Abstract
Gamma-band synchronization is a key mechanism for healthy cognitive function, yet it tends to decrease with age. EEG-based Neurofeedback (EEG-NF) is a promising tool enabling subjects to modulate their brain activity. However, its efficacy at the individual level remains unclear, which may partly explain the heterogeneity of neurofeedback outcomes. The primary objective of this study was to investigate individual neural dynamics of Gamma-band synchronization through EEG-NF training. We analyzed data from a double-blind, placebo-controlled trial using an EEG-based brain-computer interface, involving healthy older adults with subjective memory complaints, randomly assigned to a neurofeedback or a sham feedback group. Specifically, we employed a two-step unsupervised machine learning framework: first, epoch-based Agglomerative Hierarchical Clustering to identify individual-level response patterns, then Spectral Bi-Clustering to uncover higher-order structure at the population level. Results revealed a subgroup of individuals within the real neurofeedback condition who successfully enhanced their Gamma-band synchronization, with effects extending across the broader frequency spectrum. In contrast, the remaining participants in the neurofeedback group exhibited neural responses comparable to those observed in the sham group. This randomized controlled trial offers novel insights into the individual neural dynamics underlying successful Gamma EEG-NF training, highlighting its potential to promote healthy brain aging.
Similar content being viewed by others
Introduction
Brain oscillations refer to rhythmic fluctuations in neural activity, typically observed across a range of frequency bands. Functional connectivity is inferred when these fluctuations appear synchronized in two or more brain regions over time, suggesting dynamic interaction and information exchange between those regions. The human brain exhibits a high degree of synchronization, even at rest, as evidenced by the ever-changing landscape of the electroencephalogram (EEG)1. This “resting-state” synchrony is generally derived from the underlying structural connectivity2. When the brain is engaged in a particular cognitive function, the dynamics of the structures change and specific oscillation frequencies become dominant, reflecting a task-dependent modulation of neural networks3,4.
The current view is that brain oscillations in the Gamma-band frequency (range > 30 Hz) are the fundamental network mechanism for the flow of information and neural communication underpinning a wide range of cognitive processes1,5,647. For instance, accurate episodic memory has been linked to cross-regional Gamma-synchronization, which appears to be essential for the transfer of sensory information to the hippocampus during memory formation, as well as for the transfer of hippocampal representations to the prefrontal cortex during retrieval7. Episodic memory is typically disturbed from the early stages of Alzheimer’s disease (AD8), a neurodegenerative disorder characterized by the accumulation of amyloid-beta and hyperphosphorylated tau peptides in the human’s brain. These pathological changes affect excitatory and inhibitory synaptic transmission9, inducing abnormal synchronization and coupling mechanisms between cortical neurons. As a result, EEG rhythms at rest become altered1,10,11, a phenomenon consistently associated with both the progression and the severity of this neurodegenerative disorder12,13,14,15. Key findings from a mouse model of AD indicate that external stimulation of brain activity in the Gamma-band has the potential to recruit both neural and glial responses to attenuate AD-related pathology, and could therefore have a neuroprotective effect16,17.
But Gamma activity tends to decrease even in healthy aging18,19,20,21, which has been associated with age differences in cognitive performance over the lifespan22.
To counteract these aging effects, recent investigations have highlighted the potential for improving cognition through neuromodulation of Gamma oscillations, open-up new avenues for a healthy aging brain23.
Neurofeedback is one of the promising tools offering unique opportunities to modulate brain activity non-invasively24,25. In particular, EEG-Neurofeedback (EEG-NF) has attracted considerable attention due to its potential for training subjects to modulate their own brain activity, using specific real-time EEG markers as feedback, with lasting neural and cognitive/behavioral improvements as possible outcomes. Accordingly, its clinical interest has been hinted at for a wide range of neurological and psychiatric disorders25. Nevertheless, the results on neurofeedback interventions remain largely heterogeneous26. Moreover, the actual impact of neurofeedback training on neural dynamics at an individual level is far from being understood.
In our previous work27, we investigated whether cognitively healthy elderly individuals with subjective memory complaints could learn to modulate three EEG markers commonly associated with aging—namely, Peak Alpha Frequency (PAF), Theta/Beta ratio (TBR), and Gamma-band synchronization—using EEG-NF training compared to sham feedback. Specifically, we employed a supervised learning technique to assess the EEG marker that best discriminated participants receiving real EEG-NF from those in the sham condition. Gamma-band synchronization achieved the highest classification performance (71%), outperforming PAF and TBR (61.3%). Additionally, in the neurofeedback group, higher self-modulation was observed exclusively for Gamma-band synchronization, making it the only marker that participants were able to successfully modulate. However, such group-level comparisons offer limited granularity regarding the underlying neurophysiological mechanisms and dynamic patterns of modulation at the individual level. Notably, it is still unclear how these dynamics vary across subjects and may contribute to the heterogeneity of neurofeedback training outcomes.
The current study extended this initial work in order to investigate the individual neural dynamics underlying such a successful Gamma neuromodulation in cognitively healthy older adults. Specifically, our main objectives were two-fold: (1) to understand how each subject modulated their Gamma-band synchronization regardless of the group to which she/he belonged (real EEG-NF or sham feedback); and (2) to study potential broader effects of this self-neuromodulation across the entire frequency spectrum. To this end, we employed a two-step unsupervised machine learning approach that allowed clustering subjects based on their individualized responses to Gamma EEG-NF (or sham feedback) training, without relying on predefined grouping criteria. This data-driven analytical approach was designed to enable a more nuanced evaluation of neuromodulation effects by capturing individual neurophysiological modulation patterns, in order to offer deeper insights into subject-specific neural dynamics.
Methods
Study population
The study population consisted of 37 autonomous and cognitively healthy individuals over the age of 55 (mean age of 72), all reporting subjective memory complaints for at least 6 months. A power analysis, assuming α = 0.05 and 80% power, indicated that this sample size would be sufficient to detect statistically significant between-group effects. The inclusion criteria required a Mini-Mental State Examination (MMSE;28) score ≥ 27, and a Free and Cued Selective Reminding Test (FCSRT,29) total recall score ≥ 42, both within normal ranges, thereby confirming objective cognitive integrity. The exclusion criteria comprised the presence of neurological (e.g., neurodegenerative diseases, migraine, epilepsy, traumatic brain injury, stroke, tumor) or psychiatric conditions, major visual impairments, or use of medications that could affect cognitive performance (e.g., benzodiazepines, antidepressants, antipsychotics). Eligibility was confirmed by the medical investigator during the screening visit, which also served as the pre-training visit (V0), as detailed below.
The enrollment of subjects and data acquisition took place at the Institut de la Mémoire et de la Maladie d’Alzheimer (IM2A), in the Salpêtrière hospital (Paris, France), between 2018 and 2019. Written informed consent was provided by all participants prior to the experiments. The study has been conducted in accordance with the Helsinki Declaration of 1975 and was approved by the local Institutional Review Board of Ile-de-France XI (ID-RCB: 2017-A02610-53). First trial registration on May 26th, 2018.
Study design
Following a double-blind experimental design, the subjects were randomly assigned either to the real EEG-NF group (referred to as NEURO subjects) or to the sham feedback group (referred to as BIO subjects), in 1:1 allocation ratio, stratified by age, sex and education level. The assignment of subjects to each of these two groups was performed by one of the collaborators not involved in the experiments nor in the data analysis process. The investigators and the participants were all blinded with regard to the groups. Six subjects desisted at the beginning of the study for scheduling reasons, resulting in an unbalanced final sample size: 17 subjects in the NEURO group and 14 subjects in the BIO group.
As summarized in Table 1, the two groups did not differ significantly in terms of age or education level. A difference concerned gender distribution, with a slightly higher proportion of women in the NEURO group (Fisher’s exact test, p value = 0.05).
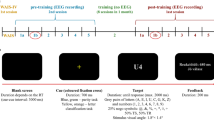
According to the protocol, each subject completed a total of 22 visits during a three-month period: one pre-training visit (V0), 20 training visits (V1 to V20), and one post-training visit (V21) (see Fig. 1).
Schematic representation of the experimental protocol 27.
At V0 and V21, resting-state EEG data was recorded for 75 s with eyes closed at a frequency sampling of 500 Hz using a 19-electrodes EEG cap (Neuroelectrics, Barcelona, Spain) with a 10–20 electrodes layout: Fp1, Fp2, F7, F3, Fz, F4, F8, T7, C3, Cz, C4, T8, P7, P3, Pz, P4, P8, O1 and O2 (see Fig. 2). A broader neuropsychological assessment was also performed at these two sessions (V0 and V21), with normal and stable results within and between groups, as previously detailed in 27.
Description of the experimental protocol and the unsupervised learning approach. (A) Brain Computer Interface (BCI) study design and resting-state EEG recordings at V0 (pre-training) and V21 (post-training). (B) At V0 and V21, pairwise synchrony was computed for each 2-s EEG epoch across channels, then averaged within the frontal, central, and parieto-occipital regions. (C) For each individual, the associated 64 EEG epochs were clustered based on their synchrony values, yielding a 3D representation of individual neural modulation dynamics per brain region. (D) Spectral bi-clustering was applied to these representations from all individuals to identify subjects exhibiting successful neural self-modulation.
During the first training session (V1), the investigator explained to each subject the EEG acquisition procedure and the visual feedback interface. Then, for subsequent training visits (from V1 to V20), the technician placed a low-density EEG system with five electrodes (Cz, Pz, Fz, C3, and C4) on the scalp and two adhesive electromyography (EMG) sensors on the trapezius muscles for all participants in both the NEURO (real EEG-NF) and BIO (sham feedback) groups. The visual feedback was provided as a picture of a tree on the screen, and the subjects were only asked to concentrate on making the tree grow. The feedback presented to subjects of the NEURO group was based on their brain dynamics, whereas in the BIO group, feedback was instead driven by their muscular activity 27.
The EEG-NF training (from V1 to V20) was based on three EEG markers that are reported to decline with age: TBR, PAF, and Gamma-band synchronization 27. Each training session consisted of three 10 min blocks per EEG marker (30 min per visit). At the beginning of each visit, one minute resting-state EEG was recorded to establish the threshold values of each subject’s markers. During training, each marker was computed in real-time from the recorded signals. If the actual computed value of the marker exceeded the threshold, the tree grew,otherwise, the tree size remained unchanged. The same protocol was followed for subjects who received the sham feedback.
In the pre-training (V0) and post-training (V21) visits, resting-state EEG signals were acquired with all 19 EEG electrodes, while during the training visits (V1 to V20), subjects received the neurofeedback through a low-density EEG system with only five electrodes. The rationale behind this choice was to test the practicality of a simple and low-cost device for potential use at home and outpatient setting. The selected electrodes (Cz, Pz, Fz, C3, and C4) were chosen based on prior experiments that we have conducted on an EEG database comprising 22 subjects with Subjective Cognitive Impairment (SCI) and 28 patients with mild AD. To distinguish between the SCI and AD groups, we tested various combinations of a limited number of electrodes out of the 30 available, calculating an average Phase Synchrony measure across different frequency bands. This average synchrony value was then used as input to a Linear Discriminant Analysis classifier to assess discrimination performance in terms of specificity (correct classification of SCI) and sensitivity (correct classification of AD). The best results (specificity = 81.8% and sensitivity = 71.4%) were achieved using the Cz, Pz, Fz, C3, and C4 electrodes 27.
EEG data pre-processing
EEG data analyses were performed on the data collected in pre- and post-training visits from the 31 subjects who completed the study. EEG signals recorded at V0 and V1 were notch-filtered at 50 Hz to eliminate the power line noise and band-pass filtered within the frequency range of 1 to 45 Hz. To remove artifacts embedded in the EEG signals, we employed Independent Component Analysis (ICA) using EEGLAB version 2024.2 30.
The cleaned EEG signals were then segmented into 2-s epochs. Epochs with a kurtosis lower than one or greater than five, or with peak-to-peak amplitude exceeding 100 µV, were excluded from the analysis 31,32. The remaining epochs were visually inspected, and bad segments were rejected. The final number of artifact-free epochs per acquisition ranged from 32 to 37 for each subject. For pre-and post-training analysis, we retained the same amount of data for each subject, i.e., 32 epochs of 2 s each at V0 and V21.
Finally, considering the 32 epochs at pre- and post-training, EEG signals were filtered into five frequency bands: Delta (1–4 Hz), Theta (4–8 Hz), Alpha (8–12 Hz), Beta (12–30 Hz), and Gamma (30–45 Hz) 48.
Intra-region synchrony estimation
To quantify the sensor-level EEG synchrony, we used the Phase-Lag Index (PLI) because of its robustness against noise and volume conduction 33. Notably, PLI ranges from 0 to 1, with values close to 0 indicating the absence of synchronization or low interaction, while values close to 1 indicate strong phase synchronization between the pairs of EEG signals examined. PLI is calculated as follows:
where, Δ(ϕ) is the phase difference at time tk between two time series, computed for all sample points per epoch; sign stands for signum function; \(\langle .\rangle\) indicates the mean value 33.
We computed the pairwise PLI on each 2 s-epoch across all channels (see Fig. 2B), and then averaged the resulting PLI values over all channel pairs within each region. In the whole, we considered three main regions: frontal (FP1, FP2, F7, F3, F4 and F8), central (Fz, Cz, Pz, T7, C3, C4, T8), and parieto-occipital (P3, P7, O1, O2, F8 and P4), as shown in Fig. 2A. Therefore, we obtained an average PLI value per region for session V0 and session V21, resulting in 64 intra-region synchrony values per individual (32 values per session), calculated for each of the three regions.
Two-step unsupervised learning approach
Given the potential heterogeneity of individual responses to feedback, we studied the EEG synchrony in each subject individually, regardless of their group assignment. Specifically, we carried out a two-step clustering procedure: first, at an individual-level, and then at a population-level, considering the intra-region synchrony values estimated at V0 and V21, in the three mentioned brain regions and the five frequency bands.
For each individual, we performed an Agglomerative Hierarchical Clustering (AHC) on all 64 PLI values computed at V0 and V21 per region (see Fig. 2C). The AHC algorithm has the advantage of not relying on random initializations 34,35. We used Ward’s linkage to merge clusters since it tends to create compact clusters that are robust to outliers 36.
For each individual, we fixed the number of clusters to three, which allowed us to consider two extreme behaviors and an intermediate one. Thus, for each individual, we obtained one cluster containing the epochs (from V0 or V21) with the highest PLI values (referred to as “H” for High), one intermediate cluster containing V0 or V21 epochs with medium PLI values (referred to “M” for Medium), and one cluster with epochs showing the lowest PLI values (referred to “L” for Low). Of note, since the AHC was performed per individual, the notions of High, Medium, and Low are subject-dependent.
Based on this individualized AHC, we analyzed the distribution of the V0 and the V21 epochs in each cluster per individual (see Fig. 2C). As such, if the synchrony increased after training, the V21 epochs would predominantly fall into “H” and “M” clusters, while the “L” cluster would contain mostly V0 epochs.
To provide a refined assessment of the connectivity change between pre- and post-training sessions for each of the 31 individuals, we calculated the difference between the number of V21 epochs and the number of V0 epochs in each cluster, resulting in the three values \(\Delta H\), \(\Delta M\) and \(\Delta L\) (see Fig. 2C). This was carried out per brain region and for each frequency band. The retrieved metrics \(\Delta H\),\(\Delta M\) and \(\Delta L\) ranged from − 32 to 32, where a positive value indicates more V21 epochs than V0 epochs in the cluster, and a negative value indicates the opposite. Each individual is thus represented by a three-dimensional pattern \((\Delta H,\text{ and }\Delta L\)) reflecting the evolution of an individual’s synchrony post-training, in a specific brain region within a frequency band. This design allowed each participant to serve as their own reference, ensuring that the extracted three-dimensional pattern represents within-subject differences on a common scale.
As previously mentioned, we focus the current study on Gamma-band synchronization, with subsequent analysis exploring the neurophysiological dynamics related to EEG-NF across the entire frequency spectrum. To this end, we used the Spectral Bi-Clustering algorithm (see Fig. 2D). Specifically, the Spectral Bi-Clustering aims at simultaneously clustering individuals and descriptors of a data matrix to identify subsets with coherent patterns across both dimensions 37,38,39. This is particularly useful when the relationships between subgroups of individuals and features are intricate and important for clusters’ interpretation.
Statistical analysis
The non-parametric Mann–Whitney U test was used to assess the significance of the differences between the 32 PLI values in V0 and the 32 PLI values in V21 for each individual. This test was chosen because the PLI values computed in V0 and V21 per region and frequency band were not normally distributed. For significant differences, we further quantified the effect size using Cliff’s delta 40,41 that estimates the amount of difference between two groups of observations beyond p-values interpretation. Cliff’s delta ranges from − 1 to 1: 0 indicates no difference, and values closer to − 1 or 1 indicate larger differences (positive, if V21 samples are greater than V0 samples; and negative otherwise). The Cliff’s delta, in absolute value, indicates a small effect when ranging from 0.15 and 0.33; a medium effect from 0.33 and 0.47; a large effect from 0.47 and 0.65; and a very large effect above 0.65 41.
Results
Gamma synchrony changes after training at the individual level
For each individual, we first applied the AHC to the PLI Gamma-band synchrony values of the 64 epochs from both V0 and V21, analyzing the frontal, central, and parieto-occipital regions separately. Figure 3 shows the distribution of the 32 V0 epochs and the 32 V21 epochs in the three clusters (H, M, L) for an individual whose synchrony increases globally from V0 to V21 in the three regions.
Distribution of V0 and V21 epochs in the three clusters (H for High, M for Medium, L for Low synchrony). This was observed for an individual whose Gamma synchrony increased at V21, in (a) frontal, (b) central and (c) parieto-occipital regions.
There were globally more V0 epochs than V21 epochs in the cluster with low synchrony values, whereas the V21 epochs predominate in the clusters with high and medium synchrony values. This was observed in the frontal, central and parieto-occipital regions. Nevertheless, among the 31 individuals, some presented a different pattern, suggesting that they did not benefit from training. For a few, we also observed an inverted pattern, reflecting a decrease in their Gamma-band synchrony.
Gamma synchrony changes at the population level
Based on individualized clustering results, we extended our analyses looking at Gamma-band synchrony changes after training at the population-level. To this end, we computed for each individual the three metrics \(\Delta H\), \(\Delta M\) and \(\Delta L\) derived from the individualized clustering for the frontal, central and parieto-occipital regions separately. This resulted in three 3D-patterns (i.e., 9 metrics per individual), describing the evolution of an individual’s synchrony post-training, in each brain region. Then, we used such 3D-patterns to cluster the 31 individuals into four subgroups with an AHC. We generated four clusters to strike a balance between a detailed analysis and a broader overview of the obtained subgroups.
Experiments showed that including the central region in our analyses—the one through which subjects received neurofeedback during the training sessions—led to similar results when considering exclusively the frontal and the parieto-occipital regions. This suggests that the neurophysiological effect resulting from Gamma self-neuromodulation was not limited to the “training” region. In any case, to avoid potential biases in the evaluation of the neurofeedback, we considered exclusively the frontal and the parieto-occipital regions in the following.
Figure 4 displays a clustering map showing the four subgroups (clusters) of individuals differentiated by three horizontal black lines. On the vertical axis, we indicated individuals belonging to each cluster (“N” for NEURO and “B” for BIO individuals). On the horizontal axis, we reported the three metrics resulting from the individual clustering, considering the frontal region (on the left of the vertical black line) and the parieto-occipital region (on the right of the vertical black line). A color scale is used to indicate the intensity of values, from blue (low values) to red (high values).
Clustering map of the 31 individuals in Gamma-band using their 3D-patterns calculated for the frontal (F) and the parieto-occipital (PO) regions. A color scale from blue (low values) to red (high values) indicates the intensity of values. The retrieved metrics \(\Delta H\),\(\Delta M\) and \(\Delta L\) ranged from -32 to 32, where a positive value indicates more V21 epochs than V0 epochs, and a negative value indicates more V0 epochs than V21 epochs.
The clusters are ordered from top to bottom, such that the first cluster (at the top) generally represents a trend towards decreased Gamma synchrony from V0 to V21 (low \(\Delta H\) values and high \(\Delta L\) values), while the fourth cluster (at the bottom) brings together individuals who showed an increase in Gamma synchrony (high \(\Delta H\) and low \(\Delta L\)). Within each cluster, individuals are ordered following the same criterion.
The first cluster (at the top) contains individuals who experienced a decrease in synchrony at V21 in both the frontal and, particularly, in the parieto-occipital region. This is characterized by strong positive values (considering the maximum is 32) in \(\Delta L\) for both regions, and weak to very weak values in \(\Delta M\) and \(\Delta H\). The second cluster below in this order also indicates a decrease in synchrony, while less pronounced than in the first cluster, and primarily affecting the parieto-occipital region. In contrast, the third cluster shows signs of increased synchrony at V21, mostly in the parieto-occipital region. Indeed, the values of \(\Delta H\) and \(\Delta M\) are sometimes positive, while the values of \(\Delta L\) are generally negative. However, this increase is still relatively modest.
Of note, these three clusters contain as many BIO as NEURO individuals and no particular trend emerges regarding their specific training. In contrast, the fourth cluster (at the bottom of Fig. 4) clearly stands out from the others and includes mainly NEURO individuals (six NEURO and two BIO individuals). This cluster exhibits a very strong increase in the synchrony post-training, revealed by the strong positive \(\Delta H\) values and strong negative \(\Delta L\) values in both the frontal and parieto-occipital regions, in sharp contrast to the previous clusters.
Also noteworthy is the fact that the individuals at the very bottom of this last cluster are all NEURO individuals, showing the highest Gamma synchrony increase in at least one of the two brain regions. This is relevant, as it is consistent with our previous study 27 showing the success of Gamma synchrony in individuals who received the real EEG-NF. There were, however, two BIO individuals (B_20 and B_31) with a similar behavior although they had not undergone the EEG neurofeedback training.
In fact, the statistical analysis revealed a significant increase in Gamma-band synchrony for five NEURO (N_14, N_02, N_12, N_23 and N_06) and two BIO (B_20 and B_31) individuals. Notably, Cliff’s delta analysis showed a large (from 0.47 and 0.65) to very large (Cliff’s delta > 0.65) effect size for these five NEURO individuals in both regions. In contrast, only one BIO (B_31) individual displayed a large effect in just one region (parieto-occipital).
Given these findings, we sought to gain a deeper understanding of the neural dynamics that might differentiate the two BIO individuals from the five NEURO individuals, by extending the unsupervised synchrony analysis to include all frequency bands, as outlined in next section.
Synchrony changes from V0 to V21 across the entire frequency spectrum
We computed the synchrony values in Delta, Theta, Alpha and Beta, exactly as done in Gamma. Then, we performed individual clusterings at each frequency band in both frontal and parieto-occipital regions.
We then applied the Spectral Bi-Clustering on all individuals incorporating simultaneously the six metrics from each of the five frequency bands, resulting in a total of 30 input variables per individual. Figure 5 displays the retrieved three clusters delineated by two horizontal black lines. The y-axis reports the individual code and the x-axis indicates the 30 variables used in the bi-clustering analysis.
Bi-clustering of the population based on the six metrics extracted in the frontal (F) and parieto-occipital (PO) regions, at Delta (\(\delta\)), Theta (\(\theta\)), Alpha (\(\alpha\)), Beta (\(\beta\)) and Gamma (\(\gamma\)). A color scale from blue (low values) to red (high values) indicates the intensity of values. The retrieved metrics \(\Delta H\),\(\Delta M\) and \(\Delta L\) ranged from -32 to 32, where a positive value indicates more V21 epochs than V0 epochs, and a negative value indicates more V0 epochs than V21 epochs.
Interestingly, there is one cluster (Cluster 3) regrouping all the five NEURO individuals previously identified, suggesting that these individuals have a distinct neurofeedback responsiveness. As shown in Fig. 5, Cluster 1 (at the top of the map) and Cluster 3 (at the bottom) are highly opposite in terms of features (descriptors); Cluster 2 is an in-between.
Notably, the Bi-clustering findings revealed that the five NEURO individuals in Cluster 3 presented a particular neural dynamic pattern, characterized by high values in \(\Delta M\) and \(\Delta H\), and consistently low values in \(\Delta L\) across all regions and frequency bands. This pattern seems to indicate the ability of such NEURO individuals to modulate their brain activity through EEG-NF training, extending beyond the Gamma band to the entire frequency spectrum.
To evaluate the robustness of the identified three-cluster structure to random initialization or algorithmic stochasticity, we performed multiple independent iterations (20 runs) of Spectral Bi-Clustering using three-cluster structure. The consistency of the resulting cluster assignments across runs was quantified using the Adjusted Rand Index (ARI), which measures the agreement between different clustering solutions. Across the multiple iterations, the clustering solutions demonstrated a high mean ARI of 0.96 (± 0.04), indicating that the three-cluster structure was highly stable and reproducible. This result confirms that the identified clusters reflect intrinsic patterns in the data rather than being artifacts of random initialization or algorithmic variability.
Figure 6 displays the synchrony values of these five NEURO individuals across the 32 epochs at V0 and V21 at all frequency bands in the frontal and parieto-occipital regions, as well as in the central region. For each individual, we displayed a top row with synchrony values at V0 and a bottom row with synchrony values at V21.
Synchrony values of the 32 epochs for the five NEURO individuals (N_02, N_06, N_12, N_14, N_23) in frontal, central and parieto-occipital regions, at different frequency bands. In each plot, an individual is represented by two rows, with the V0 row above the V21 row. A color scale ranging from blue to red indicates the intensity of the synchrony value of each epoch.
All these NEURO individuals showed increased EEG synchrony from V0 to V21 but not of the same intensity. Also, their synchrony at V0 or V21 differed and was subject-dependent. Of note, the increase of synchrony at V21 was observed in these individuals for most of the 32 epochs. Therefore, computing the synchrony per epoch for individual characterization, instead of using an average or a median synchrony value across the 32 epochs, allowed us to uncover distinct and subtle synchrony profiles.
To assess whether the observed increase in synchrony was statistically significant for these five NEURO individuals, we computed the Mann–Whitney test between the 32 synchrony values obtained at V0 and V21, on the frontal and parieto-occipital regions for each subject. Table 2 summarizes the p-values obtained for such individuals per frequency band.
Overall, we observed a significant synchrony increase across different frequency bands, at least in one of the two brain regions (frontal and parieto-occipital). Regarding Gamma, all the statistical tests are significant (p ≤ 0.05), confirming our earlier observation that these subjects were particularly able to increase their neural synchrony in this specific frequency band in both frontal and parieto-occipital regions.
The role of PAF and TBR in individuals with increased connectivity in all frequency bands
The five individuals who exhibited an increase in Gamma-band synchrony, showed a consolidation of their brain connectivity across the entire frequency spectrum. To investigate whether this broader connectivity enhancement could result from a synergic effect of EEG-NF training on the three markers explored in our double-blind study—including not only Gamma synchrony, but also PAF and TBR—we assessed the ability of these five NEURO subjects to further modulate these two specific markers.
As shown in Table 3, there were no significant differences or consistent trends regarding the modulation of these two specific biomarkers. These findings suggest that neither TBR nor PAF training played a key role in the observed enhancement of brain connectivity across the entire frequency spectrum in these five subjects.
Discussion
Advanced aging is a reflection of medical and societal progress, which is, by definition, accompanied by greater vulnerability to malfunction 42, including brain’s functional capacities that tend to decrease with age 43. Lifestyle measures, such as regular physical activity, appear to attenuate the rate of age-related decline, with evidence that it positively impacts brain functioning in a non-specific way 44,45. Neurofeedback, on the other hand, is a self-regulation training tool that helps individuals to learn to modulate the electrical activity of their brain based on specific EEG markers, such as Gamma synchrony.
The current study has two main findings. First, it confirms that elderly individuals can successfully modulate their Gamma activity through EEG-NF training. Second, it demonstrates that the effectiveness of EEG-NF targeting Gamma activity depends on individual neural dynamics across the entire EEG frequency spectrum.
This finding is particularly relevant, as it shows that group-level comparisons between EEG-NF versus sham feedback are no longer sufficient to validate the efficacy of neuro-stimulation on this EEG marker. Moreover, it suggests that group-level analyses can mask important individual variations that are essential for successful neurofeedback. Overcoming this challenge calls for the use of advanced, unsupervised machine learning methods designed to unravel the intricate neural patterns unique to each participant. By applying this approach, we identified at V21, after training, three distinct neuromodulation profiles: one showing broad Gamma-related synchronization, one with Gamma-specific increases, and one with little or no change.
Notably, we isolated a subgroup consisting exclusively of five NEURO individuals, who demonstrated cross-frequency consolidation of Gamma activity, suggesting that this pattern may serve as a neural signature of effective self-neuromodulation. In contrast, Gamma increases without broader synchronization, observed in both NEURO and BIO participants, seems to reflect spontaneous, state-dependent activity rather than a genuine training effect. These apparent false positives may help to explain the high variability often reported in the neurofeedback literature 26. The emergence of these distinct profiles likely stems from individual traits, cognitive strategies, and training intensity 46.
In summary, our findings suggest a novel neural signature of effective self-neuromodulation of Gamma activity. This is supported by a methodological approach that emphasizes the importance of accounting for individual variability in neurofeedback, which poses a specific challenge in this field.
Limitations
This work entails two main limitations. The relatively small sample size restricts the generalizability of the findings, underscoring the need for replication with larger, more diverse populations. The absence of long-term follow-up also precludes conclusions about the durability of the observed changes in Gamma-band synchronization.
Additionally, all 31 participants were cognitively healthy, exhibiting stable neuropsychological performance regardless of feedback type 27. This may account for the absence of EEG-NF effects on cognitive outcomes. Nevertheless, an important open question remains: could the ability of certain individuals to successfully self-modulate Gamma activity serve as a protective factor, potentially preventing or delaying future cognitive decline?
Conclusions
This randomized controlled trial provides unique evidence of the neural dynamics underlying successful EEG-NF training on Gamma activity in older adults at an individual level. By employing an unsupervised machine learning approach focused on individual participants, regardless of group assignment, it highlights the importance of personalized analysis in neurofeedback research. This methodological framework offers valuable guidance for future studies in the promising field of self-neuromodulation in aging.
Data availability
The data generated and analyzed in this study cannot be shared publicly. This is due to the RGPD (Europe) and French laws. Clinical data cannot be shared publicly. However, de-identified raw data could be shared upon specific requests from the Principal Clinical Investigator [Katia Andrade].
References
Babiloni, C. et al. Brain neural synchronization and functional coupling in Alzheimer’s disease as revealed by resting state EEG rhythms. Int. J. Psychophysiol.: Official J. Int. Organization Psychophysiol. 103, 88–102. https://doi.org/10.1016/j.ijpsycho.2015.02.008 (2016).
Pinotsis, D. A., Hansen, E., Friston, K. J. & Jirsa, V. K. Anatomical connectivity and the resting state activity of large cortical networks. Neuroimage 65(4), 127–138. https://doi.org/10.1016/j.neuroimage.2012.10.016 (2013).
Seguin, C., Sporns, O. & Zalesky, A. Brain network communication: concepts, models and applications. Nat. Rev. Neurosci. 24, 557–574. https://doi.org/10.1038/s41583-023-00718-5 (2023).
Yuste, R., Cossart, R. & Yaksi, E. Neuronal ensembles: Building blocks of neural circuits. Neuron 112(6), 875–892. https://doi.org/10.1016/j.neuron.2023.12.008 (2024).
Buzsáki, G. & Wang, X. J. Mechanisms of gamma oscillations. Annu. Rev. Neurosci. 35, 203–225. https://doi.org/10.1146/annurev-neuro-062111-150444 (2012).
Kucewicz, M. T., Cimbalnik, J., Garcia-Salinas, J. S., Brazdil, M., Worrell, G. A. High frequency oscillations in human memory and cognition: a neurophysiological substrate of engrams? Brain: A J. Neurol, 147(9): 2966–2982. https://doi.org/10.1093/brain/awae159 (2024)
Griffiths, B. J. & Jensen, O. Gamma oscillations and episodic memory. Trends Neurosci. 46(10), 832–846. https://doi.org/10.1016/j.tins.2023.07.003 (2023).
Dubois, B. et al. Advancing research diagnostic criteria for Alzheimer’s disease: the IWG-2 criteria. The Lancet. Neurology 13(6), 614–629. https://doi.org/10.1016/S1474-4422(14)70090-0 (2014).
Rajmohan, R. & Reddy, P. H. Amyloid-Beta and Phosphorylated Tau Accumulations Cause Abnormalities at Synapses of Alzheimer’s disease Neurons. J. Alzheimer’s Disease: JAD 57(4), 975–999. https://doi.org/10.3233/JAD-160612 (2017).
Al-Ezzi, A. et al. Disrupted brain functional connectivity as early signature in cognitively healthy individuals with pathological CSF amyloid/tau. Commun. Biol. 7, 1037. https://doi.org/10.1038/s42003-024-06673-w (2024).
Babiloni, C. et al. What electrophysiology tells us about Alzheimer’s disease: a window into the synchronization and connectivity of brain neurons. Neurobiol. Aging 85, 58–73. https://doi.org/10.1016/j.neurobiolaging.2019.09.008 (2020).
He, Y., Chen, Z., Gong, G. & Evans, A. Neuronal networks in Alzheimer’s disease. Neurosci. Rev. J. Bringing Neurobiol. Neurol. Psychiatry 15(4), 333–350. https://doi.org/10.1177/1073858409334423 (2009).
Jafari, Z., Kolb, B. E. & Mohajerani, M. H. Neural oscillations and brain stimulation in Alzheimer’s disease. Prog. Neurobiol. 194, 101878. https://doi.org/10.1016/j.pneurobio.2020.101878 (2020).
Murty, D. V. et al. Stimulus-induced gamma rhythms are weaker in human elderly with mild cognitive impairment and Alzheimer’s disease. Elife 10, e61666. https://doi.org/10.7554/eLife.61666 (2021).
Palop, J. J. & Mucke, L. Network abnormalities and interneuron dysfunction in Alzheimer disease. Nat. Rev. Neurosci. 17(12), 777–792. https://doi.org/10.1038/nrn.2016.141 (2016).
Iaccarino, H. F. et al. Gamma frequency entrainment attenuates amyloid load and modifies microglia. Nature 540(7632), 230–235. https://doi.org/10.1038/nature20587 (2016).
Murdock, M. H. et al. Multisensory gamma stimulation promotes glymphatic clearance of amyloid. Nature 627(8002), 149–156. https://doi.org/10.1038/s41586-024-07132-6 (2024).
Ishii, R. et al. Healthy and pathological brain aging: from the perspective of oscillations, functional connectivity, and signal complexity. Neuropsychobiology 75(4), 151–161. https://doi.org/10.1159/000486870 (2017).
Jiang, R. et al. A Neuroimaging signature of cognitive aging from whole-brain functional connectivity. Adv. Sci. Weinheim, Baden-Wurttemberg, Germany 9(24), e2201621. https://doi.org/10.1002/advs.202201621 (2022).
Murty, D.V., Manikandan, K., Kumar, W.S., Ramesh, R.G., Purokayastha, S., Javali, M., et al. Gamma oscillations weaken with age in healthy elderly in human EEG. NeuroImage, 215: 116826. ISSN 1053–8119, https://doi.org/10.1016/j.neuroimage.2020.116826. (2020)
Perinelli, A., Assecondi, S., Tagliabue, C.F., Mazza, V. Power shift and connectivity changes in healthy aging during resting-state EEG. NeuroImage, 256: 119247, ISSN 1053–8119, https://doi.org/10.1016/j.neuroimage.2022.119247. (2022)
Bakhtiari, A. et al. Power and distribution of evoked gamma oscillations in brain aging and cognitive performance. GeroScience 45(3), 1523–1538. https://doi.org/10.1007/s11357-023-00749-x (2023).
Ichim, A. M. et al. The gamma rhythm as a guardian of brain health. Elife 13, e100238. https://doi.org/10.7554/eLife.100238 (2024).
Hammond, D. C. What is neurofeedback: an update. J. Neurother. 15(4), 305–336. https://doi.org/10.1080/10874208.2011.623090 (2011).
Sitaram, R. et al. Closed-loop brain training: the science of neurofeedback. Nat. Rev. Neurosci. 18(2), 86–100. https://doi.org/10.1038/nrn.2016.164 (2017).
Chiasson, P. et al. Effects of neurofeedback training on performance in laboratory tasks: A systematic review. Int. J. Psychophysiol.: Official J. Int. Organization Psychophysiol. 189, 42–56. https://doi.org/10.1016/j.ijpsycho.2023.04.005 (2023).
Andrade, K. et al. Self-modulation of gamma-band synchronization through EEG-neurofeedback training in the elderly. J. Integrat. Neurosci. 23(3), 67 (2024).
Folstein, M. F., Folstein, S. E. & McHugh, P. R. Mini-mental state. A practical method for grading the cognitive state of patients for the clinician. J. Psychiatric Res. 12(3), 189–198 (1975).
Grober, E. & Buschke, H. Genuine memory deficits in dementia. Dev. Neuropsychol. 3(1), 13–36. https://doi.org/10.1080/87565648709540361 (1987).
Delorme, A., Palmer, J., Onton, J., Oostenveld, R. & Makeig, S. Independent EEG sources are dipolar. PLoS ONE 7(2), e30135. https://doi.org/10.1371/journal.pone.0030135 (2012).
Delorme, A., Sejnowski, T. & Makeig, S. Enhanced detection of artifacts in EEG data using higher-order statistics and independent component analysis. Neuroimage 34, 1443–1449. https://doi.org/10.1016/j.neuroimage.2006.11.004 (2007).
Ross, J. M. et al. A structured ICA-based process for removing auditory evoked potentials. Sci. Rep. 12, 1391. https://doi.org/10.1038/s41598-022-05397-3 (2022).
Stam, C. J., Nolte, G. & Daffertshofer, A. Phase lag index: assessment of functional connectivity from multi channel EEG and MEG with diminished bias from common sources. Hum. Brain Mapp. 28(11), 1178–1193. https://doi.org/10.1002/hbm.20346 (2007).
Jain, A. K. & Dubes, R. C. Algorithms for clustering data (Prentice Hall, 1988).
Sasirekha, K., and Baby, P., Agglomerative hierarchical clustering algorithm. A review. International journal of scientific and research publications, 3(3). ISSN: 2250–3153. (2013) http://www.ijsrp.org/research-paper-0313.php?rp=P15831
Ward, J. H. Hierarchical grouping to optimize an objective function. J. Am. Stat. Assoc. 58(301), 236–244. https://doi.org/10.1080/01621459.1963.10500845 (1963).
Kasim, A., Shkedy, Z., Kaiser, S., Hochreiter, S., Talloen, W. (Eds.), Applied Biclustering Methods for Big and High-Dimensional Data Using R (1st ed.). (Chapman and Hall/CRC, 2016). https://doi.org/10.1201/9781315373966
Kluger, Y., Basri, R., Chang, J. T. & Gerstein, M. Spectral biclustering of microarray data: coclustering genes and conditions. Genome Res. 13(4), 703–716. https://doi.org/10.1101/gr.648603 (2003).
Prelić, A. et al. A systematic comparison and evaluation of biclustering methods for gene expression data. Bioinformatics (Oxford, England) 22(9), 1122–1129. https://doi.org/10.1093/bioinformatics/btl060 (2006).
Cliff, N. Ordinal Methods for Behavioral Data Analysis 1st edn. (Psychology Press, 1996).
Meissel, K. & Yao, E. S. Using Cliff’s delta as a non-parametric effect size measure: an accessible web App and R tutorial. Pract. Assess. Res. Eval. 29(1), 2. https://doi.org/10.7275/pare.1977 (2024).
Tenchov, R., Sasso, J. M., Wang, X. & Zhou, Q. A. Aging hallmarks and progression and age-related diseases: a landscape view of research advancement. ACS Chem. Neurosci. 15(1), 1–30. https://doi.org/10.1021/acschemneuro.3c00531 (2024).
Sikora, E. et al. Cellular Senescence in Brain Aging. Front. Aging Neurosci. 13, 646924. https://doi.org/10.3389/fnagi.2021.646924 (2021).
Cabeza, R. et al. Maintenance, reserve and compensation: the cognitive neuroscience of healthy ageing. Nat. Rev. Neurosci. 19, 701–710. https://doi.org/10.1038/s41583-018-0068-2 (2018).
Erickson, K. I., Donofry, S. D., Sewell, K. R., Brown, B. M. & Stillman, C. M. Cognitive aging and the promise of physical activity. Annu. Rev. Clin. Psychol. 18, 417–442. https://doi.org/10.1146/annurev-clinpsy-072720-014213 (2022).
Kuznetsova, E. et al. Evaluation of neurofeedback learning in patients with ADHD: a systematic review. Appl. Psychophysiol. Biofeedback 48(1), 11–25. https://doi.org/10.1007/s10484-022-09562-2 (2023).
Fell, J. & Axmacher, N. The role of phase synchronization in memory processes. Nat. Rev. Neurosci. 12(2), 105–118. https://doi.org/10.1038/nrn2979 (2011).
Abhang, P. A., Gawali, B. W., & Mehrotra, S. C. 2016. Technological basics of EEG recording and operation of apparatus. In Introduction to EEG- and Speech-Based Emotion Recognition (pp. 23-51). Academic Press. https://doi.org/10.1016/B978-0-12-804490-2.00002-6
Acknowledgements
We warmly thank all the subjects who participated in this project for their strong dedication to this research. We are also grateful to the IM2A research team.
Funding
This randomized control trial was supported by Urgotech, the start-up of Group URGO, Paris, France. The present work was partially funded by Télécom SudParis, as well as IP Paris and Hi! PARIS Center on Data Analytics and Artificial Intelligence. Katia Andrade received a grant from the Fondation Recherche Alzheimer (FRA, https://alzheimer-recherche.org/) in support of her research.
Author information
Authors and Affiliations
Contributions
Nesma Houmani: Conceptualization, Formal analysis, Funding acquisition, Investigation, Methodology, Project administration, Resources, Supervision, Validation, Writing—Original Draft, Writing–review & editing. Rita Yabouri: Data curation, Formal analysis, Investigation, Methodology, Software, Visualization, Writing–review & editing. Sonia Garcia-Salicetti: Project administration, Supervision, Validation, Writing—review & editing. Maxime Bedoin: Formal analysis, Software, Writing–review & editing. Takfarinas Madani: Data curation, Resources, Validation, Writing—review & editing. Katia Andrade: Conceptualization, Funding acquisition, Project design and study conduction as Principal Clinical Investigator, Data collection, Investigation, Resources, Supervision, Validation, Writing—Original Draft, Writing–review & editing.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval and consent to participate
The study was approved by the ethical committee for the Protection of Persons of Ile-de-France XI and performed in accordance with the Declaration of Helsinki (ID-RCB: 2017-A02610-53). First trial registration on May 26th, 2018. All subjects gave written informed consent prior to the experiments.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Houmani, N., Yabouri, R., Garcia-Salicetti, S. et al. Individual neural dynamics of successful Gamma neuromodulation through EEG-neurofeedback in the aging brain. Sci Rep 16, 592 (2026). https://doi.org/10.1038/s41598-025-30212-0
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-30212-0