Abstract
Volatiles emitted from black smokers provide crucial insights into mantle and hydrothermal processes. This study investigated the Cheoeum (CVF) and MIRAE-2 (M2VF) vent fields in the 12–16°S region of the Central Indian Ridge (CIR) using RV ISABU and ROV ROPOS. We measured 3He/4He, 4He/20Ne, CH4 concentration, δ13C of CH4, and 222Rn activity in 78 seawater samples from vent fluids and the overlying plume. The end-member 3He/4He ratio is 7.94 Ra, indicating the MORB mantle source. Vent fluids show high CH4 concentrations (up to 11,578 nM/kg) and anomalously heavy δ13C values (-11.23 to -7.39 ‰), suggesting abiotic near-quantitative CO2 reduction in closed-system environments such as olivine-hosted fluid inclusions. Principal Component Analysis distinguished the hydrothermal fluids from background waters by analyzing chemical proxies and spatial information. This first volatile-geochemistry dataset from the CIR provides direct isotopic evidence for mantle-derived helium and abiotic methane, offering a key reference for understanding deep-sea carbon cycling and chemosynthetic ecosystems at slow-spreading ridges.
Similar content being viewed by others
Introduction
Mid-ocean ridges (MOR) are the most volcanically active tectonic environments on Earth, accounting for more than two-thirds of the global annual volcanic output1. In addition to erupting enormous amounts of magma to produce oceanic crust, MORs also form extensive hydrothermal systems through interactions with seawater, discharging volumes an order of magnitude greater than those from arc or back-arc settings2. These hydrothermal emissions represent a significant pathway for the transport of heat, metals, and volatiles from the Earth’s interior to the ocean3,4,5. Owing to its elongated and continuous volcanic architecture, combined with the relative absence of crustal overprint, MORs serve as natural windows into the upper mantle, allowing access to geochemical signals directly derived from the asthenosphere6.
Since the first discovery of a submarine hydrothermal vent at the Galápagos Rift in 1979 (Ref.7), seafloor hydrothermal systems have been widely investigated as significant sites for understanding the geochemical cycling and the deep biosphere. Among the various tracers of hydrothermal and mantle processes, noble gas isotopes-particularly 3He/4He-have proven to be robust indicators of mantle source characteristics8,9. Typical mid-ocean ridge basalt (MORB) values range from 7 to 9 Ra (where 1 Ra = atmospheric 3He/4He = 1.39 × 10–6)10, while higher values (> 10 Ra) suggest influence from deep-seated mantle plumes11. Additionally, when combined with other geochemical tracers-including trace elements and isotopes of volatiles such as N2, CO2, and helium-these measurements can effectively constrain interactions among the asthenospheric mantle, lithospheric mantle, and oceanic crust12,13,14,15. Methane (CH4), in particular, plays a dual role as both a key energy source for chemosynthetic microbial ecosystems and as a conservative tracer of vent-derived fluids, detectable using oxidation–reduction potential (ORP) sensors16,17. Meanwhile, 222Rn serves as a complementary tracer owing to its chemical inertness and intermediate half-life (3.82 days), enabling tracking of fluid residence times and sedimentary input18,19,20. However, 222Rn distribution can also be influenced by regional factors such as bathymetry and sediment cover, which remain poorly constrained in the CIR.
In the Indian Ocean, three main ridge systems diverge from the Rodriguez Triple Junction (Fig. 1): the Central Indian Ridge (CIR), the Southwest Indian Ridge (SWIR), and the Southeast Indian Ridge (SEIR). The CIR is of particular interest due to its slow-spreading rate (30 to 49 mm/yr)21,22 and distinct tectonic segmentation. The central region of the CIR is primarily divided into six segments by transform faults, placing several recently discovered hydrothermal vent fields between 8°S and 12°S 17. Between 8°S and 17°S in the CIR, regional differences in 3He/4He ratios of MORB glasses have been reported, with lower (7.5 to 9.2 Ra) and higher (8.6 to 11.8 Ra) values in the southern (12.25°S to 16.85°S) and northern (7.94°S to 11.53°S) segments, respectively6. These variations suggest possible mixing between a MORB-like upper mantle and a plume-derived component, such as that associated with the Réunion hotspot23. However, most of these studies have focused on basaltic rock samples, with relatively few fluid-based geochemical datasets available from the CIR24,25.
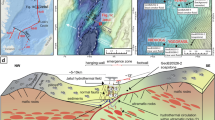
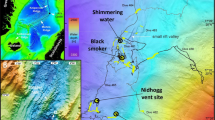
Information about the study area. (a) Map and locations of the sampling sites. Each hydrothermal site is highlighted in red. Ridge tomography was generated from internal multibeam data, and inset global map was drawn from GEBCO 2020 grid29;both were visualized using QGIS30 (b) Photos of active hydrothermal vents taken with a Remotely Operated Vehicle (ROV). (c, d) Cross-sectional bathymetry of hydrothermal vents (marked with blue stars), oriented perpendicular to the spreading center. White bands with dashed lines indicate sections where data gaps were filled by Gaussian interpolation. (e) Cross-sectional bathymetry along sampling sites with helium isotope profile overlaid. Contours are interpolated for visualization only; white dots indicate actual CTD sampling locations and yellow diamonds indicate actual ROV sampling locations.
In 2021, we conducted the first detailed visual survey of two active hydrothermal vent sites26-the Cheoeum vent field (CVF) and the Mirae-2 vent field (M2VF)-via R/V ISABU and Remotely Operated Vehicle (ROV) Remotely Operated Platform for Ocean Science ROPOS by Canadian Scientific Submersible Facility (CSSF)27. Both sites are ultramafic-hosted black smoker systems: CVF, located at segment 4, reaches a maximum vent fluid temperature of 318 °C, while M2VF, at segment 5, reaches 244 °C. However, compared with other slow-spreading ridges such as the Mid-Atlantic Ridge and the Southwest Indian Ridge-where methane and noble gas geochemistry in hydrothermal fluids have been extensively studied8,11,28-the CIR remains poorly characterized. This scarcity of fluid-based datasets, particularly for methane and its isotopes, underscores the need for new measurements to capture the full spectrum of volatile cycling and mantle processes along the CIR.
To address this gap, we present the first comprehensive dataset of volatile geochemistry from newly discovered hydrothermal vent fields along the CIR between 12°S and 16°S. Vent fluid and overlying plume water were directly sampled using a Remotely Operated Vehicle (ROV), enabling precise recovery of minimally diluted end-member fluids. This study provides new insights into the helium isotope signature of the CIR mantle, the origin of CH₄ in reducing CIR environments, and the distribution of 222Rn as a complementary tracer of hydrothermal inputs. By linking these fluid-based observations with existing rock-based geochemical data, we aim to refine our understanding of mantle heterogeneity, volatile generation mechanisms, and fluid-crust–mantle interactions in a slow-spreading ridge setting (Fig. 2).
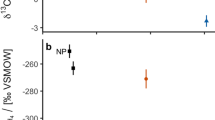
Vertical profiles of hydrothermal plume tracers from two vent fields along the Central Indian Ridge. (a) Profiles from the Cheoeum Vent Field (CVF), and (b) profiles from the MIRAE2 Vent Field (M2VF). Each panel shows the depth distribution of 3He/4He (both R/Ra and Δ3He, blue), CH4 concentration (nmol/kg, green), δ13C of CH4 (‰ vs vPDB, orange), and 222Rn activity (dpm/L, red). Samples collected by ROV are indicated by diamond symbols. Black lines connect samples from sequential depths to illustrate plume structures. Error bars represent analytical uncertainties. See Methods 5.1–5.3 for detailed sampling and measurement procedures.
Results
All measurement results are summarized in Supplementary Table S1. During five ROV dives, we collected four vent fluid samples from two vent sites (R2101-SF, R2101-PA, R2106-NIS01, and R2106-NIS-02). In addition, three bottom water samples were collected at locations showing the most significant Oxidation–Reduction Potential (ORP) drops (R2173-NIS01, R2174-NIS01, and R2175-NIS01). Above these vent sites, 8 CTD casts were performed, yielding 71 seawater samples by CTD-rosette sampling (see Method 5.1). In this study, we refer to the combination of 71 CTD samples and three bottom water samples as “clear seawater samples.” The 3He/4He and 4He/20Ne ratios of the clear seawater samples range from 1.05 to 1.36 Ra and from 0.233 to 0.501, respectively. In contrast, the vent fluid samples exhibited significantly higher and more variable values (3.99 to 6.72 Ra for 3He/4He; 0.465 to 1.307 for 4He/20Ne). The δ13C values of CH4 were broadly distributed between -71.28‰ and -7.39‰ (versus VPDB), with the vent fluid samples showing the highest δ13C values (-11.23‰ to -7.39‰). Radon (222Rn) activity was measured in 71 seawater samples. Eight samples from CTIR2105 were excluded from 222Rn analysis due to the absence of detectable hydrothermal signals in ORP readings and low radioactivity. Measured 222Rn activities range from 0.003 to 0.904 dpm/L, with an average analytical error of ± 0.075 dpm/L. Vent fluid samples exhibited higher 222Rn activities (0.594 to 0.904 dpm/L) than the other seawater samples (0.003–0.601 dpm/L). To distinguish between excess and supported 222Rn, we remeasured five samples after 10 months-three CTD samples (CTIR2101-01, CTIR2101-08, and CTIR2101R-03) and two vent fluid samples (R2101-SF and R2106-01)-allowing > 236 half-lives of 222Rnexcess to elapse. The CTD samples showed relatively uniform supported 226Ra activities (0.21 ± 0.07, 0.37 ± 0.07, and 0.10 ± 0.07 dpm/L, respectively), while the vent fluid samples exhibited lower and more variable residual activities (0.12 ± 0.06 and 0.20 ± 0.08 dpm/L), confirming the earlier presence of excess 222Rn.
Discussion
He end-member of CIR from 12°S to 16°S
By extrapolating 3He/4He to 20Ne/4He = 0 (i.e., the y-intercept of the ROV sample trend line in Fig. 3), we obtain an end-member 3He/4He value of 7.94 ± 0.12 Ra (1σ). In contrast, such extrapolation is not applicable to the CTD samples due to the large variability in 20Ne/4He relative to the amount of excess 3He resulting from equilibration with atmospheric neon. The end-member 3He/4He ratio of 7.94 Ra indicates a pure MORB-like upper mantle source (8 ± 1 Ra)11, with no detectable plume input-such as from the nearby Réunion hotspot. This strongly suggests a robust and proximal magmatic heat source sustaining high-temperature hydrothermal circulation at CVF and M2VF.The 3He/4He end-member values of hydrothermal vent fluids from other CIR sites-Dodo (7.18 Ra), Solitaire (8.96 Ra)25, and Kairei (7.90 Ra)24-also fall within the MORB range, and align with those from the CVF and M2VF. Globally, the 3He/4He values observed at CIR vent fields are consistent with MORB values from other ridge systems, such as the Mid-Atlantic Ridge and East Pacific Rise, which typically range from 7 to 9 Ra8,11. In contrast, vent fluids sampled near hot spots or plume-affected ridges, such as the Juan de Fuca Ridge and Iceland, have displayed locally elevated 3He/4He signatures (above 10 Ra), reflecting contributions from deep mantle sources11,28. The Réunion hotspot and the Mascarene basin asthenospheric reservoir (MBAR) are thought to supply plume-derived material to the CIR23. Consistent with this, elevated 3He/4He values have been observed in basaltic glasses along the CIR, reaching up to 11.8 Ra between 7.94°S and 11.53°S6 and up to 12.14 Ra between 18.65°S and 20.05°S, respectively32. However, the 3He/4He values of the CVF and M2VF vent fluids do not show such elevated 3He/4He signatures, which is consistent with basaltic glass compositions from similar latitudes. In contrast, the elevated 3He/4He observed in the Solitaire vent fluid (8.96 Ra) matches high helium isotope values reported in basaltic glasses near 19.56°S 32, suggesting possible input of plume-derived 3He into the hydrothermal system at that latitude. Therefore, expanding hydrothermal fluid surveys both northward and southward along the CIR-particularly across latitudes influenced by known plume activity-may help to further identify the spatial extent of mantle plume-derived helium in hydrothermal systems.
3He/4He versus 20Ne/4He diagram. Red diamonds represent ROV-collected samples, while blue diamonds represent CTD-collected seawater samples. The dashed line shows the mixing trend between atmospheric and MORB-like helium. The extrapolated y-intercept corresponds to the 3He/4He end-member value.
Origin of CH4
In Fig. 4a, the negative linear correlation between 1/CH4 concentration and δ13CCH4 (i.e. the positive correlation between CH4 concentration and δ13CCH4) highlights a clear mixing trend between two end-members: one with low δ13C, low CH4, and low Δ3He (i.e. percent deviation from atmospheric 3He/4He), and the other with high δ13C, high CH4, and high Δ3He. Figure 4b further supports this trend, illustrating that samples with high δ13C and high CH4/3Heexcess align along a mixing trajectory toward samples with lower CH4/3Heexcess. The δ13C values of vent fluid CH4 (-11.23 to -7.39‰) are consistent with an abiotic origin33. Two primary mechanisms for abiotic CH4 production at MORs have been proposed: (1) the Sabatier reaction and (2) the Fischer–Tropsch-type (FTT) reaction33:
CH4 and He systematics. (a) δ13CCH4 versus 1/CH4 diagram and (b) CH4/3Heexcess versus δ13C diagram. The diamonds indicate samples from ROV. In (b), mixing lines represent hypothetical trends defined by the outermost samples from three PCA-based groups: the lowest δ13C (group 1), the lowest CH4/3He (group 2), and highest CH4/3He (group 3). The shaded region indicates the typical δ13C range of microbial methane (< -50‰31).
The required H2 for both reactions is hypothesized to originate from either (1) serpentinization of ultramafic rocks or (2) the reductive pyrite-pyrrhotite-magnetite (PPM) redox buffer, both of which are plausible processes in the CIR region25,33.
The anomalously heavy δ13C values of CH4 from CVF and M2VF provide strong evidence for active abiotic methanogenesis. For comparison of CH4 characteristics with other hydrothermal plume data from different MOR settings, we compiled previous research conducted on the CIR34,35,36,37,38. Given a robust mantle helium signal (end-member 3He/4He = 7.94 Ra), the elevated CH4/3He ratios than shallow seawater samples observed at CVF and M2VF must be driven by high methane concentrations, rather than low 3He fluxes. The exceptionally heavy δ13C values observed at CVF are among the heaviest ever reported in hydrothermal systems; to our knowledge, only four samples with higher δ13C have been documented in the literature (Logachev, -6‰37; MARK/Snake Pit, -7.4 to 8.9‰; and Lau basin, -6.1‰). These values approach the upper limit defined by 1.5 times the interquartile range (IQR) for global hydrothermal methane data. The frequent association between high δ13C and high H2 in CIR25,39 suggests that H2 concentrations are likely elevated at CVF and M2VF as well.
However, the anomalously heavy δ13C values (up to -7.4‰) cannot be readily explained by conventional equilibrium or kinetic fractionation associated with either Sabatier or FTT reactions under typical hydrothermal conditions.
In hydrothermal fluids, CO and CO2 can coexist via the water–gas shift (WGS) reaction:
Given the mantle CO2 source (δ13C = -4 to -6.5‰40,41) and equilibrium WGS fractionation at our estimated vent temperatures (≥ 300 °C), where Δ13C (CO2-CO) ranges from approximately 15 to 23‰ (decreasing with increasing temperature42), the expected δ13C value of CO would be approximately -22 to -30‰.
If the system reached isotopic equilibrium via the Sabatier reaction (Eq. 1), the predicted δ13C-CH4 would be approximately -35‰ (Δ13C(CO2-CH4) ≈ 28‰ at 300 °C 42). This is far lighter than our observed values (-11.23 to -7.39‰). Similarly, kinetic fractionation during Fischer–Tropsch synthesis from CO, although producing heavier CH4 than CO2 reduction due to smaller kinetic isotope effects43, would still yield δ13C-CH4 values around -20 to -25‰ based on the estimated CO composition-substantially lighter than observed. Thus, neither FTT nor Sabatier reactions in open circulating systems can account for the observed heavy δ13C signatures.
This discrepancy strongly suggests that a reservoir effect operated during methane formation. In closed-system environments such as olivine-hosted fluid inclusions, serpentinization below ~ 340 °C can drive near-quantitative conversion of limited carbon sources to CH4, allowing the produced CH4 to inherit δ13C values approaching those of the initial mantle carbon source44. This mechanism is supported by observations at Lost City, where efficient CO2 reduction produced CH4 with δ13C = -9 to -14‰45.
Notably, CO2-CH4-bearing fluid inclusions have been documented in gabbros from the nearby Southwest Indian Ridge46. Given the ultramafic-hosted setting of CVF and M2VF47, we propose that the observed heavy δ13C-CH4 values primarily reflect abiotic methane formed via near-quantitative reduction within fluid inclusions, subsequently released into the active hydrothermal system through leaching or fracturing of the host rock.
These findings underscore the need for direct H2 measurements and further constraints on temperature and redox conditions to refine our understanding of the abiotic methane formation mechanisms operating in this region (Fig. 5).
(a) CH4/3He vs δ13C data plotted with global mid-ocean ridge (MOR) references grouped by spreading rate. (b) Box plot of δ13C values from mid-ocean ridges at different spreading rates. Data from CVF and M2VF are plotted together with the slow spreading ridge data. Each box represents the 25th and 75th percentiles (lower and upper quartiles), with the median shown as a line inside. The whiskers indicate 1.5 times the interquartile range (IQR), and outliers are shown as grey dots. All samples shown have δ13C values above the microbial CH4 field (< -50‰31).
Deconvolving source mixtures with PCA
To examine the mixing trends of seawater within the hydrothermal plume, we performed Principal Component Analysis (PCA) using five variables: 3He/4He, δ13C, CH4 concentration, 222Rn activity, and distance from the seafloor. The principal component loadings and the average values of each variable for the three resulting clusters are shown in Table 1, and the distribution of samples in the principal component space (PC1 vs. PC2) is illustrated in Supplementary Fig. S3a. Cluster 3 reflects the strongest hydrothermal influence, characterized by elevated 3He/4He ratios, δ13C values, and CH4 concentrations, whereas Clusters 1 and 2 are associated with ambient seawater. Cluster 1 corresponds to shallower waters, approximately 1,000 m above the seafloor, and is marked by low CH4 and δ13C values and air-equilibrated 3He/4He. In contrast, Cluster 2 is located closer to the seafloor and shows the highest 222Rn activity, indicating an influence from 226Ra-enriched bottom water.
As shown in the vertical profiles (Fig. 2), the depth intervals where 3He/4He, δ13C, and CH4 concentrations peak are closely aligned, indicating a common hydrothermal origin. In contrast, the peaks of 222Rn and 222Rn/3He appear at different depths, suggesting a distinct source, likely 226Ra-supported bottom water. This vertical separation supports a three-end-member mixing scenario involving hydrothermal fluid, Ra-enriched bottom water, and ambient seawater. Similar patterns of radon-influenced deep water have been reported at the TAG vent field19 and the Juan de Fuca Ridge48,49.
Compared to other hydrothermal vent sites such as the TAG, Snakepit19, or Juan de Fuca Ridge 18, the 222Rn activities at the CVF and the M2VF are relatively low, measuring 0.74 ± 0.08 dpm/L and 0.60 ± 0.09 dpm/L, respectively50. The activity levels in overlying plume waters (0.10 ± 0.06 dpm/L) are comparable to background values reported in the Indian Ocean (0.07 to 0.13 dpm/L)51. Given the similar levels of supported 222Rn activity (0.35 to 0.83 dpm/L) across the plume, the detected 222Rn is interpreted as being primarily supported by dissolved 226Ra. Since 222Rn originates exclusively from the radiogenic decay of U and Th, its flux is largely decoupled from hydrothermal activity itself and is instead controlled by the composition of the oceanic crust or that of overlying sediments. Thus, despite evident hydrothermal discharge at the CVF and the M2VF, the 222Rn signal is not sufficient to reconstruct hydrothermal flux in this case. The PCA results align well with the CH4/3He versus δ13C distribution shown in Fig. 4b, where samples form three distinguishable mixing trends. Using the outermost samples from each cluster-R2172-SF (highest CH4/3He), CTIR2108-06 (lowest CH4/3He), and CTIR2107-09 (lowest δ13C)-we drew mixing lines that outline the compositional space of the plume. In Supplementary Fig. S3b and S3c, the data points in Fig. 4a and 4b are colored according to the PCA cluster assignments. Each cluster forms a distinct distribution, supporting the three-component mixing pattern inferred from the PCA analysis. The 222Rn distribution likely reflects both hydrothermal input and regional water mass structure (e.g., Ra-supported bottom water influenced by seafloor morphology and sediment cover). However, the lack of detailed sediment distribution data for the 12–16°S CIR segment limits a comprehensive interpretation of sediment-related 222Rn sources. In light of these constraints, we prioritize 3He/4He and CH4 as tracers of mantle-derived volatiles, while 222Rn variability provides complementary information on spatial water mass structure.
Conclusions
We investigated newly discovered hydrothermal vent fields along the CIR (12°S to 16°S) using the R/V ISABU and the ROV ROPOS CSSF to constrain the sources of volatiles in this region. Based on measured 3He/4He ratios, δ13CCH4 values and 222Rn activities, we identified three different sources of fluids: (1) a hydrothermal end-member enriched in CH4 and 3He, (2) 226Ra-supported bottom water with high 222Rn activity, and (3) ambient seawater. The 3He/4He ratio of the vent fluids (7.94 Ra) corresponds to the MORB value (8 Ra) and matches previous observations from basaltic glass samples in the same region (7.7 – 7.8 Ra)11. This supports the idea that the mantle beneath the CIR has a relatively uniform MORB signature in this section. In addition, the anomalously high δ13CCH4 values (up to -7.4‰) confirm an abiotic origin52. The heavy δ13C-CH4 provide strong evidence for abiotic methanogenesis via near-quantitative CO2 reduction in closed-system environments such as fluid inclusions44, where CH4 inherits isotopic signatures close to the mantle source45,46. Although the 222Rn activity in plume waters was lower than at other vent sites, its distribution was not aligned with that of 3He and CH4. This indicates separate origins and transport pathways, and reflects the presence of multiple water masses19. Overall, our results provide new fluid-based evidence for mantle characteristics along the CIR, complementing earlier rock-based studies6. Expanding this dataset to cover a broader latitudinal range of the CIR and including additional volatile species such as H2 will further improve our understanding of CH4 formation mechanisms and mantle-crust-fluid interactions16,53. This is particularly relevant given the strongly reducing environments associated with slow-spreading ridges, where H2 may play a key role in abiotic processes16. Future work should focus on dissolved H2 and δDCH4 measurements to better constrain the mechanisms of CH4 formation in these settings.
Methods
Sampling methods
The cruise was operated with ‘RV ISABU’ by the KIOST from November to December of 2021. We conducted line surveys to detect hydrothermal vent sites and collect vent fluid samples using the ROV ‘ROPOS (Remotely Operated Platform for Ocean Sciences)’ of CSSF, Canada 27. The ROV has 4 fixed Niskin bottles attached and each of them can be triggered remotely. With monitoring the attached ORP sensor, we conducted the line survey around possible vent site. After visually identifying the hydrothermal vent site, the ROV moved through the hydrothermal fluid plume and remained for 0.5 to 1 min to flush the existing seawater inside the Niskin bottle with the plume water. and triggers the Niskin bottle in the middle of the hydrothermal plume. After recovery of the ROV, we used onboard CTD Rosette of RV ISABU to sample the hydrothermal plume above the vent site. The rosette consists of 36 Niskin bottles, and seawater was sampled from 18 different depths.
When the Niskin bottles with samples have arrived on deck, samples for noble gases are taken as soon as possible to minimize air contamination. First, we take samples in two 55 cm long copper tubes for noble gas analysis. To minimize the presence of air bubbles in the copper tubes and connecting tubing, extra caution was taken during sample collection. Seawater was flushed through the system at a volume more than 20 times that of the copper tube prior to sealing. With connected silicon tubing and plastic tubing clamp, we temporarily seal the copper tubes. Next, CH4 samples were collected into a 125 mL borosilicate bottle. The bottle was washed with 10% HCl solution before sampling and rinsed with seawater twice. Then, using a narrow-bore tubing, the seawater was injected into the bottle from the bottom to eliminate bubbles. After that, we injected saturated 0.2 mL of HgCl2 solution to sanitize the sample. The bottle is sealed with an aluminum cap with butyl rubber by a cap crimper. Lastly, the 222Rn samples are taken into a pre-vacuumed 4 L glass bottle 54. The silicon tubing can absorb radon from the seawater sample, so only the Tygon tubing was used for radon sampling. This sampling sequence was repeated for bottles of every aimed depth (6 ~ 10 bottles at each deployment) that are determined from dropped electric potential of ORP sensor of CTD. After sampling from the Niskin bottle, each end of the temporarily sealed copper tubes is completely sealed with a metal clamp. The CH4 samples were kept in a dark refrigerator until measurement, to minimize any chemical reaction.
Stable isotope analysis and excess 3He assessment
Stable noble gases such as 3He/4He and 4He/20Ne were analyzed in AORI, U-Tokyo. Volatiles from seawater samples were extracted in high-vacuum and separated into glass bottles. H2O, N2, Ar, and other reactive gases were purified by titanium getters at 400 °C and charcoal traps at liquid nitrogen temperature (77 K). After measurement of 4He/20Ne with on-line attached Pfeiffer Prisma QMS 200 Quadrupole Mass Spectrometer (QMS), Neon was trapped by cryogenic pump at 40 K. Then, purified helium was introduced into the noble gas isotopic ratio mass spectrometer (Helix SFT by ThermoFischer) to measure 3He/4He. With measured 3He/4He, 4He/20Ne combined with in-situ salinity and temperature measurements by ROV sensors, we computed excess 3He. We assumed 20Ne was in equilibrium at the given salinity and pressure since it was exposed to the air and computed the amount of 20Ne in all seawater samples 55,56. Then using measured 4He/20Ne, we acquired the 4He concentration of seawater. With 3He/4He at the equilibrium state of each sample 55,56 and each sample’s measured 3He/4He, we can compute the concentration of excess 3He. The CH4 concentration in seawater samples are very low (minimum 1.654 nmol/kg), approaching background levels (~ 2 nmol/kg)57. Therefore, the concentration and δ13C of CH4 were analyzed with a Gas Chromatography—Isotope-Ratio Mass Spectrometer (GC-IRMS) at Nagoya University58,59. After CH4 was separated from other volatiles using the AMEX (Automatic Methane Extraction System) gas chromatography purification line, it was introduced into a Finnigan MAT 252 isotope ratio mass spectrometer (IRMS).
Radon measurement
The activity of 222Rn was measured with RAD7, the portable radon-in-air monitor 54. With a vacuum pump and tubing clamps, the 4 L sampling glass bottle is prepared in the vacuumed state before sampling. When the Niskin bottles arrived on the deck, we connected the glass bottle with the Niskin bottle and opened the clamp until the sample stopped flowing naturally. For the measurement, we connect the sample bottle, the desiccant, and the RAD7 in this flow order. After the internal pump initiates airflow, the headspace of the bottle passes through a desiccant to reduce humidity to below 10%, as elevated moisture levels can interfere with the detection of 210Po by neutralizing charged particles during electrostatic collection, thereby lowering detection efficiency. As the 222Rn is the daughter isotope of 226Ra, the RAD7 detects both excess 222Rn from the hydrothermal vent and 222Rn from the decay of residual 226Ra together. To separate this, measured seawater samples were all preserved in gamma-ray sanitized polyethylene sampling bottles. After more than five half-lives of 222Rn (t1/2 = 3.8 days) had elapsed, we remeasured the samples to determine the activity of residual 226Ra only, as the excess 222Rn had already decayed.
PCA analysis
Principal Component Analysis (PCA) was performed on geochemical variables after z-score normalization. Bartlett’s Test (χ 2 = 117.60, p < 0.001)60 and Kaiser–Meyer–Olkin (KMO) test (0.399)61 confirmed the dataset’s suitability for PCA. Based on the scree plot (Supplementary Fig. S2a), two principal components with eigenvalues > 1 (PC1 = 2.61, PC2 = 1.19) and cumulative explained variance of 74.92% were retained62,63. The PCA biplot (Supplementary Fig. S3a) visualizes both sample scores and variable loadings64. K-means clustering was applied to the first two principal components (Supplementary Fig. S3a). The optimal number of clusters (k = 3) was determined through comprehensive evaluation of multiple validation metrics including the Elbow method (Supplementary Fig. S2b)65, Silhouette coefficient (k = 3: 0.385)66, Calinski-Harabasz index 67, Davies-Bouldin index68, and Gap statistic69. An intelligent recommendation algorithm considering R2 ≥ 50% and WCSS reduction < 25% confirmed k = 3 as optimal. All analyses were performed using Python 3.11 with scikit-learn70 (Pedregosa et al., 2011), pandas, numpy, and plotly.
Data availability
Data is provided within the manuscript or supplementary information files.
References
Soule, S. A. Mid-Ocean Ridge Volcanism. In The Encyclopedia of Volcanoes; Elsevier, 2015; pp 395–403. https://doi.org/10.1016/B978-0-12-385938-9.00021-3.
de Ronde, C. E. J.; Stucker, V. K. Seafloor Hydrothermal Venting at Volcanic Arcs and Backarcs. In The Encyclopedia of Volcanoes; Elsevier, 2015; pp 823–849. https://doi.org/10.1016/B978-0-12-385938-9.00047-X.
Kurz, M. D.; Jenkins, W. J.; Schilling, J. G.; Hart, S. R. Helium Isotopic Variations in the Mantle beneath the Central North Atlantic Ocean. Earth and Planetary Science Letters 1982, 58 (1), 1–14.
German, C. R.; Von Damm, K. L. Hydrothermal Processes. In Treatise on Geochemistry; Elsevier, 2003; pp 181–222. https://doi.org/10.1016/B0-08-043751-6/06109-0.
Elderfield, H.; Schultz, A. Mid-Ocean Ridge Hydrothermal Fluxes and the Chemical Composition of the Ocean. Annu. Rev. Earth Planet. Sci. 1996, 24 (1), 191–224.
Kim, J. et al. Mantle Heterogeneity in the Source Region of Mid-Ocean Ridge Basalts along the Northern Central Indian Ridge (8°S-17°S). Geochem. Geophys. Geosyst. 18(4), 1419–1434. https://doi.org/10.1002/2016GC006673 (2017).
Corliss, J. B.; Dymond, J.; Gordon, L. I.; Edmond, J. M.; Von Herzen, R. P.; Ballard, R. D.; Green, K.; Williams, D.; Bainbridge, A.; Crane, K.; Van Andel, T. H. Submarine Thermal Springs on the Galápagos Rift. Science 1979, 203 (4385), 1073–1083.
Sano, Y.; Fischer, T. P. The Analysis and Interpretation of Noble Gases in Modern Hydrothermal Systems. In The Noble Gases as Geochemical Tracers; Burnard, P., Ed.; Advances in Isotope Geochemistry; Springer Berlin Heidelberg: Berlin, Heidelberg, 2013; pp 249–317. https://doi.org/10.1007/978-3-642-28836-4_10.
Day, J. M. D.; Hilton, D. R. Heterogeneous Mantle-Derived Helium Isotopes in the Canary Islands and Other Ocean Islands. Geology 2021, 49 (2), 120–124.
Ozima, M.; Podosek, F. A. Noble Gas Geochemistry, Second Edition.
Graham, D. W. Noble Gas Isotope Geochemistry of Mid-Ocean Ridge and Ocean Island Basalts: Characterization of Mantle Source Reservoirs. Reviews in Mineralogy and Geochemistry 2002, 47 (1), 247–317.
Kim, D.; Lee, H.; Lee, W.; Kim, J.; Oh, J.; Song, J.-H.; Jung, H.; Stuart, F. M. Helium Isotopes and Olivine Geochemistry of Basalts and Mantle Xenoliths in Jeju Island, South Korea: Evaluation of Role of SCLM on the Cenozoic Intraplate Volcanism in East Asia. Lithos 2021, 390–391, 106123.
Lee, H.; Fischer, T. P.; Muirhead, J. D.; Ebinger, C. J.; Kattenhorn, S. A.; Sharp, Z. D.; Kianji, G.; Takahata, N.; Sano, Y. Incipient Rifting Accompanied by the Release of Subcontinental Lithospheric Mantle Volatiles in the Magadi and Natron Basin, East Africa. Journal of Volcanology and Geothermal Research 2017, 346, 118–133.
Lee, W.; Lee, H.; Kim, D.; Kim, J.; Oh, J.; Song, J.-H.; Kim, C. H.; Park, C. H.; Stuart, F. M. Trace Element and Helium Isotope Geochemistry of the Cenozoic Intraplate Volcanism in the East Sea (Sea of Japan): Implications for Lithosphere-Asthenosphere Interaction. Lithos 2021, 388–389, 106075.
Tardani, D.; Reich, M.; Roulleau, E.; Takahata, N.; Sano, Y.; Pérez-Flores, P.; Sánchez-Alfaro, P.; Cembrano, J.; Arancibia, G. Exploring the Structural Controls on Helium, Nitrogen and Carbon Isotope Signatures in Hydrothermal Fluids along an Intra-Arc Fault System. Geochimica et Cosmochimica Acta 2016, 184, 193–211.
Früh-Green, G. L. et al. Diversity of Magmatism, Hydrothermal Processes and Microbial Interactions at Mid-Ocean Ridges. Nature Reviews Earth and Environment 3(12), 852–871. https://doi.org/10.1038/s43017-022-00364-y (2022).
Kim, J. et al. Discovery of Active Hydrothermal Vent Fields Along the Central Indian Ridge, 8–12°S. Geochem. Geophys. Geosyst. 21(8), 1–21. https://doi.org/10.1029/2020GC009058 (2020).
Rosenberg, N. D.; Lupton, J. E.; Kadko, D.; Collier, R.; Lilley, M. D.; Pak, H. Estimation of Heat and Chemical Fluxes from a Seafloor Hydrothermal Vent Field Using Radon Measurements. Nature 1988, 334 (6183), 604–607.
Rudnicki, M. D. & Elderfield, H. Helium, Radon and Manganese at the TAG and Snakepit Hydrothermal Vent Fields, 26° and 23°N, Mid-Atlantic Ridge. Earth Planet. Sci. Lett. 113(3), 307–321. https://doi.org/10.1016/0012-821X(92)90136-J (1992).
Neff, J. M. Radium Isotopes in the Ocean. In Bioaccumulation in Marine Organisms; Elsevier, 2002; pp 191–201. https://doi.org/10.1016/B978-008043716-3/50012-9.
Pak, S. J. et al. Widespread Tectonic Extension at the Central Indian Ridge between 8°S and 18°S. Gondwana Res. 45, 163–179. https://doi.org/10.1016/j.gr.2016.12.015 (2017).
DeMets, C., Gordon, R. G., Argus, D. F. & Stein, S. Current Plate Motions. Geophys. J. Int. 101(2), 425–478. https://doi.org/10.1111/j.1365-246X.1990.tb06579.x (1990).
Barruol, G. et al. Large-Scale Flow of Indian Ocean Asthenosphere Driven by Réunion Plume. Nat. Geosci. 12(12), 1043–1049. https://doi.org/10.1038/s41561-019-0479-3 (2019).
Gamo, T.; Chiba, H.; Yamanaka, T.; Okudaira, T.; Hashimoto, J.; Tsuchida, S.; Ishibashi, J.; Kataoka, S.; Tsunogai, U.; Okamura, K.; Sano, Y.; Shinjo, R. Chemical Characteristics of Newly Discovered Black Smoker £uids and Associated Hydrothermal Plumes at the Rodriguez Triple Junction, Central Indian Ridge. EPSL 2001.
Kawagucci, S.; Miyazaki, J.; Noguchi, T.; Okamura, K.; Shibuya, T.; Watsuji, T.; Nishizawa, M.; Watanabe, H.; Okino, K.; Takahata, N.; Sano, Y.; Nakamura, K.; Shuto, A.; Abe, M.; Takaki, Y.; Nunoura, T.; Koonjul, M.; Singh, M.; Beedessee, G.; Khishma, M.; Bhoyroo, V.; Bissessur, D.; Kumar, L. S.; Marie, D.; Tamaki, K.; Takai, K. Fluid Chemistry in the Solitaire and Dodo Hydrothermal Fields of the Central Indian Ridge. Geofluids 2016, 16 (5), 988–1005.
Kim, J.; Kim, W.; Choi, S. K.; Oh, J.; Moon, I. Distribution of Diverse Styles of Hydrothermal Vent Fields along the Slow-Spreading Central Indian Ridge between 8° and 16°S. In Goldschmidt2023 abstracts; European Association of Geochemistry: Lyon, France, 2023. https://doi.org/10.7185/gold2023.14303.
Shepherd, K.; Juniper, S. K. ROPOS: Creating a Scientific Tool from an Industrial ROV. Marine Technology Society. Marine Technology Society Journal 1997, 31 (3), 48.
Hilton, D. R., Grönvold, K., Macpherson, C. G. & Castillo, P. R. Extreme 3He/4He Ratios in Northwest Iceland: Constraining the Common Component in Mantle Plumes. Earth Planet. Sci. Lett. 173(1–2), 53–60. https://doi.org/10.1016/S0012-821X(99)00215-0 (1999).
GEBCO Bathymetric Compilation Group 2020. The GEBCO_2020 Grid - a Continuous Terrain Model of the Global Oceans and Land., 2020. https://doi.org/10.5285/A29C5465-B138-234D-E053-6C86ABC040B9.
QGIS Development Team. QGIS Geographic Information System, version 3.40.6. 2025. https://www.qgis.org.
Whiticar, M. J. Carbon and Hydrogen Isotope Systematics of Bacterial Formation and Oxidation of Methane. Chemical Geology 1999, 161 (1–3), 291–314.
Füri, E.; Hilton, D. R.; Murton, B. J.; Hémond, C.; Dyment, J.; Day, J. M. D. Helium Isotope Variations between Réunion Island and the Central Indian Ridge (17°–21°S): New Evidence for Ridge–Hot Spot Interaction. J. Geophys. Res. 2011, 116 (B2), B02207.
Konn, C., Charlou, J. L., Holm, N. G. & Mousis, O. The Production of Methane, Hydrogen, and Organic Compounds in Ultramafic-Hosted Hydrothermal Vents of the Mid-Atlantic Ridge. Astrobiology 15(5), 381–399. https://doi.org/10.1089/ast.2014.1198 (2015).
Konn, C. et al. Extending the Dataset of Fluid Geochemistry of the Menez Gwen, Lucky Strike, Rainbow, TAG and Snake Pit Hydrothermal Vent Fields: Investigation of Temporal Stability and Organic Contribution. Deep Sea Res. Part I 179, 103630. https://doi.org/10.1016/j.dsr.2021.103630 (2022).
Baumberger, T. et al. Fluid Composition of the Sediment-Influenced Loki’s Castle Vent Field at the Ultra-Slow Spreading Arctic Mid-Ocean Ridge. Geochim. Cosmochim. Acta 187, 156–178. https://doi.org/10.1016/j.gca.2016.05.017 (2016).
Konn, C. et al. Organic, Gas, and Element Geochemistry of Hydrothermal Fluids of the Newly Discovered Extensive Hydrothermal Area in the Wallis and Futuna Region (SW Pacific). Geofluids 2018, 1–25. https://doi.org/10.1155/2018/7692839 (2018).
Keir, R. S. A Note on the Fluxes of Abiogenic Methane and Hydrogen from Mid‐ocean Ridges. Geophysical Research Letters 2010, 37 (24). https://doi.org/10.1029/2010gl045362.
Subseafloor Biosphere Linked to Hydrothermal Systems: Taiga Concept; Ishibashi, J., Okino, K., Sunamura, M., Eds.; Springer Open: Tokyo, 2015.
Kawagucci, S.; Okamura, K.; Kiyota, K.; Tsunogai, U.; Sano, Y.; Tamaki, K.; Gamo, T. Methane, Manganese, and Helium‐3 in Newly Discovered Hydrothermal Plumes over the Central Indian Ridge, 18°–20°S. Geochem Geophys Geosyst 2008, 9 (10). https://doi.org/10.1029/2008gc002082.
Sano, Y. & Marty, B. Origin of Carbon in Fumarolic Gas from Island Arcs. Chem. Geol. 119(1–4), 265–274. https://doi.org/10.1016/0009-2541(94)00097-R (1995).
Javoy, M. & Pineau, F. The Volatiles Record of a “Popping” Rock from the Mid-Atlantic Ridge at 14°N: Chemical and Isotopic Composition of Gas Trapped in the Vesicles. Earth Planet. Sci. Lett. 107(3–4), 598–611. https://doi.org/10.1016/0012-821X(91)90104-P (1991).
Kueter, N., Schmidt, M. W., Lilley, M. D. & Bernasconi, S. M. Experimental Determination of Equilibrium CH4–CO2–CO Carbon Isotope Fractionation Factors (300–1200 °C). Earth Planet. Sci. Lett. 506, 64–75. https://doi.org/10.1016/j.epsl.2018.10.021 (2019).
Labidi, J.; McCollom, T. M.; Giunta, T.; Sherwood Lollar, B.; Leavitt, W. D.; Young, E. D. Clumped Isotope Signatures of Abiotic Methane: The Role of the Combinatorial Isotope Effect. JGR Solid Earth 2024, 129 (7), e2023JB028194. https://doi.org/10.1029/2023JB028194.
Klein, F., Grozeva, N. G. & Seewald, J. S. Abiotic Methane Synthesis and Serpentinization in Olivine-Hosted Fluid Inclusions. Proc. Natl. Acad. Sci. U.S.A. 116(36), 17666–17672. https://doi.org/10.1073/pnas.1907871116 (2019).
Proskurowski, G.; Lilley, M. D.; Seewald, J. S.; Früh-Green, G. L.; Olson, E. J.; Lupton, J. E.; Sylva, S. P.; Kelley, D. S. Abiogenic Hydrocarbon Production at Lost City Hydrothermal Field. Science 2008, 319 (5863), 604–607.
Kelley, D. S. & Früh-Green, G. L. Abiogenic Methane in Deep-seated Mid-ocean Ridge Environments: Insights from Stable Isotope Analyses. J. Geophys. Res. 104(B5), 10439–10460. https://doi.org/10.1029/1999JB900058 (1999).
Choi, S. K. A Review on Mineralogical and Geochemical Characteristics of Seafloor Massive Sulfide Deposits in Mid-Ocean Ridge and Volcanic Arc Settings: Water-Rock Interaction and Magmatic Contribution. Econ. Environ. Geol. 55(5), 465–475. https://doi.org/10.9719/EEG.2022.55.5.465 (2022).
Baker, E. T.; Massoth, G. J. Characteristics of Hydrothermal Plumes from Two Vent Fields on the Juan de Fuca Ridge, Northeast Pacific Ocean. Earth and Planetary Science Letters 1987, 85 (1–3), 59–73.
Lupton, J. E.; Baker, E. T.; Massoth, G. J. Variable 3He/Heat Ratios in Submarine Hydrothermal Systems: Evidence from Two Plumes over the Juan de Fuca Ridge. Nature 1989, 337 (6203), 161–164.
Kadko, D. Radioisotopic Studies of Submarine Hydrothermal Vents. Reviews of Geophysics 1996, 34 (3), 349–366.
Sartandel, S. J.; Jha, S. K.; Bara, S. V.; Tripathi, R. M. Assessment of 226Ra and 228Ra Activity Concentration in West Coast of India. J Radioanal Nucl Chem 2014, 300 (2), 873–877.
Konn, C.; Charlou, J. L.; Holm, N. G.; Mousis, O. The Production of Methane, Hydrogen, and Organic Compounds in Ultramafic-Hosted Hydrothermal Vents of the Mid-Atlantic Ridge. Astrobiology 2015, 15 (5), 381–399.
Etiope, G.; Sherwood Lollar, B. ABIOTIC METHANE ON EARTH. Reviews of Geophysics 2013, 51 (2), 276–299.
Lee, J. M. & Kim, G. A Simple and Rapid Method for Analyzing Radon in Coastal and Ground Waters Using a Radon-in-Air Monitor. J. Environ. Radioact. 89(3), 219–228. https://doi.org/10.1016/j.jenvrad.2006.05.006 (2006).
Porcelli, D. P.; Ballentine, C. J.; Wieler, R. Noble Gases: In Geochemistry and Cosmochemistry; Walter de Gruyter GmbH & Co KG, 2018; Vol. 47.
Kipfer, R.; Aeschbach-Hertig, W.; Peeters, F.; Stute, M. Noble Gases in Lakes and Ground Waters. Reviews in Mineralogy and Geochemistry 2002, 47. https://doi.org/10.2138/rmg.2002.47.14.
Reeburgh, W. S. Oceanic Methane Biogeochemistry. Chem. Rev. 107(2), 486–513. https://doi.org/10.1021/cr050362v (2007).
Tsunogai, U., Yoshida, N., Ishibashi, J. & Gamo, T. Carbon Isotopic Distribution of Methane in Deep-Sea Hydrothermal Plume, Myojin Knoll Caldera, Izu-Bonin Arc: Implications for Microbial Methane Oxidation in the Oceans and Applications to Heat Flux Estimation. Geochim. Cosmochim. Acta 64(14), 2439–2452. https://doi.org/10.1016/S0016-7037(00)00374-4 (2000).
Hirota, A.; Tsunogai, U.; Komatsu, D. D.; Nakagawa, F. Simultaneous Determination of δ 15 N and δ 18 O of N 2 O and δ 13 C of CH 4 in Nanomolar Quantities from a Single Water Sample. Rapid Comm Mass Spectrometry 2010, 24 (7), 1085–1092.
Bartlett, M. S. Tests of Significance in Factor Analysis. British journal of psychology 1950.
Kaiser, H. F. A Second Generation Little Jiffy. Psychometrika 35(4), 401–415. https://doi.org/10.1007/BF02291817 (1970).
Cattell, R. B. The Scree Test For The Number Of Factors. Multivar. Behav. Res. 1(2), 245–276. https://doi.org/10.1207/s15327906mbr0102_10 (1966).
Jolliffe, I. T. Principal Component Analysis for Special Types of Data. In Principal Component Analysis; Springer Series in Statistics; Springer-Verlag: New York, 2002; pp 338–372. https://doi.org/10.1007/0-387-22440-8_13.
Gabriel, K. R. The Biplot Graphic Display of Matrices with Application to Principal Component Analysis. Biometrika 58(3), 453–467. https://doi.org/10.1093/biomet/58.3.453 (1971).
Thorndike, R. L. Who Belongs in the Family?. Psychometrika 18(4), 267–276. https://doi.org/10.1007/BF02289263 (1953).
Rousseeuw, P. J. Silhouettes: A Graphical Aid to the Interpretation and Validation of Cluster Analysis. J. Comput. Appl. Math. 20, 53–65. https://doi.org/10.1016/0377-0427(87)90125-7 (1987).
Calinski, T.; Harabasz, J. A Dendrite Method for Cluster Analysis. Comm. in Stats. - Theory & Methods 1974, 3 (1), 1–27. https://doi.org/10.1080/03610927408827101.
Davies, D. L.; Bouldin, D. W. A Cluster Separation Measure. IEEE Trans. Pattern Anal. Mach. Intell. 1979, PAMI-1 (2), 224–227. https://doi.org/10.1109/TPAMI.1979.4766909.
Tibshirani, R., Walther, G. & Hastie, T. Estimating the Number of Clusters in a Data Set Via the Gap Statistic. J. R. Stat. Soc. Ser. B Stat Methodol. 63(2), 411–423. https://doi.org/10.1111/1467-9868.00293 (2001).
Pedregosa, F.; Pedregosa, F.; Varoquaux, G.; Varoquaux, G.; Org, N.; Gramfort, A.; Gramfort, A.; Michel, V.; Michel, V.; Fr, L.; Thirion, B.; Thirion, B.; Grisel, O.; Grisel, O.; Blondel, M.; Prettenhofer, P.; Prettenhofer, P.; Weiss, R.; Dubourg, V.; Dubourg, V.; Vanderplas, J.; Passos, A.; Tp, A.; Cournapeau, D. Scikit-Learn: Machine Learning in Python. MACHINE LEARNING IN PYTHON.
Acknowledgements
We are grateful to the R/V ISABU team, especially Hyunho An and Challin Park, and ROPOS CSSF for their dedicated support during sample recovery and data collection. We also thank the members of the Biogeochemistry Group at Nagoya University for their assistance with sample analyses.
Funding
This research was supported by the Korea Institute of Marine Science and Technology Promotion (KIMST) funded by the Ministry of Oceans and Fisheries (RS-2021-KS211514) and the Korea Institute of Ocean Science and Technology (Grant PEA 0313). This study was also financially sponsored by the National Research Foundation of Korea (NRF) grant funded by the Korean Ministry of Science and ICT (RS-2022-NR070842).
Author information
Authors and Affiliations
Contributions
Heejun Kim: Writing – original draft, Methodology, Investigation. Jungpyo Hong: Wonnyon Kim: Methodology, Investigation, Funding acquisition. Jonguk Kim: Methodology, Investigation, Funding acquisition. Guebuem Kim: Methodology, Investigation. Urumu Tsunogai: Methodology, Investigation. Naoto Takahata: Methodology, Investigation. Yuji Sano: Methodology, Investigation. Hyunwoo Lee: Writing – original draft, review & editing, Methodology, Project administration, Funding acquisition.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Kim, H., Hong, J., Kim, W. et al. Emissions of mantle helium and abiotic methane at newly discovered black smokers along the Central Indian ridge (12°S–16°S). Sci Rep 15, 45736 (2025). https://doi.org/10.1038/s41598-025-32674-8
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-32674-8