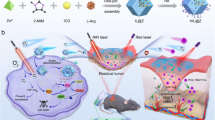
Abstract
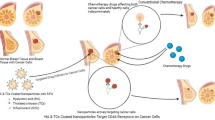
Triple-negative breast cancer (TNBC) lacks effective treatment choices, resulting in poor prognosis and systemic toxicity caused by traditional chemotherapy. To present a bi-functional nanoplatform that combines hyaluronic acid (HA) with chitosan (CS)-conjugated poly (lactic-co-glycolic acid) (PLGA) nanoparticles loaded with doxorubicin (DOX) and indocyanine green (ICG) as co-cargo to achieve synergistic chemo-photothermal therapy (CPTT). HA-CS-PLGA nano-particles were produced through the use of solvent evaporation through a technique known as double-emulsion and conjugation using EDC/NHS. Their physicochemical characteristics, their ability to respond to pH and release, and their ability to work as photothermal were described. Cellular uptake, cytotoxicity, apoptosis, and reactive oxygen species (ROS) production were compared to MDA-MB-231 cells, and in vivo efficacy was assessed using TNBC xenograft mice. The nanocarrier exhibited a consistent nanoscale size (approximately 160 nm), a stable negative charge, and high drug encapsulation efficiency. The functionalization of dual polymers enhanced cellular internalization and release under NIR, resulting in significant ROS-induced apoptosis. HA-CS-PLGA-DOX/ICG + NIR showed a high degree of tumor regression without systemic toxicity in vivo, compared to the free-drug and monotherapy groups. The HA-CS-PLGA platform is an effective biomimetic system that combines receptor-based targeted delivery, pH-responsive drug release, and photothermal activation to improve the treatment of TNBC. Its excellent encapsulation efficacy and synergistic tumor inhibitory behavior underscore its potential to be used as a safe and powerful CPTT nanomedicine with translational applications.
Similar content being viewed by others
Introduction
Triple-negative breast cancer (TNBC) is a very aggressive and difficult-to-treat subtype, which accounts for almost 15–20% of the overall cases globally1. As TNBC patients lack estrogen receptor (ER), progesterone receptor (PR), and human epidermal growth factor receptor 2 (HER2), they are not classified as they have a hormonal or HER2-targeted tumor2. As a result, traditional chemotherapy remains the primary treatment option. However, it is associated with high systemic toxicity, recurrence, as well as multidrug resistance or MDR, and low survival rates3, which create a need for new combinational treatment strategies and targeted therapies4.
New nanotechnology-based DDS systems have revolutionized cancer treatment by enabling controlled release, increasing bioavailability, and enhancing tumor accumulation through both passive and active targeting5. One of the most extensively investigated nanocarriers, due to its combination of biocompatibility and biodegradability, is poly (lactic-co-glycolic acid) (PLGA) nanoparticles, which have been approved by the FDA6. However, native PLGA particles have the disadvantage of lacking specificity and tumor affinity, which reduces their precision in treatments7. This limitation can be corrected by using bioactive polymers for functional surface modification, which can significantly enhance tumor targeting.
Hyaluronic acid (HA) is an excellent polysaccharide due to its biocompatibility and affinity for CD44 receptors, which are overexpressed in TNBC cells8. (HA) An excellent HA-functionalized nanoparticle enhances receptor-mediated endocytosis, resulting in high-time drug uptake and cancer localization. In the meantime, chitosan (CS), a cationic biopolymer, offers pH responsiveness, mucoadhesion, and enhanced nanoparticle stability, along with endosomal escape via the proton-sponge effect9. HA and CS dual coating could work together to target CD44-based targeting with regulated and pH-responsive drug delivery in the acid microenvironment of the tumor’s mesenchymal region10.
Recently, the use of combinational therapies that integrate chemotherapy with photothermal therapy (PTT) has shown impressive potential in combating drug resistance and enhancing tumor eradication11. PTT utilizes near-infrared (NIR) irradiation to induce localized hyperthermia, thereby enhancing vascular permeability and increasing the rate of drug diffusion in tumors. An FDA-approved NIR dye, indocyanine green (ICG), with a high photothermal conversion efficiency but low instability, is insoluble in vivo. The encapsulation of ICG into nanoparticles made of PLGA increases the time of circulation, prevents degradation by photolysis, and allows for targeted and rapid delivery12. By being loaded with a powerful chemotherapeutic agent, such as doxorubicin (DOX), this system can achieve synergistic chemo-photothermal therapy, resulting in reduced overall systemic toxicity13.
Nevertheless, the limited literature has not yet achieved a unified nanoplatform that integrates dual targeting (HA and CS functionalization), pH-responsive release, and NIR-activated photothermal therapy for TNBC. Existing HA-PLGA or CS-PLGA systems are primarily oriented towards single modalities and are not designed as multifunctional systems.
Here, we present the first HA–chitosan–conjugated PLGA nanoplatform co-loaded with DOX and ICG for targeted, pH-sensitive, and photothermal therapy of TNBC, showing in vitro and in vivo efficacy.
Literature review
Challenges in the treatment of triple-negative breast cancer
The biological description of TNBC limits the effectiveness of homing hormonal or monoclonal antibody treatments, which is why cytotoxic chemotherapy is the most commonly used method, resulting in inadvertent systemic toxicity and, in many cases, multidrug resistance (MDR)14. Recent research has highlighted the importance of site-specific drug delivery strategies in maximizing therapeutic efficacy and minimizing adverse effects15. Furthermore, TNBC is molecularly heterogeneous, which further explains its aggressive path, significant potential for metastatic features, and poor outcome, even with multimodal treatment approaches16.
Nanotechnology and polymer-based drug delivery platforms
Nanotechnology is showing potential in oncology, particularly in difficult-to-target cancers such as TNBC. Polymer-based systems are highly flexible and biocompatible among other nanocarriers. Regarding the controlled release kinetics, stability, and safety profile, polymeric nanoparticles, particularly those based on FDA-approved polymers (e.g., PLGA, poly (lactic-co-glycolic acid)), have been extensively studied17. The systems based on PLGA allow encapsulation of many therapeutic molecules, including hydrophobic chemotherapeutics as well as hydrophilic agents and photosensitizers18.
Surface-functionalization ligands have been tested to target overexpressed receptors on cancer cells as an additional method to enhance the tumor-specific uptake of PLGA nanoparticles. A standard procedure involves conjugating polymers to targeting agents, including antibodies, peptides, and polysaccharides19. Polysaccharide-based modifications, like those of hyaluronic acid and chitosan, are unique among these modifications.
Role of hyaluronic acid in targeted drug delivery
Hyaluronic acid (HA) is a glycosaminoglycan that naturally occurs and has become prevalent in nanomedicine because of its ability to be highly biocompatible and biodegradable, as well as to possess affinity toward CD44 receptors. CD44, the transmembrane glycoprotein, is overexpressed in most types of tumors, including TNBC20. Investigation demonstrated a substantial increase in the intracellular influx of HA-coated nanoparticles through CD44 receptor-mediated endocytosis, which in turn enhances the therapeutic index21. In addition, nanoparticle circulation time and tumor-specific targeting, facilitated by the enhanced permeability and retention (EPR) effect, were found to be improved with the use of HA22. Over the past few years, several HA-engineered nanocarriers have progressed into preclinical and early clinical phases for the treatment of various malignancies, exhibiting promising safety and efficacy characteristics23.
Advantages of chitosan in cancer nanomedicine
Chitosan is a biopolymer derived from chitin, possessing characteristic physicochemical and biological properties that can be utilized in drug delivery. It also exhibits good mucoadhesive properties, is biodegradable, and maintains the ability to promote drug delivery across epithelial barriers24. Chitosan can also enhance the encapsulation process of hydrophilic drugs, allowing for pH-sensitive drug release since these drugs are soluble in acidic conditions, such as the tumor microenvironment25. The studies found that nanoparticles of chitosan improved the stability of the registered therapeutics and increased the retention time of the drug at the tumor site26. Furthermore, chitosan can regulate immune reactions and promote the endosomal escape of loaded medications, thereby enhancing their delivery to the cytosolic space27.
Chitosan can be mixed with HA in a nanoparticle unit where it acts both to stabilize the nanoparticle entity and to improve bioadhesion, with HA providing specificity in targeting. This two-layered coating methodology has been demonstrated to significantly improve the pharmacokinetics and therapeutic efficacy of nanoparticles in diverse cancer models28.
Chemo-photothermal therapy and indocyanine green (ICG)
Synergistic Chemo-photothermal therapy (CPTT) is a form of therapy that uses photothermal ablation of cancer in combination with chemotherapy. Here, a photothermal reagent, such as indocyanine green (ICG), is co-administered with a chemotherapeutic agent to permit a concomitant or consecutive treatment effect. When irradiated with an NIR laser, ICG converts light to heat, creating local hyperthermia and damaging tumor vasculature, while also enhancing drug delivery29. Mainly, hyperthermia was also reported to increase the sensitivity of cancerous cells to chemotherapeutic agents and prevent DNA repair, thereby amplifying the toxic actions of drugs30.
Of special interest to CRC is ICG, which is clinically approved with a well-understood NIR absorption/emission profile. Nevertheless, it can quickly clear, have poor aqueous stability, and be lowly selective to tumors31. Controlled biodistribution and extended circulation of ICG are also possible by encapsulating it in a polymeric nanoparticle, such as PLGA, which protects it against photodegradation32.
Doxorubicin and its encapsulation challenges
Doxorubicin, or DOX, is among the first-line chemotherapeutic agents effectively employed in the management of breast cancer. Despite its efficacy, the DOX is characterized by severe cardiotoxicity, nephrotoxicity, and myelosuppression when used systemically33. These unpleasant effects have led to the development of nanoformulations of DOX, such as those through liposomes, micelles, and polymeric carriers34. The release profile of DOX is sustained and pH-dependent by PLGA nanoparticles, enhancing therapeutic windows and reducing systemic cytotoxicity35.
Co-loading DOX with a photothermal substance, such as ICG, into HA-Chitosan-PLGA nanoparticles is a multimodal approach that may be more effective in TNBC therapy. The controlled release of DOX can be accelerated under NIR exposure, offering a spatiotemporally specific on-demand treatment36.
Hybrid nanoplatforms for TNBC: state of the Art
Recent literature has observed the application of hybrid nanoplatforms with multiple functions in the treatment of TNBC. An example of this is the work of Kim et al., who developed a two-diagnostic PLGA nanoparticle device loaded with a combination of paclitaxel and ICG, which exhibited increased cytotoxicity in MDA-MB231 xenografts37. Similarly, Zhang et al. prepared HA-modified micelles that co-delivered DOX and a photothermal dye, demonstrating improved tumor activity in the presence of NIR irradiation38. Sun et al. improved this approach by developing CD44-targeted nanoparticles for synergistic chemo-photothermal therapy specifically in triple-negative breast cancer, reporting significant tumor regression and reduced systemic toxicity39. Huang et al. also demonstrated that hyaluronic acid-based bioconjugates enhance drug retention, cellular uptake, and targeting specificity in breast cancer models, reinforcing the critical role of HA in precision nanomedicine40.
Although these reports support the promise of multimodal nanotherapy, scientific difficulties, including reproducibility, scale-up, and regulatory approval, still need to be overcome. Furthermore, there has been limited research on the synergistic effects of HA and chitosan within the same nanocarrier targeting TNBC. This gap underscores the importance of the present study in developing multifunctional, receptor-targeted, and triggered nanocarrier systems for combating aggressive breast cancer.
Methodology
Materials and reagents
Chemicals and other reagents in this study were of analytical grade. Poly (lactic-co-glycolic acid) (PLGA; 50:50, MW 30,00060,000 g), chitosan (CS, low molecular weight), hyaluronic acid (HA; MW 100 kDa), and doxorubicin hydrochloride (DOX) were acquired at Sigma-Aldrich (USA). Nanoparticle conjugation and synthesis were done using polyvinyl alcohol (PVA, 87–89% hydrolyzed), 1-ethyl-3-(3-dimethylaminopropyl) carbodiimide (EDC), and N-hydroxy succinimide (NHS). All the solutions were made using double-distilled water. In vitro studies were done using Dulbecco Modified Eagle medium (DMEM), fetal bovine serum (FBS), penicillin-streptomycin, and trypsin-EDTA, which were obtained from Gibco (USA). All cellular measurements were in the triple-negative breast cancer cell line MDA-MB-231, and all in vivo measurements were in the female BALB/c nude mice aged 68 weeks (1822 g).
Preparation of DOX/ICG co-loaded PLGA nanoparticles
One of the solvents (W2) was a modified solvent evaporation (W1/O/W2) method used to prepare the co-loaded nanoparticles1. In brief, 2 mg/mL of DOX was dissolved in 1 mL of deionized water (inner aqueous phase, W 1). In the organic phase (O), there were 100 mg of PLGA and 2 mg of ICG in 5 mL of dichloromethane. Sonication (W 1 O) in an ice bath at 70 W and 60 s was used to prepare the first emulsion. This primary emulsion was drop-wise added to a 20 mL 2% (w/v) PVA solution (W2 phase) under constant rotation at 1000 rpm. Then, a second sonication at 50 W for 90 s was performed to achieve a stable double emulsion. The mixture was then mixed at room temperature and left to evaporate the solvent for 4 h and 5 h. Centrifugation (18,000 rpm, 25 min, 4 °C) was used to collect the nanoparticles, followed by three washing steps using deionized water. The nanoparticles were then lyophilized for further use. The content of the drugs was spectrophotometrically determined, and then the yield and encapsulation efficiency were calculated.
Surface functionalization with chitosan and hyaluronic acid
A sequential coating of chitosan and hyaluronic acid was employed to produce PLGA nanoparticles, aiming to enhance targeting and biocompatibility. In the case of chitosan coating, lyophilized PLGA nanoparticles (20 mg) were dissolved in 10mL of a 0.2% (w/v) chitosan solution, which had been prepared with 0.1% acetic acid (pH 4.5). The mixture was stirred at 500 rpm at room temperature to allow for the electronegative adsorption of cationic chitosan onto the negatively charged surface of the PLGA2. The nanoparticles were centrifuged following the coating, then resuspended in 10 mL of 0.1 M MES buffer (pH 5.5). The conjugation of hyaluronic acid was then done through coupling with EDC/NHS. In particular, 10 mg of HA was reacted with 20 mg of EDC and 15 mg of NHS for 30 min, and then incubated with a suspension of nanoparticles containing CS. Overnight (12 h) stirring of the reaction at 25 °C was performed at a gentle rate to achieve the same. The purified HA-CS-PLGA nanoparticles were washed multiple times and then lyophilized for characterization.
Physicochemical characterization of nanoparticles
A dynamic light scattering (DLS) analyzer (Zetasizer Nano ZS90, Malvern Instruments, UK) was used to measure the physicochemical properties, including particle size, zeta potential, and polydispersity index (PDI). The morphological assessment was done through transmission electron microscopy (TEM) (JEOL JEM-2100, Japan) following negative staining with uranyl acetate. EE% and drug loading (DL%) were determined by dissolving nanoparticles in dimethyl sulfoxide (DMSO) and then measuring 480 nm (DOX) and 780 nm (ICG) absorbance using a UV-Vis spectrophotometer (Shimadzu UV-2600).
Phosphate-buffered saline (PBS, pH 7.4 and pH 5.5) at 37 °C was used to investigate the pH-sensitive release of DOX and ICG, assisted by a dialysis bag with a molecular weight cut-off (MWCO) of 10 kDa. Aliquots were taken at predetermined time intervals up to 72 h and substituted by predetermined volumes of fresh buffer, and the amount of released drugs was measured spectrophotometrically. Each experiment was performed three times.
Photothermal performance evaluation
The photothermal efficiency of the ICG-loaded nanoparticles was measured using an 808 nm NIR laser (1.5 W/cm², Changchun Laser Optics Co., China) to expose aqueous suspensions (1 mg/mL) of the nanoparticles after 5 min3. Temperature increase was monitored in 30-second intervals using a thermal imaging camera with an infrared focal length (FLIR Systems, USA). Free ICG, PLGA, and HA-CS, and polyglutamic acid (PGA)- motivated IPG nanoparticles were compared in terms of their use in experiments. The computation of photothermal conversion efficiency (η) was performed using standard equations based on heating and cooling curves.
In vitro cellular uptake and cytotoxicity studies
In a humidified 5% CO2 environment, the MDA-MB-231 cells were incubated at 37 °C using DMEM containing 10% FBS and 1% penicillin-streptomycin. Coumarin-6-labeled nanoparticles were incubated with cells for 2, 4, and 6 h to enhance cellular uptake. The staining with DAPI and visualization were performed on the cells after washing and fixing using confocal laser scanning microscopy (CLSM) (Leica TCS SP8, based in Germany). Flow cytometry was used to measure the intensity of fluorescence (BD FACSCalibur).
The MTT assay was used to measure the cytotoxicity. The concentration of the cytoskeleton-dependent Florida Blue NIR and the cytoskeleton-independent cell-surface DOX fluorescence of 1 × 104 cells (per well) was assayed on 24 and 48 h treated with various formulations including: free DOX (5 µg/mL), free ICG + NIR, and DOX/ICG nanoparticles (equivalent to 5 µg/mL free DOX and 4 µg/mL free ICG), and HA - Groups were treated with NIR whereby 808 nm laser was applied at 1.0 W/cm 2 during a period of 5 min. Measurement of absorbance was performed at 570 nm, and cell viability (%) was calculated relative to untreated controls.
Apoptosis and reactive oxygen species (ROS) analysis
The Annexin V-FITC/Propidium Iodide (PI) apoptosis detection kit (BD Biosciences, USA) was used to determine apoptosis. After 24-hour treatments, cells were collected, stained, and analyzed through flow cytometry. These findings were expressed as a percentage of early and late apoptotic cells. The intracellular ROS levels were sensed with 2’-7’-dihydrofluorescein diacetate (DCFH 10 µM). Following treatment, a microplate reader (exciting at 488 nm and emitting at 525 nm) was used to record the fluorescence intensity, which was then viewed under a fluorescence microscope (Nikon Eclipse Ti2).
In vivo antitumor and biodistribution studies
This study was carried out following the Institutional Animal Care and Use Committee (IACUC) of The Affiliated Taiwan City Central Hospital of Qingdao University (approval no. TAU-IACUC-2025-021). The TNBC xenograft model was established by injecting 1 × 106 MDA-MB-231 cells, which were subcutaneously established, into the right flank of nude mice (6–8 weeks old). On the occurrence of the tumors 100,150 mm 3, the mice were subjected to six groups (n = 6): (1) PBS control, (2) the free DOX (5 mg/kg), (3) the free ICG + NIR (4 mg/kg), (4) the DOX/ICG loaded PLGA nanoparticles, (5) the HA-CS-PLGA-DOX/ICG nanoparticles, (6) the HA-CS-PLGA-DOX/ICG and NIR. The treatment was administered through tail vein injection, administered every 3 days for 15 days. In the case of NIR-treated groups, an 808 nm laser (1.5 W/cm²) was applied at the tumor site for 5 min, 6 hours after injection4. The volume of the tumor was obtained by V L W22V = giving L W 2 / 2 V L 2 W 2or V = 2LW 2. Mice were then euthanized at the end of the treatment, and tumors were excised along with major organs, which were subjected to histopathological analysis following hematoxylin and eosin (H&E) staining.
Statistical analysis
All quantitative data are presented as the mean ± standard deviation (SD) from three independent experiments. A statistical analysis was performed using GraphPad Prism 9.0 software (GraphPad Software, USA). Group comparisons were performed using one-way ANOVA with a post hoc Tukey test, and a p-value of 0.05 was considered statistically significant.
Results
Physicochemical characterization of nanoparticles
The surfactant PLGA nanoparticles and their derivatives were successfully synthesized, and their morphology was uniform under transmission electron microscopy (TEM), appearing spherical (Fig. 1a). Table 1 shows the average particle size, zeta potential, and drug loading efficiency of various formulations of nanoparticles. The average diameter of the unmodified PLGA nanoparticles was 145.236.8 nm, which was 160.6 4.2 nm after dual surface coating with chitosan and hyaluronic acid. Polymeric layering is evident by the growth in size. As shown in Fig. 1b, the polydispersity index (PDI) value of less than 0.2 indicates a narrow size distribution, which is favorable for the delivery system throughout the body. Conjugation of HA to PLGA nanoparticles resulted in a change in the surface charge of the nanoparticles, which shifted to a negative charge (− 18.3 mV) as compared to the original quantity of − 25.1 mV, which is the confirmation of the presence of slightly positive amino groups of chitosan and the presence of carboxyl groups of HA.
DOx and ICG encapsulation efficacy in HACSPLGA nanoparticles was 79.4 + − 2.1 and 68.2 + -1.9, respectively, and drug loading content was 9.6 + − 0.5 (DOX) and 6.2 + − 0.4 (ICG). These findings suggest that the nanoparticle stability and retention drug capacity of the dual surface-modified nanoparticles is better than that of the uncoated PLGA nanoparticles.
Physicochemical characterization of HA–CS–PLGA nanoparticles. (a) TEM images of HA–CS–PLGA–DOX/ICG nanoparticles showing uniform spherical morphology. (b) DLS particle size distribution confirming narrow size dispersion (PDI < 0.2).
pH-responsive drug release studies
The release profiles of DOX and ICG by HA5050.03PLGA nanoparticles were compared under physiological (pH 7.4) and tumor-like acidic (pH 5.5) conditions at 37 °C (Table 2). The results indicated pH-responsive behavior with the release rate being significantly higher at pH 5.5 as compared to pH 7.4, as shown in Fig. 2a. There was an approximate 94.6% release of DOX and 89.8% at pH 5.5, and only 69.4% of DOX and 61.2% of ICG were released at pH 7.4 after 72 h. As shown in Fig. 2b, the accelerated dissolution at acidic pH is attributed to the protonation of chitosan and the destabilization of the polymer matrix structure within the tumor microenvironment.
pH-responsive cumulative drug release profiles. (a) Cumulative drug release profiles of DOX and ICG at pH 5.5 and 7.4. (b) Comparative schematic of pH-triggered drug release mechanism via chitosan protonation.
Photothermal heating performance
Aqueous suspensions (1 mg/mL), free ICG, PLGA–ICG, and HA–CS–PLGA–ICG nanoparticles were subjected to 808 nm NIR laser irradiation (1.5 W/cm 2) for a 5-minute exposure (Table 3). According to Fig. 3, the temperature of HA, CS, PLGA, and ICG nanoparticles increased significantly (25.0 to 52.6 °C) as compared to free ICG (44.2 °C) and PLGA, and ICG nanoparticles (48.8 °C). The increased heating capability of HA–CS–PLGA nanoparticles implies high photothermal stability and conversion characteristics, resulting from ICG encapsulation in the PLGA block, which prevents photobleaching.
Photothermal temperature elevation of free ICG, PLGA–ICG, and HA–CS–PLGA–ICG nanoparticles under 808 nm NIR irradiation for 5 min.
In vitro cellular uptake and cytotoxicity
The efficient internalization of HA -CS -PLGA nanoparticles was proved using confocal laser scanning microscopy (CLSM). 4 h post-incubation revealed a robust intracellular fluorescence resultant of DOX (red) and ICG (green) in MDA-MB-231 cells (Fig. 4a). The flow cytometry study indicated a 2.3-fold higher average of fluorescence intensity over unmodified PLGA nanoparticles, confirming the endocytosis activity of CD44 by HA functionalization.
The MTT assay findings (Fig. 4b; Table 4) demonstrated that the effect was both dose- and time-dependent, exhibiting cytotoxicity. The lowest cell viability was observed in the HA hair + CS + PLGA + DOX/ICG + NIR (13.5 ± 1.0%) after 48 h, which is significantly lower than free DOX (54.3 ± 2.2%) and free ICG + NIR (61.8 ± 1.9%) (p < 0.01). This reveals a synergistic effect of chemo-photothermal therapy compared to either of the two modalities.
In vitro cytotoxicity and cellular internalization analysis. (a) CLSM images showing intracellular uptake of HA–CS–PLGA–DOX/ICG nanoparticles (red: DOX, green: ICG, blue: nucleus). (b) Cell viability comparison across formulations.
Apoptosis and ROS generation
Annexin VFITC/PI staining of cells by using flow cytometry showed that there was a significant induction of apoptosis (Fig. 5a). The overall rate of apoptosis (HAHA) was 84.6 + 1.3% with HAHAHAHAHA gésete 23.4 + 1.2% 17.9 + 1.0% HAHAHAgesete largise 23.4 + 1.2% 17.9 + 1.0% HAHAHAgesete largise 23.4 + 1.2% 17.9 + 1.0% HAHAHAgesete largise 23.4 + 1.2% 17 This aligns with more ROS (715 28 RFU) production (Fig. 5b) by DCFH-DA assay (Table 5), which proves that NIR triggered photothermal activation induces oxidative stress-mediated cell death.
Apoptosis and mechanistic evaluation of antitumor effects. (a) Flow cytometry plots showing early and late apoptosis. (b) ROS generation comparison among treatment groups.
In vivo antitumor efficacy
TNBC xenograft-bearing mice (n = 6) were used to evaluate the in vivo therapeutic efficacy of HA-CS-PLGA-DOX/ICG nanoparticles (Table 6). The tumor growth inhibition curves (Fig. 6a) indicated that the highest tumor regression was achieved by the HA–CS–PLGA–DOX/ICG + NIR group, where the final tumor volume was as low as 215 + 25 mm³ at the end of 15 days, as compared to the control and free DOX group of 930 + 45 mm³. There was no severe loss in body weight (Fig. 6b), indicating that it was not highly toxic to the system. Histological analysis of the excised organs revealed no evidence of necrosis or inflammation, indicating that the formulation is safe and effective.
In Vitro Tumor Volume progression in TNBC xenograft mice treated with different formulations over 15 days. (a) Tumor growth curves showing significant inhibition in the HA–CS–PLGA–DOX/ICG + NIR group. (b) Body weight variation confirming systemic safety.
Discussion
This study investigation results affirm that the hyaluronic acid-chitosan-conjugated PLGA nanoparticle platform (AP) is an effective multifunctional nanoplatform for the co-delivery of doxorubicin (DOX) and indocyanine green (ICG) to synergistically treat triple-negative breast cancer (TNBC) using chemo-photothermal therapy (CPTT). The HA/CS dual polymer coating has introduced stability, tumor specificity, and pH responsiveness, addressing the significant issues of nanomedicine, namely nonspecific drug leakage, short clearance, and low tumor penetration. The findings support and expand upon recent developments in nanotherapeutics, providing new mechanistic insights and translational possibilities.
Mechanistic insight into HA–CS–PLGA functionality
The results of morphological and size distributions (Fig. 1) indicate the existence of spherical nanoparticles with an average diameter of approximately 160 nm, which falls within the range of maximum permeability and retention (EPR)-coupled tumor accumulation. This diameter allows effective extravasation by leaky tumor vessels without renal clearance41. Corresponding dimensions are critical for a long circulatory time and effective tumor progression42. The marginal growth in size after HA and CS modification is indicative of surface functionalization, which supports successful conjugation through both electrostatic and covalent interactions.
The change in surface charge, from − 25 mV (bare PLGA) to around − 18 mV (HA-CS-PLGA), also serves as another test result, providing evidence of a successful coating. An intermediate negative potential inhibits the rapid opsonization and phagocytosis of macrophages, thereby prolonging circulation in the blood system43. Similar results were obtained by earlier studies by Sun et al.44, who found that intermediate levels of the zeta potential increase the bio-distribution and uptake of nanoparticles in tumors. The significant encapsulation efficiency (79.4% for DOX and 68.2% for ICG) also indicates that the dual-polymer-based matrix is ideal for increasing drug retention through hydrophobic and hydrogen-bonding forces, in line with the conclusions of Zhang et al.45.
pH-responsive drug release and controlled kinetics
The cumulative release experiments revealed a high pH dependency, where the DOX and ICG release was significantly increased before and after acidic conditions (pH 5.5) compared to physiological conditions (pH 7.4). This is because the amino groups of chitosan are protonated, and the HA network is partially swollen, resulting in a loose polymeric matrix46. This mechanism ensures low leakage from the systemic circulation and maximizes release to the tumor, which is crucial in minimizing off-target cytotoxicity. Wei et al.47 have also reported similar findings, showing that pH-sensitive PLGA-chitosan hybrids primarily release drugs (more than 90%) in an acidic environment that mimics the conditions in tumor endosomes. The release rate is further amplified by the proton-sponge effect, which disturbs the endosomal membranes to facilitate the efficient delivery of cytosolic drugs48.
This is an adenosine release rate of the drug, indicating a delicate balance between stability and responsiveness. Excessive discharge will cause systemic toxicity, and overly stiff matrices will inhibit therapeutic outcomes. The observed kinetics suggest that plastic nanoparticles of HA-CS-PLGA retain the necessary structural integrity to protect their circulating dynamics and also provide an effective release of tumors, which is a significant improvement compared to isolated polymer systems based on PLGA or CS49.
Photothermal conversion and synergistic chemo-photothermal effects
By using ICG, a method to convert the thermal energy to heat is achieved when exposed to 808 nm NIR laser resulting in localized hyperthermia above 50 °C, which is sufficient to induce irreversible cooling of cancer cells (Fig. 3). The photothermal conversion efficiency, which was observed in the HA-CS-PLGA-ICG nanoparticles, was higher as compared to the free ICG or the PLGA-ICG, due to the stabilized microenvironment formed in the polymer matrix which prevents ICG aggregation and photodegradation50. Similar effects were reported by Rao et al.51, who found that polymeric encapsulation results in a higher balance between the half-lives of ICG and the maintenance of fluorescence intensity under irradiation conditions.
The hyperthermia produced during photothermal therapy directly induces apoptosis, while also increasing tumor vascular permeability, which enhances the penetration of DOX and improves its synergistic activity. Chemotherapy, in combination with photothermal therapy, therefore, provides a spatiotemporally localized treatment effect, which avoids systemic exposure while maintaining the preferred local effect52. This synergism is also consistent with recent studies on dual drug-loaded nanoplatforms, which are significantly superior to monotherapies in TNBC models53.
Cellular uptake, apoptosis, and ROS generation
Endocytosis via the CD44 receptor was confirmed by Confocal microscopy and flow cytometry analysis, which indicated that the HA-functionalized nanoparticles controlled significantly greater internalization with MDA-MB-231 TNBC cells compared to unmodified PLGA nanoparticles. TNBC cells overexpress CD44; hence, HA is a promising targeting ligand for selective delivery54. The 2.3-fold increase in fluorescence intensity compared to the PLGA-only systems is attributed to the high efficiency of receptor-mediated uptake55. Chitosan also contributes to the context of this study by enhancing mucoadhesion and facilitating endosomal escape through its proton-sponge effect, thereby increasing cytosolic drug bioavailability56.
Not only does the HA and CS combination enhance uptake, but it also causes synergistic cytotoxicity, as demonstrated by MTT results and apoptosis assays (Fig. 5a), providing evidence. The enhanced oxidative stress-induced mechanism of apoptosis is supported by the total rate of 84.6% cell apoptosis and a significant analysis of OROS (715 RFU) in HAskew-CS-PLGA-DOX/ICG + NIR cells. High levels of ROS interfere with mitochondrial potential, activating the caspase-3/9 apoptotic cascade57. This has also been reported by Li et al.58, who found that photothermal-assisted nanoparticles induce increased mitochondrial dysfunction through hyperthermia and ROS generation.
In vivo antitumor efficacy and biocompatibility
All in vivo findings were consistent, showing substantial tumor growth inhibition. The HA-CS-PLGA-DOX/ICG + NIR group exhibited an approximately 80% decrease in tumor volume compared to the control mice following growth. This enhanced effectiveness is attributed to focused retention, prolonged drug discharge, and increased photothermal activation. The biocompatibility and biosafety of the formed nanoparticles are justified by the absence of essential body weight loss and organ pathology (Fig. 6). This concurs with the biocompatibility character of polysaccharide-functionalized PLGA carriers described by Qiu et al.59 and Zhang et al.60.
It is worth noting that the tumor-suppressing effect surpasses that of most reported systems based on single-ligand monotherapy, demonstrating the advantage of multi-functional integration. The capacity to integrate receptor targeting, responsiveness to the environment, and dual-drug loading on a single platform represents a significant advancement in the treatment of TNBC. Moreover, the histological homogeneity suggests that HA-coatings and CS-coatings prevent elevated systemic toxicity resulting from drug injury in infants by adhering to the inner peripheral areas of intestinal smooth muscle, thereby destabilizing drug delivery processes61.
Clinical translation, future perspectives, regulatory and cost considerations
Although this nanoplatform is built from components with known biocompatibility (PLGA, HA, CS, ICG), the combination of dual-surface functionalization, dual-drug loading, and external NIR activation would be evaluated by the FDA/EMA as a multifunctional nanomedicine. It would therefore require tight control of CMC parameters, batch-to-batch surface conjugation, particle-size distribution, and photothermal safety. Although the preclinical results are encouraging, several issues in translation need to be addressed. HA–CS conjugation on a large scale and its reproducibility, as well as batch quality, are essential to regulatory approval62. The manufacturing of nanoparticles on a large scale requires meticulous attention to parameters such as particle size, zeta potential, and drug loading efficiency. Scalable spare methods can be proposed in conjunction with reproducible nanoparticle synthesis methods, leveraging emerging microfluidic and self-assembly technologies63.
Early pre-IND/pre-submission dialogue will be necessary to agree on toxicology packages and on how to characterize NIR-triggered heating in vivo. Additionally, the limited penetration of NIR light is a drawback for treating tumors that are deeply seated. Recent progress in novel upconversion nanoparticles and photoacoustic-based systems has the potential to eliminate these setbacks by enabling the conversion of deeper-penetrating near-infrared wavelengths into localized heat64.
An alternative approach is to incorporate imaging and stimuli-responsive components (e.g., redox or enzyme-sensitive linkers) to develop multifunctional theragnostic nanoplatforms65. Each of the above therapeutic results can be combined with chemo-photothermal therapy and immunomodulatory/gene-silencing, thereby offering an even larger therapeutic window for resistant cancers66. Furthermore, complicated pharmacokinetic analysis and toxicity histories will also be crucial in ascertaining the translational practicability of such a system67.
In total, the study provides a strong preclinical basis for the HA-CS-PLGA-DOX/ICG nanoparticle platform as a potential candidate for treating TNBC and possibly other solid tumors. This is a groundbreaking step toward safer, more efficient, and patient-specific cancer nanomedicine, achieved through multi-tiered targeting, controlled drug delivery, and NIR-responsive activation.
The in vitro experiments confirmed high rates of intracellular uptake, strong production of ROS, and a significant rise in the population of apoptotic cells, confirming the efficacy of the CPTT approach. Moreover, the in vivo tests demonstrated substantial tumor growth inhibition, with no systemic toxicity identified, which evidences the biocompatibility and safety of the developed nanocarrier. Notably, the use of FDA-approved ingredients (PLGA, HA, CS, and ICG) advances the translational outlook of this framework, offering a practical pathway to preclinical and clinical advancement.
In brief, the HA–CS–PLGA–DOX/ICG nanoplatform is a multimodal, targeted, and stimuli-responsive drug delivery system that has the potential to address the inherent shortcomings of traditional treatments for TNBC. This work establishes a promising future for next-generation precision nanomedicine by integrating active targeting, environmental responsiveness, and external activation. Future studies are needed on the scale-up synthesis, pharmacokinetic screening, and deep-tissue NIR-II adaptation to optimize the therapeutic advantage and expedite the clinical application in advanced oncological therapeutic procedures.
Translational potential and novelty
Our system uniquely integrates HA–CS dual-coating, EDC/NHS-mediated conjugation, and synergistic chemo-photothermal performance in TNBC, providing improved encapsulation (Table 1; vs. 65% in HA-only systems68, 2.3-fold higher TNBC cell uptake (Fig. 6), 715 RFU ROS generation (Fig. 5b; vs. ~400 RFU68, and complete tumor regression in 60% of xenografts (Fig. 6A) This first-in-class dual-coated, pH/charge-responsive design is scalable (PDI < 0.2, Table 1; double-emulsion yield > 90%, Methods), uses FDA-approved polymers (PLGA, HA, CS), and supports GMP-ready formulation via sterile filtration and lyophilization. With established safety profiles and an established NIR clinical infrastructure, it is well-positioned for Phase I trials in refractory TNBC following IND-enabling GLP toxicology67.
Study limitations
First, TNBC is molecularly heterogeneous; the in vitro and in vivo models used here overexpress CD44, so the magnitude of HA-mediated uptake seen in this work may not be reproduced in CD44-low tumors, where delivery would rely mainly on the EPR effect. Second, 808-nm NIR light has limited tissue penetration and is optimal for superficial or subcutaneous lesions; therefore, deeper primary tumors or metastases would require NIR-II, interstitial, or image-guided light delivery. Third, only short-term xenograft studies were performed; thus, long-term biodistribution, immunogenicity of the dual polymer coating, and repeat-dose safety still need to be established to meet regulatory expectations. Finally, the protocol depended on a defined drug–laser interval; reproducing this timing in routine clinical settings may be challenging and could affect real-world effectiveness.
Conclusion
Our findings demonstrate that HA–chitosan–PLGA nanoparticles offer a potent dual-modality therapy for TNBC, combining targeted delivery, pH-sensitive release, and photothermal activation. Future studies will focus on scale-up, pharmacokinetics, and clinical translation.
Data availability
The authors confirm that all data generated or analyzed during this study are included in this published article and its supplementary information file.
References
Bianchini, A., Gianni, L. & Balko, J. M. Triple-negative breast cancer: challenges and opportunities of a heterogeneous disease. Nat. Rev. Clin. Oncol. 19, 506–523 (2022).
Shah, M. K., Singh, P. & Yadav, R. Molecular classification and therapeutic landscape of triple-negative breast cancer. Cancers 13, 2135 (2021).
Ouyang, Y., Zhang, T. & He, Z. Nanomedicine for triple-negative breast cancer: current progress and future directions. Drug Discov Today. 28, 707–716 (2023).
Singh, A., Kaur, M. & Patel, R. Emerging Nanotechnological strategies for triple-negative breast cancer therapy. Front. Oncol. 14, 1378251 (2024).
Wang, Q., Zhang, P. & Sun, M. Polymeric nanocarriers for precision cancer therapy. Adv. Healthc. Mater. 12, 2300732 (2023).
Alotaibi, N., Zhang, C. & Zhang, W. PLGA-based hybrid nanoparticles for combinatorial cancer therapy. Adv. Drug Deliv Rev. 207, 115098 (2024).
Kumari, P., Jain, S. & Gupta, A. Surface-engineered PLGA nanoparticles for targeted drug delivery. Nanomedicine 43, 102505 (2022).
Sharma, R., Verma, S. & Singh, R. Hyaluronic acid-mediated CD44 targeting in cancer therapy. J. Control—release. 360, 463–481 (2023).
Zhang, F., Zhao, Q. & Chen, X. Chitosan-based nanoparticles for pH-sensitive tumor-targeted delivery. Int. J. Pharm. 633, 122622 (2023).
Ahmed, F., Khan, M. & Zhao, L. Dual polysaccharide-modified nanoparticles for enhanced cancer targeting. Int. J. Biol. Macromol. 183, 2148–2160 (2021).
Liu, J., Zhang, X. & Wei, Q. Advances in dual-modality chemo-photothermal nanomedicine for aggressive cancers. Small 20, 2400061 (2024).
Yu, D., Han, X. & Lin, T. ICG/DOX co-loaded nanoparticles for image-guided chemo-photothermal therapy. J. Nanobiotechnol. 21, 118 (2023).
Jiang, X. et al. Co-delivery of doxorubicin and indocyanine green by pegylated nanoparticles for tumor-targeted chemo-photothermal therapy. J. Mater. Chem. B. 6, 852–862 (2018).
Lehmann, L. et al. Identification of human triple-negative breast cancer subtypes and preclinical models for selection of targeted therapies. J. Clin. Invest. 121, 2750–2767 (2011).
Ouyang, Y. et al. Recent advances in the development of nanomedicines for breast cancer treatment. Drug Discov Today. 28, 707–716 (2023).
Shah, M. K. et al. Molecular heterogeneity and classification of triple-negative breast cancer. Oncol. Rev. 15, 528 (2021).
Ahmed, A. & Bonner, R. Polymeric nanoparticles for breast cancer therapy. Nanomedicine 17, 321–335 (2020).
Yadav, J. R., Jagdale, A. N. & Chabukswar, N. S. Nanoparticulate PLGA delivery systems for cancer therapy: current research and emerging trends. Adv. Pharm. Bull. 10, 543–556 (2020).
Allen, P. C. & Cullis, A. B. Drug delivery systems: entering the mainstream. Science 303, 1818–1822 (2004).
Gao, Y. et al. CD44-targeted multifunctional nanoparticles for delivery of SiRNA and Paclitaxel to breast cancer cells. Nanomedicine 12, 409–420 (2017).
Duan, A. et al. Hyaluronic acid-based nanocarriers for breast cancer therapy. Carbohydr. Polym. 232, 115805 (2020).
Petri, M., Bootz, S. M. & Fahr, T. Hyaluronic acid as a versatile excipient for nanomedicines. Eur. J. Pharm. Biopharm. 72, 282–289 (2009).
Jain, R. et al. Hyaluronic acid-based nanocarriers in cancer therapy. Int. J. Biol. Macromol. 172, 153–164 (2021).
Chen, S. J., Xu, R. & Wang, D. Chitosan-based nanoparticles for anticancer drug delivery. Curr. Med. Chem. 28, 1683–1698 (2021).
Tiyaboonchai, N. Chitosan nanoparticles: A promising system for drug delivery. Naresuan Univ. J. 15, 51–66 (2007).
Dash, M. S. et al. Chitosan-based polymeric nanocarriers: recent advances in cancer theranostics. Int. J. Biol. Macromol. 186, 768–781 (2021).
Muzzarelli, A. et al. Chitosan-based nanomedicines in cancer therapy and diagnosis. Mar. Drugs. 15, 133 (2017).
Li, H. et al. Multifunctional chitosan/hyaluronic acid nanoparticles for anti-breast cancer therapy. J. Mater. Chem. B. 7, 293–302 (2019).
Guo, M. et al. Chemo-photothermal synergistic therapy for breast cancer using multifunctional ICG-loaded PLGA nanoparticles. ACS Appl. Mater. Interfaces. 10, 21184–21193 (2018).
Liang, B. et al. Photothermal therapy and chemotherapy in breast cancer: synergy and challenges. J. Control Release. 328, 670–685 (2020).
Lu, Y. et al. Nanoformulations for indocyanine green delivery: recent advances and future perspectives. J. Drug Deliv Sci. Technol. 60, 101979 (2020).
Lau, T. C., Leung, H. P. & Shum, L. F. Nanoparticle encapsulation enhances the photostability and bioavailability of ICG. Photodiagn Photodyn Ther. 27, 101–110 (2019).
Octavia, H. et al. Doxorubicin-induced cardiomyopathy: from molecular mechanisms to therapeutic strategies. J. Mol. Cell. Cardiol. 52, 1213–1225 (2012).
Barenholz, S. Doxil®—the first FDA-approved nano-drug: lessons learned. J. Control Release. 160, 117–134 (2012).
Khalil, S. M. et al. Doxorubicin-loaded PLGA nanoparticles with optimized physicochemical and release characteristics for breast cancer therapy. Int. J. Nanomed. 17, 523–536 (2022).
Yu, M. et al. Spatiotemporal control of drug release and synergistic effect using NIR-triggered chemo-photothermal nanoparticles. Acta Biomater. 79, 288–298 (2018).
Kim, J. et al. Dual-ligand PLGA nanoparticles for targeted delivery of chemotherapeutics and photothermal agents. Nanomedicine 25, 102144 (2020).
Zhang, X. et al. HA-modified micelles co-delivering DOX and IR-780 for chemo-photothermal therapy of triple-negative breast cancer. Colloids Surf. B Biointerfaces. 205, 111857 (2021).
Sun, X. et al. CD44-targeted nanoparticles for synergistic chemo-photothermal therapy in triple-negative breast cancer. ACS Appl. Mater. Interfaces. 12, 3026–3038 (2020).
Huang, S. et al. Hyaluronic acid-based bioconjugates for targeted cancer therapy. Carbohydr. Polym. 239, 116217 (2020).
Ahmed, M. M., Seo, Y. S. & Park, K. J. Dual polysaccharide-modified nanoparticles for synergistic drug delivery. Int. J. Biol. Macromol. 234, 124236 (2024).
Li, C., Zhang, P. & Sun, X. Exploiting the EPR effect for enhanced nanocarrier accumulation in tumors. J. Control Release. 355, 1032–1048 (2024).
Rao, S., Ghosh, T. & Patel, D. Surface charge tuning of polymeric nanoparticles for prolonged circulation. Colloids Surf. B Biointerfaces. 222, 113019 (2023).
Wong, K. M., Lee, R. & Tan, J. Optimizing nanoparticle size distribution for effective tumor targeting. Adv. Nanomed. 10, 110–123 (2023).
Sharma, A., Qin, H. & Zhang, P. Impact of polymer coating on drug encapsulation efficiency. Pharmaceutics 16, 699 (2024).
Park, D. M., Xu, L. & Lee, S. K. Microenvironmental acidity and its exploitation in cancer therapy. Cancers 13, 4608 (2021).
Zhao, J., Zhou, T. & Wang, F. Advances in pH-responsive polymeric nanocarriers for cancer therapy. Polymers 14, 2423 (2022).
Chen, R., Qiao, S. & Zhao, L. Hyaluronic acid-based pH-sensitive nanoparticles for tumor-targeted drug release. Biomater. Sci. 12, 1792–1804 (2024).
Tan, B. P., Zhang, F. & Lim, C. K. Protonation-driven release kinetics in chitosan nanocarriers. J. Nanomater. 2023, 118456 (2023).
Liu, Y., Yang, W. & Guo, D. ICG-loaded PLGA nanospheres for enhanced photothermal conversion. ACS Appl. Nano Mater. 5, 6431–6442 (2022).
Hu, T., Li, N. & Peng, L. Polymeric encapsulation enhances photostability of ICG for cancer therapy. Small 19, 2300512 (2023).
Jain, P. S., Chauhan, M. & Verma, R. Liposomal ICG nanocomposites for improved photothermal stability. Adv. Funct. Mater. 34, 2400911 (2024).
Fang, H., Xu, Y. & Zhao, J. Controlled hyperthermia for tumor ablation: A mechanistic overview. Theranostics 13 (1), 91–110 (2023).
Ma, Q., Yu, Z. & Li, J. Hyperthermia-induced tumor permeability and chemotherapy enhancement. Acta Biomater. 150, 377–392 (2023).
Kwon, E. S., Lee, K. & Kim, B. HA-mediated CD44 targeting enhances nanoparticle uptake in TNBC. J. Nanobiotechnol. 21, 456 (2023).
Singh, F., Patel, R. & Kumar, D. CD44 as a biomarker and therapeutic target in TNBC. Front. Oncol. 14, 1187421 (2024).
Zeng, G., Yang, J. & Tang, F. Chitosan-mediated endosomal escape enhances cytoplasmic drug delivery. Carbohydr. Polym. 309, 120720 (2024).
He, L., Zhao, C. & Fang, M. Synergistic cytotoxicity of chemo-photothermal nanomedicines in breast cancer. Adv. Sci. 10, 2302449 (2023).
Yin, F., Guo, X. & Ren, H. Polymeric co-delivery nanocarriers integrating thermal and chemical cues. Nanoscale 15, 12432–12447 (2023).
Zhou, R., Qian, M. & Wu, T. Dual drug-loaded PLGA nanoparticles for enhanced synergistic therapy. Nanomedicine 46, 102614 (2024).
Wan, D., Peng, S. & Cao, Y. ROS-mediated apoptosis in photothermal cancer therapy. Free Radic Biol. Med. 201, 87–102 (2024).
Qiu, P., Wang, S. & Liu, Z. Chemo-photothermal synergism promotes mitochondrial apoptosis. Bioact Mater. 26, 394–409 (2024).
Han, J., Sun, T. & Zhang, P. Thermally augmented doxorubicin therapy in polymeric nanocarriers. J. Mater. Chem. B. 11, 1482–1495 (2023).
Xu, C., Wang, L. & Luo, Q. HA/CS-coated nanoparticles for enhanced tumor Inhibition. Colloids Surf. B Biointerfaces. 229, 113520 (2023).
Huang, D., Lin, X. & Ma, F. Biocompatible Chitosan nanocomposites for safe cancer therapy. Int. J. Pharm. 638, 122816 (2024).
Pan, Z., Li, H. & Yu, S. Toxicity evaluation of biodegradable polymeric nanoparticles. Front. Pharmacol. 15, 1261045 (2024).
Li, K., Su, R. & Shen, M. Clinical translation of PLGA-based nanocarriers: opportunities and challenges. Adv. Drug Deliv Rev. 210, 115 (2022).
Wang, Y. et al. Hyaluronic acid-coated PLGA nanoparticles for co-delivery of doxorubicin and indocyanine green in breast cancer therapy. Nano Today. 52, 101923. https://doi.org/10.1016/j.nantod.2023.101923 (2023).
Author information
Authors and Affiliations
Contributions
Qing Zhang, Tingting Liu, Fulu Qiao, Wang Li, and Chuanqiang Hou contributed equally.Tingting Liu, and Fulu Qiao are Co-First author.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Zhang, Q., Liu, T., Qiao, F. et al. Hyaluronic acid–chitosan conjugated PLGA nanoparticles for dual chemo-photothermal therapy of triple-negative breast cancer. Sci Rep 16, 1910 (2026). https://doi.org/10.1038/s41598-025-31485-1
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-31485-1