Abstract
Acinetobacter baumannii (A. baumannii) is a pathogen that opportunistically infects patients in healthcare settings and poses a significant threat to human health due to its widespread resistance to antimicrobial agents. The temporal and regional distribution of A. baumannii in China is continually evolving, necessitating comprehensive surveillance to examine the prevalence, molecular characteristics, and clonal relationships of antimicrobial resistance determinants in burn wound A. baumannii isolates collected from six major Chinese provinces during 2016–2020. To address this need, we analyzed a total of 415 distinct A. baumannii isolates that were obtained from burn patients. Antimicrobial susceptibility testing was performed using the Kirby Bauer disk diffusion method, while genetic relatedness of the 415 isolates was assessed by utilizing multilocus sequence typing (MLST) and eBURST analysis. Additionally, the housekeeping genes and a comprehensive panel of drug resistance genes, including β-lactamases, membrane permeability factors, and efflux pump systems, were detected by multiplex PCR to elucidate their resistance mechanisms. Our analysis revealed that among the 415 isolates, 384 (92.5%) were classified as multidrug-resistant (MDR), with regional rates ranging from 88.2 to 96.0%. Additionally, 38 (9.2%) isolates were classified as extensively drug-resistant (XDR). All strains showed resistance to multiple antibiotics, except for lower resistance to Tigecycline (TGC) and Cefoperazone/sulbactam (CSL), suggesting their potential therapeutic utility. By MLST, all isolates represented 122 identified STs (sequence types). eBURST analysis identified the 415 isolates into 17 clonal complexes (CCs) in which CC1660 and CC1417 were considered large CCs with at least 100 isolates in each CC. Importantly, our investigation identified a notable epidemiological shift: CC1417 and CC1660 instead of CC92 have become the novel predominant strains in burn wound isolates of these Chinese provinces, and the predominant strain varied in separate regions. Consistently, our investigation identified ST1417, ST1660, and ST1145 instead of ST208 as predominant sequence types, indicating a notable shift in regional epidemiological patterns for burn wound A. baumannii isolates. In addition, the carrying rates of multiple antibiotic-resistant genes maintain a high level, indicating the evolution of strain genes and the severe drug resistance situation in burn wound isolates of these Chinese provinces. As a result, epidemiological surveillance and genetic evolution analysis of A. baumannii is of great significance to provide more strategies for the prevention of nosocomial infection and clinical treatment.
Similar content being viewed by others
Introduction
Acinetobacter baumannii (A. baumannii), a strictly aerobic opportunistic pathogen, has emerged as a formidable threat for burn patients1,2. In burn patients, A. baumannii demonstrates high nosocomial infection rates and frequently co-infects with other pathogens, resulting in difficult-to-treat infections associated with prolonged hospitalization and increased mortality3,4. Additionally, a significant public health issue regarding A. baumannii infection is detecting an increasing resistance rate to multiple antibiotics5. With the inappropriate use of antibiotics, the rising incidence of antimicrobial resistance has become an alarming phenomenon in clinical work6, as it limits the therapeutic efficacy of antibiotics. Therefore, continuous epidemiological monitoring and infection analysis are of great significance for the study of treatment and control strategies for A. baumannii infection in burn patients7. The resistance mechanisms of A. baumannii include β-lactamases, modifications of target sites, permeability defects, efflux pumps, and aminoglycoside-modifying enzymes8. Of them, β-lactamase is the primary mechanism9. β-lactamase enzymes hydrolyze the β-lactam ring in antibiotics, resulting in antibiotic inactivation and bacterial resistance10. In addition, bacteria possessing efflux pump genes can expel antibiotics, which is associated with many different kinds of drug resistance, especially imipenem7,11. Previously, carbapenems served as the cornerstone of A. baumannii treatment, owing to their high efficiency and low toxicity12. However, recent surveillance data indicates an alarming increase in carbapenem-resistant isolates, particularly in the ICU ward. Imipenem (IPM) and meropenem (MEM) were the two most effective carbapenem antibiotics, but studies showed their drug-resistance situation was tricky13. A 2021 report collected 94 isolates from 2016 to 2019, of which 88 (93.6%) were resistant to imipenem or meropenem, with resistance rates showing a consistent upward trajectory14. The exceptional genomic plasticity of A. baumannii facilitates horizontal gene transfer of these resistance determinants, enabling rapid acquisition of novel resistance mechanisms15.
Multilocus sequence typing can exactly show the characteristics of bacteria through housekeeping gene fragments16, and an ST identifier is composed of seven housekeeping gene loci. Its remarkable success in global epidemiological investigation makes it the most popular typing method16,17. Studies have shown that the molecular type distribution of A. baumannii is geographically related18. In China, the prevalence of CC92 clones is widespread across most provinces, indicating the significant spread of this particular clone of A. baumannii19.
Extensive research has been conducted to investigate the molecular epidemiology of this pathogen20,21. However, the epidemiology of A. baumannii in certain regions may offer new insights into its behavior and spread in the coming years. Further studies in these regions could contribute to our understanding of the disease and inform future strategies for its control and prevention.
The emergence and spread of multidrug-resistant (MDR) A. baumannii pose a significant threat to public health worldwide. In recent years, the epidemiology and resistance patterns of this bacterium have been extensively studied, with notable research conducted in different regions, including Asia. While previous studies, such as the ones by Wohlfarth et al.22 in Germany and Nodari et al.23 in South America, have shed light on the epidemiological landscape of MDR A. baumannii, the need to incorporate the latest findings from Asia, particularly China, remains pivotal. Recent research by Mo et al.24 unveiled the alarming presence of blaNDM-1 harboring MDR Acinetobacter bereziniae isolates within South China’s hospital settings, emphasizing the pressing need for comprehensive surveillance and intervention strategies in the region. Furthermore, extensive surveillance studies in China have documented the widespread distribution of OXA-type carbapenemases, with blaOXA-23 being the predominant resistance mechanism carried within Tn2007 transposons across different provinces1. Additionally, large-scale genomic analyses have revealed that carbapenem-resistant A. baumannii (CRAB) shows an isolation rate of approximately 70% in Chinese hospitals, with these strains showing consistent susceptibility only to polymyxins and tigecycline1.
Given these pressing concerns and the unique vulnerability of burn patients to A. baumannii infections2, this study seeks to explore the contemporary landscape of A. baumannii burn wound isolates, particularly emphasizing the latest regional data from six major Chinese provinces. Molecular epidemiological studies have demonstrated that A. baumannii exhibits substantial regional heterogeneity due to multiple interconnected factors including geographical isolation, distinct transmission pathways, antibiotic selection pressures, and continuous bacterial evolution25. These factors collectively contribute to region-specific dominant clonal types and resistance patterns across different healthcare settings. The epidemiology analysis of A. baumannii can help demonstrate the current clinical situation of ST typing, resistance characteristics, regional distribution differences, and genotype characteristics for further precision medicine treatments.
Material and methods
This retrospective study was carried out under the approved guidelines of the Ethics Committee of Xinqiao Hospital and in accordance with the Declaration of Helsinki. No written informed consent was required as we received anonymized isolate samples with all the personal information removed.
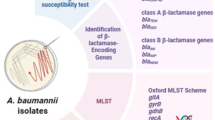
Clinical isolate collections of A. baumannii
Burn wound isolates of A. baumannii were collected from burn department of central hospital in 6 Chinese provinces between 2016 and 2020, through their active surveillance system, including Jilin (123/415), Guangdong (25/415), Henan (112/415), Shaanxi (88/415), Jiangsu (17/415), and Chongqing (50/415) (detailed information in Fig. 1). These provinces represent different geographical regions of China (north eastern, south western, eastern and south eastern), which together contain over 93% of China’s population according to the population distribution pattern defined by the Hu Huanyong Line26. Isolates were identified using standard microbiological methods including colony morphology, Gram staining, and biochemical tests27. Species identification was confirmed using MALDI-TOF mass spectrometry or 16S rRNA gene sequencing when necessary. Genomic DNA was extracted from overnight cultures using the TIANamp Bacteria DNA Kit (TIANGEN, Beijing), with quality verified by A260/A280 ratios (1.8–2.0) and 16S rRNA amplification28. The strains were stored at − 80 °C in glycerol broth for long-term preservation. For experimental procedures, isolates were retrieved from frozen stocks and cultured overnight in LB medium (Beyondtime, China) at 37 °C with shaking at 180 rpm for further studies. Resistance phenotype classification was performed according to standardized criteria. Multidrug-resistant (MDR) isolates were defined as those showing resistance to ≥ 3 antimicrobial categories with inhibition zone diameters ≤ 14 mm. Extensively drug-resistant (XDR) isolates were defined as those resistant to all tested antibiotics except 1–2 categories, determined by standard disk diffusion breakpoints.
Geographical distribution of all A. baumannii strains. (A) The highlighted area on the map is the origin of 415 quarantine points. Including Chongqing, Guangdong, Jilin, Henan, Shaanxi, and Jiangsu. (B) Pie chart distribution of 415 isolates in each province.
Multilocus sequence typing
The molecular subtypes of all A. baumannii isolates were subjected to MLST by PCR amplification and sequencing of seven housekeeping genes (gltA, gdhB, gyrB, recA, gpi, cpn60, and rpoD) as described by the Oxford scheme introduced by Bartual29,30. To carry out PCR amplification, one microliter of DNA was added to 20 µL of the reaction mixture, which contains 10 µMol/L of each primer and 10 µL of 2xTaq premix (TIANGEN, Beijing). Then PCR was performed, and 5% DMSO (Sangon Biotech, Shanghai) should be added to the reaction if necessary. The reaction products were further separated and detected on an ABI PRISM genetic analyzer 3100 (Applied Biosystems, Waltham, MA). An identifier of the ST gene was constructed by combining 7 loci, and the sequencing results of each gene were compared with the known sequences in the PubMLST database (https://pubmlst.org/). The primers used in updated Oxford MLST scheme are listed in Table S1.
eBURST and phylogenetic analysis
Following MLST analysis, goeBURST (http://www.phyloviz.net/goeburst/) was used for data grouping analysis31. All reported MLST data were queried from the PubMLST database (https://pubmlst.org/). Clonal complexes (CCs) were first assigned using the classical MLST definition, in which STs sharing 6 out of 7 housekeeping alleles were grouped into the same CC. For each ST detected in this study, the complete allelic profiles deposited in PubMLST were retrieved and used as the reference for CC determination.
Besides, goeBURST was then applied to construct the single-locus-variant (SLV) network. Unlike the 6/7-allele rule used for CC classification, the SLV-based goeBURST network links STs differing at only one allele, thereby illustrating evolutionary connections among STs. Thus, CC assignment and SLV network construction were complementary but relied on different clustering principles. After the multisequence alignments with ClustalX2 (http://www.clustal.org/clustal2/)32, an independent maximum-likelihood phylogenetic tree was generated using PhyML (http://www.atgc-montpellier.fr/phyml/)33. This phylogenetic analysis was performed to validate whether the ML-based evolutionary relationships were consistent with the population structure inferred from MLST and goeBURST.
Antimicrobial susceptibility testing
Antimicrobial susceptibility was performed by the Kirby Bauer disk diffusion method for nine antibiotics34, including sulfamethoxazole (SXT), Ciprofloxacin (CIP), Gentamicin (GEN), Ceftazidime (CAZ), Cefoperazone/sulbactam (CSL), Cefotaxime (CTX), Imipenem (IPM), Piperacillin (PIP) and Tigecycline (TGC). The selection of these nine antibiotics was based on: (1) current evidence-based therapeutic approaches for multidrug-resistant Acinetobacter baumannii infections, as outlined in international treatment guidelines35; (2) recommendations from the Chinese guidelines for carbapenem-resistant Gram-negative bacilli infections36; and (3) clinical relevance, including both conventional first-line agents and last-resort antibiotics commonly used in Chinese healthcare settings. According to the Clinical and Laboratory Standards Institute guidelines (CLSI 2017, M100-S27), the drug resistance of bacteria can be judged by comparing the diameter and breakpoint of the zone diameter37. The MIC of cefoperazone/sulbactam and tigecycline refers to the opinions of Sader HS et al.38 and Jones et al.39.
PCR-based screening for antimicrobial-resistant genes
Genomic DNA was extracted from overnight cultures using the TIANamp Bacteria DNA Kit (TIANGEN, Beijing), with quality verified by A260/A280 ratios (1.8–2.0) and 16S rRNA amplification. The 21 resistance genes were divided into 3 groups and detected by multiplex PCR40. Group1 included the β-Lactamases which can be divided into four categories: class A with blaGES, blaKPC, blaPER, blaCTX, class B with blaIMP, blaVIM, blaSIM, blaNDM, class C with blaAMP, and class D with blaOXA. Group 2 included the permeability defects with carO, ompW, and omp33-66. Group 3 included the efflux pumps with Ade. The primers used in screening these resistance genes are listed in Table S2. The conditions for PCR reactions were as follows : pre-denaturation at 95 °C for 5 min, followed by 35 cycles of denaturation at 95 °C for 1 min, annealing at 55 °C for 1 min, and elongation at 72 °C for 1 min, and a final extension step at 72 °C for 15 min.
Results
MLST and population structure analysis
Four hundred and fifteen isolates were subjected to typing using the updated Oxford Bartual scheme of MLST. The isolates were divided into 122 STs, reflecting the high clonal diversity of A. baumannii (Table 1). The study found that the six most prevalent STs of A. baumannii isolates included ST1417 (45/415, 10.8%), ST1660 (36/415, 8.7%), ST1145 (32/415, 7.7%), ST1661 (26/415, 6.3%), ST1646 (22/415, 5.3%), and ST1679 (20/415, 4.8%), collectively representing 43.6% of all isolates (Tables 1 and S3). Notably, we observed distinct regional patterns in ST distribution (Fig. 2). ST1417 dominated in Henan (18/112, 16.1%) and Shaanxi (15/88, 17.0%) provinces, while ST1679 was also prevalent in Henan (16/112, 14.3%) (Fig. 2). Jilin Province showed high prevalence of ST1660 (11/123, 8.9%) and ST1145 (15/123, 12.2%) (Fig. 2). In Shaanxi, ST1660 (14/88, 15.9%) emerged as another major strain alongside ST1417 (Fig. 2). Jiangsu exhibited co-dominance of ST1660 (5/17, 29.4%) and ST1661 (5/17, 29.4%), while Chongqing showed a unique predominance of ST368 (14/50, 28.0%) (Fig. 2).
Regional distribution of predominant A. baumannii sequence types across six Chinese provinces. Pie charts show the percentage distribution of major STs in each province. Numbers in parentheses indicate total isolates per province. “Others” includes remaining STs with lower frequencies. Maps show partial regions of China covering the six study provinces.
eBURST analysis
Using the classical MLST definition (STs sharing 6 of the 7 alleles), the 122 STs identified in this study were assigned into 17 clonal complexes based on their full PubMLST allelic profiles. Consistent with established nomenclature, several STs detected in this study (e.g., ST195, ST208, and ST368) belonged to the literature-defined CC92 lineage, even though ST92 itself was not detected among our isolates. Geographical comparison of the major CCs revealed distinct regional patterns. CC1417 was widely distributed across the sampled provinces (Fig. 3A), whereas CC1660 predominated in four provinces (Fig. 3B). CC92 exhibited strong regional specificity, with most isolates concentrated in Chongqing (Fig. 3C). Additionally, because ST1145 was one of the more common sequence types in our dataset (32/415, 7.7%), we examined its geographical distribution (Fig. 3D). ST1145 showed higher prevalence in Jilin Province (15/123, 12.2%) and was absent in Guangdong. This pattern indicates unequal distribution across provinces.
eBURST analysis of 415 Acinetobacter baumannii isolates. (A–D) Geographical distribution maps of major clonal complexes and sequence type across six Chinese provinces. Maps show isolate density by province. (A) CC1417 distribution. (B) CC1660 distribution. (C) CC92 distribution. (D) ST1145 distribution. Maps show partial regions of China covering the six study provinces. (E) Relationships among the STs were found in this study. Connections between STs indicate single-locus variants (SLVs) differing by one allele at a single locus. The blue dot denotes the founder, and the yellow dots represent the sub-founder. The size of the dot indicates the number of isolates. Central node ST1646 corresponds to CC1417, central node ST1665 corresponds to CC1660, and central node ST195 corresponds to CC92. This network demonstrates the close evolutionary relationships within each clonal complex (CC), with each CC displaying a star-like radiation pattern from central founder STs to peripheral derivative STs.
To further explore the evolutionary relationships among STs, the goeBURST algorithm was applied to construct a single-locus-variant (SLV) network. As goeBURST relies on one-allele differences rather than the 6/7 shared-allele criterion used for CC assignment, it provides complementary information by resolving evolutionary relationships. The SLV network identified three major founder-centered clusters: CC1417 (centered on ST1646), CC1660 (centered on ST1665), and the CC92 lineage (centered on ST195), illustrating the diversification pathways and genetic structure within A. baumannii populations (Fig. 3E). Finally, to independently validate the evolutionary structure inferred from MLST and the goeBURST network, a phylogenetic tree was generated using PhyML. The phylogeny showed clustering patterns consistent with the CC assignments and the SLV-based network, with no conflicting topologies, supporting the robustness of the inferred population structure.
Antimicrobial susceptibility profiles
To evaluate antimicrobial resistance profiles, we tested the isolates against nine clinically relevant antibiotics. The results revealed consistently high resistance rates across most antibiotics: ciprofloxacin (CIP, 91.57%, 380/415), gentamicin (GEN, 88.92%, 369/415), piperacillin (PIP, 90.84%, 377/415), trimethoprim/sulfamethoxazole (SXT, 82.65%, 343/415), ceftazidime (CAZ, 91.08%, 378/415), cefotaxime (CTX, 91.57%, 380/415), and imipenem (IPM, 90.60%, 376/415) (Table 2). Notably, lower resistance rates were observed for tigecycline (TGC, 37.35%, 155/415) and cefoperazone/sulbactam (CSL, 69.40%, 288/415) (Table 2). Compared with the isolates from Jiangsu, strains from Chongqing, Guangdong, Henan, Shaanxi, and Jilin showed higher resistance to most antibiotics (Table 2). Of particular clinical significance was the finding that four provinces exhibited IPM resistance rate exceeding 90% (Table 2). Furthermore, it has been observed that Jilin province exhibited the highest prevalence of TGC resistance, with a rate of 76%, which was three to nine times that of the other five regions (Table 2). The bacterial isolates originating from Jilin province have demonstrated a high level of resistance towards various antibiotics with a 100% resistance rate towards CIP and CAZ, and above 88% towards other antibiotics. Based on the comprehensive resistance patterns observed, 384 isolates (92.5%) met MDR criteria (Table 2). Guangdong showed the highest MDR rate (96.0%), while Jiangsu demonstrated the lowest (88.2%). Furthermore, 38 isolates (9.2%) exhibited XDR phenotypes. Guangdong had the highest XDR prevalence (12.0%), followed by Shaanxi (11.7%). Chongqing showed the lowest XDR rate (4.0%) (Table 2). Furthermore, our investigation has revealed the differences in antimicrobial resistance rates among major A. baumannii lineages. While the remaining five clones displayed a generally elevated drug resistance, the resistance rate of ST1679 was notably elevated in contrast to the others (Table 3). These six clones demonstrated a high degree of resistance towards a majority of antibiotics, yet exhibited a comparatively lower resistance towards TGC (Table 3).
Detection of resistance genes
The mechanisms and genes of drug resistance of carbapenems mainly include β-lactamase, multidrug efflux pump, and permeability defects. Through a comprehensive literature review, we identified 21 resistance genes. Interestingly, these were screened in all isolates by PCR. These genes were functionally categorized into three corresponding mechanisms: β-lactamase-encoding genes (including class A, B, C, and D β-lactamases), efflux pump-associated genes, and genes related to permeability defects.
For the top six STs, all of these isolates carried blaOXA-58, blaIMP, blaAmpC, and carO (Table 4). However, the carrying rates of β-lactamase blaGES-14, blaCTX-M-2, blaKPC, and blaSIM-1 varied significantly. From the perspective of regional distribution, all 415 isolates carried four genes: blaAmpC, blaOXA-58, blaIMP, and carO (Table 5). Meanwhile, over 85% of the strains from each province carried the following genes: blaVIM, blaOXA-23, blaOXA-51, omp33-36, adeA, adeB, adeC, adeI and adeJ (Table 5). Only ompW and blaSIM-1 had relatively low carrying rates (Table 5). The carrying rates of efflux pump class, carO and omp33-36 of permeability defects, blaAmpC, blaIMP, blaNDM, blaOXA-58, blaOXA-23, and blaOXA-51 of β-lactases were generally high in six provinces (Table 5). There were substantial regional differences in the individual carrying rate of resistance genes. The proportion of the strains carrying ompW in Shaanxi was 76%, which was two to three times that of the other five regions (Table 5). Meanwhile, the blaSIM-1 carriage rate in Henan was considerably lower than in other regions. The blaCTX-M-2 carrying rate is low in Jilin, Jiangsu and Shaanxi while the rate in the other provinces was more than 90% (Table 5). These findings suggest complex regional differences in selection pressures and resistance evolution patterns.
Discussion
Acinetobacter baumannii (A. baumannii), an opportunistic pathogen, mainly causes hospital-acquired infection, and it has been considered a critical-priority pathogen by the World Health Organization. An open pan-genome makes obtaining new genes and functions easier for A. baumannii. To characterize the regional distribution patterns and genetic evolution of A. baumannii and provide more references for the application of therapeutic antibiotics, the current study analyzed the molecular epidemiology of 415 A. baumannii isolates obtained in China from 2016 to 2020, including drug resistance, drug-resistant genes, and STs. These isolates were recovered from 6 provinces in China, including Jilin, Guangdong, Henan, Shaanxi, Jiangsu, and Chongqing. These provinces encompass the eastern, western, southern, and northern territories, encapsulating the geographical features prevalent throughout the nation41. This extensive coverage enables an in-depth exploration of regional variations and the influence of geographical environments on A. baumannii prevalence. Additionally, the six provinces demonstrate notable differences in population structure, economic development, healthcare standards, and infection control policies related to drug-resistant bacteria treatment42,43. These human-associated differences are well-documented in previous studies, which have shown significant regional disparities in health service development across Chinese provinces, with eastern regions (Guangdong and Jiangsu in our study) consistently demonstrating higher levels in healthcare personnel, facilities, and service provision compared to central (Henan and Shaanxi) and western regions (Chongqing)42,43. These variations are crucial for understanding the factors that contribute to the prevalence of A. baumannii.
Recently, extensively drug-resistant (XDR) and pan-drug-resistant (PDR) A. baumannii isolates have rapidly increased44. Consistent with this trend, antimicrobial susceptibility testing showed that 92.5% and 9.2% of burn wound A. baumannii isolates were MDR and XDR, respectively, posing significant therapeutic challenges. These findings align with previous studies in China, where carbapenem resistance rates reached 91–94% in Chongqing burn ICU (2018–2020)56 and 92.3% in burn-related bloodstream infections2.
Regional analysis revealed varying resistance patterns across provinces. Guangdong showed the highest MDR (96.0%) and XDR (12.0%) rates, while Jiangsu and Chongqing reported the lowest MDR (88.2%) and XDR (4%) rates, respectively (Table 2). These differences may reflect variations in healthcare infrastructure, antibiotic usage, and infection control practices42,43, though specific determinants require further investigation. Among the tested antimicrobials, most A. baumannii isolates showed high resistance to tested antibiotics, except for tigecycline (TGC) and cefoperazone/sulbactam (CSL) (Table 2). This finding is particularly significant given the limited therapeutic options available for MDR A. baumannii infections. The resistance mechanisms and genes of A. baumannii include β-lactamases, modifications of target sites, permeability defects, efflux pumps, and aminoglycoside-modifying enzymes. β-lactamase genes encode β-lactamase enzymes that hydrolyze the β-lactam ring in antibiotics, resulting in antibiotic inactivation and conferring β-lactam resistance to A. baumannii. Different countries have reported that class D contributes the most to drug resistance β-lactamase in recent years, and the prevalence of class D in China can reach 90%, especially in coastal areas45. Through a comprehensive literature review, we identified 21 resistance genes spanning β-lactamases, membrane permeability factors, and efflux pump systems46,47. These genes were screened in all isolates by PCR. The systematic categorization of these resistance determinants allowed us to examine the complex interplay between different resistance mechanisms.
A previous study has shown a significant correlation between the overexpression of the AdeABC and AdeIJK efflux pumps and resistance to tigecycline and minocycline48. Despite, the high prevalence of efflux pump genes in the 415 strains, they remained susceptible to Tigecycline (TGC), suggesting that the efflux pumps in these strains may not be involved in resistance to TGC, or their expression levels may not be sufficient to confer significant resistance. Alternatively, these strains may possess other susceptibility mechanisms, such as unknown resistance inhibitory factors, or TGC may enter the cells in a manner that is not affected by these efflux pumps. Further studies are needed to confirm these possibilities and fully understand the complex interplay between efflux pump gene carriage and antibiotic susceptibility in these strains.
Here, MLST analysis was performed using the updated Oxford scheme primers as described by Hamidian et al. (2017) to avoid previous misidentification issues associated with the original primers, particularly for the gpi gene49. This revision is specifically important for accurate identification of sequence types, notably the correct identification of ST208 which was previously misidentified as ST92 due to primer design issues. While the updated Oxford MLST scheme has certain limitations, particularly regarding homologous recombination effects, it remains valuable for differentiating isolate relatedness within clonal groups. Although the Pasteur scheme is increasingly preferred for clonal group classification, the Oxford scheme’s established presence in historical datasets makes it useful for comparative analyses across studies.
In our analysis, we employed the updated Oxford scheme to maintain consistency with existing literature and facilitate broader comparability of our findings. Due to the different environments and antibiotic selection, time and regional differences may exist in the prevalence of STs. Previous epidemiological data indicated that ST208 (initially misreported as ST92 due to limitations in the original Oxford MLST scheme) predominated in China before 2010, with ST208 and ST195 continuing to be the major epidemic types of MDR A. baumannii in recent years1,50. Regional variations in strain dominance were observed across different geographical locations, with ST208 and ST368 being prevalent in Chongqing, while ST208 (documented as ST92 in earlier studies) was predominantly identified in Henan51,52. Notably, a 12-year longitudinal epidemiological study of A. baumannii in a tertiary hospital documented a temporal shift in strain dominance: the originally prevalent ST208 and ST369 clones were replaced by ST451 in 201953.Consistently, our current investigation identified ST1417, ST1660, and ST1145 as emerging dominant sequence types, indicating a notable shift in regional epidemiological patterns. These findings collectively suggest an ongoing clonal replacement phenomenon among predominant A. baumannii strains in China. This dynamic evolutionary pattern underscores the remarkable genomic plasticity of A. baumannii populations across spatial and temporal dimensions.
While our collected main ST types (ST1417, ST1660, and ST1145) are different from a recent epidemiological study (2011–2021) focusing on A. baumannii blood isolates (ST208 and ST191 as dominant)1, the difference exemplifies the complex, multifactorial nature of A. baumannii molecular epidemiology. Previous research has demonstrated that regional variations in A. baumannii epidemiology result from the combined effects of geographical isolation, diverse transmission pathways, differential antibiotic selection pressures, and continuous bacterial evolution25. Beyond these established factors, we propose that clinical source materials may represent an additional determinant of strain distribution patterns. The referenced study primarily sampled from Zhejiang (40.23%) and Anhui (19.99%) provinces1, excluding Chongqing and Jilin, which were key regions in our investigation. Moreover, their samples were blood isolates1 rather than wound samples. However, the differences also suggest that A. baumannii evolves rapidly, highlighting the need for epidemiological surveillance. These distinctions in geographical coverage, sampling density, and strain sources account for the differences in our findings.
Previous studies on A. baumannii isolates from burn patients in China have documented distinct epidemiological patterns. In a burn ICU study in Chongqing (2013), ST368 emerged as the dominant sequence type, along with ST369, ST195, and ST19154. Similarly, research on burn-related bloodstream infections at the Chinese Burn Institute identified ST368 as the most prevalent sequence type (38%)2. These studies consistently reported high carbapenem resistance rates (91–94%) in burn isolates2,54. In contrast, our current investigation identified ST1417, ST1660, and ST1145 as the predominant sequence types in burn wound isolates, suggesting temporal and geographical variations in dominant clones. This further emphasizes the critical importance of continuous epidemiological surveillance in tracking the dynamic shifts in dominant strains in burn infections across regions and time periods. The emergence of different predominant strains in burn infection in our study underscores the evolving nature of A. baumannii epidemiology and reinforces the necessity for sustained, long-term monitoring to effectively track these changes.
Regarding resistance gene profiles, the Chongqing burn ICU study detected blaOXA-51 (100%), blaOXA-23 (83.9%), blaVIM (70.9%), blaAmpC (80.6%), and blaPER (40.3%) in patient isolates54. Our study showed a similarly high prevalence of blaOXA-51 (100%) and blaOXA-23 (96%), but notably higher rates of blaVIM (98%), blaAmpC (100%), and blaPER (94%) (Table 5). Additionally, our isolates demonstrated widespread presence of blaNDM (94%) and blaIMP (100%), which were not detected in the earlier Chongqing study (Table 5). These differences likely reflect the temporal evolution of resistance mechanisms and the increasing prevalence of metallo-β-lactamases in Chinese hospitals over the past years.
Our study provides the first comprehensive evidence of CC1417 (34.7%) and CC1660 (24.8%) emergence as major clones in burn wound isolates from several provinces of China. CC92 has been a widely prevalent clonal complex globally55. A study reported that among 92 isolates collected in the United States from 2013 to 2017, the majority (n = 59, 64%) belonged to clonal complex 92 (CC92)56. The dominance of CC92 has been documented in epidemiological studies in Malaysia and Mexico as well57,58. However, in 2023, a study collected 57 carbapenem-resistant Acinetobacter baumannii strains, with more than half (56.1%, 32/57) belonging to CC455 and over one-third (38.6%, 22/57) belonging to CC92 59. Compared to previous studies, our study indicates that CC1417 and CC1660, rather than CC92, have become the predominant strains in burn wound isolates from several provinces of China. Moreover, it is noteworthy that CC92 remains dominant in Chongqing, which is also the main distribution area of all CC92 strains. This may reflect clonal stability or distinct local selection pressures in Chongqing. The geographical analysis of ST1145 shows uneven distribution across provinces (Fig. 3D). ST1145 was more frequently detected in Jilin (12.2%), detected at lower levels in Jiangsu, and not detected in Guangdong (Fig. 3D).
The epidemiological investigation of A. baumannii holds substantial importance for the advancement of precision medicine. While precision medicine is generally considered to be related to cancer detection and treatment, employing advanced genomic, epigenomic, and other omics technologies alongside targeted molecular drugs, it often encounters limitations due to socioeconomic disparities across regions60. Investigating the epidemiology of A. baumannii can reveal risk factors for infection in diverse regions, populations, and environments, thereby informing targeted preventive measures to mitigate infection risks in specific populations. This study predicts that the predominant infection type of A. baumannii burn wound isolates in Chongqing or Southwest China is CC92, indicating the need for increased attention and research on CC92 in this region. Drug resistance in A. baumannii may exhibit variation across different regions and populations. The carrying rates of blaSIM-1 from Henan isolates and blaCTX-M-2 from Jilin, Jiangsu and Shaanxi isolates were found to be low, indicating that their drug resistance may be less associated with β-lactamase pathway. Notably, the proportion of strains carrying ompW in Shaanxi Province reached 76%, which is two to three times higher than that of the other five regions. OmpW has been identified as a virulence factor in A. baumannii, and its absence compromises bacterial adhesion and invasion of host cells while reducing biofilm formation61. Of particular significance, previous studies have demonstrated that ompW gene expression is upregulated during the development of colistin resistance in A. baumannii62. Thus, research on OmpW and its associated drug therapies holds substantial implications for the treatment of A. baumannii infections in Shaanxi Province. These regional variations in resistance mechanisms likely reflect distinct local selection pressures and underscore the necessity for region-specific treatment strategies.
The present investigation has several notable limitations. First, this study focused exclusively on burn wound isolates, which may limit the generalizability of findings to other types of A. baumannii infections. The unique environment of burn units and the specific patient population may select for particular bacterial lineages that differ from those found in other clinical settings. Although data from the six provinces provides valuable insights into regional patterns, the sample scope needs further expansion to establish a more comprehensive national profile. Additionally, the unbalanced distribution of isolates among provinces (ranging from 17 to 123 isolates per province) may affect the representativeness of regional comparisons. Moreover, a significant methodological limitation lies in our reliance on PCR-based detection of resistance genes which only identifies target resistance determinants rather than a full spectrum of genetic elements that whole-genome sequencing might reveal40. Furthermore, it is noteworthy that the quantity of strains gathered in each provinces is relatively insufficient, and thus may not be entirely representative, reminding us that epidemiological surveillance and genetic evolution analysis of A. baumannii is of great importance to provide more insights for the prevention of nosocomial infection and clinical treatment.
Conclusion
This molecular epidemiological investigation reveals significant shifts in the landscape of A. baumannii burn wound infections in selected provinces of China. Our findings demonstrate that CC1417 (34.7%) and CC1660 (24.8%) have emerged as predominant clonal complexes in burn wound isolates from several provinces of China during the study, showing different distribution patterns compared to previously reported dominant CC92, with distinct regional distribution patterns. The concomitant identification of ST1417, ST1660, and ST1145 as prevalent sequence types in burn wound infections reinforces this epidemiological transition, with ST1145 showing higher detection in northern provinces, reflecting uneven regional distribution. Our analysis demonstrates high prevalence of antimicrobial resistance, with 92.5% of isolates classified as MDR and 9.2% as XDR. Significant regional variations in resistance patterns were observed across provinces, with MDR rates ranging from 88.2% to 96.0% and XDR rates from 4 to 12.0%. However, the relatively preserved susceptibility to tigecycline (TGC) and cefoperazone/sulbactam (CSL) offers potential therapeutic options. The high prevalence of multiple resistance genes, including β-lactamases, efflux pumps, and permeability-related genes, combined with significant regional variations in resistance mechanisms, highlights the dynamic evolution of this pathogen. Of particular concern is the regional specificity of certain resistance patterns, such as the elevated ompW carriage in Shaanxi (76%) and variable distribution of β-lactamase genes across provinces. These findings specifically apply to burn wound A. baumannii isolates in selected provinces and underscore the importance of continued surveillance and molecular typing in tracking the evolution and spread of resistant strains.
Data availability
Due to the nature of MLST data, which often contains numerous repeated sequences and minor variations that don’t significantly impact the final typing results, these sequence data are not typically stored in public databases. However, to ensure maximum transparency, we have uploaded our 415 isolates’ ST typing data to PubMLST at: https://pubmlst.org/bigsdb?db=pubmlst_abaumannii_isolates&l=1&page=query and data will be provided upon request from corresponding author (Email: zichenyang@tmmu.edu.cn).
Abbreviations
- A. baumannii :
-
Acinetobacter baumannii
- CAZ:
-
Ceftazidime
- CC:
-
Clonal Complex
- CIP:
-
Ciprofloxacin
- CSL:
-
Cefoperazone/sulbactam
- CTX:
-
Cefotaxime
- GEN:
-
Gentamicin
- ICU:
-
Intensive Care Unit
- IPM:
-
Imipenem
- LB:
-
Lysogeny broth
- MDR:
-
Multidrug-Resistant
- MEM:
-
Meropenem
- MIC:
-
Minimum Inhibitory Concentration
- min:
-
Minutes
- MLST:
-
Multilocus sequence typing
- PCR:
-
Polymerase Chain Reaction
- PIP:
-
Piperacillin
- PubMLST:
-
Public databases for Molecular Typing
- ST:
-
Sequence Type
- SXT:
-
Trimethoprim/sulfamethoxazole
- TGC:
-
Tigecycline
References
Luo, Q. et al. Genomic epidemiology and phylodynamics of acinetobacter baumannii bloodstream isolates in china. Nat. Commun. 16, 3536. https://doi.org/10.1038/s41467-025-58772-9 (2025).
Huang, G. et al. Epidemiological characterization of acinetobacter baumannii bloodstream isolates from a Chinese burn institute: A three-year study. Burns J. Int. Soc. Burn Injur. 42, 1542–1547. https://doi.org/10.1016/j.burns.2016.02.021 (2016).
Munier, A. L. et al. Incidence, risk factors, and outcome of multidrug-resistant acinetobacter baumannii acquisition during an outbreak in a burns unit. J. Hosp. Infect. 97, 226–233. https://doi.org/10.1016/j.jhin.2017.07.020 (2017).
Lee, C. R. et al. Biology of acinetobacter baumannii: Pathogenesis, antibiotic resistance mechanisms, and prospective treatment options. Front. Cell. Infect. Microbiol. 7, 55. https://doi.org/10.3389/fcimb.2017.00055 (2017).
Mandell GL, Douglas RG, Bennett JE. Mandell, douglas, and bennett’s principles and practice of infectious diseases: Elsevier/Churchill Livingstone, 2010.
Helmy, Y. A. et al. Antimicrobial resistance and recent alternatives to antibiotics for the control of bacterial pathogens with an emphasis on foodborne pathogens. Antibiotics (Basel, Switzerland) https://doi.org/10.3390/antibiotics12020274 (2023).
Duan, Z. et al. Nosocomial surveillance of multidrug-resistant acinetobacter baumannii: A genomic epidemiological study. Microbiol. Spectrum 12, e02207. https://doi.org/10.1128/spectrum.02207-23 (2024).
Chang-Ro, L. et al. Biology of acinetobacter baumannii: Pathogenesis, antibiotic resistance mechanisms, and prospective treatment options. Front. Cellular Infect. Microbiol. 7, 55 (2017).
Jeon, J. H. et al. Structure of adc-68, a novel carbapenem-hydrolyzing class c extended-spectrum β-lactamase isolated from acinetobacter baumannii. Acta Crystallogr D Biol Crystallogr. 70, 2924–2936 (2014).
Lin, M. F. & Lan, C. Y. Antimicrobial resistance in acinetobacter baumannii: From bench to bedside. World J. Clin. Cases WJCC 2, 28 (2014).
Muhsin, E. A., Sajid Al-Jubori, S. & Abdulhemid, S. L. Prevalence of efflux pump and porin-related antimicrobial resistance in clinical klebsiella pneumoniae in Baghdad Iraq. Archiv. Razi Inst. 77, 785–798. https://doi.org/10.2209/ari.2022.356976.1952 (2022).
Nguyen, M. & Joshi, S. G. Carbapenem resistance in acinetobacter baumannii, and their importance in hospital-acquired infections: A scientific review. J. Appl. Microbiol. 131, 2715–2738. https://doi.org/10.1111/jam.15130 (2021).
Høg B, Korsgaard H, Sönksen U et al. Consumption of antimicrobial agents and occurrence of antimicrobial resistance in bacteria from food animals, food and humans in Denmark. 2015
Shi, X. et al. Molecular characterization and antibiotic resistance of acinetobacter baumannii in cerebrospinal fluid and blood. PLoS ONE 16, e0247418. https://doi.org/10.1371/journal.pone.0247418 (2021).
Marino, A. et al. Unveiling the secrets of acinetobacter baumannii: Resistance, current treatments, and future innovations. Int. J. Mol. Sci. 25(13), 6814. https://doi.org/10.3390/ijms25136814 (2024).
Bartual, S. G. et al. Development of a multilocus sequence typing scheme for characterization of clinical isolates of acinetobacter baumannii. J. Clin. Microbiol. 43, 4382–4390 (2005).
Ruan, Z. et al. Wide distribution of cc92 carbapenem-resistant and oxa-23-producing acinetobacter baumannii in multiple provinces of china. Int. J. Antimicrob. Agents 42, 322–328 (2013).
Zahra, F. et al. Wide distribution of carbapenem resistant acinetobacter baumannii in burns patients in iran. Front. Microbiol. 6, 1146 (2015).
Chen, T. et al. Acinetobacter baumannii strains isolated from cerebrospinal fluid (csf) and bloodstream analysed by cgmlst: The dominance of clonal complex cc92 in csf infections. Int. J. Antimicrob. Agents 58, 106404. https://doi.org/10.1016/j.ijantimicag.2021.106404 (2021).
Jiang, M. et al. Molecular epidemiology of multi-drug resistant acinetobacter baumannii isolated in Shandong, China. Front. Microbiol. 7, 1687. https://doi.org/10.3389/fmicb.2016.01687 (2016).
Zhou, K. et al. An emerging clone (st457) of acinetobacter baumannii clonal complex 92 with enhanced virulence and increasing endemicity in South China. Clin. Infect. Dis. Off. Publ. Infect. Dis. Soc. Am. 67, S179–S188. https://doi.org/10.1093/cid/ciy691 (2018).
Wohlfarth, E. et al. The evolution of carbapenem resistance determinants and major epidemiological lineages among carbapenem-resistant acinetobacter baumannii isolates in germany, 2010–2019. Int. J. Antimicrob. Agents 60, 106689. https://doi.org/10.1016/j.ijantimicag.2022.106689 (2022).
Nodari, C. S. et al. Genomic analysis of carbapenem-resistant acinetobacter baumannii isolates belonging to major endemic clones in South America. Front. Microbiol. 11, 584603. https://doi.org/10.3389/fmicb.2020.584603 (2020).
Mo, X. M. et al. First identification of multidrug-resistant acinetobacter bereziniae isolates harboring bla ndm-1 from hospitals in south china. Heliyon. 9, e12365. https://doi.org/10.1016/j.heliyon.2022.e12365 (2023).
You, Q. et al. Local characteristics of molecular epidemiolgy of acinetobacter baumannii in jilin province (northeast china). BMC Microbiol. 23, 19. https://doi.org/10.1186/s12866-023-02761-9 (2023).
Li, M. et al. Study on population distribution pattern at the county level of China. Sustainability 10(10), 3598. https://doi.org/10.3390/su10103598 (2018).
York MK. 2010, Highlights of clsi document – abbreviated identification of bacteria and yeast - m35-a2, second edition. Clinical Microbiology Newsletter. 32:65–72 https://doi.org/10.1016/j.clinmicnews.2010.04.001
Schiebelhut, L. M. et al. A comparison of DNA extraction methods for high-throughput DNA analyses. Mol. Ecol. Resour. 17, 721–729. https://doi.org/10.1111/1755-0998.12620 (2017).
Chuang, Y.-C. et al. Microbiological and clinical characteristics of acinetobacter baumannii bacteremia: Implications of sequence type for prognosis. J Infect. 78, 106–112. https://doi.org/10.1016/j.jinf.2018.10.001 (2019).
Gaiarsa, S. et al. Comparative analysis of the two acinetobacter baumannii multilocus sequence typing (mlst) schemes. Front. Microbiol. 10, 930. https://doi.org/10.3389/fmicb.2019.00930 (2019).
Francisco, A. P. et al. Global optimal eburst analysis of multilocus typing data using a graphic matroid approach. BMC Bioinform. 10, 152. https://doi.org/10.1186/1471-2105-10-152 (2009).
Larkin MA, Blackshields G, Brown NP et al. Clustal w and clustal x version 2.0. Bioinformatics. 2007;23:2947–48
S G, JF D, V L et al. New algorithms and methods to estimate maximum-likelihood phylogenies: Assessing the performance of phyml 3.0. Systematic biology. 2010;59:307–21 https://doi.org/10.1093/sysbio/syq010
Ji, S. et al. Prevalence of carbapenem-hydrolyzing class d β-lactamase genes in acinetobacter spp. Isolates in China. Eur. J. Clin. Microbiol. Infect. Dis. Off. Publ. Eur. Soc. Clin. Microbiol. 33, 989–997. https://doi.org/10.1007/s10096-013-2037-z (2014).
Bouza, E., Muñoz, P. & Burillo, A. How to treat severe acinetobacter baumannii infections. Curr. Opin. Infect. Dis. 36, 596–608. https://doi.org/10.1097/qco.0000000000000974 (2023).
Zeng, M. et al. Guidelines for the diagnosis, treatment, prevention and control of infections caused by carbapenem-resistant gram-negative bacilli. J. Microbiol. Immunol. Infection Wei mian yu gan ran za zhi 56, 653–671. https://doi.org/10.1016/j.jmii.2023.01.017 (2023).
Humphries, R. M. et al. Clsi methods development and standardization working group best practices for evaluation of antimicrobial susceptibility tests. J. Clin. Microbiol. https://doi.org/10.1128/jcm.01934-17 (2018).
Sader, H. S. et al. Antimicrobial activity of cefoperazone-sulbactam tested against gram-negative organisms from Europe, Asia-Pacific, and Latin America. Int. J. Infect. Dis. IJID Off Publ. Int. Soc. Infect. Dis. 91, 32–37. https://doi.org/10.1016/j.ijid.2019.11.006 (2020).
Jones, R. N. et al. Multicenter studies of tigecycline disk diffusion susceptibility results for acinetobacter spp.. J. Clin. Microbial. 45, 227–230. https://doi.org/10.1128/jcm.01588-06 (2007).
Ewers, C. et al. Oxa-23 and isaba1-oxa-66 class d β-lactamases in acinetobacter baumannii isolates from companion animals. Int. J. Antimicrob. Agents 49, 37–44. https://doi.org/10.1016/j.ijantimicag.2016.09.033 (2017).
Xiao, Y. H. et al. Epidemiology and characteristics of antimicrobial resistance in china. Drug Resist. Updates: Rev. Comment. Antimicrob. Anticancer Chemother. 14, 236–250. https://doi.org/10.1016/j.drup.2011.07.001 (2011).
Jin, F., Zhang, X. & Zhao, Y. Measurement, source decomposition, and formation mechanism of differences in the development of china’s health services. Front. Public Health 13, 1495077. https://doi.org/10.3389/fpubh.2025.1495077 (2025).
Ding, J. et al. Equity and efficiency of medical service systems at the provincial level of china’s mainland: A comparative study from 2009 to 2014. BMC Public Health 18, 214. https://doi.org/10.1186/s12889-018-5084-7 (2018).
Teerawattanapong, N. et al. A systematic review of the burden of multidrug-resistant healthcare-associated infections among intensive care unit patients in southeast Asia: The rise of multidrug-resistant acinetobacter baumannii. Infect Control Hosp. Epidemiol. 39(5), 525–533 (2018).
Ji, S. et al. Prevalence of carbapenem-hydrolyzing class d β-lactamase genes in acinetobacter spp. Isolates in china. Eur. J. Clin. Microbiol. Infect. Dis. 33, 989–997 (2014).
El-Shazly, S., Dashti, A., Vali, L., Bolaris, M. & Ibrahim, A. S. Molecular epidemiology and characterization of multiple-drug resistant (mdr) clinical isolates of acinetobacter baumannii. Int. J. Infect. Dis. 41, 42–49 (2015).
. D, Quiñones, I. et al. High prevalence of blaoxa-23 in acinetobacter spp. And detection of blandm-1 in a. soli in cuba: Report from national surveillance program (2010–2012). New Microbes and New Infections. 2015
Leus, I. V. et al. Inactivation of adeabc and adeijk efflux pumps elicits specific nonoverlapping transcriptional and phenotypic responses in acinetobacter baumannii. Mol. Microbiol. 114, 1049–1065. https://doi.org/10.1111/mmi.14594 (2020).
Hamidian, M., Nigro, S. J. & Hall, R. M. Problems with the oxford multilocus sequence typing scheme for acinetobacter baumannii: Do sequence type 92 (st92) and st109 exist?. J. Clin. Microbiol. 55, 2287–2289. https://doi.org/10.1128/jcm.00533-17 (2017).
Qu, J. et al. The first outbreak caused by acinetobacter baumannii st208 and st195 in china. Biomed. Res. Int. 2016, 9254907 (2016).
Huang, G. et al. Multilocus sequence typing analysis of carbapenem-resistant acinetobacter baumannii in a Chinese burns institute. Front. Microbiol. 7, 1717 (2016).
Guo, X., Cao, Z. & Zhang, Z. Dissemination and evolution of blaoxa-23 gene among carbapenem-resistant acinetobacter baumannii isolates from central china. J. Infect. Dev. Ctries. 10, 445–448 (2016).
Jiang, M. et al. Epidemiological analysis of multidrug-resistant acinetobacter baumannii isolates in a tertiary hospital over a 12-year period in china. Front. Public Health 9, 707435. https://doi.org/10.3389/fpubh.2021.707435 (2021).
Gong, Y. et al. Epidemiology and resistance features of acinetobacter baumannii isolates from the ward environment and patients in the burn icu of a chinese hospital. J. Microbiol. (Seoul, Korea) 54, 551–558. https://doi.org/10.1007/s12275-016-6146-0 (2016).
Karah, N. et al. Insights into the global molecular epidemiology of carbapenem non-susceptible clones of acinetobacter baumannii. Drug Resistance Updates : Rev. Comment. Antimicrob. Anticancer Chemother. 15, 237–247. https://doi.org/10.1016/j.drup.2012.06.001 (2012).
McKay, S. L. et al. Molecular epidemiology of carbapenem-resistant acinetobacter baumannii in the united states, 2013–2017. Microb. Drug Resistance (Larchmont, NY) 28, 645–653. https://doi.org/10.1089/mdr.2021.0352 (2022).
Mohd Rani, F. et al. Acinetobacter spp Infections in malaysia: A review of antimicrobial resistance trends, mechanisms and epidemiology. Front. Microbiol. 8, 2479. https://doi.org/10.3389/fmicb.2017.02479 (2017).
Alcántar-Curiel, M. D. et al. Carbapenem-resistant acinetobacter baumannii in three tertiary care hospitals in mexico: Virulence profiles, innate immune response and clonal dissemination. Front. Microbiol. 10, 2116. https://doi.org/10.3389/fmicb.2019.02116 (2019).
Chen, C. L. et al. Functional characterization and clinical implication of a novel epidemic carbapenem-resistant acinetobacter baumannii genetic marker. Int. J. Antimicrob. Agents 62, 106879. https://doi.org/10.1016/j.ijantimicag.2023.106879 (2023).
Nishi, A. et al. Integration of molecular pathology, epidemiology and social science for global precision medicine. Expert Rev. Mol. Diagnos. 16, 11–23. https://doi.org/10.1586/14737159.2016.1115346 (2016).
Gil-Marqués, M. L., Pachón, J. & Smani, Y. Itraq-based quantitative proteomic analysis of acinetobacter baumannii under hypoxia and normoxia reveals the role of ompw as a virulence factor. Microbiol. Spectrum 10, e02328. https://doi.org/10.1128/spectrum.02328-21 (2022).
Boral, J. et al. Resensitization to colistin results in rapid and stable recovery of adherence, serum resistance and ompw in acinetobacter baumannii. PLoS ONE 19, e0309307. https://doi.org/10.1371/journal.pone.0309307 (2024).
Funding
This work was supported by the National Natural Science Foundation of China (No. 82002051), the Natural Science Foundation of Chongqing CSTC (cstc2021jcyj-msxmX0655), and the Young Doctors program of Xinqiao Hospital (2023YQB046).
Author information
Authors and Affiliations
Contributions
SP was responsible for manuscript writing, data analysis, and figure preparation. ZL and ZY jointly supervised the project, contributed to experimental design, and participated in data analysis and figure preparation. KL, YH, YG, and JW performed bacterial isolation and cultivation procedures. ZL and ZY conceptualized the study and critically revised the manuscript. All authors have read and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval and informed consent
This is an original research article on bacteria sequencing data. For this type of study, the requirement for ethics approval was approved by the Medical Ethics Committee of the Second Affiliated Hospital (Xinqiao Hospital) of Army Medical University, PLA (Approval No.2016-170-01 ). For this particular type of research, the requirement for informed consent was waived.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Pan, S., Liu, K., Huang, Y. et al. Epidemiology and molecular typing of multidrug resistant Acinetobacter baumannii burn wound isolates from six Chinese provinces. Sci Rep 15, 45076 (2025). https://doi.org/10.1038/s41598-025-31214-8
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-31214-8