Abstract
Recent years have seen ternary metal oxide nanocomposites attract widespread research interest due to their remarkable photocatalytic properties and applications. This study introduces a novel ternary ZnO-MgO-Gd₂O₃ nanocomposite through a green synthesis protocol with Ocimum basilicum L. seed extract that avoids harmful chemicals. A comprehensive set of analytical techniques was employed to accurately describe the structural, morphological, and optical properties. XRD analysis provided insights into the crystal structure, as an increase in crystal size from 12.06 nm at 500 °C to 12.34 nm at 600 °C was calculated. FE-SEM with EDS spectroscopy showed the formation of spherical nanoparticles with uniform distribution and the elements in the nanocomposite. DRS spectroscopy confirmed an increase in the band gap with increasing temperature from 3.7 eV (500 °C) to 2.9 eV (600 °C), while FTIR provided information on the functional groups. Also, the photocatalytic performance of the nanocomposite was evaluated and showed significant ability to degrade organic dyes under specific conditions (nanocomposite concentration = 26 mg, UVA lamp power = 20 W, and λ = 365 nm). It is worth noting that the nanocomposite has a degradation rate of 99.5% for EBT (pH = 10 after 105 min), 98.5% for MB (pH = 10 after 75 min), 91.5% for MO (pH = 10 after 105 min), and 72% for RhB (pH = 2 after 105 min). These results indicate the potential of the nanocomposite as a strong photocatalyst for wastewater treatment applications.
Similar content being viewed by others
Introduction
In recent decades, water pollution has become one of the most significant environmental challenges worldwide. The release of various pollutants, including organic pigments from the textile, dyeing, and pharmaceutical industries, into water has raised serious concerns about human health and the environment, becoming a significant environmental challenge. Despite their chemical stability and high biodegradability, these dyes are difficult to remove from the environment and may have harmful effects on humans and the environment. For this reason, it is imperative to develop efficient methods for removing these pollutants. In this regard, various technologies have been developed to remove these pollutants, including physical, chemical, and biological processes1,2. However, many of these methods have limitations such as high cost, production of sludge and hazardous by-products, and inefficiency at low concentrations of pollutants3. Photocatalytic processes have attracted the attention of many researchers as a new and promising method for water purification. In this process, semiconductors such as titanium oxide (TiO2) and zinc oxide (ZnO) are used as catalysts, which, by absorbing ultraviolet or visible light, are capable of producing strong oxidizing free radicals that can decompose organic pigments and convert them into harmless substances4. ZnO is an attractive option among semiconductor materials due to its good photocatalytic properties, chemical stability, low cost, and availability, and has been widely studied5. Pure ZnO, however, has limitations, including a high band gap energy and a high electron–hole recombination rate, which reduce its photocatalytic efficiency6. To improve the photocatalytic properties of ZnO, the use of metal oxide nanocomposites has been considered. Combining ZnO with other metal oxides can lead to an increase in the specific surface area, a decrease in particle size, and the introduction of impurities that reduce the electron–hole recombination rate, thereby improving the photocatalytic efficiency7,8,9. For example, Bakre et al. (2020) showed that CuO-NiO-TiO2 nanocomposites have high catalytic efficiency in various applications10. Additionally, the method of synthesis of nanocomposites also affects their properties and effectiveness. Green synthesis methods have been proposed as a suitable alternative to traditional chemical methods due to the use of natural and non-toxic materials and agents11. Plant extracts contain organic compounds that can serve as reduction and stabilization agents in the manufacture of NPs12. Previous studies have shown that the usage of nanocomposites has been effective in the degradation of organic dyes13,14,15. For example, Kanmani and Ramachandran (2012) showed that TiO2/ZnO core–shell nanostructures have high efficiency in the degradation of MB16. Also, Sabouri et al. prepared NiO nanoparticles by a green method using Gum Arabic and investigated their cytotoxic, photocatalytic, and antibacterial effects17. In this study, a ternary metal oxide nanocomposite, ZnO-MgO-Gd2O3, was synthesized using a green synthesis method with the extract of Ocimum basilicum L. The physical and chemical properties of the produced nanocomposite were assessed using various methods such as XRD, FESEM, and UV–Vis. Also, the photocatalytic efficiency of this nanocomposite in the degradation of MB, MO, EBT, and RhB dyes was evaluated. Although numerous studies have been conducted on ZnO binary nanocomposites, reports on the green synthesis of ZnO-MgO-Gd₂O₃ (ZMG) ternary nanocomposites, particularly using basil seed extract, are very rare and lack sufficient detail. A comprehensive examination into the correlation between their structure, photonic properties, and performance is also lacking. Therefore, the primary objective of our research is to develop an environmentally friendly synthesis method and thoroughly examine the photocatalytic potential of this emerging nanocomposite.
Experimental process
Materials
Zn (NO3)2. 6H2O (98.5%), Mg (NO3)2. 6H2O (99%), Gd (NO3)3. 6H2O (99.9%), MB (C16H18ClN3S), RhB (C28H31ClN2O3), EBT (C20H12N3O7SNa), MO (C14H14N3NaO3S), HCl (98%), and NaOH were acquired from Merck Co. The solvent of choice was also distilled water. Ocimum basilicum L. seeds were commercially sourced from a supermarket in Dargaz City, Iran.
Production of Ocimum basilicum L. seed extract
A fresh aqueous Ocimum basilicum L. seed extract was prepared in our laboratory for the green synthesis of ZnO-MgO-Gd₂O₃ nanocomposite as reducing and capping agents. The seeds were commercially sourced from a supermarket in Dargaz City, Iran. Ocimum basilicum L. seed extract’s primary role was to serve as a multifunctional agent for the reduction of metal precursors and stabilization of the formed nanocomposite. After washing with distilled water and drying in the dark, 1 g of Ocimum basilicum L. seeds was mixed with 100 mL of deionized water and stirred for 2 h at 60 °C. Then, the prepared extract was concentrated using a rotary evaporator to increase the amount of active ingredient in the extract. Lastly, for the synthesis step, the filtered extract was maintained at 4 °C.
Preparation of nanocomposite
For the synthesis of the ZMG nanocomposite, aqueous solutions (0.5 M) of Zn (NO3)2·6H2O, Mg (NO3)2·6H2O, and Gd (NO3)3·6H2O were prepared. These solutions were combined in a single beaker and stirred continuously for 45 min. Following this mixing period, 40 mL of the Ocimum basilicum L. plant extract was added to the mixture solution of salts. The resulting solution was maintained at a constant temperature of 80 °C for 6 h for gel formation. The gel was subsequently dried in an oven at 80 °C for 8 h. Afterward, it was calcined in a muffle furnace at 500/600 °C for 2 h each to yield ZMG nanocomposite18. Figure 1 depicts the schematic diagram of the nanocomposite synthesis process.
Schematic of ZnO-MgO-Gd2O3 Nanocomposite synthesis steps.
Characterization
To comprehensively investigate the properties of ZMG nanocomposites, detailed characterizations were performed on their structure, morphology, and optical properties. In this regard, the crystal structure of the sample was determined using XRD (D8-ADVANCE/ Bruker axs USA) in the range of 2θ = 10–80°, and the functional groups present in it were identified by FT-IR (AVATAR 370) in the range of 400–4000 cm⁻1. The surface morphology and particle dimensions were studied using FESEM (JEOL 7600F) and TEM (LeO910/Germany). The high-resolution images obtained were analyzed with Digimizer software to determine the particle size distribution. Simultaneously, elemental analysis was performed using EDS to confirm the weight and atomic percentage of the elements and their distribution in the nanocomposite structure. Finally, the optical characteristics and band gap energy of the material were evaluated using UV–Vis-DRS (SHIMADZU 3600/ Japan).
Experiments related to photodegradation
In order to evaluate the photocatalytic efficiency of ZMG nanocomposite, its performance in the degradation of four types of organic dyes, including MB, MO, EBT, and RhB, under ultraviolet UVA light irradiation was studied. For this purpose, first, 26 mg of photocatalyst was added to 100 mL of an aqueous solution of each dye with a concentration of 10−5 M. The resulting suspension was then kept in complete darkness for 45 min under continuous stirring to establish surface adsorption–desorption equilibrium between catalyst particles and dye molecules. After this stage, the photodegradation reaction was initiated by exposing the reactor to UVA light (UVA lamp power = 20 W, and λ = 365 nm). To monitor the reaction process, 2 mL samples were taken at regular intervals of 15 min. To determine the residual dye concentration, the samples were centrifuged for 10 min to completely separate the catalyst particles, and then the resulting clear solution was analyzed with a UV–Vis spectrophotometer. Measurements were made at the maximum absorption wavelength (λmax) of each dye, including 663 nm for MB, 464 nm for MO, 550 nm for EBT, and 555 nm for RhB19,20. Finally, the dye degradation efficiency was calculated using Eq. 1, which evaluates the concentration changes over time21,22.
where A0 represents the initial absorbance and At represents the absorbance at a given time t. Also, to predict the reaction path, the kinetics of the photocatalytic process were investigated using Eq. 2.
In this equation, C0 refers to the initial concentration, while Ct denotes the concentration at a given time t, and Kobs is the observed rate constant.
Results and investigation
XRD
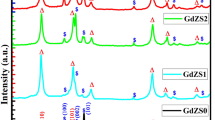
The XRD spectra of the ZMG nanocomposites were compared using X’pert High Score Plus software. X-ray diffraction patterns of the calcined nanocomposite at 500 and 600 °C are shown in Fig. 2. The diffraction peaks at angles of 31.76, 34.42, 36.25, 47.53, 56.59, 62.85, 67.94, and 69.08° correspond to crystal planes (100), (002), (101), (102), (110), (103), (112), and (201) for ZnO with hexagonal structure and space group P63mc (JCPDS No # 01–079-0206), respectively23. MgO crystallizes in a cubic structure characterized by the space group Fm-3 m. Remarkably, the XRD pattern of MgO reveals specific peak positions corresponding to crystal planes. The detected angles at 36.93, 42.91, 62.3, and 74.69°, which can be associated with (111), (200), (220), and (311) planes, respectively, for MgO, with the standard card (JCPDS No # 00–045-0946)24. On the other hand, peaks with angles of 20.10, 28.57, 33.11, 35.17, 42.59, 47.52, 52.1, 56.4, and 59.15° correspond to crystal planes (211), (222), (400), (411), (134), (440), (611), (622), and (444) of Gd2O3 with cubic structure and space group Ia3 (JCPDS No #00–043-1014)25,26. The crystallite size and strain due to lattice deformation were obtained using the Halder-Wagner method (Eq. 3). In this method, the peak broadening due to crystallite size and lattice strain is assumed to be Lorentzian and Gaussian, respectively27.
where βl and βG are the broadenings of the Lorentzian and Gaussian distributions, respectively, the advantage of the Halder-Wagner method is that it assigns more weight to the diffraction peaks at low and intermediate angles, where the overlap of the diffraction peaks is minimal. In this method, the crystal size (DH-W) and strain are obtained from the following Eq. 4.
where ε and D represent the strain and crystal size, respectively, and \({\beta }_{hkl}^{*}=\frac{{\beta }_{hkl}\text{cos}\theta }{\lambda }\) and \({d}_{hkl}^{*}=\frac{{2d}_{hkl}\text{sin}\theta }{\lambda }\)
XRD pattern of ZnO-MgO-Gd₂O₃ nanocomposites (500 and 600 °C).
In the Halder-Wagner method, first, the plot of \({\left(\frac{{\beta }_{hkl}^{*}}{{d}_{hkl}^{*}}\right)}^{2}\) is plotted against \(\frac{{\beta }_{hkl}^{*}}{\left({{d}_{hkl}^{*}}^{2}\right)}\) for the (hkl) peaks with higher intensity. Then the crystal size and strain can be obtained, respectively, by using the inverse of the slope and the width from the origin of the linear fit of the data. The Halder-Wagner curve of the ZMG nanocomposite calcined at two temperatures of 500 and 600 °C is shown in Fig. 3. According to the calculations, the crystallite size values of the ZMG nanocomposite calcined at temperatures of 500 and 600 °C were obtained as 12.06 and 12.34 nm, respectively.
Curve of variation of \({\left(\frac{{\beta }_{hkl}^{*}}{{d}_{hkl}^{*}}\right)}^{2}\) in terms of \(\frac{{\upbeta }_{\text{hkl}}^{*}}{\left({{\text{d}}_{\text{hkl}}^{*}}^{2}\right)}\) For ZMG nanocomposite calcined at temperatures of 500 °C (a) and 600 °C (b).
FT-IR
FT-IR analysis of the synthesized ZMG nanocomposite is shown in Fig. 4. The functional groups present in this nanocomposite were investigated in the range of 400 to 4000 cm-1. Under 11,000 cm-1, the peak represents the stretching vibration of the M–O bond, indicating the metal oxide nanocomposite’s excellent dispersion28. In addition, the peak identified in the range of 1300 to 1600 cm-1 is usually related to the vibrations of the C-H bonds. Also, the absorption band at 3426 cm-1 indicates the presence of the stretching vibrations of the O–H group29. The data show that a temperature increase from 500 °C to 600 °C leads to a reduction in the intensity of the C-H and O–H peaks.
FT-IR spectra of Ocimum basilicum L. seed extract and ZnO-MgO-Gd₂O₃ nanocomposites.
Morphological analysis
FE-SEM images of ZMG nanocomposites are presented in Fig. 5 (a, b). Increasing the synthesis temperature yields a nanocomposite with more uniform, highly regular spherical particles. To achieve precise measurements of particle size, the FE-SEM images were evaluated with DigiMizer software. Subsequently, the analysis was performed using IBM SPSS software, and the resulting particle size distribution curves are illustrated in Fig. 5 (c, d). The elements of the ZMG nanocomposite were identified and quantified using EDS analysis (Fig. 5 (e, f)), so that the presence of elements such as zinc, magnesium, gadolinium, and oxygen in the synthesized nanocomposite was confirmed. On the other hand, the elements’ atomic and weight percentages are inserted in a table in Fig. 5. Furthermore, the elemental map (Fig. 5 (g)) shows the nanocomposite’s elemental distribution, confirming the uniform dispersion of particles throughout the composite structure. The TEM image and particle size curve of the ZMG nanocomposite produced at 500 °C are shown in Fig. 6 (a, b). The results showed that the produced nanocomposite has a spherical morphology with a diameter of approximately 67 nm, with a homogeneous spatial distribution of particles.
FE-SEM pictures of ZnO-MgO-Gd₂O₃ nanocomposites 500 °C (a) and 600 °C (b), particle size curve 500 °C (c) and 600 °C (d), EDX spectrum 500 °C (e) and 600 °C (f), and Mapping images (g).
TEM image (a) and particle size curve (b) of ZnO-MgO-Gd₂O₃ nanocomposite at 500 °C.
UV–Vis-DRS analysis
The energy gap diagram and absorption spectrum of ZMG nanocomposite for two temperatures of 500 and 600 °C are shown in Fig. 7. The absorption edge in the absorption spectrum of the nanocomposite in the wavelength range of 380 nm indicates the presence of a band gap, which is an effective indicator in the analysis of the DRS spectrum. The energy gap values were calculated using Eqs. 5 and 6, and their values are reported in the diagram30. The results showed that the energy gap value decreases with increasing calcination temperature. The significant decrease in the band gap from 3.7 eV to 2.9 eV with increasing calcination temperature (from 500 °C to 600 °C) goes beyond being explained by a slight increase in crystallite size (12.06 nm to 12.34 nm). This phenomenon is strongly attributed to the dominance of lattice defects and impurities at 600°C due to quantum confinement. The higher temperature facilitates the formation and stabilization of oxygen vacancies (VO) and the creation of new energy levels within the band gap31. These defect levels shorten the electron–hole transfer paths and cause a strong red shift to 2.9 eV. This decrease in the band gap is highly desirable for enhancing the ability of the catalyst to absorb visible light and explains the improved photocatalytic performance.
Tauc diagrams of ZnO-MgO-Gd₂O₃ nanocomposite.
Photocatalytic activity
The photocatalytic process is the absorption of a photon by a molecule or precursor, which causes electronic excitation and the formation of a high-energy excited state in the system. Photocatalysts are known as environmentally active materials that, under the irradiation of sunlight or fluorescent light, can separate pollutants from their surface, and this light irradiation induces chemical reactions without changing the original structure of the material32,33. The main mechanism of this process is based on electron transfer on the surface of the photocatalyst. When radiation energy equal to or greater than the energy gap of the semiconductor is applied to the photocatalyst, electrons are transferred from the valence band to the conduction band. The salient features of photocatalysts include the ability to absorb natural radiant energy, low energy consumption, and environmental compatibility. The fundamental difference between the photocatalytic and catalytic processes is in their activation method; In catalysts, the reaction rate is increased by the formation of catalytic intermediate species, while in photocatalytic processes, the formation of electron–hole pairs under light irradiation accelerates chemical reactions. The positive holes (h⁺) created in the valence band react with water molecules to produce hydroxyl radicals (OH∙). On the other hand, the excited electrons in the conduction band can react with oxygen molecules to form superoxide radicals (\({{O}^{\bullet }}_{2}^{-}\)). These radical species are considered the main factors in the degradation and oxidation processes of organic compounds and can convert organic molecules to simpler products through oxidative pathways34,35,36,37. The chemical reactions (1–5) governing the photocatalytic process are summarized below, and Fig. 8 shows a schematic diagram of this process.
The mechanism of the photocatalytic process.
Considering the crucial role of the pH parameter in the efficiency of photocatalytic degradation processes, the effect of changes in pH on the degradation of four organic pollutants (MB, MO, EBT, and RhB) in the presence of the ternary ZMG nanocomposite was evaluated. The experiments were conducted in the concentration range of 10–5 M for dyes and 26 mg for the nanocomposite. In this regard, the effect of different pH levels on the degradation rate of dyes was investigated separately. For MB, MO, and EBT, pH values of 3, 7, and 10 were tested, while for RhB, values of 2, 7, and 10 were used. The pH of the dye solutions was adjusted with 2 M NaOH and 1 M HCl. The results are shown in Fig. 9. Based on the experimental results, the highest degradation efficiency was obtained for MB (98.5%), MO (91.5%), and EBT (99.5%) in an alkaline medium with pH = 10. In contrast, RhB in the acidic medium (pH = 2) showed the highest degradation percentage with a value of 72%. This difference in efficiency can be attributed to the different chemical structures of the dyes and their stability under different pH conditions. The graphs related to the investigation of the photocatalytic activity and kinetic analysis of the reactions for all dyes are presented in Fig. 10. Analysis of the kinetic data shows that the reactions adhere to a pseudo-first-order kinetic model, which indicates the dependence of the reaction rate on the initial concentration of the dyes38.
Effect of pH on the degradation of MB (a), EBT (b), MO (c), and RhB (d) dyes.
Photocatalytic activity of ZnO-MgO-Gd₂O₃ nanocomposite at optimum pH of MB (a, pH = 10), EBT (b, pH = 10), MO (c, pH = 10), and RhB (d, pH = 2), pseudo-first-order kinetics of pollutants (e).
Conclusions and prospects
In this study, ZMG nanocomposite was synthesized using Ocimum Basilicum L. extract by the green chemistry method. The results of various analyses, such as XRD, FTIR, UV–Vis/DRS, FE-SEM/EDX/Mapping, indicated the successful synthesis of the aforementioned nanocomposite. The XRD pattern of the synthesized nanocomposite confirmed its crystalline nature. Increasing the calcination temperature resulted in the formation of clearer peaks and higher intensities, indicating an increase in the crystallinity of the nanocomposite. Also, increasing the temperature increased the crystallite size of the synthesized nanocomposite, which is due to the increase in atomic mobility, nucleation, and grain growth during the calcination process. The results of FT-IR examination confirmed the presence of different functional groups within the nanocomposite. Increasing the calcination temperature caused slight changes in specific peaks, which may be due to changes in the bonding environment, lattice vibrations, or molecular interactions. The UV–Vis/DRS spectra of the ZMG nanocomposite showed absorption bands in the range of 200 to 400 nm. Also, the energy gap values of the nanocomposite were calculated according to the DRS data, and it was found that increasing the temperature resulted in a decrease in the energy gap. The larger crystallite size and smaller energy gap in calcined samples are due to temperature differences, as higher temperatures cause thermal energy diffusion and recrystallization. EDS analysis also confirmed the presence of all elements in the nanocomposite. Finally, the photocatalytic activity of the nanocomposite in the degradation of MB, MO, EBT, and RhB dyes was investigated at the optimum pH. By examining the results, it was found that MB, MO, and EBT dyes had a significant degradation percentage in basic conditions, which were 98.5, 91.5, and 99.5, respectively. Also, RhB dye had 72% degradation in acidic conditions. While the green-synthesized nanocomposite shows promising performance, the scalability of its plant-based production method requires further development. Future efforts will therefore focus on scaling the synthesis for industrial applications, such as wastewater treatment, and conducting toxicity studies to ensure environmental safety.
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- ZnO:
-
Zinc oxide
- MgO:
-
Magnesium oxide
- Gd2O3 :
-
Gadolinium oxide
- DRS:
-
Diffuse reflectance spectroscopy
- MB:
-
Methylene blue
- RhB:
-
Rhodamine B
- MO:
-
Methyl orange
- EBT:
-
Eriochrome black T
- FT-IR:
-
Fourier transform-infrared
- UV–Vis:
-
Ultraviolet–visible
- XRD:
-
X-ray diffraction
- EDS:
-
Energy-dispersive X-ray spectroscopy
- FESEM:
-
Field emission scanning electron microscopy
- TEM:
-
Transmission electron microscopy
- UVA:
-
Ultraviolet- A
References
Bopape, D. A., Ntsendwana, B. & Mabasa, F. D. Photocatalysis as a pre-discharge treatment to improve the effect of textile dyes on human health: A critical review. Heliyon 10, e39316 (2024).
Al-Tohamy, R. et al. A critical review on the treatment of dye-containing wastewater: Ecotoxicological and health concerns of textile dyes and possible remediation approaches for environmental safety. Ecotoxicol. Environ. Saf. 231, 113160 (2022).
Nkosi, N. C., Basson, A. K., Ntombela, Z. G., Dlamini, N. G. & Pullabhotla, R. V. Green synthesis, characterization and application of silver nanoparticles using bioflocculant: A review. Bioengineering 11(5), 492 (2024).
Mohamadpour, F. & Amani, A. M. Photocatalytic systems: Reactions, mechanism, and applications. RSC Adv. 14(29), 20609–20645 (2024).
Irfan, M. et al. Improving photocatalytic efficiency of ZnO nanoflowers through gold incorporation for Rhodamine B photodegradation. Opt. Mater. 154, 115681 (2024).
Yang, R., Liu, J., Sun, X., Wang, C. & Jiang, B. Enhancing photocatalytic N2 fixation efficiency through the construction of optimized hollow ZnO/Cu Mott-Schottky heterojunction microreactors. J. Alloy. Compd. 1014, 178634 (2025).
Zhu, C. & Wang, X. Nanomaterial ZnO synthesis and its photocatalytic applications: A review. Nanomaterials 15(9), 682 (2025).
Baig, A., Siddique, M. & Panchal, S. A review of visible-light-active zinc oxide photocatalysts for environmental application. Catalysts 15(2), 100 (2025).
Immanuvel, A., Pandiyarajan, C., Sivaranjana, P., Kandasamy, M. & Rameshkumar, P. Effective quenching of charge recombination in ZnO nanorods supported reduced graphene oxide for improved photocatalysis. Inorg. Chem. Commun. 179, 114878 (2025).
Bakre, P. V., Kamat, D. P., Mandrekar, K. S., Tilve, S. G. & Ghosh, N. N. CuO-NiO-TiO2 bimetallic nanocomposites for catalytic applications. Mol. Catal. 496, 111193 (2020).
Mohammadi, A. et al. Green synthesis and toxicological evaluation of zinc oxide nanoparticles utilizing Punica granatum fruit Peel extract: An eco-friendly approach. Sci. Rep. 15(1), 20853 (2025).
Shahzadi, S., Fatima, S., Shafiq, Z. & Janjua, M. R. S. A. A review on green synthesis of silver nanoparticles (SNPs) using plant extracts: A multifaceted approach in photocatalysis, environmental remediation, and biomedicine. RSC Adv. 15(5), 3858–3903 (2025).
Singh, J. et al. ‘Green’synthesis of metals and their oxide nanoparticles: Applications for environmental remediation. J. Nanobiotechnol. 16, 1–24 (2018).
Kumar, K. H., Ananda, H., Ravishankar, D., Madhu, H. & Thirumala, S. A review on nano metal oxides and their nanocomposites for photocatalytic degradation of dyes. Sustain. Chem. One World 6, 100055 (2025).
Khan, Y. et al. Photocatalytic treatment of organic dyes using metal oxides and nanocomposites: A quantitative study. Open Chem. 22(1), 20240026 (2024).
Kanmani, S. & Ramachandran, K. Synthesis and characterization of TiO2/ZnO core/shell nanomaterials for solar cell applications. Renew. Energy 43, 149–156 (2012).
Sabouri, Z., Akbari, A., Hosseini, H. A., Khatami, M. & Darroudi, M. Green-based bio-synthesis of nickel oxide nanoparticles in Arabic gum and examination of their cytotoxicity, photocatalytic and antibacterial effects. Green Chem. Lett. Rev. 14(2), 404–414 (2021).
Hazrati Saadabadi, R., Shariatmadar Tehrani, F., Sabouri, Z. & Darroudi, M. Photocatalytic activity and anticancer properties of green synthesized ZnO-MgO-Mn2O3 nanocomposite via Ocimum basilicum L seed extract. Sci. Rep. 14(1), 29812 (2024).
Mir, S. H. et al. Enhanced dye degradation through multi-particle confinement in a porous silicon substrate: A highly efficient, low band gap photocatalyst. Adv. Opt. Mater. 9(11), 2002238 (2021).
Yang, S. et al. Significantly enhanced dye removal performance of hollow tin oxide nanoparticles via carbon coating in dark environment and study of its mechanism. Nanoscale Res. Lett. 9, 1–9 (2014).
Albeladi, A. et al. Fe3O4-CdO nanocomposite for organic dye photocatalytic degradation: Synthesis and characterization. Catalysts 14(1), 71 (2024).
Selvaraj, R. et al. Eco-friendly synthesis of silver nanoparticles from rubber fig leaves and their application in the catalytic degradation of Congo Red dye. Mater. Technol. 40(1), 2498585 (2025).
Abdelghani, G. M., Ahmed, A. B. & Al-Zubaidi, A. B. Synthesis, characterization, and the influence of energy of irradiation on optical properties of ZnO nanostructures. Sci. Rep. 12(1), 20016 (2022).
Hirphaye, B. Y., Bonka, N. B., Tura, A. M. & Fanta, G. M. Biosynthesis of magnesium oxide nanoparticles using Hagenia abyssinica female flower aqueous extract for characterization and antibacterial activity. Appl. Water Sci. 13(9), 175 (2023).
Bandyopadhyay, A. et al. Dielectric study and magnetic property analysis of Gd2O3 nanorods/nanowire in combination with Monte Carlo simulation. J. Alloy. Compd. 882, 160720 (2021).
de Lima Rezende, T. K. et al. Structural and photoluminescent behavior of Rare Earths doped Gd2O3 nanoparticles embedded into SiO2 amorphous matrix obtained by sol-gel method for energy conversion. Mater. Chem. Phys. 337, 130574 (2025).
Dar, M. A. et al. Preserved crystal phase and morphology: Improving the magnetic and electrochemical performance of sulfur doped tin oxide nanoparticles synthesized via the hydrothermal method. Appl. Surf. Sci. Adv. 13, 100360 (2023).
Anjum, S. et al. Recent advances in zinc oxide nanoparticles (ZnO NPs) for cancer diagnosis, target drug delivery, and treatment. Cancers 13(18), 4570 (2021).
Alhujaily, M. et al. Au/ZnO nanocomposites prepared by laser ablation for enhancement of antibacterial activity and cytotoxic properties against cancer cells. Metals 13(4), 735 (2023).
Li, J., Xie, D., Li, M., Liu, S. & Wei, C. A. Optimal learning samples for two-constant Kubelka-Munk theory to match the color of pre-colored fiber blends. Front. Neurosci. 16, 945454 (2022).
Ajin, V. A. & Lenus, A. J. Engineering the role of oxygen vacancies in photocatalysts for environmental remediation and energy conversion applications: A comprehensive review. Mater. Sci. Semicond. Process. 197, 109705 (2025).
Li, X. et al. Challenges of photocatalysis and their coping strategies. Chem. Catal. 2(6), 1315–1345 (2022).
Sabouri, Z., Sabouri, S., Sabouri, M. & Darroudi, M. Evaluation of sensor performance, photocatalytic efficiency, and biological effects of se doped ZnO-Al₂O₃ nanocomposite synthesized with nymphaea pygmaea alba extract. Water Air Soil Pollut. 236(12), 796. https://doi.org/10.1007/s11270-025-08464-3 (2025).
He, S., Chen, Y., Fang, J., Liu, Y. & Lin, Z. Optimizing photocatalysis via electron spin control. Chem. Soc. Rev. https://doi.org/10.1039/D4CS00317A (2025).
Vinayagam, R. et al. Photocatalytic degradation of methylene blue dye using newly synthesized zirconia nanoparticles. Environ. Res. 214, 113785 (2022).
Vinayagam, R., Murugesan, G., Goveas, L. C., Varadavenkatesan, T. & Selvaraj, R. Mesoporous magnetic biochar composite through one-pot hydrothermal process for efficient degradation of methylene blue dye by Fenton-like catalysis. Mater. Technol. 40(1), 2510481 (2025).
Sabouri, Z., Sammak, S., Sabouri, S., Moghaddas, S. S. T. H. & Darroudi, M. Green synthesis of Ag-Se doped ZnO-Co3O4-NiO fivenary nanocomposite using poly anionic cellulose and evaluation of their anticancer and photocatalyst applications. Chem. Methodol. 8(3), 164–176. https://doi.org/10.48309/chemm.2024.436507.1758 (2024).
Al-Harby, N. F., Albahly, E. F. & Mohamed, N. A. Kinetics, isotherm, and thermodynamic studies for efficient adsorption of Congo Red dye from aqueous solution onto novel cyanoguanidine-modified chitosan adsorbent. Polymers 13(24), 4446 (2021).
Acknowledgements
The technical support for this work was provided by the University of Semnan and Mashhad University of Medical Sciences based on the Ph.D. thesis of Ms. R. Hazrati Saadabadi.
Author information
Authors and Affiliations
Contributions
Razieh Hazrati Saadabadi: Data acquisition, analysis, and interpretation, writing original draft. Zahra Sabouri : Data acquisition, analysis, and interpretation, Writing—Review & Editing. Fatemeh Shariatmadar Tehrani : Supervision, Project administration, Funding, and resources acquisition, Review & Editing. Majid Darroudi : Supervision, Project administration, Funding, and resources acquisition, Review & Editing.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval
For this type of study, ethical approval was not required.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Saadabadi, R.H., Sabouri, Z., Tehrani, F.S. et al. A novel ZnO-MgO-Gd₂O₃ nanocomposite synthesized with Ocimum basilicum seed extract for enhanced photocatalysis. Sci Rep 16, 2235 (2026). https://doi.org/10.1038/s41598-025-31998-9
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-31998-9