Abstract
Due to the specific reservoir characteristics of coalbed methane (CBM) in Qinshui Coalfield of Shanxi Province, the scale of gas drainage in coal mines is limited and CBM extraction remains challenging. In an effort to modify the adsorption performance of coalbed gas and enhance CBM recovery, the application of an external electric field as a stimulating method was investigated. This study focuses on #9 and #15 anthracite samples from the southern part of Qinshui Coalfield. Comprehensive coal quality analyses were conducted. An experimental device was developed to study CBM adsorption under the influence of electric fields, which served as an external physical stimulation field. Using this apparatus, adsorption isotherms, the maximum adsorbed quantity, and an empirical adsorbed constant of #9 and #15 coal samples were experimentally determined. Based on pore structure parameters, the influence of electric field enhancement on CBM adsorption was examined.The results indicate that electric fields significantly reduce CBM adsorption capacity compared with before the action of an electric field. With increasing electric field strength, the maximum adsorbed quantity decreases linearly, while the empirical adsorbed constant decreases exponentially. The reduction in CBM adsorption capacity following electric field treatment is attributed to the increase in median diameter of mesopores in coal, which lowers the surface potential energy of pores in coal, thus reducing its adsorption capacity for methane.Furthermore, the increase in the median pore diameter is more conducive to the migration and diffusion of methane gas, thereby improving the methane extraction rate and extraction efficiency. This study has certain theoretical significance for the extraction of methane from CBM reservoirs with low pressure and low permeability.
Similar content being viewed by others
Introduction
As a representative form of clean energy, the exploration and development of coalbed methane (CBM) holds the potential to partially replace coal, thereby contributing to the mitigation of environmental pollution associated with the large-scale utilization of coal and other fossil fuels1,2,3,4,5,6,7. The Qinshui Coalfield in Shanxi, one of the most significant mining regions in northern China, boasts rich CBM resources, with reserves reaching as high as 500 billion cubic meters8. The extraction and utilization of these CBM resources have become key research topics. According to available data, the CBM reservoirs in the Qinshui Coalfield are characterized by low methane pressure and permeability. These characteristics result in limited gas drainage capacity and significant fluctuations in gas concentration within the mine9,10. The challenges associated with CBM recovery are considerable. A thorough study of the adsorption behavior of coal to methane is critical for enhanced CBM recovery11.
Based on the generation, accumulation, and migration mechanisms of CBM, reduce the adsorption properties of CBM to facilitate the transformation of methane adsorbed on the inner surfaces of coal matrix pores and fractures from an adsorbed state to a free state, and to expand the migration pathways for methane, further to enable CBM to penetrate through more pores and fractures into the extraction drill holes. Currently, research aimed at reducing the adsorption of CBM mainly focuses on mechanical approaches, including methods such as water and gas injection displacement12,13,14,15,16, high-pressure hydraulic fracturing17,18,19,20, gas drainage from coal seam roof21, mining of protective layers22, depressurisation extraction23,24, and borehole drainage25. These methods can significantly reduce gas pressure during the early stages of extraction and have achieved relatively good extraction results. However, they also have the following drawbacks: in the later stages of extraction, as the gas pressure gradually declines and the driving force becomes insufficient, the adsorbed CBM on the surface of coal cannot be desorbed or discharged after reaching the depressurization limit. The number of fractures generated within the coal matrix remains limited, and the formed blocks are relatively large, meaning that the time required for complete methane dissipation is prolonged.
He et al.26,27,28 conducted studies on adsorption characteristics of CBM under the influence of electromagnetic fields and pointed out that: during the vacuum extraction phase, the electromagnetic field has no effect on methane adsorption; however, during the adsorption equilibrium phase, the electromagnetic field reduces adsorption capacity of coal for methane, increases the diffusion rate, alters the coal surface adsorption potential well, has a minor impact on the saturated adsorption capacity, but decreases the adsorption constant. The electromagnetic field increases the London dispersion potential of methane molecules, enhances their dynamic properties, and increases diffusion permeability. Nie et al.29 also studied the effect of electromagnetic fields on methane adsorption, noting that the electromagnetic field generates a thermal effect in the coal body, influencing the interactions between methane molecules and coal macro-molecules. They observed that the reduction in adsorption potential well of CBM is positively correlated with the frequency and intensity of the electromagnetic field. Liu et al.30,31 researched methane adsorption under electric field influence, with field voltages of 0.8 kV, 1.2 kV, and 1.6 kV, and concluded that: under the treatment of an electric field, the methane adsorption follows the Langmuir model, with reduced methane adsorption capacity. The electric field causes an increase in the temperature of the methane, reduces the coal surface adsorption potential well, and decreases the methane adsorption. Yi et al.32 studied methane adsorption under electric field influence at a frequency of 50 Hz and voltages of 0 V, 800 V, 1200 V, and 1600 V, and found that: methane adsorption under the electric field follows the Langmuir model, with minor changes in the maximum adsorbed quantity as the electric field voltage increases, while the empirical adsorbed constant decreases. Du33 studied methane adsorption under electric field influence and noted that: methane adsorption in the electric field follows the Langmuir model, and as the electric field strength increases, there is little change in the maximum adsorbed quantity, but the gas adsorption amount increases. Lei et al.34,35 studied the adsorption characteristics of CBM under the action of direct current electric fields and reported that: at electric field strengths of 30, 60, and 90 kV, the methane adsorption decreased by 10.9%, 15.7%, and 18.9%, respectively, and with increasing electric field strength, the initial desorption rate increased. Xu36 found that under the influence of a static electric field, the adsorption capacity of the BaiJiao mine coal sample decreases.
Upon analyzing the above studies, it is evident that non-mechanical methods, such as the application of electric fields, do influence the adsorption characteristics of CBM. However, the effects of electric fields on methane adsorption exhibit varying, and sometimes contradictory patterns. Coal is a typical porous medium, and pore structures in coal govern its adsorption and permeability properties37. The pore structure significantly affects both the adsorption capacity and the extraction recovery of methane38. In response to the low gas pressure in the southern Qinshui Coalfield, applying an external field to influence the methane adsorption process presents an innovative method, offering a new approach to enhance CBM recovery.
Currently, isothermal adsorption experiments are the most effective method for studying methane adsorption capacity in coal39,40. In this study, coal samples from two different coal seams within the same region of the southern Qinshui Coalfield were collected. Based on the coal quality analysis of experimental samples, the adsorption properties of CBM were studied under a high-pressure variable-frequency electric field. By analyzing pore structures, this research investigates how electric field enhancement influences the adsorption of CBM, thereby indirectly enhancing CBM recovery rate.
Experimental
The samples
The coal quality of samples was analyzed. The macroscopic coal characteristics are described as black to dark grey, with a bright lustrous surface, a glassy metallic sheen, and a banded structure. The coal displays a laminated texture with step-like and shell-like fractures, grey-black streaks, and poorly developed fissures. Microscopically, the coal is primarily composed of vitrinite, with inertinite being the secondary component. The samples represent anthracite coal, characterized by low volatility, low ash content, low sulfur content, and high calorific value. Collect samples from coal lumps, crush and grind them, pass them through a 0.25–0.18 mm sample sieve, weigh 200 g of the coal sample after passing through the sample sieve on a 1000− 1 analytical balance, and use it as the test coal sample. Subsequently, the samples were ground to an 80-mesh size and dried in a vacuum at 60 °C for 48 h to eliminate moisture. Table 1 presents the coal quality test results for samples.
According to Technical specification for gas drainage in coal mines41, the difficulty of coal seam gas drainage is primarily measured by two indicators: the coal seam permeability coefficient and the borehole gas flow attenuation coefficient. When the coal seam permeability coefficient is less than 0.1 m²/MPa²·d and the borehole gas flow attenuation coefficient is greater than 0.05d− 1, the coal seam is considered to be difficult for gas drainage. Table 2 presents basic gas parameters for the #9 and #15 coal samples.
From Table 2, it can be observed that the coal seam permeability coefficients of #9 and #15 experimental coal samples are 0.073 m²/MPa²·d and 0.086 m²/MPa²·d, respectively, both of which are below 0.1 m²/MPa²·d. Additionally, the borehole gas flow attenuation coefficients for the #9 and #15 samples are 1.2668 d⁻¹ and 0.9234 d⁻¹, respectively, both exceeding 0.05 d⁻¹. Based on these values, both coal seams are classified as difficult to drain.
Experimental methodology and setup
The experimental apparatus employed for investigating methane adsorption in coal under electric field treatment is depicted schematically in Fig. 1.
Schematic diagram of the experimental apparatus.
1-Gas cylinder; 2-Pressure pipeline; 3-Valve; 4-Gas container; 5-Drying agent; 6-Pressure sensor; 7-Adsorption container; 8-Anode electrode; 9-Sample; 10-Constant-temperature bath; 11-High-tension wire; 12-High Voltage Inverter Power Supply; 13-Vacuum pump; 14-Data acquisition unit; 15-Computer; 16-Rubber tube; 17-Burette; 18-Conical flask; 19-Saturated NaCl.
The experimental setup primarily comprises a methane purification system, a flow measurement system, a pressure monitoring system, a temperature monitoring system, an electric field application system, a methane adsorption data acquisition system, and adsorption isotherm plotting software. The methane purity is 99.99%, with desiccant being color-changing silica gel. Methane is fed into the storage tank via a methane cylinder, pressure-resistant gas supply pipe, and valves. The output end of the storage tank is connected to the flow meter system via a gas washing valve on one side. The other end is connected to the electric field system via a high-pressure inflation valve. The vacuum pump model is 2XZ-2, with a measurement accuracy of 2 L·s− 1. The pressure sensor exhibits a measurement accuracy of 0.2 MPa. The apparatus comprises two high-voltage frequency conversion power supplies: one supplying voltage output ranging from 0 to 10 kV with a maximum frequency of 7 kHz, and the other providing frequency output ranging from 15 to 80 kHz with a maximum voltage of 40 kV. The adsorption container, designed to house the sample, is a high-pressure vessel with an approximate volume of 300 ml. The anode electrode is a spark plug featuring an external ceramic insulator, while the cathode electrode constitutes the shell of the adsorption container. The temperature monitoring system is a constant-temperature water bath with a control accuracy of ± 0.5℃ and a measurement range of 0–99.9℃. The temperature sensor has a measurement accuracy of 0.1℃ and is model 18B20. During the test, the constant-temperature water bath operates at a temperature of 40℃, and the vacuum extraction time is 5 h.
The test coal samples were pretreated and placed in an adsorption tank. After vacuum degassing was complete, an electric field was applied to the coal samples in the adsorption tank under constant temperature conditions to simulate the average geothermal gradient of deep coal seams in China, with the temperature set to 40 °C. Figure 2 shows a schematic diagram of the adsorption system. The applied electric field voltage ranged from 0 to 5 kV, with a frequency from 0 to 80 kHz, complying with the IEEE C95.1–2019 standard for a 10-fold redundancy design for biocompatible field strengths below 3 kHz. A voltage of 5 kV was sufficient to induce pore structure reorganization in the test coal samples while avoiding breakdown. The dielectric relaxation characteristic frequency range of the test coal samples was 65–75 kHz, and the critical frequency of 80 kHz avoids significant temperature rise (< 2℃) caused by dielectric loss.
Real Image of Adsorption System Diagram.
The total pore area S, volume median pore size Dv, area median pore size Da, and average pore diameter d of experimental coal samples before and after electric field enhancement were determined using mercury intrusion porosimetry (MIP) method. The testing instrument used was Micromeritics™ Autopore IV 9500 fully automated mercury porosimeter, which includes two low-pressure stations and one high-pressure station. The lower and upper pore size limits from MIP were determined based on mercury intrusion pressure, using the empirical Washburn equation. The vacuum level of the low-pressure degassing station was 5 × 10− 3torr, and the low-pressure analysis range was 3–300 kPa, with a pore size measurement range of 3.6 nm ~ 360 μm. The high-pressure analysis pressure was 20 MPa, and the pore size measurement range was 5.5 nm ~ 1000 μm. All tests were conducted in strict compliance with the national standard GB/T 21650.1–2008.
There are various gas adsorption models, among which Langmuir’s theory of adsorption is the most widely used. Numerous experimental studies have shown that the Langmuir adsorption model effectively and correctly describes isotherm adsorption of CBM11,42. This model was employed to assess the variation in methane adsorption capacity of the coal samples. The equation is expressed as follows:
where:
P is an equilibrium pressure of the adsorbed high-pressure gas (MPa),
V is the adsorbed quantity at the equilibrium pressure P per unit mass of coal (ml·g− 1),
Vm is the maximum adsorbed quantity (ml·g− 1),
B is an empirical adsorbed constant (MPa− 1).
Experimental conditions
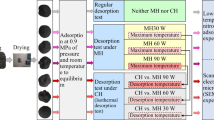
Methane adsorption characteristics under the influence of a high-voltage inverter electric field were assessed using an equilibrium volumetric method. To investigate the adsorption behaviour of methane in coal under electric field treatment, 12 experimental conditions were established. Each group consists of three test samples, for a total of 36 test coal samples, meaning that each group of tests was repeated three times, and the average value was taken for data analysis. The voltage range applied was from 0 to 10 kV, with a frequency range of 0 to 80 kHz. Specifically, voltage levels of 0, 1 kV, 3 kV, and 5 kV were selected, while the frequencies chosen were 0, 7 kHz, 40 kHz, and 80 kHz.
Results
Influence of electric field enhancement on the methane adsorption isotherm
Figure 3 shows methane adsorption isotherms of #9 and #15 experimental coal samples at an electric field frequency of 80 kHz, with applied voltages of 0 kV, 1 kV, 3 kV, and 5 kV. The fitting method adopted was nonlinear curve fitting, using the Langmuir model. The residual sum of squares ranged from 0.928 to 1.145, and the reduced chi-square values ranged from 0.155 to 0.190. The standard error ranged from 0.232 to 0.396.
CH4 Adsorption Isotherm of Coal Samples under Different Electric Voltage Field Enhancement.
In Fig. 3(a), the adsorption isotherms of #9 coal sample follows the Langmuir equation. Under voltage enhancement, as the voltage increases, the maximum adsorbed quantity significantly decreases. Compared to the 0 kV condition, at 1 kV, the maximum adsorbed quantity decreases by 9.1%; at 3 kV, it decreases by 9.3%; and at 5 kV, the decrease is 11.2%. In Fig. 3(b), the adsorption isotherms of #15 coal sample also follows the Langmuir equation. Under voltage enhancement, the maximum adsorbed quantity generally decreases as the voltage increases. The maximum adsorbed capacity decreases by 2.2%−16.6%. This indicates a significant reduction in methane adsorption capacity due to the high-voltage inverter electric field treatment.
Figure 4 shows the methane adsorption isotherms of #9 and #15 coal samples at an electric field voltage of 1 kV and frequencies of 0, 7 kHz, 40 kHz, and 80 kHz. The residual sum of squares lied within the range of 0.106 to 0.665, while the reduced chi-square values ranged from 0.018 to 0.110. The standard error spanned from 0.168 to 0.493.
CH4 Adsorption Isotherms of Coal Samples under Different Electric Frequency Field Enhancement.
In Fig. 4(a), the adsorption isotherms of #9 coal sample follow the Langmuir equation. Under frequency enhancement, as the frequency increases, the adsorption capacity significantly decreases. Compared to the 0 kHz condition, at 7 kHz, the adsorption capacity decreases by 6.0%; at 40 kHz and 80 kHz, the decrease is relatively small, with reductions of 6.3% and 9.1%, respectively. In Fig. 4(b), the adsorption isotherms of #15 coal sample also follows the Langmuir equation. Under frequency enhancement, the adsorption capacity decreases significantly as the frequency increases. Compared to 0 kHz condition, at 7 kHz, the adsorption capacity decreases by 9.1%; at 40 kHz and 80 kHz, the decrease is smaller, with reductions of 15.0% and 16.6%, respectively. From Fig. 4, it can be seen that when the electric field voltage is 1 kV and the frequencies are 0, 7 kHz, 40 kHz, and 80 kHz, the adsorption capacity of #9 and #15 coal samples decreases by a maximum of 19.2% and 25.7%, respectively. When the electric field frequency is 7 kHz, 40 kHz, or 80 kHz, the decrease in methane adsorption capacity is similar to both #9 and #15 samples.
Effect of electric field enhancement on the adsorption constants of CBM
Further analyzing the effect of electric field enhancement on methane adsorption constants. Figure 5 shows the variation of the maximum adsorbed quantity Vm with voltage and frequency under electric field enhancement for #9 and #15 coal samples, based on the Langmuir equations.
Methane Adsorption Constants under the Electric Field Enhancement.
From Fig. 5(a), it can be seen that the maximum adsorbed quantity Vm for both coal samples decreases in a linear pattern with the increase of electric field voltage at an electric field frequency of 80 kHz and voltages of 0, 1 kV, 3 kV, and 5 kV. The slopes for the #9 and #15 coal samples were − 0.677 and − 1.347, respectively, with correlation coefficients of 0.945 and 0.855.
From Fig. 5(b), it is evident that at an electric field voltage of 1 kV, as the frequency increased from 0, 7 kHz, 40 kHz, to 80 kHz, the maximum adsorbed quantity Vm of both coal samples decreased linearly, with slopes of −0.024 and − 0.046, and correlation coefficients of 0.869 and 0.851, respectively. Comparing the effects of voltage and frequency changes, it can be concluded that frequency enhancement had a relatively small impact on the maximum adsorbed quantity Vm of CBM.
Figure 6 illustrates the variation of the empirical adsorbed constant B for #9 and #15 coal samples under electric field enhancement conditions. As shown in Fig. 6(a), when the electric field frequency is 80 kHz, and the voltage increases from 0 kV to 1 kV, 3 kV, and 5 kV, the empirical adsorbed constant B decreases exponentially with increasing voltage. The correlation coefficients for #9 and #15 samples are 0.994 and 0.831, respectively. Figure 6(b) presents the changes in the empirical adsorbed constant B with a fixed electric field voltage of 1 kV and varying frequencies (0, 7 kHz, 40 kHz, and 80 kHz). In this case, B also follows an exponential decay with increasing frequency, with correlation coefficients of 0.936 and 0.964 for #9 and #15 coal samples, respectively.
The Empirical Adsorbed Constant B under the Electric Field Enhancement.
Analysis of methane adsorption in coal based on pore structures
Analysis of the pore structure in coal
The methane in coal is mainly stored in coal pores in an adsorption state, and the quantitative characterization of coal pore structure is the basis for the study of adsorption and desorption process of gases in coal43,44.
The pore structure parameters of coal samples before and after electric field enhancement were determined using the mercury intrusion porosimetry (MIP) method. Figure 7 illustrates the variation in cumulative mercury intrusion volume with pressure under voltage enhancement for #9 and #15 coal samples, while Fig. 8 presents the corresponding data under frequency enhancement conditions.
From these figures, it can be observed that under the treatment of electric fields, the mercury intrusion and extrusion curves of coal are nearly coincident, with minimal differences in mercury intrusion and extrusion volumes. This indicates the presence of a significant number of closed pores and relatively poor pore connectivity in coal. As pressure increases, no pronounced hysteresis phenomenon is observed between the intrusion and extrusion curves, suggesting minimal heterogeneity in coal matrix pores and a relatively uniform pore structure. Additionally, the extrusion curves are positioned above the intrusion curves, indicating that at the same pressure, the volume of mercury intruded into coal pores is lower than the volume extruded under equivalent pressure conditions.
Among tested samples, #15 coal sample subjected to electric fields exhibits a greater difference between mercury intrusion and extrusion volumes, implying slightly improved pore connectivity. Notably, the sample treated at 80 kHz/5 kV demonstrates the highest mercury extrusion efficiency.
Cumulative Intrusion of Coal Samples Varied with the Pressure under the Voltage Enhancement.
Cumulative Intrusion of Coal Samples Varied with the Pressure under the Frequency Enhancement.
Table 3 presents pore structure data obtained from MIP tests conducted on #9 and #15 coal samples under electric field enhancement conditions.
As shown in Table 3, when #9 coal sample is in raw state, its total pore surface area S, volume median pore diameter Dv, area median pore diameter Da and average pore diameter d are 2.977 m²·g−¹, 74.6 nm, 7.8 nm, and 18.6 nm, respectively. Under the condition of electric field enhancement, with the increase of voltage or frequency, total pore area of coal increases, and volume median pore diameter, area median pore diameter and average pore diameter are all increased correspondingly. Specifically, when the frequency is held constant at 80 kHz and the voltage varies, total pore surface area increases by 60.0–71.9.0.9%, volume median pore diameter increased by 29.1–42.0%, area median pore diameter increased by 18.1–29.4%, and average pore diameter increased by 21.3–27.9%. When the voltage is held constant at 1 kV and the frequency varies, total pore surface area increases by 55.7–60.0%, volume median pore size increased by 15.6 ~ 29.1%, area median pore size increased by 12.7 ~ 18.1%, and average pore size increased by 15.9 ~ 19.8%.
When #15 coal sample was in its original state, its total pore area S, volume median pore diameter Dv, area median pore diameter and Da average pore diameter d are 2.205 m2·g− 1, 64.2 nm, 7.5 nm and 17.2 nm, respectively. Under electric field enhancement with varying frequency and voltage, experimental data for #15 sample show a good agreement in trend with that observed in #9 coal sample. Total pore surface area increases to 4.332–5.273 m2·g− 1, representing an increase of 96.5–139%; volume median pore size increases by about 11.9 ~ 26.2%; area median pore size increases by about 23.4 ~ 38.3%; and average pore size increases by about 16.2 ~ 29.4%. These results indicate that the application of an electric field promotes the generation or opening of mesopores in coal. In particular, for #15 coal sample, total pore area was more than doubled, and the effect of electric field stimulation was remarkable.
Effect of pore structure on CBM adsorption parameters
To further explore the influence of pore structure on adsorption performance of methane, total pore area S and average pore diameter d were selected as two key pore structure parameters to be analyzed. As shown in Fig. 9(a), under the influence of electric fields, as total pore area of coal samples increases, the maximum adsorbed quantity Vm decreases exponentially. Vm measures the maximum adsorbed quantity of coal under high-pressure conditions, reflecting the overall adsorption capability of its pore system. The enhancement by electric fields expands total pore surface area of coal samples, which makes the surface potential energy of pores decrease, and the adsorption capacity of CBM decreases consequently, and its desorption capacity increases in turn.
Variation Curves of Adsorption Parameters with Total Pore Area (S) for Coal Samples.
In Fig. 9(b) the empirical adsorbed constant also decreases exponentially. The physical significance of the empirical adsorbed constant B in the methane adsorption isotherms is42:
In the equation: Ka is the adsorption rate constant; Kd is the desorption rate constant; N₀ is Avogadro’s constant; w₀ is the vibration frequency of the adsorbed gas molecules; τ₀ is the vibration time; q is the depth of the adsorption potential well; R is the universal gas constant; T is the temperature.
From equ. (2), B is the ratio of the adsorption rate constant Ka to the desorption rate constant Kd. The application of electric field enhancement reduces the empirical adsorbed constant B. It can be inferred that electric field enhancement decreases the adsorption rate of coals for methane while increasing the desorption rate. In other words, electric field enhancement leads to a reduction in the adsorption capacity of coal for methane, while the desorption amount increases.
Mechanism analysis of CBM adsorption
(1) Polarization Effect.
The adsorption force between coal and methane gas is Van Der Waals force. Van Der Waals force causes the inner surface of coal pores to form adsorption potential wells. The deeper the adsorption potential wells, the stronger the adsorption capacity of the inner surface of coal pores for methane gas. Under the conditions of electric field enhancement, CBM adsorption involves not only physical adsorption but also chemical adsorption, where the force is the surface chemical bonds formed between methane gas molecules and the coal pore surface. During the reinforcement of the electric field, on one hand, the molecules of coal and methane undergo polarization, which increases the polarization of both coal and methane molecules, thereby enhancing their electronegativity. This leads to a reduction in the depth of the adsorption potential well between the coal and methane molecules. On the other hand, the polarization of the coal and methane molecules reduces the number of free electrons and vacancies in the double electric layer of the coal pore surface, which consequently decreases the adsorption capacity of CBM45,46.
(2) Electron Vacancy Shifts Effect.
According to Table 3, the electric field enhancement led to mesoscopic rearrangement of pores in the coals, which changed the pore structure in coal. The increase in total pore area showed that the pore system of coal became more developed, and the average pore diameter increased, particularly for the pores within the range of the mesopores, which enhanced pore connectivity and created more flow channels47. These changes facilitated a smoother methane flow in the coal, accelerating the diffusion of gas within the pores, and consequently leading to rapid desorption of methane48. As the median pore size increases, the migration and diffusion of methane are facilitated due to the reduced influence of surface potential energy within the coal pores, which in turn indirectly improves the efficiency and effectiveness of CBM drainage40. The purpose of promoting the CBM recovery in coal under the reinforcing effect of the electric field is finally achieved.
Conclusions
Several experiments were conducted to investigate methane adsorption capacity in coal under electric field treatment. The adsorption properties of methane, including adsorption isotherm, maximum adsorbed quantity, and empirical adsorbed constant, were evaluated. The following conclusions can be drawn:
(1) The treatment of the electric field can significantly reduce the adsorption capacity of CBM. Voltage and frequency treatment reduces the methane adsorption of coal samples by a maximum of 25.7%.
(2) As the electric field voltage or frequency increases, the maximum adsorbed quantity Vm decreases linearly, and the empirical adsorbed constant B decreases following a negative exponential pattern.
(3) Based on the pore structure analysis, the mechanism of electric field-enhanced CBM adsorption was analyzed. The increase in total pore area S reduces the surface energy of pores in coal, thereby reducing the adsorption capacity for methane. The increase in median pore size d also facilitates the migration and diffusion of methane, making it easier to get out of control of the potential energy of the surface inside the coal pore, which makes it possible to enhance CBM recovery.
Data availability
The authors confirm that the datasets used during the current study are available from the corresponding author upon reasonable request.
References
Mohamed, T. & Mehana, M. Coalbed methane characterization and modeling: review and outlook. Energy Sour. Part A Recover. Utilization Environ. Eff. 47 (1), 2874–2896 (2020).
Jeffry, S. J. M. et al. Selection of suitable acid chemicals for matrix stimulation: A Malaysian brown field scenario. J. Petrol. Sci. Eng. 186, 106689 (2020).
Katende, A. et al. Experiments and modeling of proppant embedment and fracture conductivity for the Caney Shale, Oklahoma, USA (2022).
Katende, A. et al. A comprehensive review of proppant embedment in shale reservoirs: Experimentation, modeling and future prospects. J. Nat. Gas Sci. Eng. 95, 104143 (2021).
Katende, A. et al. Convergence of micro-geochemistry and micro-geomechanics towards Understanding proppant shale rock interaction: A Caney shale case study in Southern Oklahoma, USA. J. Nat. Gas Sci. Eng. 96, 104296 (2021).
Sheng, Z. & Derek, E. Proppant embedment in coal and shale: impacts of stress hardening and sorption. Int. J. Coal Geol. 227, 103545 (2020).
Bandara, A. et al. Crushing and embedment of proppant packs under Cyclic loading: an insight to enhanced unconventional oil/gas recovery. Geosci. Front. 12 (6), 100970 (2020).
Oil and Gas Resources Strategic Research Centre of Ministry of Land and Resources. The natural oil and gas resources evaluation. Beijing: China Land Press. in Chinese. (2010).
Lu, X. X. et al. Pore structure of deep coal seam in Southern Qinshui basin. J. Northeast Petroleum Univ. 39 (3), 41–49 (2015). in Chinese.
Katende, A. et al. Multidimensional, Experimental and Modeling Evaluation of Permeability Evolution (the Caney Shale Field Lab, 2023).
Satya, H., Basanta, K., Prusty & Dutta, P. Methane/CO2 sorption modeling for coalbed methane production and CO2 sequestration. Energy Fuels. 20, 1591–1599 (2006).
Yu, S. et al. Synergistic ECBM extraction technology and engineering application based on hydraulic Flushing combing gas injection displacement in low permeability coal seams. Fuel 318 (15), 1–15 (2022).
Gong, H. R. et al. Underground coal seam gas displacement by injecting nitrogen: field test and effect prediction. Fuel 306 (15), 1–13 (2021).
Zhang, H. B., Liu, J. S. & Elsworth, D. How sorption-induced matrix deformation affects gas flow in coal seams: A new FE model. Int. J. Rock Mech. Min. Sci. 45 (8), 1226–1236 (2008).
Fan, C. J. et al. Thermo-hydro-mechanical-chemical couplings controlling CH4 production and CO2 sequestration in enhanced coalbed methane recovery. Energy 173 (15), 1054–1077 (2019).
Wu, Y. et al. Dual poroelastic response of a coal seam to CO2 injection. Int. J. Greenhouse Gas Control. 4 (4), 668–678 (2010).
Wang, S. G., Elsworth, D. & Liu, J. S. Permeability evolution in fractured coal: the roles of fracture geometry and water-content. Int. J. Coal Geol. 87 (1), 13–25 (2011).
Cai, Y. D. et al. Permeability evolution in fractured coal-Combining triaxial confinement with X-ray computed tomography, acoustic emission and ultrasonic techniques. Int. J. Coal Geol. 122, 91–104 (2014).
Lu, Y. Y. & Xian, L. I. R. Discussion on the efficient exploitation method of deep coalbed methane with pressure relief by ground directional well hydraulic slotting. J. China Coal Soc. 46 (3), 876–884 (2021). in Chinese.
Zhao, Y. et al. Gas flow in hydraulic slotting-disturbed coal seam considering stress relief induced damage. J. Nat. Gas Sci. Eng. 75 (3), 1–14 (2020).
Yuan, L. & Liu, Z. G. Study on the technique of roof gas drainage from mine coal seam in Huainan mine area. J. China Coal Soc. 28 (2), 149–152 (2003). in Chinese.
Wang, L. et al. Safe strategy for coal and gas outburst prevention in deep-and-thick coal seams using a soft rock protective layer mining. Saf. Sci. 129 (9), 1–12 (2020). in Chinese.
Xue, J. H. et al. Current situation and development trend of pressure-relief gas extraction in the protective layer mining in coal mines in China. Coal Geol. Explor. 51 (6), 50–61 (2023). in Chinese.
Guo, M. J. et al. Position characteristics of horizontal-directional long bore holes in overlying strata and drainage mechanism of pressure-relief gas in long wall mining. J. China Coal Soc. 48 (10), 3750–3765 (2023). in Chinese.
Wang, Y. H. et al. Impact of Goaf gas drainage from surface vertical boreholes on Goaf explosive gas zones. Int. J. Coal Geol. 284 (18), 1–14 (2024).
He, X. Q. The effect of alternative electromagnetic field on adsorption of gas by coal. J. China Coal Soc. 21 (01), 63–67 (1996). in Chinese.
He, X. Q. & Zhang, L. Study on influence and mechanism of gas adsorption and desorption in electromagnetic fields exerted. J. China Coal Soc. 25 (06), 614–618 (2000). in Chinese.
He, X. Q. & Zhang, L. Effect of sine wave electromagnetic fields on gas adsorptive constant. J. China Univ. Min. Technol. 32 (05), 476–478 (2003). in Chinese.
Nie, B. S. et al. Research on the mechanisms of the influence of electromagnetic field on coalbed methane adsorption. Nat. Gas. Ind. 24 (10), 32–35 (2004). in Chinese.
Liu, B. X., Xiong, D. G. & Xian, X. F. Adsorption and seepage characteristics of coal to methane under electric field. J. Chongqing Univ. (Natural Sci. Edition). 29 (02), 83–85 (2006). in Chinese.
Liu, B. X. et al. Experimental investigation gas seepage characteristics in coal under alternating electric field. J. Chongqing Univ. (Natural Sci. Edition). 23 (S1), 41–43 (2000). in Chinese.
Yi, J., Jiang, Y. D. & Xian, X. F. The research on the traits of the absorption and desorption of coal to gas in alternating electric field or sound field. China Min. Magazine. 14 (05), 70–73 (2005). in Chinese.
Du, Y. G. Study on the Seepage Characteristics of Coal Seam Gas in Geophysical Field and the Law of Gas Emergence (ChongQing University, 1995).
Li, C. W. & Lei, D. J. Experimental research of the influence of static electric field on the characteristics of coal gas emission. J. China Coal Soc. 37 (06), 962–966 (2012). in Chinese.
Lei, D. J. & Zheng, Y. Y. Adsorption model and mechanism of coal under high-strength electrical excitation. J. Henan Polytechnic Univ. (Natural Science). 42 (1), 35–45 (2023). in Chinese.
Xu, L. J. Studies on the Ultrafine structure, Electrical properties, and Adsorption Characteristics of Coal from Prominent Areas and their Applications (ChongQing University, 1996).
Liu, X. F. & He, X. Q. Effect of pore characteristics on coalbed methane adsorption in middle-high rank coals. Adsorption 23, 3–12 (2017).
Xu, S. P. et al. Quantitative analysis of pore structure and its impact on methane adsorption capacity of coal. Nat. Resour. Res. 30 (1), 605–620 (2021).
Mohd, A. A., Mohshim, D. F., Maulianda, B. & Elraeis, K. A. Supercritical methane adsorption measurement on shale using the isotherm modelling aspect. RSC Adv. 12, 20530–20543 (2022).
Zhang, Q. et al. Methane Adsorption Capacity of Deep Buried Coal Seam Based on Full-Scale Pore Structure (Natural Resources Research, 2025).
Ministry of Coal Industry. Technical specification for gas drainage in coal mines. (2008).
Chen, S. Y. Adsorption and Catalysis (Henan Science and Technology, 2001).
Gregory, N. O. et al. Comparing the porosity and surface areas of coal as measured by gas adsorption, mercury intrusion and SAXS techniques. Fuel 141, 293–304 (2015).
Han, S. J. et al. Relationship between the geological origins of Pore-Fracture and methane adsorption behaviors in High-Rank coal. ACS Omega. 7, 8091–8102 (2022).
Liu, T. et al. Numerical simulation on enhanced coalbed methane extraction by hot flue gas based on the ECM-DFM model. Comput. Geotech. 184, 107253 (2025).
Wang, Z. Z. et al. Factors influencing the production of coalbed methane from deep reservoirs. Coal Geol. Explor. 53 (02), 84–98 (2025).
Zhang, H. J. et al. Gas emission characteristics of tectonic coal and microscopic explanation of pore structure. J. China Coal Soc. 43 (12), 3404–3410 (2018). in Chinese.
Liu, J. et al. Analysis of the influence of different pore ratios on methane adsorption characteristics of coal. Fuel 358, 130273 (2024).
Acknowledgements
The authors acknowledge that this work was supported by the State Key Laboratory of Coal and CBM Co-mining opening fund (No. 2022KF10), Patent Transformation Program of Shanxi Province (No. 202401006).
Author information
Authors and Affiliations
Contributions
Z.C. and W.H. proposed conceptualization and methodology, and provided drafts; H.L. and H.L. processed experimental data and prepared all figures and tables. All authors reviewed the manuscript and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Chen, Z., Liu, H., Han, W. et al. An experimental study of the effect of electric field enhancement on the adsorption characteristics of coalbed methane. Sci Rep 16, 2296 (2026). https://doi.org/10.1038/s41598-025-32096-6
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-32096-6