Abstract
Leptomeningeal collateral (LMC) circulation helps preserve brain tissue during acute ischemic stroke (AIS), with poor collateral status predicting worse outcomes. This study assessed the association between asymmetric dimethylarginine (ADMA), LMCs, infarct volume, and oxidative stress in 101 AIS patients with large vessel occlusion within 6 h or wake-up stroke. LMC status was graded using the Menon score (poor, intermediate, or good) on CT angiography, and recanalization was assessed by the modified Thrombolysis in Cerebral Infarction score. Serum ADMA, NADPH oxidase 2 (NOX2), and nitric oxide (NO) levels were measured at admission (< 6 h, T0), 24 h (T1), and 48 h (T2). Among patients, 43.1% had good, 35.3% intermediate, 21.6% poor LMC status. Higher admission ADMA levels were significantly associated with poor LMC at T0 (p = 0.028) and more severe neurological deficits at T1 and T2 (p = 0.005, p = 0.008). ADMA levels increased over time (p = 0.046), and correlated with NOX2 at T1 (p < 0.001). Rising NOX2 was associated with increased neutrophils (p = 0.013) and decreased lymphocytes (p = 0.006). ADMA may impair endothelial function by reducing NO availability and enhancing NOX2-driven oxidative stress. These findings support a role for the ADMA-NO-NOX2 axis in limiting collateral circulation. Targeting this axis may represent a therapeutic strategy to improve outcomes in AIS.
Similar content being viewed by others
Introduction
Ischemic stroke (IS) is the second leading cause of death worldwide1. Recent advances in reperfusion therapies - mainly intravenous thrombolysis (IVT) and endovascular thrombectomy (EVT) - have improved outcomes, with earlier treatment linked to better functional independence1,2. However, some patients do not respond effectively despite early intervention. Beyond the classic “time is brain” concept, growing evidence emphasizes the “brain tissue window”, where treatment is tailored based on the mismatch between the ischemic core (irreversibly damaged tissue) and the ischemic penumbra (hypoperfused but salvageable tissue). The extent of leptomeningeal collateral (LMC) circulation influences penumbra viability. Patients with poor collateral circulation have worse outcomes even when treated within 4.5–6 hours, compared to those with good collaterals who can benefit beyond this time window (fast and slow progressors)3. This new concept is known as the “collateral clock”4.
LMCs are involved in normal brain function, dilating in correspondence with the active brain areas at a given moment. This phenomenon is the basis of neurovascular coupling. Pressure gradients between the various arterial territories involved would activate LMCs5. Animal models classify collaterals into three types based on their persistence over time: persistent, transient, or intermediate5. Collateral compensation ceases after vessel recanalization or due to “collateral failure,” caused by endothelial damage, platelet aggregation, and increased blood viscosity, leading to microvascular obstruction (no-reflow phenomenon)6. Early ischemia also involves precapillary smooth muscle cell contraction, which worsens hypoperfusion and microthrombosis7,8.
In humans, poor collaterals correlate with older age9, metabolic syndrome10,11, and chronic hypertension12, while ipsilateral extracranial stenosis >75% associate with better collaterals13. Mechanisms driving LMC and smooth muscle cell activation remain unclear but may involve metabolic, hemodynamic, or congenital factors.
Nitric oxide (NO), produced by three nitric oxide synthase isoforms - neuronal (nNOS), endothelial (eNOS), and inducible (iNOS) - is a key vasodilator and endothelial function marker14. eNOS-derived NO preserves cerebral microcirculation, inhibits platelet aggregation, leukocyte adhesion, and smooth muscle proliferation, exerting vascular protection15. After stroke, iNOS expression peaks at 4–7 days16. NO reacts with superoxide (O₂⁻) to form peroxynitrite (ONOO⁻), causing mitochondrial damage, lipid peroxidation, protein nitrosylation, and DNA injury16. eNOS uncoupling, due to lack of cofactors like tetrahydrobiopterin, leads to ROS rather than NO production, exacerbating ischemic damage. NO also inhibits endothelin-1 (ET-1) synthesis, a potent vasoconstrictor, by reducing release of endothelin converting enzyme-1 (ECE-1)16.
Asymmetric dimethylarginine (ADMA) is an endogenous NOS inhibitor and marker of endothelial dysfunction and atherosclerosis17. ADMA levels rise after acute IS, correlating with stroke severity18. It promotes NOS uncoupling, oxidative stress, and inflammation via ROS and NF-kB pathways18. Allopurinol, a xanthine oxidase inhibitor, reduces ADMA and improves endothelium-dependent vasodilation in heart failure patients19.
The main vascular source of reactive oxygen species (ROS) is NADPH oxidase (NOX), especially the NOX2 isoform, which is upregulated in cerebral arteries after IS and contributes to oxidative stress and brain injury20,21,22,23,24. NOX inhibition reduces ischemic brain damage in experimental models24. Excessive NOX2-derived ROS also activates inflammasomes and leads to worsening inflammatory responses and ischemia-reperfusion injury25.
The primary objective of this study was to investigate the correlation between ADMA serum levels, the degree of leptomeningeal collateral circulation and the final ischemic volume in patients with AIS.
The secondary objective was to evaluate whether the levels of this biomarker were associated with increased oxidative stress, worse stroke severity and poor outcomes.
Results
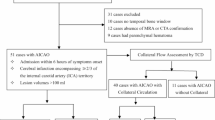
We enrolled 101 patients with LVO AIS, 37 males (36.6%) and 64 females (63.4%); mean ±SD age 76.4 ±12.4 years. Twenty-nine patients (28.5%) had a wake-up stroke or unknown onset of symptoms. Patients were divided into three groups according to the collateral status: poor, intermediate and good collaterals (Figure 1).
The figure illustrates three representative cases of leptomeningeal collateral status—good, intermediate, and poor—in patients with acute LVO stroke. The first row shows a case of LVO involving the M1 segment of the right middle cerebral artery (MCA) with good collateral circulation, as indicated by CTA (circle in A). The corresponding perfusion maps demonstrate a large ischemic penumbra supported by robust collateral flow (CBV in B; CBF in C; MTT in D; and Tmax in E). The second row presents a case with intermediate collateral circulation (circle in F). The associated perfusion maps reveal a smaller penumbra (CBV in G; CBF in H; MTT in I; and TTP in J), indicating moderate collateral support. The third row depicts a case with poor collateral circulation (circle in K). The corresponding perfusion maps show no significant ischemic penumbra, suggesting insufficient collateral perfusion. LVO: large vessel occlusion; MCA: middle cerebral artery; CBV: cerebral blood volume; CBF: cerebral blood flow; MTT: mean transit time; Tmax: time to maximum; TTP: time to peak.
Patients’ characteristics
No statistically significant differences were found among the three groups regarding demographic characteristics, prior medical therapies, blood pressure or heart rate at the three time points (Table 1, Supplementary Table S1 online). However, patients with poor collateral status presented with more severe strokes as per higher NIHSS score at T0 (poor vs. intermediate vs. good collateral status; p = 0.038).
Biomarkers
a) Venous peripheral blood samples
ADMA values significantly increase in all patients from T0 to T2 (112.55 at T0 vs 132.18 at T1 vs 146.42 at T2 p=0.047) regardless of the collateral status (p=0.046) (Figure 2.A, B; Supplementary Table S2). NOX2 serum levels also increased from T0 to T2, although not significantly (p=0.249), and the increase was sharper in poor than in intermediate and good collaterals, although the between-group difference did not reach a statistical significance (p=0.261) (Figure 2. C, D; Supplementary Table S2 online).
Temporal course of ADMA and NOX2 levels in the three collateral groups.
ADMA serum concentration at T0 also correlated with clinical severity at T1 and T2 (Spearman’s rho=0.327 and 0.323; p=0.005 and 0.008, respectively) (Supplementary Figure S1 online. A, B). This association was confirmed after considering dichotomic variable of mild stroke (NIHSS≤6) and moderate-severe stroke (NIHSS >6), respectively at T1 p=0.005 and T2 p=0.013 (Supplementary Fig. 1 online.C, D).
A significant association was found between ADMA level at T0 and LMC; specifically higher levels were associated with poor LMC (mean in overall population 112.55 ng/ml ±31.75; good 102.40 ng/ml ± 25.72 vs moderate 119.80 ng/ml ± 29.30 vs poor 123.85 ng/ml ± 42.36 LMC; p=0.028) (Figure 3).
Association between ADMA levels at T0 and collateral grades.
ADMA at T0 tended to inversely correlate with ASPECT at basal CT (Spearman’s rho= −0.217; p=0.059), although this value did not reach a statistically significance. Median ADMA serum levels at T0 were also associated with carotid atherosclerosis > 70% (86.75 ng/ml vs 115.38 ng/ml in patients with carotid stenosis ≤70%; p=0.049).
ADMA serum levels at T1 correlated with NOX2 at the same time point (Spearman’s rho=0.436; p<0.001) (Figure 4) and tended to be lower in patients who used antiplatelets prior to the index stroke (median levels, 120.76 ng/ml vs 134.68 ng/ml in patients who did not take antiplatelet prior to stroke; p=0.059).
Correlations between serum levels of ADMA and NOX2 at T1.
NOX2 serum levels at T2 were correlated with peripheral blood neutrophils count at T2 (neutrophil absolute number, Spearman’s rho= 0.322, p=0.013; neutrophil percentage, Spearman’s rho= 0.259, p=0.032), and inversely correlated with lymphocytes count at T2 (lymphocyte absolute number, Spearman’s rho=−0.353, p=0.006; lymphocyte percentage, Spearman’s rho= −0.342, p=0.008) (Supplementary Fig. S2 online).
Statin use was associated with lower median levels of NOX2 at T1 (13.36 pg/ml vs. 17.11 pg/ml; p=0.032).
No significant correlations were found between serum biomarkers levels and final lesion volume in all patients and in patients with good and moderate LMC while in patients with poor LMC, correlations between ADMA, NOX2 (positive correlations: Spearman’s rho=0.556, p=0.039; 0.557; p=0.031, respectively), and NO (inverse correlation: −0.550, p=0.034) levels and stroke volume resulted as statistically significant at T0 (Figure 5) and non-significant at the other timepoints.
Correlations between ADMA, NOX2, NO levels at T0 and follow-up lesion volume in patients with poor collaterals.
b) Intracranial arterial blood samples
The biomarker values examined, both in the pre- and post-occlusion arterial intracranial blood samples, at the three time points did not show significant differences between groups according to the collateral status (Supplementary Table S3 online).
Clinical outcome
There were no significant associations between the extent of LMC and clinical outcomes, whether measured as any mRS at 3 months, good outcome (mRS 0–2), excellent outcome (mRS 0–1), poor outcome (mRS 2–6 or 3–6), or in-hospital and 90-day mortality (Supplementary Table S4 online).
Multivariate analysis
Ordinal regression analysis identified ADMA levels at T0 as an independent variable associated with LMC status (estimates 0.019 (95% CI 0.005–0.034.005.034); OR 1.019, p=0.009) after adjustment for age, sex, NIHSS at T0, and NOX2 levels at T0 (Table 2). This result was confirmed even when statin use was included in the model (Supplementary Table S5 online). The binary logistic regression did not identify in the LMC status and the examined biomarkers (ADMA or NOX2) as independent predictors of clinical functional outcome or death at 90 days (Supplementary Tables S6 and S7 online).
Discussion
The extent of collateral circulation partly depends on good collaterogenesis influenced by genetic factors, but this alone appears insufficient to ensure effective LMC activation in LVO. This has prompted research into metabolic and hemodynamic conditions that may regulate collateral activation7,8,9,10,11,12,13. According to the rLMC scoring system, our cohort showed 43.1% with good, 35.3% with medium, and 21.6% with poor LMC status, consistent with proportions reported in the literature26. Poor LMC was significantly associated with higher NIHSS scores at admission and with higher levels of ADMA. Moreover, ADMA serum concentration at T0 correlated with clinical severity at T1 and T2 but not with final infarct volume in all study population; a significant correlation between ADMA levels at T0, and not at the other timepoints, and final infarct volume was found only in patients with poor LMC. Consistently, NO and NOX2 levels at T0 also resulted correlated with final stroke volume only in patients with poor LMC.
We found that ADMA values significantly increased in all patients from T0 to T2 regardless of collateral status—a plausible finding since ADMA is a well-established marker of endothelial dysfunction and oxidative stress27.
The association we found between higher ADMA levels at admission and poor LMC status may indicate a potential role for ADMA in impaired collateral vasodilation, possibly through its influence on NO availability. Supporting this hypothesis, previous studies demonstrated a decrease in CBF following ADMA infusion in healthy volunteers (measured by MRI perfusion)28, and an increase in anaerobic cerebral metabolism in patients with severe carotid stenosis undergoing endarterectomy, indicating that elevated ADMA may impair cerebral perfusion29. Patients presenting with elevated ADMA levels at admission exhibited more severe strokes (NIHSS > 6) at both T1 and T2, which may be attributable to compromised collateral status, in agreement with previously reported findings30. On the contrary, the significant correlation between ADMA levels at T0 and final infarct volume found in patients with poor LMC, suggests that elevated ADMA might not be a mere epiphenomenon of more severe injury and its role in influencing collateral status could not be excluded.
Recently, we observed higher NO concentrations in the early phases of stroke regardless of LMC status, followed by a progressive decrease, supporting the idea that NO plays a key role in LMC activation through collateral vasodilation, before declining later31. According to these findings, higher ADMA at admission may be associated with lower NO levels, poorer collateral status, and more severe stroke symptoms.
Higher ADMA serum levels were also associated with carotid atherosclerosis18,32. ADMA is linked to stroke risk factors and atherosclerosis, and correlates with stroke severity and poor outcomes30,32. This association may arise initially from ischemia-induced cellular proteolysis and secondarily from NOS inhibition30. Another proposed mechanism for ADMA-induced dysregulation of macrophage cholesterol metabolism involves NOX-derived ROS: oxidative stress suppresses liver X receptor alpha (LXRα) activity, downregulating ATP-binding cassette transporters A1 (ABCA1/G1) and impairing reverse cholesterol transport—promoting foam cell formation and atherosclerosis33.
NOX₂ serum levels showed a non-significant upward trend from T0 to T2, likely due to progression of the ischemic penumbra and infarction evolution. ADMA levels at T1 correlated with NOX2 at the same time point, and tended to be lower in patients on prior antiplatelet therapy. ADMA reduces NO bioavailability, contributing to endothelial dysfunction. Concurrently, elevated NOX2 activity increases ROS, exacerbating oxidative stress. This synergy suggests that ADMA may contribute to NOX2 upregulation/activation18, in part through the suppression of DDAH (dimethylarginine dimethylaminohydrolase), the key enzyme responsible for ADMA degradation. Oxidative stress, commonly present in the early phases of stroke, impairs DDAH activity, leading to the accumulation of ADMA. Elevated ADMA levels may contribute to reduced NO production by eNOS, which could diminish NO-mediated regulation of NOX2 activity. This may lead to increased NOX2-driven ROS production, which in turn could further inhibit DDAH activity, potentially establishing a self-perpetuating oxidative cycle (Figure 6).
The ADMA–NO–NOX2 loop. ADMA inhibits NO production triggering NOX2-mediated ROS generation. ROS impairs DDAH, the enzyme that breaks down ADMA inducing more ADMA accumulation. The loop amplifies oxidative injury, promotes collateral failure and worsens severity of stroke.
At T2, elevated NOX2 levels positively correlated with neutrophil counts and inversely correlated with lymphocyte counts, reflecting a possible acute pro-oxidant state largely driven by innate immune activation and neutrophil-derived oxidative bursts34. This heightened oxidative stress may induce lymphocyte apoptosis or impair function, contributing to lymphopenia and compromised adaptive immunity35. These findings underscore the clinical relevance of the neutrophil-to-lymphocyte ratio as a biomarker of systemic inflammation and immune dysregulation in acute post-stroke pathology36.
Statin use was associated with lower median NOX2 levels at T1. Statins have been shown to inhibit NADPH oxidase activation, particularly via suppression of the Rho/Rac1 pathway, thereby reducing NOX2-derived ROS production and improving endothelial redox balance37.
Main limitations of this study are the small subgroup sample sizes and the presence of missing values (although in a low percentage of patients for the majority of the variables) that may have weakened statistical power to detect differences in collateral status vs. outcome, and the single-center design that may reduce generalizability. Additionally, adjusted analyses in some of the explored correlations were not performed and corrections for multiple comparisons were not applied due to the small sample size, and findings should be considered exploratory.
The strengths of the study lie in the comprehensive biomarker analysis (peripheral/intracranial), the integration with neuroimaging, the focus on LMC activation post-AIS, and the prospective design.
In conclusion, our findings indicate that elevated ADMA levels may be associated with impaired LMC vasodilation in AIS, potentially through reduced NO bioavailability. These observations are consistent with the hypothesis that endothelial dysfunction could play a role in limiting collateral vessel recruitment. Additionally, higher ADMA levels were associated with increased NOX2 expression, which may contribute to enhanced ROS production and oxidative stress within the first 24 hours after stroke onset. This oxidative state was further supported by a correlation between NOX2 levels and changes in circulating leukocyte profiles, including increased neutrophil and decreased lymphocyte counts, suggesting a possible link to early immune activation and systemic inflammation.
Together, these findings point to a potentially multifactorial process in AIS involving disrupted endothelial function, oxidative stress, and innate immune responses that may influence collateral circulation. However, further appropriately designed studies are needed to clarify the mechanistic relationships between ADMA, NO bioavailability, NOX2 activity, and clinical outcomes. Future research should also investigate whether targeting the ADMA-NO-NOX2 pathway, ADMA-lowering strategies, or NOX inhibitors, could offer therapeutic benefits in the context of AIS.
Materials and methods
Participants
This is a prospective observational study. We enrolled consecutive AIS patients aged 18 years or older who were admitted to the Emergency Department of Umberto I Hospital – Sapienza University in Rome between November 2019 and December 2021.
Inclusion criteria were: AIS within six hours of symptom onset or wake-up stroke/unknown-onset stroke, with the presence of large vessel occlusion (LVO) in the anterior cerebral circulation (including tandem occlusion – internal carotid artery [ICA] plus middle cerebral artery [MCA], or MCA occlusion at M1 or proximal M2 segments) confirmed by multiphasic Computed Tomography Angiography (CTA).
Exclusion criteria included evidence of hemorrhagic stroke, stroke without LVO, or any contraindication to contrast media injection.
Upon admission, patients underwent a comprehensive general and neurological examination, including ECG. Clinical data were collected, including demographic characteristics and the presence of stroke risk factors such as hypertension, atrial fibrillation, diabetes, hypercholesterolemia, significant carotid stenosis, tobacco use, alcohol abuse, history of transient ischemic attack (TIA), previous stroke, myocardial infarction, renal failure, cancer, and current medication use. Functional status was assessed using the modified Rankin Scale (mRS) both at admission and at 3 months post-stroke as a measure of clinical outcome. Stroke etiology was classified according to the Trial of Org 10172 in Acute Stroke Treatment (TOAST) criteria38.
We defined three timepoints: time 0 (T0) at admission; time 1 (T1) at 24 hours; and time 2 (T2) at 48 hours. At each timepoint, stroke severity was assessed using the National Institutes of Health Stroke Scale (NIHSS), and blood samples were collected. Additionally, glucose levels, arterial pressure, heart rate, and temperature were measured at these timepoints.
Laboratory data
Blood samples were collected from patients into tubes with or without anticoagulant (3.8% sodium citrate) and centrifuged at 3000 rpm for 10 minutes at room temperature. Plasma and serum samples were immediately stored at −80 °C until analysis. Additional blood samples were taken at T0 for routine laboratory tests, including complete blood count, C-reactive protein (CRP), creatinine, glucose, and cholesterol. Some tests (blood count, CRP, and glucose) were repeated at T1 and T2.
Intracranial blood samples were obtained during the endovascular procedure at two timepoints: 1) pre-occlusion - directly from the microcatheter positioned adjacent to the intracranial site of occlusion; and 2) post-occlusion - directly from the microcatheter located beyond the site of occlusion.
sNOX2-dp, ADMA, and NO
Serum NOX2 was measured as soluble NOX2-derived peptide (sNOX2-dp) with an ELISA method as previously reported39. Values were expressed as pg/mL; intra- and inter-assay coefficients of variation were 8.95% and 9.01%, respectively.
Quantitative determination of ADMA levels was measured in serum samples by ELISA kit [TEMA ricerca srl, Castenaso (Bologna), Italy] according to the manufacturer’s instructions. The values for ADMA were expressed in ng/mL. Both intra- and inter-assay coefficients of variation were <10%.
A colorimetric assay kit (Abcam, Cambridge, UK) was used to determine the metabolites of NO (nitrites and nitrates, NOx) in 100 µL of samples under stirring conditions for 10 min at 37 ◦C. Values are expressed as µM. Intra- and inter-assay coefficients of variation were 2.9% and 1.7%, respectively.
Radiological data
All patients underwent multiphasic CTA upon admission to confirm the presence of large vessel occlusion (LVO), assess eligibility for endovascular treatment, and grade the extent of collateral circulation. Patients with wake-up or unknown-onset stroke underwent CT perfusion (CTp) or MRI perfusion (MRIp), and were included in the study only if multiphasic CTA was also performed.
Leptomeningeal collateral (LMC) scoring was conducted using the regional LMC method (rLMCm) described by Menon et al. (2011)26. This scoring system evaluates retrograde filling of leptomeningeal collaterals distal to the occluded vessel, either the middle cerebral artery (MCA) or intracranial internal carotid artery (ICA), by comparing the affected side to the healthy contralateral side. Scores were assigned separately to six MCA regions, the anterior cerebral artery (ACA) region, and the basal ganglia region. Each region was scored as 0 points if vessels were not detectable compared to the contralateral side, 1 point if less prominent, and 2 points if equal or more prominent. Higher scores were assigned to vessels in the sylvian sulcus due to their role as markers of good retrograde collateral filling, scored as 0 points if not detectable, 2 points if less prominent, and 4 points if equal or more prominent. The total LMC score was calculated by summing the scores of individual regions, ranging from 0 to 20 points. Patients were classified as having good LMC if their score was 17–20, moderate LMC for scores between 11–16, and poor LMC if 10 or lower. Based on collateral status, the study population was divided into three groups.
The Alberta Stroke Program Early CT Score (ASPECTS) was calculated on non-contrast CT40.
Patients eligible for endovascular treatment underwent digital subtraction angiography (DSA). Recanalization was assessed on DSA images using the modified Treatment in Cerebral Infarction (mTICI) score41, defined as follows: Grade 0: no perfusion, Grade 1: anterograde reperfusion past the initial occlusion with minimal or slow distal reperfusion, Grade 2a: anterograde reperfusion of less than half of the occluded artery territory (e.g., one major MCA division), Grade 2b: anterograde reperfusion of more than half of the occluded territory (e.g., two major MCA divisions), Grade 3: complete anterograde reperfusion of the previously occluded artery.
Patients were dichotomized into poor recanalization (mTICI 0–2a) and good recanalization (mTICI 2b–3).
Final ischemic lesion volume was measured on diffusion-weighted imaging (DWI) MRI sequences using the ABC/2 formula42. MRI was performed 24–48 hours after admission. For patients with MRI contraindications, follow-up CT scans were used.
Three experts in neuroimaging analysis (M.I., A.C., E.N.) independently rated the LMC status, ASPECTS, and ischemic lesion volume on multiphasic CTA. Any disagreements were resolved through consensus after joint review. If the consensus was not initially reached, a fourth neurologist expert in neuroimaging was consulted (M.D.M.).
All study participants provided written informed consent. The study was conducted according to the Declaration of Helsinki and approved by the Ethics Committee of the University Hospital Umberto I (ID: 5323).
Statistical analysis
Descriptive statistics were performed for the entire cohort and for the three groups of interest. Means or medians were calculated for continuous variables, depending on their normality distribution, while frequencies and proportions (percentages) were used for categorical variables with the exclusion of missing data from the denominator. Chi-square test or Fisher’s exact test, Student’s t test or the Mann-Whitney U test, and ANOVA were used as appropriate to compare demographic, clinical, radiological, and laboratory characteristics among subgroups of patients. Correlations between variables were assessed using Pearson’s or Spearman’s correlation coefficients. The temporal trends of molecular biomarkers at different time points were analyzed, including mean/median changes over time according to the LMC status. Ordinal and binary logistic regression multivariate analyses were performed to individuate whether any of the investigated molecular biomarkers could result as independent variables associated with LMC status and clinical outcome as per mRS 0–2 at 90 days, respectively.
A p-value of less than 0.05 was considered statistically significant.
All analyses were conducted using SPSS software (IBM Corp., SPSS Statistics for Windows, Version 25, Armonk, NY, USA).
Data availability
The data underlying this article cannot be shared publicly without restrictions due to concerns regarding the privacy of the study participants. However, the data may be made available upon reasonable request to the corresponding author, subject to approval by the local Institutional Review Board.
References
Saver, J. L. et al. Time to treatment with intravenous tissue plasminogen activator and outcome from acute ischemic stroke. JAMA 309, 2480–2488 (2013).
Saver, J. L. et al. Time to treatment with endovascular thrombectomy and outcomes from ischemic stroke: A meta-analysis. JAMA 316, 1279–1288 (2016).
Rocha, M. & Jovin, T. G. Fast versus slow progressors of infarct growth in large vessel occlusion stroke: Clinical and research implications. Stroke 48, 2621–2627 (2017).
Vagal, A. et al. Collateral clock is more important than time clock for tissue fate. Stroke 49, 2102–2107 (2018).
Wang, Z. et al. Dynamic change of collateral flow varying with distribution of regional blood flow in acute ischemic rat cortex. J. Biomed. Opt. 17, 125001 (2012).
Zhang, Y. et al. “No-reflow” phenomenon in acute ischemic stroke. J. Cereb. Blood Flow Metab. 44, 19–37 (2024).
Hill, R. A. et al. Regional blood flow in the normal and ischemic brain is controlled by arteriolar smooth muscle cell contractility and not by capillary pericytes. Neuron 87, 95–110 (2015).
Armitage, G. A. et al. Laser speckle contrast imaging of collateral blood flow during acute ischemic stroke. J. Cereb. Blood Flow Metab. 30, 1432–1436 (2010).
Arsava, E. M. et al. The detrimental effect of aging on leptomeningeal collaterals in ischemic stroke. J. Stroke Cerebrovasc. Dis. 23, 421–426 (2014).
Menon, B. K. et al. Leptomeningeal collaterals are associated with modifiable metabolic risk factors. Ann. Neurol. 74, 241–248 (2013).
Lazzaro, M. A. et al. The impact of diabetes on the extent of pial collaterals in acute ischemic stroke patients. J. Neurointerv. Surg. 3, 242–245 (2011).
Chan, S. L. et al. Pial collateral reactivity during hypertension and aging: Understanding the function of collaterals for stroke therapy. Stroke 47, 1618–1625 (2016).
Pienimäki, J. P. et al. Carotid artery stenosis is associated with better intracranial collateral circulation in stroke patients. Cerebrovasc. Dis. 49, 200–205 (2020).
Garry, P. S. et al. The role of the nitric oxide pathway in brain injury and its treatment—from bench to bedside. Exp. Neurol. 263, 235–243 (2015).
Tran, N. et al. Endothelial Nitric Oxide Synthase (eNOS) and the cardiovascular system: In physiology and in disease states. Am. J. Biomed. Sci. Res. 15, 153–177 (2022).
Minhas, R., Bansal, Y. & Bansal, G. Inducible nitric oxide synthase inhibitors: A comprehensive update. Med. Res. Rev. 40, 823–855 (2020).
Cooke, J. P. Asymmetrical dimethylarginine: The uber marker?. Circulation 109, 1813–1818 (2004).
Chen, S. et al. Asymmetric dimethylarginine as marker and mediator in ischemic stroke. Int. J. Mol. Sci. 13, 15983–16004 (2012).
Von Haehling, S. et al. Elevated levels of asymmetric dimethylarginine in chronic heart failure: A pathophysiologic link between oxygen radical load and impaired vasodilator capacity and the therapeutic effect of allopurinol. Clin. Pharmacol. Ther. 88, 506–512 (2010).
Drummond, G. R. et al. Combating oxidative stress in vascular disease: NADPH oxidases as therapeutic targets. Nat. Rev. Drug Discov. 10, 453–471 (2011).
Yoshioka, H. et al. NADPH oxidase mediates striatal neuronal injury after transient global cerebral ischemia. J. Cereb. Blood Flow Metab. 31, 868–880 (2011).
McCann, S. K. & Roulston, C. L. NADPH oxidase as a therapeutic target for neuroprotection against ischaemic stroke: Future perspectives. Brain Sci. 3, 561–598 (2013).
Duan, J. et al. Pathophysiology and therapeutic potential of NADPH oxidases in ischemic stroke-induced oxidative stress. Oxid. Med. Cell Longev. 2021, 6631805 (2021).
Chen, H., Song, Y. S. & Chan, P. H. Inhibition of NADPH oxidase is neuroprotective after ischemia-reperfusion. J. Cereb. Blood Flow Metab. 29, 1262–1272 (2009).
Chen, H. et al. NADPH oxidase is involved in post-ischemic brain inflammation. Neurobiol. Dis. 42, 341–348 (2011).
Menon, B. K. et al. Regional leptomeningeal score on CT angiography predicts clinical and imaging outcomes in patients with acute anterior circulation occlusions. AJNR Am. J. Neuroradiol. 32, 1640–1645 (2011).
Janes, F. et al. ADMA as a possible marker of endothelial damage. A study in young asymptomatic patients with cerebral small vessel disease. Sci. Rep. 9, 14207 (2019).
Kielstein, J. T. et al. ADMA increases arterial stiffness and decreases cerebral blood flow in humans. Stroke 37, 2024–2029 (2006).
Szabo, P. et al. l-Arginine pathway metabolites predict need for intra-operative shunt during carotid endarterectomy. Eur. J. Vasc. Endovasc. Surg. 52, 721–728 (2016).
Brouns, R. et al. Dimethylarginine levels in cerebrospinal fluid of hyperacute ischemic stroke patients are associated with stroke severity. Neurochem. Res. 34, 1642–1649 (2009).
Iacobucci, M. et al. Role of endothelin-1 and nitric oxide in acute ischemic stroke leptomeningeal collateral activation. Int. J. Mol. Sci. 26, 3205 (2025).
Qin, Z. et al. A systematic review of the correlation between serum asymmetric dimethylarginine, carotid atherosclerosis and ischaemic stroke. Eur. J. Clin. Invest. 51, e13558 (2021).
Chen, C. H. et al. The detrimental effect of asymmetric dimethylarginine on cholesterol efflux of macrophage foam cells: Role of the NOX/ROS signaling. Free Radic. Biol. Med. 143, 354–365 (2019).
Tu, D. et al. Activation of neuronal NADPH oxidase NOX2 promotes inflammatory neurodegeneration. Free Radic. Biol. Med. 200, 47–58 (2023).
Chen, J. et al. NADPH Oxidase 2-derived reactive oxygen species promote CD8+ T cell effector function. J. Immunol. 212, 258–270 (2024).
Zuo, L. et al. Circulating neutrophil-to-lymphocyte ratio predicts stroke-associated infection and poststroke fatigue affecting long-term neurological outcomes in stroke patients. Mediators Inflamm. 2025, 5202480 (2025).
Chen, W. H. et al. Advances in the molecular mechanisms of statins in regulating endothelial nitric oxide bioavailability: Interlocking biology between eNOS activity and L-arginine metabolism. Biomed. Pharmacother. 171, 116192 (2024).
Adams, H. P. Jr. et al. Classification of subtype of acute ischemic stroke. Definitions for use in a multicenter clinical trial. TOAST. Trial of Org 10172 in Acute Stroke Treatment. Stroke 24, 35–41 (1993).
Carnevale, R. et al. Oleuropein, a component of extra virgin olive oil, lowers postprandial glycaemia in healthy subjects. Br. J. Clin. Pharmacol. 84, 1566–1574 (2018).
Aviv, R. I. et al. Alberta stroke program early CT scoring of CT perfusion in early stroke visualization and assessment. AJNR Am. J. Neuroradiol. 28, 1975–1980 (2007).
Zaidat, O. O. et al. Recommendations on angiographic revascularization grading standards for acute ischemic stroke: A consensus statement. Stroke 44, 2650–2663 (2013).
Sims, J. R. et al. ABC/2 for rapid clinical estimate of infarct, perfusion, and mismatch volumes. Neurology 72, 2104–2110 (2009).
Acknowledgements
The authors thank the clinical staff of the Emergency Department and Stroke Unit, for their assistance in data acquisition.
Funding
This work was supported by Sapienza, University of Rome: Ricerca Ateneo Sapienza Progetti per Avvio alla ricerca (protocol number: AR120172B783068D).
Author information
Authors and Affiliations
Contributions
M.D.M. conceived and designed the study, enrolled the patients, interpreted the results, edited Figure 5 and prepared the original manuscript. M.I., E.N., and A.C. are neuroimaging experts who evaluated collateral status and measured infarct volumes. M.I. also edited Figure 1 of the manuscript. S.L. analyzed and interpreted the results, and edited Figures 2–5, all tables, and the supplementary materials. A.R., I.B., A.P., and M.B. participated in patient enrollment and data collection. R.C. and V.C. performed the serological biomarker analyses and contributed to result interpretation. S.L. and D.T. critically reviewed and edited the manuscript. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests. The authors declared no potential conflicts of interest with respect to the research, authorship, and publication of this article.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
De Michele, M., Risitano, A., Iacobucci, M. et al. The ADMA–NO–NOX2 axis and its association with leptomeningeal collateral impairment in acute ischemic stroke. Sci Rep 16, 753 (2026). https://doi.org/10.1038/s41598-025-30283-z
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-30283-z