Abstract
New poly-active magnetic-activated carbon catalysts (5% MnO2-X% Fe2O3/AC, X = 0–3) are developed to examine, for the first time, the integrated impact of varying magnetic loadings and manganese active species on the performance of catalytic oxidative desulfurization (ODS) of real heavy oil. Desulfurization reactions are conducted in a novel oscillatory baffled reactor (OBR) and a laminar flow baffled reactor (LFBR). Fast ODS is examined using a new basket central baffle in OBR to handle the solid catalyst for continuous operation and to provide easy regeneration and recyclability, compared to previous works that investigated only batch processes using solid catalytic materials in an oscillating reactor. Also, the desulfurization performance is examined in the presence (OBR) and absence (LFBR) of oscillatory mixing to investigate the importance of oscillatory parameters in enhancing mass transfer and sulfur oxidation rates. Sour heavy oil (8281 ppm of sulfur) is upgraded to produce ultraclean and eco-friendly fuel with only 30 ppm sulfur content using MFAC3 catalyst across a very short desulfurization time (12 min) under the best operational parameters (temperature of 90 °C, St of 0.103, Reo of 1168, Ren of 8.74, and Ψ of 133.6). The sulfur removal efficiency is decreased from 99.64% in OBR to 42.06% for LFBR in the absence of the oscillatory motion under the optimal conditions. This dramatic decrease indicates that the oscillatory mixing is the critical and controlling factor in producing eco-friendly fuel via ultradeep desulfurization. The new integrated catalytic system of oscillating reactor- poly-active magnetic-activated carbon catalyst is presented as a promising practical deep desulfurization technology under cost-effective consideration owing to the utilization of easily available and cheap carbonaceous catalyst across non-hydrogenation in the absence of expensive hydrogen gas and catalysts. Scalability of the new oscillating reactor can be directly achieved in the industrial refineries based on the dimensionless parameters of OBR while maintaining similar desulfurization performance.
Similar content being viewed by others
Introduction
Upon fuel combustion, toxic gaseous emissions (SOX) are released and lead to hard environmental and industrial issues such as acid rain, corrosion of industrial units, and deactivation of the expensive refinery catalysts1,2,3. Strict environmental regulations determine the allowable concentration of sulfur species in petroleum oils to less than 10 ppm4. Hydrodesulfurization (HDS) is the traditional industrial process to extract most of the sulfur components. It is not a promising technique in eliminating heterocyclic organic sulfur species, like thiophene (Th) and their sterically hindered derivatives5,6,7. Also, HDS achieves under high operational severity (high temperature and pressure), and utilizes expensive and explosive hydrogen8,9. Therefore, finding developed alternative or complementary desulfurization processes for the production of ultra-clean fuel is an urgent matter. Oxidative desulfurization (ODS) has gained more attention among all desulfurization techniques due to its mild operational parameters (P = 1 atm and T > 100 °C), and attractive performance to extract sulfur components10,11. A critical success factor for oxidative desulfurization is the selectivity of catalyst/oxidant systems and a suitable contacting mode (reactor type). Up to now, numerous homogeneous and heterogeneous catalysts with gaseous or liquid oxidants have been utilized for ODS technology, such as (Fe/ HY-zeolite + SiO2)/air12, (Fe2O3/ Al2O3 + CNF)/air13, (MoO3/Al2O3)/ t-BuOOH14, (Mn/AC)/ O215, (Fe/AC)/ O216, (Fe2O3/AC) / O217, CdO/H2O218, etc. Among the active metals of ODS catalysts, Fe and Mn catalysts are an appropriate choice for accelerating the desulfurization reactions due to the easy alteration of oxidized state, low cost, and promising reaction activity19,20. Appropriate selection of support material is critical for catalyst activity towards efficient desulfurization, which provides a suitable surface area for the spreading of active metals for the oxidation reactions21. Activated carbon proved to be the most reactive among other catalyst carriers based on attractive advantages like low cost, easy availability, significant surface area, microporous/mesoporous structure, and high physiochemical characteristics, stable morphology, and can be synthesized from different precursors like wood, charcoal, sewage sludge, etc., at a high production rate22. Kayedi et al. evaluated the activity of ODS in removing of DBT from toluene as a model oil and utilizing Fe2+ ions as active catalyst ions. They explained that an 89.39% removal efficiency was conducted23. Trisunaryanti et al. tested the catalytic reactivity of ZnO-activated carbon for DBT oxidation utilizing 30% H2O2 as oxidizing agent. The results proved that the DBT removal was 93.83% under 120 min. The high surface area and acidity of activated carbon provided better activity for desulfurization reactions24.
Up to now, the ODS process has been continuously developed using various reactors, such as, batch, flow, and oscillatory reactors. Providing a flow pattern near-plug flow reactor can be satisfied under laminar flow conditions via an oscillatory baffled reactor (OBR). It is composed of a tubular column that is filled via an internal baffle. In OBR, consistent mixing and boosting transfer rates are satisfied by interacting the internal baffles and the oscillatory motion, while maintaining flow behavior near plug flow25,26. The net Reynold’s number (Ren) in OBR is described based on the Eq. 1:
where, u: superficial net flow velocity in OBR column (m s−1), D: column diameter (m), ρ (kg/m3), and μ (kg m−1 s−1) are density anviscosity respectively. Residence time is described as:
where, V: volume of OBR reactor and v0: fluid volumetric flow rate.
And the velocity of fluid is described as:
where, Ac: cross-section area of the OBR reactor (m2).
Three dimensionless numbers can control the dynamic fluid behavior in OBR. Firstly, the oscillatory Reynold number (Reo):
where A: center-to-peak oscillatory amplitude (m), and f: oscillatory frequency (Hz)27. Reo controls the intensity of mixing. Improving the oscillatory conditions (amplitudes and frequency) can provide a wide intensity range of mixing28. Secondly, the Strouhal number (St) is:
This dimensionless group measures vortex propagation27,29,30. High values of St are obtained at lower amplitudes, providing a poor vortex. The typical values of St range from 0.01–931,32; however, it ranges from 0.15–433,34,35. Finally, the velocity ratio is presented as follow36.
OBR is developed in the present work using a unique design of basket central baffles as an alternative to traditional central baffles. The new designs of OBR are utilized for the first time to handle catalyst particles in baskets of baffles for continuous ODS. Also, this study is investigated the activity of 5%Mn/Activated carbon which is synthesized via wet impregnation method and decorated with different concentrations of magnetic active metals (0%, 1%, 2%, and 3% Fe) to produce promising poly-active magnetic-activated carbon catalyst for rapid and ultradeep desulfurization technology of real heavy oil (8281 ppm of sulfur compounds) under mild process conditions. Desulfurization of heavy oil under different oscillatory flow dimensionless groups is investigated. Also, the scalability and economic considerations for the production of ultraclean heavy oil are highlighted.
Experimental
Materials
Non-hydrogenated heavy fuel (gasoil) is utilized as a real feedstock in the present work. Heavy fuel is provided from Baji Refineries Company, Iraq. The most important physical properties of the used feedstock are as follows: Specific gravity@15 °C of 0.8173, API at 60°F of 41.6, sulfur content (ppm) of 8281, Cetane Index of 53, and boiling range of (177–350) °C. Hydrogen peroxide (H2O2) with a purity of 35% is provided by PanReach AppliChem Company, Spain, to employees as a liquid oxidizing agent. The activated carbon (AC) with particle size in the range of (1–3) mm is supplied from Applichem GmbH, Germany, to use as a catalyst support. Manganese (II) nitrate tetra-hydrate (MnN2O6.4H2O) with purity of 97% and ferric nitrate non-hydrate Fe(NO3)3.9H2O with purity of 98% are purchased from (Sigma-Aldrich company, USA) and (Thomas Baker company, India) and utilized as a hydrate salt to produce the catalyst active metals (manganese oxide and iron oxide).
Catalyst preparation
Synthesis of (5% MnO2/AC) catalyst
The preparation of MnO2 over activated carbon (5% MnO2 /AC) catalyst is conducted using the WI process. 3.6 g of hydrated manganese salt (MnN2O6.4H2O) dissolved in 100 ml of water. The prepared solution was stirred (1 h) by a hot plate magnetic stirrer at 500 rpm. After the complete dissolving of salt, 15 g of activated carbon is gradually mixed with the solution using a magnetic stirrer (1 h and 500 rpm) and then mixed utilizing an ultrasonic bath (2 h) at a power rate of 65% to improve the interaction of activated carbon with manganese salt. Prepared solution kept stagnant for 12 h to ensure efficient dispersion of Mn over activated carbon and in its pores. After that, the prepared solution is mixed and heated using a hot plate magnetic stirrer at a temperature of 85 °C for 1 h and under a constant stirring rate (500 rpm) to obtain a solid mixture by evaporation of the liquid. This solid mixture was kept in a drier at 110 °C and kept overnight. The produced dried materials were pushed into a horizontal tabular furnace and calcined under a nitrogen environment at 150 °C (1 h), 300 °C (1 h), 450 °C (1 h), and 600 °C (2 h) using a heating rate of 5 °C /min. The impregnated manganese salt over the activated carbon converted into manganese oxides during the calcination process. Thus, (5% MnO2 /AC) catalyst is synthesized and marked as MFAC0.
Decoration of (5% MnO2/AC) catalyst with different ratios of magnetic oxide
The prepared (5% MnO2/AC) catalyst was decorated with different amounts of iron oxide to enhance the magnetic properties of the composite catalyst. The (5% MnO2 /AC) catalyst impregnated via the WI method with different quantities of ferric nitrate non-hydrate salt using deionized water to produce three types of heterogeneous composite catalyst (5% MnO2- X% Fe2O3/AC). Specified amount of Fe(NO3)3.9H2O salt (1.1, 2.2, and 3.4) gm dissolved in specified amount of deionized water (65, 66, and 67 ml). The produced solution was mixed (1 h) using a hot plate stirrer under 500 rpm mixing intensity. 15 g of AC was added into the prepared homogeneous solution (manganese and iron) with stirring for 1 h using a magnetic stirrer (500 rpm) and an ultrasonic apparatus for 2 h at 65% power rate to improve the dispersion of manganese and iron salts over AC. After that, the homogeneous mixture remained stagnant overnight to ensure good dispersion of active metal oxides over the AC. Then, the mixture is primarily dried with mixing via a magnetic stirrer at 85 °C and 500 rpm for 1 h. These obtained materials were dried using an oven for 12 h at 110 °C. After that, the dried materials were calcined under a nitrogen atmosphere at 150 °C (1 h), 300 °C (1 h), 450 °C (1 h), and 600 °C (2 h) in a horizontal tabular furnace (heating rate: 5 °C/min). The calcination step converted the metal salt (Mn and Fe) to metal oxides. Finally, (5% MnO2- 1% Fe2O3/AC), (5% MnO2- 2% Fe2O3/AC), and (5% MnO2- 3% Fe2O3/AC) catalysts are synthesized and marked as MFAC1, MFAC2, and MFAC3 respectively.
Catalyst characterization
Scanning electron microscopy (SEM) and Energy Dispersive X-ray analysis (EDX) are used to measure the surface morphology and the distribution of the chemical elements for poly-active magnetic-activated carbon catalysts. Brunauer–Emmett–Teller analyzer (BET) (Micromeritics, USA) is employed to measure the surface area, pore size distribution, and total pore volume for catalysts. X-ray diffraction (XRD) is employed to characterize the crystalline phase within the synthesis catalysts and observe the phase identification based on the diffractions of catalyst components. Fourier transform infrared spectroscopy (FTIR, SHIMADZU, Japan) is conducted to measure acidity and base groups present. Thermal Gravimetric Analysis (TGA) is conducted to characterize the thermal stability of catalysts.
Experimental set up of the novel OBR
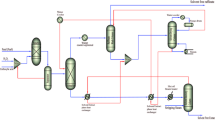
Oscillatory baffled reactor with a unique design of basket central baffles designs, installs, and operates to handle the catalysts for ultradeep catalytic desulfurization employing H2O2 as a green oxidizing agent. The unique design of the OBR unit is installed as a pilot plant, as presented in Fig. 1. The geometrical parameters of the oscillatory baffled reactor are observed in Table S1.
(A) Experimental setup of the unique OBR unit and (B) Real picture for OBCBR unit.
The oscillation motion is supplied at the bottom of the reactor tube using a local mechanical oscillatory pump. Changing the shifting of the pump plunger and the voltage value of the mechanical oscillatory pump adjusted the oscillation amplitudes and oscillation frequency, respectively. The sour heavy passed into the OBR bottom using a dosing pump (net flow: MFG. NO. 5228, Taiwan). The desired oxidant flow rate is fed into OBR at the bottom using a syringe pump (IML company, Spain) with a flow rate of 0–5.15 ml/sec. The heavy oil and hydrogen peroxide are fed in a co-current mode. An efficient heater (temperature range: 0–600 °C) is utilized as a jacket surrounding the OBR tube. The REX-C controller is employed to control the reactor heater. The glass wool insulation is used as an efficient insulator with negligible heat loss and surrounds the reactor tube. The treated heavy oil from the OBR top is fed through an efficient local cooler. This system kept the treated heavy oil in the liquid phase at room temperature. The cooled treated heavy fuel phase is withdrawn and kept in a suitable container for sulfur content analysis. OBR parts are fabricated from 316 stainless steel. The basket’s central baffle is a unique baffle compared with the conventional designs of OBR baffles, as illustrated in Fig. 2.
Major baffles design in OBR: (a) integral baffles with periodic constrictions than can be sharp or smooth (b) central axial baffles (c) round-edged helical baffles (d) sharp-edged helical baffles (e) sharp-edged helical baffles with a central rod (f) wire wool baffles (g) single-orifice baffles (h) multi-orifice baffles (i) disc and doughnut baffles, (j) multi orifice baffles, (k) Unique design of baskets central baffle.
Oxidative desulfurization in novel oscillatory baffled reactor
Dimensionless evaluation of ODS process in OBR LFBR using four types of poly-active magnetic-activated carbon catalyst under different operational parameters (Table 1) is examined. 112 ml of aqueous hydrogen peroxide solution used for each 1224 ml of real heavy oil (8281 ppm S) to provide molar ratio of H2O2 to sulfur (O/S) of 5 as recommended in most previous studies44. The experiments achieved by filling the baskets of baskets central baffles via the specified synthesis (5% MnO2- X% Fe2O3/AC) heterogeneous catalysts and inserted it inside the reactor tube. Sour heavy oil fed into reactor bottom using the dosing pump at specified net Reynold number (oxidation time). The desulfurization temperature kept at desired value using the temperature controller. Hydrogen oxide fed using the syringe pump into the oscillatory baffled reactor at the desired amount. The fluid oscillation satisfied inside the OBR using oscillatory pump, which adjusted at desired oscillation conditions (A and f). After reaching the oxidation reaction the steady state conditions, product passed through the cooler. The cooled products kept in separation funnel to obtain on two phases (aqueous and fuel). Then, the fuel phase putted in appropriate testing container and pushed for sulfur content analysis process. The total sulfur concentration in the sour and treated fuel measured using X-ray fluorescence (XRF) (Sindie-R2, Sindie Company, USA) based on standards measuring conditions (ISO 20884 and ASTM D7039). The sulfur removal calculated based on Eq. 2.
where, \(C_{Sin}\), \(C_{Sout}\) are the initial and final content of sulfur compounds in the heavy fuel.
After completing the experimental runs, OBR cooled to ambient conditions. Then, the basket’s central baffle pushed out, and nitrogen gas fed through the OBR to remove any by-product gases. Finally, ethanol was pushed into the reactor for cleaning. Each experiment was repeated twice, and the average concentration of sulfur (ppm) was discussed. The same experimental procedure was applied in a laminar flow baffled reactor (LFBR) by operating the reactor without oscillatory motion (stopping the mechanical oscillatory pump) to observe the importance of oscillation conditions in enhancing the performance of OBR toward sulfur removal efficiency. The LFBR experiments were achieved at the optimal operating conditions (T and Ren) using all types of the synthesis heterogeneous composite catalysts. Also, the desulfurization process under optimum conditions is conducted in a necked flask (150 ml) with a stirrer bar and a hot plate magnetic stirrer to use as a stirred tank reactor (STR) to exhibit the importance of OBR in enhancing the performance of ODS technology. Blank experiments without a catalyst, oxidant, or active metallic oxides (only AC without Mn and Fe oxides) were conducted to prove the importance of the reacting species in providing a fast and ultradeep oxidative desulfurization process. Under all operating conditions, oxidative desulfurization experiments are achieved with two replicates (n = 2).
Results and discussion
Catalyst characterization
FTIR analysis
FTIR spectra of MnO₂/Fe₂O₃-impregnated and activated carbon (AC) reveal dramatic surface chemistry changes, confirming the successful introduction of functional groups for adsorption and catalysis applications (Fig. 3). Hydroxyl/phenolic groups and adsorbed water are confirmed to be present by the broad O–H stretching band near 3440 cm⁻1, consistent with earlier reports on chemically activated carbon37,38. The presence of carboxyl and quinone functionalities, which contribute to polarity and metal anchoring, is confirmed by the sharp C=O stretching peak at 1630 cm⁻1, as demonstrated in earlier research on basic-activated carbon39,40,41. MnO₂ impregnation introduces a characteristic Mn–O stretching vibration near 580 cm⁻1, consistent with the characteristic δ(Mn–O) modes of MnO₂’s octahedral structures42,43. The broadening of the O–H stretching band near 3440 cm⁻1 and the decreased intensity of the C=O stretching band near 1630 cm⁻1 are indicative of interactions such as hydrogen bonding and charge transfer between MnO₂ and AC’s surface hydroxyl and carbonyl groups44,45 . Similarly, impregnation of Fe₂O₃ results in Fe–O stretching vibrations between 550–600 cm⁻1 with intensities proportional to Fe loading levels (1–3 wt%), consistent with earlier reports on Fe₂O₃/carbon composites. Weakening of the O–H stretching bands indicates coordination of the Fe3⁺ ions with surface hydroxyl groups to form Fe–OH complexes, a phenomenon responsible for the reduction of surface hydrophilicity and enhancement in Lewis acidity46,47 . Additionally, the displacement of the C=O stretching band to 1620 cm⁻1 indicates the formation of Fe–carboxylate complexes (Fe–O–C=O), stabilizing Fe₂O₃ particles against aggregation.
FTIR Analysis profile for AC and (5% MnO2- X% Fe2O3/AC, X = 0–3) heterogeneous composite catalyst.
XRD analysis
XRD studies utilize to examine phase composition, crystallinity, and the effect of metal oxide on the AC structure (Fig. 4). There are distinct peaks in 2θ values of approximately 26.5°, 43.5°, and 57.5° on the pristine activated carbon that has been functionalised with acid and basic reagents. From JCPDS No. 41-1487 it can be seen that these reflections match the (002), (100), and (110) activated carbon planes respectively48,49. The presence of short-range ordered domains typical in the case of chemically activated carbon materials is proved by these reflections. The addition of 5 wt.% MnO₂ to AC (spectrum MFAC0) causes the emergence of new diffraction peaks in 2θ ≈ 28.7°, 37.4°, 41.9°, 49.8°, 60.2°, and 65.4° corresponding to the (110), (101), (211), (310), (400), and (301) planes of α-MnO₂, respectively (JCPDS Card No. 44-0141)50,51,52. The presence of these peaks indicates the successful deposition of nanocrystalline MnO₂ on the surface of the AC matrix. At 1 wt.% Fe₂O₃(spectrum MFAC1), new 2θ angles appear near 24.1°, 33.2°, 35.6°, 40.9°, 54.1°, and 62.5° corresponding to the (012), (104), (110), (113), (116), and (214) planes of α-Fe₂O₃ hematite (α-Fe₂O₃), with conformance to JCPDS Card No. 33-066453,54. The new reflections coexist with α-MnO₂ reflections, implying a multi-phase composite that is stable. Increasing Fe₂O₃ content (2 wt.%, spectrum MFAC2)) enhances the α-Fe₂O₃ reflections, particularly the (104) and (110) reflections near 2θ ≈ 33.2° and 35.6°. At the same time, MnO₂ reflections are decreased in intensity, evidence of surface competition between the oxides. At 3 wt.% Fe₂O₃ (spectrum MFAC3), the diffraction pattern is dominated by reflections from hematite. MnO₂ reflections are strongly damped and the graphitic background is nearly unrecognizable, evidence of surface encapsulation of iron oxide or phase-preferred crystallization. The moderate peak broadening in diffraction of doped samples, particularly with lower Fe₂O₃ concentrations, indicates localized lattice distortion instead of fully amorphous character.
XRD pattern for AC and (5% MnO2- X% Fe2O3/AC, X = 0–3) heterogeneous composite catalysts.
SEM analysis
Figure 5 shows the FESEM pictures for the catalysts. The FESEM images explain that the catalyst surface showed a uniform porosity. Morphology of the AC surface is microporous with cavities and cracks (Fig. 5a, b). These cavities and cracks can be returned to thermal effects during calcination under severe temperatures (600 °C). Figure 5c–j observes that a significant distribution of MnO2 and Fe2O3 over the surface of AC. This uniform distribution boosts the oxidation reactivity of the catalyst by providing widely spread active sites over a large surface of AC. Also, small clusters of iron and manganese oxides enhance the number of active interfacial sites and boost rapid electron/oxygen transfer and oxidant activation, thus performance and selectivity in ODS upgrade63. Two magnetic and manganese active metal oxides were significantly observed in the marked white region by orange arrows and differ from the gray regions of the activated carbon surface. Figure 6 also proves that the distribution of active metal oxides increased with increasing the amount of magnetic oxide at a constant quantity of manganese oxide.
FESEM images for: (a, c) AC, (c, d) MFAC0, (e, f) MFAC1, (h, g) MFAC2, and (i, j) MFAC3.
Thermo-gravimetric analysis profile for AC and (5% MnO2- X% Fe2O3/AC, X = 0–3) heterogeneous composite catalyst.
EDX analysis
Figure S1 exhibits the EDX results. It shows different elements (Mg, Al, Si, S, P, Cu, Na, Ti, and Ca) at various percentages, whereas the major percent is for carbon and oxygen elements. These results can be attributed to the presence of some impurities at low amounts on the testing grade, which have an unobvious effect on catalyst activity. Large amounts of carbon and oxygen for AC and all synthesis catalysts proved a remarkable purity for the used materials. The results of the synthesis catalysts after loading the active metal oxides (Fig. 6b–e), it can be noticed an obvious percent of Fe and Mn (6.66% and 0% Fe), (5.63% Mn and 1.9% Fe), (5.4% and 2.64%), (5.01 Mn% and 3.74% Fe) for MFAC0, MFAC1, MFAC2, and MFAC3 catalysts which impregnated on activated carbon.
N2 adsorption–desorption analysis
The texture properties (surface areas (SA), pore volumes (PV), and pore size (PS)) for the catalyst were evaluated using N2 adsorption–desorption analysis (BET test). The obtained results are illustrated in Table S2 and Fig. S2. As presented in Table S2, loading of AC via 5% manganese oxide and decoration via different amounts of iron oxide resulted in a reduction in surface area and pore volume. These results can be assigned to the deposition of Mn and Fe species within the carbon’s porous structure, which covers and blocks some pores of activated carbon partially or completely55. Also, the decrease in the texture properties can be attributed to the pores distortion through the uploading of Mn and Fe species under the elevated calcination temperatures (600 °C)56. As well as, partial destruction in the activated carbon pore structure, there is a return to erosion of micropore walls and a reduction in the texture properties57,58. Table 8 also observes an increase in pore size after impregnating the Mn and Fe molecules on activated carbon. This increase can be assigned to significant modification and redistribution in the pore structure due to decoration with active oxide59,60. On the other hand, the elevated calcination temperature can change the pore structure and enlarge original pores or form others, thereby resulting in increasing APSs61. In ODS technology, the catalytic performance of poly-active magnetic-activated carbon catalyst significantly depends on surface area and pore accessibility. A high-surface-area, well-developed pore network improves the dispersion of manganese and iron oxides, enhancing the number of exposed redox-active sites and boosting the mass transfer rate of sulfur species and hydrogen peroxide within catalyst pores62,63.
The presented results in Fig. S2a–f illustrate a severe adsorption pattern conducted at low relative pressure (< 0.1). These isotherm data prove that catalysts are microporous with a narrow pore size distribution. Figure S2a, b proves that a high adsorbed quantity of N2 gas is achieved under lower relative pressure, whereas at intermediate and high values. Isotherm results observe H4 type hysteresis loops (micro-pores structure), and then hysteresis loops (medium-macro pores)16,64,65. Figure S2c, d) indicates to a significant increase in the pressure to pore volume ratio with increasing relative pressure. These results prove the microporous structure with narrow pore size distribution for the prepared catalysts. Figure S2e, f illustrates the BJH adsorption/desorption cumulative pore volume. The obtained results refer to a sharp decrease in pore volume at pore widths below 10 nm and then a slight decrease till 40 nm with significant stability after that. These results observe the narrow pore size distribution for the synthesis catalysts.
TGA analysis
Thermal Gravimetric Analysis determined mass loss and thermal stability of catalysts under the impact of elevated temperatures (0–1000 °C). The obtained TGA data are presented in Fig. 6. The results of thermal treatment proved that AC and the prepared four heterogeneous catalysts observed better thermal stability at low temperatures (< 100 °C). The weight loss percentages were 2.39%, 1.25%, 2.04%, 2.17%, and 1.74% for AC, MFAC0, MFAC1, MFAC2, and MFAC3, respectively, at low temperature (100 °C). These data explained that prepared catalysts can be used for sulfur oxidation at the present moderate operating conditions (30–90 °C) with minimum weight loss. Little weight loss at low temperatures is returned to evaporation of water, impurities, and volatile components66,67. The obtained TGA data with weight loss less than 2% for all prepared catalysts at temperatures less than 90 °C proved attractive stability for ODS technology. At 200–600 °C, the increase in weight losses can be assigned to the decomposition of oxygen functionalities from the surfaces of catalysts. Also, the weight loss in this region can be attributed to the degradation of hemicelluloses, lignin, and carbonaceous materials68. The high weight loss at temperatures more than 600 °C can be returned to the complete degradation of cellulose, hemicelluloses, lignin, and active salt species. Also, this high loss can be attributed the decomposition of the carbon phase at the elevated temperature (> 600 °C)67,69.
Catalytic ODS performance (non- dimensionless parameters)
Effect of temperature
The efficiency of the catalytic oxidative desulfurization process under different desulfurization temperatures (30, 60, and 90) °C is illustrated in Fig. 7a–d. At the best flow (Ren = 8.74) and intense mixing behavior (Reo = 1168), ultraclean heavy fuel is produced at short residence time (12 min) in the novel OBR by enhancing desulfurization temperature from 30 to 90 °C which leads to promising improve in oxidation rate from 77.3 to 99.94% as illustrated in Fig. 10d. The positive influence of temperature on the efficiency of ODS can assigned to the Arrhenius correlation as observed in Eq. 8, thereby increasing the oxidation temperature enhances the removal efficiency at a dramatic rate11,70,71.
Effect of desulfurization temperatures on sulfur removal efficiency using different types of catalyst in novel OBR at atmospheric pressure and: (a) Ren = 34.87 and Reo = 73, (b) Ren = 17.43 and Reo = 292, (c) Ren = 11.63 and Reo = 657, (d) Ren = 8.74 and Reo = 1168.
Increasing the desulfurization temperature improves Henry’s constant and diffusivity and lowers the density, viscosity, and surface tension of heavy oil, thereby improving transport rates (mass and heat) of reactants. As a result, this improving of mass and heat transport and physical properties satisfy dramatic enhance in the process efficiency72,73. As well as, rising the desulfurization temperature improves the mixing intensity of the reacting molecules and promotes spreading of sulfur and oxidant into the pores of catalyst, thereby ODS performance upgrades12,74. The interacting of the fluid oscillation motion and the novel baskets in the central baffle provides vortices which enhances the mixing pattern in the reactor cavities between each two baffles. Thereby, the breakage and coalescence of the reacting droplets, which lead to efficient droplet size distribution, and the generation of emulsion enhances via increasing the oxidation temperature, thus sulfur removal efficiency improves75.
Effect catalyst type
Figure 8 observes the effect of different types of catalysts on ODS performance. For example, sulfur removal efficiency is 88.1% using (5% MnO2/AC) catalyst at Ren = 8.74, T = 90 °C, and Reo = 1168 (Fig. 8d). Uploading MnO2 on activated carbon support remarkably improves the sulfur removal efficiency. This enhancement can be attributed to the fact that manganese oxide effectively activates hydrogen peroxide, causing the generation of reactive species that improve the desulfurization reaction rates76. Furthermore, the contact between MnO2 and the catalyst support promises synergistic impacts that further enhance the performance of the ODS process76.
Effect of catalyst type on removal efficiency in novel OBR at: (a) T = 30 °C and Reo = 73, (b) T = 60 °C and Reo = 1168, (c) T = 90 °C and Reo = 657, (d) the best operating conditions (T = 90 °C and Reo = 1168).
Decoration of (5% MnO2/AC) catalyst with different amounts of iron oxide remarkably upgrades the efficiency of desulfurization reactions. As observed in Fig. 8a, sulfur removal efficiency enhances from 47.4% using (5% MnO2/AC) catalyst to 59.1% via decorating the catalyst with 3% (MFAC3) of Fe2O3 at T = 30 °C, Reo = 73, and Ren = 8.74. This improvement can be assigned to the fact that Fe2O3 serves as an active catalytic site, facilitating the oxidation of sulfur77. As well as, spreading Mn and Fe oxides over a wide surface area of AC supplies a further active sites and enhanced sulfur adsorption capacity78,79. Also, manganese and iron metal oxides are modified porous structures of AC, leading to the formation of functional groups and improving catalyst reactivity80. The statistical tests of the desulfurization efficiency using different types of catalysts with two replicates of experiments (n = 2) showed the following results under best operational parameters (T = 90 °C, Reo = 1168, and Ren = 8.74): maximum desulfurization efficiency (efficiency ± SD) is (88.08 ± 0.1712) %, (92.32 ± 0.1551) %, (95.93 ± 0.1634) %, (99.63 ± 0.1607) % under 95% confidence interval (95% CI = 98.324% to 99.884%), (95% CI = 99.133% to 99.314%), (95% CI = 99.712% to 99.116%), (95% CI = 99.468% to 99.789%) for MFAC0, MFAC1, MFAC2, MFAC3, respectively.
Table 2 illustrates the significantly high desulfurization efficiency using the new synthesis poly-active magnetic-activated carbon catalysts compared to previous studies. This table also proves that the desulfurization time can be reduced dramatically in the novel design of OBR using the new catalysts. The fast and ultradeep desulfurization process can be assigned to the combination of the remarkable oxidation activity of the prepared catalyst and uniform mixing pattern formed by contact of oscillatory flow and the central baffle, thereby the catalyst adsorption capacity, the rate of mass transfer and the high desulfurization efficiency achieved at very short time compared to the other ODS processes and produced ultraclean heavy fuel. The fast and ultraclean desulfurization process in OBR confirms not only efficient removal of sulfur compounds but also improves the economic feasibility of the ODS technology81,82,83.
Effect of oscillatory flow
As presented in Fig. S3, the sulfur removal efficiency decreases from 99.64% in OBR to 42.06% in LFBR using MFAC3. Figure 9 shows that desulfurization efficiency is lowered from 99.63% using OBR to 84.45% and 42.06% using STR and LFBR, respectively. The dramatic decrease proves that the oscillatory motion is the critical and controlling factor in achieving ultradeep desulfurization through short residence time (12 min) in OBR. In LFBR, the mixing between the reacting species is produced only by the laminar flow (Ren < 35), which provides a soft mixing pattern with very weak and ineffective contact between the reactants, thereby satisfying low sulfur removal efficiency. Also, these behaviors can be assigned to the absence of vortices and shear engendered by the contacting of the fluid oscillatory and the baffle designs in LFBR, which results in lowering the coalescence and breakage of the reactant droplets and reducing the sulfur removal efficiency. In STR, non-constituent mixing due to the presence of dead zones under high local shear zones near the stirrer, leads to non-uniform mixing zones and low process efficiency. Also, STR requires high agitation speeds and energy consumption to achieve an efficient mixing pattern. As well as mass and heat transfer rates in STR are limited by poor macro-mixing or short-circuiting, thereby reducing desulfurization efficiency22,63,90. The combined impact of baffles and oscillatory motion in OBR acts to increase contacting area between the reactants and improve the process efficiency by producing vortices between each two neighboring baffles91,92. Also, the combination of the oscillatory flow and baffles enhances the coalescence and breakup processes for the reactant droplets93,94. Break-up is achieved as a result of shear produced at the baffles, which decreases the size of the liquid drops, enhances the transfer rates, and improves the desulfurization rates. As well as the residence time of the reactant droplets, radial mixing, and process efficiency increase by the produced vortices89,92,95.
Effect of reactor type on sulfur removal efficiency using MFAC3 catalyst at the optimal conditions (90 °C, Ren = 8.74, Reo = 1168).
Effect of the dimensionless group
Effect of the net Reynold number (Ren)
The flow pattern of reacting materials in OBR is controlled by the Reynolds number (Ren). The change in desulfurization performance with Ren (34.87, 17.43, 11.63, and 8.74), which corresponds to residence times (3, 6, 9, 12 min) is illustrated in Fig. 10a–d. The net Reynolds number is inversely proportional to time and directly proportional to the heavy oil flow rate (Eqs. 1–3). The results prove that the desulfurization efficiency improves with reducing flow rate (rising residence time) and decreasing Ren. At the best operating parameters (MFAC3 catalyst, T = 90 °C and Reo = 1168), a dramatic enhancement in sulfur oxidation from 84.4% to 99.64% is achieved with decreasing Ren from 34.87 to 8.74 (Fig. 10d). The numerical values of Ren (≤ 34.87) in the present study prove that the flow regime in the novel OBR is in laminar mode. As Ren decreases, the reactants’ residence time increases, thereby improving sulfur removal efficiency by enhancing the contact time between the reacting species82,96. Also, net forward flow increases via enhancing Ren, so the generated oscillatory motion is not sufficient to reverse fluid flow in the backward direction. Therefore, vortex generation only creates forward, and thus the ratio of radial to axial mixing declines, which leads to a decrease in the heat transfer rate63,97 and a lower desulfurization efficiency. The oscillatory baffled reactor is mainly designed to achieve efficient plug flow at a laminar flow regime and various oscillation intensities98,99. By generating and transporting a vortex under laminar flow media, controlled mixing is accomplished100. Therefore, the decrease in Ren enhances laminar flow behavior.
Effect of Ren on sulfur removal efficiency in novel OBR at: (a) T = 30 °C and Reo = 73, (b) T = 60 °C and Reo = 292, (c) T = 90 °C and Reo = 657, (d) the best operating conditions (T = 90 °C and Reo = 1168).
Effect of Strouhal number (St)
The impact of St (0.411, 0.206, 0.137, and 0.103), which corresponds to (3, 6, 9, and 12) mm of oscillatory amplitudes, on sulfur removal efficiency is observed in Fig. 11a–d. Desulfurization efficiency significantly decreases as St Increases. At T = 90 °C and Ren = 11.63, the results show that reducing St from 0.411 to 0.103 leads to an upgrades in sulfur removal from 67.4% to 83.9% using MFAC0 as shown in Fig. 11c. The positive effect of decreasing St can be returned to raising the oscillatory amplitude, which generates sufficient and uniform mixing of reactant molecules. Also, high St observes at small oscillatory amplitude, provides poor vortex formation, and lowers the mixing of reactants, thereby decreasing the desulfurization efficiency. As well as, kLa increases with lowering St82,91. This increase in kLa leads to improve spreading of the oxidant through the fuel and the generation of fine emulsion as a result of micro-mixing generated via oscillating. This emulsion increases the interfacial area that boosts oxidant attack on sulfur in fuel media and oxidizes it rapidly. Thus, desulfurization efficiency remarkably enhances101,102.
Effect of St on desulfurization efficiency in novel OBR at: (a) T = 30 °C and Ren = 17.43, (b) T = 60 °C and Ren = 34.87, (c) T = 90 °C and Ren = 11.63, (d) the best operating conditions (T = 90 °C and Ren = 8.74).
Effect of the oscillatory Reynolds number (Reo)
The experimental data for the present study prove that the increase in Reo significantly boosts the desulfurization efficiency, as illustrated in Fig. 12a–d. The desulfurization efficiency upgrades from 65.9% to 77.3% as Reo increases from 73 to 1168 using MFAC1 catalyst at T = 60 °C and Ren = 11.63 (Fig. 16b). The dramatic enhancement in sulfur removal efficiency can be returned to the fact that the mixing severity of reacting molecules enhances with increasing Reo, allowing desired control of desulfurization rate. Also, raising the oscillatory mixing (Reo) improves the overall desulfurization reaction rate by enhancing the contact probability between the reacting species and reducing the required time for effective reaction tendencies. Furthermore, rising Reo leads to more radial mixing by providing periodic vortices97. Also, the oscillatory conditions improve as radial and axial mixing enhance. Thus, plug flow conditions boost via rising Reo and improve desulfurization efficiency. The enhancement in oscillating flow improves kLa, thereby upgrading sulfur removal efficiency. The mixing mechanism inside OBR relates to the degree of agitation. Improving oscillation mixing increases the intensity of agitation103. The desulfurization rate controlled by contacting rates of reacting species, thereby enhancing Reo, improves the oxidation efficiency.
Effect of Reo on oxidation efficiency in novel OBR at: (a) T = 30 °C and Ren = 34.87, (b) T = 60 °C and Ren = 11.63, (c) T = 90 °C and Ren = 17.43, (d) the best operating conditions (T = 90 °C and Ren = 8.74).
Effect of the velocity ratio (Ψ)
The results of the effect Ψ on sulfur removal efficiency are illustrated in Fig. S4a–c. The results prove that desulfurization efficiency remarkably enhances with increasing Ψ due to an increase in the oscillatory Reynolds number with respect to the net flow Reynolds number. At the best oxidation temperature and using the most active catalyst (MFAC3), sulfur removal efficiency upgrades from 71.3 to 99.64% as Ψ (Reo / Ren) rises from 2.1 (73/34.87) to 133.6 (1168/8.74) with increasing Reo and decreasing Ren (Fig. S4c). This increase can be attributed to enhancing the oscillation flow condition (Reo) and lowering the effect of net flow conditions (Ren), thereby generating uniform and sufficient missing conditions. The increase in Ψ enhances the desirable degree of plug flow, which leads to dramatic upgrades in the desulfurization efficiency.
Stability and deactivation of poly-active magnetic-activated carbon catalysts
Stability and deactivation rate of catalysts (MFAC0–MFAC3) are examined across five consecutive oxidative desulfurization cycles under optimum process parameters (T = 90 °C, Ren = 8.74, Reo = 1168, St = 0.103, and Ψ = 133.6), as shown in Fig. 13. Results exhibit a slight reduction in desulfurization efficiency for all catalysts after five desulfurization cycles. Therefore, different poly-active magnetic-activated carbon catalysts exhibit high stability and low deactivation rate. Therefore, the reduction percent in desulfurization efficiency was 1.51%, 1.28%, 1.04%, and 0.62% for MFAC0, MFAC1, MFAC2, and MFAC3, respectively. These results indicate that there is no significant leaching of active metals (Mn and Fe oxides) into solution to maintain high desulfurization performance through five oxidation cycles. The slight reduction in desulfurization performance after five cycles can be attributed to the poisoning and loss of some catalytically active sites across the consecutive oxidation cycles. Also, this decrease in process efficacy can be assigned to the deposition of non-oxidized or oxidized sulfur species in pores as desulfurization time proceeds104. As well as, catalyst stability is significantly improved with increasing the amounts of iron oxides. These findings can be returned to the presence of more available catalytic active sites with increasing the amounts of spreading active oxides over catalyst carrier, thus deactivation impact is remarkablly reduced104,105.
Stability and deactivation rate of poly-active magnetic-activated carbon catalysts.
Blank experiments
Blank experiments without catalyst or oxidant and with oxidant/activated carbon (O/AC) at the optimal operating conditions (T = 90 °C, Ren = 8.74, Reo = 1168) were achieved in novel OBR and compared to the maximum desulfurization efficiency using the best catalyst in the presence of oxidant (O/MFAC3). As illustrated in Fig. 14, there is almost low desulfurization efficiency, proving that both catalyst and oxidant are indispensable. Sulfur oxidation efficiency decreases from 99.96% in the presence of all reacting species (sulfur compounds, oxidant, and catalyst) to 40.96% without (5% MnO2- 3% Fe2O3/AC) catalyst and to 45.14% without oxidant (H2O2). Therefore, the integrated impact of catalyst and oxidant is an essential factor to achieve high desulfurization efficiency in OBR. Also, activated carbon exhibits low desulfurization efficiency compared to the best catalyst (MFAC3); thus sulfur oxidation efficiency is lowered from 99.6% for MFAC3 to only 56.34% using AC in the presence of hydrogen peroxide. These findings indicat the dramatic importance of magnetic iron and manganese oxides in providing active sites over activated carbon, which are enhances the oxidation reactivity of the catalyst and accelerates the desulfurization rate106,107.
Oxidative desulfurization reactions without oxidant (O), without catalyst or catalytic active oxides.
Desulfurization kinetics
Kinetic model for oxidative desulfurization of heavy oil in oscillatory baffled reactors using the best catalyst (MFAC3) is determined by fitting the experimental data with a pseudo-first order model across the integral method. Kinetic parameters are examined under the best process parameters (Reo of 1168 and Ren of 8.74). Desulfurization efficiency is experimentally evaluated as a function of different desulfurization periods (1.5, 3, 4.5, 6, 7.5, 9, 10.5, and 12 min) under reaction temperatures (30, 60, and 90 °C), as presented in Fig. S5, to extract process kinetics. The kinetic rate for the sulfur oxidation reaction can be presented as follows:
For pseudo-first order model, the kinetic rate can be presented as follow:
Then, the integration of Eq. 10 is formed the following formula:
where, Rs: sulfur oxidation rate (mol/l. min), k: sulfur oxidation rate constant (min−1), Cso: initial sulfur level (mol/l), Cs: final sulfur level (mol/l), t: reaction period (min), and n: oxidation reaction order.
By plotting \(\left( {\ln \frac{{C_{so} }}{{C_{s} }}} \right)\) against (t) based on Eq. 12 at various reaction temperatures (30, 60, and 90 °C) and evaluating regression by coefficient of determination (R2) across linear fitting analysis strategy, k is then evaluated under each reaction temperature. The apparent activation energy can be determined from the slope of the Arrhenius plot, which is plotted based on Arrhenius equation (Eq. 8) as follow:
where, T: reaction temperature (°K), R: gas constant (kJ/mole. K), E: apparent activation energy (kJ/mol), and ko: pre-exponential factor.
By drawing of (− ln k) vs. (1/T) and using linear fitting analysis strategy across fitting the obtained results to a straight line, E is estimated under maximum R2.
Figure 15 exhibits the rate constant (k) of the sulfur oxidation reaction by fitting the obtained data to a straight line. The results show that the sulfur oxidation reaction is followed a pseudo-first order model under various temperatures, with a high R2. The pseudo-first order model is widely proven for oxidative desulfurization reactions in the literature82,104.
Linear fitting of pseudo-first order model at different temperatures (30, 60, and 90) °C.
Then, activation energy can be estimated by plotting the obtained rate constants at different temperatures based on Eq. 13, and fitting these data into a straight line, as presented in Fig. 16 to evaluate the kinetic parameters. The results exhibit that the oxidative desulfurization reaction has activation energy of 21.66 kJ/mol using the MFAC3 catalyst in the oscillating reactor. This low activation energy allows the sulfur oxidation reaction to be quickly satisfied at lower energy requirements owing to the strong reactivity of poly-active magnetic-activated carbon catalyst in accelerating desulfurization rates and reducing activation energy. Also, catalytic materials improve the electron density of sulfur species and boost the electrophilic addition of oxygen on these compounds, thus lowering the activation energy108,109. This value of activation energy is referred to the microstructural evolution due to efficient dispersion of manganese and iron oxides and the creation of crystalline Mn–Fe oxides. Furthermore, the activation energy of the MFAC3 catalyst (21.66 kJ/mol) proves the presence of mass-transfer impacts or diffusion limitations due to agglomeration of iron oxide nanoparticles, reducing dispersion, and blocking and losing some catalytic pore110,111,112,113,114
Arrhenius plot for sulfur oxidation reactions using MFAC3 catalyst in oscillating reactor.
Mechanism of desulfurization in OBCBR
The proposed catalytic oxidative desulfurization mechanism using (5% MnO2- X% Fe2O3/AC, X = 0–3) catalyst is speculative based on literature as, illustrated in Fig. 17. Firstly, both the catalyst and oxidant are introduced into the fuel, thereby, sulfur and oxidant species reach the active sites, which are attached to Fe and Mn oxides, in the pores of the catalyst, where desulfurization reactions are conducted. Catalyst with large surface area and microstructure ensured supersizing diffusion of sulfur compounds and oxidant, causing full interaction of the catalyst’s active sites with reactants. Thus, the elevated electron density of Fe and Mn and oxygen vacancies caused chemical adsorption of the hydrogen peroxide by coordinating the peroxy O–O bonds with Fe and Mn atoms, and enhances the breaking of O–O bonds, providing a large number of electrophilic active species ⋅OH115,116,117. Hydrogen peroxide decomposes into oxygen (O.), which shares in catalytic ODS, and (H2O2 or HO-OH) as a green byproduct (Eq. 14). Also, Hydrogen peroxide decomposes into ⋅OH species and contributes to the desulfurization reaction. Iron oxide acts as a Fenton-type redox catalyst to activate hydrogen peroxide (Eqs. 15 and 16). This mechanism provides hydroxyl (⋅OH) and hydroperoxyl (HO₂⋅) radicals, which are the essential oxidizing species that attack sulfur components and improve the desulfurization rate118. Manganese oxide creates surface lattice oxygen and participates in redox cycling (Mn4⁺/Mn3⁺) as presented in Eqs. 17 and 18. Manganese oxide can decompose hydrogen peroxide to provide oxygen and reactive intermediates (Eq. 19). The Mn4⁺/Mn3⁺ improves oxygen transfer and radical creation, thus catalytic oxidation enhances119. The synergistic impact of Fe3⁺/Fe2⁺ and Mn4⁺/Mn3⁺ provides a couple of interactions across electron transfer, boosting the regeneration of Fe2⁺ and maintaining continuous radical generation. This prevents iron deactivation and improves sustained peroxide activation. Also, the integrated effect of Fe–Mn facilitates heterolytic cleavage of peroxide, providing ⋅OH and ⋅O₂⁻ radicals more efficiently than either oxide alone, thereby the desulfurization rate remarkably rises120,121. Finally, sulfur compounds (such as DBT) are oxidized to sulfoxide (DBTO, Eq. 20) and rapidly oxidized to sulfones (DBTO2, Eq. 21) by ⋅OH or ⋅O species in the oxidation stage (Fig. 15)122,123,124. In the separation stage, the produced reacting mixture is pushed into separator to the separate into fuel and the aqueous phase. Finally, highly polar sulfoxide/ sulfone transfer from oil into aqueous phase82,89.
The proposed possible desulfurization reaction mechanism over novel MFACX catalyst in novel OBR.
Conclusion
In conclusion, continuous catalytic oxidative desulfurization using new synthesis heterogeneous composite catalysts (5% MnO2- X% Fe2O3/AC, X = 0–3) with wet hydrogen peroxide oxidant and novel design of oscillatory baffled reactor (OBR) with baskets central baffle is successfully used for upgrading of a highly sour heavy oil. The ODS process is examined at various catalyst types, desulfurization temperatures, net flow Reynolds’ numbers (Ren), Strouhal numbers (St), oscillatory Reynold’s numbers (Reo), and velocity ratios Ψ. Results showed that oxidation efficiency is remarkably boosted by enhancing all the operational parameters except St and Ren, which had a negative effect. Ultraclean heavy oil is produced by lowering the sulfur level from 8281 to 30 ppm using the most active synthesis catalyst (MFAC3) during a very short oxidation time (12 min) under the optimal experimental conditions (temperature of 90 °C, St of 0.103, Ren of 8.74, Reo of 1168, and Ψ of 133.6). The sulfur removal efficiency using MFAC3 decreased from 99.64% in OBR to 42.06% in the absence of the oscillatory motion through a laminar flow baffled reactor (LFBR) under the best conditions. Also, blank experiments without a catalyst or oxidant are achieved in the novel OBR under the optimal parameters. The desulfurization efficiency is lowered from 99.96% to 40.96% No catalyst and 45.14% No oxidant. The results prove that there is almost a very low oxidation rate, proving that oscillatory mixing, catalyst, and oxidant are indispensable. The present study is satisfied with an ultradeep, fast, and cost–effective ODS process that meets the global environmental standards in the production of eco-friendly and ultraclean fuel under mild conditions compared to severe traditional HDS technology. The new integrated oscillating- MFAC3 system can be directly scaled up into the petroleum industry at similar process performance based on OBR dimensionless groups.
Data availability
All data generated or analyzed during this study are included in this article.
References
Nejati, F. M., Shahhosseini, S. & Rezaee, M. Cobalt-based sandwich-type polyoxometalate supported on amino-silane decorated magnetic graphene oxide: A recoverable catalyst for extractive-catalytic oxidative desulfurization of model oil. J. Environ. Chem. Eng. 10(3), 107949 (2022).
Yu, X., Han, P. & Li, Y. Oxidative desulfurization of dibenzothiophene catalyzed by α-MnO2 nanosheets on palygorskite using hydrogen peroxide as oxidant. RSC Adv. 8(32), 17938–17943 (2018).
Jafari, Z., Mazloom, G. & Akbari, A. (Zn/Co)-ZIFs@ TiO2 composite catalysts for oxidative desulfurization: Impacts of Zn2+/Co2+ on TiO2 interactions. J. Environ. Chem. Eng. 12(1), 111874 (2024).
Guo, R., Cao, Z. & Fang, X. The development of catalysts and their stacking technology for diesel ultra-deep hydrosulfurization. Catal. Today 316, 21–25 (2018).
Kulkarni, P. S. & Afonso, C. A. Deep desulfurization of diesel fuel using ionic liquids: current status and future challenges. Green Chem. 12(7), 1139–1149 (2010).
Zhang, S., Wang, X., Li, Q. & Yang, J. Oxidative desulfurization of dibenzothiophene over V-Mo co-doped akaganeite. J. Environ. Chem. Eng. 12(6), 114267 (2024).
Jabaar, G. K., Al-Jendeel, H. A. & Alsheikh, Y. A. Desulphurization of simulated oil using SAPO-11 with CNT’s as adsorbent: A kinetic study. Iraqi J. Chem. Petrol. Eng. 24(3), 69–77 (2023).
Jin, W. et al. Ultra-deep oxidative desulfurization of fuel with H2O2 catalyzed by molybdenum oxide supported on alumina modified by Ca 2+. RSC Adv. 7(76), 48208–48213 (2017).
Humadi, J. I., Shihab, M. A., Hasan, A. A. & Mohammed, A. Experimental and ANN modeling of kerosene fuel desulfurization using a manganese oxide-tin oxide catalyst. Chem. Eng. Res. Des. 211, 160–167 (2024).
Dizaji, A. K., Mokhtarani, B. & Mortaheb, H. R. Deep and fast oxidative desulfurization of fuels using graphene oxide-based phosphotungstic acid catalysts. Fuel 236, 717–729 (2019).
Jima, B. B. & Majeed, N. S. Oxidation desulphurization of heavy naphtha improved by ultrasound waves. Iraqi J. Chem. Petrol. Eng. 21(1), 9–14 (2020).
Jarullah, A. T. et al. Production of green fuel using a new synthetic magnetite mesoporous nano-silica composite catalyst for oxidative desulfurization: Experiments and process modeling. Catalysts 14(8), 529 (2024).
Jarullah, A. T. et al. New composite mesoporous nano-catalysts for clean fuel produced by the oxidative desulfurization process. Pet. Chem. 64(4), 458–470 (2024).
Ishihara, A. et al. Oxidative desulfurization and denitrogenation of a light gas oil using an oxidation/adsorption continuous flow process. Appl. Catal. A 279(1–2), 279–287 (2005).
Mohammed, H. R. et al. Synthesis, evaluation, and optimal stability of a biowaste-based catalytic oxidative desulfurization of model fuel in a trickle bed reactor. Process Saf. Environ. Prot. 163, 513–527 (2022).
Mohammed, A. E. et al. Agricultural waste-based microporous catalysts for oxidative desulfurization of highly sour heavy gas oil. Diam. Relat. Mater. 142, 110723 (2024).
Gheni, S. A., Jarullah, A. T. & Razak, G. H. A. Oxidative catalytic desulphurization of naphtha in a trickle bed reactor. Petrol. Coal 59(2) (2017).
Zhao, M., Han, P. & Lu, X. Ultrasound assisted photocatalytic oxidative desulfurization of model diesel fuel. Pet. Sci. Technol. 36(1), 29–33 (2018).
Salman, M. S. et al. Development of an electrochemical reactor with rotating anode for fast and ultra-deep catalytic desulfurization of diesel: experimental and modeling. Chem. Eng. Commun. 211(10), 1508–1523 (2024).
Mohammed, A. E., Mohammed, W. T. & Gheni, S. A. Scale-up of oxidative desulfurization for sour diesel fuel: Modeling, simulation, and reactor design using Fe/AC catalyst. Case Stud. Chem. Environ. Eng. 11, 101024 (2025).
Mohammed, A. E., Mohammed, W. T. & Gheni, S. A. Environmental benefits of agricultural waste-derived catalysts in diesel desulfurization: A review. Clean. Mater. 13, 100262 (2024).
Humadi, J. I. & Mohammed, W. T. A Comprehensive Review on Developing of the Utilized Reactor Design for Oxidative Desulfurization Technology: Oscillatory Baffled Reactor (Johnson Matthey Technology Review, 2025).
Nazmi, N. A. S. M. et al. Catalytic oxidative desulfurisation over Co/Fe-γAl2O3 catalyst: Performance, characterisation and computational study. Environ. Sci. Pollut. Res. 29, 1009–1020 (2022).
Trisunaryanti, W. et al. ZnO-activated carbon blended as a catalyst for oxidative desulfurization of dibenzothiophene. Bull. Chem. React. Eng. Catal. 16(4), 881–887 (2021).
Phan, A. N. & Harvey, A. Development and evaluation of novel designs of continuous mesoscale oscillatory baffled reactors. Chem. Eng. J. 159(1–3), 212–219 (2010).
Rasdi, F. R. M., Phan, A. N. & Harvey, A. P. Rapid determination of reaction order and rate constants of an imine synthesis reaction using a mesoscale oscillatory baffled reactor. Chem. Eng. J. 222, 282–291 (2013).
Ni, X. et al. Mixing through oscillations and pulsations—A guide to achieving process enhancements in the chemical and process industries. Chem. Eng. Res. Des. 81(3), 373–383 (2003).
Harvey, A., Mackley, M. & Stonestreet, P. Operation and optimization of an oscillatory flow continuous reactor. Ind. Eng. Chem. Res. 40(23), 5371–5377 (2001).
Ni, X., Brogan, G., Struthers, A., Bennett, D. & Wilson, S. A systematic study of the effect of geometrical parameters on mixing time in oscillatory baffled columns. Chem. Eng. Res. Des. 76(5), 635–642 (1998).
Ni, X. & Gough, P. On the discussion of the dimensionless groups governing oscillatory flow in a baffled tube. Chem. Eng. Sci. 52(18), 3209–3212 (1997).
Brunold, C., Hunns, J., Mackley, M. & Thompson, J. Experimental observations on flow patterns and energy losses for oscillatory flow in ducts containing sharp edges. Chem. Eng. Sci. 44(5), 1227–1244 (1989).
Dickens, A., Mackley, M. & Williams, H. Experimental residence time distribution measurements for unsteady flow in baffled tubes. Chem. Eng. Sci. 44(7), 1471–1479 (1989).
Mackley, M. & Stonestreet, P. Heat transfer and associated energy dissipation for oscillatory flow in baffled tubes. Chem. Eng. Sci. 50(14), 2211–2224 (1995).
Ni, X., Gao, S., Cumming, R. & Pritchard, D. A comparative study of mass transfer in yeast for a batch pulsed baffled bioreactor and a stirred tank fermenter. Chem. Eng. Sci. 50(13), 2127–2136 (1995).
Smith, K. B. Scale-up of Oscillatory Flow Mixing (University of Cambridge, 2000).
Abbott, M., Harvey, A., Perez, G. V. & Theodorou, M. Biological processing in oscillatory baffled reactors: operation, advantages and potential. Interface Focus 3(1), 20120036 (2013).
Kunusa, W., Iyabu, H. & Abdullah. R. FTIR, SEM and XRD analysis of activated carbon from sago wastes using acid modification. In Journal of Physics: Conference Series. IOP Publishing. (2021).
Togibasa, O., Ansanay, Y. O., Dahlan, K. & Erari, M. Identification of surface functional group on activated carbon from waste sago. J. Phys. Theor. Appl. 5(1), 1–8 (2021).
Kim, J.-H. et al. Impact of the oxygen functional group of nitric acid-treated activated carbon on KOH activation reaction. Carbon Letters 29, 281–287 (2019).
Williams, N. E., Oba, O. A. & Aydinlik, N. P. Modification, production, and methods of KOH-activated carbon. ChemBioEng Reviews 9(2), 164–189 (2022).
Ali, R., Aslam, Z., Shawabkeh, R. A., Asghar, A. & Hussein, I. A. BET, FTIR, and RAMAN characterizations of activated carbon from waste oil fly ash. Turk. J. Chem. 44(2), 279–295 (2020).
Wang, L. et al. MnO2-loaded activated carbon and its adsorption of formaldehyde. BioResources 14(3), 7193–7212 (2019).
Javanbakht, M., Mahdi, F. & Shahrokhian, S. Facile In-Situ Electrosynthesis of Mno2/Rgo Nanocomposite for High Power and Energy Supercapacitors. Available at SSRN 4335419.
Riyanti, F. et al. The synthesis of MnFe2O4-activated carbon composite for removal of methyl red from aqueous solution. Molekul 13(2), 123–132 (2018).
Pego, M., Carvalho, J. & Guedes, D. Surface modifications of activated carbon and its impact on application. Surf. Rev. Lett. 26(01), 1830006 (2019).
Vasu, A.E. & Archana, A. M. S. Hydrogen peroxide treated iron oxide impregnated carbon materials for improved adsorption and photocatalytic degradation of cationic dyes. Rasayan J. Chem. 15(1) (2022).
Vaughan, R. L. Jr., Yang, J., LeMire, L. E. & Reed, B. E. Characterization and surface acidity modelling of an iron oxide-impregnated activated carbon. Adsorpt. Sci. Technol. 25(5), 295–310 (2007).
An, C. et al. Growth of graphite spheres in liquid nickel. Carbon 49(12), 3953–3957 (2011).
Chen, Z. et al. AC impedance investigation and charge–discharge performance of NaOH surface-modified natural graphite. Electrochim. Acta 102, 44–50 (2013).
Wan, X. et al. Facile synthesis of MnO2 nanoflowers/N-doped reduced graphene oxide composite and its application for simultaneous determination of dopamine and uric acid. Nanomaterials 9(6), 847 (2019).
Li, M. et al. Relationship between Surface Hydroxyl Complexation and Equi-Acidity Point pH of MnO2 and Its Adsorption for Co2+ and Ni2+. ACS Omega 7(11), 9602–9613 (2022).
Gu, H. et al. Suppressing Jahn-Teller distortion of MnO2 via B-Ni dual single-atoms integration for methane catalytic combustion. Nat. Commun. 16(1), 1008 (2025).
Chen, S. et al. Preparation and characterization of monodispersed spherical Fe2O3@ SiO2 reddish pigments with core–shell structure. J. Adv. Ceramics 8, 39–46 (2019).
Benedet, M., Barreca, D., Rizzi, G. A., Maccato, C., Wree, J. -L., Devi, A. & Gasparotto, A. Fe2O3-graphitic carbon nitride nanocomposites analyzed by XPS. Surf. Sci. Spectra. 30(2) (2023).
Meez, E., Tolkou, A. K., Giannakoudakis, D. A., Katsoyiannis, I. A. & Kyzas, G. Z. Activated carbons for arsenic removal from natural waters and wastewaters: A Review. Water 13(21), 2982 (2021).
Khelifi, A. et al. Influence of nitric acid concentration on the characteristics of active carbons obtained from a mineral coal. Fuel Process. Technol. 91(10), 1338–1344 (2010).
Yin, C. Y., Aroua, M. K. & Daud, W. M. A. W. Review of modifications of activated carbon for enhancing contaminant uptakes from aqueous solutions. Sep. Purif. Technol. 52(3), 403–415 (2007).
Pereira, L. et al. Thermal modification of activated carbon surface chemistry improves its capacity as redox mediator for azo dye reduction. J. Hazard. Mater. 183(1–3), 931–939 (2010).
Daud, W. M. A. W. & Ali, W. S. W. Comparison on pore development of activated carbon produced from palm shell and coconut shell. Biores. Technol. 93(1), 63–69 (2004).
Molina-Sabio, M. & Rodrıguez-Reinoso, F. Role of chemical activation in the development of carbon porosity. Colloids Surf., A 241(1–3), 15–25 (2004).
Chung, H.-Y., Chang, H.-M. & Wang, C.-P. Manganese Oxide-Doped Hierarchical Porous Carbon Derived from Tea Leaf Waste for High-Performance Supercapacitors. Int. J. Mol. Sci. 25(20), 10884 (2024).
Wan, M.-W. & Yen, T.-F. Enhance efficiency of tetraoctylammonium fluoride applied to ultrasound-assisted oxidative desulfurization (UAOD) process. Appl. Catal. A 319, 237–245 (2007).
Hmood, H. M. et al. Kaoline-based catalyst for a high stability desulfurization of sour heavy naphtha in a three-phase oscillatory baffled reactor. Particuology 84, 249–260 (2024).
Haw, K.-G., Bakar, W. A. W. A., Ali, R., Chong, J.-F. & Kadir, A. A. A. Catalytic oxidative desulfurization of diesel utilizing hydrogen peroxide and functionalized-activated carbon in a biphasic diesel–acetonitrile system. Fuel Process. Technol. 91(9), 1105–1112 (2010).
Kampouraki, Z. C., Giannakoudakis, D. A., Triantafyllidis, K. S. & Deliyanni, E. A. Catalytic oxidative desulfurization of a 4, 6-DMDBT containing model fuel by metal-free activated carbons: The key role of surface chemistry. Green Chem. 21(24), 6685–6698 (2019).
Jaouadi, M. & Hamzaoui, A. H. Boron adsorption onto activated carbon and amorphous carbon prepared from sucrose dehydration. Desalin. Water Treat. 149, 150–156 (2019).
Nyamful, A. et al. Processing and characterization of activated carbon from coconut shell and palm kernel shell waste by H3PO4 activation. Ghana J. Sci. 61(2), 91–104 (2020).
Sharma, S., Kaur, M., Sharma, C., Choudhary, A. & Paul, S. Biomass-derived activated carbon-supported copper catalyst: An efficient heterogeneous magnetic catalyst for base-free chan–lam coupling and oxidations. ACS Omega 6(30), 19529–19545 (2021).
Yacob, A. R., Majid, Z. A., Dewi Dasril, R. S., & Inderan, V. Comparison of various sources of high surface area carbon prepared by different types of activation. Malays. J. Anal. Sci. 12 (2008).
Dionigi, C. et al. Fabrication and properties of non-isolating γ-alumina meso-foam. J. Alloy. Compd. 666, 101–107 (2016).
Mahmood, Q. A., Abdulmajeed, B. A. & Haldhar, R. Oxidative desulfurization of simulated diesel fuel by synthesized tin oxide nano-catalysts support on reduced graphene oxide. Iraqi J. Chem. Petrol. Eng. 24(4), 83–90 (2023).
Jiang, Z., Hongying, L., Zhang, Y. & Can, L. Oxidative desulfurization of fuel oils. Chin. J. Catal. 32(5), 707–715 (2011).
Ahmed, G. S., Humadi, J. I. & Aabid, A. A. Mathematical model, simulation and scale up of batch reactor used in oxidative desulfurization of kerosene. Iraqi J. Chem. Petrol. Eng. 22(3), 11–17 (2021).
Saleh, T. A., Sulaiman, K. O., Al-Hammadi, S. A., Dafalla, H. & Danmaliki, G. I. Adsorptive desulfurization of thiophene, benzothiophene and dibenzothiophene over activated carbon manganese oxide nanocomposite: with column system evaluation. J. Clean. Prod. 154, 401–412 (2017).
Phan, A. N., Harvey, A. P. & Rawcliffe, M. Continuous screening of base-catalysed biodiesel production using new designs of mesoscale oscillatory baffled reactors. Fuel Process. Technol. 92(8), 1560–1567 (2011).
Ahmad, W. et al. Oxidative desulfurization of petroleum distillate fractions using manganese dioxide supported on magnetic reduced graphene oxide as catalyst. Nanomaterials 11(1), 203 (2021).
Kargar, H., Ghahramaninezhad, M., Shahrak, M. N. & Balula, S. S. An effective magnetic catalyst for oxidative desulfurization of model and real fuels: Fe3O4/ZIF-8/TiO₂. Microporous Mesoporous Mater. 317, 110992 (2021).
Zhu, W., Liu, X., Yang, Z. & Li, H. Synthesis of manganese-iron oxides/activated carbon as a highly effective adsorbent for sulfamerazine pollutant removal. Korean J. Chem. Eng. 39(11), 3083–3091 (2022).
Humadi, J. I. & Mohammed, W. T. Fast, ultradeep, and continuous desulfurization of heavy gasoil in novel oscillatory basket central baffled reactor using MnO2-incorparted Fe2O3-supported activated carbon catalyst. Fuel 400, 135716 (2025).
Zhang, G. et al. Comparison on surface properties and desulfurization of MnO2 and pyrolusite blended activated carbon by steam activation. J. Air Waste Manag. Assoc. 68(9), 958–968 (2018).
Kefas, H. M., Yunus, R., Rashid, U. & Taufiq-Yap, Y. H. Enhanced biodiesel synthesis from palm fatty acid distillate and modified sulfonated glucose catalyst via an oscillation flow reactor system. J. Environ. Chem. Eng. 7(2), 102993 (2019).
Humadi, J. I., Gheni, S. A., Ahmed, S. M. & Harvey, A. Dimensionless evaluation and kinetics of rapid and ultradeep desulfurization of diesel fuel in an oscillatory baffled reactor. RSC Adv. 12(23), 14385–14396 (2022).
Syam, A. M., Yunus, R., Ghazi, T. I. & Choong, T. S. Synthesis of Jatropha curcas oil-based biodiesel in a pulsed loop reactor. Ind. Crops Prod. 37(1), 514–519 (2012).
Nawaf, A. T., Gheni, S. A., Jarullah, A. T. & Mujtaba, I. M. Optimal design of a trickle bed reactor for light fuel oxidative desulfurization based on experiments and modeling. Energy Fuels 29(5), 3366–3376 (2015).
Humadi, J. I. et al. Design of new nano-catalysts and digital basket reactor for oxidative desulfurization of fuel: Experiments and modelling. Chem. Eng. Res. Des. 190, 634–650 (2023).
Ugal, J. R., Jima’a, R. B., Al-Jubori, W. M. K., Bayader, F. A. & Al-Jubori, N. M. Oxidative desulfurization of hydrotreated gas oil using Fe2O3 and Pd loaded over activated carbon as catalysts. Orient. J. Chem. 34(2), 1091 (2018).
Alwan, H. H., Ali, A. A. & Makki, H. F. Optimization of oxidative desulfurization reaction with Fe2O3 catalyst supported on graphene using box-behnken experimental method. Bull. Chem. React. Eng. Catal. 15(1), 175–185 (2020).
Nawaf, A. & Abdulmajeed, B. A synthesis of a Fe2O3-supported composite for rapid oxidative desulfurization production of environmentally friendly fuel in an OBR. Int. J. Environ. Sci. Technol. 22(6), 4923–4950 (2025).
Humadi, J. I. et al. Fast, non-extractive, and ultradeep desulfurization of diesel in an oscillatory baffled reactor. Process. Saf. Environ. Prot. 152, 178–187 (2021).
Mortazavi, H. & Pakzad, L. The hydrodynamics and mixing performance in a moving baffle oscillatory baffled reactor through computational fluid dynamics (CFD). Processes 8(10), 1236 (2020).
Hewgill, M., Mackley, M., Pandit, A. & Pannu, S. Enhancement of gas-liquid mass transfer using oscillatory flow in a baffled tube. Chem. Eng. Sci. 48(4), 799–809 (1993).
Ahmed, S. M., Phan, A. N. & Harvey, A. P. Mass transfer enhancement as a function of oscillatory baffled reactor design. Chem. Eng. Process. Process Intensif. 130, 229–239 (2018).
Oliveira, M. & Ni, X. Gas hold-up and bubble diameters in a gassed oscillatory baffled column. Chem. Eng. Sci. 56(21–22), 6143–6148 (2001).
Reis, N., Pereira, R. N., Vicente, A. A. & Teixeira, J. A. Enhanced gas−liquid mass transfer of an oscillatory constricted-tubular reactor. Ind. Eng. Chem. Res. 47(19), 7190–7201 (2008).
Pereira, F. M., Sousa, D. Z., Alves, M. M., Mackley, M. R. & Reis, N. M. CO2 dissolution and design aspects of a multiorifice oscillatory baffled column. Ind. Eng. Chem. Res. 53(44), 17303–17316 (2014).
Oday, Y., Aljendeel, H. & Shareef, Z. N. Preparation and characterization of adsorptive carbonized corncob for elimination of sulfur from sulfurized n-hexane. Iraqi J. Chem. Petrol. Eng. 26(2), 47–56 (2025).
Ahmed, S. M., Law, R., Phan, A. N. & Harvey, A. P. Thermal performance of meso-scale oscillatory baffled reactors. Chem. Eng. Process. Process Intensif. 132, 25–33 (2018).
McDonough, J. R. Process Development Using Oscillatory Baffled Mesoreactors (Newcastle University, 2018).
Humadi J. I. & Mohammed, W. T. Experimental and artificial intelligence-machine learning modeling of ultra-deep diesel desulfurization in oscillatory central bed baffled reactor using protected and unprotected dual active carbonaceous catalyst. Sustain. Chem. Clim. Action 100111 (2025).
Reis, N. et al. Application of a novel oscillatory flow micro-bioreactor to the production of γ-decalactone in a two immiscible liquid phase medium. Biotech. Lett. 28, 485–490 (2006).
Bhasarkar, J. B., Chakma, S. & Moholkar, V. S. Mechanistic features of oxidative desulfurization using sono-Fenton–peracetic acid (ultrasound/Fe2+–CH3COOH–H2O2) system. Ind. Eng. Chem. Res. 52(26), 9038–9047 (2013).
Humadi, J. I. & Mohammed, W. T. High stability diesel desulfurization for production eco-friendly fuel in new oscillatory central baffled reactor using alumina-(MnO2-Fe2O3/MAC) coated catalyst. Energy Environ. 0958305X251375943 (2025).
Ni, X. & Gao, S. Mass transfer characteristics of a pilot pulsed baffled reactor. J. Chem. Technol. Biotechnol. Int. Res. Process Environ. Clean Technol. 65(1), 65–71 (1996).
Nawaf, A. & Abdul Majeed, B. Kinetics study of oxidative desulfurization of real diesel fuel over uncoated and coated nano-catalysts in an oscillatory helical baffled reactor. J. Chem. Petrol. Eng. 58(2), 359–374 (2024).
Humadi, J. I. et al. Eco-friendly oxidative-adsorptive desulfurization for real diesel fuel using green MnO2 biowaste-extracted calcite in digital basket reactor. Processes 13, 3084 (2025).
Guo, J.-X. et al. Effects of preparation conditions on Mn-based activated carbon catalysts for desulfurization. New J. Chem. 39(8), 5997–6015 (2015).
Arcibar-Orozco, J. A., Acosta-Herrera, A. A. & Rangel-Mendez, J. R. Simultaneous desulfuration and denitrogenation of model diesel fuel by Fe-Mn microwave modified activated carbon: Iron crystalline habit influence on adsorption capacity. J. Clean. Prod. 218, 69–82 (2019).
Ahmad, W., Ahmad, I. & Yaseen, M. Desulfurization of liquid fuels by air assisted peracid oxidation system in the presence of Fe-ZSM-5 catalyst. Korean J. Chem. Eng. 33(9), 2530–2537 (2016).
Sachdeva, T. & Pant, K. Deep desulfurization of diesel via peroxide oxidation using phosphotungstic acid as phase transfer catalyst. Fuel Process. Technol. 91(9), 1133–1138 (2010).
Liu, Y.-Y. et al. Catalytic oxidative desulfurization of model and real diesel over a molybdenum anchored metal-organic framework. Microporous Mesoporous Mater. 277, 245–252 (2019).
Xue, Y., Sun, S., Wang, Q., Dong, Z. & Liu, Z. Transition metal oxide-based oxygen reduction reaction electrocatalysts for energy conversion systems with aqueous electrolytes. J. Mater. Chem. A 6(23), 10595–10626 (2018).
Long, J. W., Logan, M. S., Carpenter, E. E. & Rolison, D. R. Synthesis and characterization of Mn–FeOx aerogels with magnetic properties. J. Non Cryst. Solids 350, 182–188 (2004).
Al-Naimi, N. et al. Assessing the use of Ziziphus spina-christi as a sustainable solution for biomonitoring of urban air quality: A case study from Qatar. Sustainability 15(18), 13590 (2023).
Tepluchin, M. et al. Study on the hydrothermal and SO2 stability of Al2O3-supported manganese and iron oxide catalysts for lean CO oxidation. Catal. Today 258, 498–506 (2015).
Guo, J. et al. Preparation of porous hollow spherical MoOX/C catalyst for efficient extraction and oxidative desulfurization. Chem. Eng. J. 474, 145853 (2023).
Zou, J., Lin, Y., Wu, S., Zhong, Y. & Yang, C. Molybdenum dioxide nanoparticles anchored on nitrogen-doped carbon nanotubes as oxidative desulfurization catalysts: Role of electron transfer in activity and reusability. Adv. Func. Mater. 31(22), 2100442 (2021).
He, C. et al. Facet engineered α-MnO2 for efficient catalytic ozonation of odor CH3SH: oxygen vacancy-induced active centers and catalytic mechanism. Environ. Sci. Technol. 54(19), 12771–12783 (2020).
Gul, O., Polikarpova, P., Akopyan, A. & Anisimov, A. Bimetallic heterogeneous catalysts for the oxidation of sulfur-containing compounds with hydrogen peroxide. Kinet. Catal. 64(5), 627–634 (2023).
Islam, A. Preparation of Mixed Manganese Oxide Nano-Matrix and Studies on its Oxidative and Pathogenic Activities. (2015).
Dong, Y.-D. et al. Synthesis of Fe–Mn-based materials and their applications in advanced oxidation processes for wastewater decontamination: A review. Ind. Eng. Chem. Res. 62(28), 10828–10848 (2023).
Li, K., Li, G., Wei, B., Zhang, J. & Cao, X.-Q. Efficient abatement of organic pollutants by Fe (III)/Fe (II) cycle activating H2O2 with the assistance of sulfurized manganese-supported graphene. Sep. Purif. Technol. 344, 127240 (2024).
Humadi, J. I. et al. Evaluation the performance of the tin (IV) oxide (SnO2) in the removal of sulfur compounds via oxidative-extractive desulfurization process for production an eco-friendly fuel. Int. J. Chem. React. Eng. 21(6), 727–741 (2023).
Humadi, J. I., Razzaq, G. H. A., Razzaq, G. H. A., Ahmed, M. A. & Saeed, L. I. Improved kerosene quality with the use of a gamma alumina nanoparticles supported zinc oxide catalyst in a digital batch baffled reactor: Experiments and process modelling. Korean Chem. Eng. Res. 61(2), 226–233 (2023).
Aabid, A. A., Humadi, J. I., Ahmed, G. S., Jarullah, A. T., Ahmed, M. A. & Abdullah, W. S. Enhancement of desulfurization process for light gas oil using new zinc oxide loaded over alumina nanocatalyst. Appl. Sci. Eng. Prog. 16(3) 2023.
Acknowledgements
This work was partially supported by Chemical Engineering Department, College of Engineering, University of Baghdad, Iraq.
Author information
Authors and Affiliations
Contributions
Jasim I. Humadi wrote manuscript and methodology, Wadood T. Mohammed reviewed masuscript and supervision.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Humadi, J.I., Mohammed, W.T. Ultradeep oxidative desulfurization of heavy oil in oscillatory and laminar baffled reactors using polyactive magnetic activated carbon catalyst. Sci Rep 16, 984 (2026). https://doi.org/10.1038/s41598-025-30400-y
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-30400-y