Abstract
This study used pure silica fume nanoparticles (SF NPs) and 3-APTES-functionalized silica fume (F-SF) to improve the thermal properties of water-based acrylic paint. First, the NPs were synthesized and then their structural, chemical, and morphological studies were performed using infrared spectroscopy (FT-IR) to identify functional groups and confirm the functionalization reaction, X-ray diffraction (XRD) to determine the phase and crystal structural changes, scanning electron microscopy (SEM) to study the surface and overall morphology, energy dispersive X-ray spectroscopy/ elemental mapping (EDX/ Mapp), and transmission electron microscopy (TEM) to examine the particle size and dispersion. The addition of F-SF to water-based acrylic paint significantly improved the coating adhesion (up to 5B), hardness, and gloss, which is due to the higher dispersion of NPs and stronger interfacial interactions in the matrix. After confirming the above factors, NPs were added to water-based acrylic paint at 0.5–1.5 Wt.%. The thermal conductivity (K) properties of the samples were measured to investigate the thermal insulation performance. The K values of the different samples as base coating, FS-0.5, FS-1, FS-1.5, F-FS-0.5, F-FS-1, and F-FS-1.5 were obtained as 0.420, 0.356, 0.326, 0.342, 0.337, 0.281, and 0.300 W/mK, respectively. The analysis showed that pure SF NPs reduced the paint’s K by 22% and F-SF NPs by 33%. This decrease in K is due to the creation of porosity and increased thermal circulation paths in the paint structure, which prevents easy energy transfer and helps increase thermal insulation properties. Also, functionalization with 3-APTES improves the adhesion and dispersion of NPs in the paint matrix due to the higher decrease in K compared to pure SF, the difference in surface structure, and its effect on heat transfer. Overall, the results indicate the high potential of these NPs to improve the thermal insulation performance of acrylic paints and industrial applications in thermal protective coatings.
Similar content being viewed by others
Introduction
The major increase in the world’s population has led to an unprecedented consumption of fossil fuels, which has significantly exacerbated environmental pollution caused by industrial and human activities1,2. According to statistics, about one-third of the world’s total energy consumption is spent on human activities inside buildings3. Also, research shows that humans spend more than 90% of their time indoors, so providing comfortable conditions in these spaces is of particular importance4. The indoor environment of buildings plays a vital role in ensuring the well-being and comfort of residents, and this depends on several factors, the most important of which is temperature5. The cooling and heating processes of buildings rely on the speed and direction of heat transfer through walls and ceilings. Thermal conductivity, as one of the key factors in heat transfer between building components, is of great importance. Therefore, the use of thermal insulation as an essential tool to reduce heat transfer and optimize energy consumption in heating and cooling systems is vital6,7,8,9,10. Given the high costs of fuel and electrical energy consumed in these devices, building insulation not only helps reduce thermal load but also plays a crucial role in reducing energy consumption and, consequently, reducing greenhouse gas emissions such as carbon dioxide (CO2) and other pollutants, which are the leading causes of global warming and environmental pollution11. A significant amount of naturally generated solar heat enters the building through the roof, which creates unfavorable conditions for the occupants. In general, heat transfer from the roof occurs in three ways: conduction, convection, and radiation12,13,14,15,16,17,18,19. Heat convection cannot be controlled due to its high dependence on wind conditions. However, the other two processes, conduction and radiation, can be reduced by adopting appropriate solutions. Roof insulation significantly limits the heat transfer process. Also, to reduce the effect of thermal radiation, it is necessary to use materials with low emissivity on the internal surfaces and materials with high reflectivity on the external surface of the roof. The use of highly reflective coatings can help reduce the temperature of the external surface of the roof and, consequently, the building. Materials that have high reflectivity of solar radiation and low heat transfer to the interior of the building play an important role in reducing the energy consumption of air conditioning systems and increasing the comfort level of the occupants. Numerous studies have been conducted in the field of reducing solar heat absorption in buildings. A low k value results in reduced direct absorption of solar energy, hence saving on cooling energy7,8. The results show that buildings coated with white paint are up to 3 °C cooler than buildings painted in gray20,21,22,23,24. White electrometric coatings with a reflectance of more than 0.72 can be up to 45 °C cooler than black coatings with a reflectance of 0.0825. Also, concrete tiles painted with reflective coatings have better thermal performance than white marble and mosaic tiles, which are known as cool materials26. Non-metallic minerals with high emissivity help reduce the external surface temperature and limit the flow of heat into the building22. Recent studies on APTES-functionalized SF in coatings are limited but emerging, with some relevant work available. One 2017 study details surface modification of SF with APTES to improve dispersion and reduce NPs agglomeration, showing improved stability and reduced aging susceptibility in asphalt binders modified with APTES-functionalized SF. This work highlights chemical and rheological changes induced by APTES, such as better zeta potential, viscosity improvement, and lower viscosity aging index, demonstrating the potential coating and binder enhancement effects of APTES-functionalization on SF27.
In this paper, an experimental study on the effect of SF and F-SF as heat insulating paint on K is presented. Silica exhibits the lowest thermal conductivity of all known solid insulating materials worldwide28,29,30. Our work uniquely focuses on pure SF NPs and their functionalization with F-SF specifically in water-based acrylic paints to enhance thermal insulation. The novelty lies in the combined investigation of the functionalization effects on NP dispersion, coating adhesion, and resulting thermal conductivity reduction in this widely used paint system. Unlike other works that often focus on silica aerogels or cementitious materials, our study addresses coatings with practical applications in construction and automotive industries. This research fills the gap by demonstrating how APTES-functionalized SF NPs improve both the mechanical and thermal properties of water-based acrylic coatings, providing a new approach to producing heat-resistant and energy-saving paint products. Incorporating silica into building materials can significantly reduce their k and enable levels of thermal performance that were previously considered unattainable.
Experimental section
Materials
In this study, Commercial SF was purchased as the base material from Elkem company, Norway. Other raw materials and solvents, including acetic acid (CH3COOH, 100%), toluene (C7H8, 100%), 3-aminopropyl triethoxysilane (3-APTES, 98%), and ethanol (EtOH, 99%), were purchased with high purity from Merck, Germany.
Preparation of F-SF with 3-APTES
In this study, the method presented by Erdem et al.31 was used to F-SF, and 3-APTES was selected as the organic source of functionalization (Fig. 1). In the first step, in order to activate the surface, 20 g of SF was stirred with 100 mL of 0.01 M acetic acid solution for 1 h. After this step, the obtained sample was filtered and washed with distilled water until the pH of the medium reached 6. Next, the activated silica was transferred to a round-bottom flask containing 50 mL of toluene and 12 mL of 3-APTES. This mixture was stirred with reflux for 24 h at 110 °C under a nitrogen atmosphere. Finally, the resulting product was filtered and washed with ethanol. After removing the solvent residues, the final sample was dried at 50 °C for 24 h to complete the functionalization process.
Schematic flowchart for the functionalization of SF with 3-APTES.
Preparation of Acrylic@SF Nanocoating
In this study, acrylic paint was used as the base paint. Acrylic@SF nanocoating was produced by uniformly dispersing different percentages (0.5–1.5 wt%) of SF NPs in acrylic. For this purpose, first 0.5 g of SF NPs was stirred in 99.5 g of acrylic for 10 min using a mixer and then subjected to ultrasonic treatment for 1 h. The resulting sample was named SF-0.5. The SF-1 and SF-2 nanocoatings were also prepared in the same way, except that the amount of SF was different. The resulting nanocoatings were used to investigate thermal conductivity.
Preparation of Acrylic@F-SF Nanocoating
The 2.3 method was used to synthesize of Acrylic@F-SF nanocoating, but here F-SF NPs were used.
Result and discussion
Characterization
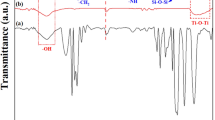
FT-IR investigated the functional groups of the synthesized samples, and the results are reported in Fig. 2. The FT-IR of SF NPs indicates stretching vibrations related to Si–O bonds in the tetrahedral [SiO₄] structure. These vibrations include symmetric and asymmetric modes that can be detected in 804 cm− 1 and 900–1300 cm− 1 (Fig. 2a; Table 1)32. In addition, a band with maximum absorption around 471–480 cm− 1 is attributed to the siloxane framework’s deformation vibrations, indicating this network’s structural flexibility32. After the functionalizing of SF NPs with 3-APTES, slight changes in the chemical environment around the Si–O groups and other surface groups occur. These changes result in small changes in the vibrational energy of the bonds and are observed as slight shifts in the positions of the FT-IR peaks. The formation of hydrogen bonds and new interactions between functional groups and other functional groups also influence these changes. These shifts are indicative of a successful synthesis31. The presence of absorption bands related to 3-APTES can be observed, which are represented by different vibrational modes associated with NH2, CH2 and Si-O bonds. The peak in the region 3300–3700 cm− 1 is related to the asymmetric and symmetric stretching modes of NH2 as well as the hydrogen stretching mode in Si-OH groups. CH2 stretching modes are detected at 2932 cm− 1 and 2883 cm− 1. The peak at 1562 cm− 1 is assigned to the NH2 of the amino groups, which weakly hydrogen bond with the silanol groups and form cyclic structures. Also, the Si-CH2 deformation mode and the asymmetric stretching modes of the Si-O-Si bond are observed, with peaks at 1410 cm− 1 and 1130 cm− 1, respectively. In addition, the siloxane groups appear with peaks at 471–480 cm− 1. These results indicate that APTES has been successfully deposited on the SF surface (Fig. 2b; Table 1).
FT-IR results of (a) SF NPs, and (b) F-SF NPs.
Figure 3 shows the XRD pattern of SF and F-SF powder, which indicates the amorphous structure of this material. In Fig. 3a, the broad and prominent peak at an angle of about 22.3° suggests the presence of nano-sized amorphous silica. Similar patterns have been observed in previous studies on nano- SF and silica NPs prepared by the sol-gel method33,34,35,36.
A similar previous peak is seen in the XRD pattern of SF after functionalization (Fig. 3b) without any apparent change because the amorphous silica NPs is generally stable, and surface functionalization does not cause any fundamental shift in the location or structure of the crystalline base. In other words, functionalization usually causes surface and chemical changes in the particles that may reduce the peak intensity. However, the peak location (angle) representing the spacing of the nanostructure layers remains unchanged, and the peak appears similar to the previous one. This stability of the peak location is due to the preservation of the amorphous structure and nanophase of silica, which indicates that the functionalization is limited to the particle surface and has not affected the overall phase structure.
XRD diffraction pattern of (a) SF NPs, and (b) F-SF NPs.
The morphology of SF and F-SF samples was examined using SEM analysis, and the results are presented in Fig. 4. As can be seen in Fig. 4a, b, the SF NPs formed a porous and interconnected structure with a honeycomb-like arrangement (hexagonal cells)37. Interestingly, after the functionalization of these NPs with 3-APTES, as shown in Fig. 4c, d, the porous and regular structure of the sample was fully preserved, indicating the high stability of the main SF skeleton against the functionalization process. In general, the functionalization of SF with 3-APTES is mainly a surface reaction, meaning that only functional groups are added to the surface of the NPs, and the overall structure or morphology of the particles does not change significantly. This process not only does not cause particles to stick or aggregate but leaves their porous network and original appearance intact. As a result, the main skeleton of SF NPs remains stable and preserved even after chemical modification. Also, the SEM measurement results showed that the silica NPs in both samples (functionalized and unfunctionalized) have a nearly similar size, ranging from 18 to 39 nm. This consistency in particle size allowed the effects of factors such as surface functionalization and better dispersion of NPs in the dye matrix to be interpreted without the complex effect of changing particle dimensions.


SEM images of (a, b) SF NPs, and (c, d) F-SF NPs.
TEM images of F-SF are presented in Fig. 5. After the 3-APTES functionalization process, it was observed that the SF NPs maintained their spherical structure and porous characteristics well, indicating these NPs remarkable morphological stability during the surface modification process38,39,40,41. The preservation of the spherical structure of the particles can be due to the reduction of the surface energy and the tendency of silica particles to maintain the shape that provides the highest energy balance, resulting in a uniform distribution of particles. 3-APTES functionalization, which is based on covalent bonds between the amine groups and the silica surface, causes chemical modification of the surface without significantly changing the physical and morphological structure of the particles, which is very important for various applications.
TEM image of F-SF at (a) 100x magnification, (b) 300x magnification.
To confirm the functionalization process’s success and investigate the distribution of elements on the surface of the NPs, EDX mapping analysis was used. This method allows for the precise observation and mapping of the elements present in the sample. It is vital for assessing the presence and uniform distribution of new elements resulting from functionalization (such as nitrogen from amine groups) (Figs. 6 and 7). The EDX mapping results showed that the expected elements were uniformly distributed on the surface of the NPs, indicating the success of the chemical modification of the surface and the preservation of the original structure of the sample after the functionalization process. Based on the results, SF sample contains about 57.47% oxygen and 42.53% silicon. After functionalization, the oxygen, carbon, silicon and nitrogen contents were reported to be 38.59%, 30.94%, 26.25%, and 4.22%, respectively. The presence of nitrogen indicates the success of the attachment of 3-APTES to the silica surface, as nitrogen is a specific element of this organic agent and its presence confirms the functionalization process.
Elemental mapping and EDX of SF NPs.
Elemental mapping and EDX of F-SF NPs.
The effect of SF and F-SF NPs on the adhesion, hardness, and gloss properties of water-based acrylic paint
The cross-cut adhesion test was performed according to ASTM D3349-A to evaluate the adhesion of the paint to the substrate (Table 2). In this test, a network of cuts is made on the paint coating and then the pressure-sensitive adhesive is stretched over it and removed. The degree of paint removal is evaluated based on a scale from 0 to 5B, with higher values indicating better adhesion. The results show that the control base paint has lower adhesion than the samples containing SF, and the use of F-SF significantly increases adhesion. By increasing the additive percentage to about 1.5%, adhesion improves and a value of 5B is observed in the samples with F-SF NPs compared to 4B in the samples without the agent, indicating improved bonding between the NPs and the paint substrate.
The hardness of the paint coatings was also measured using a Pendulum Hardness Test at a 6° angle to measure the mechanical strength of the coatings (Table 3; Fig. 8a). The impact test was conducted using a two-pound weight with a spherical bottom surface dropped from a maximum height of 100 cm. This setup was used to evaluate the sample’s resistance to sudden impact and assess its mechanical behavior under these conditions. The results of this test show that the hardness of samples containing F-SF is much higher than that of pure paint and samples containing SF. This increase in hardness is due to the improvement of the polymer network structure and better bonding of the NPs to the acrylic matrix, which results in better mechanical strength and longer durability of the paint.
Also, the gloss of the paints was measured according to ASTM D523 at a 60° angle to investigate the effect of SF on the surface appearance of the coating (Table 4; Fig. 8b). The results show that the presence of F-SF significantly increases the gloss. The addition of about 1% F-SF showed the greatest effect, which is due to a more uniform distribution of particles and proper surface interaction with the acrylic matrix in the coating. These results indicate that the use of F-SF improves not only the mechanical properties but also the appearance characteristics of water-based acrylic paints.
Comparison of (a) bardness, (b) adhesion test results of water-based acrylic paint with SF additive and F-SF NPs.
The effect of adding SF and F-SF on the thermal insulation properties of paint
In this study, SF was used as an NPs filler. First, SF was functionalized with the chemical agent 3-APTES to improve its adhesion and surface interaction with the water-based acrylic paint matrix. SF and F-SF NPs were added to water-based acrylic paint in various weight percentages (0.5–1.5 wt%), and paint samples were prepared. K tests were performed on the paint samples to evaluate the effect of adding SF and F-SF NPs on the paint’s thermal properties (Fig. 9; Table 5). According to the data of the samples, it is observed that the base K is equal to 0.420 W/mK. The addition of SF at different percentages reduces the K. For example, the FS-1 sample reduces the K to 0.326, which is equivalent to a 22% improvement. The samples containing F-FS show an even greater reduction, so that F-FS-1 with a K of 0.281 W/mK has a 33% improvement over the base sample. This reduction in K means an improvement in the paint’s thermal insulation properties and indicates the positive effect of NPs in improving the insulation properties of the coating42. This difference can be explained as follows: 3-APTES functionalization creates surface functional groups that, in addition to increasing compatibility and better bonding with the paint matrix, improve the dispersion of NPs within the acrylic paint. Better dispersion causes the particles to be more uniformly placed in the matrix, and the heat transfer paths are severely disrupted. As a result, the overall K of the functionalized paint is reduced more effectively. Functionalization may increase the relative porosity in the paint structure or create better structural cohesion between the particles and the matrix; this makes heat transfer more difficult and further reduces the K coefficient. Pure NPs usually tend to aggregate, which can facilitate heat transfer paths to a greater or lesser extent. However, in the 3-APTES functionalized sample, due to the coating of the agent on the particles, the aggregation is less, and the heat transfer becomes indirect and more resistant. The 3-APTES agent creates stronger chemical bonds between the NPs and the paint, which can create non-uniform phase boundaries and discontinuities in the thermal matrix that impede heat flow. These results agree with previous findings on using functionalized NPs to improve the thermal and mechanical properties of polymer coatings.
The effect of different percentages of SF and F-SF NPs on the K of the samples.
According to the SEM images revealed that the size of silica NPs in both SF and F-SF samples remained constant in a similar range, between 18 and 39 nm. This stability in particle size allows for a more detailed study of the effect of surface functionalization and better dispersion of NPs in the paint matrix without the influence of confounding variables such as particle size variation. Therefore, the observed decrease in thermal conductivity can be attributed mainly to improved NP distribution and enhanced surface interactions with the matrix, rather than changes in particle dimensions. As illustrated in Fig. 10, the incorporation of nanometer-sized F-SF NPs within the coating matrix significantly elongates and complicates the heat transfer path, as indicated by the black arrows, compared to a simple, unmodified coating. This increased path complexity effectively slows down the heat transfer rate from the surface to the substrate, providing a strong thermal barrier. Specifically, the uniformly dispersed F-SF NPs cause the heat transfer path to bend, deflect, and rotate around the particles, creating a tortuous route. This tortuous heat transfer path elevates the thermal resistance of the coating relative to nanoparticle-free samples. Furthermore, part of the thermal energy is scattered and collisions with the NPs reduce heat conduction efficiency, thereby lowering the overall thermal conductivity of the coating. Besides physically extending the heat transfer path, the presence of these NPs disrupts the polymer matrix’s effective thermal conductivity. Consequently, the coating structure, due to the homogeneous NP distribution, facilitates the formation of these tortuous paths, which collectively increase thermal resistance and transform the coating into an effective thermal insulator, slowing heat flow from the surface to the substrate.
Schematic of heat transfer in the water-based acrylic coating with F-SF NPs.
Conclusion
The present work showed that SF NPs, both in pure form and after functionalization with APTES, play an effective role in reducing the K of water-based acrylic paints, in other words, improving their thermal insulation properties. The 22% reduction in K in the sample equipped with pure SF NPs indicates the particles’ ability to trap and reduce heat transfer by creating a porous and heterogeneous structure in the paint, which increases thermal resistance. On the other hand, the F-SF NPs, which showed a 33% reduction. In particular, F-SF performs better than pure SF due to the increased adhesion and more uniform dispersion of the particles in the paint matrix, which is achieved through the functionalization process with APTES. This agent creates a stronger surface interaction between the NPs and the paint matrix, improving its thermal insulation properties. This highlights the importance of optimizing the type and amount of functionalizer to maintain or improve thermal insulation properties while maintaining proper dispersion. The addition of F-SF to the water-based acrylic paint significantly enhanced adhesion (up to 5B), hardness, and gloss due to improved NPs dispersion and interfacial bonding within the coating matrix. Finally, using these NPs, while increasing the thermal performance of acrylic paints, is an important step towards producing heat-resistant, high-performance, and environmentally friendly products that have applications in the construction, automotive, and energy industries. For future work, optimizing the NP concentration and the type and amount of functionalizer is essential to balance dispersion quality and thermal performance. Long-term stability studies and tests under real environmental conditions are recommended to validate practical applications. These directions will further advance the development of durable, high-performance, and environmentally friendly thermal insulating coatings suitable for use in construction, automotive, and energy industries.
Data availability
All data generated or analyzed during this study are included in this published article.
References
Saleh, A. N., Attar, A. A., Ahmed, O. K. & Mustafa, S. S. Improving the thermal insulation and mechanical properties of concrete using Nano-SiO2. Results Eng. 12, 100303 (2021).
Ali, M. M., Ahmed, O. K. & Abbas, E. F. Performance of solar pond integrated with photovoltaic/thermal collectors. Energy Rep. 6, 3200–3211 (2020).
Martínez-Molina, A., Tort-Ausina, I., Cho, S. & Vivancos, J. L. Energy efficiency and thermal comfort in historic buildings: A review. Renew. Sustain. Energy Rev. 61, 70–85 (2016).
De Giuli, V., Da Pos, O. & De Carli, M. Indoor environmental quality and pupil perception in Italian primary schools. Build. Environ. 56, 335–345 (2012).
Abed, F. M., Ahmed, O. K. & Ahmed, A. E. Effect of climate and design parameters on the temperature distribution of a room. J. Building Eng. 17, 115–124 (2018).
Darweesh, H. Influence of sun flower stalk Ash (SFSA) on the behavior of Portland cement pastes. Results Eng. 8, 100171 (2020).
Synnefa, A., Santamouris, M. & Livada, I. A study of the thermal performance of reflective coatings for the urban environment. Sol. Energy. 80, 968–981 (2006).
Rosenfeld, A. H. et al. Mitigation of urban heat islands: materials, utility programs, updates. Energy Build. 22, 255–265 (1995).
Simpson, J. & McPherson, E. The effects of roof albedo modification on cooling loads of scale model residences in Tucson, Arizona. Energy Build. 25, 127–137 (1997).
Lu, Y., Liu, Z., Li, X., Yin, X. J. & Utomo, H. D. Development of water-based thermal insulation paints using silica aerogel made from incineration bottom Ash. Energy Build. 259, 111866 (2022).
Holman, J. P. Heat Transfer (Si Units) Sie, Tata McGraw-Hill Education (2008).
Singh, G., Vasudev, H., Kumar, M., Singh, K. & Hassan, M. M. Advancements in Vision Sensing Technologies for Automated Welding Processes: A Sustainable Approach, International Conference on Emerging Materials, Smart Manufacturing and Computational Intelligence, Springer, pp. 123–132. (2024).
Choudhari, A. V. et al. Enhanced antibacterial efficacy of Ag@ ZnTiO₃ nanocomposites: green synthesis using Gaga leaf extract and mechanistic insights. J. Solgel Sci. Technol. 115, 1636–1649 (2025).
Gaikwad, M. U. et al. Fatigue analysis of electro discharge machined nitinol 60. Innov. Emerg. Technol. 10, 2340009 (2023).
Dey, D. et al. Investigation and characterization of novel aluminium matrix composites enhanced by recycled borosilicate glass. J. Inorg. Organomet. Polym Mater. 35, 3439–3453 (2025).
Deflorian, F. et al. Study of the effect of organically functionalized silica nanoparticles on the properties of UV curable acrylic coatings. Prog. Org. Coat. 72, 44–51 (2011).
Cueto-Díaz, E. J. et al. E. Mateo-Martí, Aptes-based silica nanoparticles as a potential modifier for the selective sequestration of CO2 gas molecules Vol. 11, 2893 (Nanomaterials, 2021).
Phan, M. V. et al. Controllable synthesis of Hollow silica nanoparticles using layered double hydroxide templates and application for thermal insulation coating. ACS Omega. 8, 31399–31409 (2023).
Makwana, M. et al. Influence on dynamic behaviour of single layer graphene by stone Wales and pinhole defects. Adv. Mater. Process. Technol. 10, 198–209 (2024).
Sukhummek, B., Angkaew, S., Chonkaew, W., Pongcharoen, K. & Lumpuengkul, K. The effect of titanium dioxide and additives on heat reflection and thermal reduction of paint. Key Eng. Mater. 545, 95–100 (2013).
Givoni, B. & Hoffman, M. Effect of building materials on internal temperatures, (1968).
Uemoto, K. L., Sato, N. M. & John, V. M. Estimating thermal performance of cool colored paints. Energy Build. 42, 17–22 (2010).
Gonome, H., Nakamura, M., Okajima, J. & Maruyama, S. Artificial chameleon skin that controls spectral radiation: development of chameleon cool coating (C3. Sci. Rep. 8, 1196 (2018).
Song, J. et al. The effects of particle size distribution on the optical properties of titanium dioxide rutile pigments and their applications in cool non-white coatings. Sol. Energy Mater. Sol. Cells. 130, 42–50 (2014).
Taha, H., Sailor, D. J. & Akbari, H. High-albedo materials for reducing building cooling energy use, (1992).
Synnefa, A., Santamouris, M. & Livada, I. A comparative study of the thermal performance of reflective coatings for the urban environment, Proceedings, pp. 101–108. (2005).
Abutalib, N., Karnati, S. R., Oldham, D., Zhang, L. & Fini, E. Surface modification of silica fume with amine groups to reduce agglomeration and improve asphalt resistance to oxidation. Res. Rev. J. Mater. Sci. 4, 1065–1072 (2017).
Fricke, J. & Emmerling, A. Aerogels—Preparation, properties, applications, Chemistry, Spectroscopy and Applications of Sol-Gel Glasses, Springer 2005, pp. 37–87.
Hrubesh, L. W. Aerogels: the world′ s lightest solids. ChemInform 22, no–no (1991).
Hüsing, N. & Schubert, U. Aerogels—airy materials: chemistry, structure, and properties. Angew. Chem. Int. Ed. 37, 22–45 (1998).
Marconi, E. et al. 3-APTES on dendritic fibrous mesoporous silica nanoparticles for the pH-controlled release of corrosion inhibitors. Nanomaterials 13, 2543 (2023).
Myronyuk, I., Kotsyubynsky, V., Dmytrotsa, T., Soltys, L. & Gun’Ko, V. Atomic structure and morphology of fumed silica. Phys. Chem. Solid State. 21, 325–331 (2020).
Al-Hasnawi, A. A. K. & Al-Hydary, I. A. D. The devitrification kinetics of transparent silica glass prepared by gel-casting method. Matéria (Rio De Janeiro). 24, e–12317 (2019).
Maddalena, R., Hall, C. & Hamilton, A. Effect of silica particle size on the formation of calcium silicate hydrate [CSH] using thermal analysis. Thermochim. Acta. 672, 142–149 (2019).
Wang, Y. et al. Synergistic effect of nano-silica and silica fume on hydration properties of cement-based materials. J. Therm. Anal. Calorim., 140 (2020).
Nkanpa Moffo, N. D., Mwero, J. & Gariy, Z. A. Performance of silica fume on preventing strength retrogression in hardened cement paste and mortar at elevated temperatures. Buildings 13, 1301 (2023).
Cheraghian, G. & Wistuba, M. P. Effect of fumed silica nanoparticles on ultraviolet aging resistance of bitumen. Nanomaterials 11, 454 (2021).
Milionis, A., Ruffilli, R. & Bayer, I. S. Superhydrophobic nanocomposites from biodegradable thermoplastic starch composites (Mater-Bi®), hydrophobic nano-silica and lycopodium spores. RSC Adv. 4, 34395–34404 (2014).
Zanoletti, A., Bilo, F. & Borgese, L. SUNSPACE, a porous material to reduce air particulate matter (PM). Front. Chem. 6, 534 (2018).
Jafari, V. & Allahverdi, A. Synthesis and characterization of colloidal Nanosilica via an ultrasound assisted route based on alkali leaching of silica fume. Int. J. Nanosci. Nanatechnol. 10, 145–152 (2014).
Abd El-Gawad, W. M. Utilization of affordable nanocomposites with outstanding antimicrobial activity in waterborne coatings. Sci. Rep. 15, 20288 (2025).
Silva, J. F. G., Rossi, N. R., de Menezes, B. R. C., Thim, G. P. & Junior, T. J. A. P. Effect of silver-coated silica nanoparticles on the thermal conductivity of thermally activated acrylic resin. Brazilian Dent. Sci., 25, 1–9 (2022).
Funding
The authors are thankful for the financial support accepted by the research councils of the Nanotechnology Research Center, Research Institute of Petroleum Industry.
Author information
Authors and Affiliations
Contributions
Mina Aghaee Farkoush: Doing the lab work and collecting data, writing of the original draft, literature review, andcreating graphs and tables.Alimorad Rashidi: Conceptualization, supervision, and correspondence. Mahshad Alaei: Creating figures and editing.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Farkoush, M.A., Rashidi, A. & Alaei, M. Thermal insulation of water-based acrylic coatings reinforced with APTES-functionalized silica fume nanoparticles. Sci Rep 16, 2361 (2026). https://doi.org/10.1038/s41598-025-32043-5
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-32043-5