Abstract
Remnant cholesterol and low-grade inflammation are key contributors to acute coronary syndrome (ACS) in patients with diabetes mellitus. The novel remnant cholesterol inflammatory index (NRCII), combining remnant cholesterol (RC) and the neutrophil-to-lymphocyte ratio (NLR), may enhance risk stratification, but its clinical relevance in diabetic inpatients remains unclear. This multicenter retrospective study included 3664 diabetic inpatients hospitalized between August 2019 and June 2025. Logistic regression, restricted cubic spline (RCS), subgroup, and sensitivity analyses were conducted to assess the association between NRCII and ACS. Incremental predictive value was evaluated using C-statistic, net reclassification improvement (NRI), integrated discrimination improvement (IDI), and likelihood-ratio (LR) tests. ACS occurred in 1056 participants (28.8%). Each 1-unit increase in NRCII was associated with a 21% higher risk of ACS in the fully adjusted model (OR = 1.21, 95% CI 1.13–1.29, P < 0.001). Quartile analyses showed a clear dose–response (P for trend < 0.001), with the highest quartile showing nearly three-fold greater risk (OR = 2.78, 95% CI 1.86–4.04) compared to the lowest. RCS confirmed a significant nonlinear positive association. Results remained robust after excluding patients on lipid-lowering therapy. Subgroup analyses revealed interactions with age and alcohol use. NRCII addition to the clinical model provided the greatest performance gain (C-statistic 0.751; NRI 0.271; IDI 0.020; LR 30.63; all P < 0.001) over RC or NLR alone. Higher NRCII is independently and strongly associated with ACS risk among hospitalized patients with diabetes, offering superior predictive value compared to RC or NLR alone. NRCII may serve as a simple, effective tool for ACS risk stratification in this high-risk population.
Similar content being viewed by others
Introduction
Diabetes mellitus markedly amplifies the incidence and lethality of acute coronary syndrome (ACS), yet substantial numbers of events still occur despite widespread use of guideline-directed lipid-lowering therapy and achievement of target low-density lipoprotein cholesterol (LDL-C) levels1. Contemporary evidence attributes much of this “residual” risk to two convergent processes: the atherogenicity of triglyceride-rich lipoprotein remnants, reflected by remnant cholesterol (RC), and chronic low-grade vascular inflammation, frequently indexed by the neutrophil-to-lymphocyte ratio (NLR)2,3. Elevated RC independently accelerates foam-cell formation, endothelial dysfunction and plaque progression, leading to recurrent coronary events even after intensive statin therapy4,5. In parallel, a high NLR predicts culprit-lesion complexity, infarct size and long-term mortality across the ACS spectrum, underscoring the prognostic weight of systemic innate-immune activation6,7.
Mechanistic studies indicate that metabolic and inflammatory pathways amplify each other: remnant particles enhance NLRP3-inflammasome signaling and neutrophil extracellular-trap release, whereas activated neutrophils impair lipoprotein clearance and promote oxidative modification of remnants, together fostering plaque instability8,9. Such synergy is increasingly recognized clinically; individuals with concomitantly high RC and inflammatory biomarkers experience the greatest residual event burden after ACS10,11. To capture this dual pathway more efficiently, composite indices have been proposed. Recent population studies have demonstrated that integrating RC with inflammatory markers, such as the remnant cholesterol inflammatory index (RCII), can better stratify mortality and stroke risk than either component alone12,13. However, most prior indices rely on C-reactive protein (CRP) or high-sensitivity CRP (hs-CRP), which are not routinely measured in many cardiology settings. In contrast, NLR is a simple, widely available parameter derived from routine blood counts, making the novel remnant cholesterol inflammatory index (NRCII)—which combines RC and NLR—a practical and accessible tool. To date, no studies have examined whether this composite can refine ACS risk prediction specifically in hospitalized patients with diabetes, a population at particularly high residual risk.
Accordingly, we evaluated the NRCII, defined as the product of RC and NLR, in a large multicenter cohort of hospitalized patients with diabetes. We aimed to determine the independent association between NRCII and ACS, its incremental discriminatory value over traditional metabolic and inflammatory markers, and the consistency of these relationships across clinically relevant subgroups.
Methods
Study design
This was a multicenter, retrospective observational study reported in accordance with the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines. Data were extracted from the integrated information platforms of The First Affiliated Hospital of Dali University (March 2022 to June 2025) and The Second People’s Hospital of Baoshan City Yunnan Province (August 2019 to June 2025), incorporating electronic medical records (EMR) and laboratory information systems (LIS). The study cohort included all adult patients with diabetes mellitus who were hospitalized in the Departments of Cardiology at these two hospitals during the study period. All data were fully de-identified before analysis. The study protocol was approved by the Ethics Committee of The Second People’s Hospital of Baoshan City Yunnan Province (Approval No. 2025-025-01), conducted in accordance with the Declaration of Helsinki, and exempted from written informed consent due to its retrospective nature.
Study population
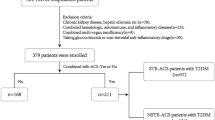
A total of 5108 hospitalized patients with a discharge diagnosis of diabetes mellitus were initially screened, including 3322 patients from The First Affiliated Hospital of Dali University and 1786 from The Second People’s Hospital of Baoshan City Yunnan Province. Patients were excluded if they (1) lacked sufficient information to calculate the NRCII, (2) presented with active infection or were receiving antibiotic therapy at admission, or (3) had a history of acute myocardial infarction (AMI) or percutaneous coronary intervention (PCI). After these exclusions, 3664 participants remained for the final analysis, of whom 2608 were classified as non-ACS and 1056 were diagnosed with ACS (Fig. 1).
Flowchart of participant selection.
Calculation of the novel remnant cholesterol inflammatory index (NRCII)
The NRCII was developed to integrate RC–related metabolic risk with systemic inflammation. RC was calculated as:
Total cholesterol (TC), high-density lipoprotein cholesterol (HDL-C), and LDL-C were measured from the first fasting blood sample obtained at admission. Systemic inflammation was assessed using the NLR, defined as the absolute neutrophil count divided by the absolute lymphocyte count, both derived from the first blood test at admission (units: ×109/L). The NRCII was then calculated as the product of RC and NLR:
NRCII was analyzed both as a continuous variable and as a categorical variable according to its quartile distribution in the study population: Q1 (NRCII ≤ 1.389), Q2 (1.389 < NRCII ≤ 2.182), Q3 (2.182 < NRCII ≤ 3.6), and Q4 (NRCII > 3.6), with Q1 serving as the reference category.
Data collection
Demographic and clinical information were extracted by trained physicians from the EMR systems of both hospitals, including sex, age, height, weight, cardiovascular risk factors, medical history, vital signs, routine laboratory results, and diagnostic codes. Fasting venous blood was obtained on the day of admission or the following morning and analyzed in the respective hospital laboratories. The ACS, including unstable angina and AMI, was identified directly from the structured discharge diagnoses in the EMR. All diagnoses were made by board-certified cardiologists during hospitalization based on contemporaneous clinical guidelines (symptoms, ECG findings, and cardiac biomarkers); no independent adjudication was performed for this retrospective analysis. Smoking status was self-reported and categorized as never (< 100 cigarettes lifetime), former (≥ 100 cigarettes lifetime but currently non-smoker), or current (≥ 100 cigarettes lifetime and smoking at present). Alcohol consumption was classified as current alcohol user or non-current alcohol user (defined as < 12 drinks lifetime or none in the past year). Lipid-lowering therapy was defined as the documented use of any lipid-lowering medication prior to hospital admission. Body mass index (BMI) was calculated as weight divided by height squared (kg/m2). Because non-statin lipid-lowering therapy (such as fibrates or ezetimibe) was prescribed in only a small proportion of participants (n = 47, < 3%), lipid-lowering therapy was treated as a binary variable (any vs. none) to maintain model stability and avoid sparse-data bias.
Statistical analysis
All participants were categorized into ACS and non-ACS groups. Continuous variables were expressed as means ± standard deviations (SD) and compared using independent sample t-tests, while categorical variables were presented as counts (percentages) and compared using chi-square tests. Logistic regression models were applied to evaluate the association between NRCII and ACS in patients with diabetes, reporting odds ratios (OR) and 95% confidence intervals (CI). Three models were constructed: Crude model, unadjusted; Model 1, adjusted for age, sex, race, education level, and marital status; and Model 2, further adjusted for smoking status, alcohol consumption, BMI, hypertension, triglycerides (TG), serum creatinine (SCR), uric acid (UA), and lipid-lowering therapy. Restricted cubic spline (RCS) regression with knots at the 10th, 50th, and 90th percentiles of NRCII was used to explore potential non-linear associations, adjusting for all covariates in Model 2. Incremental predictive value was assessed using C-statistics, net reclassification improvement (NRI), integrated discrimination improvement (IDI), and likelihood ratio (LR) tests. Subgroup analyses were conducted by sex, age (< 60 or ≥ 60 years), race, smoking status, alcohol consumption, hypertension status, and BMI category, and interactions were formally tested. Missing covariates with rates < 5% were imputed using predictive mean matching, including all variables in the imputation models. All statistical analyses were performed using R software (version 4.3.2), with two-sided P values < 0.05 considered statistically significant.
Results
Baseline characteristics of the study population
A total of 3664 hospitalized patients with diabetes mellitus were included in the analysis, comprising 1056 patients with ACS and 2608 without ACS. Among the 1056 patients with ACS, 304 (28.8%) had unstable angina, and 752 (71.2%) had AMI. Compared with the non-ACS group, patients with ACS were older and more likely to be male. No significant differences were observed in race and marital status between groups. The ACS group had a lower HDL-C, but higher neutrophil count, BMI, RC, NLR, and NRCII. Smoking status differed significantly, with current smoking more prevalent in the ACS group. A higher prevalence of hypertension was observed in the non-ACS group. There was no significant difference in the proportion receiving lipid-lowering therapy before admission (P = 0.06). Other laboratory parameters including TC, TG, LDL-C, SCR, BUN, UA, and lymphocyte count showed no significant differences between groups (Table 1).
Association between NRCII and risk of ACS
We examined the association between the NRCII and the risk of ACS using multivariable logistic regression (Table 2). When treated as a continuous variable, each 1-unit increase in NRCII was associated with a significantly higher risk of ACS in the crude model (OR = 1.22, 95% CI 1.17–1.26, P < 0.001), Model 1 (OR = 1.21, 95% CI 1.17–1.26, P < 0.001), and fully adjusted Model 2 (OR = 1.21, 95% CI 1.13–1.29, P < 0.001). Excluding marital status and uric acid from Model 2 yielded virtually identical estimates (less than a 10% change in adjusted OR), confirming that inclusion of these covariates did not materially affect the observed association between NRCII and ACS. When NRCII was categorized into quartiles, there was a clear dose–response relationship (P for trend < 0.001). Compared with participants in Q1, those in Q2 (OR = 1.38, 95% CI 0.99–1.94, P = 0.06), Q3 (OR = 2.22, 95% CI 1.58–3.13, P < 0.001), and Q4 (OR = 2.78, 95% CI 1.86–4.04, P < 0.001) showed progressively higher risks of ACS in Model 2.
The RCS analysis (Fig. 2) confirmed a significant positive association between NRCII and the risk of ACS (P overall < 0.001) and revealed evidence of a non-linear dose–response relationship (P for non-linearity = 0.0216). The curve demonstrated that ACS risk increased steeply at lower NRCII levels, followed by a more gradual rise at higher levels.
Restricted cubic spline (3 knots at the 10th, 50th, and 90th percentiles of NRCII) depicting the adjusted association between NRCII and ACS (Model-2 adjusted for age, sex, race, education level, marital status, smoking, alcohol, BMI, hypertension, TG, SCR, UA, and lipid-lowering therapy). The green line indicates adjusted log-odds; the shaded area shows the 95% CI. The red dot marks the median NRCII (reference point).
Incremental predictive value of NRCII for ACS
Table 3 summarizes the incremental discrimination achieved by adding RC, NLR, and the composite NRCII to the basic model for ACS risk prediction. The C-statistic of the basic model was 0.739 (95% CI 0.714–0.764). Inclusion of RC did not change the C-statistic (0.739, 95% CI 0.714–0.764, P < 0.001), while NLR increased it to 0.750 (95% CI 0.725–0.775, P < 0.001), and NRCII yielded the highest value at 0.751 (95% CI 0.727–0.776, P < 0.001). The NRI showed a small and non-significant improvement with RC (0.027, 95% CI − 0.081–0.136, P = 0.619), but significant increases with NLR (0.238, 95% CI 0.130–0.345, P < 0.001) and NRCII (0.271, 95% CI 0.163–0.378, P < 0.001). Similarly, the IDI was negligible for RC (0.001, 95% CI − 0.001–0.002, P = 0.977), but significant for NLR (0.017, 95% CI 0.010–0.024, P < 0.001) and NRCII (0.020, 95% CI 0.012–0.027, P < 0.001). The LR test results confirmed this pattern, showing no significant improvement with RC (LR = 0.126, P = 0.723), while NLR (LR = 27.909, P < 0.001) and NRCII (LR = 30.629, P < 0.001) significantly enhanced model fit.
Sensitivity analyses and subgroup effects
After excluding 846 participants receiving prior lipid-lowering therapy, the association between NRCII and ACS remained robust (Table 4). In the fully adjusted model, each one-unit increase in NRCII was associated with a 21% higher risk of ACS (OR = 1.21, 95% CI 1.12–1.31, P < 0.001). When analyzed as quartiles, a clear dose–response pattern persisted (P for trend < 0.001): compared with Q1, the odds of ACS increased to 1.38 (95% CI 0.93–2.04, P = 0.11) in Q2, 2.26 (95% CI 1.52–3.37, P < 0.001) in Q3, and 2.85 (95% CI 1.86–4.37, P < 0.001) in Q4.
Subgroup analyses (Fig. 3) showed that the association between NRCII and ACS was generally consistent across subgroups defined by sex, race, smoking status, drinking status, hypertension, and BMI, with no significant interactions (all P for interaction > 0.05), except for age (P for interaction = 0.015) and drinking status (P for interaction = 0.040). The association was slightly stronger in participants aged < 60 years (OR = 1.279, 95% CI 1.203–1.363) compared to those ≥ 60 years (OR = 1.154, 95% CI 1.091–1.220), and in current alcohol users (OR = 1.309, 95% CI 1.201–1.432) compared to non-current users (OR = 1.181, 95% CI 1.127–1.238). No significant association was observed in former smokers (OR = 1.107, 95% CI 0.964–1.270, P = 0.145) or participants with BMI ≥ 30 kg/m2 (OR = 1.018, 95% CI 0.640–1.444, P = 0.928), suggesting potential heterogeneity in these groups.
Subgroup analyses of the association between NRCII and risk of ACS with tests for interaction.
Discussion
Principal findings
In this multicenter retrospective study of hospitalized patients with diabetes mellitus, we found that higher levels of the NRCII were significantly associated with an increased risk of ACS. This association persisted after adjusting for multiple potential confounders, including demographic factors, cardiovascular risk factors, and lipid-lowering therapy. When analyzed as quartiles, NRCII demonstrated a clear dose–response relationship with ACS risk, and RCS analysis confirmed a significant nonlinear positive association. Furthermore, NRCII provided greater incremental predictive value for ACS compared with its individual components—RC and NLR—as evidenced by improvements in C-statistics, NRI, IDI, and LR tests. Sensitivity analyses excluding patients receiving prior lipid-lowering therapy yielded consistent results. Subgroup analyses revealed that the association was generally robust across various clinical strata, though modest interactions with age and alcohol consumption were observed.
Comparison with previous studies
Our findings further demonstrate that the combined effects of RC metabolism and low-grade systemic inflammation significantly contribute to cardiovascular risk in patients with diabetes mellitus. Earlier cohort and registry data have shown that elevated RC is independently associated with incident ACS and adverse prognosis even after LDL-C goal attainment8,14,15. Experimental work further suggests that remnant-rich lipoproteins possess pro-inflammatory properties that amplify endothelial dysfunction and plaque vulnerability16,17. Parallel clinical studies have established the NLR as a robust, easily obtained marker of systemic inflammation that predicts CAD severity, STEMI mortality and long-term events in diabetic and non-diabetic settings18,19,20. Only recently have investigators begun to explore the interaction between remnant cholesterol and residual inflammatory risk. Contemporary analyses in statin-treated ACS cohorts indicate that patients with concordant elevations in both pathways experience the highest rate of major adverse cardiovascular events, whereas isolated elevation of either component confers a more modest risk increment8,21. Our study corroborates these observations and advances the field by demonstrating that a simple multiplicative composite—NRCII—captures this dual pathway more effectively than either RC or NLR alone. The incremental gains we observed in discrimination (C-statistic), reclassification (NRI/IDI) and global model fit (LR test) parallel those reported for other integrated biomarkers that combine metabolic and inflammatory signals22. Importantly, the nonlinear dose-response curve we observed suggests that the impact of NRCII is most pronounced at lower to mid-range values, a pattern consistent with prior spline analyses of isolated RC and NLR that show steeper risk gradients at the lower ends of their distributions15,20. Collectively, these concordant findings across diverse populations reinforce the concept that simultaneous assessment of RC burden and systemic inflammation yields a more complete picture of residual cardiovascular risk than evaluating either pathway in isolation.
Possible biological mechanisms
Multiple pathophysiological pathways plausibly link a high NRCII to excess coronary risk in diabetes. First, remnant-rich lipoproteins penetrate the arterial intima more readily than LDL particles and are avidly taken up by macrophages, accelerating foam-cell formation and promoting lipid-laden, inflammation-prone plaques23,24. Within endothelial cells, RC stimulates redox-sensitive signaling that up-regulates ICAM-1, VCAM-1 and tissue factor, thereby enhancing leucocyte adhesion, thrombogenicity and endothelial apoptosis25,26. Cholesterol crystals and oxidized remnants also activate the NLRP3 inflammasome, amplifying interleukin-1β/IL-18 release and sustaining vascular inflammation27,28. Second, a raised NLR reflects a shift towards innate immune activation. Neutrophils secrete proteases, myeloperoxidase and reactive oxygen species that destabilize the fibrous cap and favor plaque rupture, whereas relative lymphocytopenia denotes impaired adaptive immune surveillance6,29. Experimental work further shows crosstalk between lipoprotein metabolism and inflammation: oxidized or remnant particles can trigger neutrophil extracellular trap formation and promote PCSK9-driven NLRP3 activation, bridging metabolic and immune pathways30,31. Under hyperglycemic conditions, these processes are accentuated through increased foam-cell lipid loading, impaired cholesterol efflux and heightened oxidative stress, providing a biologically plausible explanation for the steeper, non-linear rise in ACS risk observed at higher NRCII levels in our diabetic cohort21.
Interpreting the incremental-value findings and subgroup heterogeneity
Our incremental-discrimination analysis showed that adding RC to the basic model did not improve C-statistic, NRI, IDI or LR, whereas the composite NRCII markedly outperformed its inflammatory component alone. Biologically, RC and LDL-C share metabolic pathways and are often tightly co-modulated by statin or ezetimibe therapy, so the residual between-patient variance in RC after standard lipid-lowering is modest; this limits its ability to shift individual risk rankings and thus weakens reclassification metrics. In contrast, low-grade inflammation remains largely untreated, and NLR retains substantial inter-individual dispersion that translates into meaningful model gains32,33. NRCII magnifies predictive information because it captures the synergistic vascular damage that occurs when remnant-rich lipoproteins infiltrate the intima and simultaneously prime innate immune activation—a dual pathway repeatedly shown to accelerate plaque progression and rupture34,35. Experimental data demonstrate that RC can trigger NLRP3-inflammasome signaling and neutrophil extracellular-trap formation, processes already indexed by a high NLR; multiplying the two components therefore concentrates subjects in whom both metabolic and inflammatory drives are active, yielding steeper risk gradients and larger net reclassification gains than NLR alone12,36.
Subgroup analyses revealed broadly consistent NRCII–ACS associations, yet effect sizes were attenuated or statistically non-significant in several strata and modest interactions emerged for age and alcohol consumption. These patterns are explicable on both statistical and biological grounds. First, some strata (e.g., former smokers, BMI ≥ 30 kg m2) contained fewer events, reducing power to detect modest ORs despite similar point estimates—an issue commonly reported in inflammatory-marker studies where precision falls in low-event subgroups32. Second, age-related immunosenescence and glycation of apolipoprotein remnants may blunt the pro-inflammatory synergy captured by NRCII in older individuals, whereas younger patients display stronger neutrophil activation and triglyceride-rich lipoprotein flux, amplifying the composite risk signal34,37. Third, ethanol induces lipoprotein remodeling and modulates peripheral neutrophil counts; the stronger association observed in current drinkers aligns with data linking alcohol-driven oxidative stress to heightened RC-mediated endothelial injury35. Finally, the absence of significance in non-Han ethnicity or extremely obese strata may reflect unmeasured cultural diets, genetic variants in apolipoprotein metabolism, or dilution by competing risks, all warranting validation in larger multi-ethnic cohorts. Together, these observations reinforce that NRCII captures an integrated lipid-inflammation axis that is broadly operative yet modulated by age, metabolic milieu and lifestyle factors.
Strengths and limitations
The present study benefits from a large, contemporary, dual-center cohort of > 3,600 hospitalized people with diabetes, yielding ample statistical power and enhancing generalizability within this high-risk population. Because NRCII relies on routine fasting lipid panels and differential blood counts, our findings are readily translatable to everyday practice and complement recent work highlighting residual inflammatory-cholesterol interplay in CAD38. Methodologically, we applied comprehensive adjustment, multiple imputation and a suite of modern performance metrics (C-statistic, NRI, IDI, LR) recommended for biomarker evaluation2,39. Nevertheless, several limitations merit attention. First, the retrospective design precludes causal inference and relies on discharge diagnoses, introducing potential misclassification40. Second, single baseline measurements may not capture temporal fluctuations in RC or NLR; dynamic changes could modify risk. Third, the cohort was drawn from tertiary hospitals in southwest China, so external validation in other ethnicities and out-patient settings is still required. Unmeasured confounders—e.g., insulin resistance, dietary patterns—may persist despite extensive covariate adjustment. Finally, we acknowledge that the degree of systemic inflammatory activation differs between unstable angina and acute myocardial infarction, which may influence NRCII levels and the strength of their associations with ACS. Although our cohort allowed us to describe the distribution of these subtypes, the study was not designed for detailed subtype-specific analyses, so this potential heterogeneity should be considered when interpreting the findings.
Clinical implications and future research
Our data indicate that NRCII integrates lipid-rich remnant particles with systemic inflammation to deliver superior risk stratification over RC or NLR alone, offering clinicians a low-cost tool to pinpoint diabetic in-patients who might benefit from intensified “lower-RC plus anti-inflammatory” therapy. This supports emerging evidence that concurrent elevation of remnants and inflammatory markers drives residual events even after LDL-C optimization8,41. Future investigations should first undertake external validation of NRCII in community-based and multi-ethnic cohorts to confirm its generalizability beyond the current inpatient setting. Longitudinal studies that serially measure NRCII are also warranted to determine whether dynamic changes in the index can track treatment response and provide incremental prognostic value over established risk scores such as GRACE. In addition, mechanistic and clinical trials that target both remnant cholesterol and residual inflammatory pathways are needed to establish whether pharmacologically lowering NRCII results in fewer ACS events42,43. Finally, formal cost-effectiveness evaluations and incorporation of NRCII into electronic clinical decision-support systems will be essential steps toward its routine adoption in cardiovascular risk management.
Conclusions
In summary, the NRCII is an independent and potent predictor of ACS in hospitalized patients with diabetes. NRCII consistently outperformed RC and the NLR in risk-prediction metrics, with its prognostic value remaining stable across key subgroups and after exclusion of lipid-lowering therapy users. These results support NRCII as a practical, readily available marker for refining ACS risk stratification beyond conventional lipid or inflammatory parameters.
Data availability
The datasets generated and/or analyzed during the current study are not publicly available because of institutional data-use agreements and patient-confidentiality regulations, but they can be obtained from the corresponding author on reasonable request.
Abbreviations
- ACS:
-
Acute coronary syndrome
- AMI:
-
Acute myocardial infarction
- BMI:
-
Body-mass index
- BUN:
-
Blood urea nitrogen
- CI:
-
Confidence interval
- DM:
-
Diabetes mellitus
- ECG:
-
Electrocardiogram
- EMR:
-
Electronic medical record
- HDL-C:
-
High-density lipoprotein cholesterol
- IDI:
-
Integrated discrimination improvement
- LDL-C:
-
Low-density lipoprotein cholesterol
- LIS:
-
Laboratory information system
- LR:
-
Likelihood ratio
- NLR:
-
Neutrophil-to-lymphocyte ratio
- NRCII:
-
Novel remnant-cholesterol inflammatory index
- NRI:
-
Net reclassification improvement
- OR:
-
Odds ratio
- PCI:
-
Percutaneous coronary intervention
- RC:
-
Remnant cholesterol
- RCS:
-
Restricted cubic spline
- SCR:
-
Serum creatinine
- SD:
-
Standard deviation
- TC:
-
Total cholesterol
- TG:
-
Triglycerides
- UA:
-
Uric acid
References
Vijayaraghavan, K., Baum, S., Desai, N. R. & Voyce, S. J. Intermediate and long-term residual cardiovascular risk in patients with established cardiovascular disease treated with Statins. Front. Cardiovasc. Med. 10, 1308173 (2023).
Li, X., Li, Z. F. & Wu, N. Q. Remnant cholesterol and residual risk of atherosclerotic cardiovascular disease. Rev. Cardiovasc. Med. 26, 25985 (2025).
Ohmura, H. Contribution of remnant cholesterol to coronary atherosclerosis. J. Atheroscler Thromb. 29, 1706–1708 (2022).
Lee, J-H. et al. Remnant cholesterol as a residual risk in atherosclerotic cardiovascular disease patients under statin-based lipid-lowering therapy: A post hoc analysis of the RACING trial. J. Clin. Lipidol. 18, e905–e914 (2024).
Hao, Q-Y. et al. Remnant cholesterol and the risk of coronary artery calcium progression: insights from the CARDIA and MESA study. Circ. Cardiovasc. Imaging. 15, e014116 (2022).
Shahsanaei, F., Abbaszadeh, S., Behrooj, S., Rahimi Petrudi, N. & Ramezani, B. The value of neutrophil-to-lymphocyte ratio in predicting severity of coronary involvement and long-term outcome of percutaneous coronary intervention in patients with acute coronary syndrome: a systematic review and meta-analysis. Egypt. Heart J. 76, 39 (2024).
Kashyap, A., Ahmed, J. & Thakkar, M. Neutrophil-to-lymphocyte ratio as a predictive marker for acute coronary syndrome. MGM J. Med. Sci. 10, 241–245 (2023).
Liao, J. et al. The residual risk of inflammation and remnant cholesterol in acute coronary syndrome patients on Statin treatment undergoing percutaneous coronary intervention. Lipids Health Dis. 23, 172 (2024).
Wulff, A. B. & Nordestgaard, B. G. Residual cardiovascular risk beyond low-density lipoprotein cholesterol: inflammation, remnant cholesterol, and lipoprotein(a). Eur. Heart J. (2025).
Joshi, A., Singh, H. & Kalra, S. The acute coronary syndrome risk in medically managed subjects with type 2 diabetes Mellitus - Is the ASCVD risk score failing here? J. ASEAN Fed. Endocr. Soc. 39, 31–36 (2024).
Toso, A., Leoncini, M., Maioli, M., Villani, S. & Bellandi, F. Biomarkers of residual risk and all-cause mortality after acute coronary syndrome. Am. J. Prev. Cardiol. 21, 100934 (2025).
Wang, Y. et al. Remnant cholesterol inflammatory index and its association with all-cause and cause-specific mortality in middle-aged and elderly populations: evidence from US and Chinese National population surveys. Lipids Health Dis. 24, 155 (2025).
Chen, J. et al. Predictive value of remnant cholesterol inflammatory index for stroke risk: evidence from the China health and retirement longitudinal study. J. Adv. Res. (2024).
Bay, B. et al. Residual cholesterol and inflammatory risk in statin-treated patients undergoing percutaneous coronary intervention†. Eur. Heart J. (2025).
Liu, H. H. et al. Synergistic effect of the commonest residual risk factors, remnant cholesterol, lipoprotein(a), and inflammation, on prognosis of statin-treated patients with chronic coronary syndrome. J. Transl Med. 20, 243 (2022).
Henein, M. Y., Vancheri, S., Longo, G. & Vancheri, F. The role of inflammation in cardiovascular disease. Int. J. Mol. Sci. 23 (2022).
Alyaydin, E. et al. Cardiac allograft vasculopathy in a long-term follow-up after heart transplantation: role of remnant cholesterol in residual inflammation. Cardiol. J. 29, 782–790 (2022).
Zahorec, R. Neutrophil-to-lymphocyte ratio, past, present and future perspectives. Bratisl Lek Listy. 122, 474–488 (2021).
Ahmad, N. et al. Correlation between the Neutrophil-to-Lymphocyte ratio and the severity of coronary artery disease in patients with myocardial infarction. Cureus 16, e69061 (2024).
Sharma, D. J., Sr., Nath, H. J., Batta, A. & Goala, A. K. Neutrophil-to-Lymphocyte ratio (NLR) useful as a Cost-Effective preliminary prognostic marker in ST-Elevation myocardial infarction (STEMI): an observational study from a tertiary care hospital in Northeast India. Cureus 15, e36885 (2023).
Ghafoury, R., Malek, M., Ismail-Beigi, F. & Khamseh, M. E. Role of residual inflammation as a risk factor across Cardiovascular-Kidney-Metabolic (CKM) syndrome: unpacking the burden in people with type 2 diabetes. Diabetes Ther. 16, 1341–1365 (2025).
Bekbossynova, M. et al. Beyond cholesterol: emerging risk factors in atherosclerosis. J. Clin. Med. 14 (2025).
Cervantes, J. & Kanter, J. E. Monocyte and macrophage foam cells in diabetes-accelerated atherosclerosis. Front. Cardiovasc. Med. 10, 1213177 (2023).
Lee, J. & Choi, J. H. Deciphering macrophage phenotypes upon lipid uptake and atherosclerosis. Immune Netw. 20, e22 (2020).
Song, Y. et al. Remnant cholesterol is independently associated with an increased risk of peripheral artery disease in type 2 diabetic patients. Front. Endocrinol. (Lausanne). 14, 1111152 (2023).
Du, X. et al. Remnant cholesterol has an important impact on increased carotid intima-media thickness in non-diabetic individuals. Int. J. Cardiovasc. Imaging. 39, 2487–2496 (2023).
Elías-López, D., Kobylecki, C. J., Vedel-Krogh, S., Doi, T. & Nordestgaard, B. G. Association of low-grade inflammation and elevated remnant cholesterol with risk of ASCVD and mortality in impaired renal function. Atherosclerosis 406, 119241 (2025).
Tall, A. R. & Bornfeldt, K. E. Inflammasomes and atherosclerosis: a mixed picture. Circul. Res. 132, 1505–1520 (2023).
Li, X., Liu, M. & Wang, G. The neutrophil–lymphocyte ratio is associated with all-cause and cardiovascular mortality in cardiovascular patients. Sci. Rep. 14, 26692 (2024).
Wang, Y. et al. Interactions between PCSK9 and NLRP3 inflammasome signaling in atherosclerosis. Front. Immunol. 14, 1126823 (2023).
Wu, Y. et al. Association of remnant cholesterol with hypertension, type 2 diabetes, and their coexistence: the mediating role of inflammation-related indicators. Lipids Health Dis. 22, 158 (2023).
Fan, W. et al. The prognostic value of hematologic inflammatory markers in patients with acute coronary syndrome undergoing percutaneous coronary intervention. Clin. Appl. Thromb. Hemost. 28, 10760296221146183 (2022).
Kim, Y. H., Her, A. Y., Garg, S. & Shin, E. S. Incremental predictive value of the combined use of the neutrophil-to-lymphocyte ratio and systolic blood pressure difference after successful drug-eluting stent implantation. Cardiol. J. 30, 91–104 (2023).
Zhang, Z. et al. A synergistic effect of remnant cholesterol and C-Reactive protein on predicting the severity of coronary artery disease. J. Inflamm. Res. 17, 11291–11303 (2024).
Xu, D. et al. Association of obstructive sleep apnea with cardiovascular events in acute coronary syndrome patients with dual risk of remnant cholesterol and low-grade inflammation: a post-hoc analysis of the OSA-ACS study. Sleep. Breath. 29, 119 (2025).
Inoue, N., Morikawa, S. & Murohara, T. Comprehensive assessment of lipid markers in cardiovascular events prediction. Int. Heart J. 65, 792–799 (2024).
Xiong, C. et al. Prognostic significance of IL-18 in acute coronary syndrome patients. Clin. Cardiol. 47, e24229 (2024).
Cordero, A. et al. Remnant cholesterol in patients admitted for acute coronary syndromes. Eur. J. Prev. Cardiol. 30, 340–348 (2023).
Song, S., Chen, L., Yu, R. & Zhu, J. Neutrophil-to-lymphocyte ratio as a predictor of all-cause and cardiovascular mortality in coronary heart disease and hypertensive patients: a retrospective cohort study. Front. Endocrinol. (Lausanne). 15, 1442165 (2024).
Pan, D., Xu, L., Zhang, L. X., Shi, D. Z. & Guo, M. Associations between remnant cholesterol levels and mortality in patients with diabetes. World J. Diabetes. 15, 712–723 (2024).
Sebastian-Valles, F. et al. Impact of remnant cholesterol and triglycerides on diabetes foot and disease in type 1 diabetes: A propensity score-matched case-control study. J. Diabetes Complicat. 39, 109082 (2025).
Huh, J. H. et al. Remnant cholesterol and the risk of cardiovascular disease in type 2 diabetes: a nationwide longitudinal cohort study. Cardiovasc. Diabetol. 21, 228 (2022).
Ul Hussain, H. et al. Neutrophil to lymphocyte ratio as a prognostic marker for cardiovascular outcomes in patients with ST-segment elevation myocardial infarction after percutaneous coronary intervention: A systematic review and meta-analysis. Medicine 103, e38692 (2024).
Acknowledgements
The authors thank the physicians, nurses and administrative staff of the Departments of Cardiology at both The First Affiliated Hospital of Dali University and The Second People’s Hospital of Baoshan City for their invaluable assistance with data acquisition and patient care. We are also grateful to the Information Technology teams of both institutions for their support in extracting and anonymizing data from the electronic medical record systems.
Funding
This work was supported by the Baoshan Science and Technology Planning Project (Grant No. 2024bskjylzd007).
Author information
Authors and Affiliations
Contributions
H. L.: methodology, validation, formal analysis, writing – review & editing; W. Y.: investigation, methodology; D. Y.: data curation, investigation; Y. J.: resources, investigation; Y. Yang: data curation, formal analysis, writing – review & editing; Q. X.: visualization, validation; J. C.: resources, supervision; Z. Z.: software, project administration; C. Z.: conceptualization, writing – original draft, funding acquisition, supervision, corresponding author; X. W.: funding acquisition, supervision, writing – review & editing, corresponding author. H. L., W. Y. and D. Y. contributed equally to this work.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval and consent to participate
The study protocol was reviewed and approved by the Ethics Committee of The Second People’s Hospital of Baoshan City, Yunnan Province (Approval No. 2025-025-01). Written informed consent was waived owing to the retrospective design and use of fully de-identified data extracted from the electronic medical record systems of both The First Affiliated Hospital of Dali University and The Second People’s Hospital of Baoshan City.
Consent for publication
All authors have reviewed and approved the final manuscript and consent to its publication.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Li, H., Yang, W., Yang, D. et al. Association of the novel remnant cholesterol inflammatory index with acute coronary syndrome risk in hospitalized patients with diabetes: a two-center study. Sci Rep 16, 3237 (2026). https://doi.org/10.1038/s41598-025-33161-w
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-33161-w