Abstract
Association between estimated glucose disposal rate (eGDR) and sarcopenia among middle-aged and older adults remains unclear. To explore the relationship between eGDR and sarcopenia, and its predictive ability for sarcopenia risk among middle-aged and older adults. Data in this study were obtained from the China Health and Retirement Longitudinal Study (CHARLS) from 2011 to 2015. Logistic regression and Cox regression models were used to estimate the association of eGDR with sarcopenia. Restricted cubic splines (RCS) were employed to describe the nonlinear link between them, and subgroup analysis was conducted to ensure the stability of the results. Receiver operating characteristic curves were used to assess the capabilities of eGDR and the other six indices of insulin resistance for predicting sarcopenia risk. Four machine learning models were introduced, and the contribution of variables in the best predictive model was applied. A total of 1,627 participants participated in the cohort study. Compared with participants with eGDR ˃10.89, those with eGDR < 6.99 were at higher risk of sarcopenia, especially in individuals who are aged over 64 years and had diabetes. RCS analysis indicated a significant negative nonlinear relationship between eGDR and the risk of sarcopenia when eGDR < 7.319. A random forest model exhibited the best performance; eGDR was the key predictive factor among all variables. eGDR exhibits good capability for predicting sarcopenia risk.
Similar content being viewed by others
Introduction
Sarcopenia, characterized by a progressive and generalized loss of skeletal muscle mass and strength, often associated with aging, is increasingly being recognized as a major health concern, especially in the aging population1. Estimates suggest that sarcopenia affects approximately 10%–16% of older adults around the world, depending on factors such as age and the diagnostic criteria2. The prevalence of sarcopenia increases with age, affecting a higher proportion of individuals in their 70s and 80s. Meta-analyses have shown that the prevalence of sarcopenia varies between 10% and 27% depending on the classification and cutoff points in included studies3. On average, it is estimated that 5%–13% of individuals aged 60–70 years are affected by sarcopenia, with this number increasing to 11%–50% in those aged over 80 years4. Sarcopenia is characterized by decreased levels of three key traits: muscle strength, muscle quantity or quality, and physical performance, which can impede daily activities and negatively affect postoperative outcomes, as well as increase the risk of falls and injuries, complication rates, mortality, and morbidity in major surgical procedures5. The causes of sarcopenia are multifaceted and include factors such as lack of exercise, inadequate protein intake, chronic diseases, neuromuscular junction issues, decreased type II muscle fiber size and number, reduced serum concentrations of androgens and growth factors, genetic issues, and a blunted muscle protein synthesis response to protein or resistance exercise6,7,8,9,10. In recent years, it has been suggested that insulin resistance may be related to sarcopenia11,12. As a novel biomarker of insulin sensitivity, eGDR is derived from three clinically available variables: waist circumference, hypertension status, and glycated hemoglobin A1c (HbA1c). It was developed for type 1 diabetes and later validated in type 2 diabetes using the gold standard euglycemic hyperinsulinemic clamp13. Studies have shown that a decrease in eGDR is significantly associated with an increased risk of coronary artery disease severity and a lower risk of heart failure14,15. Compared with other indices of insulin resistance, such as the triglyceride-glucose (TyG) index and the homeostasis model assessment of insulin resistance (HOMA-IR), eGDR shows superior predictive value in terms of all-cause mortality and cardiovascular mortality15. In clinical practice, eGDR values are often interpreted categorically: values below ~ 5–6 mg/kg/min indicate significant insulin resistance, while values above ~ 8–9 mg/kg/min reflect normal insulin sensitivity16. These thresholds have been validated against hyperinsulinemic-euglycemic clamp studies and are predictive of microvascular complications and cardiovascular outcomes in diabetic populations17,18,19. However, the utility of eGDR in non-cardiometabolic conditions, particularly musculoskeletal aging, remains largely unexplored. Furthermore, eGDR does not require fasting samples or complex assays, making it highly applicable in large-scale epidemiological studies like CHARLS. There is a lack of evidence regarding the relationship between eGDR and sarcopenia, and its predictive ability. This study aimed to explore the association based on the China Health and Retirement Longitudinal Study (CHARLS) and machine learning (ML) to provide further insights into the prevention of sarcopenia.
Method
Data source and population selection
CHARLS is a large-scale interdisciplinary survey project hosted by the National School of Development at Peking University, China, and executed jointly by the China Social Science Survey Center at Peking University and the Peking University Youth League Committee. The main purpose of this project was to collect high-quality micro-data representative of Chinese families and individuals aged over 45 years to analyze the issue of the aging population in China, promote interdisciplinary research on aging, and provide a scientific basis for formulating and improving relevant policies. CHARLS uses a multi-stage sampling method, adopting probability proportional to size sampling in both county/district and village/community stages, and it pioneered the use of electronic mapping software (CHARLS-GIS) technology to create village-level sampling frames. The survey covers a wide range of information, including socioeconomic status and health conditions, to meet the needs of aging research.
The national baseline survey of CHARLS was conducted in 2011–12, followed by four rounds of regular questionnaire tracking surveys in 2013, 2015, 2018, and 2020, and a life history survey of middle-aged and elderly Chinese people in 2014. The baseline survey covered 150 national/regional areas, 450 villages/urban communities, involving 10,257 households with 17,708 people, reflecting the overall situation of China’s middle-aged and older population.
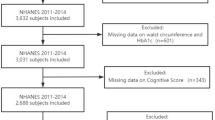
Data from 2011 to 2015, which can be publicly obtained from the CHARLS website (https://charls.pku.edu.cn/en/), were selected for analysis in this cohort study. The inclusion of participants aged ≥ 45 years aligns with the CHARLS study design, which aims to capture the transition into older adulthood—a critical period for the early development of sarcopenia and metabolic dysfunction. The exclusion criteria of participants were as follows: (1) participants in 2011 missing data of blood measurements for calculation of eGDR (waist circumference, hypertension status, and HbA1c); (2) participants missing measurement data of sarcopenia-related traits; (3) participants with sarcopenia at baseline; (4) participants missing follow-up twice. Figure 1 shows a flowchart of the selection process.
Flowchart of selection process.
Assessment of sarcopenia
The identification of sarcopenia is based on the guidelines set forth by the 2019 Asian Working Group for Sarcopenia, which categorizes the condition based on three key indicators: muscular strength, muscular mass, and physical performance20. Muscular strength was assessed by dynamometer-measured grip strength, with a decrease indicated by values ˂28 kg for males and 18 kg for females. Participants were required to hold the dynamometer with one hand while maintaining a 90°-bend at the elbow, while sitting or standing, and then grip the dynamometer with maximum force for a brief period. Each hand was measured twice, and the highest value was recorded as the individual’s grip strength. Regarding muscular mass, the CHARLS study used an estimation approach for appendicular skeletal muscle mass (ASM), based on a study by Wen et al.21. The formula for this estimation is as follows:
ASM = 0.193 * body weight (kg) + 0.107 * height (cm) − 4.157 * gender (with male coded as 1 and female as 2) − 0.037 * age − 2.631
Additionally, ASM (adjusted for height squared) was assessed in relation to height, with values falling below 7.0 kg/m2 for men and below 5.4 kg/m2 for women, signifying a reduction in muscular mass20. Physical performance was determined by either a 2.5-m walk test or a 5-repetition chair stand (CS-5) test, with a walking speed ˂ 1 m/s or a chair stand test time ≥ 12 s indicating a deterioration in physical performance. All participants aged ≥ 60 years without health conditions that restrict walking (e.g., injury) were eligible for the gait speed test and were asked to walk a straight 2.5-m flat path twice (there and back) at their usual pace. A stopwatch recorded the speed at which participants completed the gait speed test, and the average speed from both trials was calculated. A standardized armless chair (approximately 0.47 m in height) was used for the CS-5 test, and a handheld stopwatch was used for timing. The back of the chair was secured against a wall to ensure stability, and the participants were instructed to cross their arms over their chest and stand up from the chair. If the practice trial was successful, the participants were asked to perform the chair stand movement five times, as quickly as possible, without using their hands for assistance. The time from the initial seated position to the fifth complete standing position was recorded in seconds. The average speed of the CS-5 (m/s) was calculated by dividing the vertical distance covered by the center of mass by the average time taken to complete the concentric phase of the CS-5. Sarcopenia was diagnosed when there was a combination of reduced muscular mass with either diminished physical performance or weakened muscular strength.
Calculation of eGDR
eGDR serves as an indicator of insulin resistance, and it is calculated as follows22:
eGDR (mg/kg/min) = 21.158 - (3.407 * hypertension) - (0.551 * HbA1c (%)) - (0.09 * waist circumference (cm))
Here, a value of 0 indicates no hypertension and a value of 1 indicates the presence of hypertension. Hypertension is diagnosed either through self-report or by measuring an average systolic blood pressure ≥ 140 mmHg and a diastolic blood pressure ≥ 90 mmHg23.
Calculation of the other six indices of insulin resistance
The formulae for TyG, the ratio of triglyceride-to-high-density lipoprotein-cholesterol (TG/HDL), TyG adjusted for waist circumference (TyG-WC), TyG adjusted for waist-to-height ratio (TyG-WHtR), metabolic score for IR (METS-IR), and Chinese visceral adiposity index (CVAI) are as follows:
TyG = ln (Fasting Triglycerides (mg/dL) * Fasting Glucose (mg/dL)/2)
TyG-WC = TyG * Waist Circumference (cm)
TyG-WHtR = TyG * WHtR, WHtR = Waist Circumference (cm) / height (cm)
TG/HDL = Fasting triglycerides (mg/dL)/HDL cholesterol (mg/dL)
METS-IR = ln(2×Fasting Glucose (mg/dL) + Fasting Triglycerides (mg/dL)) * (BMI / ln(HDL Cholesterol (mg/dL)))
CVAI = -267.93 + 0.68 * age + 0.03 * BMI + 4.00 * WC (cm) + 22.00 * ln (TG (mmol/L)) − 16.32 * HDL-C (mmol/L) (male)
CVAI = -187.32 + 1.71 * age + 4.23 * BMI + 1.12 * WC (cm) + 39.76 * ln (TG (mmol/L)) − 11.66 * HDL-C (mmol/L) (female)
Covariates
Covariates in this study included sex, age, educational level, marital status, body mass index (BMI), waist circumference, sleep duration, smoking status, drinking status, physical activity, and diabetes. Because of the significant collinearity between hypertension status and eGDR, it was not included as a covariate. Educational level was categorized into four levels: illiterate, elementary school, middle school, and college and above. BMI was categorized into underweight (< 18.5 kg/m2), normal weight (18.5–24 kg/m2), overweight (24–28 kg/m2), and obese (≥ 28 kg/m2). Sleep duration was divided into three categories: ˂ 6 h, 6–8 h, and ≥ 8 h. Marital status was dichotomized into married or co-habitating versus unmarried or single. Smoking and drinking status were categorized into three groups: former, current, and never. Physical activity was recorded as either “active” or “inactive.” Sex, educational level, marital status, BMI, smoking status, drinking status, sleep duration, physical activity, and diabetes were considered as categorical variables, and the rest were treated as numerical variables.
Statistical analysis
Stata 18.0 and R (version 4.4.1) were used for data cleansing and analysis. The demographic and initial attributes of the participants in this research were compiled using descriptive statistics. Continuous variables that followed a normal distribution were depicted as the mean ± standard deviation (sd), and categorical variables were shown through counts and percent values. Participants were categorized into four groups for the analysis of their initial characteristics, using the quartiles of eGDR as the dividing points. Chi-square tests were used to calculate the statistical differences in categorical variables across these groups, whereas analysis of variance (ANOVA) and the Kruskal‒Wallis H test were applied to continuous variables to determine the differences among groups at the outset. Missing data were addressed through the method of multiple imputation. Logistic regression and Cox regression models were used to explore the relationship between eGDR and sarcopenia, with eGDR treated as continuous and categorical variables. Logistic regression was used to assess the cross-sectional association between eGDR and sarcopenia-related risk factors at baseline, while Cox proportional hazards models were employed to evaluate the longitudinal relationship between baseline eGDR and incident sarcopenia over the follow-up period. Restricted cubic splines (RCS) were applied to describe their nonlinear relationship. To assess whether the relationship between eGDR and sarcopenia was influenced by diabetes status, we performed stratified Cox regression analyses to support the robustness of eGDR as a predictor across glycemic statuses.Variance inflation factor (VIF) was computed to assess multicollinearity, and a VIF < 5 indicated no multicollinearity. Receiver operating characteristic (ROC) curves were used to assess the capabilities of eGDR and the other six indices of insulin resistance for predicting sarcopenia risk. P < 0.05 was considered to indicate statistical significance. The Boruta method was applied to select probable predictive factors, and decision tree (DT), random forest (RF), multi-layer perceptron (MLP), and light gradient boosting machine (LightGBM) models were introduced, and the area under the receiver operating characteristic curve (AUC), accuracy, balanced accuracy, F1 score, kappa, sensitivity (sens), specificity (spec), and SHapley Additive exPlanation (SHAP) were applied to assess the model performance.
Results
Participant characteristics
A total of 1,627 participants including 758 females and 869 males from the CHARLS database were included in this study. The average age of participants was 65.28 ± 5.19. The participants were divided into four groups based on the quartiles of eGDR: Q1 (< 6.99, n = 407), Q2 (6.99–8.48, n = 408), Q3 (8.48–10.89, n = 405), and Q4 (≥ 10.89, n = 407). The basic characteristics of the participants are shown in Table 1. The participants with the lowest level of eGDR had the following characteristics: a higher proportion of females, increased likelihood of being overweight and obese, a larger waist circumference, and increased prevalence of diabetes, compared with individuals with higher eGDR.
Multivariate logistic regression analysis of relationship between eGDR and sarcopenia
Before the logistic regression analysis, multicollinearity analysis was conducted and VIF was calculated, and all VIF values were < 5, suggesting that there was no multicollinearity (Table 2).
Subsequently, multivariate logistic regression analysis was performed to explore the association between eGDR and sarcopenia, with eGDR treated as a continuous variable and sarcopenia treated as a binary variable. During a median follow-up of 4 years, 679 out of 1,627 participants (41.733%) developed incident sarcopenia. Three models were introduced into the logistic regression analysis. Model 1 was adjusted for age, sex, educational level, marital status; Model 2 was adjusted for age, sex, educational level, marital status, BMI, waist circumference, drinking status and smoking status; Model 3 was adjusted for age, sex, educational level, marital status, BMI, waist circumference, drinking status, smoking status, diabetes, sleep duration and physical activity. Each 1-unit increase was associated with a 16.2% reduction in the odds of sarcopenia in the fully adjusted model (OR: 0.838, 95% CI: 0.711–0.985) (Table 3). Additionally, age showed a significant positive association with sarcopenia, and compared with participants who were female and of normal weight, male participants and overweight and obese participants were less likely to be affected by sarcopenia. Then, eGDR was treated as a categorical variable, and the results are shown in Table 4. A significant inverse trend across eGDR quartiles was observed in Model 1 (P for trend < 0.001) and Model 2 (P for trend < 0.001), indicating a dose-response relationship between higher eGDR levels and lower sarcopenia risk, although this trend was attenuated after full adjustment in Model 3 (P for trend = 0.671).
Multivariate cox regression analysis of relationship between eGDR and sarcopenia
To further investigate the longitude relationship between eGDR and the risk of sarcopenia, multivariate Cox regression analysis was used. The results are presented in Table 5. No covariates were regulated in Model (1) Sex, marital status, and BMI were regulated in Model 2, and sleep duration and diabetes status were increased in Model 3 based on Model (2) In Models 1 and 2, the risk of sarcopenia in the Q1 group was 1.39 times greater than that in the Q4 group (Model 1: HR: 1.329, 95% CI: 1.070–1.650, P = 0.010; Model 2: HR: 1.39, 95% CI: 1.021–1.892, P = 0.036). A trend test across eGDR quartiles revealed no significant linear trend in the fully adjusted Cox model (Model 3: P for trend = 0.478).
Subgroup analysis of relationship between eGDR and sarcopenia
To ensure the stability of the results of the longitude relationship between eGDR and sarcopenia, subgroup analysis was conducted based on gender, age, marital status, sleep duration, BMI, and diabetes status (Table 6). Among participants aged ˂ 64, compared with individuals in Q4, those in Q1 and Q3 were at higher risk of sarcopenia (HR: 2.649, 95% CI: 1.614–4.347, P < 0.001; HR: 1.633, 95% CI: 1.067–2.500, P = 0.024). Among those with diabetes, participants in Q1 had an increased likelihood of being affected by sarcopenia compared with those in Q4 (HR: 1.696, 95% CI: 1.201–2.395, P = 0.003).
Nonlinear association between eGDR and sarcopenia risk
To further validate the conclusions from logistic regression and Cox regression analyses, RCS analysis was conducted to investigate the relationship between eGDR and sarcopenia incidence. As shown in Fig. 2, after the covariates were controlled for, a significant negative relationship between eGDR and the risk of sarcopenia was found when eGDR was less than 7.319 (P for non-linearity < 0.001).
Dose‒response relationship between eGDR and sarcopenia risk. Age, sex, educational level, marital status, BMI, waist circumference, drinking status, smoking status, diabetes, sleep duration and physical activity were adjusted.
Nonlinear association between other indices of insulin resistance and sarcopenia
As shown in Fig. 3, RCS were used to investigate the nonlinear association of six insulin resistance indices with sarcopenia risk, and the results indicated the whole nonlinear effect of TyG (P-nonlinearity = 0.0069), TyG-WC (P-nonlinearity = 0.0019), TyG-WHtR (P-nonlinearity < 0.0001), TG/HDL-c (P-nonlinearity = 0.0003), METS-IR (P-nonlinearity = 0.0204), and CVAI (P-nonlinearity < 0.0001) on sarcopenia risk, and their thresholds were 8.596, 728.728, 468.130, 2.012, 33.783, and 107.510.
RCS analysis of the relationship between six IR indices and sarcopenia risk. Age, sex, educational level, marital status, BMI, waist circumference, drinking status, smoking status, diabetes, sleep duration and physical activity were adjusted. TyG, triglyceride-glucose; TyG-WC, triglyceride-glucose * waist circumference; TyG-WHtR, triglyceride-glucose/waist-to-height; TG/HDL, triglyceride/HDL-c; METS-IR, metabolic score for insulin resistance; CVAI, Chinese visceral adiposity index; OR, odds ratio; 95% CI, 95% confidence interval.
Predictive performance of eGDR and the other six insulin resistance indices for sarcopenia
As shown in Fig. 4, the effectiveness of eGDR and the other six indices in predicting sarcopenia was evaluated. It was observed that TyG-WHtR presented the highest AUC value of 0.710, followed by eGDR (AUC = 0.613), surpassing the other five indices of insulin resistance, indicating the superior performance of TyG-WHtR and eGDR for predicting sarcopenia. Notably, the best threshold of eGDR for sarcopenia prediction was 7.364, with a specificity of 0.730 and a sensitivity of 0.592.
ROC of eGDR and the other six IR indices for predicting sarcopenia. AUC, area under the curve; roc1, eGDR; roc2, TyG; roc3, TyG-WC, roc4, TyG-WHtR; roc5, TG/HDL; roc6, METS-IR; roc7, CVAI.
Model evaluation and comparison
The results of selection of variables for ML models using the Boruta algorithm are shown in Fig. 5. The shaded feature variables were selected for ML. Figure 6 shows the ROC and the heatmaps illustrating the performance of AUC, accuracy, balanced accuracy, F1 score, kappa, sens, and spec of the four ML models on test data. The LightGBM model exhibited the best interpretability. However, the RF has a simpler structure that is easier to understand and interpret than the LightGBM, and it can also reduce the impact of outliers. Therefore, RF was considered the best model.
Result of Boruta method for selection of variables for ML models.
(A) ROC of four ML models. (B) Heatmaps showing the performance of RF, MLP, LightGBM, and DT models. Red indicates the higher values, and green indicates the lower values, demonstrating the effectiveness of the four models in the test set.
Visualization of variable contribution
SHAP analysis was conducted to evaluate the contribution of each characteristic variable in the RF model prediction, as shown in Fig. 7. eGDR had the highest SHAP value among all variables and all indices of insulin resistance, and it exhibited the most important protective effect on sarcopenia risk.
(A) ROC curve of the RF model. (B) Importance of variables in the RF model; waist, waist circumference; tyg, triglyceride-glucose; tygwc, triglyceride-glucose*waist circumference; tygwhtr, triglyceride-glucose/waist-to-height; tghdl, triglyceride/HDL-c; metsir, metabolic score for insulin resistance; CVAI, Chinese visceral adiposity index.
Discussion
Multivariate logistic regression analysis showed that eGDR was negatively related to the risk of sarcopenia when treated as a continuous variable. When eGDR was classified into four groups based on quartiles, eGDR < 6.99 showed a positive effect on the risk of sarcopenia compared to that when eGDR > 10.89. The results of the Cox regression analysis were consistent with the findings of the logistic regression analysis.
At present, there is no evidence to prove the association of eGDR with sarcopenia. eGDR is a good indicator to reflect insulin resistance, which plays an important role in the progress of sarcopenia. The relationship between insulin resistance and sarcopenia is complex. The primary organ involved in insulin action is the skeletal muscle, which accounts for 40%–50% of adult lean body mass24. Glucose uptake and metabolism in skeletal muscle cells play a major role in blood glucose regulation. Insulin resistance refers to the reduced responsiveness of the skeletal muscle to insulin, which results in impaired glucose metabolism in cells throughout the body, thereby inducing the development of sarcopenia25. Muscle reduction is associated with insufficient amino acid intake and reduced insulin sensitivity. The insulin and amino acid signaling pathways work together to promote the increase of skeletal muscle protein, which highlights the role of key factors. The structure and function of mitochondria, maintained through a series of fusion and fission activities, are other factors that contribute to the risk of developing sarcopenia. Sarcopenia-related obesity downregulates the expression of the Mitofusin2 (Mfn2) gene, while a high-fat diet increases the expression of Dynamin-Related Protein 1 (Drp1) and Mitochondrial fission protein 1 (Fis1), leading to increased mitochondrial degradation in the skeletal muscle, thereby inducing insulin resistance26,27,28,29. In addition, sarcopenia may lead to increased levels of branched-chain amino acids (BCAAs), which are associated with the development of insulin resistance. BCAAs are abundant in muscles, accounting for about 30% of muscle protein content, and the skeletal muscle is the primary site for BCAA catabolism. Increased BCAA levels may contribute to insulin resistance by overactivating the mechanistic target of rapamycin complex 1 (mTORC1) and p70 ribosomal protein S6 kinase 1 (S6K1), leading to the dephosphorylation of insulin receptor substrate 1 (IRS1), or by causing mitochondrial dysfunction, which affects insulin sensitivity30,31,32,33,34,35. Sarcopenia and insulin resistance may promote each other’s progression, aggravating the outcomes of sarcopenia.
eGDR has a strong correlation with insulin resistance, as measured by the hyperinsulinemic-euglycemic clamp (HEC) technique, indicating that eGDR is a reliable indicator of insulin resistance and that it is suitable for epidemiological studies and clinical practice36. The calculation of eGDR is based on routine clinical parameters, such as HbA1c, blood pressure, and waist circumference, which are easily accessible and do not require complex laboratory equipment or techniques. As a non-invasive assessment method, eGDR is much more cost-effective than HEC technology, which requires specialized equipment and conditions37.
Studies have shown that eGDR correlates well with the degree of insulin resistance measured by HEC technology, accounting for more than 60% of insulin resistance38. In the calculation of eGDR, HbA1c serves as an indicator reflecting the average blood glucose level over the previous 2–3 months. Studies have also found that insulin resistance is positively correlated with elevated HbA1c levels. This is because insulin resistance leads to an imbalance in glucose metabolism, resulting in chronic hyperglycemia, which, in turn, leads to increased HbA1c levels39.
Insulin resistance can increase blood pressure through various mechanisms, including sodium and water retention, increased blood volume, increased sympathetic nerve activity, and elevated intracellular calcium ion concentrations40. Additionally, hypertension is closely related to insulin resistance, and research has shown that improving insulin resistance can reduce blood pressure, and vice versa41. Waist circumference is a measure of abdominal obesity, which is strongly associated with insulin resistance. A larger waist circumference indicates more abdominal fat accumulation, which is typically associated with insulin resistance42. Studies have also found that waist circumference is significantly correlated with insulin resistance indices (such as HOMA-IR) and is an independent risk factor affecting insulin resistance43. In summary, HbA1c, blood pressure status, and waist circumference are all directly related to insulin resistance and can serve as important parameters for assessing an individual’s insulin resistance status.
To date, no study has identified the relationship between eGDR and the risk of sarcopenia. This study found that eGDR, as a continuous variable, exerted a negative impact on sarcopenia risk after age, sex, BMI, and diabetes status were regulated. The results of logistic regression and Cox regression analyses demonstrate that participants with eGDR < 6.99 had an increased sarcopenia risk compared with those in Q4, while the relationships between other groups and sarcopenia were not significant, indicating the significance of a threshold of eGDR < 6.99 in evaluating the risk of sarcopenia. The observed inverse association between eGDR and sarcopenia risk was further supported by significant trend tests across eGDR quartiles in the partially adjusted logistic regression models (P for trend < 0.001). Although this trend was attenuated in the fully adjusted models (both logistic and Cox), the persistent elevation of risk in the lowest eGDR quartile (Q1) underscores the clinical importance of severe insulin resistance, as captured by an eGDR threshold below 6.99 mg/kg/min. This pattern suggests a potential non-linear or threshold effect, where the risk of sarcopenia increases markedly only after insulin resistance surpasses a critical severity, rather than following a strict linear gradient. This interpretation is consistent with the non-linear relationship identified by our RCS analysis.
Subgroup analysis identified the significant association of eGDR < 6.99 with an increased risk of sarcopenia, especially in individuals who are aged over 64 years and with diabetes. With increased age, the sensitivity of muscles to insulin declines, which is defined as a reduction in the body’s overall glucose disposal capacity in response to insulin, due to age-related disabilities, increases in muscle and central fat, mitochondrial dysfunction, inflammation, or changes in cellular signaling mechanisms44. Diabetes itself is closely related to insulin resistance, and the prevalence of sarcopenia is higher in individuals with diabetes, ranging from 18 to 66%2. While interaction between BMI, sleep duration and eGDR was insignificant, eGDR < 6.99 was associated with higher sarcopenia risk especially in participants with normal weight and short sleep duration. Individuals of normal weight may exhibit insulin resistance due to the accumulation of visceral fat, which may not be identified using the traditional BMI. The accumulation of visceral fat may lead to insulin resistance, which, in turn, affects muscle mass and function. This interaction may result in a vicious cycle between muscle reduction and fat infiltration, promoting the development of sarcopenic obesity45. Research shows that there is a U-shaped association between sleep duration and the prevalence of sarcopenia, with about 6.5 h of sleep significantly reducing the risk of sarcopenia46. In addition, short sleep duration is significantly associated with insulin resistance due to inflammatory markers, such as C-reactive protein and serum amyloid A secretion47. RCS analysis revealed that when eGDR < 7.319, eGDR exerted a significant negative effect on sarcopenia risk. The nonlinear relationship observed in the RCS analysis, showing a plateau in risk reduction beyond an eGDR of approximately 7.319, likely reflects a saturation effect in the physiological benefits of improved insulin sensitivity on skeletal muscle. Initially, as eGDR rises from severely low levels, the restoration of insulin signaling potently reverses key pathological processes: it ameliorates anabolic resistance by activating the PI3K/Akt/mTOR pathway, suppresses hyperactive proteolysis via the ubiquitin-proteasome system, and improves mitochondrial bioenergetics48,49. However, once a critical threshold of insulin sensitivity is achieved, these pathways may operate near their functional capacity; beyond this point, further improvements in insulin sensitivity yield diminishing returns for muscle health, as other non-insulin-sensitive pathways, age-related neurological decline, or inflammatory processes become the predominant limiting factors for sarcopenia risk50,51.
Notably, compared with other indices of insulin resistance, eGDR exhibited a good ability to predict sarcopenia risk, with a threshold of 7.364. These results demonstrated the significance of monitoring eGDR when evaluating the risk of sarcopenia. Among all ML models, the RF model exhibited the best performance, and eGDR had the highest SHAP value, indicating great capability of eGDR for predicting sarcopenia. However, in our machine learning framework, age was excluded as a predictive feature by the Boruta algorithm. While this might initially seem counterintuitive given the well-established correlation between aging and sarcopenia, it likely indicates that the predictive information conveyed by chronological age was effectively captured by other, more direct physiological measures in the model, such as eGDR, waist circumference, and diabetes status. This phenomenon, known as feature redundancy or non-linear mediation, suggests that the decline in insulin sensitivity and the associated metabolic dysregulation (quantified by eGDR) may serve as a key pathway through which aging exerts its effect on muscle health. Consequently, the model prioritized these mechanistically proximate variables. However, this also represents a limitation: our model is optimized for predicting risk based on a specific metabolic profile rather than providing a comprehensive assessment that explicitly includes the independent contribution of chronological aging. This may affect the model’s performance in populations where sarcopenia is driven predominantly by non-metabolic, age-related factors such as neurodegenerative processes or purely disuse atrophy.
The current study establishes several significant contributions. First, this is the first research to identify a novel inverse association between eGDR and sarcopenia risk, extending the clinical utility of eGDR beyond diabetes management into sarcopenia evaluation. Second, integrating multiple machine learning models and interpretability tools represents a methodological advancement over previous studies relying solely on conventional statistical analyses. Finally, subgroup analyses revealing age- and diabetes-dependent associations align with known pathophysiology, enhancing biological plausibility. However, several limitations warrant acknowledgment. Residual confounding may persist despite adjusting for major confounders, such as unmeasured inflammatory markers that jointly influence insulin resistance and muscle metabolism. Although we identified thresholds, the lack of serial eGDR measurements limits insight into how dynamic eGDR changes affect sarcopenia progression. Additionally, generalizability may be constrained by the single-center cohort and underrepresentation of ethnic minorities. Finally, it is important to note that eGDR incorporates HbA1c, which can be lowered by glucose-lowering therapies independent of changes in underlying insulin sensitivity. This may lead to an overestimation of eGDR and an underestimation of sarcopenia risk in treated diabetic individuals. Future studies using gold-standard measures of insulin sensitivity are needed to validate these findings in pharmacologically diverse populations.
Conclusion
eGDR < 6.99 can be considered as a risk factor of sarcopenia compared with eGDR ≥ 10.89, particularly in individuals who are aged over 64 years and with diabetes. A significant negative nonlinear relationship exists between eGDR and the risk of sarcopenia when eGDR < 7.319. Notably, eGDR exhibits a good ability to predict sarcopenia risk.
Data availability
Publicly available datasets were analyzed in this study. This data can be found here: [http://charls.pku.edu.cn/en](http:/charls.pku.edu.cn/en) .
References
Santilli, V., Bernetti, A., Mangone, M. & Paoloni, M. Clinical definition of sarcopenia. Clin. Cases Min. Bone Metab. 11 (3), 177–180 (2014).
Yuan, S. & Larsson, S. C. Epidemiology of sarcopenia: Prevalence, risk factors, and consequences. Metabolism 144, 155533. https://doi.org/10.1016/j.metabol.2023.155533 (2023).
Petermann-Rocha, F. et al. Global prevalence of sarcopenia and severe sarcopenia: a systematic review and meta-analysis. J. Cachexia Sarcopenia Muscle. 13 (1), 86–99. https://doi.org/10.1002/jcsm.12783 (2022).
von Haehling, S., Morley, J. E. & Anker, S. D. An overview of sarcopenia: facts and numbers on prevalence and clinical impact. J. Cachexia Sarcopenia Muscle. 1 (2), 129–133. https://doi.org/10.1007/s13539-010-0014-2 (2010).
Cruz-Jentoft, A. J., Sayer, A. A. & Sarcopenia Lancet ;393(10191):2636–2646. https://doi.org/10.1016/s0140-6736(19)31138-9. (2019).
Rolland, Y. et al. Sarcopenia: its assessment, etiology, pathogenesis, consequences and future perspectives. J. Nutr. Health Aging. 12 (7), 433–450. https://doi.org/10.1007/bf02982704 (2008).
Aslam, M. A., Ma, E. B. & Huh, J. Y. Pathophysiology of sarcopenia: genetic factors and their interplay with environmental factors. Metabolism 149, 155711. https://doi.org/10.1016/j.metabol.2023.155711 (2023). http://dx.doi.org/https://doi.org/
Liu, D. et al. Frontiers in sarcopenia: advancements in diagnostics, molecular mechanisms, and therapeutic strategies. Mol. Aspects Med. 97, 101270. https://doi.org/10.1016/j.mam.2024.101270 (2024).
Miao, Y. et al. Unraveling the causes of sarcopenia: roles of neuromuscular junction impairment and mitochondrial dysfunction. Physiol. Rep. 12 (1), e15917. https://doi.org/10.14814/phy2.15917 (2024).
Liu, B., Liu, R., Jin, Y., Ding, Y. & Luo, C. Association between possible sarcopenia, all-cause mortality, and adverse health outcomes in community-dwelling older adults in China. Sci. Rep. 14 (1), 25913. https://doi.org/10.1038/s41598-024-77725-8 (2024).
Chen, H. et al. The association between sarcopenia and diabetes: from pathophysiology mechanism to therapeutic strategy. Diabetes Metab. Syndr. Obes. 16, 1541–1554. https://doi.org/10.2147/dmso.S410834 (2023).
Ostler, J. E. et al. Effects of insulin resistance on skeletal muscle growth and exercise capacity in type 2 diabetic mouse models. Am. J. Physiol. Endocrinol. Metab. 306 (6), E592–605. https://doi.org/10.1152/ajpendo.00277.2013 (2014).
Meng, F-Q., Zhang, Y., Bai, X-X., Kong, F-L. & Li, F-E. Ischemic stroke and sarcopenia have an asymmetric bidirectional relationship based on a two-sample Mendelian randomization study. Front. Neurol. 15. https://doi.org/10.3389/fneur.2024.1427692 (2024).
Zhang, D. et al. Usefulness of estimated glucose disposal rate in detecting heart failure: results from National health and nutrition examination survey 1999–2018. Diabetol. Metab. Syndr. 16 (1), 189. https://doi.org/10.1186/s13098-024-01402-z (2024).
Song, J., Ma, R. & Yin, L. Associations between estimated glucose disposal rate and arterial stiffness and mortality among US adults with non-alcoholic fatty liver disease. Front. Endocrinol. (Lausanne). 15, 1398265. https://doi.org/10.3389/fendo.2024.1398265 (2024).
Sagar, R. C. et al. Insulin resistance in type 1 diabetes is a key modulator of platelet hyperreactivity. Diabetologia 68 (7), 1544–1558. https://doi.org/10.1007/s00125-025-06429-z (2025).
Guo, L. et al. The role of estimated glucose disposal rate in predicting cardiovascular risk among general and diabetes mellitus population: a systematic review and meta-analysis. BMC Med. 23 (1), 234. https://doi.org/10.1186/s12916-025-04064-4 (2025).
Zooravar, D., Radkhah, H., Amiri, B. S. & Soltani, P. Estimated glucose disposal rate and microvascular complications of diabetes mellitus type I: A systematic review and meta-analysis. Diab Vasc Dis. Res. 22 (2), 14791641251324612. https://doi.org/10.1177/14791641251324612 (2025).
Dastjerdi, P. et al. Estimated glucose disposal rate and risk of cardiovascular events in type 1 diabetes: a systematic review and meta-analysis. Diabetol. Metab. Syndr. 17 (1), 348. https://doi.org/10.1186/s13098-025-01900-8 (2025).
Chen, L. K. et al. Asian working group for sarcopenia: 2019 consensus update on sarcopenia diagnosis and treatment. J. Am. Med. Dir. Assoc. 21 (3), 300–7e2. https://doi.org/10.1016/j.jamda.2019.12.012 (2020).
Wen, X., Wang, M., Jiang, C. M. & Zhang, Y. M. Anthropometric equation for Estimation of appendicular skeletal muscle mass in Chinese adults. Asia Pac. J. Clin. Nutr. 20 (4), 551–556 (2011).
Williams, K. V., Erbey, J. R., Becker, D., Arslanian, S. & Orchard, T. J. Can clinical factors estimate insulin resistance in type 1. diabetes? Diabetes. 49 (4), 626–632. https://doi.org/10.2337/diabetes.49.4.626 (2000).
Williams, B. et al. 2018 ESC/ESH guidelines for the management of arterial hypertension. Eur. Heart J. 39 (33), 3021–3104. https://doi.org/10.1093/eurheartj/ehy339 (2018).
Liu, Z. J. & Zhu, C. F. Causal relationship between insulin resistance and sarcopenia. Diabetol. Metab. Syndr. 15 (1), 46. https://doi.org/10.1186/s13098-023-01022-z (2023).
Li, C. W. et al. Pathogenesis of sarcopenia and the relationship with fat mass: descriptive review. J. Cachexia Sarcopenia Muscle. 13 (2), 781–794. https://doi.org/10.1002/jcsm.12901 (2022).
Cleasby, M. E., Jamieson, P. M. & Atherton, P. J. Insulin resistance and sarcopenia: mechanistic links between common co-morbidities. J. Endocrinol. 229 (2), R67–81. https://doi.org/10.1530/joe-15-0533 (2016).
Marzetti, E., Calvani, R., Coelho-Junior, H. J. & Picca, A. Mitochondrial pathways and sarcopenia in the geroscience era. J. Nutr. Health Aging. 28 (12), 100397. https://doi.org/10.1016/j.jnha.2024.100397 (2024).
Marzetti, E., Calvani, R., Coelho-Júnior, H. J., Landi, F. & Picca, A. Mitochondrial quantity and quality in age-related sarcopenia. Int. J. Mol. Sci. 25 (4). https://doi.org/10.3390/ijms25042052 (2024).
Marzetti, E. et al. Restoring mitochondrial function and muscle satellite cell signaling: remedies against age-related sarcopenia. Biomolecules 14 (4). https://doi.org/10.3390/biom14040415 (2024).
Cochet, C., Belloni, G., Buondonno, I., Chiara, F. & D’Amelio, P. The role of nutrition in the treatment of sarcopenia in old patients: from restoration of mitochondrial activity to improvement of muscle performance, a systematic review. Nutrients 15 (17). https://doi.org/10.3390/nu15173703 (2023).
Liu, H. et al. Associations between sarcopenia and Circulating branched-chain amino acids: a cross-sectional study over 100,000 participants. BMC Geriatr. 24 (1), 541. https://doi.org/10.1186/s12877-024-05144-5 (2024).
Conte, E. et al. Branched-chain amino acids and L-alanine supplementation ameliorate calcium dyshomeostasis in sarcopenia: new insights for nutritional interventions. Front. Pharmacol. 15, 1393746. https://doi.org/10.3389/fphar.2024.1393746 (2024).
Cao, W. et al. Aerobic exercise attenuates insulin resistance via restoring branched chain amino acids homeostasis in obese mice. Front. Nutr. 11, 1451429. https://doi.org/10.3389/fnut.2024.1451429 (2024).
Mann, G. & Adegoke, O. A. J. Elevated BCAA catabolism reverses the effect of branched-chain ketoacids on glucose transport in mTORC1-dependent manner in L6 myotubes. J. Nutr. Sci. 13, e66. https://doi.org/10.1017/jns.2024.66 (2024).
Lim, J. J. et al. Low serum Glycine strengthens the association between branched-chain amino acids and impaired insulin sensitivity assessed before and after weight loss in a population with pre-diabetes: the PREVIEW_NZ cohort. Clin. Nutr. 43 (12), 17–25. https://doi.org/10.1016/j.clnu.2024.09.047 (2024).
Peng, J. et al. Estimated glucose disposal rate for predicting cardiovascular events and mortality in patients with non-diabetic chronic kidney disease: a prospective cohort study. BMC Med. 22 (1), 411. https://doi.org/10.1186/s12916-024-03582-x (2024).
Zhu, B. et al. The predictive value of estimated glucose disposal rate for all-cause and cardiovascular mortality in the US non-diabetic population aged ≥ 60 years: A population-based cohort study. Diabetes Metab. Syndr. 19 (1), 103182. https://doi.org/10.1016/j.dsx.2024.103182 (2024).
Yi, J., Qu, C., Li, X. & Gao, H. Insulin resistance assessed by estimated glucose disposal rate and risk of atherosclerotic cardiovascular diseases incidence: the multi-ethnic study of atherosclerosis. Cardiovasc. Diabetol. 23 (1), 349. https://doi.org/10.1186/s12933-024-02437-2 (2024).
Kumar, J. et al. A comparative analysis of clinical features of diabetes mellitus type 2 with respect to duration of diabetes. Cureus 16 (11), e74849. https://doi.org/10.7759/cureus.74849 (2024).
Shine, B. K., Choi, J. E., Park, Y. J. & Hong, K. W. The genetic variants influencing hypertension prevalence based on the risk of insulin resistance as assessed using the metabolic score for insulin resistance (METS-IR). Int. J. Mol. Sci. 25 (23). https://doi.org/10.3390/ijms252312690 (2024).
Tang, S. et al. C-reactive protein-triglyceride glucose index predicts stroke incidence in a hypertensive population: a National cohort study. Diabetol. Metab. Syndr. 16 (1), 277. https://doi.org/10.1186/s13098-024-01529-z (2024).
Huang, L. H., Liao, Y. L. & Hsu, C. H. Waist circumference is a better predictor than body mass index of insulin resistance in type 2 diabetes. Obes. Res. Clin. Pract. 6 (4), e263–346. https://doi.org/10.1016/j.orcp.2011.11.003 (2012).
Tang, Y., Li, L. & Li, J. Correlations of the triglyceride-glucose index and modified indices with arterial stiffness in overweight or obese adults. Front. Endocrinol. (Lausanne). 15, 1499120. https://doi.org/10.3389/fendo.2024.1499120 (2024).
Shur, N. F. et al. Age-related changes in muscle architecture and metabolism in humans: the likely contribution of physical inactivity to age-related functional decline. Ageing Res. Rev. 68, 101344. https://doi.org/10.1016/j.arr.2021.101344 (2021).
Han, M. et al. Initial stroke severity and discharge outcome in patients with muscle mass deficit. Sci. Rep. 14 (1). https://doi.org/10.1038/s41598-024-52381-0 (2024).
Zhang, G. et al. Association of sleep duration and prevalence of sarcopenia: A large cross-sectional study. Prev. Med. Rep. 42, 102741. https://doi.org/10.1016/j.pmedr.2024.102741 (2024).
Punjabi, N. M. et al. Sleep-disordered breathing, glucose intolerance, and insulin resistance: the sleep heart health study. Am. J. Epidemiol. 160 (6), 521–530. https://doi.org/10.1093/aje/kwh261 (2004).
Santangeli, E. et al. Pathophysiological-based nutritional interventions in cirrhotic patients with sarcopenic obesity: A state-of-the-art narrative review. Nutrients 16 (3). https://doi.org/10.3390/nu16030427 (2024).
Waye, A. A. et al. Reduced bioenergetics and mitochondrial fragmentation in human primary cytotrophoblasts induced by an EGFR-targeting chemical mixture. Chemosphere 364, 143301. https://doi.org/10.1016/j.chemosphere.2024.143301 (2024).
Tieland, M., Trouwborst, I. & Clark, B. C. Skeletal muscle performance and ageing. J. Cachexia Sarcopenia Muscle. 9 (1), 3–19. https://doi.org/10.1002/jcsm.12238 (2018).
Chen, M., Cao, Y., Ji, G. & Zhang, L. Lean nonalcoholic fatty liver disease and sarcopenia. 14–2023 https://doi.org/10.3389/fendo.2023.1217249 (2023).
Acknowledgements
Thanks to the editors and reviewers for their hard work and important comments. We thank LetPub (www.letpub.com.cn) for its linguistic assistance during the preparation of this manuscript.
Funding
This work was supported by the National Key Research and Development Program of China [grant number 2023YFC3503705].
Author information
Authors and Affiliations
Contributions
Yan Chen: writing–original draft, Lingli Gao, Xiaolei Song and Mingli Wu: writing–review and editing, Ruiqing Li, Kaiqi Su and Zhuan Lv: visualization, Jing Gao and Xiaodong Feng: supervision.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval
The CHARLS was ethically approved by the institutional review board at Peking University (Nu: IRB00001052–11015), and have therefore been performed in accordance with the ethical standards laid down in the 1964 Declaration of Helsinki and its later amendments.
Consent to participate
All participants signed written informed consent.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Chen, Y., Gao, L., Song, X. et al. The relationship between estimated glucose disposal rate and sarcopenia among middle-aged and older adults. Sci Rep 16, 1375 (2026). https://doi.org/10.1038/s41598-025-31091-1
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-31091-1









