Abstract
Malignant ovarian germ cell tumors (MOGCT) and ovarian sex cord stromal tumors (SCST) are rare ovarian tumors. Since 2013, all German federal states are obliged to record data on diagnosis, pathology, therapy, and clinical course of tumor diseases. We gathered data on MOGCT and SCST from thirteen federal cancer registries and the German Childhood Cancer Registry. Each of the participating registries provided aggregated data on MOGCT and SCST with date of diagnosis between 01.01.2016 and 31.12.2021. 629 MOGCT and 872 SCST were included, age peaks at diagnosis for MOGCT and SCST were between 20 and 24 years and between 55 and 59 years, respectively. Of all MOGCT, 23%, 22%, 13% and 12% were malignant teratoma, dysgerminoma, neuroendocrine tumors and yolk sac tumors, respectively. The large majority (94%) of SCST represented malignant granulosa cell tumors. Most tumors were diagnosed in FIGO stage I (66% of MOGCT and 85% of SCST). Surgery was reported in 58% of patients with MOGCT and in 73% of patients with SCST. Chemotherapy was administered to 26% of MOGCT patients and 8% of SCST patients. This study highlights the great potential of cancer registries as high numbers of patients are recorded, however, data are highly dependent on the reporting institutions.
Similar content being viewed by others
Introduction
Malignant ovarian germ cell tumors (MOGCT) and ovarian sex cord stromal tumors (SCST) are rare ovarian neoplasms. They account for around 1.3% (MOGCT) and 1.6% (SCST) of malignant ovarian tumors in Europe1. MOGCT are a heterogeneous group of different tumors, the most common entities include dysgerminomas, immature teratomas and yolk sac tumors2. They mainly affect young women of childbearing age3. SCST mainly include adult and juvenile granulosa cell tumors and Sertoli-Leydig cell tumors and frequently affect middle-aged women4. The treatment of choice for MOGCT includes surgical staging including stage-appropriate surgical resection after surgical staging, and, for tumors of stages higher than FIGO IA, platinum-based chemotherapy (CHT) regimens including etoposide with addition of bleomycin or ifosfamide depending on the risk assessment5. Surgical treatment for SCST is also based on the staging analogous to ovarian cancer. The extent of the surgical resection depends on the extent of the tumor and, in case no hysterectomy is performed, includes a curettage of the corpus uteri to exclude simultaneous endometrial carcinoma. The advantage of adjuvant platinum-containing CHT after complete resection is discussed controversially but may be considered for stages > FIGO IC5,6,7,8. MOGCT and SCST are usually diagnosed in stage I (57% and 64%) and are associated with a good prognosis. Studies from the US National Center for Health Services show that MOGCT have a 5-year survival probability of 94% and 88% for SCST, respectively across all stages, the 5-year cause-specific survival for diagnosis in stage I is 99% (MOGCT) and 98% (SCST)9. However, recurrences are particularly common with SCST (10–64%) and can still occur late, on average 48–57 months after initial diagnosis10.
The Cancer Early Detection and Registry Act (Krebsfrüherkennungs- und -registergesetz; KFRG) (§ 65c, Social Code, Book V) obliges all federal states in Germany to record oncologic diseases in clinical cancer registries11. In addition, epidemiologic cancer registration of pediatric tumors has been in place on a national scale since the early 1980s12.
To study the epidemiologic characteristics of these groups of rare ovarian tumors and the standards of surgical and systemic therapy in Germany we gathered aggregated data of 13 federal cancer registries and the German Childhood Cancer Registry from 2016 – 2021.
Methods
This study was based on pooled data from 13 German cancer registries i.e. clinical cancer registries of Rhineland-Palatinate, Schleswig-Holstein, Hamburg, Bremen, Mecklenburg-Western Pomerania, Brandenburg and Berlin, North Rhine-Westphalia, Lower Saxony, Hesse, Saxony-Anhalt, Saxony, Bavaria and Baden-Württemberg as well as the German Childhood Cancer Registry, representing more than 95% of the German population. Two cancer registries, Saarland and Thuringia did not deliver data for this study, data from Lower Saxony and Saxony-Anhalt were only available from 2018 to 2021 and from Hesse for the years 2016–2020. Each of the participating registries provided aggregated data on MOGCT and SCST for pooled analysis. Inclusion criteria were a primary diagnosis of ICD-10-GM C56 with one of the following ICD-O-3 histo-morphology codes (suppelement Table 1) (MOGCT: 8070/3, 8240/3, 8243/3, 8410/3, 9060/3–9065/3, 9070/3–9072/3, 9080/3- 9086/3, 9090/3, 9100/3, 9105/3 and SCST: 8600/3, 8620/3, 8630/3, 8631/3, 8634/3, 8640/3, 8650/3, 8670/3, 8810/3)13,14 and date of diagnosis between 01.01.2016 and 31.12.2021. Tumors with uncertain behavior were not included. For these patients aggregated data on age at diagnosis in five-year age groups, histo-morphology, FIGO 2014 staging classification15, grading, treatment, residual status, recurrences and vital status were provided. Numbers of 5 or less are not shown (< 6) to avoid de-anonymization. The pooled data was analyzed descriptively using R and RStudio (Version 3.6.3, 2020-02-29; Posit Software, PBC formerly RStudio, PBC, Boston, MA 02210, USA). Analyses were restricted to patients with available information for the variable of interest; we did not impute missing values. All methods were carried out in accordance with relevant guidelines and regulations, i.e. The Cancer Early Detection and Registry Act (Krebsfrüherkennungs- und -registergesetz; KFRG) (§ 65c, Social Code, Book V).
Results
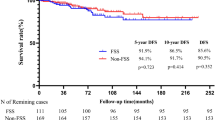
A total of 629 MOGCT and 872 SCST were included in the study. The age peaks of MOGCT and SCST were 20–24 years and 55–59 years, respectively (Fig. 1). With respect to histo-morphology about 23%, 22%, 13% and 12% of GCT were malignant teratoma (9080/3), dysgerminoma (9060/3), neuroendocrine tumors (8240/3) and yolk sac tumors (9071/3), respectively, more than 94% of SCST were malignant granulosa cell tumors (8620/3) and 2% Sertoli-Leydig cell tumors (8631/3) (Table 1).
Age distribution of MOGCT and SCST in 5-year classes: incident cases of 2016–2021 (numbers < 6 not shown).
Initial FIGO stage was available in 290 cases (46%) of MOGCT and 518 cases (59%) of SCST. Of these, 66% of MOGCT were diagnosed in FIGO stage I, 9% in FIGO II, 13% in FIGO III and 12% in FIGO IV. SCST were classified as FIGO I in 85%, FIGO II in 6%, FIGO III in 8% and FIGO IV in 2% of the patients. Tumor grading was available for 285 cases (45%) of MOGCT, and 160 cases (18%) of SCST. Medium grading was most common for MOGCT and low grading for SCST (Table 1).
Surgery was reported in 363 patients with MOGCT (58%) with laparotomy being more frequent than laparoscopy (42 vs. 26%). Diagnosis of SCST was followed by surgery in 633 cases (73%), with laparoscopy and laparotomy being similar in frequency (38 vs. 35%) (Table 2). For both types of tumors adnexectomy was most frequent (81% of MOGCT and 80% of SCST – if unilateral or bilateral adnexectomy was performed cannot be distinguished with the variables reported to the cancer registries) followed by omentectomy (40% and 53% respectively). Hysterectomy was more common in SCST than in MOGCT (51% and 23%). In rare cases (9% of MOGCT and 4% of SCST) ovarian cysts were resected. Lymphadenectomies (LNE) in general were rare, with paraaortal LNE being most frequent (8% of MOGCT and 6% of SCST) (Table 2).
Local R status was available for 76% of both, operated MOGCT and SCST. Local R0 was achieved in 92% (MOGCT) and 96% (SCST) of reported R statuses (Table 3).
CHT was reported for 26% and 8% of MOGCT and SCST, respectively (Table 2). For MOGCT etoposide was the main substance followed by cisplatin. A third substance was added less frequently (bleomycin 40% and ifosfamide 19%). For SCST carboplatin (56%) and cisplatin (13%) were the most frequent substances followed by paclitaxel (47%) (Table 2). Radiotherapy was rarely reported with 2% for MOGCT and less than 1% for SCST (Table 2).
For 8% of patients with MOGCT recurrences were reported and 6% died due to the cancer disease. In case of SCST for 5% of patients’ recurrences were reported and 2% died due to the cancer disease (Table 3).
Discussion
This study compiles data on rare ovarian tumors MOGCT and SCST. It includes data from 13 German cancer registries as well as the German Childhood Cancer Registry for the years 2016–2021, enabling the analysis of a high number of cases (n = 629 and n = 872) for those rare entities.
Data show the median ages at initial diagnosis, patients with MOGCT are considerably younger than patients with SCST (Fig. 1), a finding consistent with previous studies3,4,16. Although the initial FIGO stage is available for only half of the patients (46% of MOGCT and 59% of SCST) the available data indicate that both tumor types are predominantly diagnosed in FIGO stage I (66% and 85% respectively). This appears comparable to SEER data, although interpretation is limited due to the high rate of unknown staging information9.
Surgery was reported for 58% of MOGCT and 73% of SCST. This suggests incomplete reporting, as surgery is the treatment of choice for both tumor types5,8. Some missing surgical data may be explained by advanced, inoperable tumors, but this should only account for a minority of cases (approximately 25% MOGCT and 10% of SCST in our data).
Laparotomy was more frequent than laparoscopy in patients with MOGCT (42 vs. 26%), in patients with SCST both surgical approaches were similarly frequent (38 vs. 35%). While open surgery is generally preferred, minimally invasive staging may be appropriate in selected cases8. According to the German guideline on ovarian cancer, laparotomy is recommended for staging of SCST; however, laparoscopy is possible in early stages and in experienced centers5.
Adnexectomy was the most frequent procedure, however data of the cancer registries cannot distinguish between unilateral and bilateral adnexectomy, limiting insight into fertility sparing surgery rates.
Hysterectomy has been performed more often in patients with SCST than with MOGCT, which aligns with recommendations: hysterectomy is usually avoided in young MOGCT patients, whereas it may be omitted in patients with SCST only at stage IA8. However, cohort analysis showed that patients with SCST and unilateral adnexectomy had a worse 5-year survival than women with bilateral adnexectomy and hysterectomy6. The younger age of MOGCT patients likely explains the lower hysterectomy rates and the higher proportion of fertility-sparing procedures. In SCST, uterine curettage is obligatory, if hysterectomy is not performed, to rule out simultaneous endometrial carcinoma5; however, curettage data were not provided for this study.
Omentectomy and peritonectomy were performed in 40–50% of cases. Studies suggest higher recurrence rates in MOGCT patients without peritoneal biopsies, though without impact on overall survival17. LNE was rarely documented (MOGCT 8%, SCST 6%), consistent with guideline recommendations as LNE is not advised in the absence suspicious findings5,8,18.
R status was reported for 76% of operated patients, of whom 92% (MOGCT) and 96% (SCST) underwent complete (R0) resection.
CHT was administered to 26% of MOGCT patients and 8% of SCST patients. However, this is only comparable to a limited extent as there was a high number of patients with unknown stage. The CHT rate in MOGCT appears low given that the treatment is recommended for stages > FIGO IA. In SCST, there is no consensus regarding the benefit of CHT, although it may be considered for stages > FIGO IC5. This reflects both a potential heterogeneity and uncertainty in real-world practice and the impact of missing data. The substances most frequently used correspond to guideline recommendations: platinum-based combinations with etoposide (with or without bleomycin or ifosfamide) for MOGCT, and platinum combined mainly with paclitaxel for SCST. As expected, radiotherapy played no relevant role in treatment of MOGCT and SCST, although it is unclear whether reporting on radiotherapy was complete.
Recurrence rates were 8% (MOGCT) and 5% (SCST). These rates are slightly lower than reported in previous studies with 17.8%19 and 9%20 for MOGCT but substantially lower for SCST, where recurrence rates of 20%6,21 and up to 44% for granulosa cell tumors have been described22. The low SCST recurrence rate in our dataset is likely explained by the short observation period between 2016 and 2021. Patients may have experienced recurrence after the study period and thus remain unrecorded. Due to this limited follow-up window our study mostly reflects early events.
This study demonstrates the considerable potential of cancer registries to collect large cohorts, even for rare tumor types – numbers that are difficult to achieve even in large multicenter studies23,24,25,26,27,28,29. With these data, we can outline tumor characteristics and treatment patterns for MOGCT and SCST in Germany. However, despite the strength of the large cohort size, the substantial amount of missing information poses limitations and potential bias. Meanwhile cancer registries established several strategies to overcome this limitation30. Other limitations include the inability of pooled data to support more detailed descriptive analyses and the lack of central pathology review, despite the diagnostic complexity of these tumor types.
This is the first study to gather and publish the clinical data of nearly all German federal cancer registries on these entities into one analysis, enabling analysis of an unusually large cohort of these rare tumors. The major advantage of cancer registry data lies in its real-world nature. All inpatient and outpatient physicians are legally required to report diagnostic, treatment and follow-up information. However, the value of these data depends heavily on the quality and completeness of the submitted reports. Studies like ours may help motivate clinicians to improve reporting quality, thereby maximizing the utility of cancer registry data.
Data availability
All data generated or analysed by the cancer registries during this study are included in this published article. All data that support the findings of this study are listed in the references section.
References
Matz, M. et al. The histology of ovarian cancer: worldwide distribution and implications for international survival comparisons (CONCORD-2). Gynecol. Oncol. 144 (2), 405–413 (2017).
Smith, H. O. et al. Incidence and survival rates for female malignant germ cell tumors. Obstet. Gynecol. 107 (5), 1075–1085 (2006).
Zalel, Y. et al. Diagnosis and management of malignant germ cell ovarian tumors in young females. Int. J. Gynaecol. Obstet. 55 (1), 1–10 (1996).
Schultz, K. A. et al. Ovarian sex Cord-Stromal tumors. J. Oncol. Pract. 12 (10), 940–946 (2016).
Leitlinienprogramm Onkologie (Deutsche Krebsgesellschaft DK, AWMF). S3-Leitlinie Diagnostik, Therapie und Nachsorge maligner Ovarialtumoren. Langversion 6.1.01, 2025, AWMF-Registernummer: 032/035OL. https://www.leitlinienprogramm-onkologie.de/fileadmin/user_upload/LL_Ovarialkarzinom_Langversion_6.1.01.pdf , [Download 2025/12/17].
Seagle, B. L., Ann, P., Butler, S. & Shahabi, S. Ovarian granulosa cell tumor: A National cancer database study. Gynecol. Oncol. 146 (2), 285–291 (2017).
Vergote, I. et al. Clinical research in ovarian cancer: consensus recommendations from the gynecologic cancer intergroup. Lancet Oncol. 23 (8), e374–e84 (2022).
Ray-Coquard, I. et al. Non-epithelial ovarian cancer: ESMO clinical practice guidelines for diagnosis, treatment and follow-up. Ann. Oncol. 29 (Suppl 4), iv1–iv18 (2018).
Torre, L. A. et al. Ovarian cancer statistics, 2018. CA Cancer J. Clin. 68 (4), 284–296 (2018).
Levin, G., Zigron, R., Haj-Yahya, R., Matan, L. S. & Rottenstreich, A. Granulosa cell tumor of ovary: A systematic review of recent evidence. Eur. J. Obstet. Gynecol. Reprod. Biol. 225, 57–61 (2018).
Katalinic, A. et al. Population-Based clinical cancer registration in Germany. Cancers (Basel) ;2;15(15),3934. (2023).
Becker, C. et al. Early deaths from childhood cancer in Germany 1980–2016. Cancer Epidemiol. 65, 101669 (2020).
Carcangiu, M. L., Kurman, R. J., Carcangiu, M. L., Herrington, C. S. & WHO Classification of Tumours of Female Reproductive Organs. Lyon: International Agency for Research on Cancer; (2014). Available from: http://VH7QX3XE2P.search.serialssolutions.com/?V=1.0&L=VH7QX3XE2P&S=JCs&C=TC0001705053&T=marc&tab=BOOKS
(BfArM) BfAuM. ICD-O-3 Zweite Revision: BfArM. (2023). Available from: https://www.dimdi.de/dynamic/de/klassifikationen/icd/icd-o-3/icd03rev2html/chapter-m.htm
Wittekind, C. TNM: Klassifikation Maligner Tumoren 336 (Wiley, 2017).
Saani, I. et al. Clinical challenges in the management of malignant ovarian germ cell tumours. Int. J. Environ. Res. Public. Health ;20(12), 6089. (2023).
Mangili, G. et al. The role of staging and adjuvant chemotherapy in stage I malignant ovarian germ cell tumors (MOGTs): the MITO-9 study. Ann. Oncol. 28 (2), 333–338 (2017).
Nasioudis, D., Kanninen, T. T., Holcomb, K., Sisti, G. & Witkin, S. S. Prevalence of lymph node metastasis and prognostic significance of lymphadenectomy in apparent early-stage malignant ovarian sex cord-stromal tumors. Gynecol. Oncol. 145 (2), 243–247 (2017).
Mangili, G. et al. Outcome and risk factors for recurrence in malignant ovarian germ cell tumors: a MITO-9 retrospective study. Int. J. Gynecol. Cancer. 21 (8), 1414–1421 (2011).
Saeed Usmani, A., Yasin, I., Asif, R. B., Kahlid, N. & Syed, A. Incidence and survival rates for female malignant germ cell tumors: an institutional review. Cureus 14 (4), e24497 (2022).
Li, J. et al. Progress in the management of ovarian granulosa cell tumor: A review. Acta Obstet. Gynecol. Scand. 100 (10), 1771–1778 (2021).
Klar, M. et al. Treatment and survival of patients with malignant ovarian sex cord-stromal cell tumours: an analysis of the arbeitsgemeinschaft fur Gynakologische onkologie (AGO) study group CORSETT database. J. Surg. Oncol. 128 (1), 111–118 (2023).
Minig, L. A. O. et al. Oncological outcomes among young women with non-epithelial ovarian cancer: the YOC-Care study (Young ovarian Cancer - Care). Int. J. Gynecol. Cancer. 33 (6), 915–921 (2023).
Lenck, C. et al. The French National network dedicated to rare gynecological cancers diagnosis and management could improve the quality of surgery in daily practice of granulosa cell tumors. A TMRG and GINECO group study. Gynecol. Oncol. 157 (1), 78–84 (2020).
Sun, H. D. et al. A long-term follow-up study of 176 cases with adult-type ovarian granulosa cell tumors. Gynecol. Oncol. 124 (2), 244–249 (2012).
Sehouli, J. et al. Granulosa cell tumor of the ovary: 10 years Follow-up data of 65 patients. Anticancer Res. 24, 1223–1230 (2004).
Wilson, M. K. et al. Stage I granulosa cell tumours: A management conundrum? Results of long-term follow up. Gynecol. Oncol. 138 (2), 285–291 (2015).
Pectasides, D. et al. Adult granulosa cell tumors of the ovary: A clinicopathological study of 34 patients by the Hellenic cooperative oncology group (HeCOG). Anticancer Res. 28, 1421–1428 (2008).
Hasenburg, A. et al. The effect of fertility-sparing surgery on sexuality and global quality of life in women with malignant ovarian germ cell and sex cord stromal tumors: an analysis of the CORSETT database of the AGO study group. Arch. Gynecol. Obstet. 304 (6), 1541–1549 (2021).
Plachky, P. et al. Benefits of the Introduction of a Monitoring Program and Field Crew Service in Cancer Registration in Rhineland-Palatinate Vol. 36 (Oncology Research and Treatment, 2024).
Funding
Open Access funding enabled and organized by Projekt DEAL.
Author information
Authors and Affiliations
Contributions
DJ initiated the project and wrote the manuscript. SB analyzed and summarized the pooled data of all contributing cancer registries and provided the figure. IW and ASP provided pooled datasets for the Clinical Cancer Registry of Saxony-Anhalt. DP provided pooled datasets for the Cancer registry of Saxony. TB and MK provided pooled datasets for the Clinical Cancer Registry of Lower Saxony. SRZ and NB provided pooled datasets for the Regional Centre Würzburg, Bavarian Cancer Registry of the Bavarian Health and Food Safety Authority. AF and SH provided pooled datasets for the Epidemiological Cancer Registry Baden-Württemberg. JK and SL provided pooled datasets for the Bremen Cancer Registry. RP and BB provided pooled datasets for the Cancer Registry of Schleswig-Holstein. CE and HK provided pooled datasets for the Hamburg Cancer Registry. GR and KW provided pooled datasets for Cancer Registry of Mecklenburg-Western Pomerania. KA and SZKW provided pooled datasets for the Hessian Cancer Registry. DR and AvR provided pooled datasets for the Clinical-Epidemiological Cancer Registry Brandenburg-Berlin. FO and AS provided pooled datasets for the Cancer Registry of North Rhine-Westphalia. CR and CB provided pooled datasets for the German Childhood Cancer Registry. CJ provided datasets for the Cancer Registry of Rhineland-Palatinate, was involved in the conception of the data analysis and quality control, organized data of all contributing cancer registries and was a major contributor in writing the manuscript. AH made substantial contribution to the conception and design of the project and interpretation of the data. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Jung, D., Branscheidt, S., Wittenberg, I. et al. Epidemiology and treatment of malignant ovarian germ cell and sex cord stromal tumors in germany: a population-based cancer registry study from 2016 – 2021. Sci Rep 15, 44145 (2025). https://doi.org/10.1038/s41598-025-32998-5
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-32998-5