Abstract
Facing wild resource depletion, artificial cultivation of Gastrodia elata (G. elata) is crucial. This study compared traditional wood (TW) and artificial mushroom grow kits (AM) cultivation impacts on G. elata quality. High-Performance Liquid Chromatography (HPLC) showed similar gastrodin levels, but TW G. elata had higher 4-hydroxybenzylalcohol (0.05–0.16%) than AM G. elata (0.02–0.04%), while AM G. elata had more parishin E (0.90–1.58%) than TW G. elata (0.68–0.96%). AM G. elata exhibited elevated crude protein (8.86–10.57%) and stable total polysaccharides (38.91–39.90%), contrasting with TW G. elata. Umami amino acids (glutamic acid, aspartic acid) were significantly higher in AM G. elata (6.03–7.37 mg/g, 1.24–1.88 mg/g). Headspace Solid-Phase Microextraction coupled with Gas Chromatography-Mass Spectrometry (HS-SPME-GC-MS) identified thirteen differential volatile compounds, with AM G. elata showing increased esters and aromatics like (2S,3S)-2,3-butanediol and tetramethylpyrazine, contributing sweet/meaty notes. TW G. elata was rich in decanal and (2E,4E)-deca-2,4-dienal, yielding citrus/chicken-like aromas. AM cultivation enhances G. elata’s nutritional and aromatic profiles, supporting its refined application.
Similar content being viewed by others
Introduction
Gastrodia elata (G. elata), a perennial herb belonging to the Orchidaceae family, is a widely recognized plant with dual applications in traditional medicine and culinary practice. Initially recorded as ‘Chijian’ in the Shen Nong Ben Cao Jing (The Classic of Herbal Medicine), it was classified as a superior-grade medicinal herb. Currently, it is included in the Pharmacopoeia of the People’s Republic of China (2020 edition)1. Renowned for its neuroprotective properties, G. elata is extensively used across East Asia to treat various neurological conditions2, primarily owing to bioactive constituents such as gastrodin and polysaccharides3,4. Historically, Zhaotong in Yunnan Province was regarded as the ideal region for its cultivation. Currently, the main production areas in China include Yunnan, Guizhou, Sichuan, Anhui, Hubei, Henan, and Shaanxi provinces5.
Phenolic compounds in G. elata possess diverse pharmacological properties, such as sedative, anticonvulsant, anti-inflammatory, and antioxidant effects2, and are commonly used as markers for quality assessment. Key nutritional components, such as proteins, polysaccharides, and amino acids, play a vital role in its dual application as both a medicinal herb and a food ingredient6,7, Volatile compounds also serve as important indicators in evaluating its quality8,9.
The chemical composition of Chinese herbal medicines often varies with different cultivation practices and geographical origin9,10,11. Zeng et al. found that artificial cultivation within the natural habitat of Cordyceps sinensis significantly increased yield and improved carbon compound and nucleoside content12. Similarly, studies on the high-value orchid Dendrobium officinale demonstrated notable differences in microbial community structure and key chemical components under different simulated natural cultivation conditions13 Liu et al. compared the bioactive components in Poria cocos medicinal material under different cultivation methods using High-Performance Liquid Chromatography (HPLC), revealing consistent triterpenoid profiles but varying concentrations between substrate-based and wood-based cultivation14. In the case of Astragalus membranaceus, simulated wild cultivation led to the production of a novel polysaccharide, APS-II, which significantly potentiated the antitumor effect of cyclophosphamide15. Further research suggests that modifications in cultivation methods can alter soil physicochemical properties and microbial communities, thereby influencing plant growth and the biosynthesis of critical bioactive compounds16.
Gastrodia elata is a valuable medicinal and edible plant species whose wild resources have become increasingly scarce, leading to its classification as rare and endangered17. Currently, artificial cultivation techniques for G. elata, primarily forest-based cultivation mimicking wild growth conditions, are becoming well-established18. This forest-based cultivation model has effectively alleviated pressure on wild populations19. Gong et al. compared the quality of G. elata cultivated in forests and on barren slopes, finding that forest-cultivated G. elata contained more beneficial compounds20. Additionally, Yuan et al. reported that the use of bag cultivation in forest environments can increase land utilization rates to 60–70%21. The forest-based, semi-wild artificial cultivation method relies on robust broadleaf trees, such as oak to provide nutritional support, and co-cultivates with Armillaria (honey mushroom), employing the fixed-bed method for sexual reproduction. The cultivation of the bed not only supports the propagation of the symbiotic Armillaria but also supplies essential nutrients for the subsequent growth of G. elata22,23. However, this method requires pre-cultivation of the fungal bed a year in advance24, totaling 18 months for commercial G. elata production, resulting in a long production cycle. Furthermore, traditional cultivation methods depend on the logging of mature trees, consuming approximately 15 tons of fungal wood per 667 m225. While these methods enhance the quality of the crop, they exert irreversible impacts on the ecological environment. Based on preliminary investigations and research, previous studies have optimized cultivation techniques and developed an artificial medium for Armillaria to enhance its symbiotic growth with G. elata26. The adoption of mechanized cultivation significantly increased the propagation rate and shortened the cultivation cycle from 18 months to 8–10 months. Nutrient sources such as wheat bran, sawdust, and other materials were utilized in the artificial medium to support Armillaria growth. Furthermore, the improved process only requires small-diameter wood to provide nutrition for both Armillaria and G. elata27. In contrast to traditional methods, this artificial mushroom grow kits- cultivation uses a limited quantity of 1–2-year-old oak saplings to sustain the growth of Armillaria and promote tuber development in G. elata, thereby contributing to reduced ecological impact.
In this study, HPLC, the phenol–sulfuric acid method, the Kjeldahl method, Headspace Solid-Phase Microextraction coupled with Gas Chromatography-Mass Spectrometry (HS-SPME-GC-MS) and electronic nose (E-nose) technology were employed to compare six phenolic active compounds and key nutritional components in G. elata cultivated using traditional wood and artificial mushroom grow kits methods. Furthermore, the aroma profiles of G. elata under different cultivation methods were characterized. The findings offer substantive evidence regarding the influence of artificial mushroom grow kits cultivation on altering the quality of G. elata.
Materials and methods
Plant materials collection and cultivation methods
In April 2023, G. elata seedlings were collected from four production areas. The G. elata seedlings are for commercial purchase by Hubei Shennong Armillariella Co., Ltd. The seedlings included immature G. elata ‘Wuhong’ cultivars from Yichang, Hubei (YCWH), Suizhou, Hubei (SZWH), Mianyang, Sichuan (MYWH), and immature Gastrodia elata Bl. f. elata from Hanzhong, Shaanxi (HZH). Seedlings from each location were cultivated using both traditional wood-based and artificial mushroom grow kits-based methods under uniform environmental condition in the Dahong Mountain area of Suizhou, Hubei. Harvesting took place in November 2023. Fresh G. elata tubers were thoroughly washed, steamed until no white core remained in the center, and dried in an oven at 55 °C. The dried samples were then ground and sieved through a No. 4 sieve for subsequent analysis. Store G. elata powder in a dark place at room temperature.
Moisture correction: The moisture content of dried turmeric samples should be less than 15%. The moisture content can be determined by the standard drying method at 105 °C, which involves drying the clean weighing bottle in a 105 °C oven until it reaches constant weight, and recording its weight (M1). Precisely weigh approximately 2.0 g of turmeric powder (record the precise weight, denoted as M), and spread it evenly in the already constant-weighted weighing bottle. Open the lid of the weighing bottle, place it in a 105 °C electrically heated forced-air drying oven, and continue drying for 2 h. After taking it out, immediately close the lid, and put it in a desiccator to cool to room temperature. Then, quickly and precisely weigh it. Put the weighing bottle back into the oven to dry for another 30 min, repeat the cooling and weighing steps until the difference between two consecutive weighings is less than 0.3 mg, which indicates constant weight. Record the final weight (M2). The moisture content (%) is calculated according to the following formula: Moisture content (%) = [(M1 + M − M2)/M] × 100% (where M1 is the constant weight of the weighing bottle, M is the sample weight, and M2 is the total weight of the weighing bottle and the sample. All determinations are carried out in three parallel runs.)
Artificial mushroom grow kits (AM) cultivation: The artificial mushroom grow kit for Armillaria mellea, produced by Hubei Shennong Armillaria Biotechnology Co., Ltd., uses a dry-weight blend of sugarcane residue (10–40%), sawdust (50–60%), bran (10–15%), branches (1–5%), and corn husks (3–5%), supplemented with 1% gypsum, 0.5–1% each of KH2PO4 and MgSO4, and 1% sugar. After mixing, water is added to achieve 55–65% moisture, and the mixture is fermented for 1–3 days.The material is then bagged in polyethylene bags (30–60 cm long, 10–12 cm diameter), each containing 500–1000 g of dry material. The bags are sealed and sterilized—either at 121 °C for 1–2 h under high pressure or at normal pressure for 20–24 h. Once cooled, they are inoculated with the secondary strain of Armillaria mellea in a sterile environment and cultivated at 25 °C for 3–4 months to form finished kits. At the time of planting, artificial mushroom grow kits served as the primary nutrient source, supplemented with small-diameter Quercus glauca trees, to cultivate different cultivars of G. elata seedlings.
Traditional Wood (TW) cultivation: One year prior to planting, fixed-bed substrates were prepared using oak wood and inoculated with Armillaria spp. At the time of planting, the beds were reopened, and different cultivars of immature G. elata were planted. The schematic diagrams of the two cultivation methods are shown in Fig. 1. Sample origins and detailed cultivation conditions are summarized in Table 1.
Schematic diagram of different cultivation patterns. The artificial mushroom grow kits (AM) cultivation method (left): Immature G. elata seeds are placed around the artificial mushroom grow kits, and small materials with a diameter of 3–5 cm and a length of 5–20 cm are laid on top to provide nutrients. The traditional wood (TW) cultivation method (right): One year in advance, wood with a diameter of over 20 cm and a length of over 30 cm is used to cultivate a fixed fungal bed. In March of the following year, immature G. elata seeds are placed at intervals of 4–5 cm around the fixed fungal bed.
Sampling and experimental design
Sampling design: The G. elata materials were collected using a quintuple sampling method to ensure the sample was representative of the whole batch.
Replicates: The experiment included three independent biological replicates (n = 3). Each biological replicate originated from a separate and independently processed G. elata sample, encompassing the entire workflow from sampling to analytical measurement.
Environmental conditions: All instrumental analyses, including HPLC, E-nose, HS-SPME-GC-MS, polysaccharide, crude protein and amino acid analysis, were conducted under ambient laboratory temperature conditions (approximately 25 °C). This controlled condition ensured consistency across all analytical platforms.
Determination of gastrodin and related phenolic compounds by HPLC
The content of gastrodin, 4-hydroxybenzylalcohol, parishin E, parishin B, parishin C, and parishin in G. elata samples from two cultivation methods was determined by HPLC according to the Chinese Pharmacopoeia (2020 edition)1. All measurements were performed in triplicate.
Sample preparation: Approximately 0.50 g of G. elata powder was accurately weighed and placed into a sealed conical flask. 25 mL of 50% methanol was added, and the weight of the flask was recorded. The mixture was then sonicated for 30 min at 500 W and 40 kHz. After sonication and cooling, the loss in solvent was replenished with 50% methanol to restore the original total weight. The resulting solution was allowed to settle for 1 h, and the supernatant was filtered through a 0.22 μm membrane filter before HPLC analysis.
HPLC conditions: The analysis was conducted using an LC-2030C 3D Plus HPLC system (Shimadzu Enterprise Management Co., Ltd., China) equipped with a ZORBAX Eclipse XDB-C18 column (4.6 × 250 mm, 5 µm; Agilent Technologies, USA). Mobile Phase: 0.1% (v/v) aqueous phosphoric acid (A) and acetonitrile (B) with a gradient elution profile: 3–10% B (0–10 min), 10–12% B (10–15 min), 12–18% B (15–25 min), 18% B (25–40 min), 18–95% B (40–42 min), 95–3% B (42–55 min), and 3% B (55–60 min). The flow rate was set at 0.8 mL/min, the injection volume was 5 µL, and detection was carried out at 220 nm. The column temperature was maintained at 30 °C.
The mixed standard solution (standards purchased from Chengdu Ruifensi Biotechnology Co., Ltd.; other reagents were of HPLC or analytical grade) was analyzed using the above-described chromatographic method. Ideal chromatographic peaks with no tailing and stable baselines were obtained. Standard curves were generated based on the chromatograms, and the following linear regression equations were derived: Gastrodin: y = 20932x + 85,452 (R2 = 0.9997); 4-Hydroxybenzyl alcohol: y = 42687x + 19,372 (R2 = 0.9999); Balisaside E: y = 5578x + 3258.5 (R2 = 1.0000); Balisaside B: y = 10581x + 16,167 (R2 = 0.9993); Balisaside C: y = 10983x − 18,378 (R2 = 0.9993); Balisaside: y = 36479x + 677,238 (R2 = 0.9992). The correlation coefficients (R2) were all greater than 0.999, indicating good linearity. Representative chromatograms of the mixed standard solution and sample are shown in Fig. 2A.
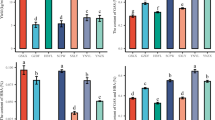
Comparison of phenolic compounds and nutritional quality components in G. elata under different cultivation methods. (A) HPLC fingerprint analysis of phenolic compounds in different G. elata sources under two cultivation methods. (B) Analysis of crude protein content in different G. elata sources under two cultivation methods. (C) Analysis of total polysaccharide content in different G. elata sources under two cultivation methods. (D) Analysis of the proportion of free amino acids contributing to taste in different G. elata sources under two cultivation methods.
Determination of G. elata polysaccharide content
The phenol–sulfuric acid method was used, with modifications based on previously reported procedures7,28. Briefly, 2.00 g of G. elata sample powder was accurately weighed and extracted with 60 mL of 80% ethanol by sonication for 30 min. After filtration, the residue was air-dried. 1.00 g of the dried residue was then transferred to a 100 mL Erlenmeyer flask, and 35 mL of ultrapure water was added. The mixture was sonicated at 45 °C for 55 min, and then the volume was adjusted to 100 mL with ultrapure water. 1 mL of the resulting solution was diluted to 10 mL to obtain the sample solution for analysis.
The reaction mixture consisted of 0.5 mL of the prepared sample solution, 1 mL of 5% phenol solution, and 5 mL of concentrated sulfuric acid. The mixture was heated in a boiling water bath for 15 min, allowed to cool to room temperature, and then analyzed by measuring the absorbance at 490 nm.
Determination of G. elata crude protein content
The crude protein content in G. elata was determined using the Kjeldahl method, according to the Chinese National Food Safety Standard GB 5009.5-2016, “Determination of Protein in Foods”29.
Briefly, 0.10 g of G. elata sample powder, 3.00 g of potassium sulfate, and 0.20 g of anhydrous copper sulfate were accurately weighed and placed into a Kjeldahl digestion tube. 8 mL of concentrated sulfuric acid was added, and the mixture was digested using the following program: 180 °C for 30 min, 280 °C for 30 min, and 420 °C for 60 min. After digestion, the protein content was determined using a K9860 automatic Kjeldahl analyzer (Jinan Hanon Instruments Co., Ltd., China).
Determination of free amino acid content
The determination of free amino acid content in G. elata was performed by Wuhan Metware Biotechnology Co., Ltd. Based on the standard samples, a MWDB (Metware Database) database was constructed, and the data from mass spectrometry analysis were subjected to qualitative analysis. After obtaining the mass spectrometry analysis data of different samples, the chromatographic peaks of all target substances were integrated, and quantitative analysis was performed using the standard curve (Supplementary Table 1).
Sample preparation: 50 mg (± 2.5 mg) of the sample was weighed into a 2 mL centrifuge tube, and 500 μL of pre-cooled (− 20 °C) 70% methanol aqueous extraction solution was immediately added. The mixture was vortexed for 3 min and centrifuged at 12,000 r/min for 10 min at 4 °C. 300 μL of the supernatant was then transferred to a 1.5 mL centrifuge tube and incubated at − 20 °C for 30 min. After a second centrifugation at 12,000 r/min for 10 min at 4 °C, 200 μL of the supernatant was transferred to a sample vial and stored at − 20 °C until analysis.
Chromatographic conditions: Column: Waters ACQUITY UPLC HSS T3 C18 column (1.8 µm, 100 mm × 2.1 mm); Mobile phase: A: Ultrapure water with 0.1% formic acid; B: Acetonitrile with 0.1% formic acid; Flow rate: 0.35 mL/min; Column temperature: 40 °C; Injection volume: 2 μL; Gradient elution (0 min: A/B = 95:5 (v/v); 8 min: A/B = 5:95 (v/v); 9.5 min: A/B = 5:95 (v/v); 9.6 min: A/B = 5:95 (v/v); 12 min: A/B = 95:5 (v/v)).
MS conditions: Ion source: Electrospray Ionization (ESI) temperature: 550 °C; Ion spray voltage: 5500 V (positive mode), − 4500 V (negative mode); Curtain Gas (CUR): 35 psi; Multiple Reaction Monitoring (MRM) was used in the Q-Trap 6500 + to scan each ion pair based on optimized Declustering Potential (DP) and Collision Energy (CE).
E-nose analysis
E-nose analysis was performed following a modified version of the method described by Li et al.30. 2.0 g of G. elata sample powder was accurately weighed and placed in a 50 mL centrifuge tube, the opening of which was sealed with two layers of Parafilm to ensure air-tightness. The tube was then heated in a water bath at 65 °C for 35 min prior to analysis.
A PEN3.5 electronic nose (Airsense Analytics GmbH, Germany) was used to analyze the headspace gas using the headspace sampling method, with stable response values as the primary indicator. The analysis parameters were as follows: injection volume, 300 mL/min; sampling interval, 1 s; cleaning time, 90 s; zero-point adjustment time, 5 s; pre-sampling time, 5 s; and acquisition time, 90 s per sample. Each sample was measured in triplicate. Electronic nose sensor information is provided in Table 2.
Determination of volatile components in G. elata
HS-SPME-GC-MS conditions were modified based on the method described by Duan et al.31. Extraction method: 0.50 g of G. elata sample powder was accurately weighed and placed in a 20 mL headspace vial, which was then capped and sealed. A manual SPME holder equipped with a 100 μm polydimethylsiloxane (PDMS) fiber was inserted into the vial, and the headspace was sampled at 80 °C for 30 min. After adsorption, the manual SPME holder was rapidly withdrawn and inserted into the gas chromatograph injection port (250 °C), where the fiber was desorbed for 5 min before data acquisition. After integrating the peak area of the product, qualitative analysis was conducted using relative percentage content.
GC conditions: Column: DB-5MS capillary column (30 m × 0.25 mm, 0.25 μm film thickness); Oven program: 40 ℃ for 2 min, then ramped to 100 °C at 30 °C/min, held for 2 min, then ramped to 160°C at 20 °C/min, held for 8 min, then ramped to 190 °C at 3 °C/min, and finally ramped to 230 °C at 8 °C/min, held for 10 min; Injection: Splitless; Carrier gas: High-purity nitrogen at a flow rate of 1 mL/min.
MS conditions: Ion source temperature: 250 °C; Electron energy: 70 eV; Injector temperature: 250 °C; Quadrupole mass analyzer; Mass scan range: 40–400 m/z.
Data processing
SPSS 25.0 software was used for data analysis. Principal component analysis (PCA) and orthogonal partial least squares discriminant analysis (OPLS-DA) were performed using Origin Pro 2024 and SIMCA 14.1 software. Differentially expressed volatile components were selected based on a variable importance in projection (VIP) value > 1. Origin Pro 2024 software was used for data visualization.
For E-nose data, radar plots were generated using Origin Pro 2024. WinMuster software was used to analyze the overall response intensity from the sensor response curves. Stable data from three replicate measurements were selected for principal component analysis (PCA).
Result
Comparison of phenolic content in G. elata under two cultivation methods
In TW cultivation, the total content of gastrodin and 4-hydroxybenzylalcohol in YCWH and HZH was > 0.25% (0.35% and 0.26%), respectively. In AM cultivation, only HZH had a total content of gastrodin and 4-hydroxybenzyl alcohol > 0.25%, at 0.30%, meeting the requirements of the Chinese Pharmacopoeia1. T-test analysis (Table 3) showed that the gastrodin content in SZWH under AM cultivation (0.13%) was significantly higher than that under TW cultivation (0.08%). The gastrodin content in MYWH and HZH under AM cultivation (0.11% and 0.27%, respectively) was also higher than that under TW cultivation (0.10% and 0.21%, respectively). However, the gastrodin content in YCWH was significantly higher under TW cultivation (0.19%) than AM cultivation (0.17%). The study also found that the content of 4-hydroxybenzylalcohol was generally higher under TW cultivation than under AM cultivation. The 4-hydroxybenzylalcohol content in YCWH, SZWH, and HZH under TW cultivation (0.16%, 0.17%, and 0.05%, respectively) was significantly higher than that under AM cultivation (0.03%, 0.04%, and 0.03%, respectively). Furthermore, analysis of parishins revealed that parishin E had the highest proportion in both cultivation modes. The content of parishin E under AM cultivation (0.90–1.58%) was significantly higher than that under TW cultivation (0.68–0.96%).
Comparison of nutritional quality components of G. elata under two cultivation methods
Comparison of protein and polysaccharide content
The protein content of G. elata under TW cultivation ranged from 6.14 to 9.12%, while under AM cultivation, it ranged from 8.86 to 10.57%. The protein content of G. elata was generally higher under AM cultivation than under TW cultivation (Fig. 2B). The polysaccharide content of G. elata under TW cultivation varied widely, ranging from 15.17 to 39.62%. In contrast, the polysaccharide content of G. elata under AM cultivation was more stable, ranging from 38.91 to 39.90%. Specifically, the polysaccharide content in YCWH and MYWH under AM cultivation (36.14% and 38.91%, respectively) was significantly higher than that under TW cultivation (15.17% and 34.59%, respectively). However, the polysaccharide content in HZH under AM cultivation (39.62%) was significantly higher than that under AM cultivation (29.91%) (Fig. 2C).
Comparison of free amino acid content
Eighteen free amino acids were detected in G. elata from different sources and under both cultivation methods (Table 4, Fig. 2D). Under AM cultivation, YCWH showed significantly higher levels of twelve free amino acids compared to TW cultivation, including umami amino acids (glutamic acid: 6.78 mg/g, asparagine: 1.28 mg/g, proline: 0.21 mg/g), sweet amino acids (glutamine: 1.53 mg/g, serine: 0.40 mg/g, alanine: 0.45 mg/g, glycine: 0.88 mg/g), acidic amino acid (aspartic acid: 1.28 mg/g), and aromatic amino acids (arginine: 0.52 mg/g, valine: 0.15 mg/g, leucine: 0.07 mg/g, phenylalanine: 0.05 mg/g, lysine: 0.13 mg/g). In SZWH, the content of umami amino acids (asparagine: 1.24 mg/g, proline: 0.25 mg/g) and aromatic amino acid (arginine: 0.62 mg/g) was significantly higher under AM cultivation, while the content of sweet amino acids (glutamine: 2.08 mg/g, threonine: 0.20 mg/g, serine: 0.35 mg/g, alanine: 0.44 mg/g), bitter amino acids (isoleucine: 0.05 mg/g, histidine: 0.18 mg/g), and aromatic amino acid (lysine: 0.15 mg/g) was significantly higher under TW cultivation. Similarly, the content of umami amino acids (asparagine: 1.88 mg/g, proline: 0.26 mg/g), sweet amino acids (threonine: 0.25 mg/g, serine: 0.41 mg/g, alanine: 0.53 mg/g, glycine: 0.96 mg/g), acidic amino acid (aspartic acid: 1.88 mg/g), and aromatic amino acids (arginine: 0.69 mg/g, valine: 0.14 mg/g, leucine: 0.08 mg/g, phenylalanine: 0.05 mg/g, lysine: 0.14 mg/g) was significantly elevated in MYWH under AM cultivation compared to TW cultivation, whereas the content of sweet amino acid (glutamine: 2.27 mg/g) and bitter amino acid (histidine: 0.17 mg/g) was significantly higher under TW cultivation. Furthermore, AM cultivation of HZH resulted in significantly increased levels of umami amino acids (glutamic acid: 7.37 mg/g, asparagine: 1.61 mg/g, proline: 0.23 mg/g), sweet amino acids (glutamine: 2.46 mg/g, threonine: 0.33 mg/g, serine: 0.52 mg/g, alanine: 0.80 mg/g, glycine: 1.49 mg/g), acidic amino acid (aspartic acid: 1.61 mg/g), and aromatic amino acids (arginine: 0.79 mg/g, valine: 0.21 mg/g, leucine: 0.09 mg/g, phenylalanine: 0.06 mg/g, lysine: 0.16 mg/g) compared to TW cultivation.
Compared to TW cultivation, AM cultivation significantly increased the content of two umami amino acids, glutamic acid and asparagine, in G. elata from all four sources. Additionally, AM cultivation significantly elevated the content of four sweet amino acids (glutamine, serine, alanine, and glycine), four aromatic and bitter amino acids (arginine, valine, leucine, and phenylalanine), and one acidic amino acid (aspartic acid) in G. elata from YCWH, MYWH, and HZH.
Comparative analysis of G. elata aroma under two cultivation methods using E-nose
PCA analysis revealed that the E-nose could effectively differentiate the aroma profiles of G. elata under the two cultivation methods (Fig. 3A–D). For YCWH, the first principal component (PC1) accounted for 98.877% of the variance, and the second principal component (PC2) accounted for 0.85484%, with a cumulative contribution of 99.732%. For SZWH, PC1 accounted for 96.025% of the variance, and PC2 accounted for 3.9575%, with a cumulative contribution of 99.983%. For MYWH, PC1 accounted for 99.475% of the variance, and PC2 accounted for 0.51637%, with a cumulative contribution of 99.992%. For HZH, PC1 accounted for 99.584% of the variance, and PC2 accounted for 0.38867%, with a cumulative contribution of 99.973%. These results demonstrate the ability of E-nose technology to distinguish G. elata based on cultivation method.
Comparative analysis of volatile components of G. elata by electronic nose detection under traditional wood planting and artificial mushroom grow kits planting.
The PEN 3.5 electronic nose used in this study is equipped with 10 heated sensors capable of precisely detecting the presence and concentration of volatile compounds, exhibiting different selectivity and sensitivity to these compounds. As shown in the radar plots of sensor response values (G/G0) (Fig. 3E and F), the profiles were similar across the two cultivation methods. All 10 sensors of the E-nose responded to the aroma of G. elata, with the response values of W1W, W2W, and W5S being significantly higher than the other 7 sensors (Supplementary Table 2). This suggests that the major volatile components common to both cultivation methods are terpenes, aromatics, and nitrogen oxide compounds. Furthermore, the electronic nose was more sensitive to the aroma of substrate-cultivated G. elata, indicating that AM cultivation results in a richer aroma profile compared to TW cultivation.
Analysis of volatile components in G. elata under two cultivation methods
The volatile components of four G. elata samples under two different cultivation methods were analyzed by HS-SPME-GC-MS, and a total of 53 components were identified based on average relative content. These included 4 alcohols, 11 aromatics, 3 phenols, 1 nitrile, 1 aldehyde, 1 acid, 1 ketone, 4 alkanes, 1 alkene, and 1 ester (Supplementary Table 3). The percentage content of alcohols (1.25–5.82%), aromatics (6.5–10.93%), and acids (0.91–25.07%) was generally higher under substrate cultivation than under traditional cultivation (0.13–2.13%, 0.31–3.7%, and 1.22–21.02%, respectively). Conversely, the aldehyde content was significantly higher under traditional cultivation (10.18–19.37%) than under substrate cultivation (1.18–11.33%) (Fig. 4A–D, Supplementary Table 4). This is generally consistent with the electronic nose results, indicating that substrate cultivation results in a richer volatile profile and a higher total relative content of volatile compounds.
Comparison of total relative content of volatile compound classes in YCWH (A), MYWH (B), SZWH (C), and HZH (D).
To further investigate the differential volatile compounds between the two cultivation methods, OPLS-DA analysis was performed. Using a VIP value > 1 as the selection criterion, 13 differential compounds were identified, mainly including 2 alcohols, 2 aromatics, 3 aldehydes, 2 acids, 3 ketones, and 1 ester. The model exhibited good fit and predictive ability, with R2X = 0.947, R2Y = 1, and Q2 = 0.853. A permutation test with 200 iterations yielded a Q2 regression line intercept > 0.5, indicating no overfitting and validating the model for distinguishing G. elata under different cultivation methods (Fig. 5A and B). Cluster analysis of the 13 differential compounds revealed that YCWH under substrate cultivation clustered with the four G. elata sources under traditional cultivation, while SZWH, MYWH, and HZH under substrate cultivation clustered together (Fig. 6A).
Scatter plot from OPLS-DA analysis (A) and permutation test plot (B).
Analysis of volatile components in G. elata under traditional wood cultivation and artificial mushroom grow kits cultivation: (A) Hierarchical clustering heat map of relative percentage content of differential compounds. (B) Key differential volatile compounds and their aroma profiles in G. elata under different cultivation methods.
To characterize the aroma of G. elata under different cultivation methods, further analysis of key volatile compounds was conducted. The results indicated that G. elata under substrate cultivation was enriched in (2S,3S)-2,3-butanediol (1.25–2.32%), tetramethylpyrazine (2.97–8.95%), 1-(3,4-dihydro-2H-pyrrol-5-yl)ethanone (0.00–1.76%), and 3-hydroxy-2-methyl-4H-pyran-4-one (1.70–11.25%), with higher percentage content than G. elata under traditional cultivation (0.00–0.77%, 0.00–2.23%, 0.00%, and 1.39–7.91%, respectively). These compounds contribute to a sweet32, meaty33, roasted grain-like34, and fruity35 aroma in substrate-cultivated G. elata. Conversely, G. elata under traditional cultivation was enriched in decanal (1.13–1.77%), (2E,4E)-deca-2,4-dienal (2.11–4.25%), 5-(hydroxymethyl) furfural (3.82–9.64%), and pentadecan-2-one (4.05–5.38%), with higher percentage content than G. elata under substrate cultivation (0.00–0.74%, 0.00–1.20%, 0.00–6.57%, and 0.44–2.82%, respectively). These compounds contribute to a citrusy36, chicken-like37, chamomile-like38, and celery-like aroma in traditionally-cultivated G. elata (Fig. 6B).
Discussion
Ecological cultivation of Traditional Chinese Medicine emphasizes minimizing the use of chemical fertilizers and pesticides while promoting the use of organic fertilizers and biopesticides39,40. To ensure the quality and safety of TCM materials41. Specific practices include ecological site selection, targeted cultivation, crop rotation and intercropping, and scientific management42–45. For example, cultivating G. elata using artificial mushroom grow kits cultivation can significantly reduce timber consumption and shorten the cultivation cycle25.
This study aimed to investigate the impact of traditional wood -based cultivation (TW) and artificial mushroom grow kits cultivation (AM) on the quality of different G. elata sources. The results indicated that AM, while maintaining the level of the main active compound, gastrodin, significantly affected the accumulation of minor active compounds, nutritional components, and aroma compounds. This provides new insights into the artificial cultivation and quality improvement of G. elata.
Although the impact on gastrodin was limited, the content of 4-hydroxybenzyl alcohol and parishin E differed significantly between the two cultivation methods. Both cultivation methods rely on the Armillaria to provide nutrients for the G. elata. As long as the Armillaria grows well and can effectively infect the G. elata, it lays a similar foundation for the synthesis of gastrodin. Previous studies have shown that there are differences in yield and quality among different varieties of G. elata17. The experiment standardized the genetic background of the immature G. elata, which might be one of the reasons for the relatively small differences in gastrodin content. Given that 4-hydroxybenzyl alcohol and gastrodin influence the central nervous system via similar pathways and may undergo interconversion46, and considering that parishin compounds may be metabolically converted to gastrodin47, The AM cultivation conditions may have accelerated the conversion and consumption of 4-hydroxybenzyl alcohol, the reduction in 4-hydroxybenzyl alcohol under AM may be partially compensated by the elevated presence of parishin E. The observed reduction in 4-hydroxybenzyl alcohol under AM cultivation suggests a directed shift in metabolic flux, potentially towards the synthesis of downstream metabolites such as its dimer, which may possess enhanced defensive efficacy. This dynamic response underscores the role of 4-hydroxybenzyl alcohol as a sensitive metabolic node, indicative of the plant’s altered physiological state rather than merely a change in a fixed chemical constituent. Previous studies have shown that genetic variation among G. elata populations exceeded that within populations48, this is consistent with the differences in phenolic substances found in different sources of G. elata in this study. However, under the AM cultivation, the G. elata may have inhibited the accumulation of certain intermediates, but at the same time promoted the synthesis of downstream products that are more valuable to the plant itself. This demonstrates the adaptability of the ginseng to the specific growth environment and its metabolic reorganization.
AM cultivation significantly increased the crude protein content in G. elata, likely due to the richer nitrogen source provided by artificial mushroom grow kits38, which facilitates enhanced protein synthesis. As an essential nutrient for the human body, protein plays a crucial role in tissues and organs development as well as in maintaining physiological functions7. Furthermore, the total polysaccharide content was also more stable under AM. Polysaccharides are important active components of both medicinal and edible TCMs, possessing a wide range of pharmacological effects and biological activities49. The significant increase in the content of umami and sweet amino acids further suggests that AM can improve the nutritional quality and taste of G. elata. The analysis of amino acid content and the evaluation of nutritional value can provide an important basis for the selection of fungal materials in G. elata cultivation50.
Aroma component analysis revealed significant differences in the volatile composition of G. elata under TW and AM. The enrichment of esters and aromatic compounds in AM G. elata, along with the increased levels of characteristic aroma components such as (2S,3S)-2,3-butanediol, tetramethylpyrazine, 1-(3,4-dihydro-2H-pyrrol-5-yl)ethanone, and 3-hydroxy-2-methyl-4H-pyran-4-one, conferred a richer sweet, meaty, roasted grain-like, and fruity aroma. In contrast, the enrichment of characteristic aroma components such as decanal, (2E,4E)-deca-2,4-dienal, 5-(hydroxymethyl)furfural, and pentadecan-2-one in TW G. elata resulted in a unique citrusy, chicken-like, chamomile-like, and celery-like aroma. These differences not only affect the sensory qualities of G. elata but may also influence its pharmacological activity. It is worth noting that the significant increase in aromatic compounds in the volatile components of substrate-cultivated G. elata synergistically improved its aroma and taste, in conjunction with the increased levels of umami and sweet amino acids.
In conclusion, the AM method offers advantages in enhancing certain nutritional components and improving the aroma characteristics of G. elata, while also providing more stable total polysaccharide content. These advantages give G. elata grown under AM broader application prospects in fields such as clinical medicine, health product development, and food processing. However, this study did not monitor the soil microbial communities under substrate cultivation, and the potential impact of factors such as soil environment and temperature/humidity was not fully considered. Therefore, future research should focus on optimizing the technical details of the substrate cultivation method, such as finely adjusting the matrix ratio, improving soil structure, and precisely controlling temperature and humidity conditions, while also monitoring changes in soil microorganisms. By comprehensively optimizing the cultivation environment, it is expected to further enhance the overall quality of artificial mushroom grow kits-cultivated G. elata, providing a more sufficient scientific basis for the comprehensive development and utilization of G. elata resources.
Conclusion
This study confirms that artificial mushroom grow kits cultivation demonstrates significant advantages in G. elata cultivation compared to traditional wood cultivation. This method enhances the content of the active compounds gastrodin and parishin E, stabilizes polysaccharide content, increases crude protein and umami/sweet amino acid content, and improves aroma characteristics, making it more promising in terms of nutritional value and sensory qualities. However, to comprehensively improve the quality of artificially substrate-cultivated G. elata, future research should focus on optimizing substrate formulations and growth environment control, thereby providing a more solid scientific basis for the application of G. elata in the pharmaceutical, health, and food sectors.
Data availability
Data will be made available on request.
References
Pharmacopoeia of the People’s Republic of China. (China Medical Science & Technology, 2020).
Guo, H., Cheng, D., Zhang, C. & Xue, F. The pharmacological effects of gastrodin and the progress in the treatment of neurological disorders. Am. J. Chin. Med. 53, 803–831. https://doi.org/10.1142/s0192415x25500302 (2025).
Guan, D. et al. Exploring the potential protective mechanism of Gastrodia elata Blume in Parkinson’s disease using LC-MS/MS-based striatal metabolomics. J Ethnopharmacol 352, 120094. https://doi.org/10.1016/j.jep.2025.120094 (2025).
Zhao, F. et al. Extraction, purification, characteristics, bioactivities, application, and toxicity of Gastrodia R. Br. polysaccharides: A review. Int. J. Biol. Macromol. 301, 140084. https://doi.org/10.1016/j.ijbiomac.2025.140084 (2025).
Liu, Y. et al. Research progress in the field processing and quality evaluation of Gastrodiae Rhizoma. World Chin. Med. 19, 727–735 (2024).
Yang, L., Qin, S. H. & Zi, C. T. Research progress of Gastrodia elata Blume polysaccharides: A review of chemical structures and biological activities. Front. Chem. 12, 1395222. https://doi.org/10.3389/fchem.2024.1395222 (2024).
Li, X. Y. et al. Main nutrients content in different specifications of Gastrodiae Rhizoma. China J. Chin. Mater. Med. 49, 4572–4577. https://doi.org/10.19540/j.cnki.cjcmm.20240216.104 (2024).
Duan, H., Zhou, S., Guo, J. & Yan, W. HS-GC-IMS Analysis of Volatile Organic Compounds in Different Varieties and Harvesting Times of Rhizoma gastrodiae (Tian Ma) in Yunnan Province. Molecules 28, https://doi.org/10.3390/molecules28186705 (2023).
Zhang, B. B. et al. Evaluation of quality characteristics of 15 Gastrodia elata germplasm resources based on HS-SPME-GC-MS, HPLC and electronic nose techniques. Food Biosci. 71, https://doi.org/10.1016/j.fbio.2025.107158 (2025).
Sun, J. et al. Metabonomics analysis reveals the influence mechanism of three potassium levels on the growth metabolism and accumulation of medicinal components of Bupleurum scorzonerifolium Willd. (Apiaceae). Biology (Basel) 14, 452. https://doi.org/10.3390/biology14050452 (2025).
Zhang, W. et al. Evaluation of different ecological regions for cultivation of best quality Bupleurum: A case study from Shanxi China. BMC Plant Biol. 25, 542. https://doi.org/10.1186/s12870-025-06479-w (2025).
Zeng, Z. et al. Different cultivation environments affect the yield, bacterial community and metabolites of Cordyceps cicadae. Front. Microbiol. 12, 669785. https://doi.org/10.3389/fmicb.2021.669785 (2021).
Zhu, M., Chen, H., Si, J. & Wu, L. Effect of cultivation mode on bacterial and fungal communities of Dendrobium catenatum. BMC Microbiol. 22, 221. https://doi.org/10.1186/s12866-022-02635-6 (2022).
Liu, C. L., Hu, Y. D. & Xu, L. Comparative study on triterpenoids of Poria cocos with different cultivation modes. Res. Pract. Chin. Med. 35, 11–14. https://doi.org/10.13728/j.1673-6427.2021.05.003 (2021).
Zhang, Y. et al. Structural characterization and anti-tumor immunomodulatory effects of polysaccharides from Astragalus mongholicus with different cultivation modes. Int. J. Biol. Macromol. 318, 145233. https://doi.org/10.1016/j.ijbiomac.2025.145233 (2025).
Zhang, Y., Hou, H., Li, Y., Li, K. & Lin, H. Differential analysis of the quality and soil microhabitat of Epimedium koreanum Nakai under different cultivation methods. Front. Microbiol. 16, 1556173. https://doi.org/10.3389/fmicb.2025.1556173 (2025).
Jiang, X. et al. Comprehensive quantitative evaluation and mechanism analysis of influencing factors on yield and quality of cultivated Gastrodia elata Blume. Sci. Rep. 15, 18457. https://doi.org/10.1038/s41598-025-99706-1 (2025).
Li, J. Effect of Gastrodia elata Bl cultivation under forest stands on runoff erosion and nutrient loss. Forests 15, 1127. https://doi.org/10.3390/f15071127 (2024).
Zhang, J. Q. et al. Analysis of ecological recycling modes for Gastrodia elata cultivation. China J. Chin. Mater. Med. 45, 2036–2041. https://doi.org/10.19540/j.cnki.cjcmm.20200302.109 (2020).
Gong, Y. et al. Effects of different cultivation patterns on the active ingredients of Gastrodia elata Bl. f. glauca S. Chow. Natural Product Research and Development, 1–15.
Yuan, Z. T. et al. Analysis of ecological bag planting technology under the Gastrodia elata forest in Xuefeng Mountain. Rural Sci. Technol. 16, 94–97. https://doi.org/10.19345/j.cnki.1674-7909.2025.08.020 (2025).
Huang, X. M., Chen, S. F. & Qi, C. Planting technology in sexual reproduction of Gastrodia elata BI. J. Zhaotong Univ. 33, 22–24 (2011).
Xie, X. Q. Simple culture techniques for Germinationpromoting fungi and Armillariella mellea in artificial planting of Gastrodia elata. Agric. Sci. Technol. 17, 2005 (2016).
Sun, Z., Tao, Y. & Su, H. Interplanting method for Polyporus umbellatus and Gastrodia elata. (2011).
Jiang, W. K. et al. Thoughts and suggestions on ecological cultivation of Gastrodia elata. China J. Chin. Materia Med. 47, 2277–2280. https://doi.org/10.19540/j.cnki.cjcmm.20220218.101 (2022).
Liu, D. H., Yan, H. Y., Xue, C., Zhang, B. H. & Du, H. Z. A factory production system for artificial fungus sticks of Gastrodia elata symbiotic Armillaria mellea. China patent (2022).
Wang, L. et al. Effects of Armillaria artificial stick substrate formula C:N on the yield and nutritional quality of Gastrodia elata. J. Food Saf. Qual. 15, 50–57. https://doi.org/10.19812/j.cnki.jfsq11-5956/ts.20240102001 (2024).
Cheng, L. J., Peng, J. H., Hu, Z. F., Gao, Y. & Cha, Y. Determination of polysaccharide content of Gastrodia Elata in Zhaotong and optimization of ultrasound-assisted extraction technology. Anhui Agric. Sci. Bull. 25, 28–29. https://doi.org/10.16377/j.cnki.issn1007-7731.2019.z1.011 (2019).
Vol. GB5009.5-2016 (China Standard Publishing House, Beijing, 2016).
Li, H. et al. Identification of Bletilla striata and related decoction pieces: a data fusion method combining electronic nose, electronic tongue, electronic eye, and high-performance liquid chromatography data. Front. Chem. 11, 1342311. https://doi.org/10.3389/fchem.2023.1342311 (2023).
Duan, H., Zhou, Y., Wang, D. & Yan, W. Differences in volatile organic compounds in Rhizoma gastrodiae (Tian Ma) of different origins determined by HS-GC-IMS. Molecules 28, 4883. https://doi.org/10.3390/molecules28134883 (2023).
Zhang, L. et al. Cloning, expression and characterization of glycerol dehydrogenase involved in 2,3-butanediol formation in Serratia marcescens H30. J. Ind. Microbiol. Biotechnol. 41, 1319–1327. https://doi.org/10.1007/s10295-014-1472-x (2014).
Li, Y. et al. Dynamics of physicochemical properties, volatile aroma compounds and non-volatile metabolites of Zhaiji millet vinegar during aging: A systematic elucidation by GC-IMS coupled with UPLC-MS/MS untargeted metabolomics. Food Chem 493, 146009. https://doi.org/10.1016/j.foodchem.2025.146009 (2025).
Majcher, M. A., Olszak-Ossowska, D., Szudera-Kończal, K. & Jeleń, H. H. Formation of key aroma compounds during preparation of pumpernickel bread. J. Agric. Food Chem. 68, 10352–10360. https://doi.org/10.1021/acs.jafc.9b06220 (2020).
Wang, J., Zhao, M., Xie, N., Huang, M. & Feng, Y. Community structure of yeast in fermented soy sauce and screening of functional yeast with potential to enhance the soy sauce flavor. Int. J. Food Microbiol. 370, 109652. https://doi.org/10.1016/j.ijfoodmicro.2022.109652 (2022).
de Siqueira, A. C. P. et al. Volatile profiles of Murcott and Ponkan mandarins obtained by stir bar sorptive extraction technique and their contributions to the fruit aroma. J. Food Sci. 89, 4823–4838. https://doi.org/10.1111/1750-3841.17232 (2024).
Warner, S. & Munafo, J. P. Jr. Characterization of key odorants in chardonnay seeds. J. Agric. Food Chem. 70, 16316–16322. https://doi.org/10.1021/acs.jafc.2c06119 (2022).
Wang, S., Bu, Q. & Feng, Y. Non-targeted metabolomics reveals substrates and products of maillard reaction related to changes of color and aroma of dried sweet potato during storage. Food Chem. X 31, 103031. https://doi.org/10.1016/j.fochx.2025.103031 (2025).
Cao, X. et al. Wild wisdom meets cultivation: comparative rhizomicrobiome analysis unveils the key role of Paraburkholderia in growth promotion and disease suppression in Coptis chinensis. Microbiome 13, 150. https://doi.org/10.1186/s40168-025-02136-4 (2025).
Li, J. et al. Artemisia argyi essential oil exerts herbicidal activity by inhibiting photosynthesis and causing oxidative damage. Ind. Crops Prod. https://doi.org/10.1016/j.indcrop.2023.116258 (2023).
Yang, D. Y. et al. Selection and reflection on ecological fine manufacturing model of traditional Chinese medicine under “dual carbon” goals. China J. Chin. Mater. Med. 49, 6549–6557. https://doi.org/10.19540/j.cnki.cjcmm.20240923.303 (2024).
Zhou, T. et al. Potential risk, spatial distribution, and soil identification of potentially toxic elements in Lycium barbarum L. (Wolfberry) fruits and soil system in Ningxia, China. Int. J. Environ. Res. Public Health 19, 16186. https://doi.org/10.3390/ijerph192316186 (2022).
Wang, W., Xu, J., Fang, H., Li, Z. & Li, M. Advances and challenges in medicinal plant breeding. Plant Sci. 298, 110573. https://doi.org/10.1016/j.plantsci.2020.110573 (2020).
Zhang, L. et al. Maize-soybean rotation and intercropping increase maize yield by influencing the structure and function of rhizosphere soil fungal communities. Microorganisms 12, 1620. https://doi.org/10.3390/microorganisms12081620 (2024).
Yan, H. Y., Liu, F., Jiang, J. Y., Guo, L. P. & Liu, D. H. Analysis and evaluation of paddy-upland rotation of Gastrodia elata-Oryza sativa in Dabie Mountains. Zhongguo Zhong Yao Za Zhi 47, 3686–3692. https://doi.org/10.19540/j.cnki.cjcmm.20220315.103 (2022).
Zhang, Z. L. et al. Research progress on mechanism of gastrodin and p-hydroxybenzyl alcohol on central nervous system. China J. Chin. Mater. Med. 45, 312–320. https://doi.org/10.19540/j.cnki.cjcmm.20190730.401 (2020).
Zhang, J., Song, N. L. & Ma, K. J. Research progress on the pharmacological effects and in vivo processes of Prashins components in Gastrodia elata. Chin. Tradit. Pat. Med. 44, 2223–2229 (2022).
Zhang, Z. et al. Ecological factors impacting genetic characteristics and metabolite accumulations of Gastrodia elata. Chin. Herb. Med. 17, 562–574. https://doi.org/10.1016/j.chmed.2024.09.002 (2025).
Zhao, J. H., Zhu, Z. W., Wang, L. L., Zhu, P. S. & Miao, M. L. Research advances on anti-inflammatory and anti-oxidation effect of medicinal and edible herbs liver-protecting Chinese medicine. Chin. J. Exp. Tradit. Med. Formulae 31, 293–301. https://doi.org/10.13422/j.cnki.syfjx.20241312 (2025).
Luo, X. L., Wu, X. F., Liu, Q., Tao, S. Z. & Wang, B. Y. Amino acid composition and nutritional value evaluation of Gastrodia elata cultivated with different woods materials. Chin. Wild Plant Resour. 43, 28–34 (2024).
Funding
This work was supported by grants from the Seed Industry High-quality Development Project of Hubei Province (Grant HBZY2023B00503), Project of Hubei Province Traditional Chinese Medicine Innovation Team (ZY2025J002), the National Key Research and Development Plan (2023YFC3503804), Hubei Provincial Science and Technology Program Project (2025BCA007), Joint supported by Hubei Provincial Natural Science Foundation and Traditional Chinese Medicine Innovation and Development of China (2024AFD234) and Doctoral Research Funding of Hubei University of Chinese Medicine (2023ZXB010).
Author information
Authors and Affiliations
Contributions
Conceptualization and methodology, Zhang, B. B. and Zhu, H. J.; sample preparation, Zhang, B. B., Zhu, H. J. and Hua, L.; formal analysis, Zhang, B. B. and Zhu, H. J.; data curation, Zhang, B. B.; writing original draft and visualization, Zhang, B. B.; writing review and editing, Liu, D. H., Luo, M., Li, X. H., Zhu, H. J., and Hua, L.; funding acquisition, Liu, D. H. and Li, X. H.. All authors have read and agreed to the published version of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Zhang, B., Zhu, H., Hua, L. et al. Quality comparison of Gastrodia elata grown using artificial mushroom cultivation kits and traditional wood-based cultivation. Sci Rep 16, 2264 (2026). https://doi.org/10.1038/s41598-025-32164-x
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-32164-x