Abstract
The surgical approach for papillary thyroid cancer (PTC) by either lobectomy or total thyroidectomy (TT) has long been a topic of debate, especially for patients with intermediate-risk PTC, such as T1 and T2 stage patients with more than five positive lymph node metastases (LNM). This study analyzed a population-based retrospective cohort of T1 and T2 PTC patients with more than five positive LNM from the SEER database (2004–2017), comparing clinicopathologic features and survival outcomes between those undergoing lobectomy and TT. Cox proportional hazards regression analysis to explore prognostic factors of survival. Propensity score matching (PSM) was used to balance covariates. The study included 5,610 patients, with 5,322 (94.87%) receiving TT and 288 (5.13%) undergoing lobectomy. Patients in the TT group had higher rates of N1b stage diagnoses, multifocal lesions, and more lymph nodes examined and more lymph nodes positive. Survival analysis showed no difference in overall survival (OS) between the groups (p = 0.177), but a significant difference in cancer-specific survival (CSS) (p < 0.001). After 1:1 PSM, there were no significant difference in OS (p = 0.089) and CSS (p = 0.350). Additionally, stratified analysis showed no significant difference in OS and CSS between patients treated with TT plus radioactive iodine (RAI) ablation and those who underwent lobectomy (both p > 0.05). This cohort study suggests that T1 and T2 PTC patients with more than five positive LNM have no additional survival benefit of TT over lobectomy. If RAI ablation is not planned, lobectomy may be an effective alternative.
Similar content being viewed by others
Introduction
Papillary thyroid carcinoma (PTC) constitutes over 90% of all thyroid neoplasms and is typically characterized by an indolent disease course and favorable prognosis1,2,3,4. With the growing emphasis on risk-stratified management, guideline-recommended approaches have evolved to permit low-intensity treatment options for low-risk patients5,6. A significant modification in treatment protocols is the reduced utilization of total thyroidectomy (TT) and postoperative radioactive iodine (RAI) ablation in patients with T1 and T2 PTC5,7.
For PTC patients with lymph node metastasis, TT followed by RAI ablation is recommended despite limited direct evidence. Guidelines suggest this approach due to the higher recurrence risk, particularly in T1 and T2 intermediate-risk PTC patients with more than five positive lymph nodes. However, it’s uncertain if TT is better than lobectomy for these cases.
Therefore, this study employed population-based data from the Surveillance, Epidemiology, and End Results (SEER) database to compare overall survival (OS) and cancer-specific survival (CSS) in T1 and T2 PTC with more than five positive lymph node metastases (LNM) treated with either lobectomy or TT using propensity score matching (PSM) analysis.
Materials and methods
Data sources
SEER*STAT software version 8.4.0 was used for obtaining the study population. The study cohort was obtained from the Surveillance, Epidemiology, and End Results (SEER) from 17 regions in the United States, consisting of data between 2004 and 2017, representing approximately 26.5% of the US population.
The protocol for this study was approved by the Ethics Committee of Chengdu Fifth People’s Hospital (The Second Clincal Medical College, Affiliated Fifth People’s Hospital of Chengdu University of Traditional Chinese Medicine). As the SEER database was publicly accessible, the present study did not require informed patient consent.
Patient selection
We extracted the clinicopathological characteristics of patients with papillary thyroid carcinoma (PTC, ICD-O-3 codes 8050/3, 8260/3, 8341/3, 8342/3, 8343/3, and 8344/3), and follicular variant of papillary carcinoma (PTCF, ICD-O-3 codes 8340/3) in SEER database. All the patients enrolled in the study were unilateral PTC with ipsilateral neck metastasis, which ensured a similar tumor stage and surgical extent. The detailed study design and inclusion and exclusion criteria are described in Fig. 1. According to the different surgical procedures, all patients were divided into two groups: TT and Lobectomy.
Flowchart of Patient Inclusion and Exclusion.
Evaluation of variables clinicopathological variable assessment
The clinicopathological characteristics, including age at diagnosis, age group ( ≦ 55 y, > 55 y), sex (female, male), race, marital status at diagnosis, T stage, N stage, multifocal, number of LN Examined, number of LN Positive and RAI ablation (yes, no) were analyzed. The primary endpoints were thyroid cancer-specific survival (CSS, defined as the time from diagnosis to death from thyroid cancer) and overall survival (OS, defined as survival time until death by any reason). 1:1 PSM (exact match, match tolerance = 0) was performed to further compare the CSS and OS by adjusting for independent risk factors of survival.
Statistical analysis
The research group was divided into two groups based on therapy, TT versus lobectomy. Differences in categorical variables were compared by using the Chi-square test and Fisher’s exact test, while that in continuous variables were compared using Student’s t-test. OS and CSS were estimated by the Kaplan-Meier method and compared with the log-rank test. Univariable and multivariable Cox proportional hazard regression was used to determine independent prognostic factors, with hazard ratios (HR) and 95% confidence intervals (95%CI).
For the statistical analyses, we employed IBM SPSS Statistics 24.0 (IBM Corporation, Armonk, NY, USA) and R Studio version 4.3.0 software (The R Foundation for Statistical Computing, Austria, Vienna). A p value less than 0.05 was considered to have statistical significance.
Results
Baseline demographic and clinicopathological characteristics
After excluding those who did not fulfill the inclusion criteria, a total of 5,610 PTC patients were eligible for analysis (Fig. 1). Among them, 5,322 (94.87%) patients received TT, and 288 (5.13%) patients underwent lobectomy. The clinicopathological characteristics of patients in the two groups are shown in Table 1. Compared to patients in the lobectomy group, patients in TT the group were diagnosed with a higher proportion of N1b stage (49.31% vs. 57.85%, p = 0.004), multifocal lesions (54.64% vs. 42.36%, p < 0.001), and higher number of lymph node (LN) Examined and higher number of LN Positive (both p < 0.001).
To confirm the conclusion and balance the bias from other variables, a 1:1 (lobectomy and TT) matched case-control analysis by PSM was carried out to balance the differences of baseline characteristics in the two group. After PSM, 287 pairs of patients were available for analysis (Fig. 1). Compared with patients in the TT group, patients in the lobectomy group were less likely to be female (194 [67.60%] vs. 199 [69.34%]), slightly younger (< 55 years, 247 [86.06%] vs. 245 [85.37%], have a larger tumor (T2, 131 [45.64%] vs. 126 [43.90%], have less N1b stage (142 [49.48%] vs. 164 [57.14%]), and have less multifocality (122 [42.51%] vs. 140 [48.78%]. Similar numbers of LN examined (median [IQR], 18.00 [10.00, 33.00]) and similar numbers LN Positive (median [IQR], 8.00 [6.00, 11.00]) were observed between 2 groups; however, none of these differences were significant (Table 2).
Comparison of survival outcomes
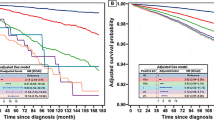
The median follow-up period for the SEER cohort was 89 months. Figure 2 presents the survival curves according to surgical intervention using the Kaplan-Meier method. Overall, OS showed no significant difference between the two groups (HR, 0.640; 95% CI, 0.334–1.229; P = 0.177), while CSS showed significant difference between the two groups (HR, 0.242; 95% CI, 0.098–0.596; P < 0.001).
OS and CSS before PSM.
After PSM, 287 patients from each group were enrolled in the analysis. There was still no difference in OS between the two groups (HR, 0.343; 95% CI, 0.094–1.248; P = 0.089). What’s important, there was no difference in CSS (HR, 0.522; 95% CI, 0.130–2.087; P = 0.350) between the two groups (Fig. 3).
OS and CSS after PSM.
OS and CSS in TT + RAI ablation group and counterparts.
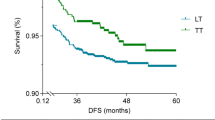
Stratified analysis was performed in patients treated with RAI ablation in the TT group (n = 106) and counterparts in the lobectomy group (n = 106). Similarly, no significant difference in OS (HR, 0.994; 95% CI, 0.139–7.117; P = 0.997) and CSS (HR, 0.523; 95% CI, 0.047–5.816; P = 0.593) between the lobectomy and TT plus RAI ablation groups were found (Figure 4).
Univariate and multivariate Cox regression analyses for survival
Tables 3 and 4 display the results of the univariate and multivariate Cox regression analyses. Univariate analysis of all patients revealed that OS was associated with age, sex, race, marital status, and N stage, while CCS was associated with age, sex, race, marital status, number of LN examined and number of LN positive. Multivariable analysis demonstrated that age, sex and marital status were independent risk factors for OS. Meanwhile, age and marital status were independent risk factors for CSS. Overall, surgical extent (lobectomy and TT) was neither an independent risk factor for OS nor for CSS.
Discussion
The optimal surgical approach selection for intermediate risk of PTC patients, especially for T1 and T2 stage with more than five positive lymph nodes remains controversial, and high quality prospective studies are still lacking. In the present cohort study, we compared OS and CSS for those patients with intermediate risk of PTC after lobectomy vs. TT. To create a comparable case series, we admitted patients with unilateral PTC with ipsilateral neck metastasis, which ensured a similar tumor stage and surgical extent. Moreover, PSM analysis was performed to minimize the influence of related factors. The results of our large-scale population-based study revealed that TT does not confer an additional survival advantage over lobectomy, which may provide a meaningful reference to assist clinicians in treatment decision-making.
It is generally accepted that lobectomy is only an optimal option for patients with but not for patients with intermediate-high–risk PTC5,8,9. However, low-risk PTC such as T1 or T2 patients with cN0 preoperative evaluation may turn to be intermediate–risk after final paraffin pathological examination, due to the number of positive LN exceed five. Clinicians have long puzzled over whether this group of people should have additional resection of the remaining half of the gland lobe, for the sake of further iodine-131 therapy to reduce the risk of recurrence10,11.
Every coin has two sides. Advocates of TT argue it allows for RAI ablation to detect and treat residual or metastatic disease. While ATA guidelines recommend considering RAI ablation for intermediate-risk PTC patients post-TT, data supporting this are limited and debated12. One analyses from the SEER database suggest that RAI ablation is associated with a 29% reduction in the risk of death, with a hazard risk of 0.75 in patients with intermediate-risk PTC, which is the first nationally representative study of intermediate-risk PTC patients and RAI therapy demonstrating an association of RAI with improved overall survival13. However, a retrospective study from a Chinese Cancer Center suggest that patients with PTC and N1b LNM (No. of metastatic lymph nodes, median [IQR]: 85,6,7,8,9,10,11,12,13 had a similar RFS after lobectomy vs. those who had TT14. Simiarly, another retrospective study from Memorial Sloan-Kettering Cancer Center examining RFS also suggested no significant benefit in patients with more advanced-stage tumors or regional metastases have low rates of recurrence and high rates of survival when managed without RAI ablation15.
Currently, no consensus has been reached through previous studies regarding the optimal surgical extent for T1 and T2 stage PTC with more than five positive lymph nodes. The present study revealed that TT did not confer improved OS and CSS compared to lobectomy, which is similar to several previous studies14,15. Additionally, this study endpoints were OS and CSS, which was a supplement to the previous study endpoints of RFS. Although 63.01% of patients in the TT group in our study did not receive RAI ablation, which might have potentially influenced the survival results, we think the 2 groups are comparable based on the conflicting results of the previously mentioned studies.
Moreover, when we performed further stratified analyses in patients treated with lobectomy and TT plus RAI ablation, no significant difference was found in OS and CSS, which conformed with result from the whole cohort. Certainly, the stratified comparison did not have enough power to examine the effect of RAI ablation on tumor recurrence subject to the limitation of sample size and case selection. Further study is needed on this topic.
There are some previous stduies reported that no advantages of TT over lobectomy for intermediate-risk PTC. A study by Matsuura et al. of 3756 patients with low-intermediate risk node-negative thyroid cancer found no significant difference in 10-year recurrence-free survival (RFS) between lobectomy and TT (99.5% vs. 98.3%; P = 0.08) after adjusting for various factors16. Similarly, Adam et al. analyzed data from 61,775 patients with papillary thyroid cancer and found that the extent of surgery did not independently affect survival, regardless of the presence of extrathyroidal extension, TNM staging, or distant metastasis17.
In our study, PTC patients with more than five positive LNM had a similar OS and CSS after lobectomy vs. those who had TT after PSM. However, Bilimoria et al.18 first to demonstrate that TT results in lower recurrence rates and improved survival for PTC > or = 1.0 cm compared with lobectomy using data of 52,173 PTC patients from National Cancer Data Base (1985–1998). Suman et al. also19 reported that patients with cN1, lymphovascular invasion, ETE, positive margins, poorly differentiated PTC, and M1 disease in the TT group maintained a survival advantage compared with those in the lobectomy group after PSM (5 years: 96.5% vs. 94.7% and 10 years: 90.9% vs. 88.5%, respectively, P = 0.03). The differences could be due to several factors, including inclusion and exclusion criteria, differences between the 2 groups, the criteria used to define disease recurrence, and possibly statistical methods.
There are several limitations in the present study. First, the rate of recurrence of PTC may be a better indicator to evaluate the effect of surgical procedures. However, because the SEER database does not include information on the recurrence of diseases, differences in recurrence rates between the two groups cannot be examined. Thus, further prospective studies would be needed to answer this question. Second, due to the retrospective nature of the SEER database, missing information could not be verified, and thus selective bias was unavoidable. In addition, since the size of lymph node was not clarified in the SEER database, there may be an undetected effect on the prognosis analysis.
Conclusions
In conclusion, the findings of this cohort study indicate that patients with T1 and T2 stage PTC who have more than five positive lymph node metastases have no additional survival benefit of TT over lobectomy after PSM. These results imply that the presence of more than five positive lymph node metastases should not be considered an absolute indication for TT. In cases where RAI ablation is not planned, lobectomy may serve as an effective alternative for appropriately selected patients, potentially optimizing their quality of life.
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Code availability
The software application generated during and/or analyzed during the urrent study are available from the corresponding author on reasonable request.
Change history
05 March 2026
The original online version of this Article was revised: In the original version of this Article the Tables were ordered incorrectly. The original article has been corrected.
References
Chen, W. et al. Cancer statistics in China, 2015. Ca-Cancer J. Clin. 2, 115–132 (2016).
Tuttle, R. M. Controversial issues in thyroid cancer management. J. Nucl. Med. 8, 1187–1194 (2018).
SiegelRL, MillerKD, WagleNS & Jemal, A. Cancer Statistics, 2023. Ca-Cancer J. Clin. 1, 17–48 (2023).
Megwalu, U. C. & Moon, P. K. Thyroid cancer incidence and mortality trends in the united states: 2000–2018. Thyroid 5, 560–570 (2022).
Haugen, B. R. et al. 2015 American thyroid association management guidelines for adult patients with thyroid nodules and differentiated thyroid cancer: the American thyroid association guidelines task force on thyroid nodules and differentiated thyroid cancer. Thyroid 1, 1–133 (2016).
Chen, D. W., Lang, B., McLeod, D., Newbold, K. & Haymart, M. R. Thyroid cancer. Lancet 10387, 1531–1544 (2023).
Pasqual, E. et al. Trends in the management of localized papillary thyroid carcinoma in the united States (2000–2018). Thyroid 4, 397–410 (2022).
Sanabria, A. et al. Current therapeutic options for Low-Risk papillary thyroid carcinoma: Scoping evidence review. Head Neck-J Sci. Spec. 1, 226–237 (2022).
Haddad, R. I. et al. NCCN guidelines insights: Thyroid Carcinoma, version 2.2018. J. Natl. Compr. Canc Ne. 12, 1429–1440 (2018).
Nam, S. H. et al. Nodal factors predictive of recurrence after thyroidectomy and neck dissection for papillary thyroid carcinoma. Thyroid 1, 88–95 (2018).
Jianyong, L. et al. A nomogram based on the characteristics of metastatic lymph nodes to predict papillary thyroid carcinoma recurrence. Thyroid 3, 301–310 (2018).
Lamartina, L., Durante, C., Filetti, S. & Cooper, D. S. Low-Risk differentiated thyroid cancer and radioiodine remnant ablation: A systematic review of the literature. J. Clin. Endocr. Metab. 5, 1748–1761 (2015).
Ruel, E. et al. Adjuvant radioactive iodine therapy is associated with improved survival for patients with Intermediate-Risk papillary thyroid cancer. J. Clin. Endocr. Metab. 4, 1529–1536 (2015).
Xu, S. et al. Comparison of lobectomy vs total thyroidectomy for Intermediate-Risk papillary thyroid carcinoma with lymph node metastasis. Jama Surg. 1, 73–79 (2023).
Nixon, I. J. et al. The results of selective use of radioactive iodine on survival and on recurrence in the management of papillary thyroid cancer, based on memorial Sloan-Kettering cancer center risk group stratification. Thyroid 6, 683–694 (2013).
Matsuura, D. et al. Surgical management of Low-/Intermediate-Risk node negative thyroid cancer: A Single-Institution study using propensity matching analysis to compare thyroid lobectomy and total thyroidectomy. Thyroid 1, 28–36 (2022).
Adam, M. A. et al. Extent of surgery for papillary thyroid cancer is not associated with survival: An analysis of 61,775 patients. Ann. Surg. 4, 601–605 (2014).
Bilimoria, K. Y. et al. Extent of surgery affects survival for papillary thyroid cancer. Ann. Surg. 3, 375–381 (2007).
Suman, P. et al. Thyroid lobectomy for T1B-T2 papillary thyroid cancer with High-Risk features. J. Am. Coll. Surg. 1, 136–144 (2020).
Funding
This work received financial support from the Chengdu High-level Key Clincal Specialty Construction Projiect.
Author information
Authors and Affiliations
Contributions
J.H.H and J.Y.L designed the study, performed further data analysis, and drafted the manuscript. J.Q.D and J.Y.T interpreted the data and assisted in writing. Y.L and Y.G assisted in writing and revising the manuscript. Z.Y.Y and J.Y.T performed data extraction and initial analysis. All authors have read and agreed to the published version of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval
The protocol for this study was approved by the Ethics Committee of Chengdu Fifth People’s Hospital (The Second Clincal Medical College, Affiliated Fifth People’s Hospital of Chengdu University of Traditional Chinese Medicine).
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Huang, J., Tan, J., Guo, Y. et al. Comparative survival outcomes of lobectomy versus total thyroidectomy in T1 and T2 papillary thyroid cancer with more than five positive lymph nodes. Sci Rep 16, 1445 (2026). https://doi.org/10.1038/s41598-025-31110-1
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-31110-1