Abstract
The relationship between the systemic immune-inflammation index (SII) and disease activity in rheumatoid arthritis (RA) remains unclear. This study aimed to clarify this association in a cohort of hospitalized RA patients overwhelmingly with moderate-to-high disease activity. We consecutively included 1,191 RA patients hospitalized at Xingtai People’s Hospital from March 2022 to December 2024. SII was calculated as (neutrophils × platelets)/lymphocytes. Disease activity was assessed using the Disease Activity Score 28-erythrocyte sedimentation rate (DAS28ESR) and C-reactive protein (DAS28-CRP). Multiple linear regression and generalized additive models were employed to explore the association between SII and disease activity. For regression analysis, SII underwent natural logarithm transformation to improve normality. After strict eligibility criteria, 997 individuals (mean age 57.66 years; 77.93% female, 95.6% moderate-to-high disease activity) were included for the final analysis. A nonlinear relationship was identified with a threshold at LnSII = 5.56 (SII = 260.36). Above this cutoff, LnSII showed positive correlations with both DAS28-ESR (β = 0.53, 95% CI: 0.44–0.63) and DAS28-CRP (β = 0.63, 95% CI: 0.53–0.72). No significant association was found below this threshold (DAS28-ESR: β = -0.56, 95% CI: -1.13– 0.02; DAS28-CRP: β = -0.35, 95% CI: -0.91–0.20). These findings suggest SII levels were independently positive associated with DAS28-ESR and DAS28-CRP among RA patients with SII > 260.36, supporting its potential as a useful biomarker for evaluating disease activity in RA patients with predominantly moderate-to-high disease activity.
Similar content being viewed by others
Introduction
Rheumatoid arthritis (RA) is a common chronic systemic autoimmune disease, primarily marked by persistent inflammation in multiple joints, progressive joint deformity, and gradual loss of function1,2. Beyond joint involvement, RA is frequently associated with systemic complications, including cardiovascular disease and interstitial lung disease, which contribute significantly to impaired quality of life and reduced life expectancy3. Epidemiological evidence indicates a global prevalence of RA ranging from 0.5% to 1%, with estimates projecting an increase to 31.7 million cases by 2050, emphasizing its role as a critical public health issue4. Thus, precise evaluation of disease activity and inflammatory burden is essential for optimizing treatment strategies and improving patient outcomes.
In clinical practice, traditional inflammatory markers such as erythrocyte sedimentation rate (ESR) and C-reactive protein (CRP) remain widely used to evaluate RA disease activity5. However, their specificity is limited and they are susceptible to confounding factors (such as infection and anemia)5, which restrict their ability to comprehensively reflect the complex inflammatory environment of RA. Although cytokines like tumor necrosis factor-alpha (TNF-α) and interleukin-6 (IL-6) are more closely correlated with disease activity2, their high cost and technical demands restrict their routine clinical application. Therefore, there is an urgent need to identify cost-effective, accessible, and reliable biomarkers that better reflect inflammatory status and aid prognostic evaluation in RA.
The systemic immune-inflammation index (SII), derived from neutrophil, lymphocyte, and platelet counts, has recently emerged as a promising biomarker of systemic immune and inflammatory status6. This index is simple, non-invasive, easily accessible, and cost-effective for routine clinical use. Increasing evidence supports the utility of SII across a range of conditions, including malignancies6,7, cardiovascular diseases8,9 and autoimmune disorders10,11,12,13,14,15. Indeed, during the active phase of RA, elevated neutrophil and platelet counts, accompanied by a reduced lymphocyte count, are commonly observed16,17. By integrating these parameters, SII may offer a more comprehensive indicator of the inflammatory status in RA patients. Besides, this index is simple, non-invasive, easily accessible, and cost-effective for routine clinical use. Several studies have demonstrated that SII has been linked to inflammatory markers such as ESR, CRP, and Disease Activity Score in 28 joints (DAS28)18,19,20. However, these findings are limited by small sample sizes and reliance on unadjusted correlation analyses, leaving the relationship between SII and RA disease activity insufficiently defined.
To address these gaps, the present study expanded the sample size and adjusted for multiple confounders to more accurately evaluate the association between SII and disease activity. Furthermore, we further explored potential nonlinear and threshold effects, thereby providing deeper insight into the clinical value of SII in assessing RA disease activity.
Materials and methods
Study design
This was a cross-sectional study using data collected from March 2022 to December 2024 at Xingtai People’s Hospital. The study was designed to investigate the association between SII and disease activity in patients with RA. The study protocol and reporting adhered to the STROBE (Strengthening the Reporting of Observational Studies in Epidemiology) statement guidelines. The study was conducted in accordance with the Declaration of Helsinki and approved by the Ethics Committee of Xingtai People’s Hospital (approval number: 2025[031]). Given the retrospective design of the study and the anonymization of patient data, the requirement for informed consent was waived by the Ethics Committee of Xingtai People’s Hospital, consistent with previous reports21,22.
Study population
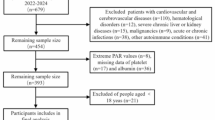
We consecutively enrolled 1,191 patients with RA who were hospitalized at Xingtai People’s Hospital between March 2022 and December 2024. Patients were primarily hospitalized due to active disease or suboptimal control, requiring inpatient assessment and therapeutic adjustment. All participants met the 2010 ACR/EULAR classification criteria for RA23. Patients were excluded if they were younger than 18 years, had active or chronic infections, other autoimmune diseases, severe hepatic or renal impairment, malignancies, or incomplete DAS28 data. Ultimately, 997 patients with RA were included in the study. A flowchart detailing the patient screening and enrollment process is shown in Fig. 1. All data were retrospectively extracted from the hospital’s electronic medical records.
A flowchart of the screening process for RA patients.
Clinical data collection
Demographic and clinical data were collected including age, sex, body mass index (BMI), smoking status, alcohol consumption history, past medical history (Hypertension, diabetes mellitus [DM], coronary heart disease [CHD]), disease duration of RA, and recent medication use (including nonsteroidal anti-inflammatory drugs [NSAIDs], glucocorticoids, conventional synthetic disease-modifying antirheumatic drugs [csDMARDs], biological DMARDs [bDMARDs], and targeted synthetic DMARDs [tsDMARDs]). In addition, swollen joint count in 28 joints (SJC28), tender joint count in 28 joints (TJC28), and patient global health assessment (GH) and evaluator global assessment (EGA) measured by a 100-mm visual analogue scale (VAS) were recorded. All information was extracted from the hospital’s electronic medical records and was originally collected through face-to-face interviews and physical examinations conducted by rheumatologists on the day of admission.
Laboratory data were obtained from the electronic medical record system, including complete blood count parameters (including white blood cell [WBC] count, neutrophil count, lymphocyte count, red blood cell [RBC] count, hemoglobin, and platelet count), ESR, CRP, rheumatoid factor (RF), and autoantibodies such as anti-cyclic citrullinated peptide antibody (ACPA), anti-perinuclear factor (APF), and anti-keratin antibody (AKA). All laboratory measurements were performed according to standardized protocols in the hospital laboratory using fasting peripheral blood samples collected on the day of admission or the following day.
The SII was calculated using absolute counts of neutrophils, platelets, and lymphocytes, all expressed as ×10⁹/L, with the following formula: SII = (neutrophil count × platelet count) ÷ lymphocyte count. Disease activity was assessed using DAS28, calculated by two formulas: DAS28-ESR = 0.56 × √(TJC28) + 0.28 × √(SJC28) + 0.70 × ln(ESR) + 0.014 × GH; DAS28-CRP = 0.56×√(TJC28) + 0.28 × √(SJC28) + 0.36×ln(CRP + 1) + 0.014×GH + 0.9620; Clinical Disease Activity Index (CDAI) = TJC28 + SJC28 + GH + EGA; Simplified Disease Activity Index (SDAI) = TJC28 + SJC28 + GH + EGA + CRP (mg/dL)19. Disease activity was classified based on DAS28 as follows: high activity (≥ 5.1), moderate activity (≥ 3.2 to < 5.1), low activity (≥ 2.6 to < 3.2), and remission (< 2.6).
Statistical analysis
Continuous variables were expressed as mean ± standard deviation (SD) or median (interquartile range, IQR) and analyzed using one-way ANOVA or the Kruskal-Wallis test, respectively. Categorical variables were presented as counts (percentages) and compared using the chi-square (χ²) test. Since the missing data were small (< 1%), such as BMI (n = 5) and RF (n = 9), the corresponding means or medians were used to supplement them, respectively. Details of missing covariates were provided in Supplementary Table S1.
Correlations between variables were determined using the Spearman test (ρ). The relationship between SII and DAS28 was assessed using multivariable linear regression models constructed in three distinct models: Model 1 was unadjusted; Model 2 was adjusted for sex, age, hemoglobin, monocyte count, RF, and csDMARDs; Model 3 was further included smoking status, hypertension, glucocorticoid use, bDMARD, and tsDMARDs. These covariates were selected on a change in effect of more than 10% (model 2) or a regression coefficient p-value less than 0.1 (model 3). Additionally, SII, RF, and disease duration were natural logarithm transformed to regression analyses to improve normality. To assess the robustness of the results, we performed sensitivity analyses by categorizing LnSII into tertiles and quartiles. The cut-off points for tertiles were defined by the 33.3rd and 66.7th percentiles, and for quartiles by the 25th, 50th, and 75th percentiles of the LnSII distribution. Trends were evaluated by treating the median LnSII value of each tertile or quartile as a continuous variable. Generalized additive models were used to explore potential nonlinear associations between SII and DAS28. When nonlinear relationships were identified, two-piecewise linear regression models were further employed to analyze threshold effects, with inflection points determined by smoothing spline plots combined with a recursive algorithm to optimize model fit.
All statistical analyses were conducted using R (http://www.r-project.org, The R Foundation) and EmpowerStats (http://www.empowerstats.com, X&Y Solutions, Inc., Boston, MA). Statistical significance was defined as a two-sided P value < 0.05.
Results
Study population characteristics
This study included 997 patients with RA (77.93% female), with a mean age of 57.66 ± 12.82 years, and a median disease duration of 48.00 months (IQR: 12.00–132.00). The cohort was defined by its inpatient nature, with an overwhelming majority (95.6%, n = 953) of patients presenting with moderate-to-high disease activity (DAS28-ESR ≥ 3.2). As presented in Table 1, when stratified by DAS28-ESR, patients with high disease activity (≥ 5.1, n = 540) were more likely to be male and older compared to those with moderate disease activity (≥ 3.2 to < 5.1, n = 413) or low disease activity/remission(< 3.2, n = 44). The high activity group also exhibited significantly elevated inflammatory markers, including ESR, CRP, RF, WBC, neutrophils, and platelets, alongside reduced RBC counts and hemoglobin levels (all P < 0.05). Notably, SII values were markedly higher in this group. In terms of treatment, patients with high disease activity were less frequently treated with methotrexate, leflunomide, types of csDMARDs, and tsDMARDs, while bDMARD usage was lower in both the low and high activity groups. No significant differences were observed regarding BMI, smoking status, alcohol consumption, disease duration, NSAIDs or glucocorticoid use, lymphocyte counts, or autoantibody status.
As shown in Table 2, patients in the highest SII tertile (T3) exhibited characteristics similar to those with high disease activity. This included a higher proportion of males and hypertension, alongside decreased lymphocyte counts and hemoglobin levels. Furthermore, these patients had elevated inflammatory markers and higher disease activity scores, as measured by DAS28-ESR, DAS28-CRP, CDAI, and SDAI. This group was also less likely to receive leflunomide and types of csDMARDs. No significant differences were found in terms of BMI, smoking, drinking, disease duration, autoantibody status, NSAIDs, glucocorticoids, tsDMARDs, and bDMARDs.
Univariate analysis
As shown in Supplementary Table S2, univariate analysis revealed that male, age, smoking, hypertension, and DM were significantly associated with higher RA disease activity. Elevated disease activity was also correlated with increased inflammatory markers, including WBC count, neutrophil count, platelet count, RF, and SII. In contrast, higher RBC count and hemoglobin levels were associated with lower disease activity. Use of methotrexate, leflunomide, types of csDMARDs, as well as tsDMARDs, showed a negative association with disease activity. No significant relationships were observed for BMI, alcohol consumption, CHD, lymphocyte count, or the use of NSAIDs, glucocorticoids, or bDMARDs.
Correlation between SII and disease activity parameters
As shown in Supplementary Table S3, Spearman correlation analysis revealed that SII was moderately and positively correlated with C-reactive protein (ρ = 0.53) and SDAI (ρ = 0.48). Significant correlations were also found with other composite scores, including DAS28-CRP (ρ = 0.44), DAS28-ESR (ρ = 0.37), and CDAI (ρ = 0.36) (all P < 0.001).
Association of SII with disease activity in rheumatoid arthritis
As presented in Table 3, multivariable linear regression analysis revealed a consistent positive association between SII and disease activity across all models. In the fully adjusted model, each unit increase in LnSII was associated with a 0.46-point rise in DAS28-ESR (95% CI: 0.38–0.55) and a 0.55-point increase in DAS28-CRP (95% CI: 0.47–0.63). This association was further confirmed by sensitivity analyses, which showed a significant dose-response relationship when LnSII was analyzed as tertiles (p for trend < 0.0001) and a consistent trend across quartiles (Supplementary Table S4). To further validate these findings, we performed additional analyses using other composite indices. As shown in Supplementary Table S5, LnSII remained independently and positively associated with CDAI (β = 2.62, 95% CI: 2.10–3.15) and SDAI scores (β = 5.11, 95% CI: 4.35–5.87), with significant dose-response trends (p for trend < 0.0001 for both). These results collectively confirm the robust association between SII and RA disease activity, independent of the specific composite index used.
Model 1 was adjusted for none; Model 2 was adjusted for sex, age, hemoglobin, monocyte count, rheumatoid factor, and conventional disease-modifying antirheumatic drugs; Model 3 was adjusted for sex, age, smoking, hypertension, hemoglobin, monocyte count, rheumatoid factor, glucocorticoids, conventional disease-modifying antirheumatic drugs, biological disease-modifying antirheumatic drugs, and targeted synthetic disease-modifying antirheumatic drugs.
Nonlinear relationship of SII with disease activity in RA
As shown in Fig. 2, a nonlinear association between SII and disease activity was found. To further explore the relationship, we employed a two-piecewise linear regression model. As shown in Table 4, a significant inflection point was identified at LnSII = 5.56 (corresponding to SII = 260.36). Above this threshold, LnSII is significantly positively associated with disease activity (DAS28-ESR: β = 0.53, 95% CI: 0.44–0.63; DAS28-CRP: β = 0.63, 95% CI: 0.53–0.72). In contrast, below this threshold, the association is not significant (DAS28-ESR: β = −0.56, 95% CI: −1.13–0.02; DAS28-CRP: β = −0.35, 95% CI: −0.91–0.20). These findings provide robust evidence for a nonlinear threshold effect of SII on disease activity in RA patients.
a. A nonlinear relationship of SII with DAS28-ESR. Note: The model was adjusted for sex, age, smoking, hypertension, hemoglobin, monocyte count, rheumatoid factor, glucocorticoids, conventional disease-modifying antirheumatic drugs, biological disease-modifying antirheumatic drugs, and targeted synthetic disease-modifying antirheumatic drugs. b. A nonlinear relationship of SII with DAS28-CRP. Note: The model was adjusted for sex, age, smoking, hypertension, hemoglobin, monocyte count, rheumatoid factor, glucocorticoids, conventional disease-modifying antirheumatic drugs, biological disease-modifying antirheumatic drugs, and targeted synthetic disease-modifying antirheumatic drugs.
Discussion
This cross-sectional study involving 997 RA patients revealed a nonlinear relationship between SII and disease activity, as assessed by both DAS28-ESR and DAS28-CRP, independent of potential confounders. A threshold effect was identified at a SII value of 260.36. Above this threshold, SII was positively associated with both DAS28-ESR and DAS28-CRP, whereas no significant association was observed below this threshold. These findings indicate that SII may serve as a potential biomarker for assessing disease activity in RA patients.
Emerging evidence highlights the potential of SII as a marker of inflammation and treatment response in RA19,20,24,25. A cross-sectional study of 58 patients found positive correlations between SII and conventional inflammatory markers such as CRP, ESR, and the neutrophil-to-lymphocyte ratio24. Another investigation involving 109 RA patients demonstrated that SII could distinguish active disease from remission and healthy controls, suggesting its usefulness for monitoring disease activity20. A larger study with 257 participants confirmed strong associations of SII with disease activity scores including DAS28, the simplified disease activity index (SDAI), and the clinical disease activity index (CDAI) (all p < 0.01), with an area under the curve (AUC) of 0.703 for SII—outperforming traditional hematological markers19. However, these previous studies have been limited by small sample sizes and inadequate adjustment for confounding factors. In contrast, our study leverages a substantially larger sample, applies more rigorous inclusion and exclusion criteria, and comprehensively adjusts for multiple confounders, thereby providing stronger and more reliable evidence. Additionally, a retrospective analysis of 154 RA patients indicated that SII was superior to other inflammatory indices in predicting response to TNF-α inhibitors, underscoring its potential for guiding therapy26. Furthermore, a large population-based analyses utilizing National Health and Nutrition Examination Survey (NHANES) data demonstrated a positive, nonlinear association between elevated SII and RA risk in U.S. adults (OR = 1.167, 95% CI: 1.025–1.328), with a threshold effect at SII > 578.2527. Another NHANES study involving 2,070 RA patients found that a SII ≥ 919.75 was independently associated with increased all-cause and cardiovascular mortality, highlighting its prognostic value28.
An increasing number of studies are exploring the potential of SII as a marker in various autoimmune diseases. For instance, a retrospective study of 136 ankylosing spondylitis (AS) patients and 63 controls demonstrated that SII was significantly elevated in active AS patients and showed strong positive correlations with Bath Ankylosing Spondylitis Disease Activity Index (rs = 0.667, p < 0.001), and further ROC analysis identified an optimal cutoff of 513.2, with 83.3% sensitivity and 86.8% specificity for distinguishing active disease15. Similarly, research on psoriatic arthritis revealed that elevated SII was closely linked to disease activity scores and could effectively differentiate moderate to severe cases from milder forms13,14. Beyond joint diseases, SII has also shown promise in predicting adverse pregnancy outcomes in women with Systemic Lupus Erythematosus (SLE), as well as in forecasting SLE onset and detecting nephritis10,11. Additionally, a study of 166 Behçet’s disease patients demonstrated that SII was significantly higher during active disease phases, with an optimal cutoff of 552 providing 81% sensitivity and 82% specificity for assessing disease activity12.
The pathogenesis of RA involves complex interactions between innate and adaptive immunity. SII integrates neutrophil, platelet, and lymphocyte counts, offering a a comprehensive measure of systemic inflammation and immune status. Neutrophils play a pivotal role in initiating and driving joint inflammation, migrating early into the joint cavity in response to cytokines such as interleukin (IL)−8, IL-17, and Tumor Necrosis Factor-alpha (TNF-α), which activate them to release pro-inflammatory mediators29,30,31,32,33. They also contribute to autoimmunity through forming neutrophil extracellular traps (NETs), which expose citrullinated proteins and promote the production of autoantibodies such as ACPA34,35,36,37. The inflammatory microenvironment, rich in granulocyte colony-stimulating factor (G-CSF), granulocyte-macrophage colony-stimulating factor (GM-CSF), IL-6, and IL-1β, prolongs neutrophil survival by delaying apoptosis, thereby exacerbating inflammation38,39,40,41. NETs serve as autoantigens, further activating immune responses and creating a vicious cycle that accelerates joint tissue destruction29,30.
Similarly, platelets play a significant role in the pathogenesis of RA. In addition to their well-known function in blood clotting, activated platelets release cytokines such as IL-1α, Chemokine (C-X-C motif) ligand (CXCL) 4, and CXCL7, as well as microparticles that further amplify local and systemic inflammation42,43,44,45. They interact with a variety of immune cells (including neutrophils, monocytes, T, and B lymphocytes) enhancing immune activation and promoting the formation of immune complexes that contribute to joint tissue damage42,43. Platelet-derived microparticles also activate the complement system, creating a self-perpetuating inflammatory cycle44. Moreover, activated platelets facilitate thrombosis, establishing a direct link between systemic inflammation and increased cardiovascular risk in RA patients46. The decline lymphocytes in peripheral blood observed during active RA may result from multiple mechanisms. Chemokines like CXCL12 attract lymphocytes into inflamed joints, reducing their numbers in the bloodstream. Meanwhile, cytokines such as TNF-α and pathways involving Fas/FasL promote lymphocyte apoptosis. Elevated levels of IL-6 also impair lymphocyte production in the bone marrow2,47. Additionally, decreases in regulatory T cells (Tregs) and B-cell depletion further disrupt lymphocyte subset balance48. These immune alterations reflect an ongoing imbalance between systemic inflammation and local immune activity, closely associated with disease activity and progression in RA.
This study has several advantages. First, compared to prior research with smaller sample sizes, we included a larger patient cohort and collected more comprehensive clinical data. Second, as an observational study, controlling for confounding factors is crucial. Previous similar studies have generally only performed basic correlation analyses between SII and inflammation markers such as ESR, CRP, or DAS28, without adequately controlling for potential confounders. In contrast, we rigorously adjusted for a broad range of potential confounders, including demographics, comorbidities, laboratory parameters, and medication use. Third, beyond evaluating the association between SII and DAS28, we employed generalized additive models to explore their potential nonlinear relationship, providing a more precise understanding of their true relationship.
Nevertheless, this study has several limitations. First, the cross-sectional design only allows for the assessment of the association between SII and DAS28, but it cannot establish a causal relationship. Second, the lack of longitudinal follow-up data means we cannot evaluate how SII and DAS28 change over time after treatment. Third, a notable limitation stems from the exclusive inclusion of hospitalized individuals, which inherently limited the sample size of the low disease activity/remission subgroup. This restricts the direct applicability of our conclusions to patients with moderate-to-high disease activity. Therefore, additional validation in cohorts with a higher proportion of outpatients or those in sustained remission is required to fully elucidate the role of SII across all disease stages. Lastly, although we adjusted for known potential confounders, residual confounding or unmeasured factors may still influence the results. Future multi-center prospective studies are necessary to validate these findings.
In conclusion, a nonlinear relationship was observed between SII and DAS28, with a threshold at SII = 260.36. Among RA patients with SII levels above this threshold, SII was independently and positively associated with both DAS28-ESR and DAS28-CRP, supporting its potential as a promising biomarker for assessing disease activity in RA, particularly in patients with moderate-to-high disease activity.
Data availability
The original data supporting the findings of this study are not publicly available due to sensitivity reasons but can be obtained from the corresponding author upon reasonable request.
References
Di Matteo, A., Bathon, J. M. & Emery, P. Rheumatoid arthritis. Lancet 402 (10416), 2019–2033 (2023).
Weyand, C. M. & Goronzy, J. J. The immunology of rheumatoid arthritis. Nat. Immunol. 22 (1), 10–18 (2021).
Schattner, A. The cardiovascular burden of rheumatoid Arthritis - Implications for treatment. Am. J. Med. 136 (12), 1143–1146 (2023).
GBD 2021 Rheumatoid Arthritis Collaborators. Global, regional, and National burden of rheumatoid arthritis, 1990–2020, and projections to 2050: a systematic analysis of the global burden of disease study 2021. Lancet Rheumatol. 5 (10), e594–e610 (2023).
Orr, C. K. et al. The utility and limitations of CRP, ESR and DAS28-CRP in appraising disease activity in rheumatoid arthritis. Front. Med. 5, 185 (2018).
Hu, B. et al. Systemic immune-inflammation index predicts prognosis of patients after curative resection for hepatocellular carcinoma. Clin. Cancer Res. 20 (23), 6212–6222 (2014).
Tian, B. W. et al. Systemic immune-inflammation index predicts prognosis of cancer immunotherapy: systemic review and meta-analysis. Immunotherapy 14 (18), 1481–1496 (2022).
He, L., Xie, X., Xue, J., Xie, H. & Zhang, Y. Association of the systemic immune-infammation index with all-cause mortality in patients with arteriosclerotic cardiovascular disease. Front. Cardiovasc. Med. 9, 952953 (2022).
Wang, H., Nie, H., Bu, G., Tong, X. & Bai, X. Systemic immune-inflammation index (SII) and the risk of all-cause, cardiovascular, and cardio-cerebrovascular mortality in the general population. Eur. J. Med. Res. 28 (1), 575 (2023).
Sahin, R. et al. The role of first-trimester NLR (neutrophil to lymphocyte ratio), systemic immune-inflammation index (SII), and, systemic immune-response index (SIRI) in the prediction of composite adverse outcomes in pregnant women with systemic lupus erythematosus. J. Reprod. Immunol. 158, 103978 (2023).
Ozdemir, A., Baran, E., Kutu, M., Celik, S. & Yılmaz, M. Could systemic immune inflammation index be a new parameter for diagnosis and disease activity assessment in systemic lupus erythematosus? Int. Urol. Nephrol. 55 (1), 211–216 (2023).
Tanacan, E., Dincer, D., Erdogan, F. G. & Gurler, A. A cutoff value for the systemic Immune-Inflammation index in determining activity of Behçet disease. Clin. Exp. Dermatol. 46 (2), 286–291 (2021).
Kelesoglu Dincer, A. B. & Sezer, S. Systemic immune inflammation index as a reliable disease activity marker in psoriatic arthritis. J. Coll. Physicians Surg. Pak. 32 (6), 773–778 (2022).
Yorulmaz, A., Hayran, Y., Akpinar, U. & Yalcin, B. Systemic Immune-Inflammation index (SII) predicts increased severity in psoriasis and psoriatic arthritis. Curr. Health Sci. J. 46 (4), 352–357 (2020).
Wu, J., Yan, L., & Chai, K. Systemic immune-inflammation index is associated with disease activity in patients with ankylosing spondylitis. J. Clin.Lab. Anal. 35 (9), e23964 (2021).
Cecchi, I. et al. Neutrophils: novel key players in rheumatoid Arthritis. Current and future therapeutic targets. Autoimmun. rev. 17 (11), 1138–1149 (2018).
Targońska-Stępniak, B., Grzechnik, K. & Zwolak, R. The relationship between platelet indices and Ultrasound, Clinical, laboratory parameters of disease activity in patients with rheumatoid arthritis. J. Clin. Med. 10 (22), 5259 (2021).
hoe, J. Y. & Kim, S. K. Association between hematological indicesand disease activity in patients with rheumatoid arthritis treated with Janus kinase inhibitors for 24 weeks. Med. (Kaunas). 58 (3), 426 (2022).
Choe, J. Y., Lee, C. U. & Kim, S. K. Association between novel hematological indices and measures of disease activity in patients with rheumatoid arthritis. Med. (Kaunas). 59 (1), 117 (2023).
Satis, S. New inflammatory marker associated with disease activity in rheumatoid arthritis: the systemic Immune-Inflammation index. Curr. Health Sci. J. 47 (4), 553–557 (2021).
Aletaha, D. et al. 2010 rheumatoid arthritis classification criteria: an American college of Rheumatology/European league against rheumatism collaborative initiative. Arthritis Rheum. 62 (9), 2569–2581 (2010).
Filion, K. B. et al. A multicenter observational study of Incretin-based drugs and heart failure. N Engl. J. Med. 374 (12), 1145–1154 (2016).
Geleris, J. et al. Observational study of hydroxychloroquine in hospitalized patients with Covid-19. N Engl. J. Med. 382 (25), 2411–2418 (2020).
Dervisevic, A. et al. Systemic Immune-inflammation index in evaluation of inflammation in rheumatoid arthritis patients. Medeni Med. J. 39 (3), 183–191 (2024).
Başaran, P. Ö. & Dogan, M. The relationship between disease activity with pan-immune-inflammatory value and systemic immune-inflammation index in rheumatoid arthritis. Medicine 103 (9), e37230 (2024).
Bai, J., Tian, Y., Wang, F., Dang, J. & Wang, H. Role of systemic immune-inflammation index (SII) in assessing clinical efficacy of TNF-α inhibitors for rheumatoid arthritis. Am. J. Transl Res. 15 (11), 6524–6533 (2023).
Liu, B., Wang, J., Li, Y. Y., Li, K. P. & Zhang, Q. The association between systemic immune-inflammation index and rheumatoid arthritis: evidence from NHANES 1999–2018. Arthritis Res. Ther. 25 (1), 34 (2023).
Yin, X., Zhang, Y., Zou, J. & Yang, J. Association of the systemic immune-inflammation index with all-cause and cardiovascular mortality in individuals with rheumatoid arthritis. Sci. Rep. 14 (1), 15129 (2024).
Chen, W., Wang, Q., Ke, Y. & Lin, J. Neutrophil function in an inflammatory milieu of rheumatoid arthritis. J Immunol. Res 8549329 (2018).
Wright, H. L., Moots, R. J. & Edwards, S. W. The multifactorial role of neutrophils in rheumatoid arthritis. Nature reviews. Rheumatology 10 (10), 593–601 (2014).
Verri, W. A. et al. IL-33 induces neutrophil migration in rheumatoid arthritis and is a target of anti-TNF therapy. Ann. Rheum. Dis. 69 (9), 1697–1703 (2010).
den Broeder, A. A. et al. Neutrophil migration and production of reactive oxygen species during treatment with a fully human anti-tumor necrosis factor-alpha monoclonal antibody in patients with rheumatoid arthritis. J. Rheumatol. 30 (2), 232–237 (2003).
Lemos, H. P. et al. Prostaglandin mediates IL-23/IL-17-induced neutrophil migration in inflammation by inhibiting IL-12 and IFNgamma production. Proc. Natl. Acad. Sci. U S A. 106 (14), 5954–5959 (2009).
Apel, F., Zychlinsky, A. & Kenny, E. F. The role of neutrophil extracellular traps in rheumatic diseases. Nat. Rev. Rheumatol. 14 (8), 467–475 (2018).
Demoruelle, M. K. et al. Antibody responses to citrullinated and noncitrullinated antigens in the sputum of subjects with rheumatoid arthritis and subjects at risk for development of rheumatoid arthritis. Arthritis Rheumatol. 70 (4), 516–527 (2018).
Khandpur, R. et al. NETs are a source of citrullinated autoantigens and stimulate inflammatory responses in rheumatoid arthritis. Sci. Transl Med. 5 (178), 178ra40 (2013).
Gorlino, C. V. et al. Association between levels of synovial anti-citrullinated peptide antibodies and neutrophil response in patients with rheumatoid arthritis. Eur. J. Immunol. 48 (9), 1563–1572 (2018).
Mitchell, T. S., Moots, R. J. & Wright, H. L. Janus kinase inhibitors prevent migration of rheumatoid arthritis neutrophils towards interleukin-8, but do not inhibit priming of the respiratory burst or reactive oxygen species production. Clin. Exp. Immunol. 189 (2), 250–258 (2017).
Wright, H. L., Chikura, B., Bucknall, R. C., Moots, R. J. & Edwards, S. W. Changes in expression of membrane TNF, NF-{kappa}B activation and neutrophil apoptosis during active and resolved inflammation. Ann. Rheum. Dis. 70 (3), 537–543 (2011).
Ottonello, L. et al. Differential regulation of spontaneous and immune complex-induced neutrophil apoptosis by Proinflammatory cytokines. Role of oxidants, Bax and caspase-3. J. Leukoc. Biol. 72 (1), 125–132 (2002).
Chowdhury, K. et al. Synovial IL-9 facilitates neutrophil survival, function and differentiation of Th17 cells in rheumatoid arthritis. Arthritis Res. Ther. 20 (1), 18 (2018).
Andia, I. Rheumatoid arthritis: the Ins and outs of platelets in RA. Nat. Rev. Rheumatol. 13 (5), 262–264 (2017).
Knijff-Dutmer, E. A., Koerts, J., Nieuwland, R., Kalsbeek-Batenburg, E. M. & van de Laar M. A. Elevated levels of platelet microparticles are associated with disease activity in rheumatoid arthritis. Arthritis Rheum. 46 (6), 1498–1503 (2002).
Olumuyiwa-Akeredolu, O. O., Page, M. J., Soma, P. & Pretorius, E. Platelets: emerging facilitators of cellular crosstalk in rheumatoid arthritis. Nat. Rev. Rheumatol. 15 (4), 237–248 (2019).
Harifi, G. & Sibilia, J. Pathogenic role of platelets in rheumatoid arthritis and systemic autoimmune diseases. Perspectives and therapeutic aspects. Saudi Med. J. 37 (4), 354–360 (2016).
von Hundelshausen, P. & Weber, C. Platelets as immune cells: bridging inflammation and cardiovascular disease. Circ. Res. 100 (1), 27–40 (2007).
Wang, S., Yang, N. & Zhang, H. Metabolic dysregulation of lymphocytes in autoimmune diseases. Trends Endocrinol. Metab. 35 (7), 624–637 (2024).
Jiang, Q., Yang, G., Liu, Q., Wang, S. & Cui, D. Function and role of regulatory T cells in rheumatoid arthritis. Front. Immunol. 12, 626193 (2021).
Acknowledgements
The authors express their gratitude to all patients who participated in this study.
Funding
This study was supported by Key Research and Development Program of Xingtai City (No. 2025ZC074).
Author information
Authors and Affiliations
Contributions
1. Y.Q. and X.L.: conception and design, statistical analysis and result interpretation. X.L., L.L.,Y.H., J.Z, and J.S.: data collection and cleaning. Y.Q. and L.L.: validation and supervision. X.L. drafted the manuscript. Y.Q. revised the manuscript. All authors reviewed and approved the final manuscript submitted.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Li, X., Leng, L., Han, Y. et al. Association between systemic immune-inflammation index and disease activity score 28 in patients with rheumatoid arthritis: a cross-sectional study. Sci Rep 16, 557 (2026). https://doi.org/10.1038/s41598-025-30053-x
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-30053-x