Abstract
The combination of immune checkpoint inhibitors (ICIs) with anti-angiogenic agents has demonstrated efficacy in the clinical treatment of advanced hepatocellular carcinoma (HCC). This study seeks to elucidate the underlying mechanisms that contribute to the enhanced therapeutic effects of apatinib when administered in conjunction with ICIs for the treatment of advanced HCC. The effects of apatinib on the viability, clonal formation, and apoptosis of HCC cells were evaluated through in vitro experiments. Meanwhile, in vivo experiments were conducted to substantiate these findings and further investigate the synergistic effects of apatinib with PD-1 inhibitors on the immune microenvironment, particularly by activating the signal transducer and activator of transcription 1 (STAT1)/natural killer (NK) cell axis. In vitro experiments demonstrated that apatinib significantly suppressed HCC cell viability, colony formation capacity, and induced apoptosis. In tumor-bearing mouse models, the combination of apatinib with PD-1 inhibitors showed superior tumor growth inhibition compared to monotherapy (combination group exhibited the smallest tumor volume and 100% survival rate vs. 0% in PBS group, p < 0.001). Western blot and immunohistochemical analyses revealed STAT1/NK axis activation through combination therapy (upregulated STAT1 expression with increased CD8+T cell and NK cell infiltration, p < 0.001). In the mechanism discussion, STAT1-overexpressing Hepa1-6 cells confirmed the antitumor effect of STAT1 in the combination therapy. Subsequently, we validated our findings using the STAT1 inhibitor fludarabine or the NK cell-depleting agent Asialo GM1. Furthermore, combination therapy remodeled the tumor microenvironment by reducing CA IX (hypoxia marker), CD31 (angiogenesis marker), and α-SMA (stromal activation marker) expression (p < 0.05). Apatinib enhances the efficacy and responsiveness of PD-1 inhibition via the STAT1/NK axis, while the combination therapy remodels the tumor microenvironment to potentiate anti-tumor effects.
Similar content being viewed by others
Introduction
Hepatocellular carcinoma, the predominant form of liver cancer1, is highly malignant and is the third leading cause of cancer-related mortality worldwide2. While surgical resection remains the cornerstone of curative treatment, over 70% of patients present with unresectable disease at diagnosis due to vascular invasion or extrahepatic metastasis. Immunotherapy has achieved significant success in the treatment of HCC, particularly with immune checkpoint inhibitors (ICIs) such as PD-1 and PD-L1, significantly improving patient survival rates3. However, primary and acquired resistance to ICIs remains a critical challenge, as evidenced by the majority of patients deriving limited clinical benefit4. This therapeutic heterogeneity is closely linked to the immunosuppressive tumor microenvironment (TME), characterized by dysfunctional cytotoxic T cells, abundant regulatory T cells (Tregs), and impaired antigen presentation5. Recent efforts to overcome resistance have focused on remodeling the TME through combination strategies, particularly with anti-angiogenic agents that may reverse vascular exclusion of immune cells6,7.
A key regulator of TME immunogenicity is the signal transducer and activator of transcription 1 (STAT1) pathway. STAT1 activation promotes antitumor immunity by enhancing interferon-γ (IFN-γ) signaling – a mechanism conserved across multiple malignancies8,9. In melanoma, STAT signaling pathways correlates with increased CD8⁺ T cell infiltration and prolonged progression-free survival (PFS) under anti-PD-1 therapy10,11. Similarly, breast cancer patients with STAT1-high tumors exhibit higher objective response rates to ICIs compared to STAT1-low counterparts12. Mechanistically, STAT1 orchestrates the transcription of MHC class I/II molecules and chemokines, facilitating dendritic cell maturation and NK cell recruitment12,13. Therefore, promoting the activation of STAT1 may effectively enhance the immune response and therapeutic efficacy of ICIs14. Despite these advances, the role of STAT1 in HCC immunobiology remains underexplored. Methods to activate the STAT1 pathway and overcome PD-1 resistance, as well as their effectiveness, still require further investigation.
Apatinib, a selective VEGFR2 inhibitor approved for advanced HCC, demonstrates unique immunomodulatory properties beyond angiogenesis inhibition. Research has reported that apatinib can alter interferon-γ (IFN-γ) levels15,16. IFN-γ is a key activator of STAT, and activated STAT1 has been shown to enhance the antitumor function of natural killer (NK) cells against HCC17. In patients with extensive-stage small cell lung cancer treated with apatinib combine PD-1 inhibitors, STAT1 activation demonstrated a significant correlation with elevated NK cell levels and prolonged PFS. Targeted sequencing further identified the STAT1 signaling pathway as a critical biomarker for therapeutic efficacy of this combined regimen18. These findings align with clinical observations from the CARES-310 trial, where apatinib plus camrelizumab achieved unprecedented median overall survival (22.1 months vs. 15.2 months with sorafenib, HR = 0.62)19.However, clinical combination therapies are largely based on empirical evidence, with limited studies investigating the synergistic mechanisms of immunotherapy from preclinical data20. Based on these research findings, we propose that apatinib-mediated activation of the STAT1/NK pathway represents a potential mechanism to overcome PD-1 inhibitor resistance.
Given the resistance to PD-1 inhibitors and the potential failure of combination therapies, it is imperative to investigate the underlying mechanisms of these issues. Previous studies have shown the STAT1-overexpressing (STAT1-OE) cell line could confirm the role of STAT1 in antitumor effects. The NK cell-depleting agent and STAT1 inhibitor can further validate the roles of NK cells and STAT1. The focus of this study is to explore the potential mechanisms by which apatinib enhances the efficacy of PD-1 inhibitors and to validate these findings.
Materials and methods
All methods were performed in accordance with the relevant guidelines and regulations.
The study was approved by the Ethics Committee of the Mengchao Hepatobiliary Hospital of Fujian Medical University (IRB: MCHH-AEC-2024-009). All experimental procedures strictly followed the ARRIVE guidelines and applicable legal requirements.
Cell culture
HCC cell lines (Hepa1-6 and Hep 53.4) were obtained from the Chinese Academy of Sciences. The above cells were cultured in DMEM (containing 10% fetal bovine serum and 100 U/mL penicillin and streptomycin) in 5% CO2 at 37 ℃. Exponential growth phase cells were used in the experiment. A full-length mouse STAT1 cDNA sequence was cloned into the lentiviral vector pCDH-CMV-MCS-EF1-Puro. Lentiviruses were produced in HEK293T cells by co-transfecting the transfer plasmid with packaging plasmids psPAX2 and pMD2.G. Hepa1-6 cells were then transduced with the resulting lentivirus. Stable polyclonal populations were selected and maintained in complete medium containing 2 µg/mL puromycin. STAT1 overexpression was confirmed by Western blot analysis.
Drug source
Apatinib and were originally produced by Hengrui Pharmaceutical Co. Ltd (Jiangsu, China). Apatinib stock solution was prepared using dimethyl sulfoxide (DMSO) as a reagent for in vitro experiments. In vivo studies, apatinib (0.25 g per tabel) and anti-PD-1 antibodies (200 mg per bottle) were diluted with 5% glucose solution.
Cell viability assay
First, a dose-response experiment was performed to determine the half-maximal inhibitory concentration (IC₅₀) of apatinib, which was confirmed to be 500 nM for tumor cell cytotoxicity. Cells were seeded in 6-well plates at a density of approximately 8 × 10⁴ cells per well. After the cells adhered, the experimental groups were treated with 500 nM apatinib. The cells were then cultured at 37 °C in a 5% CO₂ atmosphere for 24 h, followed by cell counting and imaging.
Colony formation assay
Hepa1-6 and Hep 53.4 cells were treated with apatinib (500 nM) and seeded into double-layer agar cultures in 6-well plates at a concentration of about1000 cells per well. Cells were cultured in 5% CO2 atmosphere at 37 °C for 15 days. After cultivation, the cells were fixed in methanol for 15 min and stained with crystal violet for at least 30 min. Colony formation was analyzed using an inverted phase-contrast microscope (Nikon Eclipse Ti, Japan).
Western blotting
Total proteins were extracted from cells and tumor tissues using RIPA lysis buffer (Beyotime Biotechnology) supplemented with 100× protease inhibitor cocktail (TargetMol) and 50× phosphatase inhibitor cocktail (Bimake). Protein concentrations were determined using a BCA assay kit (Beyotime Biotechnology) following manufacturer’s protocols. Equal amounts of protein (30 µg/lane) were resolved on 10% SDS-polyacrylamide gels (Epizyme) and transferred to polyvinylidene fluoride (PVDF) membranes (Millipore) using a wet transfer system at 100 V for 90 min. Membranes were blocked with 5% bovine serum albumin (BSA) in TBST for 2 h at room temperature, followed by overnight incubation at 4 °C with primary antibodies: STAT1 (Rabbit polyclonal), β-Actin (Mouse monoclonal). After TBST washes, membranes were incubated for 1 h with HRP-conjugated secondary antibodies: Anti-Rabbit IgG (Cell Signaling Technology), Anti-Mouse IgG (Abcam). Signals were developed using ECL Prime Western Blotting Detection Reagent (Cytiva) and quantified with ImageJ software.
Flow cytometry analysis of cell apoptosis
To analyze apoptosis and NK cell activation, Jurkat T cells were harvested by centrifugation at 300 ×g for 5 min at 4 °C, washed twice with ice-cold PBS containing 2% FBS, and assessed for viability using Trypan Blue exclusion (> 95% viable cells). For apoptosis detection, cells were resuspended in Annexin V Binding Buffer and stained with FITC-conjugated Annexin V antibody (Biolegend) and Propidium Iodide (PI, Biolegend) for 15 min in the dark. For NK cell activation, cells were stained with APC-conjugated anti-mouse CD3ε (Biolegend) and PE-conjugated anti-mouse CD69 (Biolegend) in PBS containing 2% FBS for 30 min at 4 °C, followed by washing and fixation with 1% paraformaldehyde. Flow cytometry was performed using a BD FACSCanto II system, with specific filters for Annexin V (530/30 nm), PI (670/14 nm), CD3 (660/20 nm), and CD69 (575/26 nm). Gating strategies were employed to identify viable, apoptotic, and activated NK cell populations. All experiments data analyzed using GraphPad Prism 9.5 with one-way ANOVA followed by Tukey’s post-hoc test.
Construction of mice HCC model
C57BL/6 mice were derived from Shanghai Slack Laboratory Animal Co., LTD. C57BL/6 mice (3–4 weeks old) were placed in specific pathogen free facilities. These experimental plans involving animals had been approved by the Clinical Research and Animal Experimental Institution Ethics Committee of Mengchao Hepatobiliary Hospital of Fujian Medical University. Hepa1-6 or Hep 53.4 cells (3 × 106 cells, 200µL) inject into the armpits of the mice21. After 4 days, the mice were randomly divided into 4 groups (n = 5): (1) physiological saline (negative control); (2) Apatinib; (3) PD-1; (4) Apatinib and PD-1, 5 in each group. Oral administration of physiological saline and apatinib, 3 times a week, at a dose of 50 mg/kg; Anti PD-1 antibody is administered intraperitoneally three times a week at a dose of 10 mg/kg. Measure the tumor size weekly according to formula a × Measure the maximum diameter (a) and its short diameter (b) of the tumor, and estimate the tumor volume. Draw swelling based on tumor volume and implantation time. At the conclusion of the experiment or when the tumor volume in mice reaches 2000mm3, euthanasia will be performed using the cervical dislocation method.
Immunohistochemistry assay
HCC tumor tissues from mice were fixed with 4% paraformaldehyde for 24 h and embedded in paraffin. For immunohistochemistry, sections were deparaffinized, rehydrated, and subjected to antigen retrieval using citrate buffer (pH 6.0) at 95 °C for 20 min. Endogenous peroxidase activity was blocked with 3% hydrogen peroxide for 10 min. After blocking with 5% bovine serum albumin (BSA) for 1 h at room temperature, sections were incubated with primary antibodies overnight at 4 °C. The primary antibodies used were as follows: anti-CD8 antibody: Rat monoclonal anti-mouse CD8a (Clone 4SM15, eBioscience), anti-NK cell antibody: Rat monoclonal anti-mouse CD161/NK1.1, anti-STAT1 antibody: Rabbit monoclonal anti-STAT1 (Cell Signaling Technology), anti-CD31 (Dianova DIA-310, Germany, 1:100 dilution) for vascular endothelial cell staining, anti-α-Smooth Muscle Actin (SMA) (Abcam, UK, 1:200 dilution) for pericytes, anti-PD-L1 (Cell Signaling Technology, USA, 1:100 dilution) for PD-L1 expression, and anti-Carbonic Anhydrase IX (CAIX) (Abcam, UK, 1:200 dilution) for hypoxia detection. After washing with PBS, sections were incubated with horseradish peroxidase (HRP)-conjugated secondary antibodies (Dako, Denmark, 1:500 dilution) for 1 h at room temperature. The signal was visualized using 3,3’-diaminobenzidine (DAB) substrate (Vector Laboratories, USA), and sections were counterstained with hematoxylin.
For immunofluorescence, paraffin-embedded sections were deparaffinized and rehydrated as described above. Antigen retrieval was performed using citrate buffer (pH 6.0) at 95 °C for 20 min. Sections were blocked with 5% BSA for 1 h at room temperature and then incubated with primary antibodies overnight at 4 °C. The primary antibodies used were anti-CD31 (Millipore, USA, 1:100 dilution), anti-SMA (Abcam, UK, 1:200 dilution), anti-PD-L1 (Cell Signaling Technology, USA, 1:100 dilution), and anti-CAIX (Abcam, UK, 1:200 dilution). After washing with PBS, sections were incubated with fluorochrome-conjugated secondary antibodies (Cy3, Alexa Fluor 647; Jackson ImmunoResearch, USA, 1:500 dilution) for 1 h at room temperature in the dark. Nuclei were counterstained with 4’,6-diamidino-2-phenylindole (DAPI; Thermo Fisher Scientific, USA, 1:1000 dilution) for 5 min. Sections were mounted with anti-fade mounting medium (Vector Laboratories, USA) and imaged using a fluorescence microscope (Nikon Eclipse Ti2, Japan). For CD8 + T cells: Armenian Hamster monoclonal anti-mouse CD8a (BioLegend), rat monoclonal anti-mouse CD3ε (Clone 17A2, Tonbo Biosciences, catalog # 40 − 0032), diluted in blocking buffer. For NK cells: Rat monoclonal anti-mouse CD161 (Clone 2D12), diluted in blocking buffer.
The total number of blood vessels, the number of blood vessels surrounded by pericytes, and the proportion of hypoxic areas within the tumor were quantified by randomly selecting 5 fields of view at 400× magnification. Image analysis was performed using ImageJ and GraphPad Prism software.
Tumor tissues from mice were fixed in 4% paraformaldehyde for 24 h, embedded in paraffin, and sectioned at 4 μm thickness. Sections were stained with hematoxylin (Sigma-Aldrich) for 5 min and eosin (Sigma-Aldrich ) for 3 min following standard protocols. Stained slides were imaged under a light microscope (Nikon Eclipse E100) at 10× and 20× magnification. Tumor necrosis was quantified independently by two pathologists who were blinded to the treatment groups. The percentage of necrotic area per tumor section was calculated and used to determine the tumor necrosis score.
Statistical analysis
The statistical analysis was conducted using GraphPad Prism 6.0 software. The data were presented as mean ± standard deviation. All in vitro and in vivo experiments were replicated a minimum of three times, with at least three samples collected during each replication. Two-tailed Student’s t-tests, one-way analysis of variance (ANOVA), or two-way ANOVA were performed to compare the differences between treatment groups. Significant statistical difference was defined as p-values < 0.05.
Results
Inhibitory effect of apatinib for HCC cells
First, the inhibitory effect of apatinib for HCC cells was evaluated. As shown in Fig. 1A, the cell viability was analyzed by calculating the alive cells’ number after using apatinib from 0 h to 24 h. Initially (0 h), the number of the HCC cells in the control group and the treated group (apatinib group) was basically the same. After 24 h, the hepa1-6 cells in the control group grew from about 7 × 104 cells/mL to nearly 1.5 × 105/mL. However, the number of hepa1-6 cells in the treated group was decreased from about 7 × 104/mL to approximately 5 × 105/mL. Compared to the control group, the cell number of the treated group was markedly reduced after 24 h (p < 0.001). Apart from the cells number, it was obvious that the condition of hepa1-6 cells deteriorated following apatinib treatment.
Experimental Study on the Inhibition of Cell Proliferation and Promotion of Apoptosis in Hepatocellular Carcinoma Cells by Apatinib. (A) Apatinib exhibits cytotoxic effects on Hepa1-6 hepatocellular carcinoma cells within 24 h. (B) Apatinib inhibits the proliferation of Hepa1-6 hepatocellular carcinoma cells. (C) Apatinib can induce significant apoptosis in Hepa1-6 hepatocellular carcinoma cells. (D) Apatinib inhibits the proliferation of Hep 53.4 hepatocellular carcinoma cells. (E) Apatinib can induce significant apoptosis in Hep 53.4 hepatocellular carcinoma cells.
Then, the effect of apatinib on the colony-forming ability of hepa1-6 cells was investigated. Cells were seeded at a density of 1000 cells per well in 6-well plates and treated with 500 nM apatinib or vehicle for 10 days. The colonies were then fixed, stained with crystal violet, and counted. As demonstrated in Fig. 1B, the number of colonies in the apatinib-treated group decreased significantly from 172 colonies per well in the control group to 157 colonies per well (p < 0.001), indicating that apatinib suppressed the colony-forming capacity of hepa1-6 cells.
Following that, in order to analyze the relationship between the cell apoptosis and the use of apatinib, flow cytometry was used to evaluate the index of the apoptosis. Figure 1C illustrates that the apoptotic cell rate escalated from approximately 6% to nearly 16%. Consequently, apatinib exerted an inhibitory effect on hepa1-6 cell viability and colony formation, while simultaneously promoting cell apoptosis (p < 0.001).
To further validate the above conclusions, we repeated the experiments using the hep53.4 cell line. The cell colony formation results showed that the number of colonies in the apatinib-treated group decreased significantly (Fig. 1D). Flow cytometry analysis revealed that tumor cells in the apatinib-treated group also underwent significant apoptosis (Fig. 1E).
Apatinib enhancing the efficacy of PD-1 inhibitors
To establish a subcutaneous Hepa1-6 and Hep 53.4 tumor-bearing mice model (3*106 cells/200 µL, Fig. 2A), mice were treated with four different regimens: PBS, apatinib, PD-1, and a combination of apatinib and PD-1. The mouse models treated with PBS (control), apatinib (50 mg/kg, oral gavage, 3 times a week), PD-1 inhibitor (10 mg/kg, intraperitoneal injection, 3 times a week), or a combination of apatinib and PD-1 inhibitor for 2 weeks (Fig. 2B). Tumor volume was measured every 3 days using calipers, and mice were euthanized at the end of the treatment period for tumor dissection. As shown in Fig. 2C, Hepa1-6 tumor growth was significantly inhibited in all treatment groups compared to the PBS group, with the combination group showing the most pronounced reduction in tumor volume. A 30-day survival observation was conducted on mice, revealing that the PBS group had the poorest survival rate, while no deaths occurred in the combination therapy group during the observation period, with the difference being statistically significant (p < 0.001) (Fig. 2D). To further verify the therapeutic efficacy, we conducted validation using the hep53.4 cell line. The results showed that the tumor volume reduction was most significant in the combination group (Fig. 2E). Moreover, in the survival analysis, no deaths were observed in mice from the combination group (Fig. 2F). The photographs of tumor sizes dissected after euthanizing the mice confirmed that the combine group represent the most significant and rapid reduction in Hepa1-6 tumor volume (Fig. 2G). To validate the role of the STAT1/NK axis in the effects of apatinib and PD-1, Western blot analysis was further performed to detect STAT1 expression in each treatment group, and immunohistochemistry was utilized to assess the expression of CD8-positive T cells and NK cells in each group. The western blot results confirmed that STAT1 expression was significantly elevated in the apatinib treatment group and the combination treatment group relative to the control and PD-1 monotherapy groups (Fig. 2H).
The efficacy of apatinib in combination with PD-1 inhibitors in vitro subcutaneous tumor models for the treatment of HCC. (A) Create a model diagram for the in vitro subcutaneous tumor model for HCC. (B) Treatment cycle diagram. (C) Changes in tumor volume for each treatment group in Hepa1-6. (D) Survival rate curves for each treatment group. (E) Changes in tumor volume for each treatment group in Hep 53.4. (F) Survival rate curves for each treatment group in Hep 53.4. (G) Photos of Hepa1-6 tumor volume changes for each treatment group. (H) Western blotting to detect the expression of STAT1 protein in Hepa1-6 tumor tissue of each treatment group.
Combination therapy induces tumor necrosis and activates STAT1/NK axis
Tumor necrosis scoring was performed on Hepa1-6 tumor sections from each group, and the differences in scores between the combination therapy group, monotherapy groups, and control group were compared. The results showed that the necrosis score in the combination therapy group was significantly higher than that in the other groups, providing strong support for the efficacy of the combination therapy (Fig. 3A). Compared to the control group and the PD-1 monotherapy group, the expression of CD8-positive T cells was also significantly higher in the apatinib treatment group and the combination treatment group (Figs. 3B). Immunofluorescence further validated this conclusion. (Figs. 3C). Flow cytometry analysis of the percentage of NK cells in tumors from each group revealed that the combination group exhibited a significantly higher proportion of NK cells (Figs. 3D). Immunofluorescence further validated this conclusion. (Figs. 3E). The STAT-positive score further validated the activation effect of the combination therapy on STAT1 (Figs. 3F). In the above results, the p-values for Apa + PD-1 vs. other groups were all less than 0.001. These findings suggested that apatinib activated STAT1, possibly enhancing the efficacy of PD-1 through the STAT1/NK axis.
Evaluation of immune cell expression in the subcutaneous tumor model of HCC after combination therapy with apatinib and PD-1. (A) HE staining of tumor tissues in each treatment group. (B) Immunohistochemistry method to detect the infiltration of CD8-positive T cells in tumor tissues of each treatment group. (C) Immunofluorescence method to detect the infiltration of CD8-positive T cells in tumor tissues of each treatment group. (D) Flow cytometry method to detect the infiltration of NK cells in tumor tissues of each treatment group. (E) Immunofluorescence method to detect the infiltration of NK cells in tumor tissues of each treatment group. (F) Immunohistochemistry method to detect the expression of STAT1 protein in tumor tissue of each treatment group.
Validation of STAT1/NK axis mediates the anti-tumor efficacy of combination therapy
To validate the role of the STAT1/NK axis in the tumor-suppressive effects of the combination therapy, We established a STAT1-OE Hepa1-6 cell line. Then, the subcutaneous tumor-bearing mice model was hereby re-established (Fig. 4A). The treatment protocol is shown in Fig. 4B. As depicted in Figs. 4C, the combination treatment group presented the lowest tumor volume, and the tumor-killing effects of apatinib and PD-1 were significantly enhance in STAT1-OE Hepa1-6 tumor moedel. As indicated by Fig. 4D, after STAT1 overexpression, the survival of mice in each group was significantly improved, while the PBS group had the lowest survival rate. The photographs of tumor sizes dissected after euthanizing the mice confirmed that the tumor-killing effect of the combination therapy was significantly enhance in STAT1-OE Hepa1-6 cell line (Fig. 4E). As shown in Figs. 4F, immunofluorescence showed that in the STAT1-OE Hepa1-6 cell line, the number of NK cells significantly increased in the combined treatment group. These results further confirmed that stat1demonstrated a strong effect in anti-tumor activity. Subsequently, we treated mice with the fludarabine group (a STAT1 inhibitor) and the combination treatment with the Asialo GM1 group (an NK cell-depleting antibody) to further validate our conclusion. The treatment protocol is shown in Fig. 5A. As depicted in Figs. 5B, the combination treatment group presented the lowest tumor volume, and the tumor-killing effects of apatinib and PD-1 were significantly reduced upon treatment with fludarabine and Asialo GM1. As indicated by Fig. 5C, the combination treatment group exhibited the highest survival rate, while the PBS group had the lowest. The survival rate decreased following treatment with fludarabine and Asialo GM1. The photographs of tumor sizes dissected after euthanizing the mice confirmed that the tumor-killing effect of the combination therapy was significantly reduced following the administration of inhibitors (Fig. 5D). After administering the inhibitors, the tumor necrosis score in the combination treatment group significantly decreased, indicating a notably weakened therapeutic killing effect(Fig. 5E). Immunohistochemistry confirmed that NK cells were reduced in the combination treatment group after administering the inhibitors (Fig. 5F). Immunofluorescence results further confirmed that NK cells were significantly reduced in the combination treatment group after administering the inhibitors(Fig. 5G).
In STAT1-OE hepa 1–6 cells, the anti-tumor efficacy of apatinib combined with PD-1 is enhanced. (A) Create a model for the in vitro subcutaneous tumor model of STAT1-OE hepa 1–6 cells. (B) Treatment cycle diagram. (C) Changes in tumor volume for each treatment group. (D) Survival rate curves for each treatment group. (E) Photos of tumor volume changes for each treatment group. (F) Immunofluorescence method to detect the infiltration of NK cells in tumor tissues of each treatment group.
Inhibition of the STAT1/NK Axis reduces the anti-tumor efficacy of apatinib and PD-1 combination therapy. (A) Create a tumor model for HCC and treatment cycle diagram. HE staining of tumor tissues in each treatment group. (B) Changes in tumor volume for each treatment group. (C) Survival rate curves for each treatment group. (D) Photos of tumor volume changes for each treatment group. (E) Immunohistochemistry method to assess tumor necrosis score in tumor tissues of each treatment group. (F) Immunohistochemistry method to detect the infiltration of NK cells in tumor tissues of each treatment group. (G) Immunofluorescence method to detect the infiltration of NK cells in tumor tissues of each treatment group.
Combined therapy reshaping the tumor microenvironment in HCC
To gain a deeper understanding of the changes in the immune microenvironment induced by the combination of apatinib and PD-1, the infiltration of immune-related cells in each treatment group was hereby evaluated. Immunofluorescence staining was employed to detect the expression of carbonic anhydrase IX (CA IX) in tumor tissues from each treatment group. The expression of CA IX was found to be significantly reduced in the combination treatment group (Figs. 6A). Regarding angiogenesis, the expression of CD31 in the tumor tissues of the combination treatment group was significantly lower compared to the other groups (Figs. 6B). Additionally, in the tumor stroma, the expression levels of α-smooth muscle actin (α-SMA) were notably decreased in the tumor tissues of the combination treatment group (Figs. 6C). In the above results, the p-values for Apa + PD-1 vs. other groups were all less than 0.05. These findings suggested that the combination of apatinib and PD-1 not only modulated the immune microenvironment by reducing the expression of CA IX, but also inhibited angiogenesis and the activation of stromal cells, which collectively enhanced the overall antitumor effects.
Expression of various indicators in the immune microenvironment of each treatment group. (A) Immunofluorescence method to detect the expression of CA9 in tumor tissues of each treatment group. (B) Immunofluorescence method to detect the expression of CD31 in tumor tissues of each treatment group. (C) Immunofluorescence method to detect the expression levels of α-smooth muscle actin in tumor tissues of each treatment group.
Discussion
The clinical application of the combination of apatinib and PD-1 in the treatment of advanced HCC has shown promising results19,22. However, despite these encouraging outcomes, the exact molecular mechanisms underlying the enhanced antitumor efficacy of this combination remain elusive. In the current study, in vitro experiments demonstrated that apatinib significantly inhibited HCC cell viability and colony formation while inducing apoptosis23. Complementing these findings, the present in vivo studies using mice models confirmed that apatinib enhanced the efficacy of PD-1 through STAT1 activation. Concurrently, the combination with apatinib elicited a synergistic antitumor response via the STAT1/NK axis.
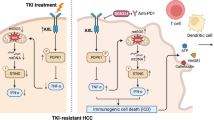
STAT1 activation is a key driver of the tumor microenvironment associated with the response to ICIs24. Previous studies have indicated that apatinib can upregulate IFN-γ expression, a cytokine pivotal in tumor cell proliferation inhibition, angiogenesis suppression, and the promotion of tumor cell apoptosis15. IFN-γ activates STAT1 in tumor cells, leading to the regulation of cyclin-dependent kinase inhibitor 1 expression and ultimately resulting in cell cycle arrest17. Gene set enrichment analysis has also demonstrated that IFN-γ is a critical positive regulator of STAT1 activation25. The significant upregulation of STAT1 in the combination therapy group (Fig. 2F) aligns with its established role in driving interferon-stimulated genes (e.g., CXCL10, IRF1) that recruit cytotoxic immune cells such as NK cells and CD8+ T cells26. This is further supported by our observation of enhanced NK cell infiltration (Fig. 3D) and tumor necrosis (Fig. 3A), which are hallmarks of STAT1-mediated anti-tumor immunity.
Sensitization therapy induces a PD-1-responsive phenotype and is dependent on NK cells27. Apatinib attracts conventional NK cells to the tumor microenvironment in a STAT1-dependent manner, where they synergize with ICIs to kill tumor cells28,29. NK cells not only directly eliminate cancer cells but also modulate the immune system, thereby promoting dendritic cell (DC) maturation and increasing the infiltration of cytotoxic T cells into the tumor site30. Besides, studies have shown that tumor cells that do not express PD-L1 can actually stimulate NK cells to highly express PD-L131. Furthermore, other research has indicated the stronger ability of NK cells to recognize and attack tumor cells that do not express PD-L132. Herein, the results suggested that apatinib increased PD-1 sensitivity and synergized with PD-1 to exert a more potent antitumor effect, facilitating comprehensive tumor eradication. In our research, the level of STAT1 was not significantly increased in PD-1 monotherapy, suggesting that tumor regression in PD-1 monotherapy may involve alternative mechanisms, such as: (1) Reactivation of exhausted T cells; (2) Reduction of regulatory T cells or myeloid-derived suppressor cells in the tumor microenvironment. These pathways have been well-documented in previous studies33. The significant increase in STAT1 in the combination therapy group indicates a synergistic interaction between APA and PD-1 inhibitors, further confirming the role of the combination therapy in modulating the STAT1/NK axis.
The combination therapy has significant implications for angiogenesis, with studies indicating that blood vessel proliferation can occur post-treatment with apatinib or anti-PD-1. Previous analysis of angiogenesis biomarkers, including CA9, CD31, and α-smooth muscle actin (α-SMA), revealed that these markers were inhibited after combination therapy, indicating that angiogenesis was suppressed. However, a limitation of this study is the lack of direct measurement of STAT1 downstream targets. Future work will include profiling STAT1-regulated genes (e.g., IRF1, CXCL10) to further elucidate its functional role in the combination therapy’s efficacy. Moreover, the experiments were limited to a single cell line, which, without cross-validation on multiple cell lines, might affect the broader applicability of the findings.
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- ICIs:
-
immune checkpoint inhibitors
- TME:
-
tumor microenvironment
- HCC:
-
hepatocellular carcinoma
- STAT1:
-
transcription 1
- NK:
-
natural killer
- Tregs:
-
abundant regulatory T cells
- IFN-γ:
-
alter interferon-γ
- PFS:
-
progression-free survival
- OS:
-
overall survival
- PD-1:
-
programmed cell death protein 1
- α-SMA:
-
α-smooth muscle actin
- PBS:
-
Phosphate-Buffered Saline
References
Kulik, L. & El-Serag, H. B. Epidemiology and management of hepatocellular carcinoma. Gastroenterology 156, 477–491e1 (2019).
Bray, F. et al. Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 74, 229–263 (2024).
Li, Q., Han, J., Yang, Y. & Chen, Y. PD-1/PD-L1 checkpoint inhibitors in advanced hepatocellular carcinoma immunotherapy. Front. Immunol. 13, 1070961 (2022).
Rizzo, A., Brunetti, O. & Brandi, G. Hepatocellular carcinoma immunotherapy: predictors of Response, Issues, and challenges. Int. J. Mol. Sci. 25, 11091 (2024).
Gao, M. et al. PD-1 regulation in immune homeostasis and immunotherapy. Cancer Lett. 588, 216726 (2024).
Oura, K., Morishita, A., Tani, J. & Masaki, T. Tumor immune microenvironment and immunosuppressive therapy in hepatocellular carcinoma: A review. Int. J. Mol. Sci. 22, 5801 (2021).
Xing, R., Gao, J., Cui, Q. & Wang, Q. Strategies to improve the antitumor effect of immunotherapy for hepatocellular carcinoma. Front. Immunol. 12, 783236 (2021).
Palakurthi, B. et al. Targeting CXCL16 and STAT1 augments immune checkpoint Blockade therapy in triple-negative breast cancer. Nat. Commun. 14, 2109 (2023).
Li, Y-J., Zhang, C., Martincuks, A., Herrmann, A. & Yu, H. STAT proteins in cancer: orchestration of metabolism. Nat. Rev. Cancer. 23, 115–134 (2023).
Khushalani, N. I. et al. Phase II clinical and immune correlate study of adjuvant nivolumab plus ipilimumab for high-risk resected melanoma. J. Immunother Cancer. 10, e005684 (2022).
Horowitch, B. et al. Subsets of IFN signaling predict response to immune checkpoint Blockade in patients with melanoma. Clin. Cancer Res. 29, 2908–2918 (2023).
Wong, G. L., Manore, S. G., Doheny, D. L. & Lo, H-W. STAT family of transcription factors in breast cancer: pathogenesis and therapeutic opportunities and challenges. Semin Cancer Biol. 86, 84–106 (2022).
Verhoeven, Y. et al. The potential and controversy of targeting STAT family members in cancer. Semin Cancer Biol. 60, 41–56 (2020).
Zheng, X. et al. Tumor cell-intrinsic SETD2 inactivation sensitizes cancer cells to immune checkpoint Blockade through the NR2F1-STAT1 pathway. J. Immunother Cancer. 11, e007678 (2023).
Yang, Y. et al. Apatinib prevents natural killer cell dysfunction to enhance the efficacy of anti-PD-1 immunotherapy in hepatocellular carcinoma. Cancer Gene Ther. 28, 89–97 (2021).
Han, J., Wu, M. & Liu, Z. Dysregulation in IFN-γ signaling and response: the barricade to tumor immunotherapy. Front. Immunol. 14, 1190333 (2023).
Jeong, W-I., Park, O., Radaeva, S. & Gao, B. STAT1 inhibits liver fibrosis in mice by inhibiting stellate cell proliferation and stimulating NK cell cytotoxicity. Hepatology 44, 1441–1451 (2006).
Liu, M. et al. Induction chemotherapy followed by camrelizumab plus apatinib and chemotherapy as first-line treatment for extensive-stage small-cell lung cancer: a multicenter, single-arm trial. Signal. Transduct. Target. Ther. 10, 65 (2025).
Qin, S. et al. Camrelizumab plus Rivoceranib versus Sorafenib as first-line therapy for unresectable hepatocellular carcinoma (CARES-310): a randomised, open-label, international phase 3 study. Lancet 402, 1133–1146 (2023).
Farkona, S., Diamandis, E. P. & Blasutig, I. M. Cancer immunotherapy: the beginning of the end of cancer? BMC Med. 14, 73 (2016).
Chen, H. et al. Personalized neoantigen vaccine combined with PD-1 Blockade increases CD8 + tissue-resident memory T-cell infiltration in preclinical hepatocellular carcinoma models. J. Immunother Cancer. 10, e004389 (2022).
Zhang, T-Q. et al. Camrelizumab (a PD-1 inhibitor) plus apatinib (an VEGFR-2 inhibitor) and hepatic artery infusion chemotherapy for hepatocellular carcinoma in Barcelona clinic liver cancer stage C (TRIPLET): a phase II study. Signal. Transduct. Target. Ther. 8, 413 (2023).
Li, H. et al. Apatinib: A novel antiangiogenic drug in monotherapy or combination immunotherapy for digestive system malignancies. Front. Immunol. 13, 937307 (2022).
Tao, Q. et al. Mefloquine enhances the efficacy of anti-PD-1 immunotherapy via IFN-γ-STAT1-IRF1-LPCAT3-induced ferroptosis in tumors. J. Immunother Cancer. 12, e008554 (2024).
Zemek, R. M. et al. Sensitization to immune checkpoint Blockade through activation of a STAT1/NK axis in the tumor microenvironment. Sci. Transl Med. 11, eaav7816 (2019).
Jandl, K. et al. Impairment of the NKT-STAT1-CXCL9 axis contributes to vessel fibrosis in pulmonary hypertension caused by lung fibrosis. Am. J. Respir Crit. Care Med. 206, 981–998 (2022).
Contribution of NK cells. to immunotherapy mediated by PD-1/PD-L1 blockade - PubMed [Internet]. [cited 2024 Dec 16]. Available from: https://pubmed.ncbi.nlm.nih.gov/30198904/
Raja, R. et al. PP4 Inhibition sensitizes ovarian cancer to NK cell-mediated cytotoxicity via STAT1 activation and inflammatory signaling. J. Immunother Cancer. 10, e005026 (2022).
Meissl, K. et al. STAT1 isoforms differentially regulate NK cell maturation and Anti-tumor activity. Front. Immunol. 11, 2189 (2020).
Barry, K. C. et al. A natural killer-dendritic cell axis defines checkpoint therapy-responsive tumor microenvironments. Nat. Med. 24, 1178–1191 (2018).
Lin, M. et al. Pembrolizumab plus allogeneic NK cells in advanced non-small cell lung cancer patients. J. Clin. Invest. 130, 2560–2569 (2020).
Samareh Salavatipour, M., Poursalehi, Z., Hosseini Rouzbahani, N., Mohammadyar, S. & Vasei, M. CRISPR-Cas9 in basic and translational aspects of cancer therapy. Bioimpacts 14, 30087 (2024).
Geels, S. N. et al. Interruption of the intratumor CD8 + T cell:treg crosstalk improves the efficacy of PD-1 immunotherapy. Cancer Cell. 42, 1051–1066e7 (2024).
Funding
This study was supported by Fujian Provincial Natural Science Foundation (2021J011290); China Hepatitis Prevention and Treatment Foundation Tianqing Liver Disease Research Fund (TQGB20210098); Fujian Provincial Natural Science Foundation (2024J011222).
Author information
Authors and Affiliations
Contributions
Study concept and design: Zi-sen Lai. Acquisition of data and prepared figures1-3: Linsheng Cui, Meng-ru Wei, Jun Fu. Statistical analysis and prepared figures4-5: Jie-ru Guo, Qiao Ke, Qi-zhen Huang, Luo-bin Guo, You-shi Zheng. Drafting of the manuscript: Ting-feng Huang. Obtained funding: Zi-sen Lai. Study supervision: Zi-sen Lai.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval and consent to participate
The study was approved by the Ethics Committee of the Mengchao Hepatobiliary Hospital of Fujian Medical University (IRB: MCHH-AEC-2024-009). All experimental procedures strictly followed the ARRIVE guidelines and applicable legal requirements.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Cui, Ls., Wei, Mr., Fu, J. et al. Apatinib-Induced STAT1/NK axis activation augments PD-1 inhibitor efficacy in advanced Hepatocellular Carcinoma. Sci Rep 16, 2497 (2026). https://doi.org/10.1038/s41598-025-32228-y
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-32228-y