed. (C,D) Examples of responses obtained during mapping of receptive fields while the LED was OFF I or on (D). The gray lines below the PSTHs indicate the time when a visual stimulus was presented. Green and light blue ellipses indicate 2D Gaussian fits of the ON and OFF response distributions, respectively. (E) Average response PSTHs taken from the receptive field area; green indicates an ON response, while black indicates an OFF response. (F) Gray color-coded response distributions in the mapped area; the broken line ellipses indicate 2D Gaussian fits of the ON and OFF response distributions. (A) and (B) are modified from Baranauskas et al.10.
Therefore, by employing optogenetic stimulation of layer IV–VI pyramidal neurons, we tested whether the properties of ON and OFF responses are modulated independently, i.e., whether feedback connections of V1 affect the properties of ON and OFF responses independently. Our results indicate that while there was a clear correlation between changes in ON and OFF response amplitudes, the receptive field (RF) area was modulated independently.
Results
We activated mainly pyramidal neurons in deep layers IV-VI of the rat primary visual cortex by employing optogenetic methods during visual stimulus presentation (Fig. 1A,B). To this end, modified channelrhodopsin-2 (ChR2) with the H134R mutation (ChR2-H134R14, controlled by a short CaMKII promoter15, was expressed in V1 pyramidal neurons following AAV injections performed ~ 1000 μm below the cortical surface (see “Methods” for details). We have shown previously that such injections result in the expression of ChR2 mainly in cortical layer IV–VI pyramidal neurons13. ChR2 channels were activated by 465 nm LED light delivered ~ 200 μm above the recording site via a 100 μm wide optical fiber attached to the recording tetrode13. Trains of 18–20 ~ 10 ms long pulses delivered each 110 ms were used to optogenetically stimulate pyramidal neurons. All but one recording weas made between 890 μm and 1930 μm below the cortical surface, mostly corresponding to cortical layers V and VI16. One recording (3 single units) was performed 670 μm below cortical surface, corresponding to layer IV. Although these layer inferences are indirect, in our previous study, the same range of depths was used for recordings (690 μm – 1940 μm) and lesions were generated for recording location identification; it was found that all recordings were made within layers IV to VI13.
In this study, we identified 116 single units (neurons; see “Methods” for details) during recordings in 8 rats. All the recorded single units were divided into putative pyramidal neurons (PPNs) and putative interneurons (PINs) according to the action potential (AP) width and the time interval between the trough and the peak13,17. However, we have shown previously that the effects of optogenetic pyramidal neuron stimulation are similar for PPNs and PINs; therefore, all the results will be presented for pooled data, while the main results will also be given separately for PPNs and PINs.
In agreement with our previous results, such local optogenetic stimulation of V1 pyramidal neurons resulted in the suppression of spontaneous activity in most neurons (95 out of 116, 81.9%; Supplementary material, Fig. S1A,B), presumably due to activation of local inhibitory neurons13,18; however, in a minority of neurons (16 out of 116, 13.8%; Supplementary material, Fig. S1A–C), LED light pulses induced stimulus-locked APs; thus, these neurons were optically tagged13,19. All such optically tagged neurons were PPNs, confirming that mainly pyramidal neurons were activated during LED stimulation. In 62.5% of these neurons (10 out of 16), the LED-induced response started ≥ 1 ms after the response of distal neurons detected as low-amplitude multiunit APs. Usually, only APs from neurons < 70 μm away from the recording electrode can be used for sorting, while low-amplitude unsorted APs can originate from neurons up to 300 μm away20. Therefore, neurons close to the tip of the optic fiber located ~ 200 μm above the recording electrode contributed to the low-amplitude multiunit signal, and some of these neurons were directly activated by the LED light, providing the start time of the LED-induced response in neurons directly activated by the LED light; thus, later onset suggests activation via multisynaptic pathway. This notion is supported by the large jitter of these subsequently activated neurons (Supplementary material, Fig. S1C). The remaining 6 out of 16 PPNs (37.5%) were activated simultaneously with the multiunit signal, suggesting direct activation by LED light; the decreased jitter of APs in these neurons supports this notion (Supplementary material, Fig. S1C,D). In this study the fraction of presumably directly activated PPNs was much greater than that in a previous study (4 out 27, 15%)13, apparently due to the closer location of the optical fiber tip, ~ 200 μm in this study, compared with the ~ 400 μm in the previous the study13. This comparison suggested that the area of directly activated pyramidal neurons was very small, probably ~ 200 μm wide, or < 0.01 mm3. Assuming that the density of pyramidal neurons in layers IV-VI of the rat visual cortex is close to 100,000 in 1 mm316,21,22, the estimated number of pyramidal neurons directly activated by LED stimulation is likely much less than 1000. The main effect of such trains of LED light pulses was inhibition, and the only source of inhibition in the visual cortex was local inhibitory interneurons23. The absence of recorded inhibitory neurons active during trains of LED light pulses can be explained by several reasons. First, a very low number of inhibitory interneurons was sufficient to produce such an inhibitory effect, and all these neurons were very close to the tip of the optical fiber and thus undetectable as single units. Second, somatostatin interneurons could be responsible for inhibition, and these neurons may produce wide APs24.
In the remaining 100 neurons, LED stimulation usually reduced spontaneous activity (in 95 out of 100 units, 95%), while in a minority of units, there was an increase in spontaneous activity (5 out of 100, 5%). Since in this study we were interested in the simultaneous modulation of ON and OFF responses, only neurons with detectable ON and OFF responses were analyzed (60 in total, 51.7% of all neurons, 45 PPNs, and 15 PINs). We excluded 16 neurons in which LED light induced APs; in addition, we excluded 16 neurons (13.8%) in which our stimulation protocol did not induce detectable visual responses. Furthermore, in 16 neurons, only ON responses could be evoked (13.8%), and in 4 neurons, the RF area of either OFF or ON responses was outside the mapping area. We also excluded 2 units with very slow OFF responses (> 3 s long), and in 2 units, the OFF response was undetectable during LED stimulation.
To evaluate visual response changes during 465 nm LED stimulation, we mapped the RF area by flashing 15° wide bright spots for 0.9 s (Fig. 1C,D). Although moving gratings or moving bars are typically used to identify RF in cortical visual area V1, we opted for flashed stationary spots to distinguish between ON and OFF responses (in our case, OFF responses were generated by the offset of bright spots rather than the onset of dark spots that are usually used for OFF-response generation). In addition, flashing of bright spots allowed mapping of the RF area (Fig. 1F). LED stimulation was switched on ~ 300 ms before visual stimulus presentation and switched off ~ 700 ms after the stimulus offset (Fig. 1E).
No correlation was found between LED-induced changes in the RF area during ON or OFF responses. (A) Summary box plots of RF area changes for OFF and ON responses. (B) The distribution of RF area changes for ON and OFF responses. The Kolmogorov–Smirnov test indicated no statistically significant difference (p < 0.4). C. Changes in the RF area of the ON and OFF responses of single neurons are plotted.
As previously shown for ON responses, the RF area of both ON and OFF responses was reduced during LED stimulation (Fig. 2A). For OFF responses, the median RF area during LED stimulation was 60.7% of that during control stimulation (Q1 = 45.1%, Q3 = 92.2%, n = 60, p < 0.001; Wilcoxon signed rank test), while for ON responses, the median was 77.9% (Q1 = 56.0%, Q3 = 93.1%, n = 60, p < 0.001; Wilcoxon signed rank test). No statistically significant difference was found between changes in ON- and OFF-response RF area (p > 0.17, Kruskal––Wallis test). No difference in the distribution of these changes was detected (Fig. 2B; p > 0.4, Kolmogorov‒Smirnov test).
Surprisingly, despite the similarity in the RF area changes for ON and OFF responses, there was no correlation between RF area changes in individual neurons (Fig. 2C; Spearman’s rank correlation coefficient ρ = 0.144, p > 0.27; n = 60). No correlation was found when only PPNs (ρ = 0.094, p > 0.5, n = 45) or PINs (ρ = 0.196, p > 0.47, n = 15) were considered. This result was also maintained when the outliers were excluded from the PPN data (ρ = 0.137, p > 0.37, n = 43). Since the exclusion of outliers resulted in a distribution that passed the Shapiro‒Wilk test for normality, a linear correlation test could be applied. However, the linear correlation test yielded the same negative result (Pearson correlation coefficient r = 0.139, p > 0.37, n = 43). Approximately 77% of recorded cells could be classified as complex (46 out of 60)9. No correlation between ON and OFF response RF area changes was found when only complex cells were considered (ρ = 0.171, p > 0.25, n = 46) or only PPNs with complex RFs were included (ρ = 0.078, p > 0.5, n = 34). Since the absence of correlation could be produced by noisy measurements, we compared ranked changes in the OFF-response RF area and compared the first quartile of neurons (Q1_OFF), in which the OFF-response RF area was reduced the most, with the fourth quartile of neurons (Q4_OFF), in most of which the OFF-response RF area was not reduced but increased. We reasoned that despite noisy measurements, these two groups of neurons are clearly distinct regarding their changes in the OFF-response RF area: median values were 33% and 121% of the control for changes in the Q1_OFF and Q4_OFF groups of neurons respectively. Thus, if there were any correlation between changes in the RF areas of ON and OFF responses, we should also have a clear difference between changes in the ON response RF area for these two groups of neurons. However, there was almost no such difference: the median value was 78% of the control for Q1_OFF and 84% of the control for Q4_OFF. Note, that for all neurons, the median change in the ON response RF area was 77.9% (Q1 = 56.1%, Q3 = 93.1%); thus, the difference between Q1_OFF and Q4_OFF was negligible, as confirmed by the Kruskal–Wallis test (p = 0.29).
Next, we analyzed changes in the ON and OFF response amplitudes. To this end, the power (squared RMS) was measured for control and LED-modulated visual responses. The amplitudes of both the ON and OFF responses were modestly reduced: for ON responses, the median during LED stimulation was 79.6% of the control (Q1 = 66.9%, Q3 = 92.6%, n = 60), while for OFF responses, the median was 84.6% of the control (Q1 = 66.1%, Q3 = 97.9%, n = 60; Fig. 3A). Again, no differences were found between the ON or OFF response amplitude changes (p > 0.15, Kruskal‒Wallis test) or between the distributions of these changes (p > 0.4, Kolmogorov–Smirnov test; Fig. 3B). However, these changes correlated significantly in individual neurons: ρ = 0.486, p < 0.0003, n = 60 (Fig. 3C). This correlation was also present when PPNs were considered alone (ρ = 0.440, p < 0.003, n = 45) and following removal of the outliers from the PPN data (ρ = 0.439, p < 0.004, n = 44), the latter group passed the Shapiro–Wilk test for normality and thus could be used for the test of linear correlation that yielded a significant correlation (r = 0.401, p < 0.007, n = 44). When PINs were considered alone, the correlation did not reach statistical significance (ρ = 0.436, p > 0.1; n = 15), probably because of the low number of such units.
LED-induced changes in ON and OFF response amplitudes correlated in single neurons. (A) Summary box plots of amplitude changes for OFF and ON responses. (B) The distribution of amplitude changes for ON and OFF responses. The Kolmogorov–Smirnov test indicated no statistically significant difference (p < 0.4). C. Changes in the ON and OFF response amplitudes for single neurons are plotted.
The absence of correlation between changes in the RF area of ON and OFF responses maybe caused by more noisy measurements as compared to the response amplitude measurements. Therefore, by employing bootstrap simulations, we tested how much noise should be added to the amplitude data to obtain the same absence of correlation that was observed in the RF area measurements. These simulations showed that the added noise SD should correspond to 26% of the original value (Supplementary material, Fig. S2A). Such noise SD means that in ~ 1/3d of measurements the added error exceeded 30% of the original value (Supplementary material, Fig. S2B). Taking into account that this is an additional error and it should be independent for both control and stimulation conditions and for ON and OFF responses, it seems to be very unlikely cause of the absence of correlation in the RF area measurements.
Finally, we analyzed changes in the signal-to-noise ratio (SNR) for both ON and OFF responses. As in our previous report, we found an increase in the SNR of visual responses during pyramidal neuron stimulation, by a median value of 96% for ON responses (Q1 = 29.8%, Q3 = 224%, n = 60, p < 0.001, Wilcoxon signed rank test) and by a median value of 242% for OFF responses (Q1 = 27.2%, Q3 = 279%, n = 60, p < 0.001, Wilcoxon signed-rank test). We also tested whether LED-induced changes in the ON and OFF response amplitudes correlated with changes in spontaneous activity. Changes in both the ON and the OFF-response amplitudes were correlated with changes in the RMS spontaneous activity (ρ = 0.476, p < 0.0003, n = 60 for OFF responses and ρ = 0.424, p < 0.0008, n = 60 for ON responses). However, only changes in the ON response RF area were correlated with the LED-induced changes in spontaneous activity (ρ = 0.397, p < 0.002; n = 60), while for OFF-responses such correlation was absent (ρ = 0.215, p > 0.1; n = 60).
Discussion
We demonstrated that, in the rat primary visual cortex, optogenetic stimulation of pyramidal neurons in deep layers IV–VI independently modulates the RF area of ON and OFF responses. Meanwhile, changes in the amplitudes of ON and OFF responses were correlated in single neurons. We have shown previously that such modulation of the RF area during local pyramidal neuron stimulation is mediated through local inhibitory neurons—the optogenetic stimulation of parvalbumin interneurons reproduced these effects13. Therefore, the results presented here suggest that synaptic connections that form negative feedback loops between pyramidal neurons via local inhibitory cells are at least to a degree segregated for ON and OFF responses. Although our methods do not allow identification of the exact pathway of this negative feedback, several observations indicate that this feedback is mediated by the transthalamic corticocortical pathway via dLGN (for detailed discussion see below). Furthermore, average changes in the RF area were similar for ON and OFF responses, suggesting that there is a mechanism that maintains a balance between ON and OFF responses.
Parallel processing of ON and OFF responses starts in the retina and is maintained at least up to dLGN afferents in V1 where they segregate into ON and OFF domains in all studied species4,5,6. Interestingly, this type of segregation is also found in mouse V1 even though there is no clear functional lamination in the mouse dLGN26. How far this segregation is maintained through all cortical circuitry remains the matter of debate. Human evidence and theoretical considerations suggest that it is maintained in most cortical circuits27,28; however, additional direct evidence is lacking. Our data suggest that the feedback circuitry, that connects locally pyramidal neurons, is also segregated, at least to some degree, since it allows independent modulation of the RF area but not the amplitude of the ON and OFF responses. We propose that such independent modulation of ON and OFF response RF area arises through a transthalamic feedback loop, i.e. via dLGN. However, first, we need to discuss what kind of activity is induced by local optogenetic stimulation of layer IV-VI pyramidal neurons.
The activity of the cortical network during local optogenetic pyramidal neuron stimulation was analyzed in detail in our recent study13, in this study we had fewer neurons activated by the LED light pulses, no PINs among them, thus the analysis was more limited. Nevertheless, the combined results allow to have a relatively clear picture about what neurons are activated locally when an optic fiber of 100 μm in diameter is inserted into layers IV–VI of V1 for optogenetic stimulation of pyramidal neurons transduced with ChR2 by employing short CaMKII promoter containing rAAV. In both studies all recorded neurons could be subdivided into those that responded to LED stimulation with stimulus-locked APs (‘optically tagged neurons’) and the remaining cells, in most of which the background activity was strongly suppressed while visual response amplitude was only modestly reduced. It should be stressed that most ‘optically tagged’ putative pyramidal neurons, i.e., PPNs excited by the LED stimulation, were activated indirectly. The LED light, delivered through an optical fiber located above the tetrode used for the recordings, could directly activate only neurons close to the tip of the fiber; meanwhile, at the recording site few hundred microns below, the LED light was dissipated so much by the tissue that it was unable to activate neurons directly29. Thus, all recorded excitatory activity produced by the LED stimulation close ( < ~ 70 µm20) to the recording site was the consequence of synaptic activation of these neurons by the pyramidal neurons that were sufficiently close to the optical fiber tip ( < ~ 100 μm) and only these cells were directly activated by the LED light; these neurons were too far away (> 150 μm) from the recording site to produce any detectable single APs. However, a low amplitude multi-unit signal could be detected during LED stimulation confirming an early, direct activation of pyramidal neurons near the tip of the optical fiber. Moreover, since in this study the recording electrode was only 200 μm away from the optical fiber tip and 62.5% of PPN were activated indirectly even at this distance from the tip, we can infer that most pyramidal neurons activated by the LED light were < 150 μm from the tip, corresponding to the brain volume of < 0.01 mm3, thus fewer than 1000 pyramidal neurons in layers IV or V were activated by our stimulation protocol.
In the study presented here, in our sample of recorded neurons, fewer than 15% of the neurons responded to LED stimulation with a stimulus-locked APs, while in ~ 95% of the remaining neurons spontaneous activity was suppressed, indicating the occurrence of an overall inhibitory effect. This inhibitory effect is likely mediated by local negative feedback circuitries, at least in part, since we have shown in our recent study that local activation of parvalbumin inhibitory interneurons produced a similar effect13. There are at least two distinct mechanisms that could explain our results without the need of dedicated ON and OFF local feedback circuits in the primary visual cortex. Below, these two potential mechanisms are outlined in detail, see also Fig. 4.
At first glance, the most likely explanation of independent modulation of the ON and OFF response RF area is the presence of at least partially segregated negative feedback circuitry inside the cortical area V1 (Fig. 4A). Indeed, in mice a small but significant fraction of interneurons, between 15% and 28%, are monocontrast, i.e., they respond exclusively or almost exclusively to either an increase or a decrease in stimulus brightness, thus producing either ON or OFF responses only24,25,30. Although no such evidence exists for rats, it is reasonable to assume that these features are similar in these two closely related species since dLGN provides the main input to V1 and dLGN structure and function is similar in many respects in rats and mice26,31,32,33,34 (see also below). However, bootstrap simulations of the RF area modulation (Supplementary material, Fig. S2B) show that the magnitude of the modulation component, not correlated between ON and OFF responses, is equal to at least ~ 1/3rd of the total RF area of the corresponding response. Thus, fewer than 1/3rd of monocontrast interneurons present in V1 may be insufficient to explain the observed result.
Two alternative negative feedback loops that can explain the observed independence of ON and OFF response RF area modulation. (A) Local, entirely cortical negative feedback loop could result into partially independent modulation of ON and OFF response RF areas because of the presence of monocontrast interneurons (OFF and ON interneurons in the diagram). (B) A corticothalamic loop that includes OFF- and ON-center relay neurons in dLGN could be sufficient to explain the observed lack of correlation between changes in the ON and OFF response RF areas. Such OFF- and ON-center neurons have been reported to be present in rats (~ 40% of all dLGN neurons, Suresh et al., 2016). For more references and details see the main text. Note that the location of synapses on depicted neurons is symbolic, i.e., no attention was paid whether these synapses are predominantly dendritic, somatic or axonal, see, for instance, Sherman SM, 2016, in Nature Neuroscience, 19(4), 533–541.
An alternative and, in our view, more likely explanation is based on the fact that many cortico-cortical connections are made through cortico-thalamic loops12 (Fig. 4B). dLGN neurons are heavily connected to area V1, and the activation of layer IV–VI pyramidal neurons inevitably activates dLGN neurons and their feedforward connections to the cortex. These feedforward axons form synapses on both pyramidal neurons and interneurons23; therefore, they can produce such an overall inhibitory effect, and these effects are quite rapid since the peak amplitude of visual responses can be affected without any clear change in their timing35. The specific properties of dLGN neurons and their projections can explain our results.
dLGN neurons have very stereotyped ON and OFF response fields32,36,37: in primates and carnivores most dLGN neurons have either ON or OFF central RF area surrounded by an annulus of the opposite sign. Moreover, the opposite sign stimulus produces an inhibitory response, the so-called push-pull structure of excitation and inhibition38. It has been shown that in rodents, both mice and rats, ~ 40% of dLGN neurons have the same ON- and OFF-center RF fields and a similar push-pull arrangement for excitation and inhibition32. Since a large body of evidence shows that this type of dLGN neurons is responsible for ON and OFF domain formation in dLGN afferents in the cortex of cats4,7, and these domains are present also in other species, including mink39, ferret40, monkey41 and mouse6, it is reasonable to assume that also in rats such segregation of dLGN afferents exists although no data is yet available (note, that in mice no dLGN lamination according to ON and OFF responses is found indicating that such a lamination is not necessary for ON and OFF domain formation in the dLGN afferents). As mentioned above, simulations show that our results can be reproduced when the magnitude of independent RF area modulation for ON and OFF responses is equivalent to ~ 1/3rd of the original RF area value. Since in rats ~ 40% of dLGN neurons are either ON- or OFF-center cells, such cortico-thalamo-cortical feedback can explain the observed result.
It should be noted that the main V1 output to the dLGN is formed by layer VI pyramidal neurons10,42 and we stimulated mainly layer IV and V cells. However, there are excitatory inter-layer connections between layer IV–VI pyramidal neurons23, that is supported by the presence in our recordings of PPNs stimulated indirectly by the LED light29. Thus, it is very likely that our stimulation protocol produced activation of layer VI pyramidal neurons that in turn activated the whole circuit from V1 to dLGN and then back to V1 (Fig. 4B).
Finally, it should be noted that we cannot entirely rule out the possibility that the measurement error was much larger for the RF area than for the response amplitude and this larger error prevented the detection of correlation between changes in the ON and OFF response RF area. However, bootstrap simulations show that the minimal error amplitude should be such that in ~ 1/3rd of measurements the error exceeds 30% of the real RF area value (Supplementary material, Fig. S2B). Moreover, the errors should be independent for ON and OFF responses measured for the same unit (correlated errors would increase correlation strength). Thus, limitations related to the measurement procedure such as grid size used for the visual stimulus presentation or the visual stimulus size will not affect the results since these limitations apply equally to the ON and the OFF response area measurements. This notion is supported by the fact that no correlation in the RF area changes were detected also when only neurons with the strongest modulation of their area were considered. Therefore, it is difficult to imagine a situation when RF area measurements produce such a large error independently for ON and OFF responses.
One important conclusion that can be drawn from our results is that modulation of the RF area by pyramidal neuron stimulation is unlikely due to the ‘iceberg effect’. Indeed, this hypothesis posits that activation of local interneurons, directly or via pyramidal neuron stimulation, hyperpolarizes the membrane potential of a neuron; therefore, stronger synaptic inputs are required to reach the AP threshold44,45. Since, for a single neuron, such an increased synaptic drive for AP generation will be the same for ON and OFF responses, one expects at least some degree of correlation between ON and OFF response modulation. In fact, for the response amplitude, such a correlation was present (Fig. 3). This correlation can likely be attributed to the iceberg effect since there was a strong correlation between changes in the amplitude of both the ON and OFF responses and the LED-induced changes in the spontaneous activity, an indicator of the strength of the LED-induced inhibitory interneuron activation. In contrast, only changes in the ON response RF area correlated with LED-induced changes in spontaneous activity. Thus, it is likely that several effects, both post- and presynaptic, such as modulation of cortico-thalamic connections, contribute to the RF area modulation induced by the local optogenetic stimulation of pyramidal neurons.
Conclusions
We demonstrated that, in the deep layers of V1, local optogenetic stimulation of pyramidal neurons modulates the RF area of ON and OFF responses independently. Thus, the feedback circuits between cortical pyramidal neurons for ON and OFF responses are at least partially separate. This effect is likely due to the transthalamic corticocortical feedback connection acting via the thalamic nucleus of the dLGN. These results demonstrated that even cortical neurons can shape their ON and OFF response properties independently. Our results provide further evidence that much of the circuitry in the visual cortex processes ON and OFF responses in parallel.
Methods
In vivo injection of viral particles
All procedures were carried out following the European Communities Council Directive of 22 September 2010 on the protection of animals used for scientific purposes (2010/63/EEC) and were approved by the Animal Care and Use Committee of the State Food and Veterinary Service of Lithuania (No. G2-147 of May 13, 2020). All methods involving animals were carried out following ARRIVE guidelines46,47,48. In this type of research, there was no control group of animals because we compared the effects of the 465 nm LED on and off the same animal. No 465 nm LED effects were ever observed in noninjected animals or in animals in which sham virus injections were performed, i.e., all surgical procedures for virus injections but a phosphate-buffered saline without a virus was injected.
For channelrhodopsin expression mostly in pyramidal neurons, a short CaMKII promoter was used15; the promoter sequence was obtained by subcloning the plasmid pAAV-CaMKIIa-mCherry, which was a gift from Karl Deisseroth (Addgene plasmid #114469; http://n2t.net/addgene:114469; RRID: Addgene_114469). In addition, the Pv poly(A) sequence was substituted with a standard hHG poly(A) signal. Viral particles were purchased from Creative Biogene (Shirley, NY 11967, USA), and the concentration used was 10^13 vg/mL.
Standard procedures were used for virus injections
For virus injections, 8 Wistar rats of both sexes (4 males, weighing 290 to 500 g, 2–4 months old, and 4 females, weighing 240 to 290 g, 2.5–3 months old), were anesthetized with a mixture of xylazine chloride (Eurovet Animal Health B.V., Bladel, Netherlands, 10 mg kg− 1), ketamine (Richter Pharma AG, Wels, Austria, 60 mg kg− 1) and butorphanol (Richter Pharma AG, Wels, Austria, 0.4 mg kg− 1), which were delivered intraperitoneally. The depth of anesthesia was monitored by testing for the absence of a hind limb withdrawal reflex following a pinch of the paw. To maintain the depth of anesthesia, additional doses of the mixture were used as needed for the duration of the experiment. Under anesthesia, the body temperature was maintained at 36–38 °C with a heating pad. The anesthetized animal was placed in a stereotaxic apparatus (World Precision Instruments, Sarasota, FL, USA). A small craniotomy (approximately 1 × 1 mm) was made in the parietal bone immediately above the visual cortex areas V1/V2 (the injection coordinates were ~ 2 mm rostral to the lambda and ~ 3 mm lateral to the midline). The dura was left intact. A volume of 0.8 µL of the particle mixture was injected (1013 particles/mL, corresponding to a total of 8 × 109 injected viral particles). Injections were performed with a pipette made from a borosilicate glass capillary and attached to a 10 µL Hamilton syringe (CH-7402 Bonaduz, GR, Switzerland); the pipette tip size was ~ 50 μm. The injection depth was between 700 μm and 1200 μm. Following pipette withdrawal, the scar was removed, and the scalp was closed with sutures.
The rats were returned to their home cages after the injection. Care was taken to avoid any infections. Recordings were performed 19 to 38 days postinjection. The time postinjection affects the expression level of transduced cells, and after the initial increase, the expression usually plateaus after 4 to 6 weeks49. No correlation was found between the age or sex of the rats. Therefore, data from all rats of different ages and sexes were pooled.
Extracellular recording procedures
All procedures were carried out in accordance with the European Communities Council Directive of 22 September 2010 on the protection of animals used for scientific purposes (2010/63/EEC) and were approved by the Animal Care and Use Committee of the State Food and Veterinary Service of Lithuania (No. G2-147 of May 13, 2020).
Wistar rats, purchased from the Vivarium of the Veterinary Academy of the Lithuanian University of Health Sciences (https://lsmu.lt/en/about-lsmu/structure/general-departments-of-the-university/other-departments/vivarium/), were used in this study. Although Wistar rats are albino and, compared to those of pigmented rats, their visual acuity is approximately twofold lower50, the receptive field sizes are similar to those reported for pigmented rats51,52,53. In addition, albino rats exhibit greater sensitivity to dim stimuli, and visual responses of superior colliculus neurons are largely the same in both pigmented and albino rats54,55. We are not aware of a similar comparison of neuronal responses in the visual cortex.
Standard procedures were used for single-unit recordings56,57. Briefly, rats were anesthetized with urethane (Alfa Aesar—Thermo Fisher, Karlsruhe, Germany; 1.5–1.8 g kg− 1) aided with butorphanol (Richter Pharma AG, Wels, Austria; 0.4 g kg− 1), both of which were delivered intraperitoneally58. The depth of anesthesia was monitored by testing for the absence of a hind limb withdrawal reflex following a pinch of the paw. To maintain the depth of anesthesia, additional doses of butorphanol were used during the duration of the experiment. Under anesthesia, the body temperature was maintained at 36–38 °C with a heating pad. The anesthetized animal was placed in a modified stereotaxic apparatus (World Precision Instruments, Sarasota, FL, USA) that enabled an unobstructed view of the left eye. Eye gel (Artelac Nighttime Gel, Baush & Lomb Incorporated, Berlin, Germany) was applied to avoid eye drying. Although eye movements are rarely problematic in anesthetized rats59, to prevent eye movements and maintain lid opening, miniature hooks were inserted between the conjunctiva of the inner eyelids and the sclera and then attached to the stereotaxic frame with a thread. Preliminary experiments showed that such hooks completely eliminated occasional drifts in the receptive field position that were sometimes observed without hooks; differences in response amplitude to stimuli at centers as little as 1 degree apart could be detected57. To dilate the pupils, 0.5% atropine (Sigma–Aldrich Chemie GmbH, Taufkirchen, Germany) was applied to the cornea surface. Eye dilation increases light scattering, which may confound the measurements of the receptive field size. However, pupil dilation or systemic atropine or D-turbocurarine, which both also result in pupil dilation, are often applied during such experiments, allowing us to compare our data with those of other studies straightforwardly51,53,60,61,62.
For recording, a small craniotomy (approximately 2 × 2 mm) was made in the parietal bone, and a small incision was made in the dura mater. Tetrodes from Thomas Recording (Giessen, Germany) were used to acquire action potential data, while a blue PlexBright LED measuring 465 nm (Plexon, Dallas, Texas, USA) was used to excite neurons expressing channelrhodopsins. The LED light was delivered via a 105 μm optic fiber (Thorlabs GmbH, Munich, Germany) glued to the tetrode such that the fiber tip was located ~ 200 μm above the tetrode tip (maximal nominal power at the tip ~ 5 mW, the measured maximal irradiance 130 mW/mm2 at the fiber tip; the actual power was adjusted as described in the Results section). We have shown previously that, in such an arrangement of the optical fiber and the recording tetrode, no optoelectrical artifacts were noted when rats were injected with a virus containing an eGFP transgene but not ChR229. Electrodes were typically placed 1.5–2.5 mm rostral to lambda and 2.5–3.5 mm lateral to the midline and then lowered perpendicular to the cortical surface by employing a microdrive to a depth of 890–1600 μm. We have shown previously that lesions at the end of the experiment confirmed that all such recordings were in deep layers of the cortex (layers IV–VI)29.
For data acquisition, a 4-channel differential amplifier was used (EX4-400, DAGAN, Minneapolis, MN, USA) with bandpass filters set to 300 − 10,000 Hz. The data were acquired via a National Instrument DAQ card PCI-MIO-16E-1 (6070E) connected to a PC at a 40 kHz sampling frequency and visualized by employing a custom program written in the LabVIEW environment (National Instruments, Austin, TX, USA). Although the sampling rate of 40 kHz in theory is excessive, tests have shown that this approach improves the signal-to-noise ratio, presumably because the real signal has very high frequency components that are not completely cancelled out by the analogous filters of the amplifier.
Visual stimulation
An LED backlit LCD monitor (frame rate 60 Hz, 58 cm by 28 cm) was used for image presentation and was placed 16 cm from the right eye. The bottom of the monitor was slightly (~ 5 cm) below the rat eye level. The monitor was inclined at a 45° angle to the rat’s longitudinal axis in the horizontal plane. In the vertical plane, the monitor was inclined at 30° toward the rat to cover a wider range of vertical angles. The full screen subtended 110° horizontally and approximately 80° vertically. At the center of the screen, 1 cm corresponded to ~ 3.6° of the visual angle. During the experiments, the stimulus size was entered as a fraction of the monitor height; 1/10th of the monitor height was considered to correspond to 10° of the visual angle (actual calculations show that it is 2.8 cm × 3.4° cm-1 = 10.08°). Rat eyes are normally emmetropic and can see sufficiently well from 7 cm to infinity63. Apparently, rat vision is mainly used to discern large predators64 and has low acuity even for short distances65. The monitor had 1920 × 1080 image pixels or > 30 pixels/cm corresponding to a minimal stimulus size of < 0.1°, which is well below the visual acuity of ~ 0.5°–1° found in rodents65. Images were generated by employing the open-source software package PsychoPy, which is controlled by an in-house program written in Python and synchronized with recordings via the LabVIEW environment (National Instruments, Austin, TX, USA).
All the visual stimuli used were bright images (~ 30–45 cd/m2 at the rat’s eye level) presented on a dark gray background (~ 0.3–0.45 cd/m2).
The following visual stimulation protocol was used. The receptive field size and location were determined by flashing 15° wide bright round spots for 900 ms followed by a 3500 ms gap (4.4 s interstimulus interval, ISI) on an 18 × 11 grid in a quasi-random fashion; in some cases, smaller grids were used. Usually, the grid spacing was ~ 15°. Clearly, the use of such a large stimulus of 15° was chosen to enable distinguishable visual responses, but the disadvantage was that the minimal estimated RF size was approximately 15° wide. However, in rats, receptive fields are rarely less than 10° in diameter51,53; moreover, we observed a reduction in the receptive field area upon 465 nm LED stimulation; thus, if the actual size of the reduced receptive field was < 10°, our data underestimate only the 465 nm LED stimulation effects. Each grid location was tested 5 to 16 times. Following a preliminary estimate of the receptive field location and size, 465 nm LED stimulation (Plexon LED module driven by PlexBright LD-1, Plexon, Dallas, Texas, USA) was applied during visual stimulus presentation in a quasi-random fashion during the same run as the control visual stimulus presentations.
465-nm LED stimulation
The 465 nm LED was controlled by the analog signal generated by LabView software and fed into the PlexBright LD-1 analog control input. Typically, the 465 nm LED stimulus started 250–350 ms before visual stimulus onset and lasted for ~ 2000 ms. We mostly used 10 ms long 465 nm LED pulses delivered every 110 ms (a 100 ms interval between pulses). We previously showed that 465 nm LED stimulation had no effect on rats injected with a virus carrying only fluorescent probe with no channelrhodopsin transgen13.
Spike detection and sorting
Most analyses of recorded traces were performed with custom-written routines by employing Igor Pro software (version 6.3.7.2, Wavemetrics, Lake Oswego, Oregon, USA; https://www.wavemetrics.com/). MATLAB (version R2015b/8.6.0.267246, MathWorks, Natick, MA, USA; https://www.mathworks.com/products/matlab.html) was used for principal component analysis (PCA), while data clustering analysis was performed with the publicly available KlustaKwik software (http://klustakwik.sourceforge.net). Single units were first detected as threshold-crossing events, while the threshold was set approximately 4 standard deviations from the baseline. The threshold was lowered by 30% when synchronous events occurred on 2 or more traces. Care was taken that no artifacts were present in the trace. For each detected event between 1.1 ms and 1.6 ms from each channel, data were collected, for a total of 44–62 data points: 14–20 before and 30–45 after the negative peak. The window duration was determined by the shape of the action potentials. For spike sorting, each channel’s data were reduced by principal component analysis (PCA) via MATLAB software. Then, spike classes were determined by finding clusters of 2–5 PCAs from each channel by employing KlustaKwik software. The quality of sorting was verified by the occurrence of a refractory period with auto and cross-correlograms66.
Data analysis
A response to a visual stimulus was considered to be present/detectable if the frequency of action potentials during visual stimulus presentation exceeded the background firing frequency by > 3 standard deviations of the background noise. The background firing rate and standard deviation were calculated for a 1 s time interval before visual stimulus presentation, and 100 ms binning was applied during these calculations.
The average spike rates during visual stimulus presentation were calculated for each location of the stimulus. Then, a two-dimensional Gaussian function was used to fit the spatial distribution of these spike rate values60:
where x’ and y’ are the polar transformations of space coordinates x and y, respectively, along which the Gaussian distribution is oriented; a and b are standard deviations of the Gaussian function in the direction of the x′ and y′ axes, respectively; and A is the predicted maximal spike rate in the RF center. The RF area was calculated as π\(\:*\)a\(\:*\)b (the ellipse area with semiaxes a and b).
Simple and complex cells were identified according to the overlap between ON and OFF receptive fields. To quantify the separation between these fields, an overlap index (OI) was calculated for cells exhibiting both ON and OFF responses30:
where d is the distance between the centers of two ellipses and W1 and W2 are the widths of them, respectively, which are the segments of the line that connects the two centers intercepted by the ellipses. For complex cells OI was > 0.330.
Noise and signal-to-noise ratio calculations
The signal power was assessed as the mean square of individual action potential numbers during visual stimulus presentation, that is, the mean square of all responses during visual stimulus presentation in a location inducing a maximum visual response. The number of such presentations varied between 5 and 16. If the average number of action potentials during visual response differed by less than 20% for two different locations, then the response to visual stimulus presentation in both locations was used to calculate the signal power. Only spontaneous noise (when no visual stimulus was presented) was considered in this study. In this case, at least 300 brief sections of ~ 0.5 s of spontaneous activity just before visual stimulus presentation were used for calculations, and the noise power was calculated as the mean square of action potential numbers.
Experimental design and statistical analyses
Unless stated otherwise, all the results are presented as the average ± SD plus the median for nonnormally distributed data. Most of the data presented here are from repeated measurements or matched pairs (465 nm LED on/off). The Shapiro–Wilk test of normality showed significant deviation from a normal distribution for a few data sets. Therefore, a nonparametric Wilcoxon signed-ranks 2-tailed test was used for matched pairs. For independent samples, a nonparametric Kruskal––Wallis 2-tailed test for a fixed-effects model was used. In the figures, the population data are presented as box plots (Q1, Q2, and Q3; whiskers indicate the 5–95% range).
Data availability
The author declares that all the data necessary for evaluation of the statements of this paper are present in the manuscript. However, any additional reasonable requests for data will be granted by the corresponding author.
References
Baden, T. et al. The functional diversity of retinal ganglion cells in the mouse. Nature 529, 345–350. https://doi.org/10.1038/nature16468 (2016).
Schiller, P. H. The ON and OFF channels of the visual system. Trends Neurosci. 15, 86–92. https://doi.org/10.1016/0166-2236(92)90017-3 (1992).
Kim, U. S., Mahroo, O. A., Mollon, J. D. & Yu-Wai-Man, P. Retinal ganglion cells-diversity of cell types and clinical relevance. Front. Neurol. 12, 661938. https://doi.org/10.3389/fneur.2021.661938 (2021).
Reid, R. C. & Alonso, J. M. Specificity of monosynaptic connections from thalamus to visual cortex. Nature 378, 281–284. https://doi.org/10.1038/378281a0 (1995).
Smith, G. B., Whitney, D. E. & Fitzpatrick, D. Modular representation of luminance polarity in the superficial layers of primary visual cortex. Neuron 88, 805–818. https://doi.org/10.1016/j.neuron.2015.10.019 (2015).
Tring, E., Duan, K. K. & Ringach, D. L. ON/OFF domains shape receptive field structure in mouse visual cortex. Nat. Commun. 13, 2466. https://doi.org/10.1038/s41467-022-29999-7 (2022).
Kremkow, J., Jin, J., Wang, Y. & Alonso, J. M. Principles underlying sensory map topography in primary visual cortex. Nature 533, 52–57 (2016).
Ji, X. Y. et al. Thalamocortical innervation pattern in mouse auditory and visual cortex: laminar and cell-type specificity. Cereb. Cortex. 26, 2612–2625. https://doi.org/10.1093/cercor/bhv099 (2016).
Li, Y. T., Liu, B. H., Chou, X. L., Zhang, L. I. & Tao, H. W. Synaptic basis for differential orientation selectivity between complex and simple cells in mouse visual cortex. J. Neurosci. 35, 11081–11093. https://doi.org/10.1523/jneurosci.5246-14.2015 (2015).
Busse, L. In Handbook of Behavioral Neuroscience, vol. 27, 53–68 (Elsevier, 2018).
Lamme, V. A., Supèr, H. & Spekreijse, H. Feedforward, horizontal, and feedback processing in the visual cortex. Curr. Opin. Neurobiol. 8, 529–535. https://doi.org/10.1016/s0959-4388(98)80042-1 (1998).
Sherman, S. M. & Guillery, R. W. Distinct functions for direct and transthalamic corticocortical connections. J. Neurophysiol. 106, 1068–1077. https://doi.org/10.1152/jn.00429.2011 (2011).
Baranauskas, G., Rysevaite-Kyguoliene, K., Sabeckis, I., Tkatch, T. & Pauza, D. H. Local stimulation of pyramidal neurons in deep cortical layers of anesthetized rats enhances cortical visual information processing. Sci. Rep. 14, 22862. https://doi.org/10.1038/s41598-024-73995-4 (2024).
Nagel, G. et al. Light activation of channelrhodopsin-2 in excitable cells of caenorhabditis elegans triggers rapid behavioral responses. Curr. Biol. 15, 2279–2284. https://doi.org/10.1016/j.cub.2005.11.032 (2005).
Dittgen, T. et al. Lentivirus-based genetic manipulations of cortical neurons and their optical and electrophysiological monitoring in vivo. Proc. Natl. Acad. Sci. U. S. A. 101, 18206–18211. https://doi.org/10.1073/pnas.0407976101 (2004).
Narayanan, R. T., Udvary, D. & Oberlaender, M. Cell type-specific structural organization of the six layers in rat barrel cortex. Front. Neuroanat. 11, 91. https://doi.org/10.3389/fnana.2017.00091 (2017).
Barthó, P. et al. Characterization of neocortical principal cells and interneurons by network interactions and extracellular features. J. Neurophysiol. 92, 600–608. https://doi.org/10.1152/jn.01170.2003 (2004).
Butovas, S. & Schwarz, C. Spatiotemporal effects of microstimulation in rat neocortex: a parametric study using multielectrode recordings. J. Neurophysiol. 90, 3024–3039. https://doi.org/10.1152/jn.00245.2003 (2003).
Kvitsiani, D. et al. Distinct behavioural and network correlates of two interneuron types in prefrontal cortex. Nature 498, 363–366. https://doi.org/10.1038/nature12176 (2013).
Henze, D. A. et al. Intracellular features predicted by extracellular recordings in the hippocampus in vivo. J. Neurophysiol. 84, 390–400. https://doi.org/10.1152/jn.2000.84.1.390 (2000).
Keller, D., Erö, C. & Markram, H. Cell densities in the mouse brain: A systematic review. Front. Neuroanat. 12, 83. https://doi.org/10.3389/fnana.2018.00083 (2018).
Bjerke, I. E. et al. Densities and numbers of Calbindin and parvalbumin positive neurons across the rat and mouse brain. iScience 24, 101906. https://doi.org/10.1016/j.isci.2020.101906 (2021).
Binzegger, T., Douglas, R. J. & Martin, K. A. A quantitative map of the circuit of Cat primary visual cortex. J. Neurosci. 24, 8441–8453. https://doi.org/10.1523/jneurosci.1400-04.2004 (2004).
Ma, W. P. et al. Visual representations by cortical somatostatin inhibitory neurons–selective but with weak and delayed responses. J. Neurosci. 30, 14371–14379. https://doi.org/10.1523/jneurosci.3248-10.2010 (2010).
Mesik, L. et al. Functional response properties of VIP-expressing inhibitory neurons in mouse visual and auditory cortex. Front. Neural Circuits. 9 https://doi.org/10.3389/fncir.2015.00022 (2015).
Kerschensteiner, D. & Guido, W. Organization of the dorsal lateral geniculate nucleus in the mouse. Vis. Neurosci. 34, E008. https://doi.org/10.1017/s0952523817000062 (2017).
Clark, D. A. et al. Flies and humans share a motion estimation strategy that exploits natural scene statistics. Nat. Neurosci. 17, 296–303. https://doi.org/10.1038/nn.3600 (2014).
Wei, J. et al. Understanding contrast perception in amblyopia: a psychophysical analysis of the ON and OFF visual pathways. Front. Psychol. 15, 1494964. https://doi.org/10.3389/fpsyg.2024.1494964 (2024).
Baranauskas, G., Rysevaite-Kyguoliene, K., Sabeckis, I. & Pauza, D. H. Saturation of visual responses explains size tuning in rat collicular neurons. Eur. J. Neurosci. 57, 285–309. https://doi.org/10.1111/ejn.15877 (2023).
Liu, B. H. et al. Visual receptive field structure of cortical inhibitory neurons revealed by two-photon imaging guided recording. J. Neurosci. 29, 10520–10532. https://doi.org/10.1523/jneurosci.1915-09.2009 (2009).
Reese, B. E. Hidden lamination’ in the dorsal lateral geniculate nucleus: the functional organization of this thalamic region in the rat. Brain Res. 472, 119–137. https://doi.org/10.1016/0165-0173(88)90017-3 (1988).
Suresh, V. et al. Synaptic contributions to receptive field structure and response properties in the rodent lateral geniculate nucleus of the thalamus. J. Neurosci. 36, 10949–10963 (2016).
Peters, A. & Feldman, M. L. The projection of the lateral geniculate nucleus to area 17 of the rat cerebral cortex. I. General description. J. Neurocytol. 5, 63–84. https://doi.org/10.1007/bf01176183 (1976).
Peters, A. & Kara, D. A. The neuronal composition of area 17 of rat visual cortex. IV. The organization of pyramidal cells. J. Comp. Neurol. 260, 573–590. https://doi.org/10.1002/cne.902600410 (1987).
Spacek, M. A. et al. Robust effects of corticothalamic feedback and behavioral state on movie responses in mouse dLGN. Elife. 11. https://doi.org/10.7554/eLife.70469 (2022).
Fukuda, Y., Sumitomo, I., Sugitani, M. & Iwama, K. Receptive-field properties of cells in the dorsal part of the albino rat’s lateral geniculate nucleus. Jpn. J. Physiol. 29, 283–307 (1979).
Zhao, X., Chen, H., Liu, X. & Cang, J. Orientation-selective responses in the mouse lateral geniculate nucleus. J. Neurosci. 33, 12751–12763 (2013).
Hirsch, J. A. Synaptic physiology and receptive field structure in the early visual pathway of the Cat. Cereb. Cortex. 13, 63–69. https://doi.org/10.1093/cercor/13.1.63 (2003).
McConnell, S. K. & LeVay, S. Segregation of on- and off-center afferents in Mink visual cortex. Proc. Natl. Acad. Sci. U. S. A. 81, 1590–1593. https://doi.org/10.1073/pnas.81.5.1590 (1984).
Zahs, K. R. & Stryker, M. P. Segregation of ON and OFF afferents to ferret visual cortex. J. Neurophysiol. 59, 1410–1429. https://doi.org/10.1152/jn.1988.59.5.1410 (1988).
Li, P., Garg, A. K., Zhang, L. A., Rashid, M. S. & Callaway, E. M. Cone opponent functional domains in primary visual cortex combine signals for color appearance mechanisms. Nat. Commun. 13, 6344. https://doi.org/10.1038/s41467-022-34020-2 (2022).
Olsen, S. R., Bortone, D. S., Adesnik, H. & Scanziani, M. Gain control by layer six in cortical circuits of vision. Nature 483, 47–52. https://doi.org/10.1038/nature10835 (2012).
Kullander, K. & Topolnik, L. Cortical disinhibitory circuits: cell types, connectivity and function. Trends Neurosci. 44, 643–657. https://doi.org/10.1016/j.tins.2021.04.009 (2021).
Carandini, M. Melting the iceberg: contrast invariance in visual cortex. Neuron 54, 11–13. https://doi.org/10.1016/j.neuron.2007.03.019 (2007).
Shapiro, J. T., Gosselin, E. A. R., Michaud, N. M. & Crowder, N. A. Activating parvalbumin-expressing interneurons produces iceberg effects in mouse primary visual cortex neurons. Neurosci. Lett. 786, 136804. https://doi.org/10.1016/j.neulet.2022.136804 (2022).
Kilkenny, C., Browne, W., Cuthill, I. C., Emerson, M. & Altman, D. G. Animal research: reporting in vivo experiments: the ARRIVE guidelines. Br. J. Pharmacol. 160, 1577–1579. https://doi.org/10.1111/j.1476-5381.2010.00872.x (2010).
Percie du Sert. Reporting animal research: explanation and elaboration for the ARRIVE guidelines 2.0. PLoS Biol. 18, e3000411. https://doi.org/10.1371/journal.pbio.3000411 (2020).
Percie du Sert. The ARRIVE guidelines 2.0: updated guidelines for reporting animal research. J. Cereb. Blood Flow. Metab. 40, 1769–1777. https://doi.org/10.1177/0271678x20943823 (2020).
Bedbrook, C. N., Deverman, B. E. & Gradinaru, V. Viral strategies for targeting the central and peripheral nervous systems. Annu. Rev. Neurosci. 41, 323–348. https://doi.org/10.1146/annurev-neuro-080317-062048 (2018).
Prusky, G. T., Harker, K. T., Douglas, R. M. & Whishaw, I. Q. Variation in visual acuity within pigmented, and between pigmented and albino rat strains. Behav. Brain Res. 136, 339–348. https://doi.org/10.1016/s0166-4328(02)00126-2 (2002).
Shaw, C., Yinon, U. & Auerbach, E. Receptive fields and response properties of neurons in the rat visual cortex. Vis. Res. 15, 203–208. https://doi.org/10.1016/0042-6989(75)90208-4 (1975).
Burne, R. A., Parnavelas, J. G. & Lin, C. S. Response properties of neurons in the visual cortex of the rat. Exp. Brain Res. 53, 374–383. https://doi.org/10.1007/bf00238168 (1984).
Girman, S. V., Sauvé, Y. & Lund, R. D. Receptive field properties of single neurons in rat primary visual cortex. J. Neurophysiol. 82, 301–311. https://doi.org/10.1152/jn.1999.82.1.301 (1999).
de Herreros, P., Green, D. G. & Muñoz Tedó, C. Visual thresholds in albino and pigmented rats. Vis. Neurosci. 9, 409–414. https://doi.org/10.1017/s0952523800010816 (1992).
Thomas, B. B., Aramant, R. B., Sadda, S. R. & Seiler, M. J. Light response differences in the superior colliculus of albino and pigmented rats. Neurosci. Lett. 385, 143–147. https://doi.org/10.1016/j.neulet.2005.05.034 (2005).
Bytautiene, J. & Baranauskas, G. Rat superior colliculus neurons respond to large visual stimuli flashed outside the classical receptive field. PLoS One. 12, e0174409 (2017).
Bytautiene, J. & Baranauskas, G. Experimentally derived model shows that adaptation acts as a powerful Spatiotemporal filter of visual responses in the rat collicular neurons. Sci. Rep. 8, 8942. https://doi.org/10.1038/s41598-018-27331-2 (2018).
Lomas, L. M., Barrett, A. C., Terner, J. M., Lysle, D. T. & Picker, M. J. Sex differences in the potency of kappa opioids and mixed-action opioids administered systemically and at the site of inflammation against capsaicin-induced hyperalgesia in rats. Psychopharmacol. (Berl). 191, 273–285. https://doi.org/10.1007/s00213-006-0663-1 (2007).
Humphrey, N. K. Responses to visual stimuli of units in the superior colliculus of rats and monkeys. Exp. Neurol. 20, 312–340 (1968).
Wang, L., Sarnaik, R., Rangarajan, K., Liu, X. & Cang, J. Visual receptive field properties of neurons in the superficial superior colliculus of the mouse. J. Neurosci. 30, 16573–16584. https://doi.org/10.1523/jneurosci.3305-10.2010 (2010).
Razak, K. A. & Pallas, S. L. Neural mechanisms of stimulus velocity tuning in the superior colliculus. J. Neurophysiol. 94, 3573–3589. https://doi.org/10.1152/jn.00816.2004 (2005).
Li, X., Sun, C. & Shi, L. Comparison of visual receptive field properties of the superior colliculus and primary visual cortex in rats. Brain Res. Bull. 117, 69–80. https://doi.org/10.1016/j.brainresbull.2015.07.007 (2015).
Powers, M. K. & Green, D. G. Single retinal ganglion cell responses in the dark-reared rat: grating acuity, contrast sensitivity, and defocusing. Vis. Res. 18, 1533–1539 (1978).
Wallace, D. J. et al. Rats maintain an overhead binocular field at the expense of constant fusion. Nature 498, 65–69. https://doi.org/10.1038/nature12153 (2013).
Prusky, G. T., West, P. W. & Douglas, R. M. Behavioral assessment of visual acuity in mice and rats. Vis. Res. 40, 2201–2209 (2000).
Harris, K. D., Henze, D. A., Csicsvari, J., Hirase, H. & Buzsaki, G. Accuracy of tetrode Spike separation as determined by simultaneous intracellular and extracellular measurements. J. Neurophysiol. 84, 401–414 (2000).
Acknowledgements
This work was supported by the Faculty of Medicine of the Lithuanian University of Health Sciences.
Author information
Authors and Affiliations
Contributions
G.B. designed and performed the research, analyzed the data, and wrote the paper. J.K. and E.S. analyzed the data and edited the manuscript. All the authors approved the final version of the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Kozal, J., Simkute, E. & Baranauskas, G. Optogenetic stimulation of pyramidal neurons in the rat primary visual cortex modulates ON- and OFF- receptive field areas independently. Sci Rep 15, 44838 (2025). https://doi.org/10.1038/s41598-025-29091-2
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-29091-2