Abstract
Lily seeds are commonly used for commercial production and genetic breeding, and their germination rates under different light conditions can be improved by chemical treatments. However, there is a lack of research to systematically screen the pre-treatment methods that are most suitable for various lily species. The study selected six lily species (Lilium pumilum DC., Lilium davidii var. unicolor, Lilium auratum var. platyphyllum, Lilium lancifolium Thunb., Lilium distichum Nakai, and Lilium pensylvanicum Ker Gawl.) and employed a randomized complete block three-factor factorial design, with three factors being species, chemical treatments (0.1% KNO₃, 0.1% K3PO4, and different concentrations of GA3), and light duration (0 h/d, 12 h/d, and 24 h/d). Each treatment included three replicates. Before sowing, the seeds were disinfected with 2% sodium hypochlorite solution for 15 min and rinsed with distilled water, then soaked in the corresponding solution for 1 h. Treated seeds were placed in petri dishes and incubated in an artificial climate chamber at 25 °C, with moist filter paper maintained and light conditions set accordingly. Germination was monitored continuously for 40 days, and the germination start time, completion time, and germination rate were recorded. Germination was defined as radicle emergence through the seed coat by 1–2 mm. The results showed that different lily species responded significantly differently to chemical treatment and light conditions. GA3 treatment generally improved germination performance, and some species also showed a promoting effect under KNO3 or K3PO4 treatment. Comprehensive analysis revealed that L. lancifolium performed consistently well under multiple treatment conditions and can be selected as the preferred species for stimulation treatment.
Similar content being viewed by others
Introduction
Seed germination is a critical stage in the plant life cycle, directly affecting seedling establishment and final yield formation1. Seed priming, a widely used pre-sowing treatment method, has been extensively applied in horticulture and crop production. By activating seed metabolism without triggering full germination, priming improves germination rate, uniformity, and seedling vigor. Studies have shown that different types of priming agents promote germination by regulating endogenous hormones, enhancing water uptake, and activating hydrolytic enzymes2,3.
Lily (Lilium spp.), an important ornamental and medicinal plant, often exhibits physiological dormancy or slow germination, leading to poor seedling quality and low propagation efficiency4. Previous research has indicated that chemical priming treatments can improve lily seed germination by increasing osmotic potential or supplementing essential nutrients5. For example, potassium nitrate (KNO3), as a nitrate-based nitrogen source, regulates hormonal balance and promotes seed coat softening. Phosphate compounds participate in energy metabolism and play a key regulatory role during early germination6,7. However, the response patterns of different lily species to these treatments remain unclear, and comparative studies in this area are still limited.
In addition, light is another key factor affecting seed germination, often acting as a promoter for many photophilic seeds8. However, studies have also shown that light can have inhibitory or neutral effects in certain plant species9. In particular, for species strongly regulated by gibberellins (GA) and abscisic acid (ABA), the light-induced germination mechanisms involve phytochrome-mediated signaling pathways and complex hormone interactions10,11. Nevertheless, current research on the light responsiveness of lily seeds remains limited, and there is a lack of systematic analysis regarding the synergistic effects of light and priming treatments.
Based on this, this study selects six representative lily seed types and conducts a three-factor factorial experiment to systematically investigate the effects of different chemical priming treatments (KNO3, K3PO4, GA3) and light conditions (0 h/d, 12 h/d, 24 h/d) on their germination process. The goal is to elucidate the regulatory mechanisms of various treatment combinations on lily seed germination behavior and to provide theoretical support and technical references for the future propagation of lily seeds.
Results and analysis
Effect of distilled water treatment on the germination time and rate of Lily seeds
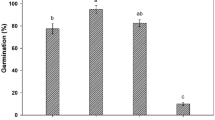
Six types of lily seeds were each treated with distilled water for 1 h as the control check group (CK), and the results are shown in Figs. 1 and 2. Under 0 h/d light conditions, the time required for complete germination of most lily species was shorter than under 12 h/d and 24 h/d light conditions. However, L. pensylvanicum took longer to germinate under 0 h/d light than under 12 h/d and 24 h/d light, which may be due to its stronger light requirement during the seed germination phase. Additionally, most species exhibited the highest germination rates under 24 h/d light, while L. davidii var. unicolor and L. distichum had significantly higher germination rates under 12 h/d light compared to the other two treatments. The germination rate of L. pensylvanicum did not show significant differences between 12 h/d and 24 h/d light conditions. Therefore, considering both the time for complete germination and the germination rate, the optimal treatment combinations are as follows: L. pumilum, L. auratum var. platyphyllum, and L. lancifolium performed best under 24 h/d light conditions, while L. davidii var. unicolor, L. distichum, and L. pensylvanicum showed better performance under 12 h/d light conditions.
Effect of control treatment on germination rate of lily seeds.
Chemical treatment
The effects of different chemical treatments and light conditions on the complete germination time of six lily seed species are shown in Figs. 3 and 4. Under 12 h/d light conditions, the 0.1% KNO3 treatment significantly shortened the germination time for most lily species and improved germination rates, with some species achieving germination rates close to 100%, significantly outperforming the CK and 0.1% tripotassium phosphate (K3PO4) treatments. Similarly, the 0.1% K3PO4 treatment also showed a noticeable promoting effect on germination rates for several species, which was higher than that of the CK in most cases, except for L. distichum. Under 24 h/d light conditions, the 0.1% KNO3 treatment continued to exhibit good germination promoting effects in most species, characterized by significantly shorter germination times and substantially improved germination rates, particularly in light-responsive species such as L. pumilum, L. auratum var. platyphyllum and L. distichum. In addition, the 0.1% K3PO4 treatment also exhibited a clear promoting effect under 24 h/d light conditions, with higher germination rates compared to the CK in several species. Under 0 h/d light conditions, the 0.1% KNO3 treatment resulted in germination rates of nearly 100% for all species, indicating a strong promoting effect, although it also prolonged the time required for complete germination. Considering both germination rate and the time required for complete germination, the optimal treatment conditions for each lily seed under chemical treatment are as follows: L. pumilum, L. auratum var. platyphyllum and L. distichum should be treated with 0.1% KNO3 for 1 h under 24 h/d light conditions; L. davidii var. unicolor and L. lancifolium performed best under 24 h/d light conditions with either 0.1% KNO3 or 0.1% K3PO4. L. pensylvanicum should be treated with 0.1% K3PO4 under 24 h/d light conditions.
Effect of different chemical treatments on the complete germination time of lily seeds.
Effect of different chemical treatments on the germination rate of lily seeds.
Gibberellin treatment
The complete germination time and germination rate of six lily seeds treated with different concentrations of gibberellin solution are shown in Figs. 5 and 6. The optimal treatment method: L. pumilum and L. distichum are suitable for treatment with 200 mg/L gibberellin (GA3) for 1 h under 12 h/d light conditions. L. davidii var. unicolor is suitable for treatment with 150 mg/L GA3 for 1 h under 0 h/d light conditions. L. auratum var. platyphyllum is suitable for treatment with 50 mg/L GA3 for 1 h under 24 h/d light conditions. L. lancifolium is suitable for treatment with 400 mg/L GA3 for 1 h under 24 h/d light conditions. L. pensylvanicum is suitable for treatment with 400 mg/L GA3 for 1 h under 0 h/d light conditions.
Effect of GA3 treatment on the complete germination time of lily seeds.
Effect of GA3 treatment on the germination rate of lily seeds.
Three-way factorial experiment
The study used a three-way factorial experimental design to investigate the effects of lily seed species, chemical treatments, and light conditions on the germination of six lily seeds. The details are shown in Table 1. Different lily species (A), chemical treatment methods (B), and light conditions (C) all significantly affect seed germination rates. Among these, the main effect of lily species was the most significant (p < 0.001), with an F-value of 16.99, indicating significant differences in germination rates among different species; Chemical treatment methods (p = 0.003, F = 14.24) and light conditions (p = 0.015, F = 10.70) also showed significant effects, indicating that pre-treatment agents and light exposure time are important factors influencing seed germination rates. In addition, a significant interaction was detected between species and treatment method (p = 0.042), demonstrating that different lily species respond differently to specific chemical treatments. In contrast, the interactions between species and light exposure (p = 0.082), treatment and light exposure (p = 0.107), and the three-way interaction (p = 0.216) were not significant. Therefore, only the species–treatment interaction had a notable influence, while the overall effects of other interactions were relatively minor. In summary, germination rate is primarily influenced by single factors, and when optimizing treatment strategies, the focus should be on matching different species with appropriate agents.
Summary and discussion
Summary
The results showed that GA3 treatment significantly improved the germination rate and shortened the germination time of most lily species, with the most pronounced effects observed in L. lancifolium and L. distichum. KNO3 also promoted germination in several species, such as L. auratum var. platyphyllum and L. pumilum, while K3PO4 showed only limited improvement. Regarding light response, L. davidii var. unicolor and L. pensylvanicum germinated best in darkness, indicating light inhibition. L. auratum var. platyphyllum and L. lancifolium showed higher germination capacity under continuous light, suggesting light promotion. Sensitivity to both chemical treatments and light conditions varied significantly among species. Overall, GA3 was the most effective treatment, with L. lancifolium showing the strongest response.
Discussion
The results showed that chemical treatments and light conditions significantly affected lily seed germination, with clear differences between varieties. First, GA3 treatment significantly increased germination rates and shortened germination time in most species, with L. lancifolium and L. distichum showing the most pronounced responses. This suggests that these two seed types are highly dependent on exogenous GA3, consistent with the physiological mechanism by which GA3 promotes cell elongation and activates amylase synthesis to break dormancy. Related studies have pointed out that GA3 can break dormancy and promote germination by promoting seedling cell elongation and the synthesis of hydrolytic enzymes such as α-amylase12.
KNO3 showed clear promoting effects in multiple species, whereas K3PO4 exerted limited improvement, mainly in L. distichum and L. lancifolium. KNO3, as a typical nitrate nitrogen source, acts as a signal molecule in plants to regulate endogenous hormone balance, thereby promoting seed coat softening and water absorption13. K3PO4, as an efficient phosphorus source, enhances early seed energy metabolism, promotes radicle emergence, and improves germination uniformity14. Research results indicate that in L. distichum and L. lancifolium, germination rate and germination time under K3PO4 treatment were slightly superior to the control group, demonstrating a limited but species-specific adaptability.
Regarding light conditions, studies have also validated their important regulatory role in the germination process of lily seeds. Overall, L. davidii var. unicolor and L. pensylvanicum exhibited optimal germination under 0 h/d light conditions, suggesting that these species may belong to the negative phototropic seed type. In contrast, L. auratum var. platyphyllum and L. lancifolium demonstrated higher germination potential under 24 h/d light conditions, exhibiting typical positive phototropic responses. Previous studies have shown that light can regulate the relative content of GA and ABA by activating light-sensitive pigment-mediated signalling pathways, thereby influencing seed germination behaviour15]– [16. The results indicate that different lily seed species exhibit significant differences in their responses to light induction, and the effects of light conditions on seed germination are species-specific.
Additionally, in the interaction between GA3 and light, synergistic effects were observed in some treatments. For example, L. distichum treated with 200 mg/L GA3 under 12 h/d light conditions exhibited a significant advantage, indicating that moderate light can enhance the stimulating effect of GA3. This synergistic mechanism may be related to the cross-regulation between photopigments and the gibberellin signal pathway17.
In summary, lily seed responses to stimulants and light are strongly species-dependent, suggesting that targeted pre-sowing strategies should be developed to enhance germination efficiency and seedling quality.
Materials and methods
Test materials
Among the six lily seed types listed in Table 2, the species are as follows: (1) L. pumilum, (2) L. davidii var. unicolor, (3) L. auratum var. platyphyllum, (4) L. lancifolium, (5) L. distichum, and (6) L. pensylvanicum. All seeds were collected from the same region in Tieling City, Liaoning Province, China, with the exception of L. pensylvanicum, which was cultivated in the medicinal plant garden of Jilin Agricultural University. Images of these seeds are presented in Fig. 7, and their physical characteristics are detailed in Table 2. All seeds used in this study were harvested in 2022.
Picture of lily seed (a: L. pumilum; b: L. davidii var. unicolor; c: L. auratum var. platyphyllum; d: L. lancifolium; e: L. distichum; f: L. pensylvanicum).
Chemicals and nutrients
KNO3, K3PO4, GA3, and sodium hypochlorite solution (NaClO), all of analytical grade, were purchased from domestic suppliers in China and used in this study.
Main testing instruments and equipment
Shanghai Sanfa Scientific Instrument Co., Ltd., artificial climate incubator, LHP.250 type; Yadu ultrasonic humidifier, YC-D205S type; HH-6 digital display constant temperature water bath pot; Ohus Instruments (Shanghai) Co., Ltd., electronic balance, AR323CN model.
Experimental design
The experiment was conducted in November 2023 using a randomized complete block design with three main factors: lily species, chemical treatments, and light conditions. A three-factor factorial experiment was implemented. Each treatment had three replicates, and five lily seeds were placed in each petri dish. Before sowing, all seeds were disinfected with 2% NaClO solution for 15 min and then rinsed three times with distilled water18.
A total of 17 pre-treatment combinations were set up, as detailed in Table 3. Distilled water treatment for 1 h served as the control group (CK). Chemical pre-treatments included 0.1% KNO3 and 0.1% K3PO4, while hormone pre-treatments used GA3 at different concentrations ranging from 50 to 400 mg/L. All treatments were applied by soaking the seeds for 1 h.
After pre-treatment, seeds were placed in 9 cm petri dishes lined with two layers of moist filter paper and incubated in an artificial climate chamber. Different light conditions were set according to Table 4 (0 h/d, 12 h/d, 24 h/d), with the temperature maintained at a constant 25 °C. During the 40 d incubation period, the moisture of the filter paper was checked daily and supplemented with distilled water as needed to maintain a stable environment. Moldy seeds were promptly removed.
A seed was considered germinated when the radicle emerged through the seed coat and extended 1 to 2 mm. Germination was recorded daily. The observation period ended when all seeds had either germinated or decayed.
Measured traits
The start time of germination is recorded, including the completion time of germination for all seeds and the daily germination amount under different treatments, and then the germination rate is calculated. The germination start time refers to the time when the first lily seed germinates in each treatment. The completion time of all germination refers to the time when the last lily seed germinates in the petri dish. Germination rate = (Number of germinated seeds / Total number of seeds) × 100%.
Statistical analysis method
Data were analyzed using SPSS 22.0 software. A three-way factorial analysis of variance (ANOVA) was conducted to assess the effects of lily species, chemical treatment, and light condition on germination rate and germination time. The significance of main effects and interactions was evaluated at a significance level of p < 0.05. Duncan’s multiple range test was used for post-hoc comparisons among means. Significance tests were conducted within each species to compare different treatments under the same light conditions. Letters were used in figures and tables to indicate significant differences, where the means sharing the same letter were not significantly different at the 0.05 level.
Data availability
The datasets used and/or analysed during the current study available from the corresponding author on reasonable request.
References
Shen, L. et al. Study on the off-site reproduction and conservation of wild fagopyrum cymosum germplasm resources. J. Plant. Genetic Resour. 24, 889–895. https://doi.org/10.13430/j.cnki.jpgr.20221012001 (2023).
Tian, W. et al. Effect of simulated digestion in vitro on the rheology and biological activity of lilium Davidii var.unicolor Salisb polysaccharide. J. Plant Biochem. Biotechnol. 32, 587–596. https://doi.org/10.1007/s13562-023-00843-3 (2023).
Guan, H., Chen, S., Chenglin, E., Wang, X. & Xiao, J. Research progress on genetic diversity of snap bean (phaseolus vulgaris) germplasm resources in China. Agric. Biotechnol. 11, 19–21 (2022).
Guo, R. et al. Investigation and taxonomy of wild Fragaria resources in the South of Qinling mountain in Hanzhong city, Shaanxi Province of China. J. Plant. Genetic Resour. 23, 1393–1399. https://doi.org/10.13430/j.cnki.jpgr.20220225002 (2022).
Li, S., Gao, W. & Cui, Z. Renal protective effects of Lanzhou Lily (lilium Davidii var. unicolor) polysaccharide and aerobic exercise in streptozotocin-induced diabetic rats. Food Sci. 44, 165–180. https://doi.org/10.7506/spkx1002-6630-20220630-346 (2023).
Blossei, J., Gbelein, R., Hammann, T. & Uptmoor, R. Late blight resistance in wild potato species-resources for future potato (solanum tuberosum) breeding. Plant. Breed. 141 https://doi.org/10.1111/pbr.13023 (2022).
Osawa, S. Habitat characteristics of shortia Uniflora as a local resource plant with the flower landscape in tomiyama-funato district, Nakagawa town, Tochigi Prefecture. J. Jpn. Inst. Landsc. Archit. 85, 645–650. https://doi.org/10.5632/jila.85.645 (2022).
Dong, S., Li, L., Chen, W., Wang, Y. & Wang, S. Evaluation of heavy metal speciation distribution in soil and the accumulation characteristics in wild plants: a study on naturally aged abandoned farmland adjacent to tailings. Sci. Total Environ. 917, 170594. https://doi.org/10.1016/j.scitotenv.2024.170594 (2024).
Khatri, K., Negi, B., Bargali, K. & Bargali, S. S. Effects of different concentrations of leaf residues of ageratina adenophora on seed germination and growth behavior of two native tree species of Kumaun himalaya, India. Waste Biomass Valoriz. 15, 923–943. https://doi.org/10.1007/s12649-023-02213-5 (2024).
Al-Turki, T. A., Davy, A. J., Al-Ammari, B. S. & Basahi, M. A. Seed germination characteristics of some medicinally important desert plants from the Arabian Peninsula. J. Arid Environ. 198, 104689. https://doi.org/10.1016/j.jaridenv.2021.104689 (2022).
Meghana, A., Kalleshwaraswamy, C. M., Deshmukh, S. S. & Mallikarjuna, H. B. Effect of whorl application of sand mixed Chlorantraniliprole 18.5 SC on the reproductive biology of invasive fall armyworm, spodoptera Frugiperda (J. E. Smith) (lepidoptera: noctuidae) in maize. Cereal Res. Commun. 52, 683–690. https://doi.org/10.1007/s42976-023-00402-5 (2024).
Taratima, W., Rohmah, K. N., Plaikhuntod, K., Maneerattanarungroj, P. & Trunjaruen, A. Optimal protocol for in vitro polyploid induction of cymbidium aloifolium (L.) Sw. BMC Plant Biol. 23, 295. https://doi.org/10.1186/s12870-023-04314-8 (2023).
Ivanchenko, T. & Belikina, A. Research results on the use of plant growth regulator triacontanol in winter wheat crops in the conditions of the lower volga region, in IOP Conference Series: Earth and Environmental Science 979. https://doi.org/10.1088/1755-1315/979/1/012004 (2022).
Jacob, S. S. et al. Bringing back a forgotten legume-sensory profiles of australian native wattleseeds reveal potential for novel food applications. J. Food Sci. 89 (2024). https://doi.org/10.1111/1750-3841.16860 (2024).
Gupta, A. et al. SlDREB3, a negative regulator of ABA responses, controls seed germination, fruit size and the onset of ripening in tomato. Plant Sci. Int. J. Exp. Plant Biol. 319. https://doi.org/10.1016/j.plantsci.2022.111249 (2023).
Wang, Y. et al. Identification of a novel transcription factor under long-term drought resistance in Highland barley: A DNA affinity purification sequencing-based transcriptomic analysis. Chem. Biol. Technol. Agric. 10 https://doi.org/10.1186/s40538-022-00376-2 (2023).
Fu, J. et al. Effect of gibberellic acid on photosynthesis and oxidative stress response in maize under weak light conditions. Front. Plant Sci. 14, 1128780. https://doi.org/10.1186/s40529-021-00329-2 (2023).
Yiamthaisong, N., Jampeetong, A. & Tiansawat, P. Surface sterilization and moist storage conditions for recalcitrant seeds of three Asian tree species. For. Sci. Technol. 20 (4), 410–417. https://doi.org/10.1080/21580103.2024.2409916 (2024).
Funding
The research is supported by: Jilin Provincial Department of Science and Technology key research and development plan project (medical and health field), Breeding and construction of provenance base of new lily variety (No. 20210204169YY).
Author information
Authors and Affiliations
Contributions
H.M.L. and Y.Z.H. processed the numerical attribute linear programming of communication big data, and the mutual information feature quantity of communication big data numerical attribute was extracted by the cloud extended distributed feature fitting method. Y.B. and H.Z. combined with fuzzy C-means clustering and linear regression analysis, the statistical analysis of big data numerical attribute feature information was carried out, and the associated attribute sample set of communication big data numerical attribute cloud grid distribution was constructed. Y.H.X. and J.S.B. did the experiments, recorded data, and created manuscripts. All authors read and approved the final manuscript.
Permissions
The permission to collect lily seed was obtained from Jilin Agricultural University.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Liu, H., Han, Y., Bao, Y. et al. Insight of the seed germination of Lily species under different chemical treatments and light conditions. Sci Rep 16, 110 (2026). https://doi.org/10.1038/s41598-025-29364-w
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-29364-w