Abstract
To investigate dosimetry-derived tumor-to-perfused normal tissue ratio (T/N) and corresponding clinical outcomes of patients receiving yttrium-90 radioembolization with glass microspheres for tumors identified as hypovascular on arterial-phase cross-sectional imaging. Patients with hypovascular lesions undergoing 90Y-RE at a single institution were included. Inclusion criteria were hypodense or hypoattenuating tumors relative to normal liver on arterial phase CT or MRI, excluding necrotic tumors. Pre-treatment planning mapping angiography 99mTc-MAA SPECT/CT was used for multi-compartment dosimetry for T/N estimation. The final cohort comprised 27 patients (median age 68, range 52–85 years), including 13 with Child-Pugh A cirrhosis, and ECOG status of 0 (n = 15), 1 (n = 11), and 2 (n = 1). Tumor types were hepatocellular carcinoma (n = 11), intrahepatic cholangiocarcinoma (n = 8), and liver metastases (n = 8). Treatment strategies included lobar (19%), lobar plus segmentectomy (22%), single segmentectomy (33%) and multiple segmentectomies (30%). The mean index tumor size was 5.7 ± 3.2 cm. Mean tumor and perfused normal tissue doses were 480 ± 314 Gy and 270 ± 138 Gy, respectively. Median T/N ratio on planning SPECT/CT was 1.56 (range 0.28–4.79). Response assessments using mRECIST criteria for index lesions were complete response (41%), partial response (33%), stable disease (22%), and progressive disease (4%). Nine patients experienced Grade 1 adverse events (ALP, ALT, or AST serum markers) with no other adverse events. Calculated T/N ratios were greater than 1.00 for most patients with hypovascular tumors, indicating that tumor uptake of 90Y may be greater than perceived from arterial-phase cross-sectional imaging. Post-treatment follow-up demonstrated favorable outcomes for this patient population.
Similar content being viewed by others
Introduction
Trans-arterial Yttrium-90 radioembolization (90Y-RE) tumor therapy relies on accurate characterization and utilization of flow dynamics to maximize dose to target lesions, while minimizing damage to surrounding normal liver parenchyma1,2. While many liver tumors have increased arterial hypervascularity compared to background liver, some are inherently hypovascular on arterial-phase CT-imaging. Tumor hypovascularity is not considered an absolute contraindication to 90Y-RE, as it has been shown that hypovascularity on contrast-enhanced imaging and hepatic angiography does not affect survival after radioembolization3. It does, however, increase technical challenges due to increased toxicity to perfused background liver and may hinder clinical response4. Some hepatic tumors, such as liver metastases from colorectal cancer (CRC), are notorious for being hypovascular and are better suited for resection or ablation with adequate margin when possible5,6. When these are treated with 90Y-RE, increased hepatic arterial enhancement compared to portal vein enhancement on CT imaging can predict better response to treatment5.
Multi-compartment voxel-based dosimetry using 99mTc-MAA SPECT/CT for tumor-to-normal liver ratio (T/N) estimation is increasingly used for 90Y-RE treatment planning to predict treatment response and hepatotoxicity and shows general correlation with perfusion angiography7,8,9. It is unknown whether liver tumors deemed hypovascular on CT imaging also have low T/N with multi-compartment voxel-based dosimetry. This study aims to quantify dosimetry-derived tumor-to-normal liver ratios (T/N) in patients with both primary and secondary hepatic malignancies demonstrating hypovascularity on pre-procedural cross-sectional imaging. Secondary outcomes include mRECIST criteria for treatment response.
Materials and methods
Study cohort selection
Institutional review board approved this retrospective study of patients who underwent radioembolization with 90Y glass microspheres (Boston Scientific Corporation, Marlborough, MA). The final cohort was derived from patients identified with hypovascular tumors after reviewing all 90Y-RE treatments at a single institution between January 2022 and December 2023. For inclusion criteria, hypovascularity of the tumors was defined as the presence of tumoral enhancement less than background liver enhancement during the arterial phase of contrast on MRI or CT, followed by gradual enhancement or no enhancement during the portal venous and delayed phases relative to the normal liver (Fig. 1a, b). Exclusion criteria included tumors with pre-existing necrosis. Tumor hypovascularity was confirmed by 3 board-certified radiologists with 18, 13, and 8 years of experience, respectively. Patients were excluded if they did not undergo MAA SPECT/CT imaging, or the imaging did not include uptake surrounding the index lesion. All methods were carried out in accordance with relevant guidelines and regulations, and are in accordance with the Declaration of Helsinki.
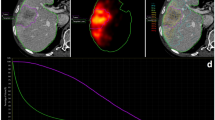
Pre-treatment CT axial (a) and coronal (b) of a 66 year-old female with pathology-proven poorly differentiated carcinoma favoring intra-hepatic cholangiocarcinoma (ICC) measuring 5.6 cm x 5.2 cm. Arterial-phase CT demonstrates decreased enhancement compared to background liver. (c) Reconstruction software demonstrates iso-lines of Tc99m-MAA uptake overlaid on pre-treatment contrast-enhanced CT of the tumor. Axial (d) and coronal (e) MR scan of tumor demonstrating complete response and central necrosis 29 days following treatment.
90Y-RE technique
All procedures were performed after obtaining informed consent from subjects and/or their legal guardian(s). Suitable candidates for 90Y-RE underwent a pretreatment planning visceral arteriogram with 99mTc-MAA and were sent to nuclear medicine for SPECT/CT imaging and lung shunt fraction calculation. In a subsequent procedure, Therasphere 90Y glass microspheres were administered by the authorized user (AU) interventional radiologist. Personalized multi-compartment voxel-based dosimetry was employed using Simplicity90Y (Simplicit90Y, Boston Scientific Corporation). Dosimetry followed a “personalized” regimen, meaning there was no pre-defined prescribed dose based on efficacy or safety, rather it was at the discretion of the AU based on uptake on the 99mTc-MAA SPECT/CT within each region of interest. Treatments were intended to be either curative or palliative depending on the patient and tumor distribution. Patients were monitored and then discharged home the same day. Patients with imaging evidence of residual or recurrent disease underwent further treatment if deemed medically appropriate.
Dosimetry
All patients were injected with 99mTc-MAA during pre-treatment angiographic mapping and subsequently imaged with planar and SPECT/CT acquisitions (window: 140.5 ± 10.5 keV, 60 projections, 180°, 128 × 128, 15 s/projection) (Symbia T16; Siemens) (Fig. 2). SPECT data was reconstructed using an iterative method (ordered-subset expectation maximization: 20 iterations, 8 subsets). The 99mTc-MAA matrix size was 128 × 128, and the attenuation map was CT-based, and scatter correction was energy window-based with a CT-based attenuation map and scatter correction. A 9.0 mm FWHM Gaussian filter was used for smoothing. The distribution of the 99mTc-MAA particles was used to determine extrahepatic shunting, calculate the lung shunt fraction (LSF) (Eq. 1), and estimate mean absorbed dose to various segmented compartments. The T/N ratio for each patient was extracted from technetium-99mTc-MAA single-photon emission computed tomography (SPECT)/computed tomography (CT) performed for multicompartment dosimetry. The Tc99m MAA map was converted into a voxel dose map using known activity in the perfused volume and correlating the counts in the same region with Simpicit90Y hand-drawn segmentation, using the local deposition model. Therapeutic activity in this cohort was determined through this SPECT/CT voxel dose map with hand-drawn segmentation. Pure physical decay and permanent sphere trapping are assumed in all Y90 dosimetry. T/N was defined as ratio between perfused tumor tissue and perfused normal tissue uptake (Eq. 2) on the pre-treatment 99mTc-MAA scan calculated using Simplicity90Y software (Simplicit90Y, Boston Scientific Corporation) (Fig. 1c). Median T/N is reported rather than mean to reduce any potential mathematical artifact from outliers. The difference between T/N and a T/N equal to 1.00 was used to display values in the waterfall plot in Fig. 3.
Nuclear Medicine pre-treatment Tc99m-MAA raw data demonstrating hypovascular tumor compared to background liver in coronal, axial, and sagittal reconstruction-images.
Clinical outcomes
Patients were followed with triphasic computed tomography (CT) or magnetic resonance imaging (MRI) at 1-month post-procedure and then at 3-month intervals after radioembolization (Fig. 1d,e). Index tumor response to treatment, evaluated using mRECIST criteria, was classified as complete response (CR), partial response (PR), stable disease (SD), or progressive disease (PD)10. Despite tumors being hypoenhancing to background liver on arterial phase of contrast, there was still some degree of enhancement on either arterial or delayed phase imaging, and thus non-enhancing tissue on post-procedure imaging could still be defined separately from untreated hypovascular tumor. Digital subtraction sequences on MR and CT were also used to arbitrage difficult cases. Treatment response with the use of mRECIST criteria was determined by a board-certified radiologist with 9 years of experience.
Adverse events (AEs) were defined using the CTCAE v5.0 grading values for hepatobiliary serum marker changes during a period of 3 months following-treatment11. Serum markers monitored included Bilirubin, ALP, AST, ALT, albumin, and INR. Increases in serum markers were not recorded as AEs if increases were only seen on the day of treatment and returned to baseline within 3 days. Serious adverse events (SAEs) included but were not limited to jaundice, ascites, radiation pneumonitis, radioembolization induced liver disease (REILD), and hospitalizations/admissions directly related to the 90Y-RE procedure.
Statistical analysis
Descriptive statistics were employed to assess patient demographics, tumor characteristics, and dosimetry. Recording of continuous variables included the mean/median, standard deviation, interquartile range (IQR), minimum values, and maximum values, whereas categorical variables included fractions and percentages.
Results
Study cohort
From a total of 228 90Y-RE treatments at a single institution between January 2022 and December 2023, 27 patients (10 female, 17 male, median age 68, range 52–85) were found to have hypovascular liver tumors without pre-existing regions of necrosis and with sufficient imaging data to perform dosimetry. Mean index tumor size was 5.7 ± 3.2 cm (median 4.3, range 1–12). All patients were Child-Pugh A. Thirteen patients had cirrhosis. ECOG status was 0 (n = 15), 1 (n = 11), or 2 (n = 1).
Additionally, patient tumors were classified as hepatocellular carcinoma (n = 11), intrahepatic cholangiocarcinoma (ICC) (n = 8), or metastatic disease (n = 8), which included colorectal cancer (CRC) (n = 4), malignant melanoma of ciliary body (n = 1), neuroendocrine (NET) (n = 1), leiomyosarcoma (n = 1), and gastrointestinal stromal tumor (GIST) (n = 1) based on varying combinations of imaging, lab markers, and biopsy (Table 1). The mean index tumor size was 5.7 ± 3.2 cm. Six patients had received a previous 90Y-RE treatment using glass microspheres, one of whom underwent an initial resin microsphere 90Y-RE. Treatment for this cohort included single segmentectomy radioembolization (8/27, 33%), single lobectomy radioembolization (5/27, 19%), lobectomy plus segmentectomy radioembolization (6/27, 22%), and multiple segmentectomy radioembolization (8/27, 20%) (Table 2). The mean LSF for the patient cohort was 5.5% ± 3.3%. Perfused liver volumes ranged from 40 to 1296 cc, with a mean of 379 ± 314 cc. Prescribed perfused volume dose ranged from 100 to 800 Gy, with a mean of 283 ± 154 Gy (Table 2). Prescribed mean activity administered was 2.6 ± 1.6 GBq, median 2.1 (range 0.1–5.2).
Tumor to normal ratios
Voxel-based dosimetry revealed that the median T/N for this cohort was 1.56, with values ranging from 0.28 to 4.79 across the cohort. Notably, 81% (22/27) of the patients exhibited T/N greater than 1 (Fig. 3). The mean tumor dose was 480 ± 314 Gy, and the mean dose to perfused normal liver was 270 ± 138. The whole-liver mean absorbed dose (i.e. averaged across the entire liver) was 79 ± 43 Gy (Table 3).
Waterfall comparison between cohort T/N and a T/N equal to 1.00. Twenty-two of the twenty-seven treatments in the cohort resulted in dosimetric T/N values greater than 1.00.
Outcomes
Best response to treatment using mRECIST included 11/27 (41%) CR, 9/27 (33%) PR, 6/27 (22%) SD, and 1/27 (4%) with PD. The median number of days to best treatment response was 62 (range 28–170). At the time of most recent imaging, 11/27 (41%) patients had CR, 8/27 (30%) had PR, 6/27 (22%) had SD, and 2/27 had PD (7%) with one patient progressing from PR to PD at 183 days after treatment. The median number of days from treatment to most recent imaging was 246 (range 51–736) (Table 4). Of 5 patients with a T/N ratio less than 1.00, 3/5 (60%) had stable disease (SD) as their best response while one had a partial response (PR). The patient with the highest T/N ratio in this subgroup (0.97) had complete response (CR).
Adverse events
For adverse events following treatment, 7/27 (26%) of patients experienced a Grade 1 increase in ALP defined as an ALP value greater than the upper limit of normal (ULN) to 2.5x the ULN according to the CTCAE guidelines11. The median number of days to ALP marker increase following treatment was 47.5 days (range 2–101), and 2 of these patients returned to baseline, pre-treatment serum marker values at 190 and 238 days following the initial increase. There were no CTCAE adverse events of grade 3 or higher, and no other post-treatment events or complications were observed.
Discussion
In this single institutional retrospective study, most patients (81%) with hypovascular tumors on pre-procedure cross-sectional arterial-phase contrast imaging demonstrated T/N ratios greater than 1.00 on voxel-based Tc-99 MAA SPECT/CT (Fig. 3), and experienced high rates of treatment response on imaging with low rates of adverse events within an aggressive treatment paradigm of radiation segmentectomy or lobectomy. Though the median T/N value in this cohort (1.56) is much lower than reported in non-hypovascular HCC tumors measured similarly (2.28–2.96)7,12, the findings support previously published data that hypovascular tumors are amenable to radioembolic therapy and may have more favorable T/N on Tc99m MAA SPECT/CT than initially perceived on cross-sectional arterial phase imaging3.
Tumor “hypovascularity” can be assessed using various imaging surrogates to help predict the biodistribution of glass microspheres during radioembolization. These surrogates include tissue density (e.g., hypodense), vascularity (as indicated by uptake of contrast), and particle distribution (e.g., Tc-99 m MAA SPECT/CT). Each method has its own strengths and limitations, and none offers a perfect prediction of the actual therapeutic scenario. Nevertheless, the T/N ratio derived from the planning Tc99m MAA SPECT/CT is presently used as the standard method to predict absorbed doses to tumor and non-tumor hepatic tissues13,14,15. Many apparent “paradoxes” observed in radioembolization dosimetry can often be explained by the inherent biophysical differences among these imaging surrogates. In this study, we chose to correlate the two clinically most useful imaging surrogates, which are apparent “hypovascularity” on pre-procedural arterial phase contrast enhanced cross-sectional imaging to particle distribution on Tc-99 m MAA SPECT/CT.
Use of mRECIST for treatment response in hypovascular tumors is challenging since tumors are already hypoenhancing on baseline MRI and/or CT. Tumors that are hypoenhancing compared to background liver on arterial phase of contrast still showed some enhancement, especially on delayed phase imaging, and thus could be distinguished from completely non-enhancing tissue on post-procedure imaging, which show no difference on arterial phase or delayed phase imaging. Digital subtraction imaging was used to arbitrage enhancement versus non-enhancement when available. In this context, this study reports objective tumor response (CR, PR) of 74%, comparable to recently reported data, including the LEGACY study and DOSISPHERE-01 trial, that demonstrated objective tumor response in 71–90% percent of patients with HCC16,17. Additionally, five patients had a calculated T/N ratio less than 1.00 on SPECT/CT and this subgroup experienced comparatively worse outcomes: three had stable disease (SD) while one had a partial response (PR).
During long-term follow-up, two patients, including one with PD, experienced Grade 1 adverse event increases in ALP serum markers without returning to baseline levels. Although no post-treatment Grade 2 + adverse events were found in patients with a T/N less than 1.00 on pre-treatment mapping, their small sample size and lower rate of achieving PR/CR does still warrant careful consideration of whether 90Y-RE is appropriate.
The use of voxel-based dosimetry may further aid in prognostic stratification. Previous studies investigating patient outcomes after 90Y-RE have demonstrated that achieving a tumor dose of at least 205 Gy while limiting the dose to the normal liver parenchyma to a maximum of 120 Gy, producing a T/N of 1.71, exhibits 100% sensitivity and 75% specificity in predicting favorable treatment outcomes for patients with HCC18. Similar data was reported in another study demonstrating that a T/N greater than 1.00 predicted significant positive outcomes in patients with metastatic liver lesions with a sensitivity of 89%, specificity of 65% and positive predictive value of 71%19.
Study limitations include the retrospective design, small sample size, and comparison over multiple tumor types. Although the heterogeneity of tumor types does not affect dosimetry data with all tumors being hypovascular and undergoing the same pre-treatment planning, this does limit the ability to compare treatment outcomes. Additionally, dosimetry was at the discretion of the AU without a pre-defined criteria based on efficacy or safety, which limits generalizability. Use of mRECIST for treatment response in hypovascular tumors is another limitation, since tumors are already hypoenhancing on pre-procedural imaging.
In conclusion, this study indicated that both primary and secondary hepatic tumors that are hypovascular on pre-procedural arterial-phase imaging most often have T/N values greater than 1 and have appropriate clinical outcomes after 90Y-RE treatment with low adverse event rates. Routine incorporation of compartment-based dosimetry should be strongly considered, particularly for this population of hypovascular tumors.
Data availability
Original data sets for this study are not publicly available. The data that support the findings of this study are not openly available due to reasons of sensitivity and are available from the corresponding author upon reasonable request. Data are located in controlled access data storage within the University of Colorado.
References
Stella, S. F., Noel-Lamy, M., Rogalla, P., Beecroft, R. & Rajan, D. K. Hepatic arterial blood flow modulation in patients with hepatocellular carcinoma: A pilot study of the influence of intraarterial norepinephrine assessed with CT perfusion. J. Vasc Interv Radiol. 32(2), 204–210. https://doi.org/10.1016/j.jvir.2020.08.006 (2021).
McGregor, H. et al. Quantitative assessment of the hemodynamic effects of Intra-Arterial nitroglycerin on hepatocellular carcinoma using Two-Dimensional perfusion angiography. J. Vasc Interv Radiol. 32(2), 198–203. https://doi.org/10.1016/j.jvir.2020.10.023 (2021).
Sato, K. T. et al. The role of tumor vascularity in predicting survival after yttrium-90 radioembolization for liver metastases. J. Vasc Interv Radiol. 20(12), 1564–1569. https://doi.org/10.1016/j.jvir.2009.08.013 (2009).
Kim, H. O. et al. Change in perfusion angiography during transcatheter arterial chemoembolization for hepatocellular carcinoma predicts Short-Term outcomes. AJR Am. J. Roentgenol. 213(4), 746–754. https://doi.org/10.2214/AJR.18.20499 (2019).
Boas, F. E. et al. Quantitative measurements of enhancement on preprocedure triphasic CT can predict response of colorectal liver metastases to radioembolization. AJR Am. J. Roentgenol. 207(3), 671–675. https://doi.org/10.2214/AJR.15.15767 (2016).
Boas, F. E., Bodei, L. & Sofocleous, C. T. Radioembolization of colorectal liver metastases: Indications, Technique, and outcomes. J. Nucl. Med. 58(Suppl 2), 104S–11S. https://doi.org/10.2967/jnumed.116.187229 (2017).
Brunson, C. P. et al. Measurement of the Tumor-to-Normal ratio for radioembolization of hepatocellular carcinoma: A prospective study comparing 2-Dimensional perfusion Angiography, Technetium-99m macroaggregated Albumin, and Yttrium-90 SPECT/CT. J. Vasc Interv Radiol. 35(1), 94–101. https://doi.org/10.1016/j.jvir.2023.09.023 (2024).
Watanabe, M. et al. Voxel-Based dosimetry predicts hepatotoxicity in hepatocellular carcinoma patients undergoing radioembolization with (90)Y glass microspheres. J. Nucl. Med. 64(7), 1102–1108. https://doi.org/10.2967/jnumed.122.264996 (2023).
Orcajo Rincon, J. et al. Maximum tumor-absorbed dose measured by voxel-based multicompartmental dosimetry as a response predictor in yttrium-90 radiation segmentectomy for hepatocellular carcinoma. EJNMMI Phys. 10(1), 7. https://doi.org/10.1186/s40658-022-00520-9 (2023).
Lencioni, R. & Llovet, J. M. Modified RECIST (mRECIST) assessment for hepatocellular carcinoma. Semin Liver Dis. 30(1), 52–60. https://doi.org/10.1055/s-0030-1247132 (2010).
Common Terminology. Criteria for Adverse Events (CTCAE);Published online 2017.
Villalobos, A. et al. Tumor-to-Normal ratio relationship between planning Technetium-99 macroaggregated albumin and posttherapy Yttrium-90 Bremsstrahlung SPECT/CT. J. Vasc Interv Radiol. 32(5), 752–760. https://doi.org/10.1016/j.jvir.2020.12.023 (2021).
Gnesin, S. et al. Partition Model-Based 99mTc-MAA SPECT/CT predictive dosimetry compared with 90Y TOF PET/CT posttreatment dosimetry in radioembolization of hepatocellular carcinoma: A quantitative agreement comparison. J. Nucl. Med. 57(11), 1672–1678. https://doi.org/10.2967/jnumed.116.173104 (2016).
Levillain, H. et al. International recommendations for personalised selective internal radiation therapy of primary and metastatic liver diseases with yttrium-90 resin microspheres. Eur. J. Nucl. Med. Mol. Imaging. 48(5), 1570–1584. https://doi.org/10.1007/s00259-020-05163-5 (2021).
Weber, M. et al. EANM procedure guideline for the treatment of liver cancer and liver metastases with intra-arterial radioactive compounds. Eur. J. Nucl. Med. Mol. Imaging. 49(5), 1682–1699. https://doi.org/10.1007/s00259-021-05600-z (2022).
Garin, E. et al. Personalised versus standard dosimetry approach of selective internal radiation therapy in patients with locally advanced hepatocellular carcinoma (DOSISPHERE-01): a randomised, multicentre, open-label phase 2 trial. Lancet Gastroenterol. Hepatol. 6(1), 17–29. https://doi.org/10.1016/S2468-1253(20)30290-9 (2021).
Salem, R. et al. Yttrium-90 radioembolization for the treatment of Solitary, unresectable HCC: the LEGACY study. Hepatology 74(5), 2342–2352. https://doi.org/10.1002/hep.31819 (2021).
Garin, E. et al. Dosimetry based on 99mTc-macroaggregated albumin SPECT/CT accurately predicts tumor response and survival in hepatocellular carcinoma patients treated with 90Y-loaded glass microspheres: preliminary results. J. Nucl. Med. 53(2), 255–263. https://doi.org/10.2967/jnumed.111.094235 (2012).
Flamen, P. et al. Multimodality imaging can predict the metabolic response of unresectable colorectal liver metastases to radioembolization therapy with Yttrium-90 labeled resin microspheres. Phys. Med. Biol. 53(22), 6591–6603. https://doi.org/10.1088/0031-9155/53/22/019 (2008).
Author information
Authors and Affiliations
Contributions
P.T., J.L., and L.C. contributed to the study conception and design. Material preparation, data collection, and analysis were performed by M.M., L.E., and G.E. The first draft of the manuscript was written by M.M. and G. E. Both A.A. and L.E. made significant contributions to the manuscript. K.M., J.H., J.L., P.T., and L.C. commented on previous versions of the manuscript. All authors read and approved of the final manuscript.
Corresponding author
Ethics declarations
Competing interests
Premal Trivedi has received consulting fees and honoraria from Boston Scientific, with payments made to his institution. Jonathan Lindquist has received consulting fees and honoraria from Boston Scientific Corp, and serves on the advisory board for Boston Scientific Corp, with payments made to his institution Leigh Casadaban has received consulting fees and honoraria from Boston Scientific Corp, with payments made to her institution.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Malavia, M., Eliason, G., Eggers, L.A. et al. Yttrium-90 radioembolization for primary and metastatic liver tumors exhibiting arterial-phase hypovascularity. Sci Rep 16, 4588 (2026). https://doi.org/10.1038/s41598-025-34619-7
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-34619-7