Abstract
Chronic rhinosinusitis with nasal polyps (CRSwNP) is a chronic inflammatory disease that significantly impairs quality of life. Dupilumab, a monoclonal antibody targeting IL-4 and IL-13 signaling, has emerged as a highly effective therapy for patients with severe, uncontrolled CRSwNP. This retrospective real-life cohort study assessed the impact of dupilumab on symptom severity using both subdomain and individual item scores of the validated Sino-Nasal Outcome Test-22 (SNOT-22). Eighty-nine patients with CRSwNP were evaluated before and after six months of dupilumab treatment. Significant and clinically meaningful improvements were observed in all four SNOT-22 domains—nasal, otologic/facial pain, sleep, and emotional—with the most pronounced reduction in nasal symptoms. Item-level analysis revealed the greatest improvements in decreased sense of smell/taste, nasal congestion, thick nasal discharge, post-nasal drip, and the need to blow the nose—symptoms also rated most severe at baseline. These findings underscore dupilumab’s effectiveness in alleviating the most burdensome aspects of CRSwNP. Domain- and item-level SNOT-22 assessment may offer a clearer view of disease burden and treatment impact than the total score alone.
Similar content being viewed by others
Introduction
Chronic Rhinosinusitis (CRS) is a heterogenous chronic inflammation of the nose and paranasal sinus defined by rhinologic symptoms (nasal blockage, rhinorrhea, facial pain impaired sense of smell) and inflamed sinonasal mucosa, lasting for more than 12 weeks. CRS is traditionally categorized into two main phenotypes: CRS without nasal polyps (CRSsNP) and CRS with nasal polyps (CRSwNP), with CRSwNP being the more severe and recurrent form1. CRSwNP accounts for about 25% of CRS cases and is frequently associated with comorbidities including bronchial asthma and non-steroidal anti-inflammatory drug-exacerbated respiratory disease (N-ERD), due to shared immunopathogenesis1,2,3. Recently, an endotype-based classification of CRS has emerged, based on the underlying inflammatory pathways: Type 1 inflammation is driven by the canonical cytokines IFN-γ and IL-12, type 2 by IL-4, IL-5, IL-13; and type 3 by IL-17 A and IL-22. Clinically, the key distinction lies between type 2 and non–type 24. In Western countries, around 85% of CRSwNP cases are associated with type 2 inflammation, whereas approximately 50% of CRSsNP cases also exhibit a type 2 profile5. The pathophysiological mechanisms of CRS include epithelial barrier dysfunction, impaired mucociliary clearance, dysregulated immune response, and extensive tissue remodeling. The exact cause for these alterations remains elusive; however, host factors and exposure to pathogens, allergens and pollutants are considered to contribute4,6. Standard treatment of CRS includes intranasal corticosteroids (INCS), short-term systemic corticosteroids (SCOS) and endoscopic sinus surgery (ESS)1. Nevertheless, despite standard therapy, a considerable proportion of CRSwNP patients continue to experience severe symptoms and polyp recurrence7,8. The recent introduction of monoclonal antibodies targeting key type 2 inflammatory mediators has revolutionized the management of severe uncontrolled CRSwNP1,9. Currently approved biologics for CRSwNP are dupilumab, mepolizumab and omalizumab10,11,12. The EUFOREA group recently recommended dupilumab as first-choice biologic agent in CRSwNP, based on network meta-analysis and/or expert opinion13. Dupilumab is a fully human monoclonal antibody that blocks IL-4 and IL-13 signaling by targeting the IL-4 receptor α-subunit, thereby reducing type 2 inflammation. The efficacy of dupilumab treatment in CRSwNP has been demonstrated in clinical trials10 and further supported by numerous real-world studies14,15.
The recommended patient-reported outcome measure (PROM) for evaluation of response to biologic treatment in CRSwNP is the sinonasal-outcome-test-22 (SNOT-22)9. The SNOT-22 is a validated 22-item questionnaire that assesses the severity of CRS symptomatology and CRS-specific quality of life (QoL)16. The SNOT-22 can be categorized into different domains, each reflecting specific symptom types (such as nasal symptoms, otologic/facial pain, sleep disturbances, and emotional issues)17. As distinct outcome measures, the SNOT-22 domains offer additional clinical value beyond the overall total score18,19,20,21,22. Available real-life data on improvements in SNOT-22 domains during dupilumab treatment for CRSwNP remain limited23,24,25,26. Moreover, recent post-hoc analyses of the phase III SINUS trials further evaluated treatment-related changes of individual SNOT-22 items, indicating the importance of item-level assessment27,28. To date, no real-life study in CRSwNP has specifically examined the impact of dupilumab therapy on all individual SNOT-22 item scores. Therefore, the objectives of this study were to evaluate the effects of dupilumab on both the SNOT-22 domain and individual item scores in patients with CRSwNP.
Materials and methods
Study design and subjects
This retrospective, longitudinal real-life cohort study was carried out at the Department of Otorhinolaryngology, Medical University of Graz, Austria. We included all consecutive adult patients with CRSwNP, who received dupilumab treatment (300 mg biweekly) between January 2020 and February 2025, and completed the 6-month follow-up visit. Patients were identified through the institutional electronic medical record system. Exclusion criteria were: non-primary forms of CRSwNP, age < 18 years, and incomplete clinical data. Ninety-two eligible patients were initially included and three patients were excluded due to incomplete documentation of the study variables of interest.
Dupilumab prescription and clinical assessments
Dupilumab was initiated at the Department’s Rhinology outpatient clinic. All included patients were treated as part of routine clinical practice. Dupilumab was prescribed according to the national reimbursement guidelines established by the Austrian Federation of Social Insurances29: It was initiated as add-on therapy to INCS in adults with bothersome, recurrent CRSwNP after surgical treatment (or medical contraindication to surgery) and after failure, intolerance, or contraindication to SCOS. Clinical evaluations were conducted at baseline and again at the 6-month follow-up. These assessments included completion of the German SNOT-22 questionnaire30, nasal endoscopy to assess polyp burden, and blood sampling for measurement of serum eosinophil count (EOS) and total immunoglobulin E (IgE) levels. At the baseline visit, patients’ medical histories were obtained, documenting age, sex, prior ESS, and comorbid conditions including asthma and NERD. Nasal polyp size was assessed by using the well-known Meltzer nasal polyp score (NPS), which grades the extent of nasal polyps on a scale from 0 to 4 for each nasal cavity, yielding a total score ranging from 0 to 831. At the 6-month follow-up visit, dupilumab treatment outcome was evaluated. A reduction of > 1 point in the total NPS is required to qualify for continued reimbursement under national insurance criteria29.
Study aims
The main objectives of this study were to evaluate changes in the SNOT-22 domains and individual SNOT-22 item scores after six months of dupilumab therapy in patients with CRSwNP.
Study variables of interest
The SNOT-22 is a 22-item symptom-questionnaire to assess the impact of CRS on patients’ QoL (SNOT-22: All rights reserved. Copyright 2006 by Jay F. Piccirillo, M.D. Washington University in St. Louis, Missouri). Each of the items is rated on a scale from 0 to 5: 0 = no problem, 1 = very mild problem, 2 = mild or slight problem, 3 = moderate problem, 4 = severe problem, and 5 = as bad as it can be. The total score ranges from 0 to 110, with higher scores reflecting greater symptom burden. The minimal clinically important difference (MCID) of the total SNOT-22 for medically managed CRS patients has been determined as ≥ 12 points32. Based on the validated model by Feng et al.17, the following four SNOT-22 domains can be distinguished: nasal domain (items: need to blow nose, sneezes, runny nose, cough, post-nasal drip, thick nasal discharge, decreased sense of smell/taste, blockage/congestion of nose; score range: 0–40 points; MCID: 4), otologic/facial pain domain (items: ear fullness, dizziness, ear pain, facial pain/pressure; score range: 0–20; MCID: 2), sleep domain (items: difficulty falling asleep, wake up at night, lack of a good night’s sleep, wake up tired, fatigue during the day, reduced productivity, reduced concentration, frustrated/restless/irritated; score range: 0–40 points; MCID: 4), emotional domain (items: sad, embarrassed; score range: 0–10 points; MCID: 1)33. Adopting the item severity methodology from Hopkins et al.28, the five most severe items were further classified into mild (score 0 and 1), moderate (score 2 and 3) and severe (score 4 and 5). The proportions of severity grade improvements (improvement of ≥ 1 severity grade, e.g., from “severe” to “moderate”) were then assessed.
Statistical analyses
Statistics were performed using SPSS © software, version 29.0 (IBM©, Armonk, NY, USA). Continuous variables are presented as mean ± standard deviation or as median with interquartile range (IQR), while categorical variables are reported as absolute numbers and percentages. Longitudinal comparisons were conducted using paired t-tests for NPS, total SNOT-22, and SNOT-22 domain scores, and the Wilcoxon signed-rank test for individual SNOT-22 item scores. In an exploratory analysis, outcomes were also compared between patients with and without comorbid asthma, showing no significant differences. Detailed results are provided in Supplementary Table S1 and S2. Effect sizes were expressed as Cohen’s d and Rosenthal’s r for significant results. A two-sided p-value < 0.05 was considered statistically significant. As SNOT-22 domains and items represent interrelated components of a single validated questionnaire, no formal Bonferroni correction was applied. Nevertheless, to confirm the robustness of the results, all p-values were post hoc verified using the Benjamini–Hochberg false discovery rate (FDR) correction, applied across all 26 main comparisons (4 domains and 22 items). Adjusted p-values ranged from 0.001 to 0.026, all remaining below the 0.05 significance threshold.
Ethics
The study was conducted in accordance with the Declaration of Helsinki, and approved by the Institutional Review Board of Medical University of Graz (1115/2025). Due to the retrospective nature of this study, requirement of patient’s informed consent was waived by the Institutional Review Board of Medical University of Graz.
Results
Demographics and baseline characteristics
This retrospective analysis included 89 patients with CRSwNP, who received six months dupilumab treatment at our academic institution. The study population consisted of 38 females (43%) and 51 males (57%), with a mean age of 51.4 ± 14.1 years. Patients had undergone a median of 2 prior ESS, (range: 0–7). Two individuals (2%) had not received ESS due to medical contraindications. A comorbid asthma bronchiale was present in 57 patients (64%). Fifteen patients (17%) had a history of N-ERD. At baseline, the mean EOS was 0.4 ± 0.3 × 10⁹/L and 79 patients (89%) had EOS of > 0.15. The mean total serum IgE level at baseline was 199.2 ± 226.8 kU/L.
NPS and total SNOT-22 outcomes
The mean NPS declined significantly by 3.5 ± 2.1 points from 4.9 ± 1.7 at baseline to 1.3 ± 1.6 after six months of treatment [p < 0.001, d = 1.7]. Notably, 75 participants (84%) exhibited an NPS reduction of > 1 at the follow-up visit. Similarly, mean total SNOT-22 decreased significantly by 29.2 ± 21.3 points from 53.2 ± 21.6 prior-treatment to 24 ± 19.1 post-treatment [p < 0.001, d = 1.3]. A clinically meaningful improvement in SNOT-22 score, defines as a reduction of ≥ 12 points, was achieved by 72 patients (81%). Individual changes in total NPS and SNOT-22 scores are illustrated in Fig. 1.
Individual changes in (a) the total nasal polyp score (NPS) and (b) the total Sino-Nasal Outcome Test-22 (SNOT-22) score from baseline to 6-month follow-up under dupilumab therapy. Each line represents an individual patient.
SNOT-22 domains outcome
Significant improvements were observed in all four SNOT-22 domains following six months of dupilumab treatment (p < 0.05). The mean improvements in each domain exceeded the respective MCID (p < 0.05). Comprehensive results are presented in Table 1. The MCID was met for the nasal domain by 82 subjects (92%), for the extra-nasal domain by 54 subjects (61%), for the sleep domain by 68 subjects (76%), and for the emotional domain by 60 subjects (67%). Individual changes for each SNOT-22 domain are shown in Fig. 2.
Individual baseline and 6-month post-dupilumab treatment scores for each SNOT-22 domain—nasal (a), otologic/facial pain (b), sleep (c), and emotional (d). Each line represents an individual patient.
Individual SNOT-22 items outcome
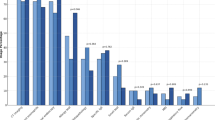
All item scores exhibited a significant reduction after six months dupilumab treatment (p < 0.001, detailed results are presented in Table 2). The most effective reductions were observed for blockage/congestion of nose (r = 0.89), need to blow nose (r = 0.83), decreased sense of smell/taste (r = 0.82), thick nasal discharge (r = 0.80) and post-nasal drip (r = 0.78), respectively. At baseline, the five lowest scored SNOT-22 items were ear pain (median: 0 [IQR 0–2]), facial pain/pressure (median: 0 [IQR 0–2), dizziness (median: 1 [IQR 0–2]), sad (median: 1 [IQR 0–3]), and embarrassed (median: 2 [IQR 0–3]), respectively. In contrast, the five most severe items at baseline were decreased sense of smell/taste (median: 5 [IQR 4–5]), blockage/congestion of nose (median: 4 [IQR 3–5]), need to blow nose (median: 3 [IQR 2.5-4]), post-nasal drip (median: 3 [IQR 2–4]) and thick nasal discharge (median: 3 [IQR 2–4]), respectively. Severity grade outcomes of these items are displayed in Fig. 3. An improvement of ≥ 1 severity grade was achieved by 63 subjects (71%) for blockage/congestion of nose, by 60 subjects (67%) for thick nasal discharge, by 59 subjects (66%) for decreased sense of smell/taste, by 55 subjects (61%) for need to blow nose, and by 53 subjects (60%) for post-nasal drip, respectively.
Severity outcomes of the five highest ranked SNOT-22 items at baseline.
Discussion
The introduction of biologic medications has significantly transformed the therapeutic landscape of uncontrolled CRSwNP, offering targeted options for patients who previously had limited alternatives. Dupilumab became the first biologic agent approved for this indication in 2019. Its clinical efficacy has been well established through randomized controlled trials10 and further validated by a growing body of real-world evidence14,15. A recent meta-analysis has synthesized the sinonasal outcomes of 26 real-life studies evaluating the effectiveness of dupilumab in CRSwNP15. The authors reported a pooled mean NPS decrease of 3.6, and SNOT-22 improvement of 37.2, highlighting the substantial benefit observed in routine practice. Our study revealed a comparable mean NPS and SNOT-22 improvement of 3.5 and 29.2 points, respectively, confirming that our patient cohort reflects the typical population that benefits from dupilumab therapy.
The SNOT-22 is one of the most common used PROMs in the field of CRS9. The questionnaire covers various aspects of CRS-specific QoL including sinonasal and otologic symptom severity, craniofacial pain, emotional and social impact, reduced productivity, and sleep disturbances16. While the total SNOT-22 score appropriately reflects overall disease impact, a more detailed insight is essential to fully capture the impact of CRS on the different dimensions on patients’ QoL. As a result, some research groups have proposed domain structures within the SNOT-22 to better capture distinct symptom profiles in patients with CRS17,34,35,36. By using principal component analysis and exploratory factor analysis (EFA), DeConde et al.34 and later Crump et al.35 identified five underlying symptom factors within the SNOT-22, reflecting the sleep, nasal, otologic/facial, extranasal and emotional domains. Notably, the two studies proposed different interpretations of the extranasal symptom group. In contrast, Feng et al.17 determined the same four domains using EFA, without including an extranasal factor. Compared to the other two studies, they performed a confirmatory factor analysis for construct validity and a subsequent goodness of fit measurement, to strengthen their domain model. Moreover, their study featured a geographically more diverse patient population, supporting greater generalizability. In a follow-up study, the same group established MCID values for each of the identified domains30. The most recent attempt to refine the domain structure of the SNOT-22 was made by Khan et al.36, who proposed a five-domain structure comprising nasal, otologic/facial pain, sleep, function, and emotion domains. Unlike earlier models, they excluded an extranasal factor and introduced a distinct function domain that includes fatigue, reduced productivity, and concentration. Their model was specifically developed for CRSwNP and showed strong psychometric properties. However, the lack of consistency among proposed SNOT-22 domain models remains a significant barrier to their broader adoption as standardized tools in both clinical practice and research.
Current real-life data on how individual SNOT-22 domains respond to dupilumab treatment in CRSwNP remains scarce23,24,25,26. Therefore, we aimed in the present study to evaluate how dupilumab affects the different symptom domains of the SNOT-22 in 89 patients with CRSwNP treated at our academic center. We selected the model proposed by Feng et al.17 based on its strong methodological validity, broad adoption in the field, and the availability of established MCID thresholds for each subdomain. We observed a clinically significant reduction in all four SNOT-22 domains, with mean scores exceeding the MCID of each domain. The most effective reduction was seen in the nasal domain (d = 1.4, 92% met MCID), followed by the sleep domain (d = 1.1, 76% met MCID). Similar results were reported from Boscke et al.26, who also applied the Feng domain model with established MCIDs. In their study of 41 patients, the MCIDs were met after 12 months dupilumab treatment for the nasal-, sleep-, emotional-, and otologic/facial pain domain in 97.7%, 71%, 71% and 69.2%, respectively. Another study by Campion et al.24 observed significant SNOT-22 domain score reductions comparable to our results, however, they did not assess proportions of reached MCIDs. Two further real-life dupilumab studies reported as well significant improvements across all SNOT-22 domains, but they have used different domain models making direct comparisons difficult23,25.
Evaluating how patients perceive and respond to their condition offers valuable insight into the broader impact of CRSwNP on both their overall health and daily life. Individual item scores provide an understanding of which aspects of CRSwNP are most disruptive from the patient’s perspective. To our knowledge, this is the first real-world study to assess all individual SNOT-22 item responses in CRSwNP patients treated with dupilumab. Our findings revealed significant improvements across all items following six months of therapy. The most effective reductions were observed in nasal items, particularly decreased sense of smell/taste, nasal blockage, thick nasal discharge, need to blow nose, and post-nasal drip. Notably, these symptoms were also ranked as most severe items at baseline, highlighting the effectiveness of dupilumab in targeting the most impactful aspects of the disease. Hopkins et al.28 evaluated in a post-hoc analysis of the SINUS trials the five SNOT-22 items, that patients identified as having the greatest impact on their health. Unfortunately, we did not ask our patients to individually select the five most troublesome items. Nevertheless, the five highest-rated items in our cohort closely align with the five most frequently reported items in the study by Hopkins et al.28: decreased sense of smell (87.4%), nasal blockage (82.4%), post-nasal drip (39.5%), thick nasal discharge (37.1%), and wake up at night (25.7%). They further classified these items according to their severity into mild (score 0 and 1), moderate (score 2 and 3), and severe (score 4 and 5), and determined the proportions of severity grade improvements. We adopted this methodology and observed similar results for decreased sense of smell (66% vs. 73%), nasal blockage (71% vs. 76%), post-nasal drip (60% vs. 66%), and thick nasal discharge (67% vs. 73%).
This study has several limitations. First, its retrospective design and the exclusion of a few patients with incomplete data may have introduced selection bias. Second, the study was conducted at a single academic center, potentially limiting the generalizability of our findings to broader populations. Third, the relatively short follow-up duration of six months limits the assessment of long-term sustainability of symptom improvements achieved with dupilumab. Fourth, we did not perform objective olfactory/gustatory assessments to complement subjective patient-reported improvements in smell and taste. Fifth, our evaluation was limited to the SNOT-22 questionnaire and did not incorporate broader, general QoL measures (e.g., EQ-5D), which could have provided additional insights into overall patient well-being. Sixth, the study lacked sufficient power for subgroup analyses. Although an exploratory comparison between patients with and without asthma was performed, further stratification by blood eosinophil levels or N-ERD was limited by the very small subgroup sizes. Finally, patients were not asked to individually select their five most bothersome symptoms, as original integrated part of the SNOT-22. Despite these limitations, a notable strength of this study is that it represents the most comprehensive real-life evaluation of SNOT-22 item-level responses to dupilumab therapy in patients with CRSwNP to date.
Conclusions
This study demonstrated that dupilumab treatment in routine clinical practice leads to significant improvements across all SNOT-22 domains and individual item scores in patients with CRSwNP. The most substantial effects were seen in the nasal domain and nasal items, particularly those most severe ranked at baseline, including decreased sense of smell/taste and nasal congestion. These real-world findings underscore dupilumab’s effectiveness in targeting the core symptoms that drive patient discomfort and disease burden. Assessing SNOT-22 domain and item scores helps identify which specific aspects of CRSwNP have the greatest impact on patients’ daily lives, and may offer a more complete picture of disease burden than looking at the total SNOT-22 score alone. This approach also allows for a clearer evaluation of how the most burdensome symptoms respond to treatment.
Data availability
The datasets generated during and analyzed during the current study are available from the corresponding author on reasonable request.
References
Fokkens, W. J. et al. European position paper on rhinosinusitis and nasal polyps 2020. Rhinology 58 (Suppl S29), 1–464 (2020).
Chaaban, M. R., Walsh, E. M. & Woodworth, B. A. Epidemiology and differential diagnosis of nasal polyps. Am. J. Rhinol Allergy. 27 (6), 473–478 (2013).
Kato, A. & Kita, H. The immunology of asthma and chronic rhinosinusitis. Nat. Rev. Immunol.. 16 https://doi.org/10.1038/s41577-025-01159-0 (2025).
Kato, A. et al. Endotypes of chronic rhinosinusitis: relationships to disease phenotypes, pathogenesis, clinical findings, and treatment approaches. Allergy 77 (3), 812–826 (2022).
Stevens, W. W. et al. Associations between inflammatory endotypes and clinical presentations in chronic rhinosinusitis. J. Allergy Clin. Immunol. Pract. 7 (8), 2812–2820e3 (2019).
Kato, A., Schleimer, R. P. & Bleier, B. S. Mechanisms and pathogenesis of chronic rhinosinusitis. J. Allergy Clin. Immunol. 149 (5), 1491–1503 (2022).
Calvanese, L. et al. Polyps’ extension and recurrence in different endotypes of chronic rhinosinusitis: A series of 449 consecutive patients. J. Clin. Med. 13 (4), 1125 (2024).
DeConde, A. S. et al. Prevalence of polyp recurrence after endoscopic sinus surgery for chronic rhinosinusitis with nasal polyposis. Laryngoscope 127 (3), 550–555 (2017).
Fokkens, W. J. et al. EPOS/EUFOREA update on indication and evaluation of biologics in chronic rhinosinusitis with nasal polyps 2023. Rhinology 61 (3), 194–202 (2023).
Bachert, C. et al. Efficacy and safety of dupilumab in patients with severe chronic rhinosinusitis with nasal polyps (LIBERTY NP SINUS-24 and LIBERTY NP SINUS-52): Results from two multicentre, randomised, double-blind, placebo-controlled, parallel-group phase 3 trials. Lancet 394, 1638–1650 (2019).
Gevaert, P. et al. Efficacy and safety of omalizumab in nasal polyposis: 2 randomized phase 3 trials [published correction appears in J Allergy Clin Immunol. 2020;146(3):595–605. (2021).
Han, J. K. et al. Mepolizumab for chronic rhinosinusitis with nasal polyps (SYNAPSE): A randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Respir Med. 9 (10), 1141–1153 (2021).
Fokkens, W. J. et al. Pocket guide: Biologics in upper and lower airways in adults. Rhinology 63 (2), 242–244 (2025).
Seys, S. F. et al. Real-world effectiveness of dupilumab in a European cohort of chronic rhinosinusitis with nasal polyps (CHRINOSOR). J. Allergy Clin. Immunol. 155 (2), 451–460 (2025).
Rodriguez-Iglesias, M. et al. Effect of dupilumab in CRSwNP sinonasal outcomes from real life studies: A systematic review with meta-analysis. Curr. Allergy Asthma Rep. 25 (1), 13 (2025).
Hopkins, C., Gillett, S., Slack, R., Lund, V. J. & Browne, J. P. Psychometric validity of the 22-item sinonasal outcome test. Clin. Otolaryngol. 34 (5), 447–454 (2009).
Feng, A. L. et al. A validated model for the 22-item Sino-Nasal outcome test subdomain structure in chronic rhinosinusitis. Int. Forum Allergy Rhinol. 7 (12), 1140–1148 (2017).
Hoehle, L. P. et al. Symptoms of chronic rhinosinusitis differentially impact general health-related quality of life. Rhinology 54 (4), 316–322 (2016).
Speth, M. M. et al. Changes in chronic rhinosinusitis symptoms differentially associate with improvement in general health-related quality of life. Ann. Allergy Asthma Immunol. 121 (2), 195–199 (2018).
Sedaghat, A. R., Hoehle, L. P. & Gray, S. T. Chronic rhinosinusitis control from the patient and physician perspectives. Laryngoscope Investig Otolaryngol. 3 (6), 419–433 (2018).
Mattos, J. L. et al. Symptom importance, patient expectations, and satisfaction in chronic rhinosinusitis. Int. Forum Allergy Rhinol. 9 (6), 593–600 (2019).
Campbell, A. P. et al. Depression symptoms and lost productivity in chronic rhinosinusitis. Ann. Allergy Asthma Immunol. 118 (3), 286–289 (2017).
Kilty, S. J. & Lasso, A. Canadian real-world study of access and clinical results using dupilumab for chronic rhinosinusitis with polyps. J. Otolaryngol. Head Neck Surg. 51 (1), 17 (2022).
Campion, N. J. et al. The real life efficacy of dupilumab is independent of initial polyp size and concomitant steroids in CRSwNP. J. Otolaryngol. Head Neck Surg. 52 (1), 56 (2023).
Ottaviano, G. et al. The impact of dupilumab on work productivity and emotional health in crswnp: A multicentric study in Northeast Italy. J. Pers. Med. 14 (5), 468 (2024).
Boscke, R., Heidemann, M. & Bruchhage, K. L. Dupilumab for chronic rhinosinusitis with nasal polyps: real-life retrospective 12-month effectiveness data. Rhinology 61 (3), 203–213 (2023).
Lee, S. E. et al. Dupilumab improves health related quality of life: Results from the phase 3 SINUS studies. Allergy 77 (7), 2211–2221 (2022).
Hopkins, C. et al. Impact of dupilumab on sinonasal symptoms and outcomes in severe chronic rhinosinusitis with nasal polyps. Otolaryngol. Head Neck Surg. 170 (4), 1173–1182 (2024).
Österreichische Sozialversicherung Infotool zum Erstattungskodex. (2025). Available from: https://www.sozialversicherung.at/oeko/views/index.xhtml. Accessed on May 1.
Riedl, D. et al. Assessment of health-related quality-of-life in patients with chronic rhinosinusitis—Validation of the German Sino-Nasal outcome Test-22 (German-SNOT-22). J. Psychosom. Res. 140, 110316 (2021).
Meltzer, E. O. et al. Rhinosinusitis: Developing guidance for clinical trials. Otolaryngol. Head Neck Surg. 135 (5 Suppl), S31–S80 (2006).
Phillips, K. M., Hoehle, L. P., Caradonna, D. S., Gray, S. T. & Sedaghat, A. R. Minimal clinically important difference for the 22-item sinonasal outcome test in medically managed patients with chronic rhinosinusitis. Clin. Otolaryngol. 43 (5), 1328–1334 (2018).
Singerman, K. W., Phillips, K. M. & Sedaghat, A. R. Minimal clinically important difference for subdomains of the 22-item Sino-Nasal outcome test in medically managed chronic rhinosinusitis patients. Int. Forum Allergy Rhinol. 12 (9), 1196–1199 (2022).
DeConde, A. S., Bodner, T. E., Mace, J. C. & Smith, T. L. Response shift in quality of life after endoscopic sinus surgery for chronic rhinosinusitis. JAMA Otolaryngol. Head Neck Surg. 140 (8), 712–719 (2014).
Crump, R. T., Liu, G., Janjua, A. & Sutherland, J. M. Analyzing the 22-item Sino-Nasal outcome test using item response theory. Int. Forum Allergy Rhinol. 6 (9), 914–920 (2016).
Khan, A. H. et al. Development of sinonasal outcome test (SNOT-22) domains in chronic rhinosinusitis with nasal polyps. Laryngoscope 132 (5), 933–941 (2022).
Funding
The APC was funded by the State Styria, Department 12 Economy, Tourism, Science and Research (Grant number: ABT12-128252/2025).
Author information
Authors and Affiliations
Contributions
All authors have provided substantial contributions to the conception or design of the work acquisition, analysis or the interpretation of data for the work. All worked on the draft or revised it critically for important intellectual content. The final version was approved for publishing by all authors. The authors agree on accountability for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Lang, A., Habenbacher, M., Moser, U. et al. Impact of dupilumab on SNOT-22 domain and individual item scores in chronic rhinosinusitis with nasal polyps. Sci Rep 16, 3772 (2026). https://doi.org/10.1038/s41598-025-33742-9
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-33742-9